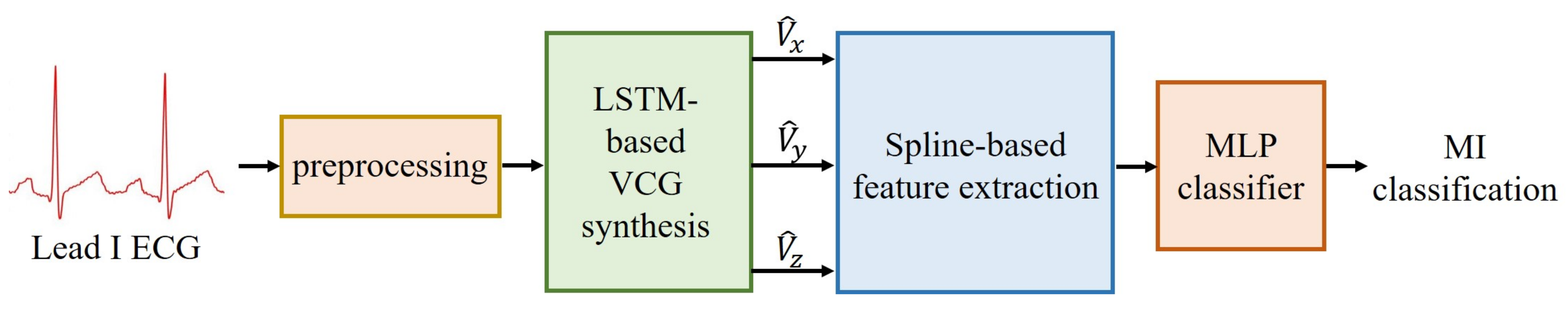
1. Introduction
Myocardial infarction (MI) has long been recognized as the main cause of death worldwide. According to the data from the World Health Organization (WHO) [
1], cardiovascular diseases, including MI, were estimated to account for 31% of deaths worldwide in 2017. In the United States, about 110,000 Americans died of MI in 2015 and the estimated annual incidence of MI is 605,000 new attacks [
2]. MI results from an occlusion of the coronary artery and insufficient blood supply to the myocardium. It can be further classified into various subtypes depending on the localization of infarcted area. In clinical setting, MI is diagnosed using 12-lead electrocardiography (ECG) [
3] as well as 3-lead vectorcardiography (VCG) [
4]. ECG signals are recorded from different locations of the body to capture the three-dimensional view of the human heart. The standard ECG has 12 leads, including six limb leads (I, II, III, aVR, aVL, aVF) and six chest leads (
to
).
Figure 1 shows the three-dimensional view of 12 standard leads on the
-coordinate axis system. According to electrode positioning, the 12 ECG leads can be used to localize different types of MI, such as inferior leads (II, III, aVF), septal leads
, anterior leads
, and lateral leads (I, aVL,
,
). A typical waveform of the ECG beat consists of a P wave, a QRS-complex, and a T wave. These characteristic waves correspond to the sequence of depolarization and repolarization of the atria and ventricles. ECG signs suggestive of MI include ST-segment deviation or changes in the shapes of Q-wave and T-wave, using which physicians can localize damage to specific areas of the heart. However, it may be noted that 12-lead ECG requires ten electrodes for recording and some of the leads contain redundant information. Instead, VCG requires a minimum of four electrodes and it monitors cardiac electrical activity in three orthogonal planes of the body [
5]. Generally, Frank leads
scanned in orthogonal xyz axes are used for VCG measurements. The main advantage of VCG is that it uses fewer leads than 12-lead ECG for medical diagnostic applications. Moreover, different studies [
6,
7,
8] have demonstrated that VCG provides a higher sensitivity for the diagnosis of MI as well as ischemic heart diseases. In this study, VCG signal is processed to extract clinically significant features that will allow for MI classification.
