Assessment of Machine Learning Models to Identify Port Jackson Shark Behaviours Using Tri-Axial Accelerometers
Abstract
1. Introduction
2. Materials and Methods
2.1. Captive Observations
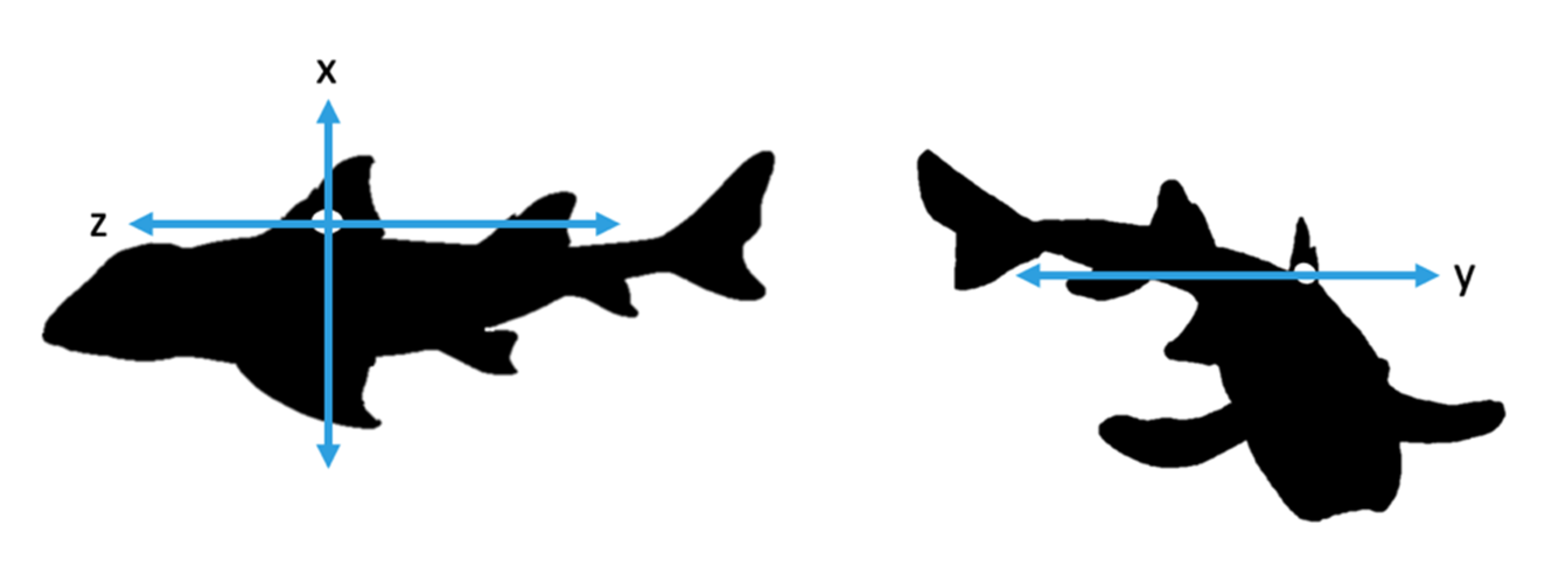
2.2. Accelerometer Attachment and Specifications
2.3. Video Observations
2.4. Data Processing and Feature Analysis
2.5. Swim Column and Swim Floor Behaviours
2.6. Classification Models
2.7. Model Performance Assessment
3. Results
3.1. Sampling
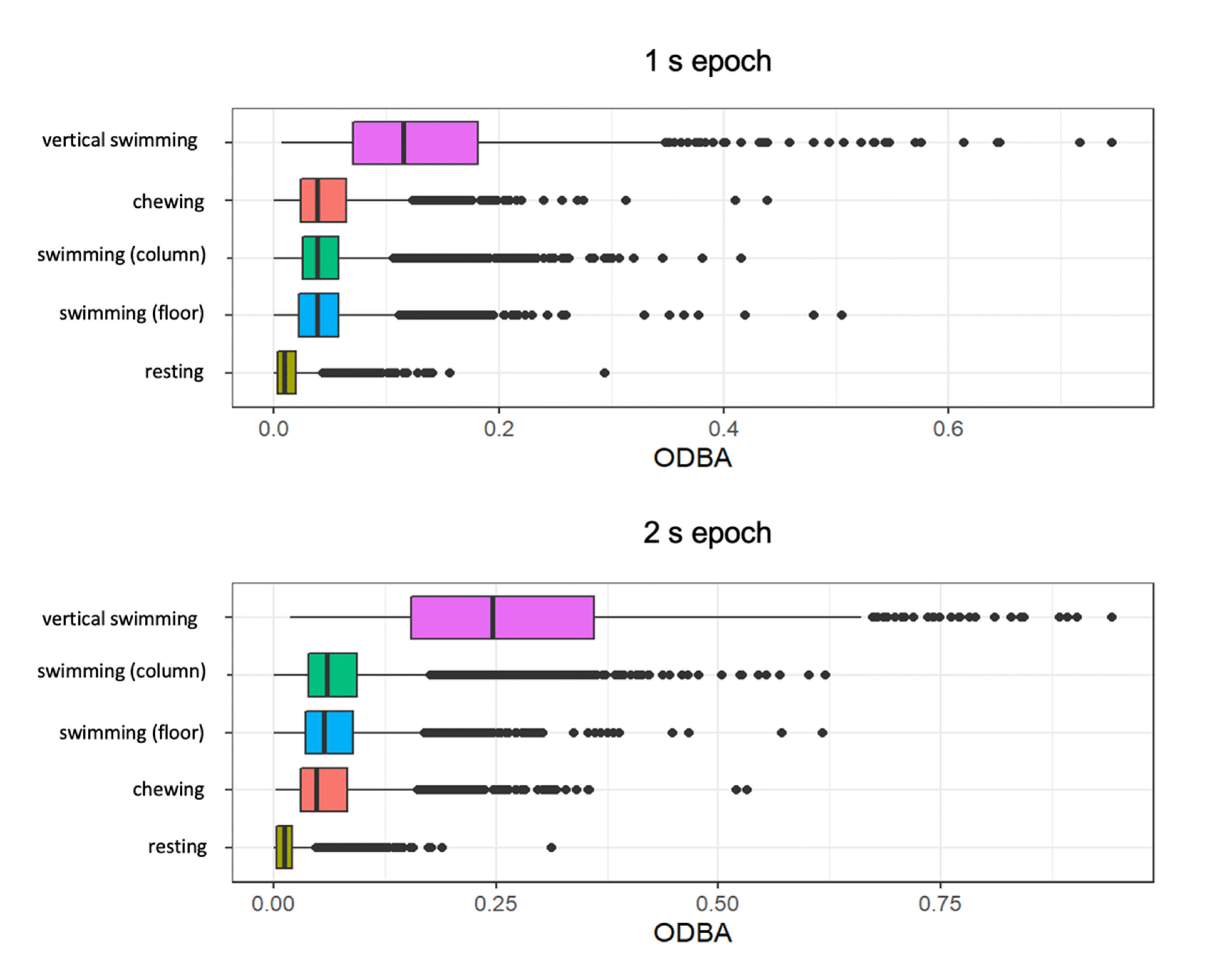
3.2. ODBA for All Behaviours
3.3. Swimming in the Water Column vs. Swimming on the Floor
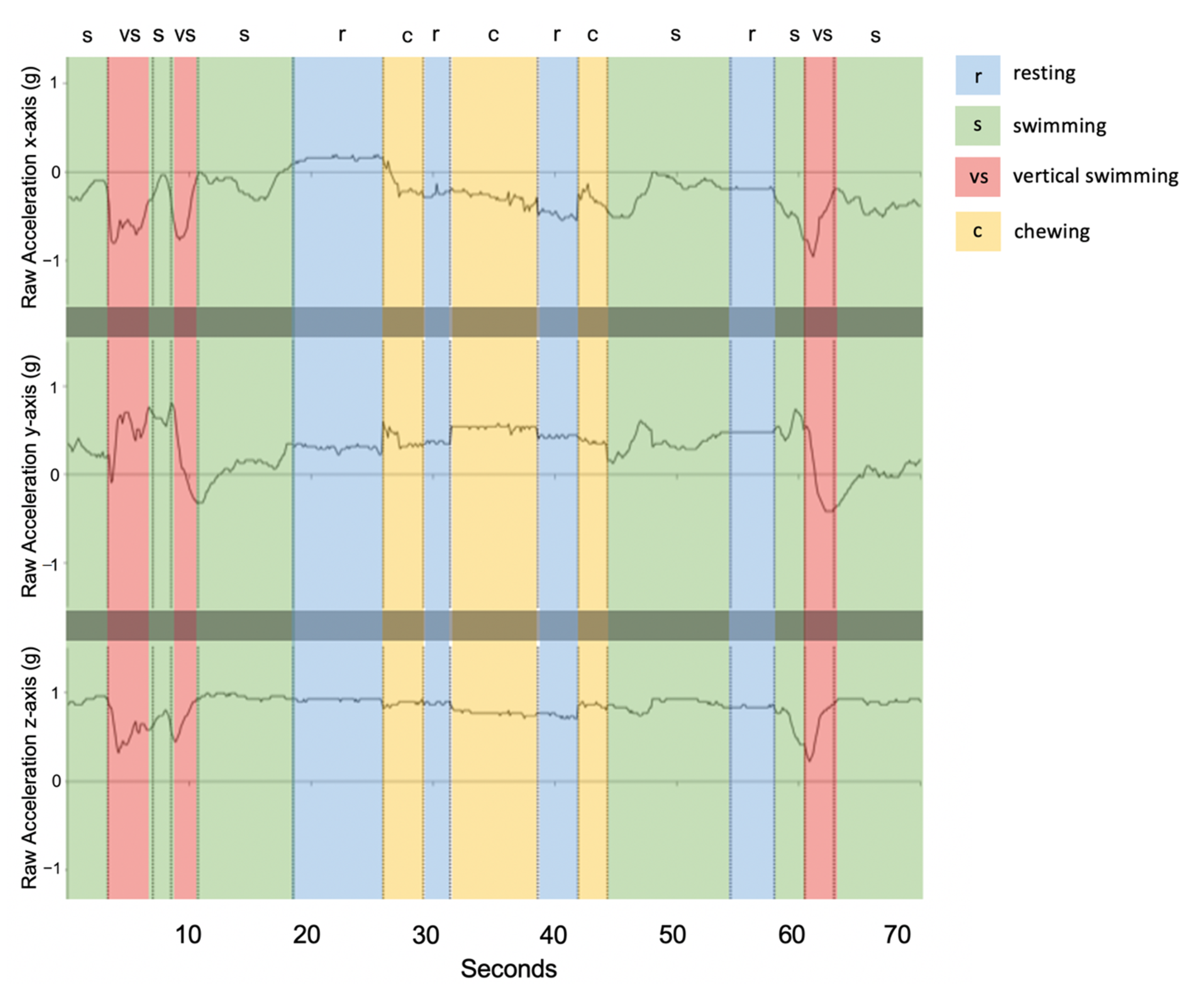
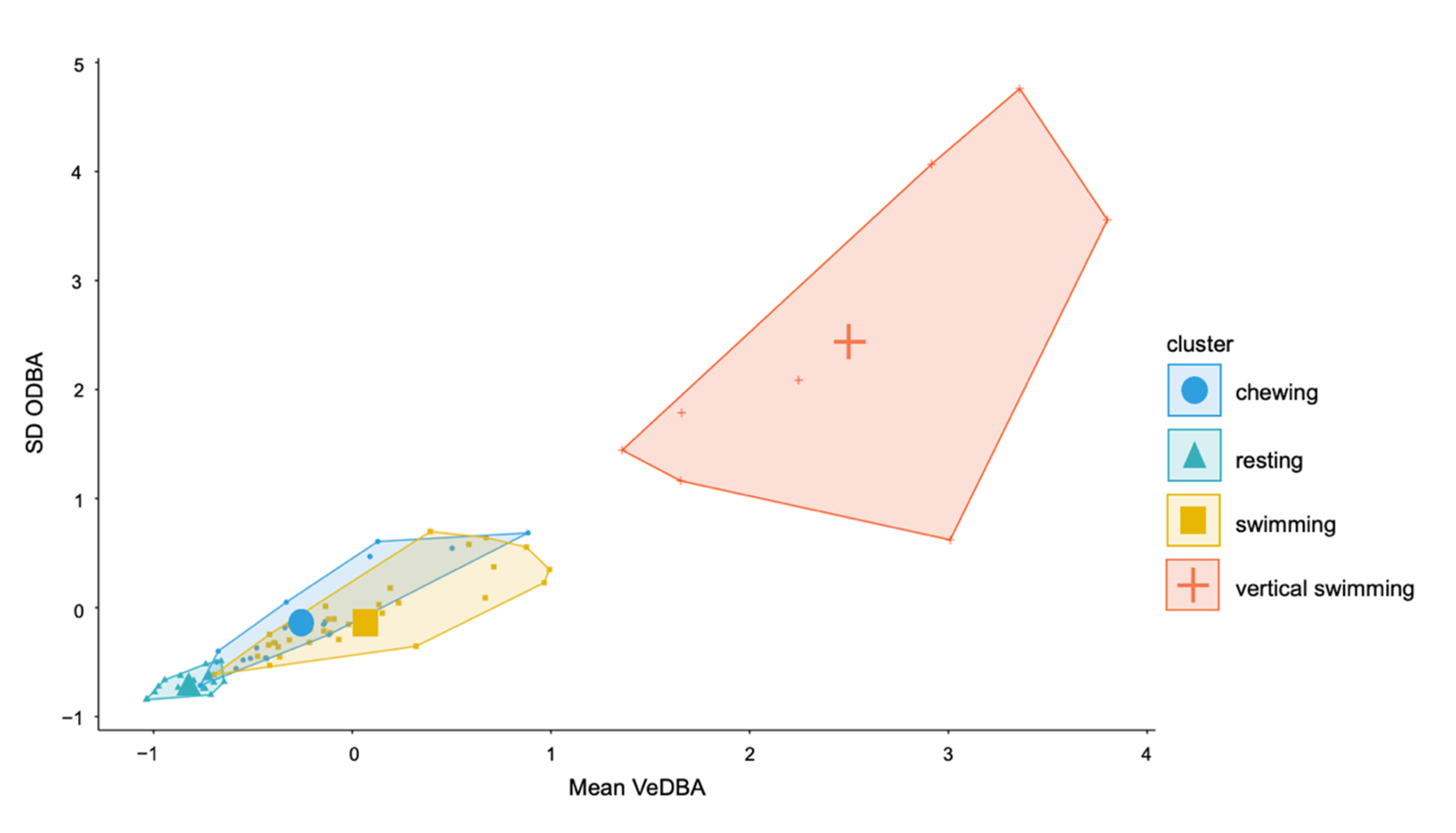
3.4. Description of Behaviours
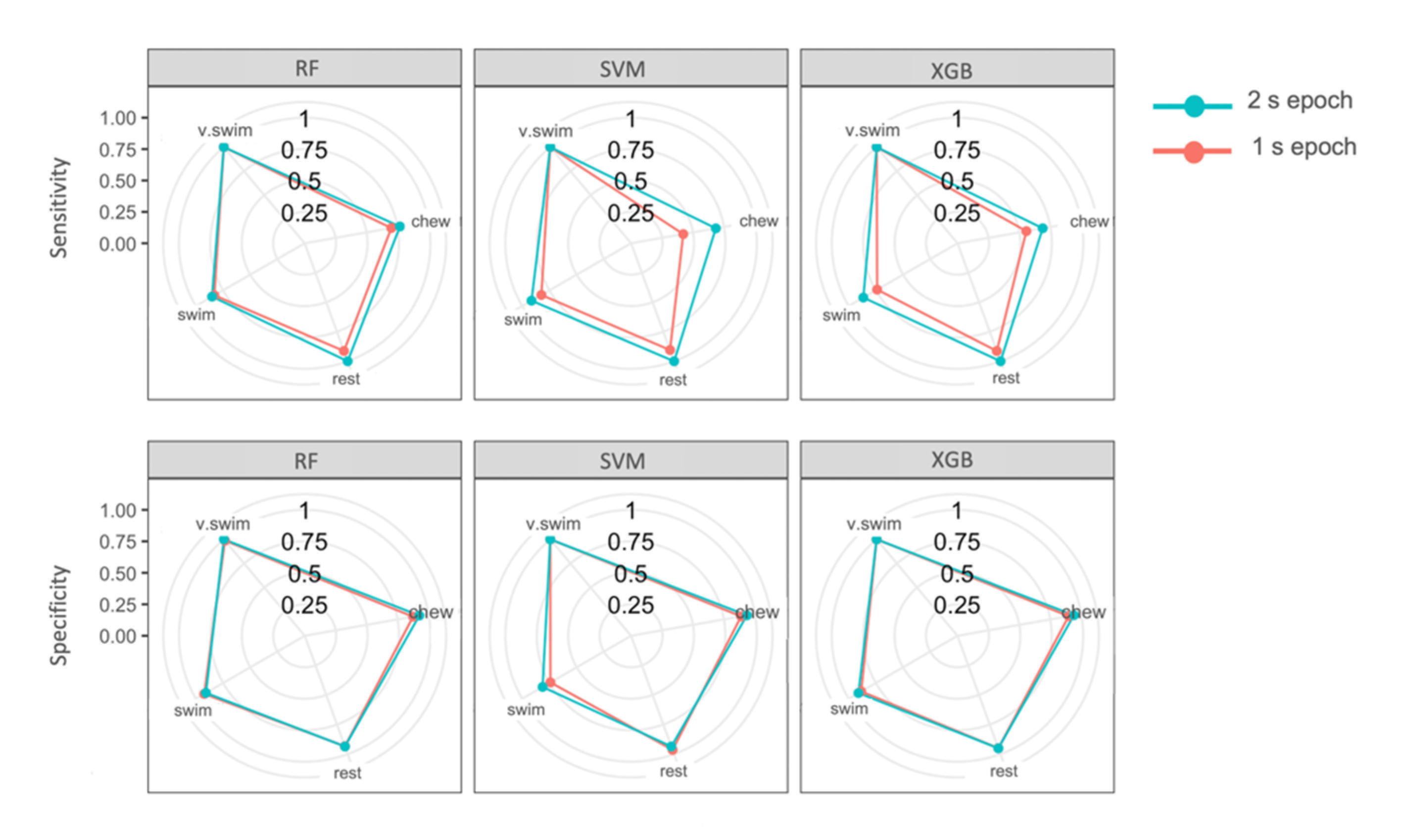
3.5. Model Classification
3.6. Feature Importance
4. Discussion
4.1. Machine Learning
4.2. Ethogram
4.3. Epochs
4.4. Summary Feature Importance
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lea, J.S.E.; Humphries, N.E.; Von Brandis, R.G.; Clarke, C.R.; Sims, D.W. Acoustic telemetry and network analysis reveal the space use of multiple reef predators and enhance marine protected area design. Proc. R. Soc. B Biol. Sci. 2016, 283, 20160717. