The Impact of a Novel Immersive Virtual Reality Technology Associated with Serious Games in Parkinson’s Disease Patients on Upper Limb Rehabilitation: A Mixed Methods Intervention Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Quantitative Intervention Phase Design
2.1.1. Sampling and Participants
2.1.2. Intervention
2.1.3. Data Collection and Outcome Measures
2.1.4. Data Analysis
2.2. Qualitative Phase Design
2.2.1. Sampling and Participants
2.2.2. Data Collection
2.2.3. Data Analysis
2.3. Ethical Considerations of the Mixed Methods Intervention
2.4. Embedded Integration Procedure for Quantitative and Qualitative Content
3. Results
3.1. Quantitative Findings
3.2. Qualitative Findings
3.2.1. Results of the Thematic Analysis
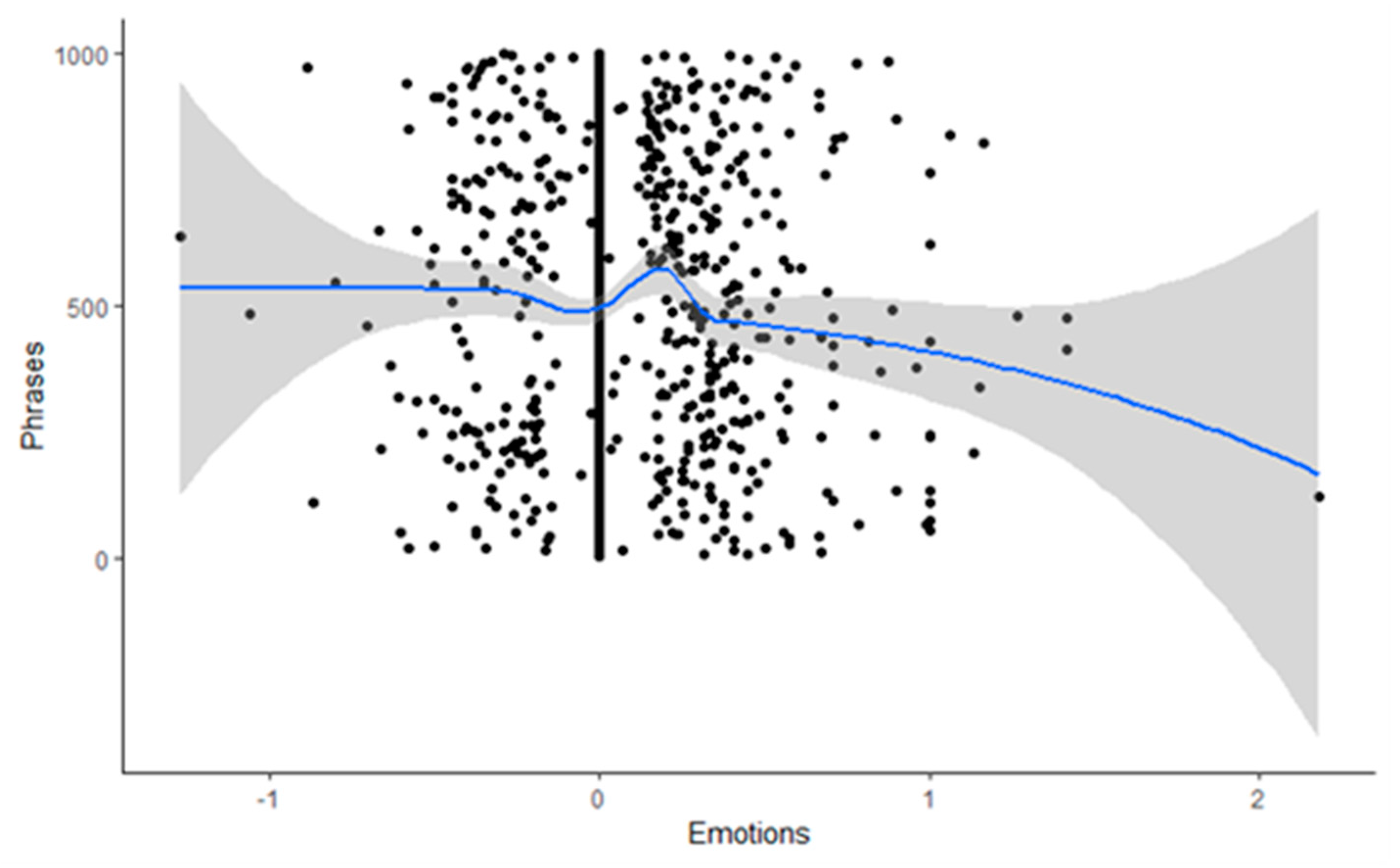
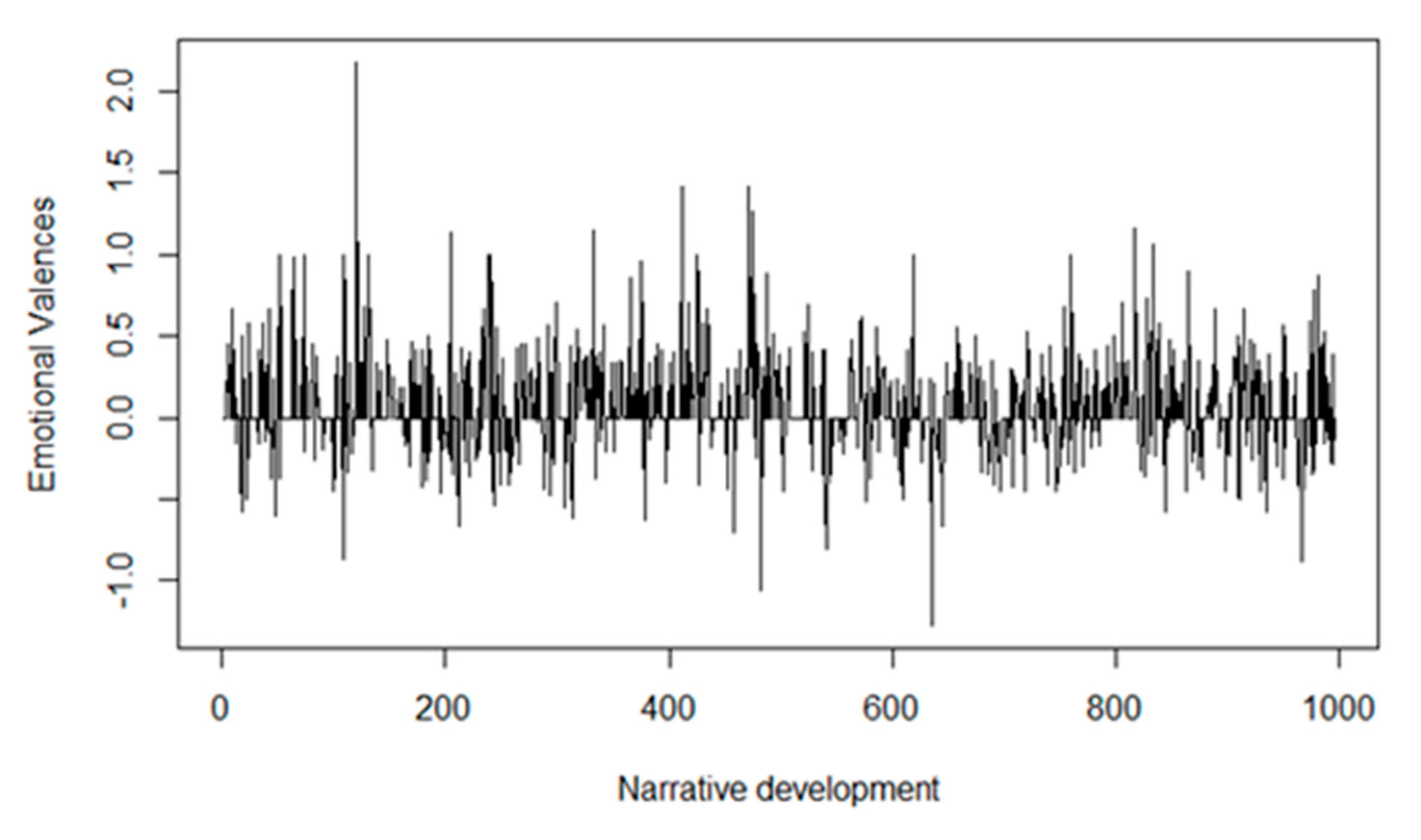
3.2.2. Results of the Acceptance-Rejection (Polarity) Analysis
3.3. Mixed Methods Findings (Integration)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ARAT | Action research arm test | BBT | Box and block test |
| CSQ-8 | Client satisfaction questionnaire | FG | Flip game |
| GG | Grab game | MMI | Mixed methods intervention |
| MMR | Mixed methods research | OR2-LMC | Oculus Rift 2 plus leap motion controller |
| PD | Parkinson’s disease | PPT | Purdue pegboard test |
| QUAL | qualitative | QUAN | quantitative |
| QUART | Qualitative research in trials | RG | Reach game |
| SG | Sequence game | TIDieR | Template for intervention description and replication |
| VR | virtual reality |
References
- Cano-De-La-Cuerda, R.; Vela-Desojo, L.; Miangolarra-Page, J.C.; Macías-Macías, Y.; Muñoz-Hellín, E. Axial rigidity and quality of life in patients with Parkinson’s disease: A preliminary study. Qual. Life Res. 2010, 20, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Lebel, K.; Duval, C.; Goubault, E.; Bogard, S.; Blanchet, P.J. Can We Predict the Motor Performance of Patients with Parkinson’s Disease Based on Their Symptomatology? Front. Bioeng. Biotechnol. 2020, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Fernández-González, P.; Carratalá-Tejada, M.; Monge-Pereira, E.; Collado-Vázquez, S.; Baeza, P.S.-H.; Cuesta-Gómez, A.; Oña-Simbaña, E.D.; Jardón-Huete, A.; Molina-Rueda, F.; De Quirós, C.B.-B.; et al. Leap motion controlled video game-based therapy for upper limb rehabilitation in patients with Parkinson’s disease: A feasibility study. J. Neuroeng. Rehabil. 2019, 16, 133. [Google Scholar] [CrossRef] [PubMed]