MI is also known as a silent heart attack that usually occurs without clear symptoms. Hence, early diagnosis and timely treatment are crucial to improve the recovery rate of MI patients. In recent years, several computer-aided diagnostic methods have been proposed for automatic MI detection and localization [
9,
10,
11,
12,
13,
14,
15,
16,
17,
18]. Most of these approaches extract the clinically significant features from the ECG signal and then apply an appropriate classifier in the classification stage. Various informative features have been extracted to represent the ECG beats, such as morphological features [
10] as well as frequency and wavelet-based features [
11,
12]. Moreover, some studies have attempted to use directly measured or derived VCG to identify changes in the VCG morphology such as the QRS and T-wave loops [
16,
17,
18]. For classification, different machine learning algorithms have been investigated, including k-nearest neighbors (KNN) [
10,
12], artificial neural network (ANN) [
11], recurrent neural network (RNN) [
13] and convolutional neural network (CNN) [
14,
15]. Furthermore, several researchers [
13,
14,
15] have proposed end-to-end approaches for MI detection and localization. These methods obviate the need to extract features at the cost of higher computational complexity. ECG abnormalities due to MI may be observed in the ST-segment deviation or changes in the shapes of T-wave and Q-wave. Generally, it is a prerequisite to identify characteristic waves of ECG beats before performing the feature extraction. Although various methods have been proposed for ECG wave delineation [
19,
20,
21,
22], they still have some limitations for characterization of MI beats. To address this constraint, we apply spline curve fitting [
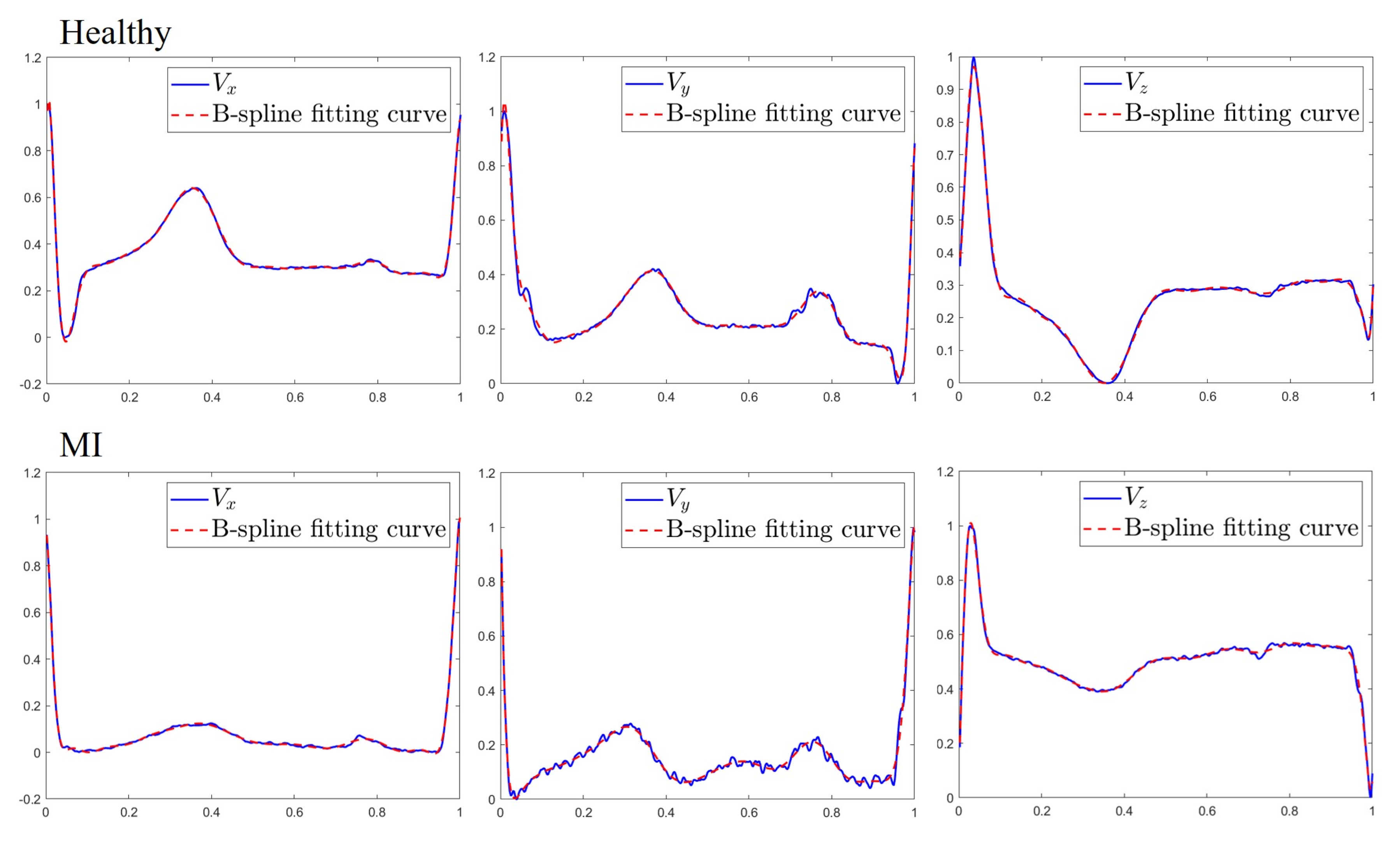
23,
24] to the entire heartbeat to model all of the characteristic waves and use fitted coefficients as features. The advantage of using the entire heartbeat is that the QRS complexes and P and T waves can be included in the curve fitting so that poor quality features resulting from delineation errors can be avoided. Moreover, the VCG signal is semiperiodic in nature and has numerous clinically relevant turning points in each heartbeat. Such signals require a higher-order polynomial to fit, leading to severe oscillations of the fitted curve which cause the overfitting problem [
25]. By contrast, the spline’s flexibility in approximating curves with different degrees of smoothness at different locations is ideal for representing the semiperiodic VCG signal.
