The Rehapiano—Detecting, Measuring, and Analyzing Action Tremor Using Strain Gauges
Abstract
1. Introduction
- an introduction of the Rehapiano device for fast detection and quantification of action tremor using strain gauges,
- validation of the system by the comparison of measurements made by the Rehapiano to those made using optical encoders,
- an experimental analysis of the Rehapiano on healthy subjects and patients with PD, and
- an adaptation of an algorithm [9] that was previously developed for use with accelerometers and gyroscopes to asses tremor severity.
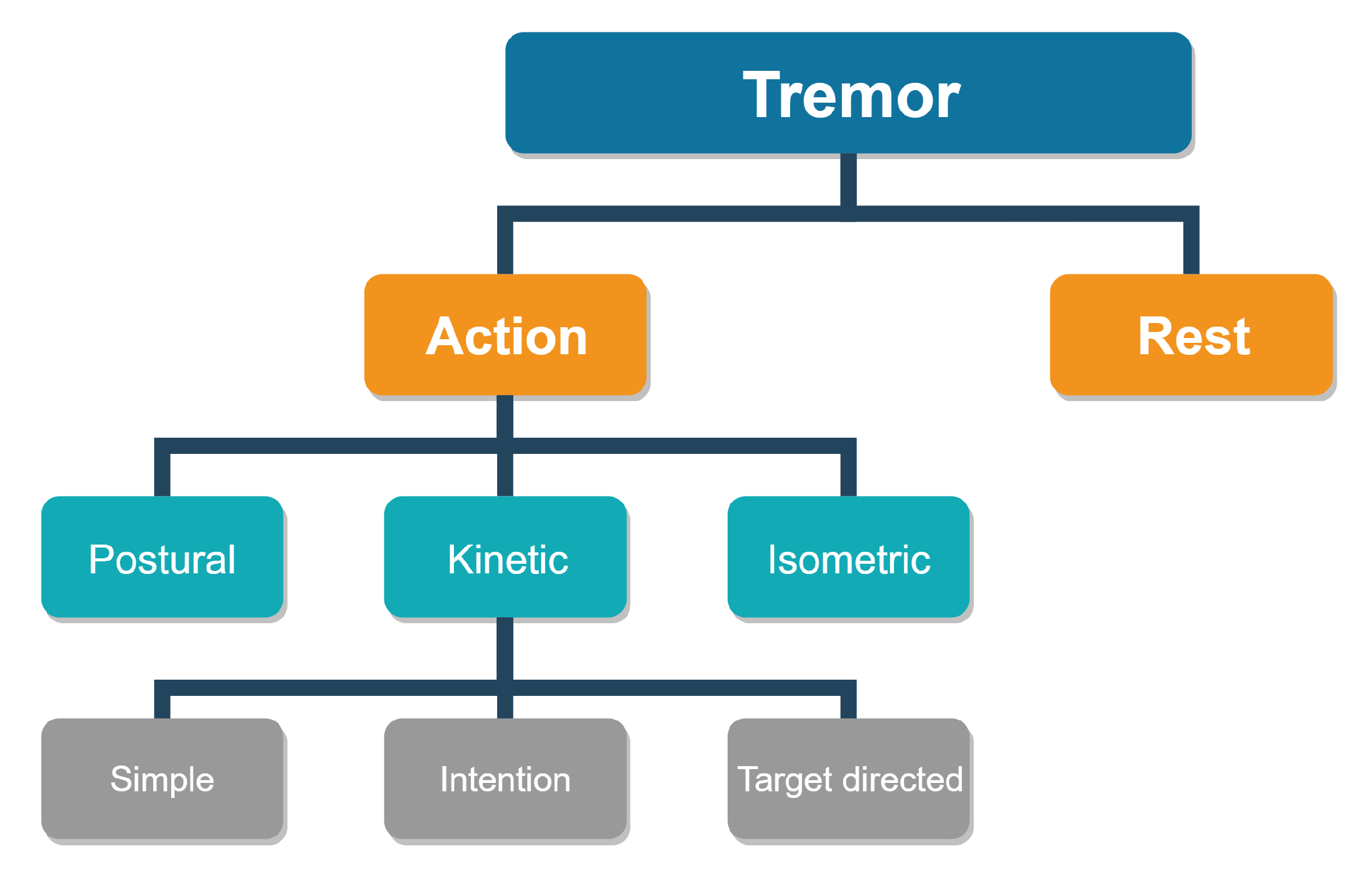
2. Problem Statement
3. Background and Related Works
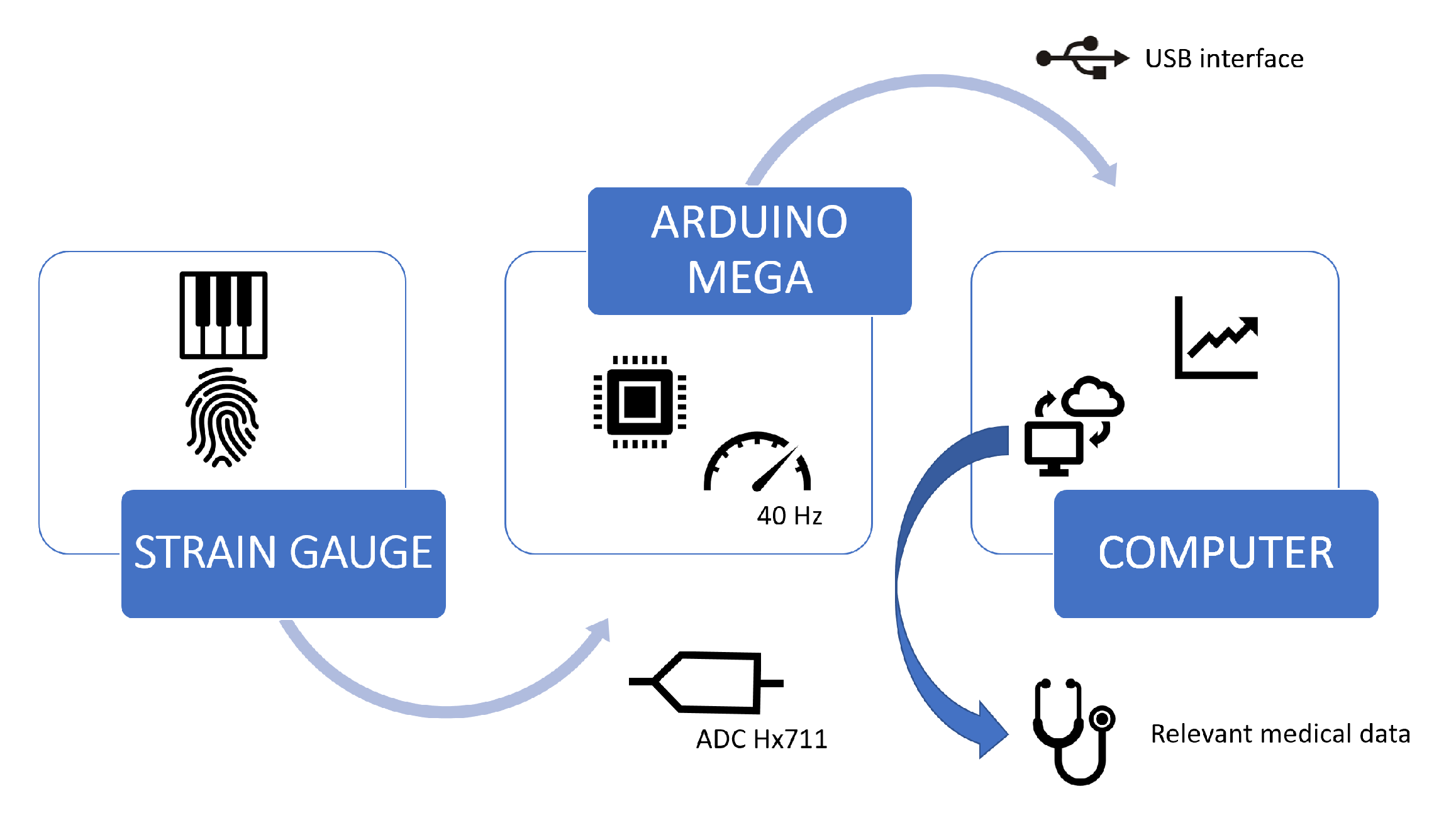
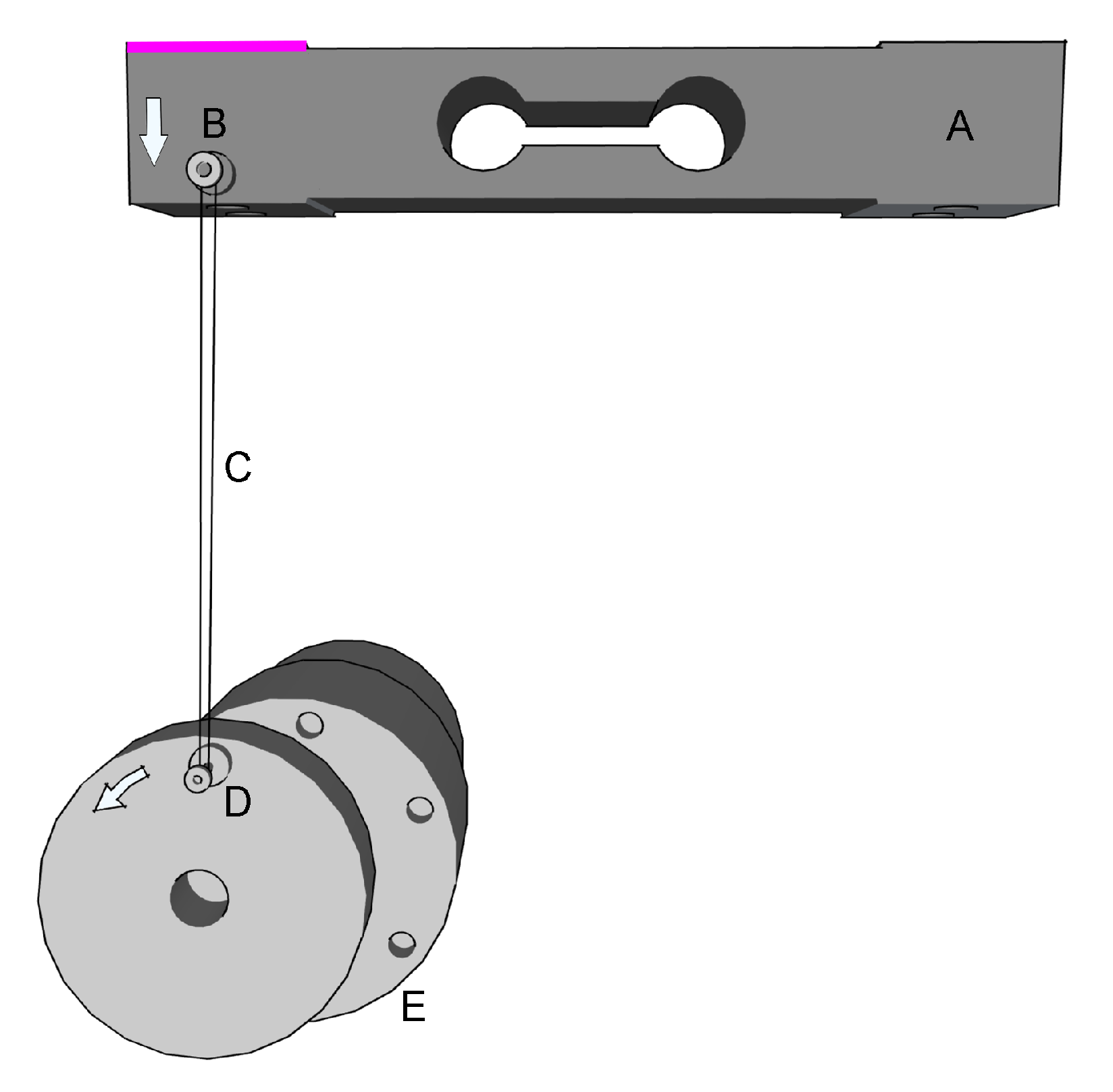
4. The Rehapiano
5. Methods
- The Rehapiano is able to detect force changes with frequencies between 1 and 20 Hz.