- Van Der Marck, M.A.; Munneke, M.; Mulleners, W.; Hoogerwaard, E.M.; Borm, G.F.; Overeem, S.; Bloem, B.R. Integrated multidisciplinary care in Parkinson’s disease: A non-randomised, controlled trial (IMPACT). Lancet Neurol. 2013, 12, 947–956. [Google Scholar] [CrossRef]
- Patel, S.; Meek, C.; Clarke, C.E.; Stowe, R.; Shah, L.; Sackley, C.M.; Deane, K.H.O.; Herd, C.; Wheatley, K.; Ives, N. Physiotherapy versus placebo or no intervention in Parkinson’s disease. Cochrane Database Syst. Rev. 2012, 7, CD002817. [Google Scholar]
- Tomlinson, C.L.; Patel, S.; Meek, C.; Herd, C.; Clarke, C.E.; Stowe, R.; Shah, L.; Sackley, C.M.; Deane, K.H.O.; Wheatley, K.; et al. Physiotherapy intervention in Parkinson’s disease: Systematic review and meta-analysis. BMJ 2012, 345, e5004. [Google Scholar] [CrossRef]
- Riva, G.; Castelnuovo, G.; Mantovani, F. Transformation of flow in rehabilitation: The role of advanced communication technologies. Behav. Res. Methods 2006, 38, 237–244. [Google Scholar] [CrossRef]
- Oña, E.D.; Balaguer, C.; Cano-de la Cuerda, R.; Collado-Vázquez, S.; Jardón, A. Effectiveness of Serious Games for Leap Motion on the Functionality of the Upper Limb in Parkinson’s Disease: A Feasibility Study. Comput. Intell. Neurosci. 2018, 2018, 7148427. [Google Scholar] [CrossRef]
- Ramírez-Nieto, M.; Ortiz-Gutiérrez, R.; Cano-De-La-Cuerda, R. Eficacia de los videojuegos comerciales en el tratamiento del equilibrio y la marcha en la enfermedad de Parkinson. Rehabilitación 2018, 52, 114–124. [Google Scholar] [CrossRef]
- O’Cathain, A.; Thomas, K.J.; Drabble, S.; Rudolph, A.; Goode, J.; Hewison, J. Maximising the value of combining qualitative research and randomised controlled trials in health research: The QUAlitative Research in Trials (QUART) study—A mixed methods study. Health Technol. Assess. 2014, 18, 1. [Google Scholar] [CrossRef]
