Advances in Non-Invasive Blood Pressure Monitoring
Abstract
1. Introduction
1.1. Current Standard of Care for Blood Pressure Monitoring
1.1.1. Invasive Arterial Catheters
1.1.2. Inflatable Cuff Measurements
1.2. Current Alternatives in Blood Pressure Measurement
1.2.1. Cuff-Based Devices
1.2.2. Cuffless Devices
2. Materials and Methods
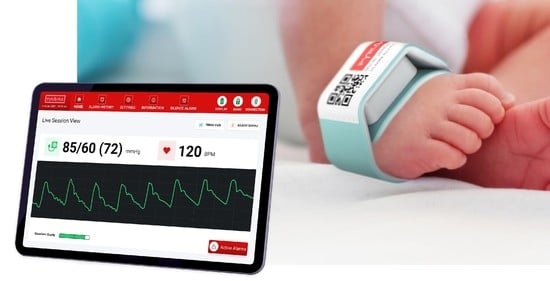
2.1. PyrAmes’ Solution
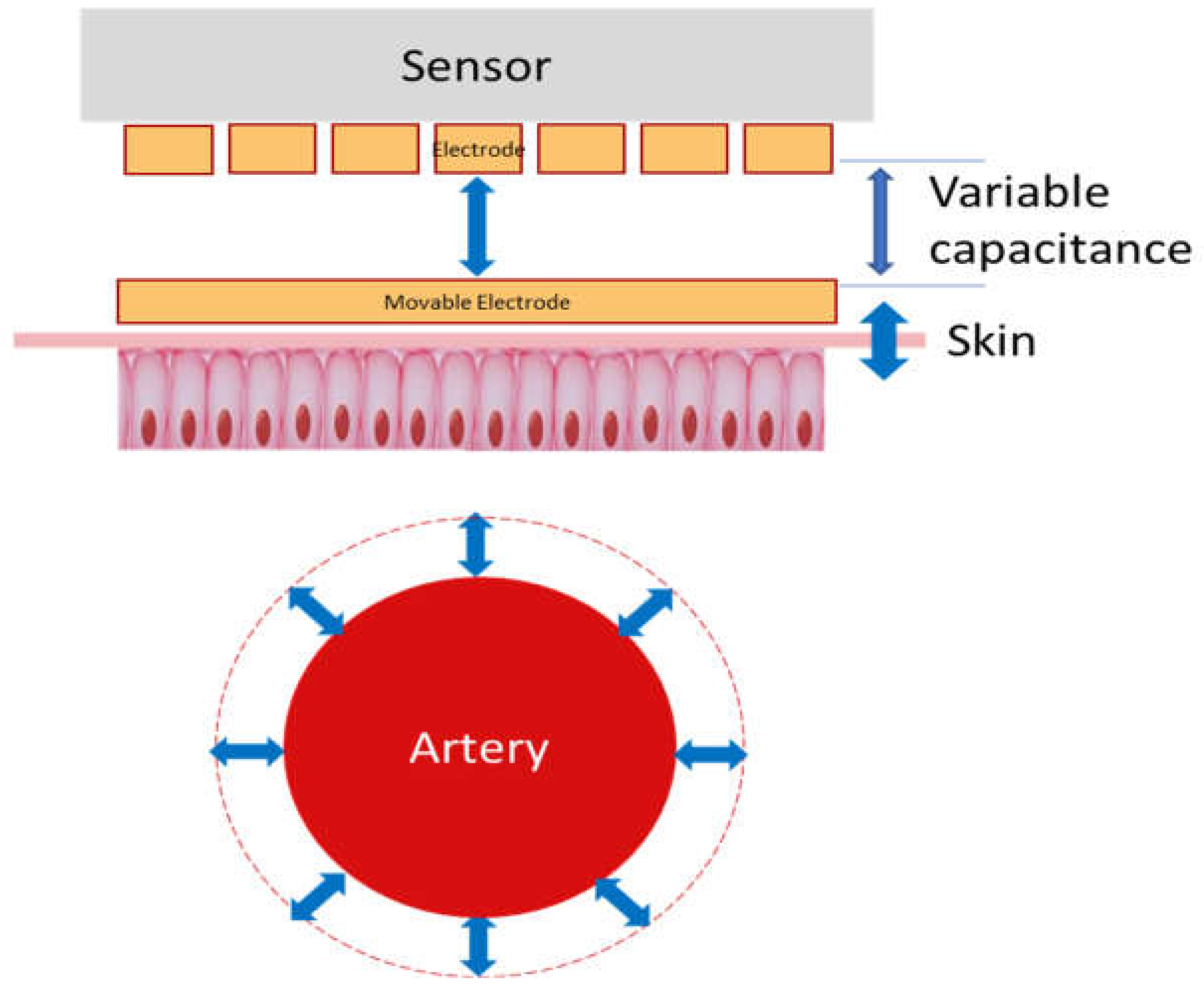
2.2. How It Works
3. Results and Feasibility Studies
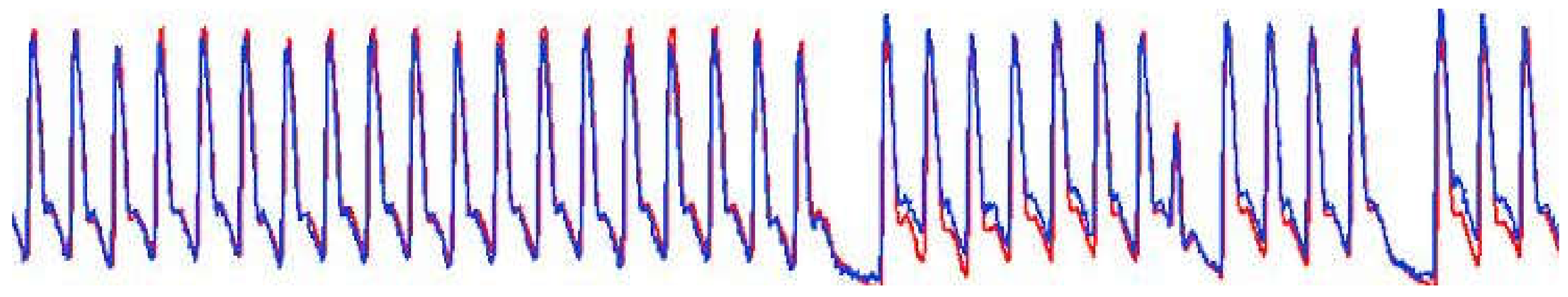
3.1. Proof of Concept
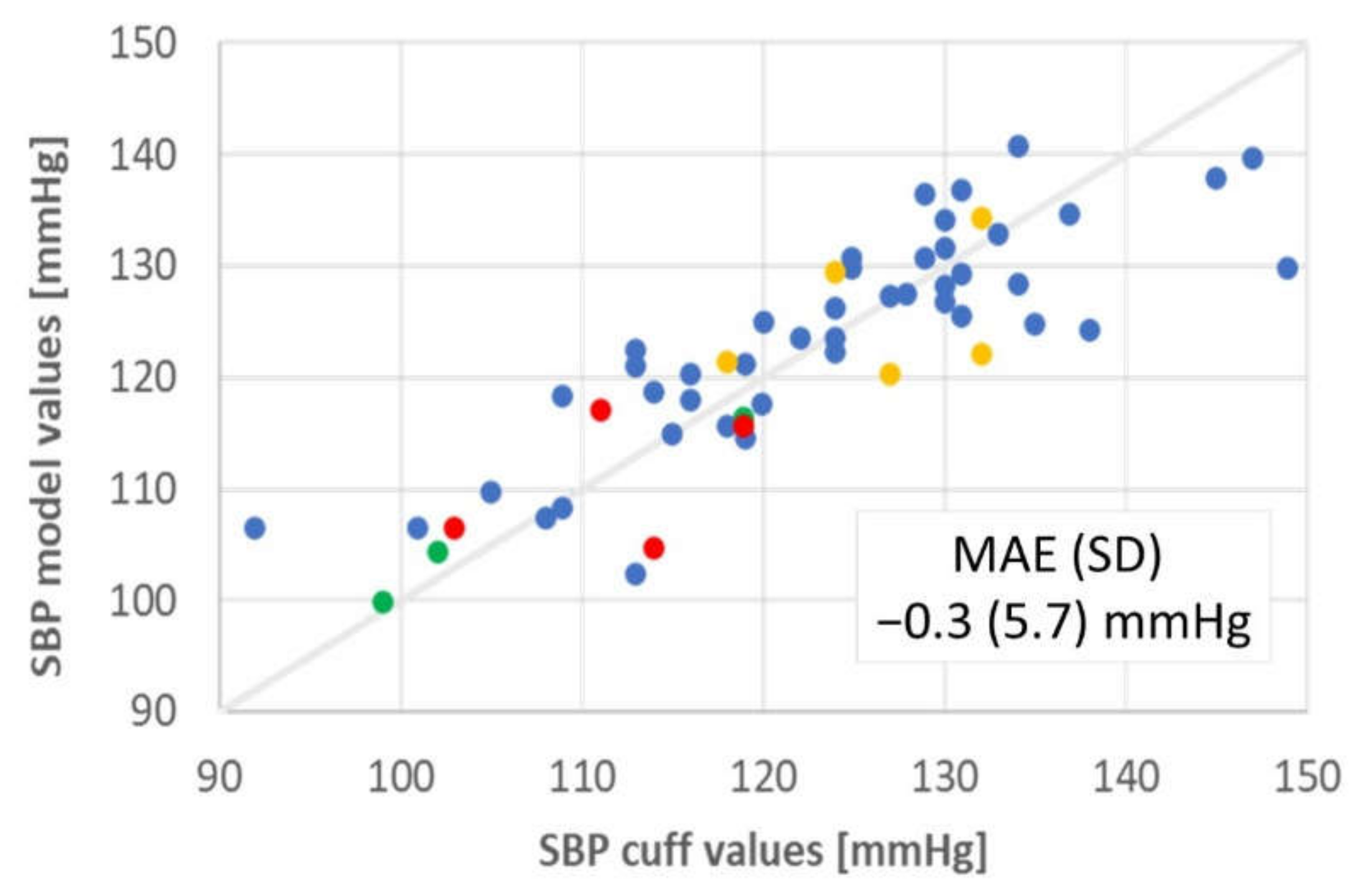
3.2. Ambulatory Adults
3.3. Critically Ill Patients
4. Discussion
Study Limitations
5. Future Work
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Muntner, P.; Carey, R.M.; Gidding, S.; Jones, D.W.; Taler, S.J.; Wright, J.T., Jr.; Whelton, P.K. Potential US population impact of the 2017 ACC/AHA high blood pressure guideline. Circulation 2018, 137, 109–118. [Google Scholar] [CrossRef]
- Vranas, K.C.; Jopling, J.K.; Scott, J.Y.; Badawi, O.; Harhay, M.O.; Slatore, C.G.; Ramsey, M.C.; Breslow, M.J.; Milstein, A.S.; Kerlin, M.P. The Association of ICU with Outcomes of Patients at Low Risk of Dying. Crit. Care Med. 2018, 46, 347. [Google Scholar] [CrossRef]
- Halpern, N. Critical Care Statistics. Available online: https://www.sccm.org/Communications/Critical-Care-Statistics (accessed on 24 May 2021).
- CDC. National Center for Health Statistics. Emergency Department Visits. 2021. Available online: https://www.cdc.gov/nchs/fastats/emergency-department.htm (accessed on 24 May 2021).