- Rehapiano measurements can be used to detect tremors of Parkinson patients.
- The Rehapiano is sensitive enough to enable quantification of PT.
5.1. Verification of Hypothesis 1
5.2. Verification of Hypothesis 2
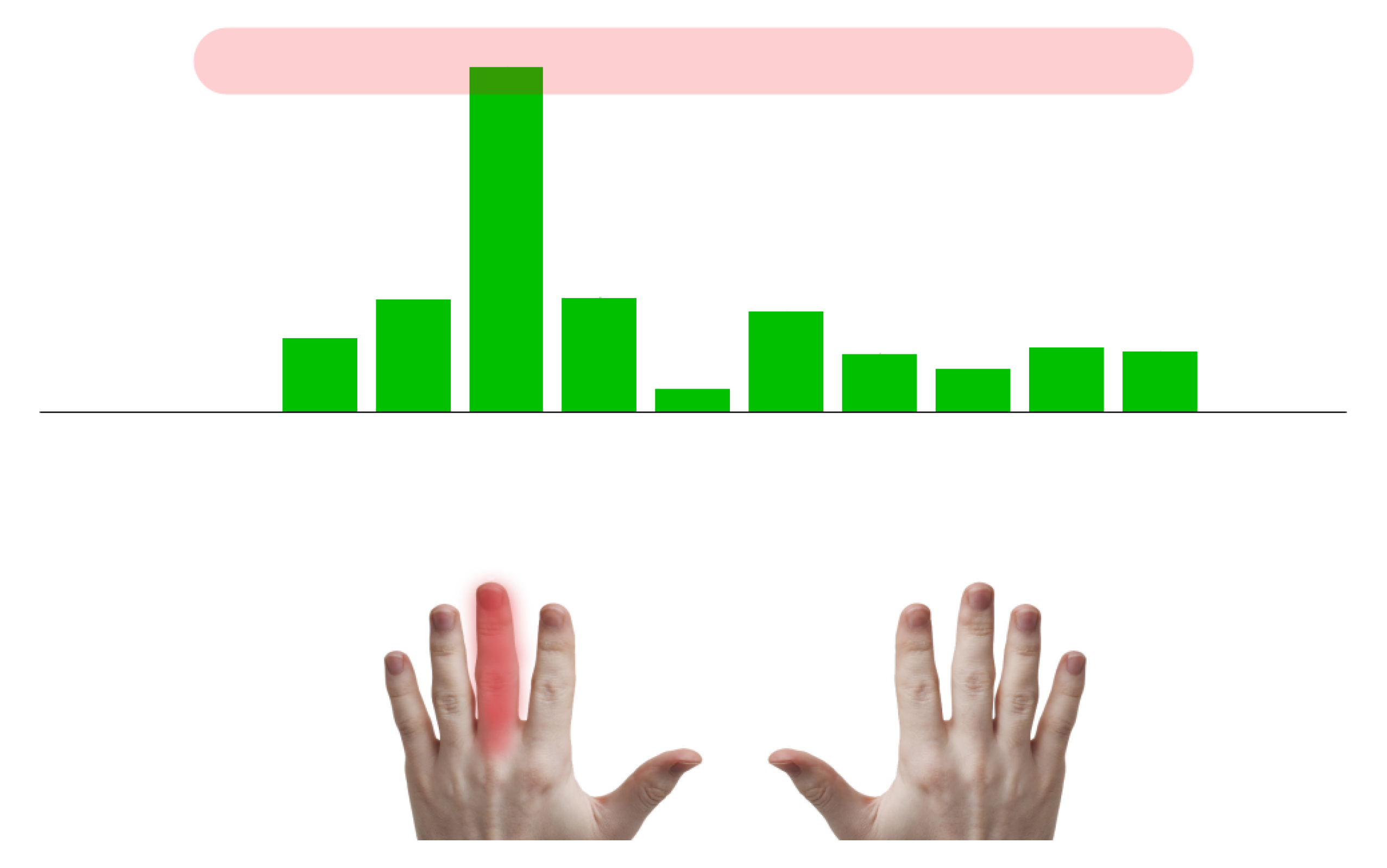
- The target value—set to 300 g—is the force produced by pressing on the strain gauge. All PD subjects could exert a finger force of 450 g on average. The experimental target value was set to of 450 g—300 g for all fingers.
- The hold time—set to 3 s—is the period during which a patient should keep the force around the target value. This value was selected based on two aspects: we assumed that a longer exercise than 3 s for all fingers would lead to fatigue and that a shorter exercise would not contain enough data to evaluate the tremor.
- The sequence of fingers represents the sequence of fingers without repetition from the left little finger to the right little finger. In these experiments, we chose the most simple sequence to iterate through the fingers from left to right to make the exercise as simple as possible.