- Curry, L.A.; Krumholz, H.M.; O’Cathain, A.; Clark, V.L.P.; Cherlin, E.; Bradley, E.H. Mixed methods in biomedical and health services research. Circ. Cardiovasc. Qual. Outcomes 2013, 6, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Creswell, J. The Use of “Mixing” Procedure of Mixed Methods in Health Services Research. Med. Care 2013, 51, e51–e57. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, B.L.; Van Puymbroeck, M.; Walter, A.; Sharp, J.; Woshkolup, K.; Urrea-Mendoza, E.; Revilla, F.; Schmid, A.A. Perceived Activities and Participation Outcomes of a Yoga Intervention for Individuals with Parkinson’s Disease: A Mixed Methods Study. Int. J. Yoga Ther. 2018, 28, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Murdoch, K.C.; Larsen, D.; Edey, W.; Arsenault, C.; Howell, A.; Joyce, A.; Sandham, T.; Miyasaki, J.M. The efficacy of the Strength, Hope and Resourcefulness Program for people with Parkinson’s disease (SHARP-PWP): A mixed methods study. Park. Relat. Disord. 2019, 70, 7–12. [Google Scholar] [CrossRef]
- Curry, L.; Nunez-Smith, M. Mixed Methods in Health Sciences Research: A Practical Primer; SAGE Publications: Thousand Oaks, CA, USA, 2015. [Google Scholar]
- Peterson, E.D.; Harrington, R.A. Evaluating Health Technology Through Pragmatic Trials: Novel Approaches to Generate High-Quality Evidence. JAMA 2018, 320, 137–138. [Google Scholar] [CrossRef]
- Tuzzio, L.; Larson, E.B.; Chambers, D.A.; Coronado, G.D.; Curtis, L.H.; Weber, W.J.; Zatzick, D.; Meyers, C.M. Pragmatic clinical trials offer unique opportunities for disseminating, implementing, and sustaining evidence-based practices into clinical care: Proceedings of a workshop. Healthc (Amst) 2019, 7, 51–57. [Google Scholar] [CrossRef]
- Litvan, I.; Kieburtz, K.; Tröster, A.I.; Aarsland, D. Strengths and challenges in conducting clinical trials in Parkinson’s disease mild cognitive impairment. Mov. Disord. 2018, 33, 520–527. [Google Scholar] [CrossRef]
- Fetters, M.D.; Curry, L.A.; Creswell, J.W. Achieving Integration in Mixed Methods Designs-Principles and Practices. Health Serv. Res. 2013, 48, 2134–2156. [Google Scholar] [CrossRef]
- NIH Office of Behavioral and Social Sciences. Best Practices for Mixed Methods Research in the Health Sciences, 2nd ed.; National Institutes of Health: Bethesda, DC, USA, 2018. Available online: https://www.obssr.od.nih.gov/wp-content/uploads/2018/01/Best-Practices-for-Mixed-Methods-Research-in-the-Health-Sciences-2018-01-25.pdf (accessed on 26 February 2020).