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.C.; Mansfield, K.L.; Thums, M.; Meekan, M.G. Satellite Tracking Technologies and Their Application to Shark Movement Ecology. In Shark Research: Emerging Technologies and Applications for the Field and Laboratory; Carrier, J., Heithaus, M.R., Simpendorfer, C.A., Eds.; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Nakamura, I.; Watanabe, Y.; Papastamatiou, Y.; Sato, K.; Meyer, C. Yo-yo vertical movements suggest a foraging strategy for tiger sharks Galeocerdo cuvier. Mar. Ecol. Prog. Ser. 2011, 424, 237–246. [Google Scholar] [CrossRef]
- Hussey, N.; Kessel, S.; Aarestrup, K.; Cooke, S.J.; Cowley, P.D.; Fisk, A.T.; Harcourt, R.G.; Holland, K.N.; Iverson, S.J.; Kocik, J.F.; et al. Aquatic animal telemetry: A panoramic window into the underwater world. Science 2015, 348, 1255642. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.D.; Kays, R.; Wikelski, M.; Wilson, R.; Klimley, A.P. Observing the unwatchable through acceleration logging of animal behavior. Anim. Biotelem. 2013, 1, 20. [Google Scholar] [CrossRef]
- Valletta, J.J.; Torney, C.; Kings, M.; Thornton, A.; Madden, J. Applications of machine learning in animal behaviour studies. Anim. Behav. 2017, 124, 203–220. [Google Scholar] [CrossRef]