Another problem which requires further investigation is to test the feasibility of single-lead ECG in classifying different types of MI. Several wearable devices which use single-lead ECG to facilitate continuous ambulatory monitoring have recently appeared on the market [
26]. While these devices make regular ECG recording possible, their practical applicability for cardiac diagnostics remain limited. This is because physicians need checking ECG patterns to diagnose by correlating information from two or more ECG leads. For example, abnormalities in chest leads (
to
) are suggestive of a problem in the posterior wall of the heart and no abnormalities will be detected by a single lead [
27]. The ability to transform from single-lead ECG to 12-lead ECG enables the wider use of wearable devices for clinical diagnostic applications. However, prior attempts to synthesize 12-lead ECG or 3-lead VCG from a single lead have not been successful. Most existing lead transformation approaches require at least two synchronously acquired leads [
28,
29,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39,
40], hampering their applicability to the present context. This has motivated our investigation into trying to synthesize the 3-lead VCG from single-lead ECG signal. Since lead I is provided by most wearable devices, we propose a derived VCG system by considering the lead I ECG signal as input and three Frank leads as output of the system.
A lot of emphases have been recently put on derived ECG systems due to the increasing demand of personalized healthcare applications. The methods of lead synthesis can be categorized in terms of reconstruction algorithms and lead configuration. The lead configuration for ECG synthesis can be divided into two groups: use of subsets of 12-lead ECG [
28,
29,
30,
31] and use of Frank VCG leads [
32,
33,
34,
35,
36,
37,
38,
39,
40]. A common assumption in previous works was that the heart-torso electrical system is linear and quasi-static, which allows for the use of linear transformation to derive the 3-lead VCG from reduced-lead set of the 12-lead ECG. These can either be patient-specific or generic transformation of which the former is learned using data from a single patient, while the latter requires data from a group of patients. Previous studies have shown the possibility to derive the 12-lead ECG from the three Frank XYZ leads through Dower transformation [