- O’Cathain, A.; Murphy, E.; Nicholl, J. The Quality of Mixed Methods Studies in Health Services Research. J. Health Serv. Res. Policy 2008, 13, 92–98. [Google Scholar] [CrossRef]
- Hoffmann, T.; Glasziou, P.; Boutron, I.; Milne, R.; Perera, R.; Moher, D.; Altman, D.; Barbour, V.; Macdonald, H.; Johnston, M.; et al. Better reporting of interventions: Template for intervention description and replication (TIDieR) checklist and guide. BMJ 2014, 348, g1687. [Google Scholar] [CrossRef]
- Alvarez, G.; Cerritelli, F.; Úrrutia, G. Using the template for intervention description and replication (TIDieR) as a tool for improving the design and reporting of manual therapy interventions. Man. Ther. 2016, 24, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Tong, A.; Sainsbury, P.; Craig, J. Consolidated criteria for reporting qualitative research (COREQ): A 32-item checklist for interviews and focus groups. Int. J. Qual. Health Care 2007, 19, 349–357. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, B.C.; Harris, I.B.; Beckman, T.J.; Reed, D.A.; Cook, D.A. Standards for Reporting Qualitative Research. Acad. Med. 2014, 89, 1245–1251. [Google Scholar] [CrossRef] [PubMed]
- Creswell, J.W.; Poth, C.N. Qualitative Inquiry and Research Design. Choosing among Five Approaches, 4th ed.; Sage: Thousand Oaks, CA, USA, 2018. [Google Scholar]
- Korstjens, I.; Moser, A. Series: Practical guidance to qualitative research. Part 4: Trustworthiness and publishing. Eur. J. Gen. Pr. 2017, 24, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.J.; E Daniel, S.; Kilford, L.; Lees, A.J. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: A clinico-pathological study of 100 cases. J. Neurol. Neurosurg. Psychiatry 1992, 55, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression, and mortality. Neurology 1967, 17, 427. [Google Scholar] [CrossRef] [PubMed]
- Schwab, R.S.; England, A.C., Jr. Projection techniques for evaluating surgery in Parkinson’s Disease. In Proceedings of the Third Symposium on Parkinson’s Disease, Royal College of Surgeons, Edinburgh, UK, 20–22 May 1968; E. & S. Livingstone Ltd.: Edinburgh, UK, 1969; pp. 152–157. [Google Scholar]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. Mini-mental state. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Mathiowetz, V.; Weber, K.; Volland, G.; Kashman, N. Reliability and validity of grip and pinch strength evaluations. J. Hand Surg. 1984, 9, 222–226. [Google Scholar] [CrossRef]
- Villafañe, J.H.; Valdes, K.; Buraschi, R.; Martinelli, M.; Bissolotti, L.; Negrini, S. Reliability of the Handgrip Strength Test in Elderly Subjects With Parkinson Disease. HAND 2016, 11, 54–58. [Google Scholar] [CrossRef]
- Desrosiers, J.; Bravo, G.; Hébert, R.; Dutil, Élisabeth; Mercier, L. Validation of the Box and Block Test as a measure of dexterity of elderly people: Reliability, validity, and norms studies. Arch. Phys. Med. Rehabil. 1994, 75, 751–755. [Google Scholar] [CrossRef]
- Mathiowetz, V.; Volland, G.; Kashman, N.; Weber, K. Adult Norms for the Box and Block Test of Manual Dexterity. Am. J. Occup. Ther. 1985, 39, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Agnew, J.; Bolla-Wilson, K.; Kawas, C.H.; Bleecker, M.L. Purdue pegboard age and sex norms for people 40 years old and older. Dev. Neuropsychol. 1988, 4, 29–35. [Google Scholar] [CrossRef]
- Yozbatiran, N.; Der-Yeghiaian, L.; Cramer, S.C. A Standardized Approach to Performing the Action Research Arm Test. Neurorehabilit. Neural Repair 2007, 22, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Song, C.-S. Intrarater Reliability of the Action Research Arm Test for Individuals with Parkinson^|^rsquo;s Disease. J. Phys. Ther. Sci. 2012, 24, 1355–1357. [Google Scholar] [CrossRef][Green Version]