- Leos-Barajas, V.; Photopoulou, T.; Langrock, R.; Patterson, T.A.; Watanabe, Y.Y.; Murgatroyd, M.; Papastamatiou, Y.P. Analysis of animal accelerometer data using hidden Markov models. Methods Ecol. Evol. 2017, 8, 161–173. [Google Scholar] [CrossRef]
- Wang, G. Machine learning for inferring animal behavior from location and movement data. Ecol. Inform. 2019, 49, 69–76. [Google Scholar] [CrossRef]
- Ladds, M.A.; Thompson, A.P.; Slip, D.J.; Hocking, D.P.; Harcourt, R.G. Seeing It All: Evaluating Supervised Machine Learning Methods for the Classification of Diverse Otariid Behaviours. PLoS ONE 2016, 11, e0166898. [Google Scholar] [CrossRef]
- Chakravarty, P.; Cozzi, G.; Ozgul, A.; Aminian, K. A novel biomechanical approach for animal behaviour recognition using accelerometers. Methods Ecol. Evol. 2019, 10, 802–814. [Google Scholar] [CrossRef]
- Nathan, R.; Spiegel, O.; Fortmann-Roe, S.; Harel, R.; Wikelski, M.; Getz, W.M. Using tri-axial acceleration data to identify behavioral modes of free-ranging animals: General concepts and tools illustrated for griffon vultures. J. Exp. Biol. 2012, 215, 986–996. [Google Scholar] [CrossRef]