34] and vice versa through the inverse Dower transformation [
35]. Similarly, Kors et al. [
36] derived the transformation matrix using the regression analysis method. In [
37], Dawson et al. derived the linear affine transformation between 3-lead VCG and 12-lead ECG, which achieved higher accuracy than Kors and inverse Dower transformation. Another strategy can be seen in [
31,
32], where nonlinear methods such as ANN were used to synthesize the 12-lead ECG and 3-lead VCG from leads I, II, and
. It was found that nonlinear transformation are appropriate for ECG data with diversity resulting from variation in individuals and measurement positions. A weakness for majority of the reviewed methods is that they only exploited the inter-lead correlation between spatially aligned samples of the lead signals. It is important to note that, in addition to spatially correlated information in different leads, temporally correlated information can also be found between different waves within a single lead. System design approaches that consider both intra- and inter-lead correlation are expected to provide better solutions to the VCG synthesis problem. This task can be accomplished by using RNN [
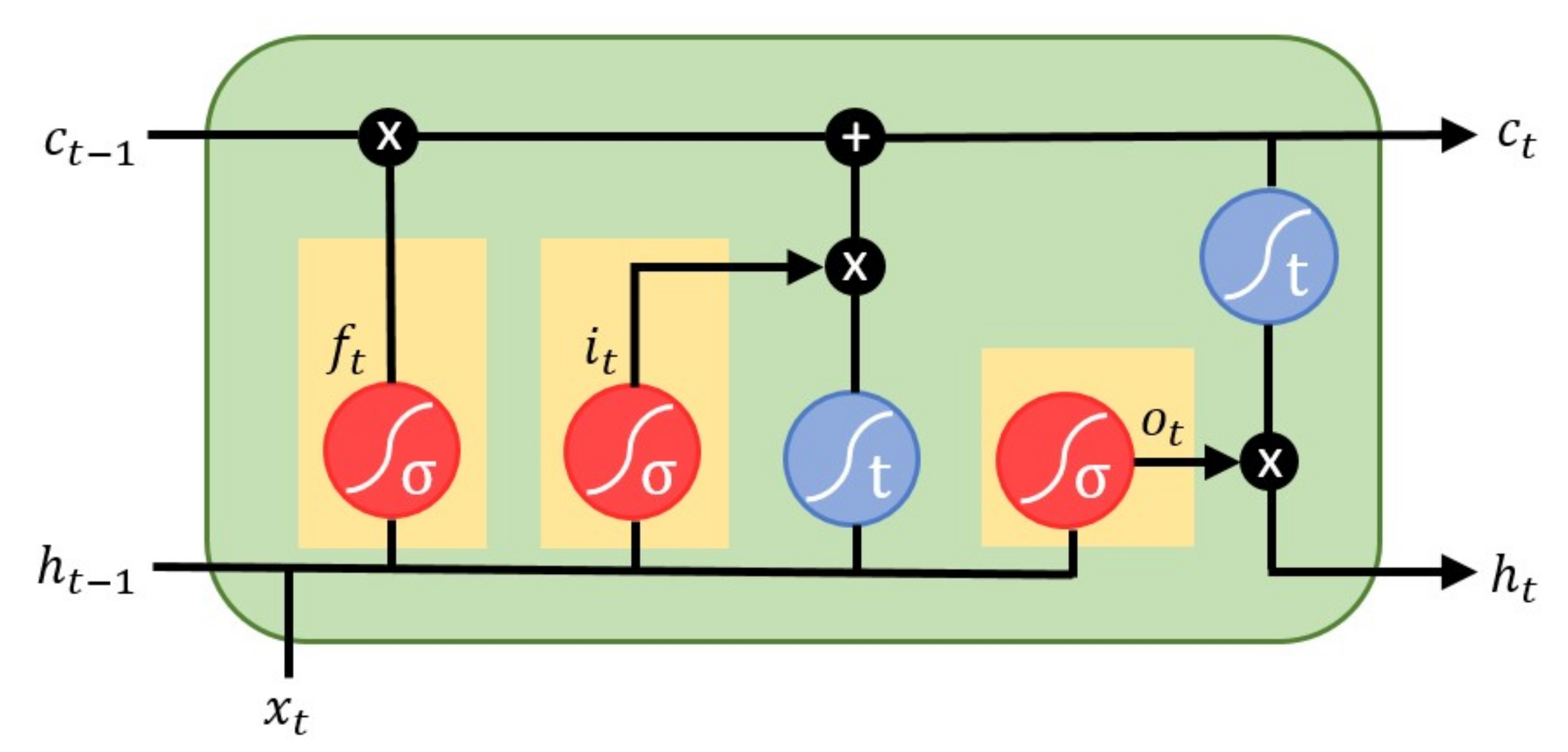
41] as it can use the learning capabilities of ANN and could further improve it by representing the spatio-temporal correlations between the lead signals. In this work, we proposed a patient-specific transformation for VCG synthesis by applying a long short-term memory (LSTM) network [
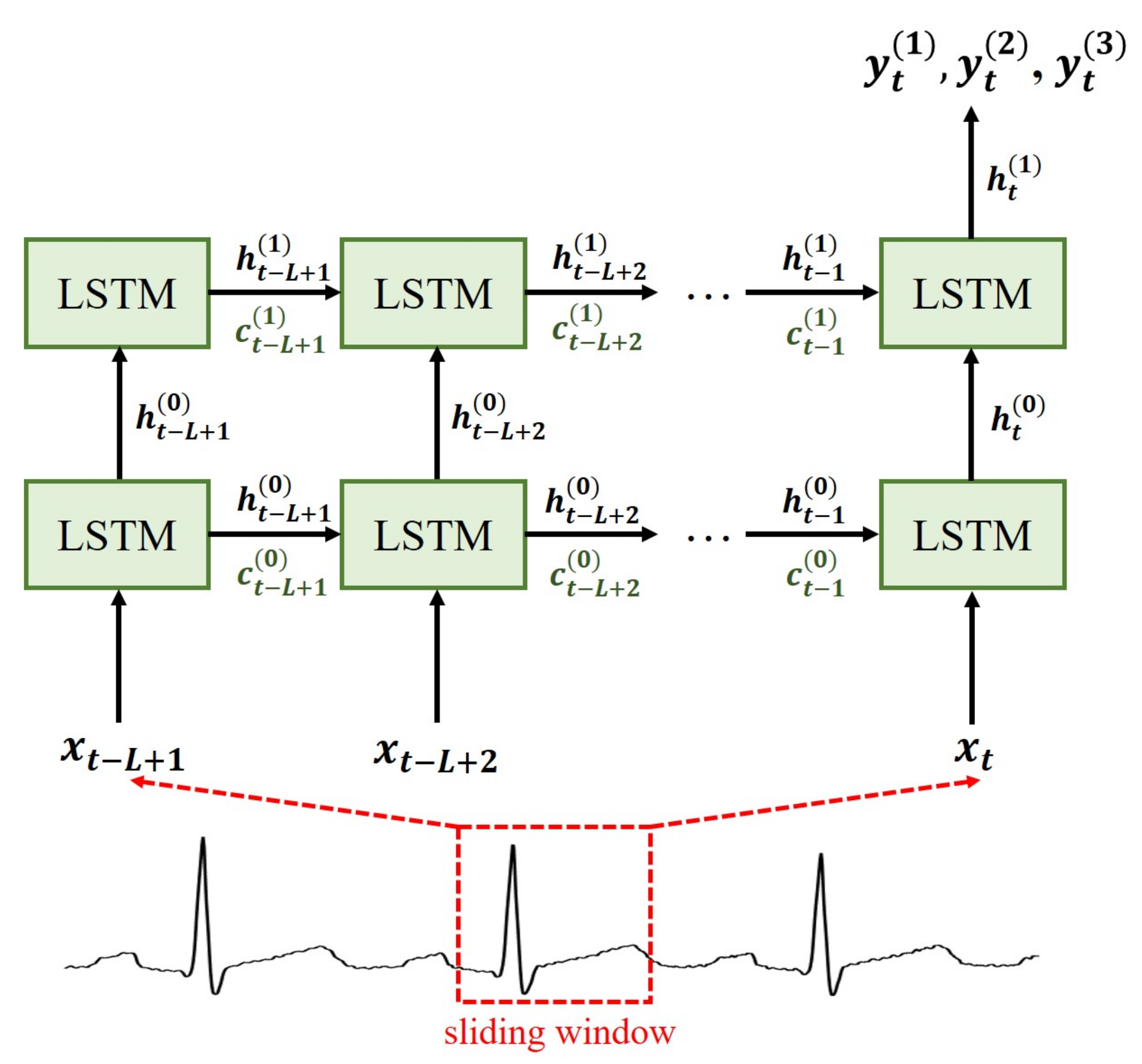
42] with sliding window approach.
This study focuses on two issues: synthesis of 3-lead VCG and extraction of VCG features, to develop an MI classifier that is suitable for wearable devices with only a single lead recording. The first part of this study focuses on developing a method of VCG reconstruction from lead I ECG using a LSTM network to exploit both intra- and inter-lead correlations of ECG signals. The second part of this study develops a novel spline framework for parametrically representing the derived Frank lead signals. After extracting features by the spline approximation, a classifier is used for the classification of healthy and 11 types of MI.
3. Evaluation Parameters
In this study, ECG records were taken from the Physikalisch-TechnischeBundesanstalt (PTB) [