5.3. Verification of Hypothesis 3
6. Experimental Results
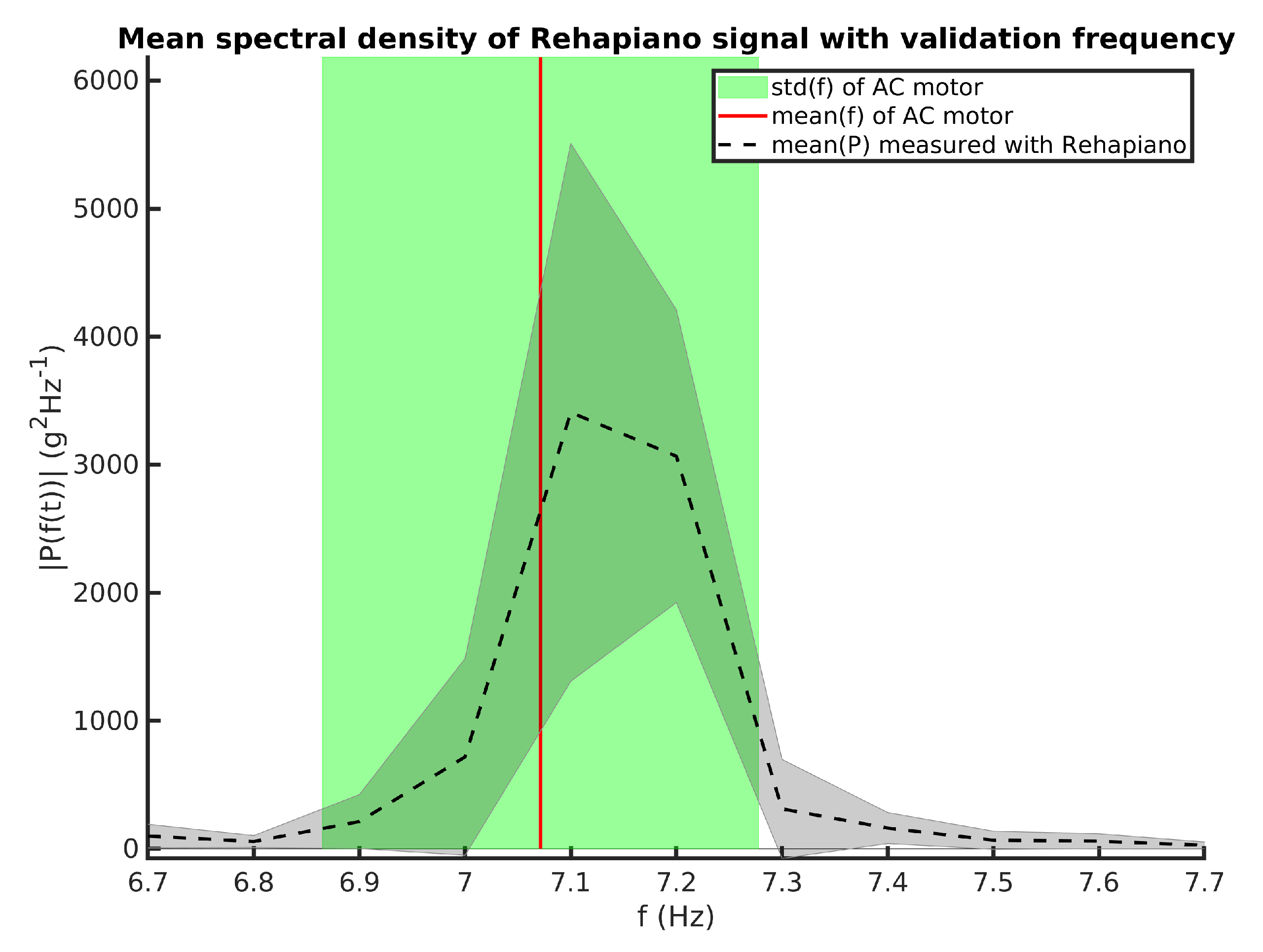
6.1. Validation of the Rehapiano
- : Mean frequency measured by the optical encoder and by the Rehapiano is equal.
- : Mean frequency measured by optical encoder and by the Rehapiano is not equal.
6.2. Distinction between Healthy Population and Patients with PT
- Measurements from the Rehapiano contain detectable tremor information. The performance metrics of a classifier that detects tremor should meet the following requirements.