- Gabayan, G.Z.; Gould, M.K.; Weiss, R.E.; Derose, S.F.; Chiu, V.Y.; Sarkisian, C.A. Emergency department vital signs and outcomes after discharge. Acad. Emerg. Med. 2017, 24, 846–854. [Google Scholar] [CrossRef]
- Zakaria, E.R.; Joseph, B. Traumatic brain injury: An update. Surg. Res. Open J. 2017, 4, e1–e5. [Google Scholar] [CrossRef]
- De Wall, J. The ABCs of TBI. Evidence-based guidelines for adult traumatic brain injury care. J. Emerg. Med. Serv. 2010, 35, 54–61. [Google Scholar]
- Seamon, M.J.; Feather, C.; Smith, B.P.; Kulp, H.; Gaughan, J.P.; Goldberg, A.J. Just one drop: The significance of a single hypotensive blood pressure reading during trauma resuscitations. J. Trauma Acute Care Surg. 2010, 1, 1289–1295. [Google Scholar] [CrossRef]
- Spaite, D.W.; Hu, C.; Bobrow, B.J.; Chikani, V.; Barnhart, B.; Gaither, J.B.; Denninghoff, K.R.; Adelson, P.D.; Keim, S.M.; Viscusi, C.; et al. Association of out-of-hospital hypotension depth and duration with traumatic brain injury mortality. Ann. Emerg. Med. 2017, 70, 522–530. [Google Scholar] [CrossRef]
- Spaite, D.W.; Hu, C.; Bobrow, B.J.; Chikani, V.; Sherrill, D.; Barnhart, B.; Gaither, J.B.; Denninghoff, K.R.; Viscusi, C.; Mullins, T.; et al. Mortality and prehospital blood pressure in patients with major traumatic brain injury: Implications for the hypotension threshold. JAMA Surg. 2017, 152, 360–368. [Google Scholar] [CrossRef]
- Joffe, R.; Duff, J.; Guerra, G.G.; Pugh, J.; Joffe, A.R. The accuracy of blood pressure measured by arterial line and non-invasive cuff in critically ill children. Crit. Care 2016, 20, 1–9. [Google Scholar] [CrossRef]
- Cuper, N.J.; de Graaff, J.C.; Hartman, B.J.; Verdaasdonk, R.M.; Kalkman, C.J. Difficult arterial cannulation in children: Is a near-infrared vascular imaging system the answer? Br. J. Anaesth. 2012, 109, 420–426. [Google Scholar] [CrossRef]
- Raising Children Network (Australia) Ltd. Your Premature Baby’s Appearance. Available online: https://raisingchildren.net.au/newborns/premature-babies/development/premature-appearance (accessed on 21 May 2021).
- Aouad-Maroun, M.; Raphael, C.K.; Sayyid, S.K.; Farah, F.; Akl, E. Ultrasound-guided arterial cannulation for paediatrics. Cochrane Database Syst. Rev. 2016, 9, CD011364. [Google Scholar] [CrossRef]
- Schindler, E.; Kowald, B.; Suess, H.; Niehaus-Borquez, B.; Tausch, B.; Brecher, A. Catheterization of the radial or brachial artery in neonates and infants. Pediatr. Anesth. 2005, 15, 677–682. [Google Scholar] [CrossRef]
- CDC. High Blood Pressure. Facts About Hypertension. 2020. Available online: https://www.cdc.gov/bloodpressure/facts.htm (accessed on 25 May 2021).
- Dempsey, E.M.; Al Hazzani, F.; Barrington, K.J. Permissive hypotension in the extremely low birthweight infant with signs of good perfusion. Arch. Dis. Child. Fetal Neonatal Ed. 2009, 94, F241–F244. [Google Scholar] [CrossRef]
- Spaite, D.W.; Bobrow, B.J.; Stolz, U.; Sherrill, D.; Chikani, V.; Barnhart, B.; Sotelo, M.; Gaither, J.B.; Viscusi, C.; Adelson, P.D.; et al. Evaluation of the impact of implementing the emergency medical services traumatic brain injury guidelines in Arizona: The Excellence in Prehospital Injury Care (EPIC) study methodology. Acad. Emerg. Med. 2014, 21, 818–830. [Google Scholar] [CrossRef]
- Carteron, L.; Taccone, F.S.; Oddo, M. How to manage blood pressure after brain injury? Minerva Anestesiol. 2016, 83, 412–421. [Google Scholar] [PubMed]
- Chesnut, R.M. Secondary brain insults after head injury: Clinical perspectives. New Horiz. 1995, 3, 366–375. [Google Scholar]
- Fortunato, J.; (Northwestern University Feinberg School of Medicine, Lake Forest, IL, USA). Personal communication, 22 May 2019.
- Wright, J.T., Jr.; Williamson, J.D.; Whelton, P.K.; Snyder, J.K.; Sink, K.M.; Rocco, M.V.; Reboussin, D.M.; Rahman, M.; Oparil, S.; Lewis, C.E.; et al. A randomized trial of intensive versus standard blood-pressure control. N. Engl. J. Med. 2015, 373, 2103–2116. [Google Scholar]
- Prospective Studies Collaboration. Age-specific relevance of usual blood pressure to vascular mortality: A meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002, 360, 1903–1913. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Hypertension Cascade: Hypertension Prevalence, Treatment and Control Estimates Among US Adults Aged 18 Years and Older Applying the Criteria From the American College of Cardiology and American Heart Association’s 2017 Hypertension Guideline—NHANES 2013–2016 External Icon; US Department of Health and Human Services: Atlanta, GA, USA, 2019. [Google Scholar]
- CDC Division for Heart Disease and Stroke Prevention. National Hypertension Control Roundtable. 2020. Available online: https://www.cdc.gov/dhdsp/programs/hypertension-roundtable.htm (accessed on 21 May 2021).
- Huppertz, B. Placental origins of preeclampsia: Challenging the current hypothesis. Hypertension 2008, 51, 970–975. [Google Scholar] [CrossRef] [PubMed]
- Ghulmiyyah, L.; Sibai, B. Maternal mortality from preeclampsia/eclampsia. Semin. Perinatol. 2012, 36, 56–59. [Google Scholar] [CrossRef]
- Center for Disease Control and Prevention. High Blood Pressure in The United States; Division for Heart Disease and Stroke Prevention: Washington, DC, USA, 2016. Available online: https://www.cdc.gov/dhdsp/data_statistics/fact_sheets/fs_bloodpressure.htm (accessed on 8 June 2018).