- Roberts, R.E.; Atrkisson, C.C.; Mendias, R.M. Assessing the Client Satisfaction Questionnaire in English and Spanish. Hisp. J. Behav. Sci. 1984, 6, 385–396. [Google Scholar] [CrossRef]
- Larsen, D.L.; Attkisson, C.; Hargreaves, W.A.; Nguyen, T.D. Assessment of client/patient satisfaction: Development of a general scale. Eval. Program Plan. 1979, 2, 197–207. [Google Scholar] [CrossRef]
- Korstjens, I.; Moser, A. Series: Practical guidance to qualitative research. Part 2: Context, research questions and designs. Eur. J. Gen. Pr. 2017, 23, 274–279. [Google Scholar] [CrossRef]
- Moser, A.; Korstjens, I. Series: Practical guidance to qualitative research. Part 3: Sampling, data collection and analysis. Eur. J. Gen. Pr. 2017, 24, 9–18. [Google Scholar] [CrossRef]
- Malterud, K.; Siersma, V.D.; Guassora, A.D. Sample Size in Qualitative Interview Studies. Qual. Health Res. 2016, 26, 1753–1760. [Google Scholar] [CrossRef]
- Mammen, J.R.; Elson, M.J.; Java, J.J.; Beck, C.A.; Beran, D.B.; Biglan, K.; Boyd, C.M.; Schmidt, P.N.; Simone, R.; Willis, A.W.; et al. Patient and Physician Perceptions of Virtual Visits for Parkinson’s Disease: A Qualitative Study. Telemed. e-Health 2018, 24, 255–267. [Google Scholar] [CrossRef]
- De Vries, N.M.; Smilowska, K.; Hummelink, J.; Abramiuc, B.; Van Gilst, M.; Bloem, B.R.; De With, P.; Overeem, S. Exploring the Parkinson patients’ perspective on home-based video recording for movement analysis: A qualitative study. BMC Neurol. 2019, 19, 71. [Google Scholar] [CrossRef] [PubMed]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual Res Psicol 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Liu, B. Sentiment Analysis and Subjectivity. In Handbook of Natural Language Processing, 2th ed.; Chapman and Hall/CRC Press: Boca Raton, FL, USA, 2010; pp. 627–666. [Google Scholar]
- Brooke, J.; Tofiloski, M.; Taboada, M. Cross-Linguistic Sentiment Analysis: From English to Spanish. In Proceedings of the International Conference RANLP-2009, Borovets, Bulgaria, 14–16 September 2009; pp. 50–54. [Google Scholar]
- Vilares, D.; Alonso Pardo, M.; Gómez-Rodríguez, C. Polarity classification of opinionated Spanish texts using dependency parsing. Proces. Leng. Nat. 2013, 50, 13–20. [Google Scholar]
- Li, J.; Liu, M.; Li, X.; Liu, X.; Liu, J.; Sander, U.; Lavin, M.; Bidmon, S. Developing Embedded Taxonomy and Mining Patients’ Interests From Web-Based Physician Reviews: Mixed-Methods Approach. J. Med. Internet Res. 2018, 20, e254. [Google Scholar] [CrossRef] [PubMed]
- Moessner, M.; Feldhege, J.; Wolf, M.; Bauer, S. Analyzing big data in social media: Text and network analyses of an eating disorder forum. Int. J. Eat. Disord. 2018, 51, 656–667. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association Declaration of Helsinki. JAMA 2013, 310, 2191. [CrossRef]
- Johnson, S.L. Impact, Growth, Capacity-building of Mixed Methods Research in the Health Sciences. Am. J. Pharm. Educ. 2019, 83, 7403. [Google Scholar]
- Guetterman, T.C.; Fetters, M.D.; Creswell, J.W. Integrating Quantitative and Qualitative Results in Health Science Mixed Methods Research Through Joint Displays. Ann. Fam. Med. 2015, 13, 554–561. [Google Scholar] [CrossRef]
- Chen, C.-A.; Lee, H.-Y.; Huang, Z.-S.; Lo, C.-C.; Chen, J.-J.J.; Wang, Y.-L.; Lin, K.-C. Movement Training and Assessment with 3D Virtual Reality for Parkinson’s Disease Patient. In Proceedings of the World Congress on Medical Physics and Biomedical Engineering 2006, Seoul, Korea, 27 August–1 September 2006; Springer Science and Business Media LLC.: Seoul, Korea, 2015; Volume 51, pp. 1120–1123. [Google Scholar]
