Tracking Fluorescent Dye Dispersion from an Unmanned Aerial Vehicle
Abstract
1. Introduction
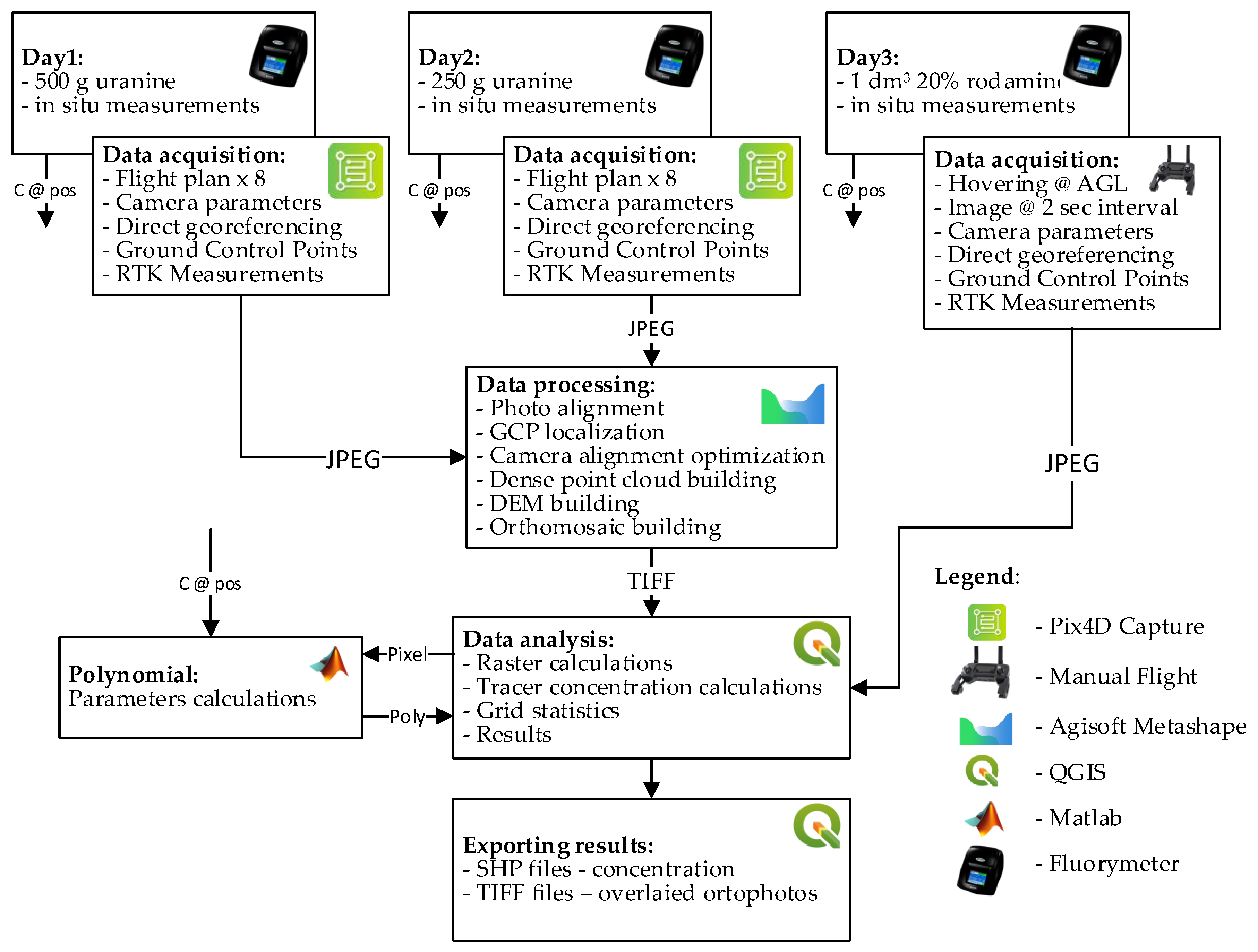
2. Materials and Methods
2.1. Method for Image-Based Measurement of Dye Concentration
2.2. Research Plan
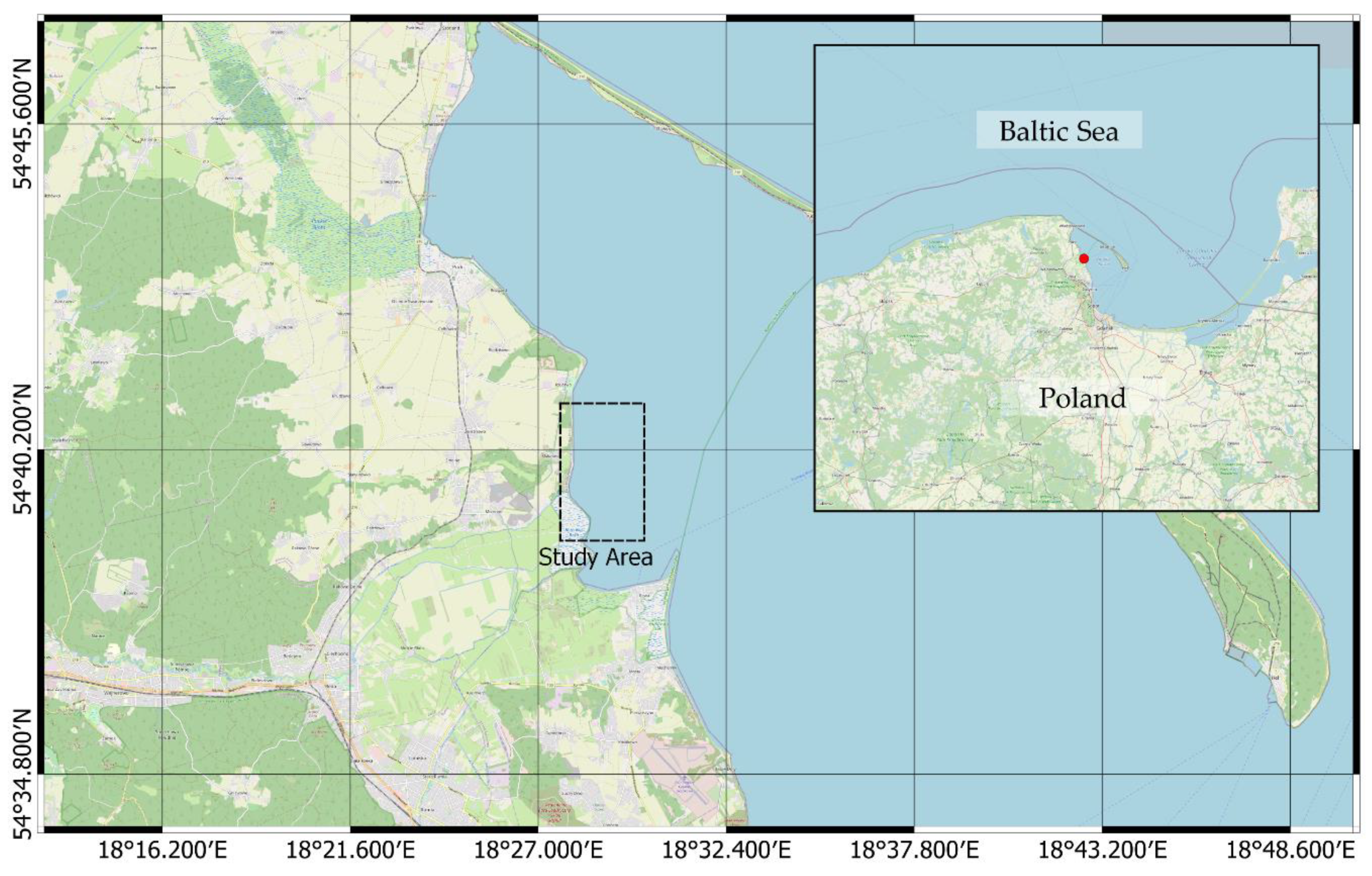
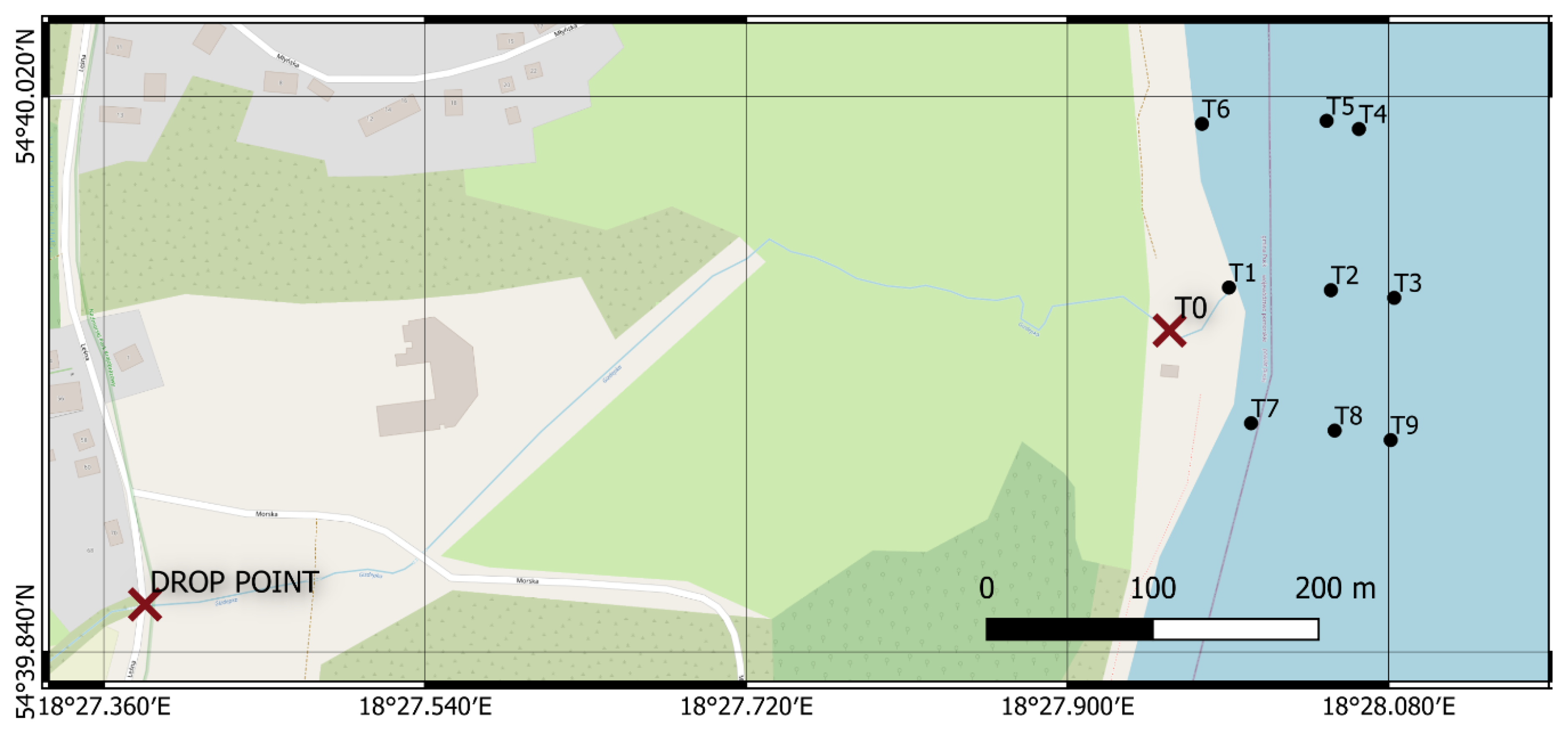
2.3. Measured Object
2.4. Fluorometer Measurement
2.5. UAV Measurement
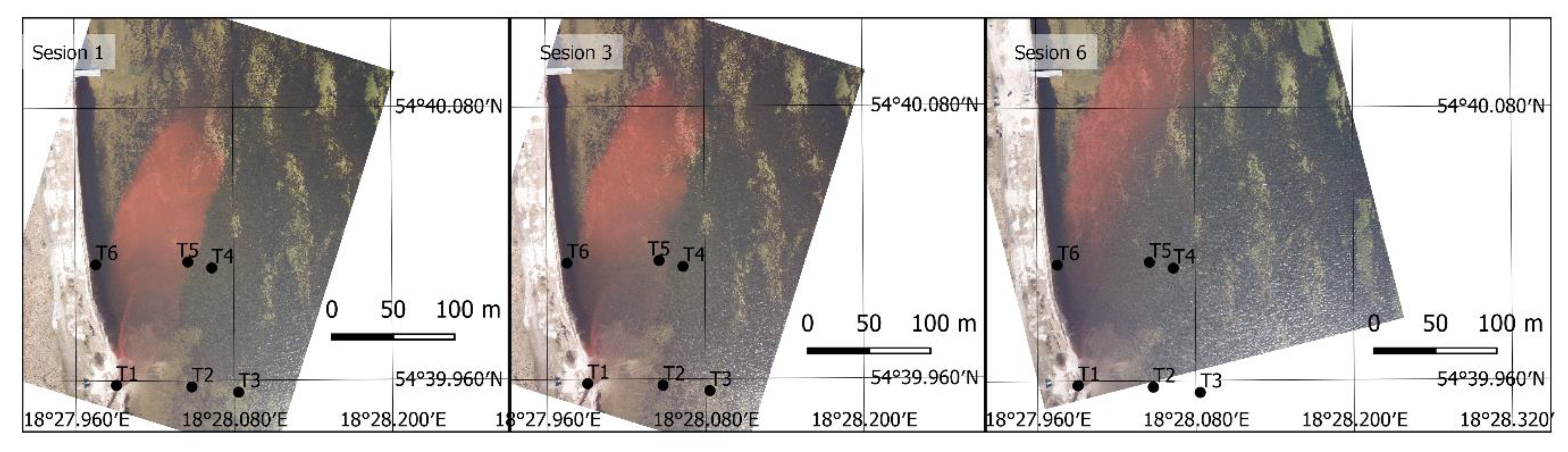
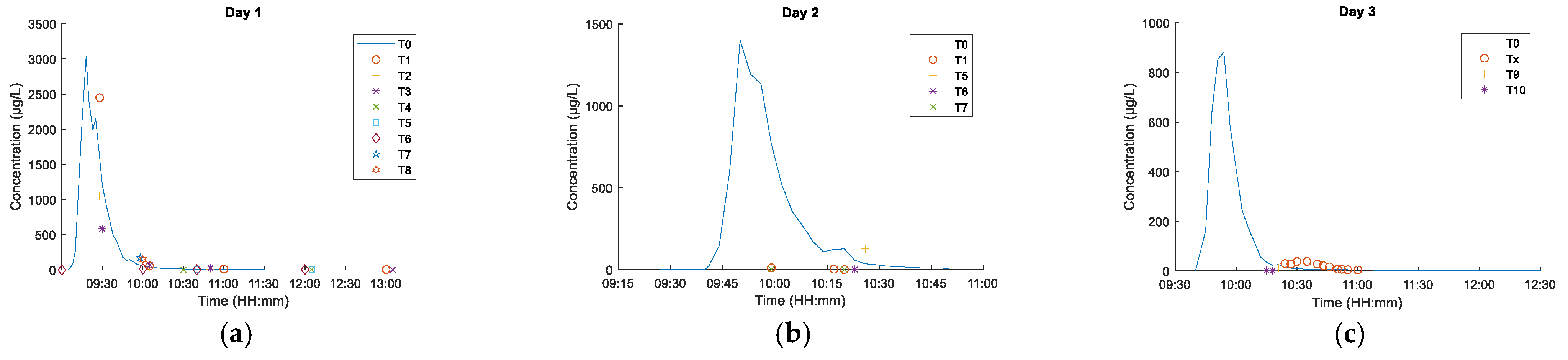
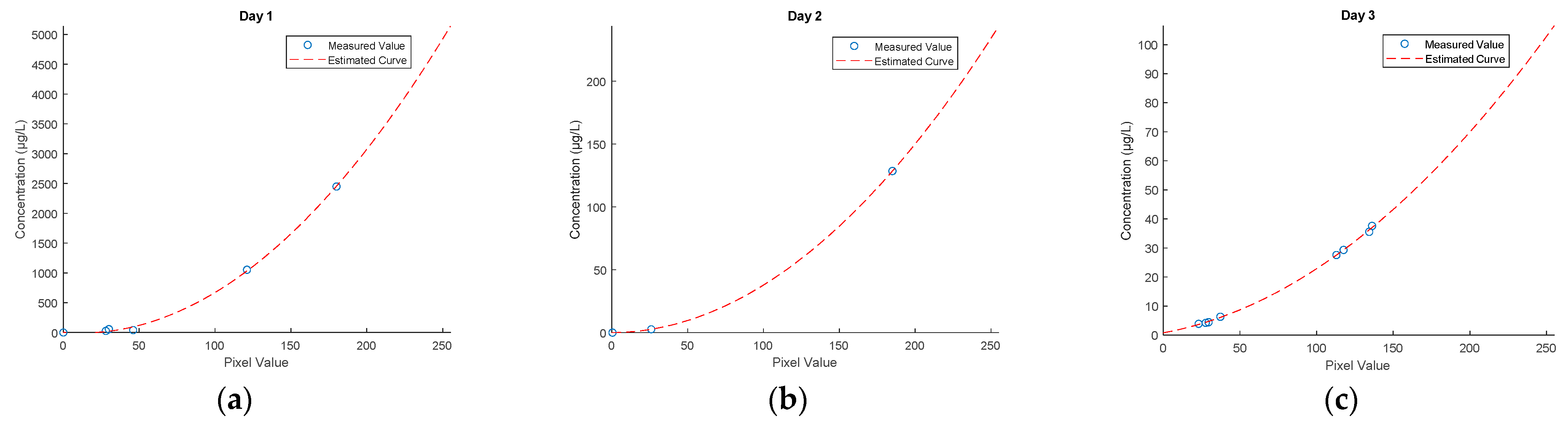
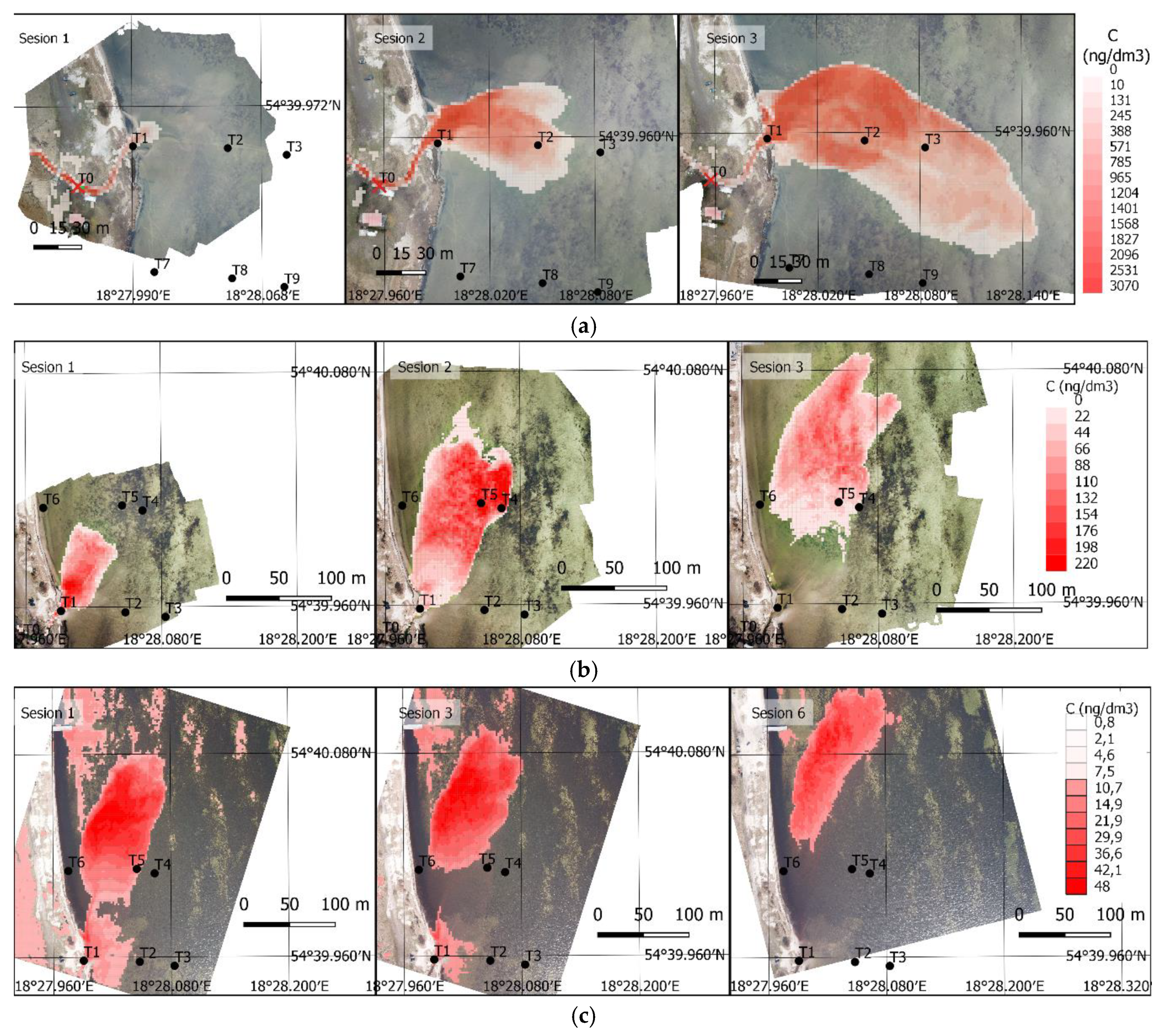
3. Results
3.1. Fluorometer
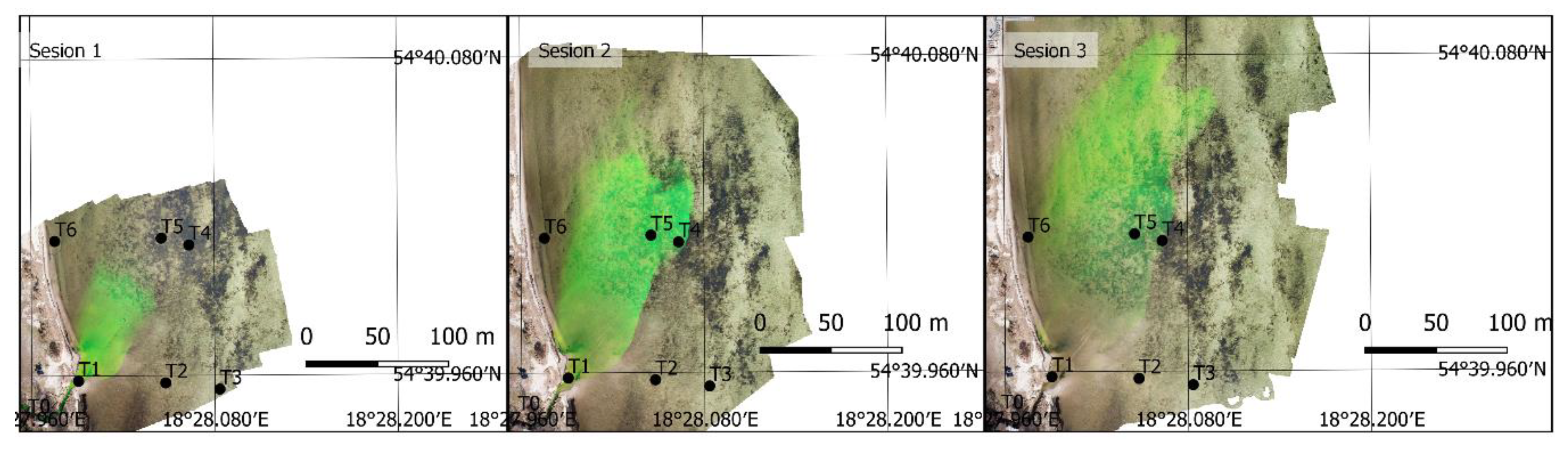
3.2. UAV Measurements
3.3. Final Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Remondino, F.; Barazzetti, L.; Nex, F.; Scaioni, M.; Sarazzi, D. Uav Photogrammetry for Mapping and 3d Modeling–Current Status And Future Perspectives. ISPRS Int. Arch. Photogramm. Remote Sens. Spat. Inf. Sci. 2012, 38, 25–31. [Google Scholar] [CrossRef]
- Specht, C.; Świtalski, E.; Specht, M. Application of an Autonomous/Unmanned Survey Vessel (ASV/USV) in Bathymetric Measurements. Pol. Marit. Res. 2017. [Google Scholar] [CrossRef]
- Stateczny, A.; Burdziakowski, P. Universal Autonomous Control and Management System for Multipurpose Unmanned Surface Vessel. Pol. Marit. Res. 2019, 26, 30–39. [Google Scholar] [CrossRef]
- Taddia, Y.; Russo, P.; Lovo, S.; Pellegrinelli, A. Multispectral UAV Monitoring of Submerged Seaweed in Shallow Water. Appl. Geomat. 2020, 12, 19–34. [Google Scholar] [CrossRef]
- Segales, A.; Gregor, R.; Rodas, J.; Gregor, D.; Toledo, S. Implementation of a Low Cost UAV for Photogrammetry Measurement Applications. In Proceedings of the 2016 International Conference on Unmanned Aircraft Systems (ICUAS), Arlington, VA, USA, 7–10 June 2016; pp. 926–932. [Google Scholar]
