Demonstrating the Applicability of Smartwatches in PM2.5 Health Impact Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. PM Monitoring
2.3. HR Monitoring
2.4. Statistical Analysis
3. Results
3.1. PM2.5 Concentration, HR, and Activity Intensities
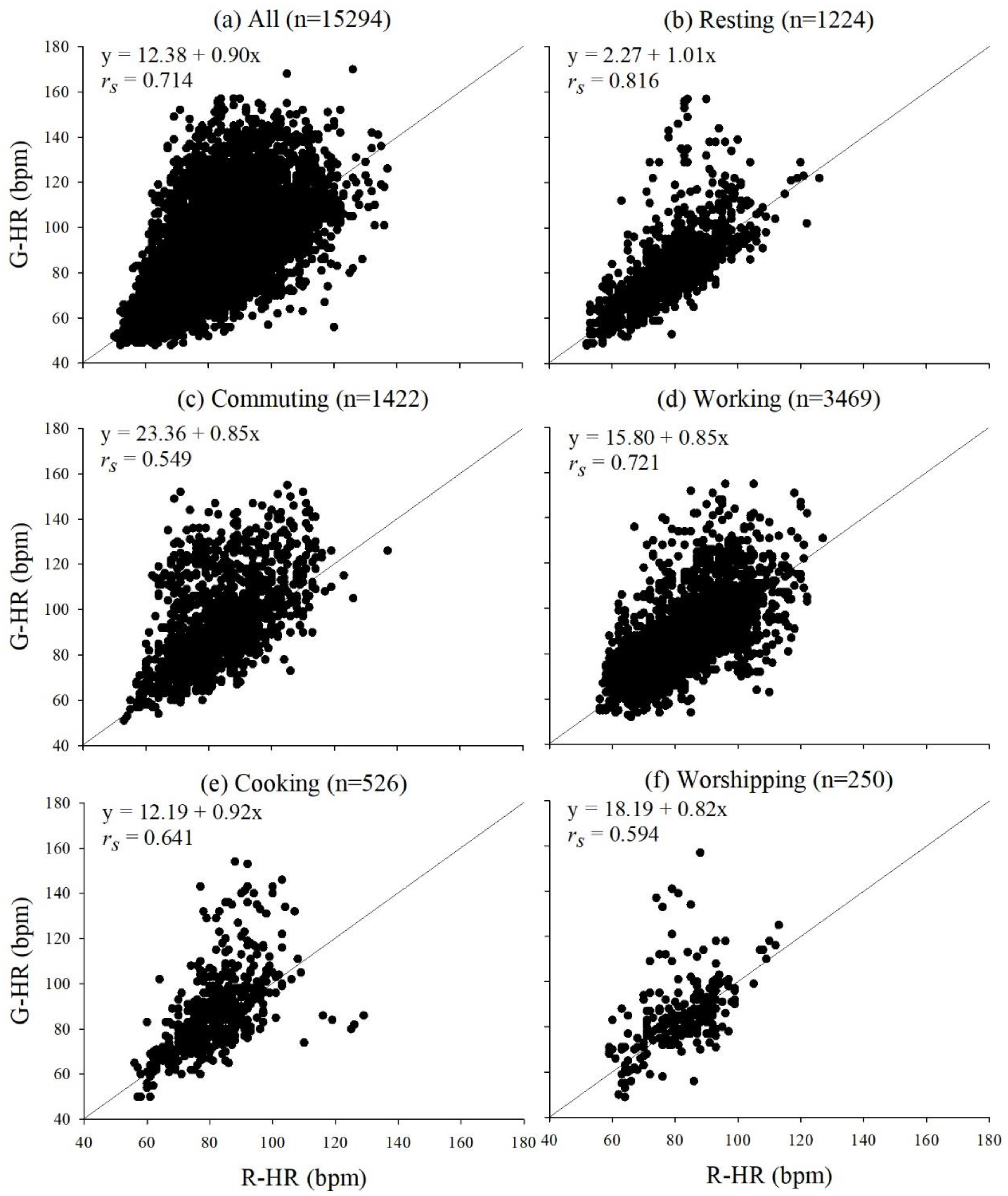
3.2. Correlations between G-HR and R-HR
3.3. Evaluation of Impacts of PM2.5 on G-HR and R-HR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mordor Intelligence, Smartwatch Market—Growth, Trends, COVID-19 Impact, and Forecasts (2021–2026). Available online: https://www.mordorintelligence.com/industry-reports/smartwatch-market (accessed on 23 March 2021).