- Butt, A.H.; Rovini, E.; Dolciotti, C.; Bongioanni, P.; De Petris, G.; Cavallo, F. Leap motion evaluation for assessment of upper limb motor skills in Parkinson’s disease. In Proceedings of the 2017 International Conference on Rehabilitation Robotics (ICORR), London, UK, 17–20 July 2017; pp. 116–121. [Google Scholar]
- Cikajlo, I.; Potisk, K.P. Advantages of using 3D virtual reality based training in persons with Parkinson’s disease: A parallel study. J. Neuroeng. Rehabil. 2019, 16, 119. [Google Scholar] [CrossRef]
- Shumway-Cook, A.; Woollacott, M.H. Motor Control: Translating Research into Clinical Practice, 4th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2012; p. 612. [Google Scholar]
- Peters, D.M.; McPherson, A.K.; Fletcher, B.; McClenaghan, B.A.; Fritz, S.L. Counting Repetitions. J. Neurol. Phys. Ther. 2013, 37, 105–111. [Google Scholar] [CrossRef]
- Tsai, L.L.Y.; McNamara, R.; Dennis, S.M.; Moddel, C.; Alison, J.A.; McKenzie, D.K.; McKeough, Z.J. Satisfaction and Experience With a Supervised Home-Based Real-Time Videoconferencing Telerehabilitation Exercise Program in People with Chronic Obstructive Pulmonary Disease (COPD). Int. J. Telerehabilitation 2016, 8, 27–38. [Google Scholar] [CrossRef] [PubMed]
- A Coventry, P.; Bower, P.; Blakemore, A.; Baker, E.; Hann, M.; Li, J.; Paisley, A.; Gibson, M. Satisfaction with a digitally-enabled telephone health coaching intervention for people with non-diabetic hyperglycaemia. NPJ Digit. Med. 2019, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- Kraepelien, M.; Svanborg, C.; Lallerstedt, L.; Sennerstam, V.; Lindefors, N.; Kaldo, V. Individually tailored internet treatment in routine care: A feasibility study. Internet Interv. 2019, 18, 100263. [Google Scholar] [CrossRef] [PubMed]
- Kringle, E.A.; Setiawan, I.M.A.; Golias, K.; Parmanto, B.; Skidmore, E.R. Feasibility of an iterative rehabilitation intervention for stroke delivered remotely using mobile health technology. Disabil. Rehabil. Assist. Technol. 2019, 1–9. [Google Scholar] [CrossRef]
- Mawhinney, G.; Thakar, C.; Williamson, V.; A Rothenfluh, D.; Reynolds, J. Oxford Video Informed Consent Tool (OxVIC): A pilot study of informed video consent in spinal surgery and preoperative patient satisfaction. BMJ Open 2019, 9, e027712. [Google Scholar] [CrossRef]
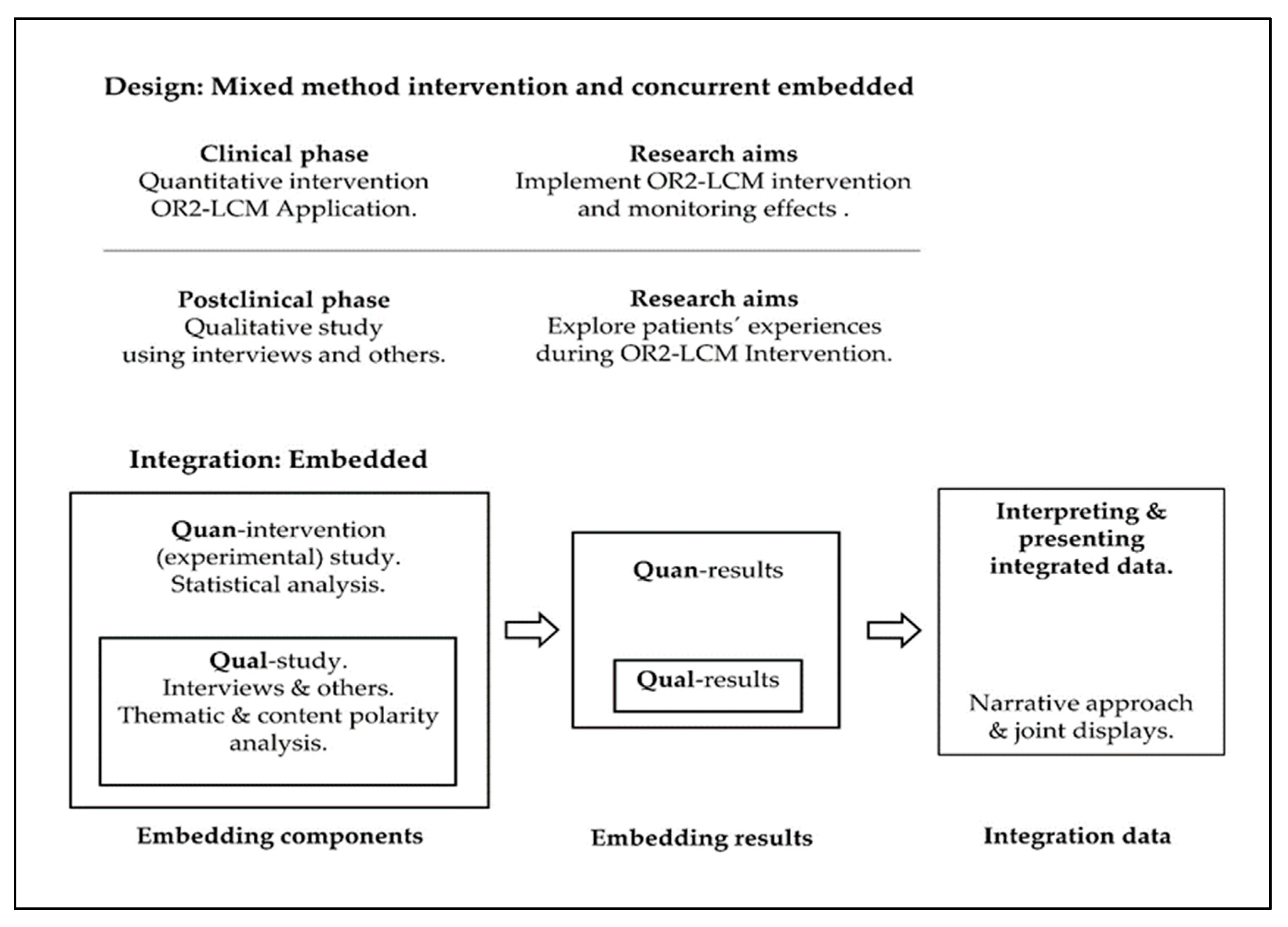
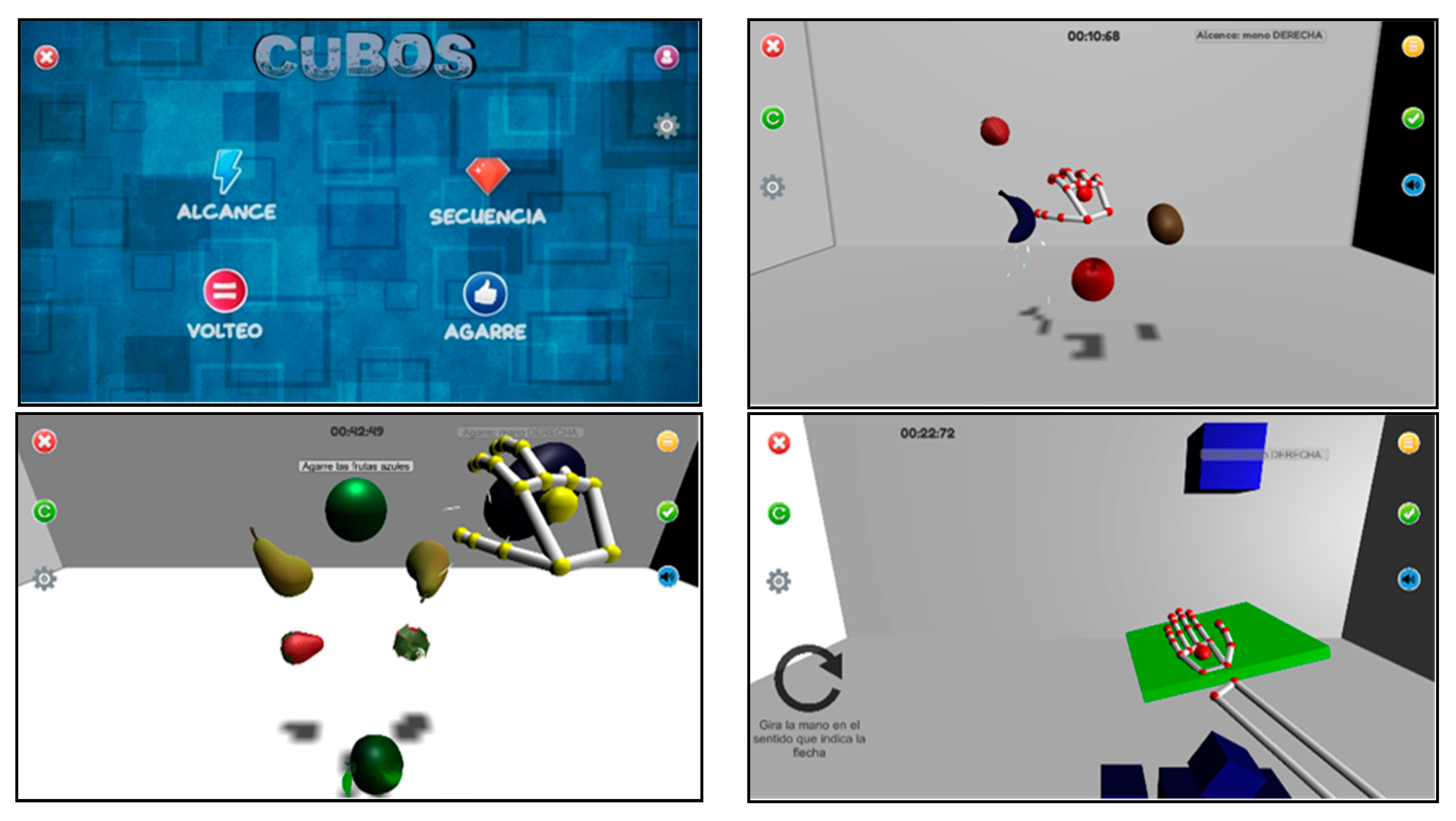
| Component | Sampling | Participants | Intervention | Data Collection | Analysis | |
|---|---|---|---|---|---|---|
| Main study | OR2-LMC novel technology intervention (non-randomized) | Non-probabilistic sampling of non-consecutive cases | Patients with PD (diagnostic criteria of the Brain Bank of the United Kingdom; stages II, III, and IV of the Hoehn and Yahr scale; >60% Schwab and England functionality scale | Oculus Rift 2 with the leap motion controller intervention using virtual reality | A Jamar® hydraulic hand dynamometer (grip strength), the block and box test (unilateral gross manual dexterity), the Purdue pegboard test (coordination, speed of movement, and fine motor dexterity), the action research arm test (upper extremity performance), and the client satisfaction questionnaire (the satisfaction of health service users) | The statistical analysis was performed using the SPSS statistical software system. The Shapiro-Wilk’s test and the Kolmogorov-Smirnov test were used to screen all data for normality of distribution. Additionally, the Wilcoxon test for related samples was used to compare variables. |