- −
- Cross validation accuracy > 90%
- −
- Precision > 95%
- −
- Recall > 95%
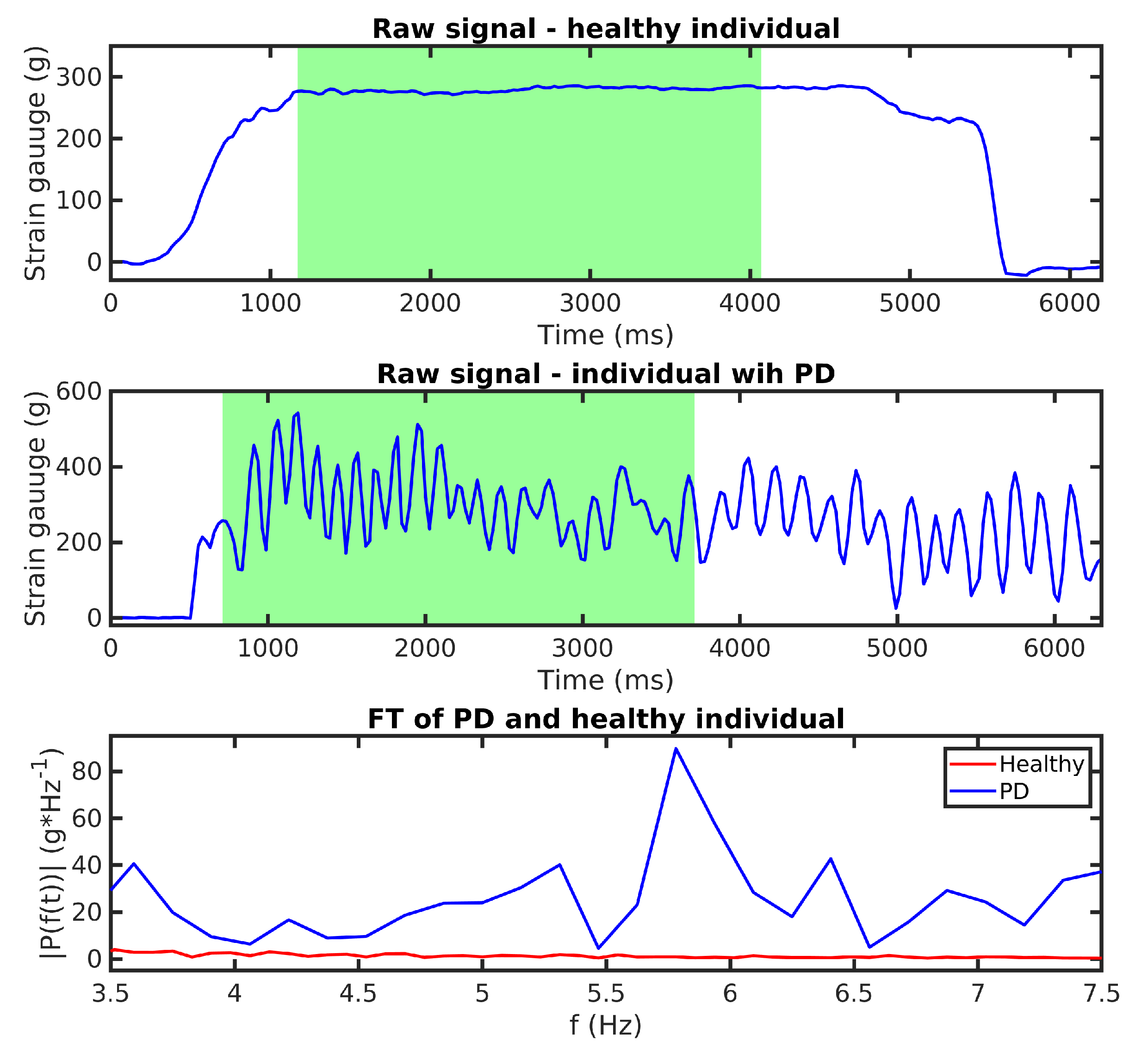
- First, our algorithm filters the raw signal with an outlier filter that replaces values below the 1.25th percentile of the distribution using linear interpolation.
- It then applies a band-pass filter that keeps frequencies between Hz and Hz.
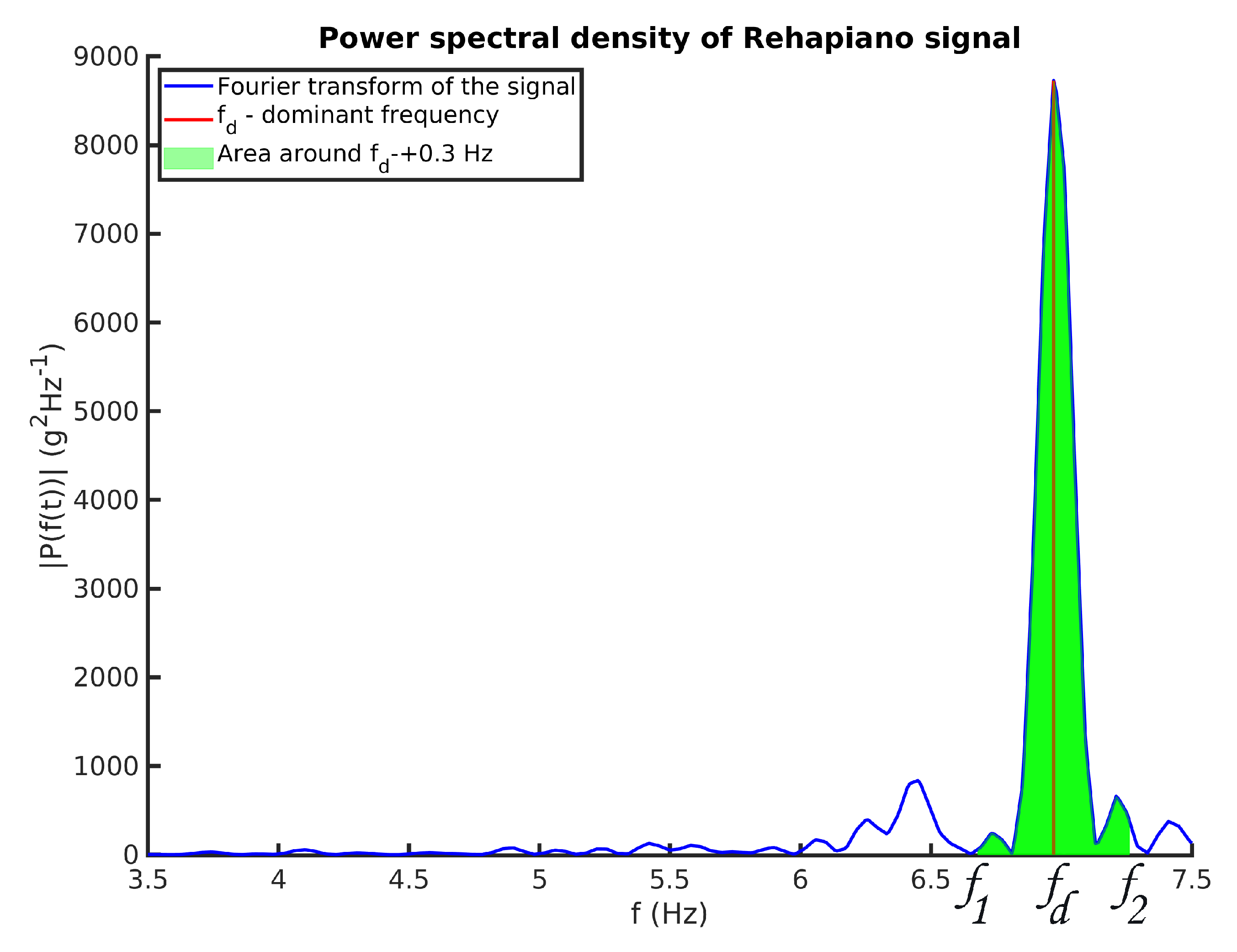
- Next, the algorithm calculates a one-sided amplitude spectrum of a 3 s signal, where the patient reached the desired force (Figure 8). After that, it resamples the result of the FT at Hz between and Hz, creating a vector with 41 values describing the FT amplitude of the signal at specific frequencies.