- Mohammadzadeh, N.; Gholamzadeh, M.; Saeedi, S.; Rezayi, S. The application of wearable smart sensors for monitoring the vital signs of patients in epidemics: A systematic literature review. J. Ambient. Intell. Humaniz. Comput. 2020, 1–15. [Google Scholar] [CrossRef]
- Reeder, B.; David, A. Health at hand: A systematic review of smart watch uses for health and wellness. J. Biomed. Inform. 2016, 63, 269–276. [Google Scholar] [CrossRef]
- Hu, D.; Henry, C.; Bagchi, S. The Effect of Motion on PPG Heart Rate Sensors. In Proceedings of the 2020 50th Annual IEEE-IFIP International Conference on Dependable Systems and Networks-Supplemental Volume (DSN-S), Valencia, Spain, 29 June–2 July 2020; pp. 59–60. [Google Scholar]
- Boudreaux, B.D.; Hebert, E.P.; Hollander, D.B.; Williams, B.M.; Cormier, C.L.; Naquin, M.R.; Gillan, W.W.; Gusew, E.E.; Kraemer, R.R. Validity of Wearable Activity Monitors during Cycling and Resistance Exercise. Med. Sci. Sports Exerc. 2018, 50, 624–633. [Google Scholar] [CrossRef]
- Nelson, B.W.; Allen, N.B. Accuracy of Consumer Wearable Heart Rate Measurement During an Ecologically Valid 24-Hour Period: Intraindividual Validation Study. JMIR mHealth uHealth 2019, 7, e10828. [Google Scholar] [CrossRef]
- Wang, R.; Blackburn, G.; Desai, M.; Phelan, D.; Gillinov, L.; Houghtaling, P.; Gillinov, M. Accuracy of Wrist-Worn Heart Rate Monitors. JAMA Cardiol. 2017, 2, 104–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evenson, K.R.; Spade, C.L. Review of Validity and Reliability of Garmin Activity Trackers. J. Meas. Phys. Behav. 2020, 3, 170–185. [Google Scholar] [CrossRef] [PubMed]
- Dooley, E.E.; Golaszewski, N.M.; Bartholomew, J.B. Estimating Accuracy at Exercise Intensities: A Comparative Study of Self-Monitoring Heart Rate and Physical Activity Wearable Devices. JMIR mHealth uHealth 2017, 5, e34. [Google Scholar] [CrossRef]
- Brook, R.D.; Rajagopalan, S.; Pope, C.A., 3rd; Brook, J.R.; Bhatnagar, A.; Diez-Roux, A.V.; Holguin, F.; Hong, Y.; Luepker, R.V.; Mittleman, M.A.; et al. Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation 2010, 121, 2331–2378. [Google Scholar] [CrossRef] [Green Version]
- Tadic, M.; Cuspidi, C.; Grassi, G. Heart rate as a predictor of cardiovascular risk. Eur. J. Clin. Investig. 2018, 48, e12892. [Google Scholar] [CrossRef] [Green Version]
- Dalal, J.; Dasbiswas, A.; Sathyamurthy, I.; Maddury, S.R.; Kerkar, P.; Bansal, S.; Thomas, J.; Mandal, S.C.; Mookerjee, S.; Natarajan, S.; et al. Heart Rate in Hypertension: Review and Expert Opinion. Int. J. Hypertens. 2019, 2019, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Münzel, T.; Hahad, O.; Gori, T.; Hollmann, S.; Arnold, N.; Prochaska, J.H.; Schulz, A.; Beutel, M.; Pfeiffer, N.; Schmidtmann, I.; et al. Heart rate, mortality, and the relation with clinical and subclinical cardiovascular diseases: Results from the Gutenberg Health Study. Clin. Res. Cardiol. 2019, 108, 1313–1323. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.-J.; Barywani, S.B.; Hansson, P.-O.; Thunström, E.Ö.; Rosengren, A.; Ergatoudes, C.; Mandalenakis, Z.; Caidahl, K.; Fu, M.L. Impact of changes in heart rate with age on all-cause death and cardiovascular events in 50-year-old men from the general population. Open Heart 2019, 6, e000856. [Google Scholar] [CrossRef]
- Tang, C.-S.; Wu, T.-Y.; Chuang, K.-J.; Chang, T.-Y.; Chuang, H.-C.; Lung, S.-C.C.; Chang, L.-T. Impacts of In-Cabin Exposure to Size-Fractionated Particulate Matters and Carbon Monoxide on Changes in Heart Rate Variability for Healthy Public Transit Commuters. Atmosphere 2019, 10, 409. [Google Scholar] [CrossRef] [Green Version]
