The Influence of Plasticizers on the Response Characteristics of the Surfactant Sensor for Cationic Surfactant Determination in Disinfectants and Antiseptics
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Membrane Preparation and Surfactant Sensors Fabrication
2.3. Response Measurements
2.4. Potentiometric Titrations
3. Results and Discussion
3.1. Plasticizer Properties
3.2. Potentiometric Response
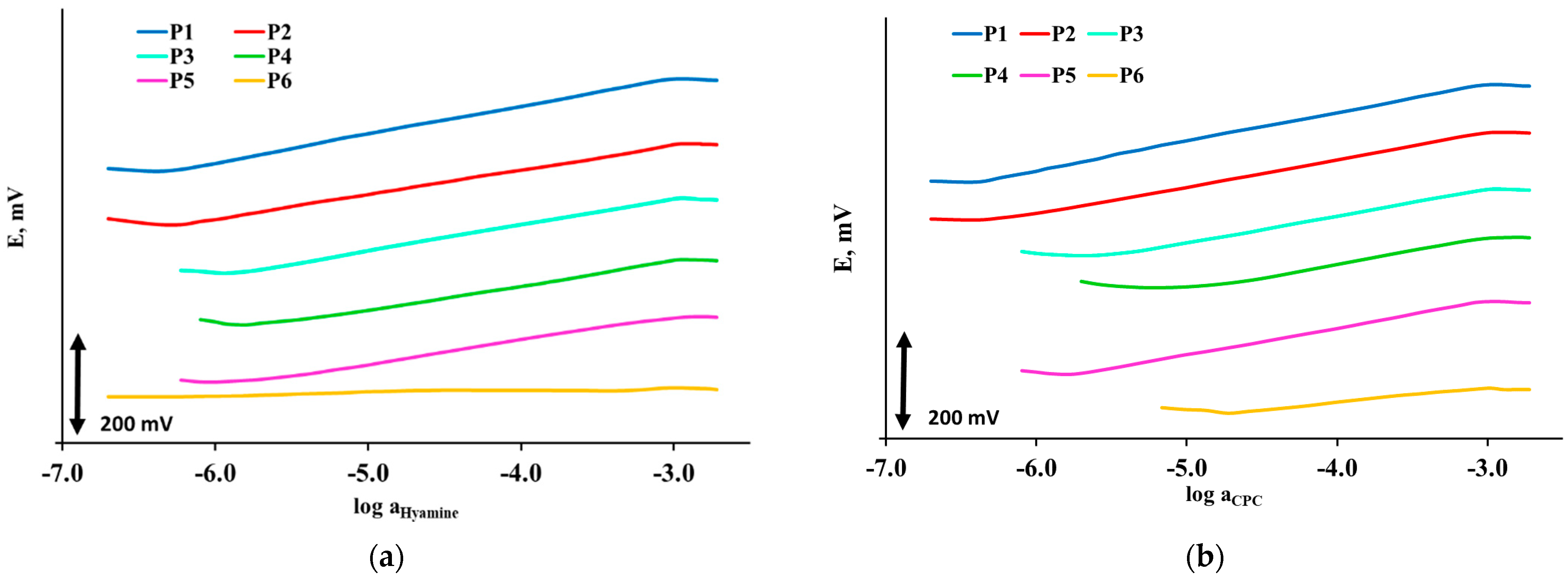
3.2.1. Surfactant Sensors Responses to Hyamine 1622
3.2.2. Surfactant Sensors Responses to CPC
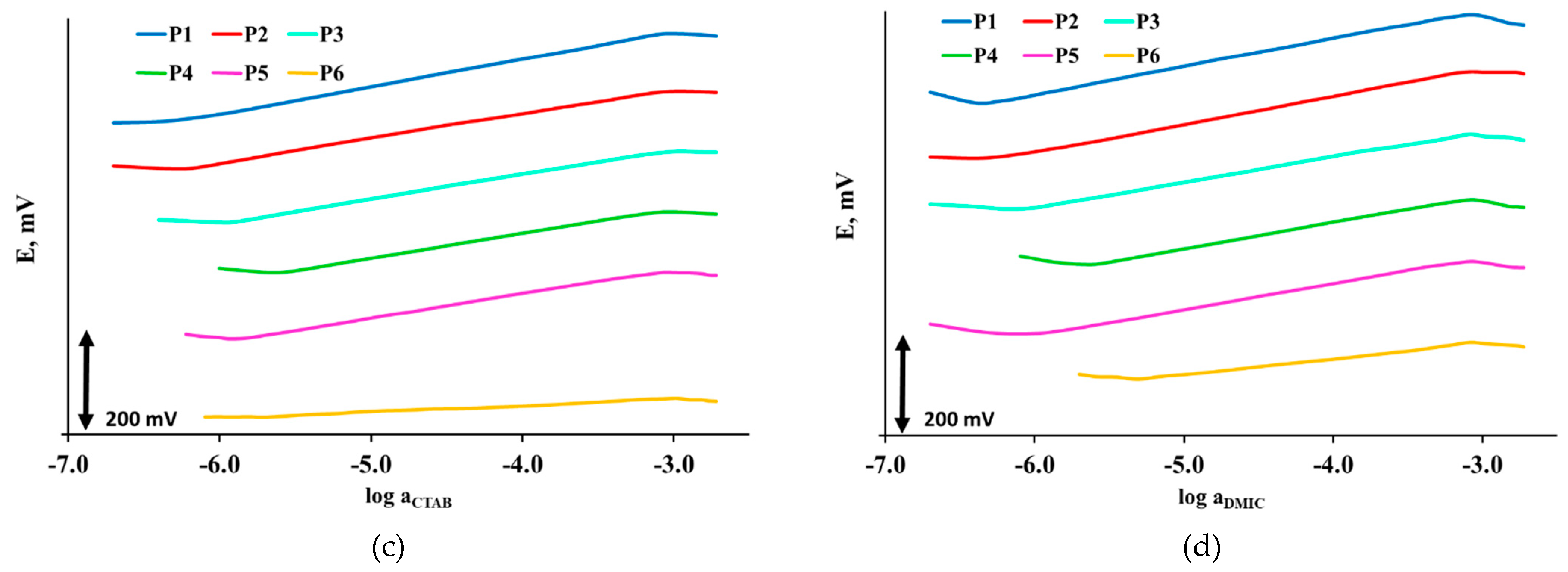
3.2.3. Surfactant Sensors Responses to CTAB
3.2.4. Surfactant Sensors Responses to DMIC
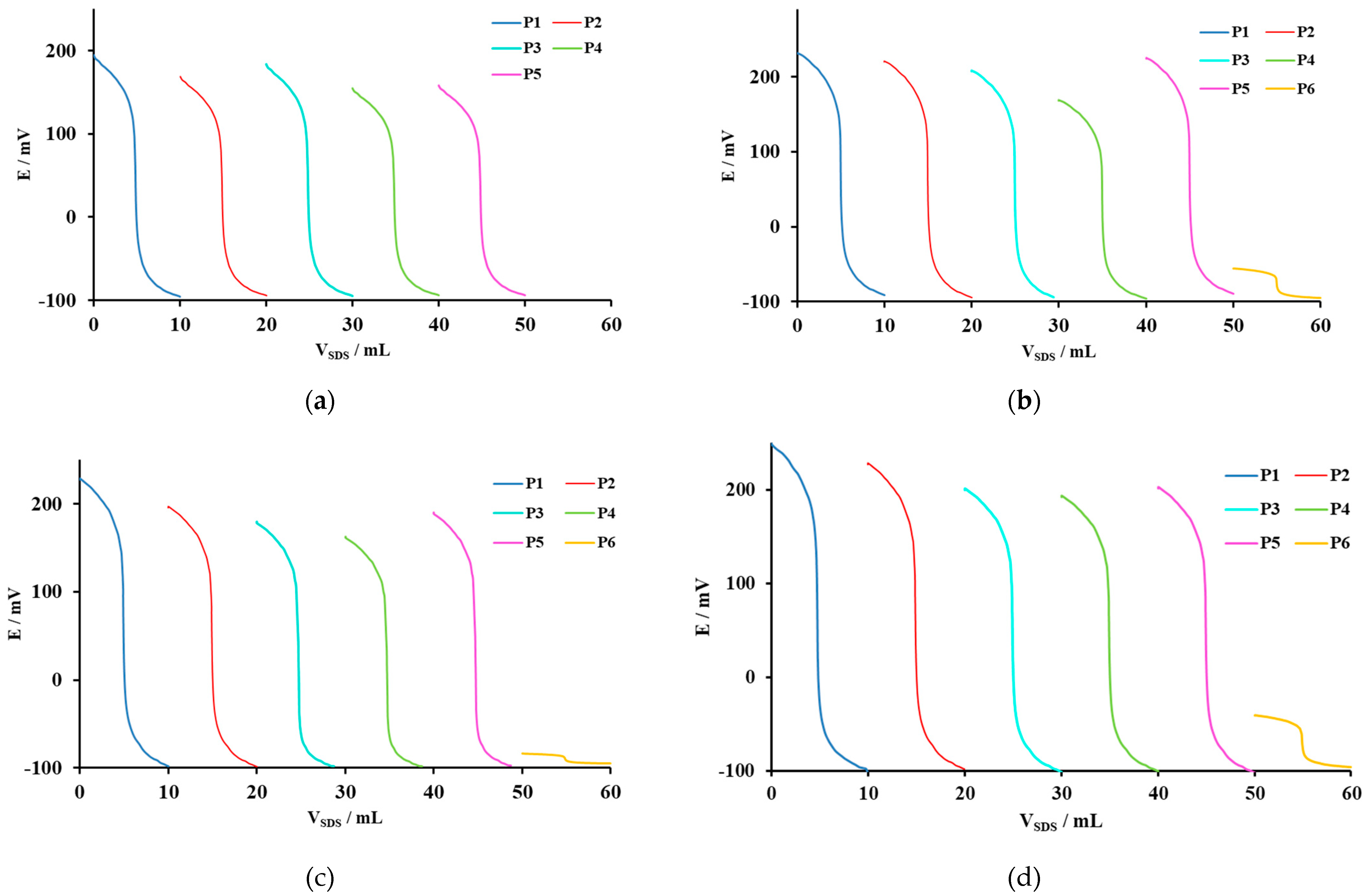
3.3. Potentiometric Titrations
3.4. Titration of Commercial Disinfectants and Antiseptics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frend, A.J.; Moody, G.J.; Thomas, J.D.R.; Birch, B.J. Flow injection analysis with tubular membrane ion-selective electrodes in the presence of anionic surfactants. Analyst 1983, 108, 1357. [Google Scholar] [CrossRef]
- Sakač, N.; Jozanović, M.; Karnaš, M.; Sak-Bosnar, M. A New Sensor for Determination of Anionic Surfactants in Detergent Products with Carbon Nanotubes as Solid Contact. J. Surfactants Deterg. 2017, 20, 881–889. [Google Scholar] [CrossRef]