- Yang, B.; Hawthorne, T.L.; Torres, H.; Feinman, M. Using Object-Oriented Classification for Coastal Management in the East Central Coast of Florida: A Quantitative Comparison between UAV, Satellite, and Aerial Data. Drones 2019, 3, 60. [Google Scholar] [CrossRef]
- Burdziakowski, P.; Specht, C.; Dabrowski, P.S.; Specht, M.; Lewicka, O.; Makar, A. Using UAV Photogrammetry to Analyse Changes in the Coastal Zone Based on the Sopot Tombolo (Salient) Measurement Project. Sensors 2020, 20, 4000. [Google Scholar] [CrossRef]
- Mancini, F.; Dubbini, M.; Gattelli, M.; Stecchi, F.; Fabbri, S.; Gabbianelli, G. Using Unmanned Aerial Vehicles (UAV) for High-Resolution Reconstruction of Topography: The Structure from Motion Approach on Coastal Environments. Remote Sens. 2013, 5, 6880–6898. [Google Scholar] [CrossRef]
- Wyngaard, J.; Barbieri, L.; Thomer, A.; Adams, J.; Sullivan, D.; Crosby, C.; Parr, C.; Klump, J.; Raj Shrestha, S.; Bell, T. Emergent Challenges for Science SUAS Data Management: Fairness through Community Engagement and Best Practices Development. Remote Sens. 2019, 11, 1797. [Google Scholar] [CrossRef]
- Stateczny, A.; Gronska, D.; Motyl, W. Hydrodron—New Step for Professional Hydrography for Restricted Waters. In Proceedings of the 2018 Baltic Geodetic Congress (BGC Geomatics), Olsztyn, Poland, 21–23 June 2018; pp. 226–230. [Google Scholar]
- Chahl, J. Unmanned Aerial Systems (UAS) Research Opportunities. Aerospace 2015, 2, 189–202. [Google Scholar] [CrossRef]
- Inniss, L.; Simcock, A. The First Global Integrated Marine Assessment; United Nations, Ed.; Cambridge University Press: Cambridge, MA, USA, 2017; ISBN 9781108186148. [Google Scholar]
- VanderZwaag, D.L.; Powers, A. The Protection of the Marine Environment from Land-Based Pollution and Activities: Gauging the Tides of Global and Regional Governance. Int. J. Mar. Coast. Law 2008. [Google Scholar] [CrossRef]