- Lionakis, N.; Mendrinos, D.; Sanidas, E.; Favatas, G.; Georgopoulou, M. Hypertension in the elderly. World J. Cardiol. 2012, 4, 135–147. [Google Scholar] [CrossRef]
- Jesus, N.S.D.; Nogueira, A.D.R.; Pachu, C.O.; Luiz, R.R.; Oliveira, G.M.M.D. Blood pressure treatment adherence and control after participation in the ReHOT. Arq. Bras. Cardiol. 2016, 107, 437–445. [Google Scholar] [CrossRef]
- Kronish, I.M.; Kent, S.; Moise, N.; Shimbo, D.; Safford, M.M.; Kynerd, R.E.; O’Beirne, R.; Sullivan, A.; Muntner, P. Barriers to conducting ambulatory and home blood pressure monitoring during hypertension screening in the United States. J. Am. Soc. Hypertens. 2017, 11, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Castellano, P.; Lurato, J.; Howell, C.; Edwards, M. Top 5 Considerations for Implementing ABPM in Clinical Trials Med Device Online. 2019. Available online: https://www.ert.com/blog/top-5-considerations-for-implementing-abpm-in-clinical-trials/ (accessed on 21 December 2019).
- Chung, E.; Chen, G.; Alexander, B.; Cannesson, M. Non-invasive continuous blood pressure monitoring: A review of current applications. Front. Med. 2013, 7, 91–101. [Google Scholar] [CrossRef]
- Pickering, T.G.; Shimbo, D.; Haas, D. Ambulatory blood-pressure monitoring. N. Engl. J. Med. 2006, 354, 2368–2374. [Google Scholar] [CrossRef]
- Banegas, J.R.; Ruilope, L.M.; de la Sierra, A.; Vinyoles, E.; Gorostidi, M.; de la Cruz, J.J.; Ruiz-Hurtado, G.; Segura, J.; Rodríguez-Artalejo, F.; Williams, B. Relationship between Clinic and Ambulatory Blood-Pressure Measurements and Mortality. N. Engl. J. Med. 2018, 378, 1509–1520. [Google Scholar] [CrossRef]
- Burr, M.L.; Dolan, E.; O’Brien, E.W.; O’Brien, E.T.; McCormack, P. The value of ambulatory blood pressure in older adults: The Dublin outcome study. Age Ageing 2008, 37, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Weiner, R.E.; Yohannes-Tomicich, J. Arterial Line Monitoring and Placement. In Critical Care; Oropello, J.M., Pastores, S.M., Kvetan, V., Eds.; McGraw-Hill: New York, NY, USA, 2016; Available online: https://accessmedicine.mhmedical.com/content.aspx?bookid=1944§ionid=143522170 (accessed on 21 May 2021).
- Holt, T.; Davinia, R.; Withington, E.; Mitchell, E. Which pressure to believe? A comparison of direct arterial with indirect blood pressure measurement techniques in the pediatric intensive care unit. Pediatric Crit. Care Med. Soc. Crit. Care Med. 2011, 12, e391–e394. [Google Scholar] [CrossRef]
- Brzezinski, M.; Luisetti, T.; London, M.J. Radial artery cannulation: A comprehensive review of recent anatomic and physiologic investigations. Anesth. Analg. 2009, 109, 1763–1781. [Google Scholar] [CrossRef]
- McAdams, R.M.; Winter, V.T.; McCurnin, D.C.; Coalson, J.J. Complications of umbilical artery catheterization in a model of extreme prematurity. J. Perinatol. 2009, 29, 685–692. [Google Scholar] [CrossRef][Green Version]
- Nuttall, G.; Burckhardt, J.; Hadley, A.; Kane, S.; Kor, D.; Marienau, M.S.; Schroeder, D.R.; Handlogten, K.; Wilson, G.; Oliver, W.C. Surgical and Patient Risk Factors for Severe Arterial Line Complications in Adults. Anesthesiology 2016, 124, 590–597. [Google Scholar] [CrossRef]
- Froehler, M.T.; Chitale, R.; Magarik, J.A.; Fusco, M.R. Comparison of a pressure-sensing sheath and radial arterial line for intraoperative blood pressure monitoring in neurointerventional procedures. J. Neurointerv. Surg. 2018, 10, 784–787. [Google Scholar] [CrossRef]
- Brotschi, B.; Hug, M.I.; Latal, B.; Neuhaus, D.; Buerki, C.; Kroiss, S.; Spoerri, C.; Albisetti, M. Incidence and predictors of indwelling arterial catheter-related thrombosis in children. J. Thromb. Haemost. 2011, 9, 1157–1162. [Google Scholar] [CrossRef] [PubMed]
- Vasquez, P.; Burd, A.; Mehta, R.; Hiatt, M.; Hegyi, T. Resolution of Peripheral Artery Catheter-induced Ischemic Injury Following Prolonged Treatment with Topical Nitroglycerin Ointment in a Newborn: A Case Report. J. Perinatol. 2003, 23, 348–350. [Google Scholar] [CrossRef]
- Gleich, S.J.; Wong, A.V.; Handlogten, K.S.; Thum, D.E.; Nemergut, M.E. Major Short-term Complications of Arterial Cannulation for Monitoring in Children. Anesthesiology 2021, 134, 26–34. [Google Scholar] [CrossRef]
- Furfaro, S.; Gauthier, M.; Lacroix, J.; Nadeau, D.; Lafleur, L.; Mathews, S. Arterial Catheter—Related Infections in Children A 1-Year Cohort Analysis: A 1-Year Cohort Analysis. Am. J. Dis. Child. 1991, 145, 1037–1042. [Google Scholar] [CrossRef]