- Seguí, M.J.; Lizondo-Sabater, J.; Benito, A.; Martínez-Máñez, R.; Pardo, T.; Sancenón, F.; Soto, J. A new ion-selective electrode for anionic surfactants. Talanta 2007, 71, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Fizer, M.; Fizer, O.; Sidey, V.; Mariychuk, R.; Studenyak, Y. Experimental and theoretical study on cetylpyridinium dipicrylamide—A promising ion-exchanger for cetylpyridinium selective electrodes. J. Mol. Struct. 2019, 1187, 77–85. [Google Scholar] [CrossRef]
- Fizer, O.; Fizer, M.; Sidey, V.; Studenyak, Y. Predicting the end point potential break values: A case of potentiometric titration of lipophilic anions with cetylpyridinium chloride. Microchem. J. 2021, 160, 105758. [Google Scholar] [CrossRef]
- Kovács, B.; Csóka, B.; Nagy, G.; Ivaska, A. All-solid-state surfactant sensing electrode using conductive polymer as internal electric contact. Anal. Chim. Acta 2001, 437, 67–76. [Google Scholar] [CrossRef]
- Zareh, M.M. Blank membranes versus ionophore-based membranes for the selective determination of H+. Anal. Sci. 2009, 25, 1131–1135. [Google Scholar] [CrossRef][Green Version]
- Pechenkina, I.A.; Mikhelson, K.N. Materials for the ionophore-based membranes for ion-selective electrodes: Problems and achievements (review paper). Russ. J. Electrochem. 2015, 51, 93–102. [Google Scholar] [CrossRef]
- Carey, C. Plasticizer Effects in the PVC Membrane of the Dibasic Phosphate Selective Electrode. Chemosensors 2015, 3, 284–294. [Google Scholar] [CrossRef]