- Tsou, M.-C.M.; Lung, S.-C.C.; Shen, Y.-S.; Liu, C.-H.; Hsieh, Y.-H.; Chen, N.; Hwang, J.-S. A community-based study on associations between PM2.5 and PM1 exposure and heart rate variability using wearable low-cost sensing devices. Environ. Pollut. 2021, 277, 116761. [Google Scholar] [CrossRef]
- Lung, S.-C.C.; Chen, N.; Hwang, J.-S.; Hu, S.-C.; Wang, W.-C.V.; Wen, T.-Y.J.; Liu, C.-H. Panel study using novel sensing devices to assess associations of PM2.5 with heart rate variability and exposure sources. J. Expo. Sci. Environ. Epidemiol. 2020, 30, 937–948. [Google Scholar] [CrossRef]
- Wang, W.-C.V.; Lung, S.-C.C.; Liu, C.H.; Shui, C.-K. Laboratory Evaluations of Correction Equations with Multiple Choices for Seed Low-Cost Particle Sensing Devices in Sensor Networks. Sensors 2020, 20, 3661. [Google Scholar] [CrossRef]
- Lung, S.-C.C.; Wang, W.-C.V.; Wen, T.-Y.J.; Liu, C.-H.; Hu, S.-C. A versatile low-cost sensing device for assessing PM2.5 spatiotemporal variation and quantifying source contribution. Sci. Total Environ. 2020, 716, 137145. [Google Scholar] [CrossRef]
- Garmin, Garmin: Forerunner® 35. Available online: https://buy.garmin.com/en-US/US/p/552962 (accessed on 15 March 2021).
- Garmin, Activity Tracking and Fitness Metric Accuracy. Available online: http://www.garmin.com/en-US/legal/atdisclaimer (accessed on 20 March 2021).
- Rooti Labs, RootiCare: The Future of Medical Care. Available online: https://www.rooticare.com/ (accessed on 15 March 2021).
- Karaoğuz, M.R.; Yurtseven, E.; Aslan, G.; Deliormanlı, B.G.; Adıgüzel, Ö.; Gönen, M.; Li, K.-M.; Yılmaz, E.N. The quality of ECG data acquisition, and diagnostic performance of a novel adhesive patch for ambulatory cardiac rhythm monitoring in arrhythmia detection. J. Electrocardiol. 2019, 54, 28–35. [Google Scholar] [CrossRef]
- Kroll, R.R.; Boyd, J.G.; Maslove, D.M. Accuracy of a Wrist-Worn Wearable Device for Monitoring Heart Rates in Hospital Inpatients: A Prospective Observational Study. J. Med. Internet Res. 2016, 18, e253. [Google Scholar] [CrossRef] [PubMed]
- Stahl, S.E.; An, H.-S.; Dinkel, D.M.; Noble, J.M.; Lee, J.-M. How accurate are the wrist-based heart rate monitors during walking and running activities? Are they accurate enough? BMJ Open Sport Exerc. Med. 2016, 2, e000106. [Google Scholar] [CrossRef] [Green Version]
- Cadmus-Bertram, L.; Gangnon, R.; Wirkus, E.J.; Thraen-Borowski, K.M.; Gorzelitz, J. The Accuracy of Heart Rate Monitoring by Some Wrist-Worn Activity Trackers. Ann. Intern. Med. 2017, 166, 610–612. [Google Scholar] [CrossRef]
- Soares, A.; Catita, C.; Silva, C. Exploratory Research of CO2, Noise and Metabolic Energy Expenditure in Lisbon Commuting. Energies 2020, 13, 861. [Google Scholar] [CrossRef] [Green Version]
- TFESC; NASPE. Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef] [Green Version]
- Lim, Y.-H.; Bae, H.-J.; Yi, S.-M.; Park, E.; Lee, B.-E.; Hong, Y.-C. Vascular and cardiac autonomic function and PM2.5 constituents among the elderly: A longitudinal study. Sci. Total Environ. 2017, 607–608, 847–854. [Google Scholar] [CrossRef]