50] diagnostic database. The PTB database consists of 549 ECG records from 290 subjects and each record contains 12 ECG leads and 3 Frank VCG leads. From the database, a total of 26,080 heartbeats from 52 healthy subjects and 143 MI patients were included in the analysis.
Table 1 shows the number of heartbeats for each type of MI and healthy subjects in this study. These data were further divided into 12 classes of ECG beats: anterior (AMI), anterior-lateral (ALMI), anterior-septal (ASMI), anterior-septal-lateral (ASLMI), inferior (IMI), inferior-lateral (ILMI), inferior-posterior (IPMI), inferior-posterior-lateral (IPLMI), lateral (LMI), posterior (PMI), posterior-lateral (PLMI), and healthy control (HC).
Root-mean-square-error (RMSE) and correlation coefficient (CC) were chosen to test the accuracy of derived VCG by the individual methods in relation to the measured VCG. RMSE measures the similarity of two recordings and it is defined as Equation (
10), where
V is the original value of the measured VCG,
is the value of the derived VCG, and
N is the number of samples. Instead, CC is a statistic that measures the correlation between two recordings, which is defined in Equation (
11).
To test the feasibility of the proposed MI classifiers, the performance analysis is based on the accuracy (ACC), sensitivity (SEN), and specificity (SPE) represented in the form of confusion matrix [
51]. These performance metrics are related to the number of true positives (TP), true negatives (TN), false positives (FP), and false negatives (FN). The accuracy is the proportion of correctly classified samples to the total number of samples, and it is defined as Equation (
12). Sensitivity, defined in Equation (
13), measures the proportion of positives that are correctly identified. Instead, specificity measures the proportion of negatives that are correctly identified and defined as Equation (
14).
4. Results
Computer simulations were conducted to evaluate the validity of the proposed method in differentiating 11 types of MI and healthy subjects. A preliminary experiment was first conducted to examine the performance dependence of VCG synthesis on the sliding window size
L employed in constructing the LSTM models. In this experiment, ECG recordings from 20 HC subjects and 20 MI patients were used.
Table 2 presents the RMSE and CC between measured and derived Frank XYZ leads. Based on the results, we empirically chose
in the sequel.