- Finally, we expect that FT amplitudes of the PD patients will be significantly different from the healthy population, and the data are labeled based on this assumption (PD patient: 1; Healthy Subject: 0).
6.3. Quantitative Assessment of the Tremor
- Measurements from the Rehapiano provide quantitative information about tremor. Subsequent measurements of the same subject output the same tremor frequency.
- −
- Standard deviation of the measurements is less than Hz.
7. Discussion
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dorsey, E.R.; Elbaz, A.; Nichols, E.; Abd-Allah, F.; Abdelalim, A.; Adsuar, J.C.; Ansha, M.G.; Brayne, C.; Choi, J.Y.J.; Collado-Mateo, D. Global, regional, and national burden of Parkinson’s disease, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 939–953. [Google Scholar] [CrossRef]
- Moisan, F.; Kab, S.; Mohamed, F.; Canonico, M.; Le Guern, M.; Quintin, C.; Carcaillon, L.; Nicolau, J.; Duport, N.; Singh-Manoux, A.; et al. Parkinson disease male-to-female ratios increase with age: French nationwide study and meta-analysis. J. Neurol. Neurosurg. Psychiatry 2016, 87, 952–957. [Google Scholar] [CrossRef] [PubMed]
- Politis, M.; Wu, K.; Molloy, S.; Bain, P.G.; Chaudhuri, K.R.; Piccini, P. Parkinson’s disease symptoms: The patient’s perspective. Mov. Dis. 2010, 25, 1646–1651. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, K.R.; Healy, D.G.; Schapira, A.H. Non-motor symptoms of Parkinson’s disease: Diagnosis and management. Lancet Neurol. 2006, 5, 235–245. [Google Scholar] [CrossRef]
- Abdo, W.F.; Van De Warrenburg, B.P.; Burn, D.J.; Quinn, N.P.; Bloem, B.R. The clinical approach to movement disorders. Nat. Rev. Neurol. 2010, 6, 29. [Google Scholar] [CrossRef]
- Bendat, J.S.; Piersol, A.G. Random Data: Analysis and Measurement Procedures; John Wiley & Sons: Hoboken, NJ, USA, 2011; Volume 729. [Google Scholar]
- Haubenberger, D.; Abbruzzese, G.; Bain, P.G.; Bajaj, N.; Benito-León, J.; Bhatia, K.P.; Deuschl, G.; Forjaz, M.J.; Hallett, M.; Louis, E.D. Transducer-based evaluation of tremor. Mov. Dis. 2016, 31, 1327–1336. [Google Scholar] [CrossRef]
- Rovini, E.; Maremmani, C.; Cavallo, F. How wearable sensors can support Parkinson’s disease diagnosis and treatment: A systematic review. Front. Neurosci. 2017, 11, 555. [Google Scholar] [CrossRef]
- Dai, H.; Zhang, P.; Lueth, T. Quantitative assessment of parkinsonian tremor based on an inertial measurement unit. Sensors 2015, 15, 25055–25071. [Google Scholar] [CrossRef]
- Samuel, M.; Torun, N.; Tuite, P.J.; Sharpe, J.A.; Lang, A.E. Progressive ataxia and palatal tremor (PAPT) Clinical and MRI assessment with review of palatal tremors. Brain 2004, 127, 1252–1268. [Google Scholar] [CrossRef]
- Ahlskog, J.E. Slowing Parkinson’s disease progression: Recent dopamine agonist trials. Neurology 2003, 60, 381–389. [Google Scholar] [CrossRef]
- Sanger, T.D.; Chen, D.; Fehlings, D.L.; Hallett, M.; Lang, A.E.; Mink, J.W.; Singer, H.S.; Alter, K.; Ben-Pazi, H.; Butler, E.E. Definition and classification of hyperkinetic movements in childhood. Mov. Dis. 2010, 25, 1538–1549. [Google Scholar] [CrossRef]
- Brittain, J.S.; Probert-Smith, P.; Aziz, T.Z.; Brown, P. Tremor suppression by rhythmic transcranial current stimulation. Curr. Biol. 2013, 23, 436–440. [Google Scholar] [CrossRef]
- Choi, S.M. Movement disorders following cerebrovascular lesions in cerebellar circuits. J. Mov. Dis. 2016, 9, 80. [Google Scholar] [CrossRef] [PubMed]
- Bötzel, K.; Tronnier, V.; Gasser, T. The differential diagnosis and treatment of tremor. Deutsch. Ärztebl. Int. 2014, 111, 225. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Deuschl, G.; Bain, P.; Brin, M.; Committee, A.H.S. Consensus statement of the movement disorder society on tremor. Mov. Dis. 1998, 13, 2–23. [Google Scholar] [CrossRef] [PubMed]
- Elble, R.J. Tremor. In Neuro-Geriatrics; Springer: Cham, Switzerland, 2017; pp. 311–326. [Google Scholar]