- Buregeya, J.M.; Apparicio, P.; Gelb, J. Short-Term Impact of Traffic-Related Particulate Matter and Noise Exposure on Cardiac Function. Int. J. Environ. Res. Public Health 2020, 17, 1220. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Breitner, S.; Cascio, W.E.; Devlin, R.B.; Neas, L.M.; Diaz-Sanchez, D.; Kraus, W.E.; Schwartz, J.; Hauser, E.R.; Peters, A.; et al. Short-term effects of fine particulate matter and ozone on the cardiac conduction system in patients undergoing cardiac catheterization. Part. Fibre Toxicol. 2018, 15, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Qin, G.; Xia, J.; Zhang, Y.; Guo, L.; Chen, R.; Sang, N. Ambient fine particulate matter exposure induces reversible cardiac dysfunction and fibrosis in juvenile and older female mice. Part. Fibre Toxicol. 2018, 15, 1–14. [Google Scholar] [CrossRef]
- Qi, Z.; Yang, C.; Liao, X.; Song, Y.; Zhao, L.; Liang, X.; Su, Y.; Chen, Z.-F.; Li, R.; Dong, C.; et al. Taurine reduction associated with heart dysfunction after real-world PM2.5 exposure in aged mice. Sci. Total Environ. 2021, 782, 146866. [Google Scholar] [CrossRef]
- Cui, L.; Shi, L.; Li, D.; Li, X.; Su, X.; Chen, L.; Jiang, Q.; Jiang, M.; Luo, J.; Ji, A.; et al. Real-Ambient Particulate Matter Exposure-Induced Cardiotoxicity in C57/B6 Mice. Front. Pharmacol. 2020, 11. [Google Scholar] [CrossRef]
- Zhong, J.; Trevisi, L.; Urch, B.; Lin, X.; Speck, M.; Coull, B.A.; Liss, G.; Thompson, A.; Wu, S.; Wilson, A.; et al. B-vitamin Supplementation Mitigates Effects of Fine Particles on Cardiac Autonomic Dysfunction and Inflammation: A Pilot Human Intervention Trial. Sci. Rep. 2017, 7, 45322. [Google Scholar] [CrossRef] [Green Version]
- Xie, X.; Wang, Y.; Yang, Y.; Xu, J.; Zhang, Y.; Tang, W.; Guo, T.; Wang, Q.; Shen, H.; Zhang, Y.; et al. Long-term exposure to fine particulate matter and tachycardia and heart rate: Results from 10 million reproductive-age adults in China. Environ. Pollut. 2018, 242, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Breen, M.; Cascio, W.; Moyer, J.; Schwartz, J.; Di, Q.; Pfaff, E.; Devlin, R.; Diaz-Sanchez, D.; Ward-Caviness, C. Short-term Effects of Fine Particulate Matter on Heart Rate in Heart Failure Patients. In Proceedings of the 32nd Annual Conference of the International Society for Environmental Epidemiology (ISEE), 24–27 August 2020. Virtual Conference. [Google Scholar]
- Paceli, R.; Cardoso, A.; De Sá, M.C.; Vaisberg, M.W.; Nakagama, N.K.; Terra-Filho, M.; Zanetta, D.M.T.; Santos, U.D.P. Outdoor exercise with different PM2.5concentrations and heart rate variability effects in healthy individuals, São Paulo, Brazil. PLoS ONE 2016, 48, PA4286. [Google Scholar] [CrossRef]
- Chen, X.; Chen, W.; Wang, Y.; Han, Y.; Zhu, T. Responses of healthy young males to fine-particle exposure are modified by exercise habits: A panel study. Environ. Health 2018, 17, 88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, M.; Ma, Y.; Song, J.; Lai, C.-F.; Hu, B. Smart Clothing: Connecting Human with Clouds and Big Data for Sustainable Health Monitoring. Mob. Netw. Appl. 2016, 21, 825–845. [Google Scholar] [CrossRef]
- Maeda, Y.; Sekine, M.; Tamura, T. The Advantages of Wearable Green Reflected Photoplethysmography. J. Med. Syst. 2010, 35, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Kheirkhahan, M.; Nair, S.; Davoudi, A.; Rashidi, P.; Wanigatunga, A.A.; Corbett, D.B.; Mendoza, T.; Manini, T.M.; Ranka, S. A smartwatch-based framework for real-time and online assessment and mobility monitoring. J. Biomed. Inform. 2019, 89, 29–40. [Google Scholar] [CrossRef]