- Wielgat, P.; Kalinowska, D.; Szymkiewicz, A.; Zima, P.; Jaworska-Szulc, B.; Wojciechowska, E.; Nawrot, N.; Matej-Lukowicz, K.; Dzierzbicka-Glowacka, L.A. Towards a Multi-Basin SWAT Model for the Migration of Nutrients and Pesticides to Puck Bay (Southern Baltic Sea). PeerJ 2021, 9, e10938. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.-M.; Whitfield, M. The Significance of the River Input of Chemical Elements to the Ocean. In Trace Metals in Sea Water; Springer: Boston, MA, USA, 1983; pp. 265–296. ISBN 0306411652. [Google Scholar]
- Wojciechowska, E.; Pietrzak, S.; Matej-Łukowicz, K.; Nawrot, N.; Zima, P.; Kalinowska, D.; Wielgat, P.; Obarska-Pempkowiak, H.; Gajewska, M.; Dembska, G.; et al. Nutrient Loss from Three Small-Size Watersheds in the Southern Baltic Sea in Relation to Agricultural Practices and Policy. J. Environ. Manag. 2019. [Google Scholar] [CrossRef]
- Rodrigues, M.; Guerreiro, M.; David, L.M.; Oliveira, A.; Menaia, J.; Jacob, J. Role of Environmental Forcings on Fecal Contamination Behavior in a Small Intermittent Coastal Stream: Case Study of the Aljezur Coastal Stream, Portugal. J. Environ. Eng. 2016, 142, 05016001. [Google Scholar] [CrossRef]
- Bailly Du Bois, P.; Garreau, P.; Laguionie, P.; Korsakissok, I. Comparison between Modelling and Measurement of Marine Dispersion, Environmental Half-Time and 137Cs Inventories after the Fukushima Daiichi Accident. Ocean Dyn. 2014. [Google Scholar] [CrossRef]
- Schmidt, W.E.; Woodward, B.T.; Millikan, K.S.; Guza, R.T.; Raubenheimer, B.; Elgar, S. A GPS-Tracked Surf Zone Drifter. J. Atmos. Ocean. Technol. 2003, 20, 1069–1075. [Google Scholar] [CrossRef]
- Spydell, M.S.; Feddersen, F.; Suanda, S. Inhomogeneous Turbulent Dispersion across the Nearshore Induced by Surfzone Eddies. J. Phys. Oceanogr. 2019, 49, 1015–1034. [Google Scholar] [CrossRef]