- Brewster, L.R.; Dale, J.J.; Guttridge, T.L.; Gruber, S.H.; Hansell, A.C.; Elliott, M.; Cowx, I.G.; Whitney, N.M.; Gleiss, A.C. Development and application of a machine learning algorithm for classification of elasmobranch behaviour from accelerometry data. Mar. Biol. 2018, 165, 62. [Google Scholar] [CrossRef]
- Riaboff, L.; Poggi, S.; Madouasse, A.; Couvreur, S.; Aubin, S.; Bédère, N.; Goumand, E.; Chauvin, A.; Plantier, G.; Poggi, S.; et al. Development of a methodological framework for a robust prediction of the main behaviours of dairy cows using a combination of machine learning algorithms on accelerometer data. Agriculture 2020, 169, 105179. [Google Scholar] [CrossRef]
- Mansbridge, N.; Mitsch, J.; Bollard, N.; Ellis, K.; Miguel-Pacheco, G.; Dottorini, T.; Kaler, J. Feature Selection and Comparison of Machine Learning Algorithms in Classification of Grazing and Rumination Behaviour in Sheep. Sensors 2018, 18, 3532. [Google Scholar] [CrossRef]
- Chen, T.; Guestrin, C. XGBoost: A Scalable Tree Boosting System. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; pp. 785–794. [Google Scholar]
- Leoni, J.; Tanelli, M.; Strada, S.C.; Berger-Wolf, T. Ethogram-based automatic wild animal monitoring through inertial sensors and GPS data. Ecol. Inform. 2020, 59, 101112. [Google Scholar] [CrossRef]