| Embedded study | A qualitative case study | Purposeful sampling and information power criteria | The same ncludi ncluding at main study | Non-applicable | Semi-structured interviews based on a question guide and researcher field notes | Thematic inductive analysis and content analysis of free text using The Bing Dictionary and SODictionariesV1.11Spa2 to obtain a description of the polarity (acceptance or rejection). |
| Patients (n) | Age (Years) Mean (± Standard Deviation) | Gender | Hoenhn and Yahr | More Affected Side | Schwab and England Score (%) Mean (±Standard Deviation) |
|---|---|---|---|---|---|
| 6 patients | 74.50 (±4.72) | 5 Male | II (2) | 2 Right | 71.66 (±9.83) |
| 1 Female | III (4) | 4 Left |
| Variable | Median (Interquartile Range) | p-Value | Cohen’s d | |||
|---|---|---|---|---|---|---|
| Jamar | More affected | Pre | 26.85 (10.67) | Pre-Post | 0.028 * | 0.53 |
| Post | 31.16 (9.25) | Pre-Follow-Up | 0.093 | 0.32 | ||
| Follow-up | 28.00 (10.00) | Post-Follow-Up | 0.062 | 0.21 | ||
| Less affected | Pre | 23.13 (13.33) | Pre-Post | 0.028 * | 0.54 | |
| Post | 30.66 (14.33) | Pre-Follow-Up | 0.028 * | 0.15 | ||
| Follow-up | 29.83 (15.00) | Post-Follow-Up | 0.136 | 0.47 | ||
| BBT | More affected | Pre | 42.50 (22.00) | Pre-Post | 0.039 * | 0. 57 |
| Post | 46.00 (16.50) | Pre-Follow-Up | 0.916 | 0.10 | ||
| Follow-up | 44.50 (11.50) | Post-Follow-Up | 0.058 | 0.21 | ||
| Less affected | Pre | 50.00 (9.25) | Pre-Post | 0.343 | 0.18 | |
| Post | 49.00 (14.25) | Pre-Follow-Up | 0.684 | 0.16 | ||
| Follow-up | 48.00 (15.25) | Post-Follow-Up | 0.715 | 0.25 | ||
| PPT | More affected | Pre | 7.83 (4.92) | Pre-Post | 0.027 * | 0.57 |
| Post | 8.66 (4.50) | Pre-Follow-Up | 0.073 | 0.20 | ||
| Follow-up | 8.16 (4.75) | Post-Follow-Up | 0.109 | 0.26 | ||
| Less affected | Pre | 8.66 (2.50) | Pre-Post | 0.028 * | 0.54 | |
| Post | 9.83 (2.75) | Pre-Follow-Up | 0.400 | 0.37 | ||
| Follow-up | 8.16 (5.66) | Post-Follow-Up | 0.686 | 0.11 | ||
| PPT both hands | Pre | 11.00 (5.17) | Pre-Post | 0.168 | 0.15 | |
| Post | 11.33 (6.00) | Pre-Follow-Up | 0.026 * | 0.11 | ||
| Follow-up | 11.99 (5.17) | Post-Follow-Up | 0.715 | 0.19 | ||
| PPT assemblies | Pre | 12.16 (8.92) | Pre-Post | 0.028 * | 0.57 | |
| Post | 13.83 (10.33) | Pre-Follow-Up | 0.600 | 0.26 | ||
| Follow-up | 12.49 (5.33) | Post-Follow-Up | 0.416 | 0.14 | ||
| ARAT total score | More affected | Pre | 52.50 (8.25) | Pre-Post | 0.180 | 0.15 |
| Post | 53.50 (4.50) | Pre-Follow-Up | 0.276 | 0 | ||
| Follow-up | 53.50 (5.25) | Post-Follow-Up | 0.679 | 0.14 | ||
| Less affected | Pre | 52.50 (6.25) | Pre-Post | 0.596 | 0.10 | |
| Post | 53.00 (2.50) | Pre-Follow-Up | 0.891 | 0.21 | ||
| Follow-up | 54.00 (6.25) | Post-Follow-Up | 0.914 | 0.24 | ||
| Variable | Patients Punctuation |
|---|---|
| 1. Quality of service | 3.66 (0.51) |
| 2. Type of service | 3.33 (0.51) |
| 3. Needs met | 3.66 (0.51) |
| 4. Recommend to a friend | 3.66 (0.51) |
| 5. Amount of help | 4 (0) |
| 6. Deal with problems | 3.33 (0.51) |
| 7. Overall satisfaction | 4 (0) |
| 8. Come back | 3.66 (0.51) |
| Total Score | 3.66 (0.18) |
| Outcomes | Quantitative Findings | Qualitative Findings | Observations |
|---|---|---|---|
| Jamar: measures grip strength | Significant improvements on the Jamar post-treatment and at follow-up | There was no narrative regarding strength improvement | Patients initially reported no perceived short-term benefits from the intervention, although later on at home, patients did report improvements in their activities of daily living. Patients believed the intervention was aimed at achieving mental and cognitive (concentration, reaction rate, memory, etc.) improvements. Patients reported their experiences using the OR2-LMC system, describing it as a process that began with nervousness, fear, and surprise with the virtual environment. Through adaptation to the virtual world and confronting these new challenges, they eventually gained control of the tests and treatment. This process resulted in satisfaction (through overcoming the challenges and limitations), frustration (needing to train more), or boredom (monotony of the games). |