- Ren, C.; Liang, R.; Yu, C.; Bai, Y. A Numerical Model with Stokes Drift for Pollutant Transport within the Surf Zone on a Plane Beach. Acta Oceanol. Sin. 2019, 38, 102–112. [Google Scholar] [CrossRef]
- Hally-Rosendahl, K.; Feddersen, F.; Clark, D.B.; Guza, R.T. Surfzone to Inner-Shelf Exchange Estimated from Dye Tracer Balances. J. Geophys. Res. Ocean. 2015. [Google Scholar] [CrossRef]
- Feddersen, F.; Clark, D.B.; Guza, R.T. Modeling Surf Zone Tracer Plumes: 1. Waves, Mean Currents, and Low-Frequency Eddies. J. Geophys. Res. Ocean. 2011. [Google Scholar] [CrossRef]
- Clark, D.B.; Feddersen, F.; Guza, R.T. Modeling Surf Zone Tracer Plumes: 2. Transport and Dispersion. J. Geophys. Res. Ocean. 2011. [Google Scholar] [CrossRef]
- Oral, H.V.; Carvalho, P.; Gajewska, M.; Ursino, N.; Masi, F.; Hullebusch, E.D.; van Kazak, J.K.; Exposito, A.; Cipolletta, G.; Andersen, T.R.; et al. A Review of Nature-Based Solutions for Urban Water Management in European Circular Cities: A Critical Assessment Based on Case Studies and Literature. Blue-Green Syst. 2020, 2, 112–136. [Google Scholar] [CrossRef]
- Faustine, A.; Mvuma, A.N. Ubiquitous Mobile Sensing for Water Quality Monitoring and Reporting within Lake Victoria Basin. Wirel. Sens. Netw. 2014, 6, 257–264. [Google Scholar] [CrossRef]
- Zima, P. Simulation of the Impact of Pollution Discharged by Surface Waters from Agricultural Areas on the Water Quality of Puck Bay, Baltic Sea. Euro-Mediterr. J. Environ. Integr. 2019, 4, 16. [Google Scholar] [CrossRef]
- Zima, P. Numerical Analysis of an Impact of Planned Location of Sewage Discharge on Natura 2000 Areas—The Dead Vistula Region Case Study. Pol. Marit. Res. 2019. [Google Scholar] [CrossRef]
- Croucher, A.E.; O’Sullivan, M.J. Numerical Methods for Contaminant Transport in Rivers and Estuaries. Comput. Fluids 1998, 27, 861–878. [Google Scholar] [CrossRef]