- Veldman, A.; Nold, M.F.; Behnke, I.M. Thrombosis in the critically ill neonate: Incidence, diagnosis, and management. Vasc. Health Risk Manag. 2008, 4, 1337–1348. [Google Scholar]
- Cohen, M.M.; Cameron, C.B.; Duncan, P.G. Pediatric anesthesia morbidity and mortality in the perioperative period. Anesth. Analg. 1990, 70, 160–167. [Google Scholar] [CrossRef]
- Blume-Peytavi, U.; Tan, J.; Tennstedt, D.; Boralevi, F.; Fabbrocini, G.; Torrelo, A.; Soares-Oliveira, R.; Haftek, M.; Rossi, A.B.; Thouvenin, M.D.; et al. Fragility of epidermis in newborns, children and adolescents. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 3–56. [Google Scholar] [CrossRef]
- Rizzi, M.; Goldenberg, N.; Bonduel, M.; Revel-Vilk, S.; Amankwah, E.; Albisetti, M. Catheter-related arterial thrombosis in neonates and children: A systematic review. Thromb. Haemost. 2018, 118, 1058–1066. [Google Scholar] [CrossRef]
- Mosalli, R.; Elbaz, M.; Paes, B. Topical Nitroglycerine for Neonatal Arterial Associated Peripheral Ischemia following Cannulation: A Case Report and Comprehensive Literature Review. Case Rep. Pediatr. 2013, 2013, 1–7. [Google Scholar] [CrossRef]
- Tareerath, M.; Wongyingsinn, M. Comparison of the Incidences of Cuff-Related Trauma after Non-invasive Arterial Blood Pressure Measurement with and without Padding in Patients Undergoing Elective Surgery. J. Med. Assoc. Thail. 2018, 101, 1. [Google Scholar]
- Jeon, Y.S.; Kim, Y.S.; Lee, J.A.; Seo, K.H.; In, J.H. Rumpel-Leede phenomenon associated with noninvasive blood pressure monitoring-A case report. Korean J. Anesthesiol. 2010, 59, 203. [Google Scholar] [CrossRef]
- Elmatite, W.; Mangla, C.; Upadhyay, S.; Yarmush, J. Perioperative Automated Noninvasive Blood Pressure- (NIBP-) Related Peripheral Nerve Injuries: An Anesthetist’s Dilemma—A Case Report and Review of the Literature. Case Rep. Anesthesiol. 2020, 2020, 1–6. [Google Scholar] [CrossRef]
- Diprose, G.K.; Evans, D.H.; Archer, L.N.; Levene, M.I. Dinamap fails to detect hypotension in very low birthweight infants. Arch. Dis. Child. 1986, 61, 771–773. [Google Scholar] [CrossRef] [PubMed]
- Tzong, K.Y.; Han, S.; Roh, A.; Ing, C. Epidemiology of pediatric surgical admissions in US children: Data from the HCUP kids inpatient database. J. Neurosurg. Anesthesiol. 2012, 24, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Schulman, J.; Braun, D.; Lee, H.C.; Profit, J.; Duenas, G.; Bennett, M.V.; Dimand, R.J.; Jocson, M.; Gould, J.B. Association between neonatal intensive care unit admission rates and illness acuity. JAMA Pediatrics 2018, 172, 17–23. [Google Scholar] [CrossRef] [PubMed]
- CDC National Center for Health Statistics. Births and Natality. 2021. Available online: https://www.cdc.gov/nchs/fastats/births.htm (accessed on 24 May 2021).
- Mohammadi, A.; Mehdizade, A.; Ghasemi-Rad, M. Bilateral Tardus-Parvus Waveforms in a Patient With Aortic Coarctation. J. Ultrasound Med. 2011, 30, 1030–1031. [Google Scholar] [CrossRef]
- Mayo Clinic. Coarctation of the Aorta. Available online: https://www.mayoclinic.org/diseases-conditions/coarctation-of-the-aorta/diagnosis-treatment/drc-20352535 (accessed on 24 May 2021).
- Ahmed, M. Aortic Stenosis a Tight Aortic Valve—A Comprehensive Patient Guide. 2016. Available online: https://myheart.net/articles/aortic-stenosis-a-tight-aortic-valve-a-comprehensive-patient-guide/ (accessed on 23 May 2021).
- Kutty, P.K.; Krishnaswamy, G. The Link: Pediatric History Taking and Physical Examination; Kutty, P.K., Krishnaswamy, G., Eds.; Jaypee Brothers: New Delhi, India, 2016. [Google Scholar]
- Dhandare, A. Cardiovascular System Examination. 2017. Available online: https://www.slideshare.net/AshishDhandare2/cardiovascular-system-examination-75965094 (accessed on 23 May 2021).
- Fang, J.; O’Gara, P. The History and Physical Examination.An Evidence-Based Approach.2016. Available online: https://thoracickey.com/the-history-and-physical-examination/#s0060 (accessed on 23 May 2021).
- Gevers, M.; Van Der Mooren, K.; Stergiopulos, N.; Van Genderingen, H.R.; Lafeber, H.N.; Hack, W.W.; Westerhof, N. Bisferiens peaks in the radial artery pressure wave during patent ductus arteriosus in newborn infants: Relationship with ascending aortic flow. Pediatric Res. 1996, 40, 163–168. [Google Scholar] [CrossRef]
- Moses, S. Arterial Pulse. 2021. Available online: https://fpnotebook.com/CV/Exam/ArtrlPls.htm (accessed on 23 May 2021).