- Karan, S.; Meese, E.N.; Yang, Y.; Yeh, H.G.; Lowe, C.G.; Zhang, W. Classification of Shark Behaviors using K-Nearest Neighbors. In Proceedings of the 2019 IEEE Green Energy and Smart Systems Conference, IGESSC 2019, Long Beach, CA, USA, 4–5 November 2019; Institute of Electrical and Electronics Engineers Inc.: Piscataway, NJ, USA, 2019. [Google Scholar]
- Kadar, J.; Ladds, M.; Mourier, J.; Day, J.; Brown, C. Acoustic accelerometry reveals diel activity patterns in premigratory Port Jackson sharks. Ecol. Evol. 2019, 9, 8933–8944. [Google Scholar] [CrossRef]
- Bass, N.C.; Mourier, J.; Knott, N.A.; Day, J.; Guttridge, T.; Brown, C. Long-term migration patterns and bisexual philopatry in a benthic shark species. Mar. Freshw. Res. 2017, 68, 1414–1421. [Google Scholar] [CrossRef]
- Last, P.R.; Stevens, J.D.; Swainston, R.; Davis, G. Sharks and Rays of Australia, 2nd ed.; CSIRO Australia, Ed.; CSIRO Publishing: Collingwood, Australia, 2009; ISBN 9780643094574 (hbk.). [Google Scholar]
- Powter, D.M.; Gladstone, W.; Platell, M. The influence of sex and maturity on the diet, mouth morphology and dentition of the Port Jackson shark, Heterodontus portusjacksoni. Mar. Freshw. Res. 2010, 61, 74. [Google Scholar] [CrossRef]