- Oliveira, A.; Rodrigues, M.; Fortunato, A.B.; Guerreiro, M. Impact of Seasonal Bathymetric Changes and Inlet Morphology on the 3D Water Renewal and Residence Times of a Small Coastal Stream. J. Coast. Res. 2011, 64, 1555–1559. [Google Scholar]
- Sawicki, J.M.; Zima, P. Influence of Mixed Derivatives on the Mathematical Simulation of Pollutants Transfer. In Proceedings of the Fourth International Conference on Water Pollution Modelling, Measuring and Prediction, Bled, Slovenia, 18–20 June 1997. [Google Scholar] [CrossRef]
- Inman, D.L.; Tait, R.J.; Nordstrom, C.E. Mixing in the Surf Zone. J. Geophys. Res. 1971, 76, 3493–3514. [Google Scholar] [CrossRef]
- Spydell, M.; Feddersen, F.; Guza, R.T.; Schmidt, W.E. Observing Surf-Zone Dispersion with Drifters. J. Phys. Oceanogr. 2007, 37, 2920–2939. [Google Scholar] [CrossRef]
- Peregrine, D.H. Surf Zone Currents. Theor. Comput. Fluid Dyn. 1998. [Google Scholar] [CrossRef]
- Ren, J.; Li, W.; Gao, Y. Analyzing Dynamic Characteristics of River Plume in the Modaomen Mouth, Pearl River Estuary. J. Oceanogr. 2020, 76, 247–258. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, Z.; Lee, K. Modelling the Dispersion of Wastewater Discharges from Offshore Outfalls: A Review. Environ. Rev. 2011, 19, 107–120. [Google Scholar] [CrossRef]
- Zimmerman, M.; Dyson, S.; Murray, R. Comparison of Radiographic and Scintigraphic Findings of the Spinous Processes in the Equine Thoracolumbar Region. Vet. Radiol. Ultrasound 2011. [Google Scholar] [CrossRef] [PubMed]
- Buscombe, J.R.; Cwikla, J.B.; Thakrar, D.S.; Hilson, A.J.W. Scintigraphic Imaging of Breast Cancer a Review. Nucl. Med. Commun. 1997, 18, 698–709. [Google Scholar] [CrossRef]
- Naiki, H.; Higuchi, K.; Hosokawa, M.; Takeda, T. Fluorometric Determination of Amyloid Fibrils In Vitro Using the Fluorescent Dye, Thioflavine T. Anal. Biochem. 1989. [Google Scholar] [CrossRef]