- Giese, E.A.; O’Connor, F.G.; Brennan, F.H., Jr.; Depenbrock, P.J.; Oriscello, R.G. The Athlete Preparticipation Evaluation: Cardiovascular Assessment. Am. Fam. Physician 2007, 75, 1008–1014. [Google Scholar]
- Flynn, J.T.; Kaelber, D.C.; Baker-Smith, C.M.; Blowey, D.; Carroll, A.E.; Daniels, S.R.; de Ferranti, S.D.; Dionne, J.M.; Falkner, B.; Flinn, S.K.; et al. Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics 2017, 140. [Google Scholar] [CrossRef]
- Ilic, A.; Ilic, D.; Papović, J.; Stojsic, S.; Milovancev, A.; Grkovic, D.; Stojsic-Milosavljevic, A.; Redzek-Mudrinic, T.; Bjelica, A.; Rankov, O.; et al. Non-Dipping Patten of Blood Pressure and Gestational Hypertension. Blood Press. Bench Bed 2018. [Google Scholar] [CrossRef]
- Desai, R.; Park, H.; Dietrich, E.A.; Smith, S.M. Trends in ambulatory blood pressure monitoring use for confirmation or monitoring of hypertension and resistant hypertension among the commercially insured in the U.S., 2008–2017. Int. J. Cardiol. Hypertens. 2020, 6, 100033. [Google Scholar] [CrossRef]
- Taira, D.; Sentell, T.; Albright, C.; Lansidell, D.; Nakagawa, K.; Seto, T.; Stevens, J.M. Insights in Public Health: Ambulatory Blood Pressure Monitoring: Underuse in Clinical Practice in Hawaii. J. Med. Public Health 2017, 76, 314–317. [Google Scholar]
- Ghuman, N.; Campbell, P.; White, W.B. Role of ambulatory and home blood pressure recording in clinical practice. Curr. Cardiol. Rep. 2009, 11, 414–421. [Google Scholar] [CrossRef]
- Olejnikov, V.E.; Eliseeva, I.V.; Tomashevskaya, Y.A.; Borisova, N.A.; Fadeeva, S.S. The Efficacy Of Antihypertensive Therapy In Elderly Patients And Treatment Compliance Analysis. Ration. Pharm. Cardiol. 2014, 10, 391–396. [Google Scholar] [CrossRef]
- Setia, S.; Subramaniam, K.; Teo, B.W.; Tay, J.C. Ambulatory and home blood pressure monitoring: Gaps between clinical guidelines and clinical practice in Singapore. Int. J. Gen. Med. 2017, 10, 189–197. [Google Scholar] [CrossRef] [PubMed]
- BIOPAC Systems. Available online: https://www.biopac.com/ (accessed on 8 June 2021).
- Finapres Medical Systems: Enschede. Available online: https://www.finapres.com/ (accessed on 8 June 2021).
- ADIntruments: Dunedin. Available online: https://www.adinstruments.com/ (accessed on 8 June 2021).
- Edwards Lifesciences Corporation: Irvine. Available online: https://www.edwards.com/ (accessed on 8 June 2021).
- CNAP: Graz. Available online: https://www.cnsystems.com/ (accessed on 8 June 2021).
- Caretaker Medical: Charlottesville. Available online: https://caretakermedical.net/ (accessed on 8 June 2021).
- Hodsden, S. Omron Introduces Smartwatch-size Blood Pressure Monitor. Med Device Online. 2019. Available online: https://www.meddeviceonline.com/doc/omron-introduces-smartwatch-size-blood-pressure-monitor-0001 (accessed on 8 June 2018).
- H2CARE. Available online: http://charmcare.com/ (accessed on 8 June 2021).
- Dionne, J.M.; Bremner, S.A.; Baygani, S.K.; Batton, B.; Ergenekon, E.; Bhatt-Mehta, V.; Dempsey, E.; Kluckow, M.; Koplowitz, L.P.; Apele-Freimane, D.; et al. Method of blood pressure measurement in neonates and infants: A systematic review and analysis. J. Pediatrics 2020, 221, 23–31. [Google Scholar] [CrossRef]
- Association for the Advancement of Medical Instrumentation. American National Standard. Manual, Electronic or Automated Sphygmomanometers ANSI/AAMI SP10-2002. 3330 Washington Boulevard; AAMI: Arlington, VA, USA, 2003. [Google Scholar]
- ISO 81060-2:2019. Non-Invasive Sphygmomanometers—Part 2: Clinical Investigation Of In-termittent Automated Measurement Type; ISO: Washington, DC, USA, 2019. [Google Scholar]
- Tamura, T. Cuffless Blood Pressure Monitors: Principles, Standards and Approval for Medical Use. IEICE Trans. Commun. 2021, 104, 580–586. [Google Scholar] [CrossRef]
- Bruining, N.; Caiani, E.; Chronaki, C.; Guzik, P.; van der Velde, E. Acquisition and analysis of cardiovascular signals on smartphones: Potential, pitfalls and perspectives: By the Task Force of the e-Cardiology Working Group of European Society of Cardiology. Eur. J. Prev. Cardiol. 2014, 21 (Suppl. 2), 4–13. [Google Scholar] [CrossRef]
- Matthys, K.; Verdonck, P. Development and modelling of arterial applanation tonometry: A review. Technol. Health Care 2002, 10, 65–76. [Google Scholar] [CrossRef]
- Tensys Medical Inc.: San Diego, CA, United States. Available online: https://www.dicardiology.com/company/tensys-medical-inc (accessed on 8 June 2021).
- Bard, D.M.; Joseph, J.I.; Van Helmond, N. Cuff-Less Methods for Blood Pressure Telemonitoring. Front. Cardiovasc. Med. 2019, 6, 40. [Google Scholar] [CrossRef]
- Stojanova, A.; Koceski, S.; Koceska, N. Continuous Blood Pressure Monitoring as a Basis for Ambient Assisted Living (AAL)—Review of Methodologies and Devices. J. Med. Syst. 2019, 43, 24. [Google Scholar] [CrossRef]
- Pandit, J.A.; Lores, E.; Batlle, D. Cuffless Blood Pressure Monitoring: Promises and Challenges. Clin. J. Am. Soc. Nephrol. 2020, 15, 1531–1538. [Google Scholar] [CrossRef]
- Samsung. Samsung Expands Vital Blood Pressure and Electrocardiogram Tracking to Galaxy Watch3 and Galaxy Watch Active2 in 31 More Countries. 2021. Available online: https://news.samsung.com/global/samsung-expands-vital-blood-pressure-and-electrocardiogram-tracking-to-galaxy-watch3-and-galaxy-watch-active2-in-31-more-countries (accessed on 25 May 2021).
- Vybornova, A.; Polychronopoulou, E.; Wurzner-Ghajarzadeh, A.; Fallet, S.; Sola, J.; Wuerzner, G. Blood pressure from the optical Aktiia Bracelet: A 1-month validation study using an extended ISO81060-2 protocol adapted for a cuffless wrist device. Blood Press. Monit. 2021, 30. [Google Scholar] [CrossRef]
- Nachman, D.; Gepner, Y.; Goldstein, N.; Kabakov, E.; Ben Ishay, A.; Littman, R.; Azmon, Y.; Jaffe, E.; Eisenkraft, A. Comparing blood pressure measurements between a photoplethysmography-based and a standard cuff-based manometry device. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Sensifree. A New Standard of Care in Blood Pressure Monitoring. Available online: https://sensifree.com (accessed on 25 May 2021).