| BBT: measures unilateral gross manual dexterity | Significant and positive results for the affected side post-treatment | Patients reported improvement in activities such as handling dishes and cutlery | |
| PPT: measures coordination, speed of movement, and fine motor dexterity | Significant improvements for both hands post-treatment, both hands at follow-up, and assembly capacity post-treatment | Patients reported improved fine movements in activities such as accepting a purchase ticket. Coordination and the ability to overcome obstacles were also improved, although no improvements in the speed of movements were reported | |
| ARAT: measures upper limb performance | No significant results | Patients reported improved movements when handling dishes, objects, and reaction speeds | |
| CSQ-8: measures patient satisfaction | High degree of satisfaction; patients obtained a mean of 3.66 (0.18) points out of the maximum of 4 | Polarity results showed a general acceptance of OR2-LMC therapy, although some patients pointed out the necessity of more clearly explaining the treatment and realistic expectations for its use; some patients were unaware of the objectives of the study or expected greater effects from therapy |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Herrera-Baeza, P.; Cano-de-la-Cuerda, R.; Oña-Simbaña, E.D.; Palacios-Ceña, D.; Pérez-Corrales, J.; Cuenca-Zaldivar, J.N.; Gueita-Rodriguez, J.; Balaguer-Bernaldo de Quirós, C.; Jardón-Huete, A.; Cuesta-Gomez, A. The Impact of a Novel Immersive Virtual Reality Technology Associated with Serious Games in Parkinson’s Disease Patients on Upper Limb Rehabilitation: A Mixed Methods Intervention Study. Sensors 2020, 20, 2168. https://doi.org/10.3390/s20082168
Sánchez-Herrera-Baeza P, Cano-de-la-Cuerda R, Oña-Simbaña ED, Palacios-Ceña D, Pérez-Corrales J, Cuenca-Zaldivar JN, Gueita-Rodriguez J, Balaguer-Bernaldo de Quirós C, Jardón-Huete A, Cuesta-Gomez A. The Impact of a Novel Immersive Virtual Reality Technology Associated with Serious Games in Parkinson’s Disease Patients on Upper Limb Rehabilitation: A Mixed Methods Intervention Study. Sensors. 2020; 20(8):2168. https://doi.org/10.3390/s20082168
Chicago/Turabian StyleSánchez-Herrera-Baeza, Patricia, Roberto Cano-de-la-Cuerda, Edwin Daniel Oña-Simbaña, Domingo Palacios-Ceña, Jorge Pérez-Corrales, Juan Nicolas Cuenca-Zaldivar, Javier Gueita-Rodriguez, Carlos Balaguer-Bernaldo de Quirós, Alberto Jardón-Huete, and Alicia Cuesta-Gomez. 2020. "The Impact of a Novel Immersive Virtual Reality Technology Associated with Serious Games in Parkinson’s Disease Patients on Upper Limb Rehabilitation: A Mixed Methods Intervention Study" Sensors 20, no. 8: 2168. https://doi.org/10.3390/s20082168
APA StyleSánchez-Herrera-Baeza, P., Cano-de-la-Cuerda, R., Oña-Simbaña, E. D., Palacios-Ceña, D., Pérez-Corrales, J., Cuenca-Zaldivar, J. N., Gueita-Rodriguez, J., Balaguer-Bernaldo de Quirós, C., Jardón-Huete, A., & Cuesta-Gomez, A. (2020). The Impact of a Novel Immersive Virtual Reality Technology Associated with Serious Games in Parkinson’s Disease Patients on Upper Limb Rehabilitation: A Mixed Methods Intervention Study. Sensors, 20(8), 2168. https://doi.org/10.3390/s20082168