- Abdel-Dayem, H.M.; Scott, A.; Macapinlac, H.; Larson, S. Tracer Imaging in Lung Cancer. Eur. J. Nucl. Med. 1994, 21, 57–81. [Google Scholar] [CrossRef]
- Matwiyoff, N.A.; Ott, D.G. Stable Isotope Tracers in the Life Sciences and Medicine. Science 1973, 181, 1125–1133. [Google Scholar] [CrossRef]
- Miller, L.L. Isotopic Tracers in Biology. An Introduction to Tracer Methodology. J. Am. Chem. Soc. 1958, 80, 5579–5580. [Google Scholar] [CrossRef]
- Kasnavia, T.; Vu, D.; Sabatini, D.A. Fluorescent Dye and Media Properties Affecting Sorption and Tracer Selection. Ground Water 1999, 37, 376–381. [Google Scholar] [CrossRef]
- Lange, J.; Olsson, O.; Sweeney, B.; Herbstritt, B.; Reich, M.; Alvarez-Zaldivar, P.; Payraudeau, S.; Imfeld, G. Fluorescent Tracers to Evaluate Pesticide Dissipation and Transformation in Agricultural Soils. Sci. Total Environ. 2018. [Google Scholar] [CrossRef]
- Leibundgut, C.; Maloszewski, P.; Klls, C. Tracers in Hydrology; John Wiley & Sons, Ltd.: Chichester, UK, 2009; ISBN 9780470747148. [Google Scholar]
- Rowiński, P.M.; Chrzanowski, M.M. Influence of Selected Fluorescent Dyes on Small Aquatic Organisms. Acta Geophys. 2011. [Google Scholar] [CrossRef]
- Nevins, T.D.; Kelley, D.H. Front Tracking for Quantifying Advection-Reaction-Diffusion. Chaos Interdiscip. J. Nonlinear Sci. 2017, 27, 043105. [Google Scholar] [CrossRef]
- Legleiter, C.J.; McDonald, R.R.; Nelson, J.M.; Kinzel, P.J.; Perroy, R.L.; Baek, D.; Seo, I.W. Remote Sensing of Tracer Dye Concentrations to Support Dispersion Studies in River Channels. J. Ecohydraulics 2019, 4, 131–146. [Google Scholar] [CrossRef]
- Clark, D.B.; Lenain, L.; Feddersen, F.; Boss, E.; Guza, R.T. Aerial Imaging of Fluorescent Dye in the near Shore. J. Atmos. Ocean. Technol. 2014, 31, 1410–1421. [Google Scholar] [CrossRef]