- Zhang, J.; Xing, Y.; Ma, X.; Feng, L. Differential diagnosis of Parkinson disease, essential tremor, and enhanced physiological tremor with the tremor analysis of EMG. Parkinson’s Dis. 2017, 2017, 1597907. [Google Scholar] [CrossRef]
- Chen, W.; Hopfner, F.; Becktepe, J.S.; Deuschl, G. Rest tremor revisited: Parkinson’s disease and other disorders. Transl. Neurodegener. 2017, 6, 16. [Google Scholar] [CrossRef]
- Koller, W.C.; Vetere-Overfield, B.; Barter, R. Tremors in early Parkinson’s disease. Clin. Neuropharmacol. 1989, 12, 293–297. [Google Scholar] [CrossRef]
- Lance, J.W.; Schwab, R.S.; Peterson, E.A. Action tremor and the cogwheel phenomenon in Parkinson’s disease. Brain 1963, 86, 95–110. [Google Scholar] [CrossRef]
- Deuschl, G.; Krack, P.; Lauk, M.; Timmer, J. Clinical neurophysiology of tremor. J. Clin. Neurophysiol. 1996, 13, 110–121. [Google Scholar] [CrossRef]
- Lee, H.J.; Lee, W.W.; Kim, S.K.; Park, H.; Jeon, H.S.; Kim, H.B.; Jeon, B.S.; Park, K.S. Tremor frequency characteristics in Parkinson’s disease under resting-state and stress-state conditions. J. Neurol. Sci. 2016, 362, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Perumal, S.V.; Sankar, R. Gait and tremor assessment for patients with Parkinson’s disease using wearable sensors. ICT Express 2016, 2, 168–174. [Google Scholar] [CrossRef]
- Bhidayasiri, R. Differential diagnosis of common tremor syndromes. Postgrad. Med. J. 2005, 81, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Zach, H.; Dirkx, M.; Bloem, B.R.; Helmich, R.C. The clinical evaluation of Parkinson’s tremor. J. Parkinson’s Dis. 2015, 5, 471–474. [Google Scholar] [CrossRef]
- Benito-León, J.; Domingo-Santos, Á. Orthostatic tremor: An update on a rare entity. Tremor Other Hyperkinet. Mov. 2016, 6, 411. [Google Scholar]
- Sharott, A.; Marsden, J.; Brown, P. Primary orthostatic tremor is an exaggeration of a physiological response to instability. Mov. Dis. Off. J. Mov. Dis. Soc. 2003, 18, 195–199. [Google Scholar] [CrossRef]
- Thenganatt, M.A.; Jankovic, J. Psychogenic tremor: A video guide to its distinguishing features. Tremor Other Hyperkinet. Mov. 2014, 4, 253. [Google Scholar]
- Elble, R.J.; McNames, J. Using portable transducers to measure tremor severity. Tremor Other Hyperkinet. Mov. 2016, 6, 375. [Google Scholar]
- Hurtado, J.M.; Gray, C.M.; Tamas, L.B.; Sigvardt, K.A. Dynamics of tremor-related oscillations in the human globus pallidus: A single case study. Proc. Natl. Acad. Sci. USA 1999, 96, 1674–1679. [Google Scholar] [CrossRef]
- Elble, R.J.; Pullman, S.L.; Matsumoto, J.Y.; Raethjen, J.; Deuschl, G.; Tintner, R. Tremor amplitude is logarithmically related to 4-and 5-point tremor rating scales. Brain 2006, 129, 2660–2666. [Google Scholar] [CrossRef]
- Niazmand, K.; Tonn, K.; Kalaras, A.; Fietzek, U.M.; Mehrkens, J.H.; Lueth, T.C. Quantitative evaluation of Parkinson’s disease using sensor based smart glove. In Proceedings of the 2011 24th International Symposium on Computer-Based Medical Systems (CBMS), Bristol, UK, 27–30 June 2011; pp. 1–8. [Google Scholar]
- Khan, F.M.; Barnathan, M.; Montgomery, M.; Myers, S.; Côté, L.; Loftus, S. A wearable accelerometer system for unobtrusive monitoring of parkinson’s diease motor symptoms. In Proceedings of the 2014 IEEE International Conference on Bioinformatics and Bioengineering, Boca Raton, FL, USA, 10–12 November 2014; pp. 120–125. [Google Scholar]
- Bhavana, C.; Gopal, J.; Raghavendra, P.; Vanitha, K.; Talasila, V. Techniques of measurement for Parkinson’s tremor highlighting advantages of embedded IMU over EMG. In Proceedings of the 2016 International Conference on Recent Trends in Information Technology (ICRTIT), Chennai, India, 8–9 April 2016; pp. 1–5. [Google Scholar]
- AVIA Semiconductors. 24-Bit Analog-to-Digital Converter (ADC) for Weigh Scales. 2006. Available online: https://www.mouser.com/datasheet/2/813/hx711_english-1022875.pdf (accessed on 23 January 2020).