- Mahajan, R.K.; Shaheen, A. Effect of various additives on the performance of a newly developed PVC based potentiometric sensor for anionic surfactants. J. Colloid Interface Sci. 2008, 326, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Guilbault, G.G.; Durst, R.A.; Frant, M.S.; Freiser, H.; Hansen, E.H.; Light, T.S.; Pungor, E.; Rechnitz, G.; Rice, N.M.; Rohm, T.J.W.; et al. Recommendations for nomenclature of ion-selective electrodes. Pure Appl. Chem. 1976, 48, 127–132. [Google Scholar]
- Zahran, E.M.; New, A.; Gavalas, V.; Bachas, L.G. Polymeric plasticizer extends the lifetime of PVC-membrane ion-selective electrodes. Analyst 2014, 139, 757–763. [Google Scholar] [CrossRef]
- Karnaš, M.; Sakač, N.; Jozanović, M.; Tsakiri, M.; Kopriva, M. The Influence of Plasticisers on Response Characteristics of Anionic Surfactant Potentiometric Sensor. Int. J. Electrochem. Sci. 2017, 12, 5921–5933. [Google Scholar] [CrossRef]
- Abramova, N.; Bratov, A. Application of Photocured Polymer Ion Selective Membranes for Solid-State Chemical Sensors. Chemosensors 2015, 3, 190–199. [Google Scholar] [CrossRef]
- Dinten, O.; Spichiger, U.E.; Chaniotakis, N.; Gehrig, P.; Rusterholz, B.; Morf, W.E.; Simon, W. Lifetime of neutral-carrier-based liquid membranes in aqueous samples and blood and the lipophilicity of membrane components. Anal. Chem. 1991, 63, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Eugster, R.; Rosatzin, T.; Rusterholz, B.; Aebersold, B.; Pedrazza, U.; Rüegg, D.; Schmid, A.; Spichiger, U.E.; Simon, W. Plasticizers for liquid polymeric membranes of ion-selective chemical sensors. Anal. Chim. Acta 1994, 289, 1–13. [Google Scholar] [CrossRef]
- Harsányi, G. Polymer Films in Sensor Applications, 1st ed.; Technomic Publishing Co. Inc.: Lancaster, UK; Basel, Switzerland, 1995; ISBN 9780203742907. [Google Scholar]
- Madunić-Čačić, D.; Sak-Bosnar, M.; Matešić-Puač, R.; Grabarić, Z. Determination of anionic surfactants in real systems using 1,3-Didecyl-2-methyl-imidazolium-tetraphenylborate as sensing material. Sens. Lett. 2008, 6, 339–346. [Google Scholar] [CrossRef]
- Bakshi, U.; Bakshi, A.; Bakshi, K. Electrical Measurement and Instrumentation; Technical Publications: Pune, India, 2009; ISBN 9788184312553. [Google Scholar]
- Sakač, N.; Marković, D.; Šarkanj, B.; Madunić-Čačić, D.; Hajdek, K.; Smoljan, B.; Jozanović, M. Direct Potentiometric Study of Cationic and Nonionic Surfactants in Disinfectants and Personal Care Products by New Surfactant Sensor Based on 1,3-Dihexadecyl−1H-benzo[d]imidazol−3-ium. Molecules 2021, 26, 1366. [Google Scholar] [CrossRef] [PubMed]
- ISO 2871-2:2010 Surface Active Agents—Detergents—Determination of Cationic-Active Matter Content—Part 2: Cationic-Active Matter of Low Molecular Mass (between 200 and 500); International Organization for Standardization: Geneva, Switzerland, 2010.