We next compare the reconstruction performance of using MLP [
32] and LSTM for learning the derived VCG models. All the experiments were based on the evaluation of RMSE and CC and experimental results were obtained by five-fold cross-validation. ECG recordings from 52 HC subjects are denoted as dataset DS1, and ECG recordings from 143 MI patients are denoted as dataset DS2. For comparison purposes, the MLP consists of one input layer with 150 neurons, one output layer with three neurons, two hidden layers and 150 neurons per hidden layer. The results of VCG synthesis by five-fold cross-validation are presented in
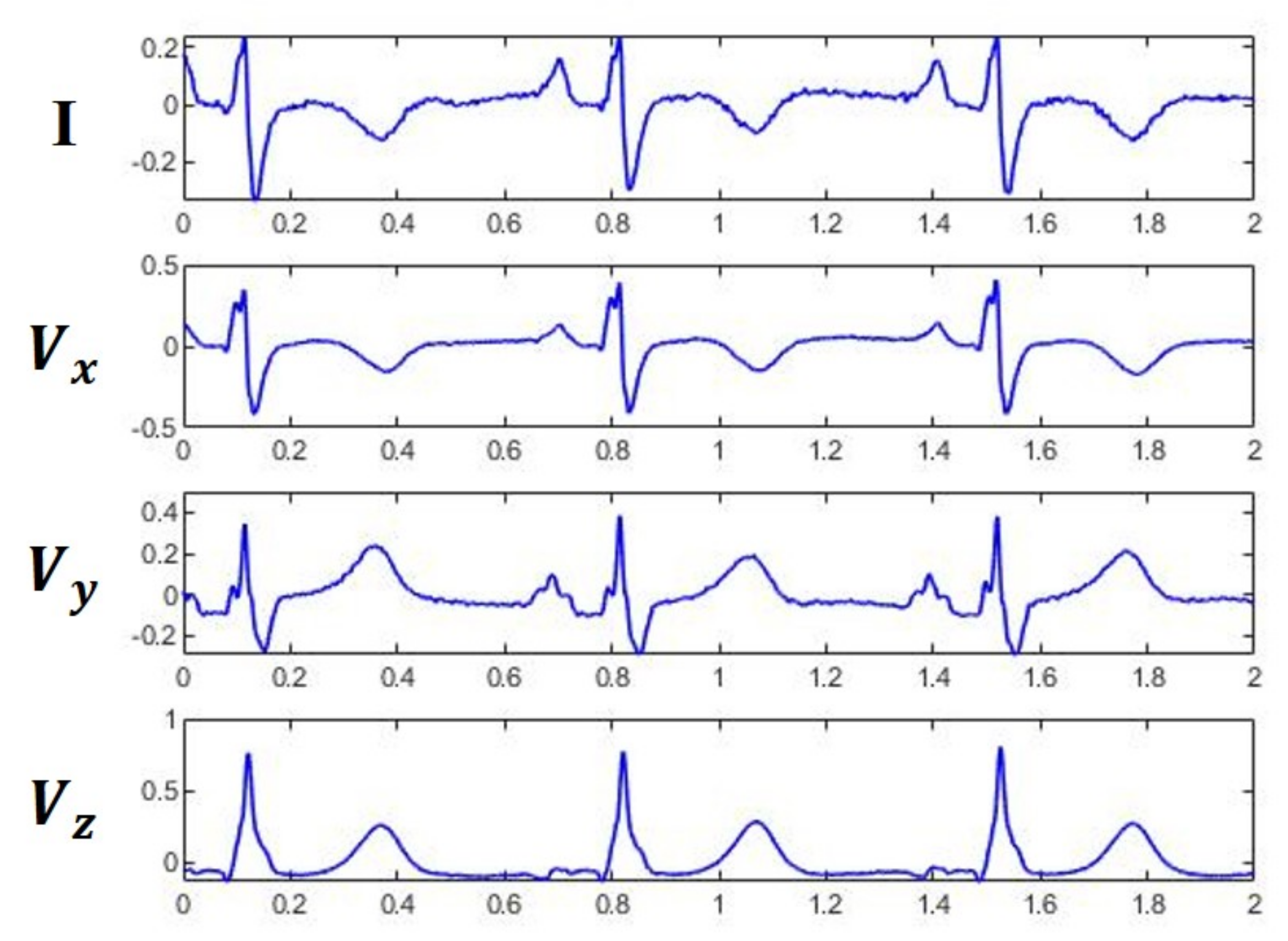