- Baruch, M.C.; Kalantari, K.; Gerdt, D.W.; Adkins, C.M. Validation of the pulse decomposition analysis algorithm using central arterial blood pressure. Biomed. Eng. Online 2014, 13, 1–19. [Google Scholar] [CrossRef]
- Munir, S.; Guilcher, A.; Kamalesh, T.; Clapp, B.; Redwood, S.; Marber, M.; Chowienczyk, P. Peripheral Augmentation Index Defines the Relationship Between Central and Peripheral Pulse Pressure. Hypertension 2008, 51, 112–118. [Google Scholar] [CrossRef]
- Donley, D.A.; Fournier, S.B.; Reger, B.L.; DeVallance, E.; Bonner, D.E.; Olfert, I.M.; Frisbee, J.C.; Chantler, P.D. Aerobic Exercise Training Reduces Arterial Stiffness in Metabolic Syndrome. J. Appl. Physiol. 2014, 116, 1396–1404. [Google Scholar] [CrossRef]
- Lee, B.-K. Computational fluid dynamics in cardiovascular disease. Korean Circ. J. 2011, 41, 423. [Google Scholar] [CrossRef]
- TensorFlow Developers, Version v1.14.0; Zenodo: Geneva, Switzerland, 2021.
- Visscher, M.; Taylor, T. Pressure ulcers in the hospitalized neonate: Rates and risk factors. Sci. Rep. 2014, 4, 1–6. [Google Scholar] [CrossRef]
- Seattle Children’s Hospital. Improving Neonatal Skin Care. Available online: https://www.seattlechildrens.org/globalassets/documents/healthcare-professionals/neonatal-briefs/neonatal-skin-care.pdf (accessed on 26 May 2021).
- Wu, E.; Wu, K.; Daneshjou, R.; Ouyang, D.; Ho, D.E.; Zou, J. How medical AI devices are evaluated: Limitations and recommendations from an analysis of FDA approvals. Nat. Med. 2021, 27, 582–584. [Google Scholar] [CrossRef]



| Method | Measurement | Advantages | Disadvantages | Example Companies w/FDA/EU Clearance | Example Companies w/Development Programs | |
|---|---|---|---|---|---|---|
| Cuff | Finger, tabletop (volume clamp technology) | Continuous | Validated for adults, can be self-calibrated | Restricted mobility, bulky, expensive (>$15 K) | BMEYE, Finapres, ADI, Biopac, Edwards, CNAP | |
| Finger, wearable (volume clamp technology) | Continuous | Validated for adults, can be self-calibrated | Expensive ($5 K), uncomfortable, restricts movement of hand, high power | Caretaker | ||
| Wrist | Intermittent | Validated for adults; established technology | Concerns about accuracy, bulky, high power | Omron, H2Care | ||
| Cuffless | PPG | Continuous | Wearable technology widely used for heart rate; low cost; can pick up signal almost anywhere on body | Noise from ambient light, skin color; uncomfortable; high power; periodic calibration | Aktiia, BioBeat, Aura | Apple, ASUS, Samsung, Sensifree, |
| PWV, PTT | Continuous | Widely studied technique; can use many combinations of sensor technology | Requires multiple sensors to determine pulse wave velocity (PWV) and pulse transit time (PTT), large training dataset, high power; many of the same issues as PPG | Sotera, Somnometics | Vital Insite, Quanttus, Scanadu, Blumio, Sibel | |
| Tonometer | Continuous | Established technique; validated for adults | Requires calibration, expensive, uncomfortable, restricts movement | Tensys, HealthStat | ||