| Plasticizer Name | Abbreviation | Mr g/mol | Lipophilicity * | Dielectric Constant (ε) ** | Resistance (MΩ) | Membrane Visual Appearance |
|---|---|---|---|---|---|---|
| 2-nitrophenyl-octyl-ether | P1 | 251.32 | 5.80 | 21 | 55 | opaque |
| bis(2-ethylhexyl) phthalate | P2 | 390.56 | 7.60 | 5.17 | 57 | milky |
| bis(2-ethylhexyl) sebacate | P3 | 426.67 | 10.08 | 3.89 | 62 | milky |
| 2-nitrophenyl phenyl ether | P4 | 215.20 | 3.39 | 9.8 | 61 | transparent |
| dibutyl phthalate | P5 | 278.34 | 4.79 | 6.43 | 59 | opaque |
| dibutyl sebacate | P6 | 314.46 | 6.30 | 4.6 | 74 | opaque |
| Parameters | Surfactant Sensor with Plasticizer | |||||
|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | |
| Slope, mV/decade of activity | 58.78 ± 0.6 | 53.61 ± 1.1 | 56.61 ± 0.5 | 50.6 ± 1.0 | 50.67 ± 1.1 | -/- |
| Standard error | 0.4 | 0.6 | 0.3 | 0.7 | 0.7 | -/- |
| Correlation coefficient, R2 | 0.9935 | 0.9993 | 0.9992 | 0.9964 | 0.9965 | -/- |
| Detection limit, M | 2.1 × 10−7 | 4.2 × 10−7 | 0.1 x 10−6 | 0.2 × 10−6 | 0.1 x 10−6 | -/- |
| Useful conc. Range, M | 3.2 × 10−3–3.9 × 10−7 | 3.2 × 10−3–5.9 x 10−7 | 3.2 × 10−3–1.2 × 10−6 | 3.2 × 10−3–1.6 × 10−6 | 3.2 × 10−3–1.2 × 10−6 | -/- |
| Parameters | Surfactant Sensor with Plasticizer | |||||
|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | |
| Slope, mV/decade of activity | 59.1 ± 0.5 | 57.8 ± 0.6 | 55.8 ± 0.4 | 55.9 ± 1.3 | 57.47 ± 0.6 | 31.9 ± 0.2 |
| Standard error | 0.4 | 0.4 | 0.4 | 0.2 | 0.3 | 0.5 |
| Correlation coefficient, R2 | 0.9945 | 0.9997 | 0.9986 | 0.9968 | 0.9996 | 0.9955 |
| Detection limit, M | 2.1 × 10−7 | 4.2 × 10−7 | 1.8 × 10−6 | 0.1 × 10−5 | 0.2 × 10−6 | 0.2 × 10−5 |
| Useful conc. Range, M | 3.2 × 10−3–3.9 × 10−7 | 3.2 × 10−3–5.9 × 10−7 | 3.2 × 10−3–3.5 × 10−6 | 3.2 × 10−3–1.3 × 10−5 | 3.2 × 10−3–1.9 × 10−6 | 3.2 × 10−3–1.8 × 10−5 |
| Parameters | Surfactant Sensor with Plasticizer | |||||
|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | |
| Slope, mV/decade of activity | 59.2 ± 0.7 | 53.5 ± 0.5 | 52.8 ± 0.5 | 53.01 ± 0.6 | 52.9 ± 0.5 | 14.1 ± 0.1 |
| Standard error | 0.7 | 0.7 | 0.4 | 0.3 | 0.5 | 0.044 |
| Correlation coefficient, R2 | 0.9996 | 0.9991 | 0.9992 | 0.9984 | 0.9999 | 0.9986 |
| Detection limit, M | 2.1 × 10−7 | 4.2 × 10−7 | 0.1 × 10−6 | 0.9 × 10−6 | 0.2 × 10−6 | 1.9 × 10−6 |
| Useful conc. Range, M | 3.2 × 10−3–3.9 × 10−7 | 3.2 × 10−3–5.9 × 10−7 | 3.2 × 10−3–1.2 × 10−6 | 3.2 × 10−3–2.6 × 10−6 | 3.2 × 10−3–1.6 × 10−6 | 3.2 × 10−3–3.9 × 10−6 |
| Parameters | Surfactant Sensor with plasticizer | |||||
|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | |
| Slope, mV/decade of activity | 58.4 ± 0.6 | 59.6 ± 0.7 | 54.6 ± 0.7 | 55.1 ± 0.7 | 55.9 ± 0.5 | 32.9 ± 0.4 |
| Standard error | 0.2 | 0.4 | 0.2 | 0.4 | 0.7 | 0.6 |
| Correlation coefficient, R2 | 0.9994 | 0.9997 | 0.9998 | 0.9997 | 0.9999 | 0.9994 |
| Detection limit, M | 2.1 × 10−7 | 4.2 × 10−7 | 5.7 × 10−7 | 0.9 × 10−6 | 0.1 × 10−6 | 2.9 × 10−6 |
| Useful conc. Range, M | 3.2 × 10−3–3.9 × 10−7 | 3.2 × 10−3–5.9 × 10−7 | 3.2 × 10−3–7.9 × 10−7 | 3.2 × 10−3–2.6 × 10−6 | 3.2 × 10−3–1.2 × 10−6 | 3.2 × 10−3–4.9 × 10−6 |