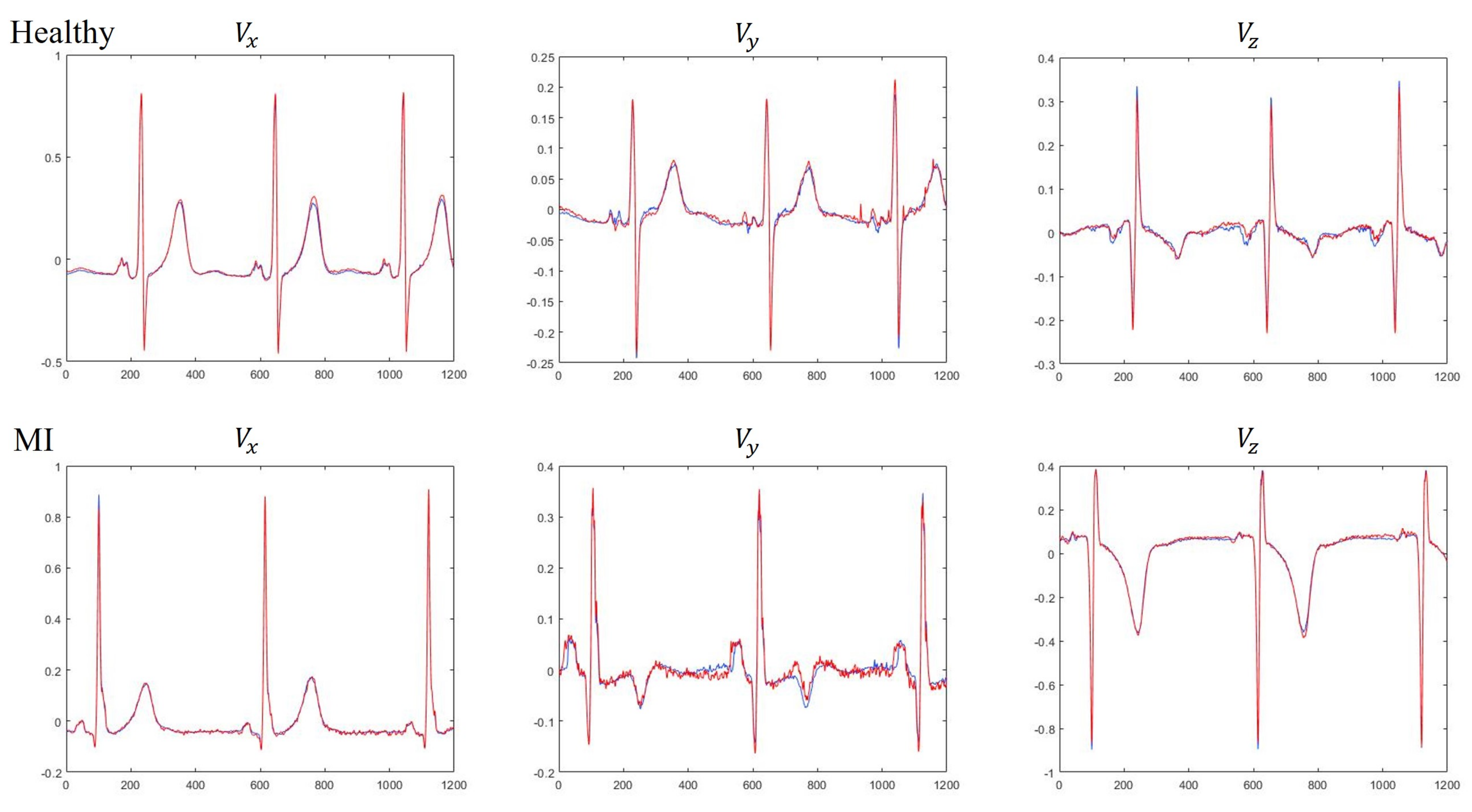
Table 3. The results clearly demonstrate that the LSTM is preferred to MLP for use in constructing the VCG synthesizer because the LSTM can exploit both intra- and inter-lead correlations of ECG signals. Further analysis indicates that the average CC of three Frank leads using LSTM were 0.9943 and 0.9807 for dataset DS1 and DS2, respectively, suggesting that the MI patient data was less accurately reconstructed than HC subjects. Visual inspection of the reconstructed signals showed that the derived VCG signals were not significantly different from the measured signals. A typical example for measured and derived Frank XYZ leads is depicted in
Figure 7.