- Hounslow, J.L.; Brewster, L.R.; Lear, K.O.; Guttridge, T.L.; Daly, R.; Whitney, N.M.; Gleiss, A.C. Assessing the effects of sampling frequency on behavioural classification of accelerometer data. J. Exp. Mar. Biol. Ecol. 2019, 512, 22–30. [Google Scholar] [CrossRef]
- Chen, K.Y.; Bassett, D.R., Jr. The Technology of Accelerometry-Based Activity Monitors: Current and Future. Med. Sci. Sports Exerc. 2005, 37, S490–S500. [Google Scholar] [CrossRef]
- Shepard, E.; Wilson, R.; Halsey, L.; Quintana, F.; Gómez Laich, A.; Gleiss, A.; Liebsch, N.; Myers, A.; Norman, B. Derivation of body motion via appropriate smoothing of acceleration data. Aquat. Biol. 2008, 4, 235–241. [Google Scholar] [CrossRef]
- Jeantet, L.; Planas-Bielsa, V.; Benhamou, S.; Geiger, S.; Martin, J.; Siegwalt, F.; Lelong, P.; Gresser, J.; Etienne, D.; Hiélard, G.; et al. Behavioural inference from signal processing using animal-borne multi-sensor loggers: A novel solution to extend the knowledge of sea turtle ecology. R. Soc. Open Sci. 2020, 7, 200139. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Therneau, T.; Atkinson, B. rpart: Recursive Partitioning and Regression Trees; R Package Version 4.1-15; 2019; Available online: https://cran.r-project.org/web/packages/rpart/rpart.pdf (accessed on 15 October 2020).