| DMI-TPB Sensor Plasticizer Type | |||||||
|---|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | ||
| Hyamine 1622 with SDS | ΔE, mV | 290.1 | 263.4 | 278.2 | 248.6 | 252.4 | -/- |
| dE/dV | 61.5 | 59.1 | 43:5 | 43.1 | 55.3 | -/- | |
| SDS consumption in EP, mL | 4.83 | 4.83 | 4.79 | 5.07 | 4.79 | -/- | |
| SD of EP | 0.013 | 0.023 | 0.018 | 0.054 | 0.018 | -/- | |
| CPC with SDS | ΔE, mV | 321.7 | 314.9 | 304.0 | 263.8 | 313.3 | 39.4 |
| dE/dV | 69.4 | 67.7 | 46.4 | 45.0 | 62.2 | 9.6 | |
| SDS consumption in EP, mL | 4.78 | 4.76 | 4.69 | 4.79 | 4.72 | 4.86 | |
| SD | 0.016 | 0.018 | 0.016 | 0.059 | 0.019 | 0.035 | |
| CTAB with SDS | ΔE, mV | 325.7 | 294.7 | 276.2 | 260.2 | 286.1 | 11.6 |
| dE/dV | 71.5 | 67.9 | 54.8 | 56.9 | 65.2 | 13.9 | |
| SDS consumption in EP, mL | 4.97 | 4.92 | 4.92 | 4.91 | 4.94 | 4.96 | |
| SD | 0.024 | 0.015 | 0.037 | 0.035 | 0.038 | 0.027 | |
| DMIC with SDS | ΔE, mV | 357.6 | 325.2 | 300.3 | 292.5 | 303.2 | 54.6 |
| dE/dV | 66.6 | 61.4 | 52.4 | 46.2 | 59.1 | 33.8 | |
| SDS consumption in EP, mL | 4.73 | 4.70 | 4.69 | 4.75 | 4.71 | 4.96 | |
| SD | 0.027 | 0.015 | 0.014 | 0.026 | 0.015 | 0.025 | |
| Commercial Sample | CPC Taken/Mol | CPC Found/Mol |
|---|---|---|
| 1 | 1 × 10−5 | 0.989 × 10−5 |
| 1 × 10−4 | 0.982 × 10−5 | |
| 2 | 1 × 10−5 | 0.996 × 10−5 |
| 1 × 10−4 | 0.992 × 10−5 | |
| 3 | 1 × 10−5 | 0.989 × 10−5 |
| 1 × 10−4 | 0.991 × 10−5 |
| Product | CATIONIC SURFACTANT CONTENT * | ||||
|---|---|---|---|---|---|
| DMI-TPB Sensor with P1 | Surfactant Sensor ** | Two-Phase Titration | |||
| % | RSD (%) | % | RSD (%) | % | |
| A | 4.38 | 0.36 | 4.32 | 0.41 | 4.33 |
| B | 5.26 | 0.38 | 5.13 | 0.62 | 5.15 |
| C | 4.70 | 0.31 | 4.76 | 0.42 | 4.71 |
| D | 4.80 | 0.35 | 4.72 | 0.48 | 4.77 |
| E | 0.06 | 0.26 | 0.07 | 0.35 | 0.07 |
| F | 0.15 | 0.45 | 0.15 | 0.25 | 0.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakač, N.; Madunić-Čačić, D.; Karnaš, M.; Đurin, B.; Kovač, I.; Jozanović, M. The Influence of Plasticizers on the Response Characteristics of the Surfactant Sensor for Cationic Surfactant Determination in Disinfectants and Antiseptics. Sensors 2021, 21, 3535. https://doi.org/10.3390/s21103535
Sakač N, Madunić-Čačić D, Karnaš M, Đurin B, Kovač I, Jozanović M. The Influence of Plasticizers on the Response Characteristics of the Surfactant Sensor for Cationic Surfactant Determination in Disinfectants and Antiseptics. Sensors. 2021; 21(10):3535. https://doi.org/10.3390/s21103535
Chicago/Turabian StyleSakač, Nikola, Dubravka Madunić-Čačić, Maja Karnaš, Bojan Đurin, Ivan Kovač, and Marija Jozanović. 2021. "The Influence of Plasticizers on the Response Characteristics of the Surfactant Sensor for Cationic Surfactant Determination in Disinfectants and Antiseptics" Sensors 21, no. 10: 3535. https://doi.org/10.3390/s21103535
APA StyleSakač, N., Madunić-Čačić, D., Karnaš, M., Đurin, B., Kovač, I., & Jozanović, M. (2021). The Influence of Plasticizers on the Response Characteristics of the Surfactant Sensor for Cationic Surfactant Determination in Disinfectants and Antiseptics. Sensors, 21(10), 3535. https://doi.org/10.3390/s21103535