Next, we assess the performance of MLP classifiers for the classification of normal and 11 MI classes. One problem with the PTB database is the high imbalance between the number of heartbeats belonging to each ECG class. Training an MLP classifier with unbalanced data usually leads to a certain bias towards the majority class. Recognizing this, we applied the Synthetic Minority Over-sampling Technique (SMOTE) [
52] before starting the training process. Moreover, we used 5-fold cross-validation technique to train and test the MLP classifiers. We began testing the MLP classifiers for the situation where MI classes were identified solely by means of single-lead feature data. The classification performance for each ECG class is summarized in
Table 4. Simulation results indicated that using lead I yielded an overall accuracy of
, suggesting that it cannot provide sufficient cues for reliable classification. To elaborate further, we show in
Table 5 the confusion matrix of all the 12 classes for lead I ECG beats. It was found that the notably low classification accuracy can be attributed to the high confusions made across anterior MI group (AMI, ALMI, ASMI, ASLMI) and inferior MI group (IMI, ILMI, IPMI, IPLMI). For instance,
of the ECG beats notated in ASMI were classified as representing IMI and
of the IMI beats were classified as being ASMI. Furthermore, results indicate that derived Frank leads
and
are preferred to lead I ECG for use in constructing the MI classifier. Notably, the use of lead
yielded an overall accuracy of
, compared with
for lead I and
for
. The confusion matrices obtained using derived Frank lead
and
are shown in
Table 6 and
Table 7, respectively. Further analysis indicates that anterior and inferior MI groups are dominant in the MI groups that benefited the most from exploitation of derived VCG leads. In case of inferior MI group, the average sensitivity has increased from
in lead I to
in lead
. Similarly in case of anterior MI group, average sensitivity obtained for lead I and
is
and
, respectively. We speculate that this might be attributed to the difference in closeness between Frank leads and 12 ECG leads. Support for such a speculation can be found in [
39], where the authors showed that Frank lead
is most likely associated with inferior leads (II, III, aVF), and Frank lead
is closest to subset of anteroseptal leads
. We can also see from
Figure 1 that leads
and
are located near the negative Z-axis in the sagittal plane. Similarly, it can be found that leads II, III and aVF are oriented along the Y-axis.
Next, we examine whether combining multiple derived Frank leads would improve the classification performance.
Table 8 shows the MLP classifier results for healthy and 11 types of MI ECG beats obtained using various lead configurations. Our proposed method yielded the best performance with an overall accuracy of
, sensitivity of
and specificity of
in MI classification, by using 52 features obtained from the derived Frank XYZ leads. The results also indicate that the ability of derived VCG to correctly identify the MI classes is almost identical to that of measured VCG.
Table 9 shows the confusion matrix of all classes obtained using MLP classifier on the derived Frank XYZ leads. A comparison between
Table 5 and
Table 9 indicates that the improvement can be seen in the following areas. First, the derived VCG can reduce a significant portion of confusions across anterior and inferior MI groups. For instance, only
of ECG beats notated in ASMI were misclassified as representing IMI, the corresponding value for lead I being
. Second, the derived VCG significantly increased the sensitivity of healthy subjects to
, compared with
for lead I,
for
, and
for
. The results clearly demonstrate that MI classification by computational means is significantly improved when clinically significant features relating to the derived VCG are taken into account.
5. Discussion
In recent years, numerous approaches were proposed to identify various types of MI from ECG records. The numbers of ECG leads and MI classes are important factors correlated with diagnosis efficiency, and should be noted when comparing their relative performances.
Table 10 summarizes the studies employing different techniques in MI classification with the same PTB database. Arif et al. [
10] used 12 lead ECG signal and time domain features such as T-wave amplitude, Q-wave and ST-level elevation, reporting overall accuracy of
on ten different MI classes with a KNN classifier. Alternatively, Noorian et al. [
11] used ANN classifier and wavelet coefficients as features extracted from the derived VCG. Acharya et al. [
12] have evaluated ten MI classes with 12 types of nonlinear features based on wavelet transform. They obtained an accuracy of 98.74%, sensitivity of
, and specificity of
by only using lead V3 ECG signal. Lui et al. [
13] combined the power of CNN and RNN, and achieved
sensitivity and
specificity for classification of MI as well as other cardiovascular diseases. Baloglu et al. [
14] proposed an end-to-end approach based on deep CNN and reported an overall accuracy of
by using 12 lead ECG signal for classification into 11 types of ECG beats. In [
15], a multi-lead attention mechanism integrated with CNN and bidirectional gated recurrent unit was applied for MI classification based on six classes of 12-lead ECG records, namely HC, AMI, ALMI, ASMI, IMI, and ILMI. Towards addressing the challenges in identifying MIs using wearable devices, our work, as well as some earlier studies [
12,
13], was focused on single-lead rather than 12-lead exploration. Results reported in this paper are generally better than those of MI classifiers in the literature, with its performance only slightly lower than that of [
14]. However, our proposed method applies single-lead derived VCG for classification into 12 types of ECG beats, in which ASLMI with larger necrotic area is ignored in [
14]. Overall, the proposed method obtained an accuracy of
, sensitivity of
and specificity of
. With this performance, our proposed model has the potential to provide an early and accurate diagnosis of MI in wearable ECG monitoring devices.