- Liaw, A.; Wiener, M. Classification and Regression by RandomForest. R News 2002, 2, 18–22. [Google Scholar]
- Meyer, D.; Dimitriadou, E.; Hornik, K.; Weingessel, A.; Leisch, F. e1071: Misc Functions of the Department of Statistics, Probability Theory Group (Formerly: E1071) TU Wien, R package version 1.7-4; 2020; Available online: https://cran.r-project.org/web/packages/e1071/index.html (accessed on 15 October 2020).
- Kuhn, M.; Quinlan, R. C50: C5.0 Decision Trees and Rule-Based Models; R Package Version 0.1.3.1; 2020; Available online: https://cran.r-project.org/web/packages/C50/C50.pdf (accessed on 15 October 2020).
- Greenwell, B.; Boehmke, B.; Cunningham, J. GBM Developers. gbm: Generalized Boosted Regression Models, R package version 2.1.8; 2020; Available online: https://cran.r-project.org/web/packages/gbm/gbm.pdf (accessed on 15 October 2020).
- Ripley, B.; Venables, W. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, USA, 2002; ISBN 0-387-95457-0. [Google Scholar]
- Kuhn, M. caret: Classification and Regression Training, R package version 6.0-86; 2020; Available online: https://cran.r-project.org/web/packages/caret/caret.pdf (accessed on 15 October 2020).