- Powers, C.; Hanlon, R.; Schmale, D. Tracking of a Fluorescent Dye in a Freshwater Lake with an Unmanned Surface Vehicle and an Unmanned Aircraft System. Remote Sens. 2018, 10, 81. [Google Scholar] [CrossRef]
- Sasano, M.; Imasato, M.; Yamano, H.; Oguma, H. Development of a Regional Coral Observation Method by a Fluorescence Imaging LIDAR Installed in a Towable Buoy. Remote Sens. 2016, 8, 48. [Google Scholar] [CrossRef]
- Baek, D.; Seo, I.W.; Kim, J.S.; Nelson, J.M. UAV-Based Measurements of Spatio-Temporal Concentration Distributions of Fluorescent Tracers in Open Channel Flows. Adv. Water Resour. 2019, 127, 76–88. [Google Scholar] [CrossRef]
- Bueno Hernández, D.; Mishra, R.K.; Muñoz, R.; Marty, J.L. Low Cost Optical Device for Detection of Fluorescence from Ochratoxin A Using a CMOS Sensor. Sens. Actuators B Chem. 2017, 246, 606–614. [Google Scholar] [CrossRef]
- Oh, S.W.; Brown, M.S.; Pollefeys, M.; Kim, S.J. Do It Yourself Hyperspectral Imaging with Everyday Digital Cameras. In Proceedings of the 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Las Vegas, NV, USA, 27–30 June 2016; pp. 2461–2469. [Google Scholar]
- Jiang, J.; Liu, D.; Gu, J.; Susstrunk, S. What Is the Space of Spectral Sensitivity Functions for Digital Color Cameras? In Proceedings of the 2013 IEEE Workshop on Applications of Computer Vision (WACV), Clearwater Beach, FL, USA, 15–17 January 2013; pp. 168–179. [Google Scholar]
- Deglint, J.; Kazemzadeh, F.; Cho, D.; Clausi, D.A.; Wong, A. Numerical Demultiplexing of Color Image Sensor Measurements via Non-Linear Random Forest Modeling. Sci. Rep. 2016. [Google Scholar] [CrossRef] [PubMed]
- Weisstein, E.W. Least Squares Fitting—Polynomial. Available online: https://mathworld.wolfram.com/LeastSquaresFittingPolynomial.html (accessed on 23 February 2021).