| Capacitance | Continuous | Demonstrated for neonates; highly detailed pulse waveforms; requires minimal contact with skin for less irritation; lower power | New technology | PyrAmes, Vena Vitals |
| Features | Study #1: Ambulatory Adults | Study #2a: Critically Ill Adults & Children | Study #2b: NICU Infants |
|---|---|---|---|
| Study participants | n = 104,
Age 21- > 89 y, 62% F | n = 124, Age 4–87 y, 42% F | n = 16,
Age 1–8 days, 33%F GA 25–40 w, Weight 0.7–3.6 kg |
| PyrAmes Data duration | ~10 min per subject (6 h total) ABPM subjects 4–24 h ea | ~5 h per patient (500 h) | ~10 h per patient (360 h) |
| Device | V2 Prototype (training data) Boppli Band (test data) | V3 Prototype | Boppli Band |
| Sensor/Electronics size | Area of 4-sensor array: 6 × 10 mm2, Electronics: 2.1 × 6.3 cm, >30 g | Area of 4-sensor array: 6 × 10 mm2, Electronics: 3.5 × 3.8 cm, >30 g | Area of 4-sensor array: 15 × 7 mm2, Electronics: 2 × 2.5 cm, 12 g |
| PyrAmes device location | Wrist | Wrist and foot | |
| Source of training data | 3–100 cuff msmts per patient. | Stanford invasive arterial line (IAL) data (n = 899,
age 1 day ~53 year, Weight 0.83 Kg~114.8 kg, 45% F), Children’s National Hospital IAL data (n = 40, age 1–2 day, Weight 0.5 kg~4.7 kg, 40% F, GA 23–40 wk) (~9000 h total) | |
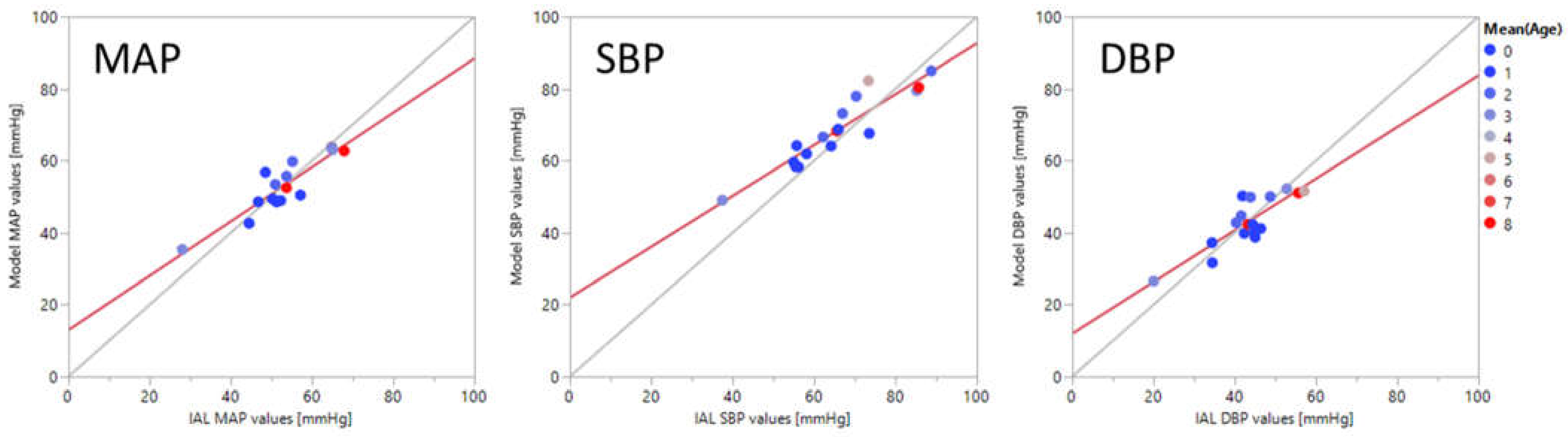
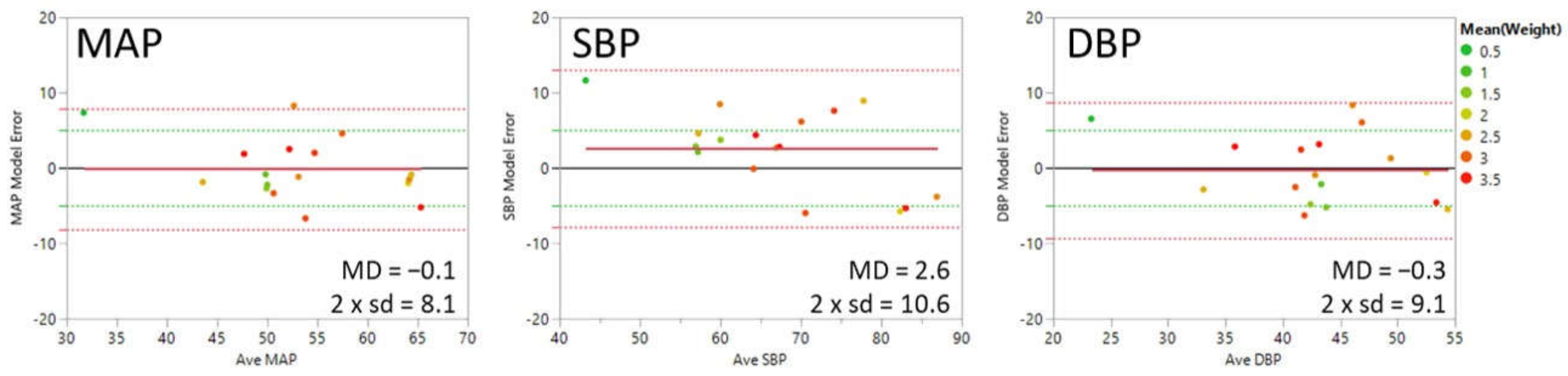
| Outcome: Meets FDA accuracy criteria? [84] MAE (sd) < ±5 (8) mmHg | SBP: −0.3 (5.7) mmHg | SBP: −3 to 3 (>12) mmHg (no successful models) | SBP: 2.8 (5.5) mmHg DBP: −0.2 (4.6) mmHg MAP: 0.1 (4.1) mmHg |
| Clinical collaborators | Drs. Vivek Bhalla, Tara Chang, Sandra Tsai | Drs. Anita Honkanen, Chandra Ramamoorthy, Archana Varma, Alexandria Joseph | Drs. William Rhine, Anoop Rao |
| Stanford study site | Cardiology & Hypertensive clinics | Intensive Care Unit (ICU), Pediatric ICU (PICU), Cardiovascular ICU (CVICU) | Neonatal (ICU), CVICU |
| MAP | SBP | DBP | FDA Guidelines [84] | ||||
|---|---|---|---|---|---|---|---|
| MAE | sd | MAE | sd | MAE | sd | MAE | sd |
| 0.1 | 4.1 | 2.8 | 5.5 | −0.2 | 4.6 | <±5 | <8 mmHg |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quan, X.; Liu, J.; Roxlo, T.; Siddharth, S.; Leong, W.; Muir, A.; Cheong, S.-M.; Rao, A. Advances in Non-Invasive Blood Pressure Monitoring. Sensors 2021, 21, 4273. https://doi.org/10.3390/s21134273
Quan X, Liu J, Roxlo T, Siddharth S, Leong W, Muir A, Cheong S-M, Rao A. Advances in Non-Invasive Blood Pressure Monitoring. Sensors. 2021; 21(13):4273. https://doi.org/10.3390/s21134273
Chicago/Turabian StyleQuan, Xina, Junjun Liu, Thomas Roxlo, Siddharth Siddharth, Weyland Leong, Arthur Muir, So-Min Cheong, and Anoop Rao. 2021. "Advances in Non-Invasive Blood Pressure Monitoring" Sensors 21, no. 13: 4273. https://doi.org/10.3390/s21134273
APA StyleQuan, X., Liu, J., Roxlo, T., Siddharth, S., Leong, W., Muir, A., Cheong, S.-M., & Rao, A. (2021). Advances in Non-Invasive Blood Pressure Monitoring. Sensors, 21(13), 4273. https://doi.org/10.3390/s21134273