- Hernando, D.; Roca, S.; Sancho, J.; Alesanco, Á.; Bailón, R. Validation of the Apple Watch for Heart Rate Variability Measurements during Relax and Mental Stress in Healthy Subjects. Sensors 2018, 18, 2619. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaffer, F.; Ginsberg, J.P. An Overview of Heart Rate Variability Metrics and Norms. Front. Public Health 2017, 5, 258. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| (a) | PM2.5 (μg/m3) | G-HR (bpm) a | R-HR (bpm) b | |||
| Characteristics | nc | Mean ± SD d (Midian) | nc | Mean ± SD d (Midian) | nc | Mean ± SD d (Midian) |
| Age (years) | ||||||
| 40 to 64 years (27) e | 35,846 | 21.8 ± 12.8 * (20.3) | 35,846 | 85.7 ± 16.2 * (83.0) | 7731 | 83.0 ± 12.4 * (83) |
| 65 to 75 years (22) e | 27,215 | 21.2 ± 9.8 (20.0) | 27,215 | 84.2 ± 16.9 (82.0) | 7563 | 80.9 ± 12.2 (80.0) |
| Gender | ||||||
| Male (20) e | 24,844 | 21.4 ± 12.1 * (19.5) | 24,844 | 85.0 ± 16.1 (82.0) | 6170 | 83.0 ± 12.9 * (82.0) |
| Female (29) e | 38,217 | 21.6 ± 11.3 (20.6) | 38,217 | 85.1 ± 16.8 (83.0) | 9124 | 81.2 ± 11.9 (81.0) |
| Body mass index (BMI, kg/m2) | ||||||
| <24 (19) e | 25,811 | 21.8 ± 10.0 * (20.5) | 25,811 | 82.4 ± 15.8 * (80.0) | 5811 | 78.4 ± 11.5 * (77.0) |
| ≥24 (30) e | 37,250 | 21.3 ± 12.6 (20.0) | 37,250 | 86.9 ± 16.7 (84.0) | 9483 | 84.1 ± 12.3 (83.0) |
| (b) | Activity intensity (AS-LUNG-P) f (mG g) | Activity Intensity (RootiRx) h (mG g) | ||||
| Characteristics | nc | Mean ± SD d (Midian) | nc | Mean ± SD d (Midian) | ||
| Age (years) | ||||||
| 40 to 64 years (27) e | 15,006 i | 2140 ± 1160 * (1920) | 7731 | 1970 ± 450 * (1950) | ||
| 65 to 75 years (22) e | 10,963 i | 2090 ± 1070 (1900) | 7563 | 1900 ± 430 (1890) | ||
| Gender | ||||||
| Male (20) e | 12,527 i | 1950 ± 1030 * (1950) | 6170 | 1910 ± 440 * (1910) | ||
| Female (29) e | 1344 i | 2280 ± 1180 (2090) | 9124 | 1950 ± 440 (1920) | ||
| Body mass index (BMI, kg/m2) | ||||||
| <24 (19) e | 11,376 i | 2140 ± 1110 * (1960) | 5811 | 1890 ± 410 * (1880) | ||
| ≥24 (30) e | 14,593 i | 2100 ± 1130 (1880) | 9483 | 1960 ± 460 (1950) | ||
| R-HR | G-HR | |||||
|---|---|---|---|---|---|---|
| Coefficient c | 95% CI d | p-Value | Coefficient c | 95% CI d | p-Value | |
| PM2.5 | 0.229 | 0.127, 0.332 | <0.001 | 0.234 | 0.0801, 0.389 | 0.003 |
| Age | −2.50 | −8.23, 3.59 | 0.412 | −1.83 | −6.47, 3.04 | 0.454 |
| BMI | 5.78 | −0.800, 12.8 | 0.086 | 6.50 | 1.33, 11.9 | 0.013 |
| Gender | 2.03 | −4.23, 8.70 | 0.534 | −1.31 | −6.12, 3.74 | 0.604 |
| Activity | G-HR | ||
|---|---|---|---|
| Coefficient b | 95% CI d | p-Value | |
| Resting (n = 1296) c | 0.617 | –0.117, 1.36 | 0.100 |
| Low-intensity (n = 23909) | 0.219 | 0.0606, 0.378 | 0.007 |
| Moderate- to high-intensity (n = 764) | 4.53 | 1.46, 7.70 | 0.004 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsou, M.-C.M.; Lung, S.-C.C.; Cheng, C.-H. Demonstrating the Applicability of Smartwatches in PM2.5 Health Impact Assessment. Sensors 2021, 21, 4585. https://doi.org/10.3390/s21134585
Tsou M-CM, Lung S-CC, Cheng C-H. Demonstrating the Applicability of Smartwatches in PM2.5 Health Impact Assessment. Sensors. 2021; 21(13):4585. https://doi.org/10.3390/s21134585
Chicago/Turabian StyleTsou, Ming-Chien Mark, Shih-Chun Candice Lung, and Chih-Hui Cheng. 2021. "Demonstrating the Applicability of Smartwatches in PM2.5 Health Impact Assessment" Sensors 21, no. 13: 4585. https://doi.org/10.3390/s21134585
APA StyleTsou, M.-C. M., Lung, S.-C. C., & Cheng, C.-H. (2021). Demonstrating the Applicability of Smartwatches in PM2.5 Health Impact Assessment. Sensors, 21(13), 4585. https://doi.org/10.3390/s21134585