- Heida, T.; Wentink, E.C.; Marani, E. Power spectral density analysis of physiological, rest and action tremor in Parkinson’s disease patients treated with deep brain stimulation. J. NeuroEng. Rehabil. 2013, 10, 70. [Google Scholar] [CrossRef]
- Kumar, H.; Jog, M. A patient with tremor, part 1: Making the diagnosis. CMAJ 2011, 183, 1507–1510. [Google Scholar] [CrossRef][Green Version]
- Stacy, M.A.; Elble, R.J.; Ondo, W.G.; Wu, S.C.; Hulihan, J.; Group, T.S. Assessment of interrater and intrarater reliability of the Fahn–Tolosa–Marin Tremor Rating Scale in essential tremor. Mov. Dis. 2007, 22, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Western, D.G.; Neild, S.A.; Jones, R.; Davies-Smith, A. Personalised profiling to identify clinically relevant changes in tremor due to multiple sclerosis. BMC Med. Inform. Decis. Mak. 2019, 19, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Hssayeni, M.D.; Jimenez-Shahed, J.; Burack, M.A.; Ghoraani, B. Wearable Sensors for Estimation of Parkinsonian Tremor Severity during Free Body Movements. Sensors 2019, 19, 4215. [Google Scholar] [CrossRef] [PubMed]
- Vivar-Estudillo, G.; Ibarra-Manzano, M.A.; Almanza-Ojeda, D.L. Tremor Signal Analysis for Parkinson’s Disease Detection Using Leap Motion Device. In Mexican International Conference on Artificial Intelligence; Springer: Cham, Switzerland, 2018; pp. 342–353. [Google Scholar]
- Kostikis, N.; Hristu-Varsakelis, D.; Arnaoutoglou, M.; Kotsavasiloglou, C. A smartphone-based tool for assessing parkinsonian hand tremor. IEEE J. Biomed. Health Inform. 2015, 19, 1835–1842. [Google Scholar] [CrossRef]
- Manzanera, O.M.; Elting, J.W.; van der Hoeven, J.H.; Maurits, N.M. Tremor detection using parametric and non-parametric spectral estimation methods: A comparison with clinical assessment. PLoS ONE 2016, 11, e0156822. [Google Scholar] [CrossRef]
- Raethjen, J.; Austermann, K.; Witt, K.; Zeuner, K.E.; Papengut, F.; Deuschl, G. Provocation of Parkinsonian tremor. Mov. Dis. Off. J. Mov. Dis. Soc. 2008, 23, 1019–1023. [Google Scholar] [CrossRef]
- Jankovic, J. Distinguishing essential tremor from Parkinson’s disease. Pract. Neurol. 2012, 36–38. [Google Scholar]

| PD Patient | Tremor | Handwriting | Sessions |
|---|---|---|---|
| Patient 1. | 1 | 2 | 2 |
| Patient 2. | 2 | 3 | 1 |
| Patient 3. | 3 | 3 | 1 |
| Patient 4. | 3 | 4 | 1 |
| Patient 5. | 3 | 2 | 7 |
| Patient 6. | 3 | 2 | 1 |
| Patient 7. | 4 | 3 | 1 |
| Freq | ||||||
|---|---|---|---|---|---|---|
| Low | 1.56 | 0.17 | 1.42 | 0.053 | −2.51 | 1 |
| Medium | 2.96 | 0.09 | 2.94 | 0.021 | −0.97 | 0 |
| High | 7.07 | 0.2 | 7.16 | 0.028 | 1.28 | 0 |
| Class. | ValAccuracy | Sensitivity | Specificity | Precision | |
|---|---|---|---|---|---|
| SVM | 0.9311 | 0.798 | 0.9965 | 0.9965 | 0.8863 |
| NB | 0.9464 | 0.875 | 0.9722 | 0.9557 | 0.9136 |
| DT | 0.9638 | 0.975 | 0.9861 | 0.9872 | 0.9811 |
| KNN | 0.9285 | 0.9326 | 0.9756 | 0.9872 | 0.9537 |
| Name | Device | Scale | Method | Type | Accuracy | Sensitivity | Specificity |
|---|---|---|---|---|---|---|---|
| Our | Rehapiano | FTMTRS | DT | Binary | 0.9638 | 0.975 | 0.9861 |
| [41] | Gyroscope + Acc | UPDRS | LSTM/GTB | Multi | 0.84/0.96 * | - | - |
| [42] | Leap Motion | UPDRS | BCT | Binary | 0.99 | 0.99 | 0.99 |
| [43] | Smartphone | UPDRS | RF | Binary | - | 0.90 | 0.82 |
| [44] | Accelerometers | Binary | Welch (2) | Binary | 0.95 | 0.98 | 0.69 |
| Left Hand Measurements | Right Hand Measurements | |||||
|---|---|---|---|---|---|---|
| Valid rel | Valid abs | Valid rel | Valid abs | |||
| 0.5 | 0.8571 | 24 | 0.3928 | 11 | ||
| 0.6 | 0.6071 | 17 | 0.2857 | 8 | ||
| 0.7 | 0.4285 | 12 | 0.0714 | 2 | ||
| 0.8 | 0.3214 | 9 | 0 | 0 | - | |
| 0.9 | 0 | 0 | - | 0 | 0 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferenčík, N.; Jaščur, M.; Bundzel, M.; Cavallo, F. The Rehapiano—Detecting, Measuring, and Analyzing Action Tremor Using Strain Gauges. Sensors 2020, 20, 663. https://doi.org/10.3390/s20030663
Ferenčík N, Jaščur M, Bundzel M, Cavallo F. The Rehapiano—Detecting, Measuring, and Analyzing Action Tremor Using Strain Gauges. Sensors. 2020; 20(3):663. https://doi.org/10.3390/s20030663
Chicago/Turabian StyleFerenčík, Norbert, Miroslav Jaščur, Marek Bundzel, and Filippo Cavallo. 2020. "The Rehapiano—Detecting, Measuring, and Analyzing Action Tremor Using Strain Gauges" Sensors 20, no. 3: 663. https://doi.org/10.3390/s20030663
APA StyleFerenčík, N., Jaščur, M., Bundzel, M., & Cavallo, F. (2020). The Rehapiano—Detecting, Measuring, and Analyzing Action Tremor Using Strain Gauges. Sensors, 20(3), 663. https://doi.org/10.3390/s20030663