- Wojciechowska, E.; Nawrot, N.; Matej-Łukowicz, K.; Gajewska, M.; Obarska-Pempkowiak, H. Seasonal Changes of the Concentrations of Mineral Forms of Nitrogen and Phosphorus in Watercourses in the Agricultural Catchment Area (Bay of Puck, Baltic Sea, Poland). Water Sci. Technol. Water Supply 2019. [Google Scholar] [CrossRef]
- Matej-Lukowicz, K.; Wojciechowska, E.; Nawrot, N.; Dzierzbicka-Głowacka, L.A. Seasonal Contributions of Nutrients from Small Urban and Agricultural Watersheds in Northern Poland. PeerJ 2020, 8, e8381. [Google Scholar] [CrossRef]
- McInerney, D.; Kempeneers, P. Image (Re-)projections and Merging. In Open Source Geospatial Tools; McInerney, D., Kempeneers, P., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 99–127. ISBN 978-3-319-01824-9. [Google Scholar]
- Wilson, J.F.; Cobb, E.D.; Kilpatrick, F.A. Fluorometric Procedures for Dye Tracing; Department of the Interior, U.S. Geological Survey: Reston, WV, USA, 1986. [CrossRef]
- Buch, B.; Dittrich, P.-G.; Guo, S.; Zhang, C.; Radtke, L.; Rosenberger, M.; Notni, G. A Comparative Study of Methods and Algorithms for Spatially Resolved Spectral Value Reconstruction with Multispectral Resolving Filter-on-Chip CMOS Sensors. In Proceedings of the Physics and Simulation of Optoelectronic Devices XXVII, San Francisco, CA, USA, 26 February 2019; p. 34. [Google Scholar]



| Name | Date | Tracer | Tracer Concentration |
|---|---|---|---|
| Day 1 | 17 February 2019 | Uranine | 500 g/10 dm3 H2O |
| Day 2 | 2 March 2019 | Uranine | 250 g/10 dm3 H2O |
| Day 3 | 23 March 2019 | Rhodamine WT | 1 dm3 @ 20% |
| Parameter | Rhodamine WT | Uranine |
|---|---|---|
| Minimum detected concentration | 0.01 ppb | 0.01 ppb |
| Linear range of concentration—fluorescence relationship | 0–500 ppb | 0–200 ppb |
| Excitation electromagnetic wavelength | 550 nm | 485 nm |
| Recorded electromagnetic wavelength | 610 nm | 540 nm |
| Sesion | Time | T1 | Pixel Value | Pixel Value | Pixel Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 09:43 | 715 | 103 | 718 | 7081 | 1 | 0 | 3 | 2,83 | 1 | 0 | 3 | 3 |
| 2 | 09:55 | 2449 | 180 | 2454 | 24,532 | 1052 | 121 | 1032 | 1042 | 1 | 0 | 3 | 3 |
| 3 | 10:07 | 2069 | 166 | 2062 | 2051 | 1331 | 135 | 1315 | 1322 | 584 | 93 | 568 | 567 |
| 4 | 10:28 | 55 | 30 | 21 | 201 | 40 | 46 | 95 | 84 | 285 | 69 | 278 | 270 |
| 5 | 10:58 | 12 | 25 | 7 | 3 | 12 | 0 | 3 | 3 | 70 | 42 | 72 | 62 |
| 6 | 11:26 | 10 | 0 | 3 | 3 | 8 | 0 | 3 | 3 | 26 | 28 | 15 | 21 |
| 7 | 12:02 | 7 | 0 | 3 | 3 | 7 | 0 | 3 | 3 | 1 | 0 | 3 | 3 |
| 8 | 13:03 | 6 | 0 | 3 | 3 | 5 | 0 | 3 | 3 | 1 | 0 | 3 | 3 |
| Sesion | Time | T1 | Pixel Value | Mean | |
|---|---|---|---|---|---|
| 1 | 09:43 | 10:25 | 2,932,284 | 1176 | 30,002,168 |
| 2 | 09:55 | 10:28 | 2,758,726 | 1129 | 28,145,903 |
| 3 | 10:07 | 10:31 | 37,611,736 | 1362 | 37,911,632 |
| 4 | 10:28 | 10:34 | 35,541,976 | 1343 | 37,062,427 |
| 5 | 10:58 | 10:52 | 6,331,872 | 372 | 6,160,352 |
| 6 | 11:26 | 10:55 | 4,430,896 | 295 | 4,758,875 |
| 7 | 12:02 | 10:58 | 3,906,064 | 23 | 36,958 |
| 8 | 13:03 | 11:01 | 4,237,472 | 276 | 4,436,768 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burdziakowski, P.; Zima, P.; Wielgat, P.; Kalinowska, D. Tracking Fluorescent Dye Dispersion from an Unmanned Aerial Vehicle. Sensors 2021, 21, 3905. https://doi.org/10.3390/s21113905
Burdziakowski P, Zima P, Wielgat P, Kalinowska D. Tracking Fluorescent Dye Dispersion from an Unmanned Aerial Vehicle. Sensors. 2021; 21(11):3905. https://doi.org/10.3390/s21113905
Chicago/Turabian StyleBurdziakowski, Pawel, Piotr Zima, Pawel Wielgat, and Dominika Kalinowska. 2021. "Tracking Fluorescent Dye Dispersion from an Unmanned Aerial Vehicle" Sensors 21, no. 11: 3905. https://doi.org/10.3390/s21113905
APA StyleBurdziakowski, P., Zima, P., Wielgat, P., & Kalinowska, D. (2021). Tracking Fluorescent Dye Dispersion from an Unmanned Aerial Vehicle. Sensors, 21(11), 3905. https://doi.org/10.3390/s21113905