- Viera, A.J.; Garrett, J.M. Understanding Interobserver Agreement: The Kappa Statistic. Fam. Med. 2005, 37, 360–363. [Google Scholar]
- Vázquez Diosdado, J.A.; Barker, Z.E.; Hodges, H.R.; Amory, J.R.; Croft, D.P.; Bell, N.J.; Codling, E.A. Classification of behaviour in housed dairy cows using an accelerometer-based activity monitoring system. Anim. Biotelem. 2015, 3, 15. [Google Scholar] [CrossRef]
- Hammond, T.T.; Springthorpe, D.; Walsh, R.E.; Berg-Kirkpatrick, T. Using accelerometers to remotely and automatically characterize behavior in small animals. J. Exp. Biol. 2016, 219, 1618–1624. [Google Scholar] [CrossRef]
- Nishizawa, H.; Noda, T.; Yasuda, T.; Okuyama, J.; Arai, N.; Kobayashi, M. Decision tree classification of behaviors in the nesting process of green turtles (Chelonia mydas) from tri-axial acceleration data. J. Ethol. 2013, 31, 315–322. [Google Scholar] [CrossRef]
- Bom, R.A.; Bouten, W.; Piersma, T.; Oosterbeek, K.; Van Gils, J.A. Optimizing acceleration-based ethograms: The use of variable-time versus fixed-time segmentation. Mov. Ecol. 2014, 2, 6. [Google Scholar] [CrossRef]
- White, B.J.; Amrine, D.E.; Larson, R.L. Big data analytics and precision animal agriculture symposium: Data to decisions. J. Anim. Sci. 2018, 96, 1531–1539. [Google Scholar] [CrossRef]
- Carroll, G.; Slip, D.; Jonsen, I.; Harcourt, R. Supervised accelerometry analysis can identify prey capture by penguins at sea. J. Exp. Biol. 2014, 217, 4295–4302. [Google Scholar] [CrossRef]
- Campbell, H.A.; Gao, L.; Bidder, O.R.; Hunter, J.; Franklin, C.E. Creating a behavioural classification module for acceleration data: Using a captive surrogate for difficult to observe species. J. Exp. Biol. 2013, 216, 4501–4506. [Google Scholar] [CrossRef]
- Fehlmann, G.; O’Riain, M.J.; Hopkins, P.W.; O’Sullivan, J.; Holton, M.D.; Shepard, E.L.C.; King, A.J. Identification of behaviours from accelerometer data in a wild social primate. Anim. Biotelem. 2017, 5, 6. [Google Scholar] [CrossRef]
- Fogarty, E.S.; Swain, D.L.; Cronin, G.M.; Moraes, L.E.; Trotter, M. Behaviour classification of extensively grazed sheep using machine learning. Comput. Electron. Agric. 2020, 169, 105175. [Google Scholar] [CrossRef]
- Rutz, C.; Hays, G.C. New frontiers in biologging science. Biol. Lett. 2009, 5, 289–292. [Google Scholar] [CrossRef]
- Wilson, A.D.M.; Wikelski, M.; Wilson, R.P.; Cooke, S.J. Utility of biological sensor tags in animal conservation. Conserv. Biol. 2015, 29, 1065–1075. [Google Scholar] [CrossRef] [PubMed]
- Jeanniard-du-Dot, T.; Guinet, C.; Arnould, J.P.Y.; Speakman, J.R.; Trites, A.W. Accelerometers can measure total and activity-specific energy expenditures in free-ranging marine mammals only if linked to time-activity budgets. Funct. Ecol. 2017, 31, 377–386. [Google Scholar] [CrossRef]
- Mori, T.; Miyata, N.; Aoyama, J.; Niizuma, Y.; Sato, K. Estimation of metabolic rate from activity measured by recorders deployed on Japanese sea bass Lateolabrax japonicus. Fish. Sci. 2015, 81, 871–882. [Google Scholar] [CrossRef]
- Gleiss, A.C.; Dale, J.J.; Holland, K.N.; Wilson, R.P. Accelerating estimates of activity-specific metabolic rate in fishes: Testing the applicability of acceleration data-loggers. J. Exp. Mar. Biol. Ecol. 2010, 385, 85–91. [Google Scholar] [CrossRef]
- Lowe, C. Metabolic rates of juvenile scalloped hammerhead sharks (Sphyrna lewini). Mar. Biol. 2001, 139, 447–453. [Google Scholar] [CrossRef]
- Gleiss, A.C.; Morgan, D.L.; Whitty, J.M.; Keleher, J.J.; Fossette, S.; Hays, G.C. Are vertical migrations driven by circadian behaviour? Decoupling of activity and depth use in a large riverine elasmobranch, the freshwater sawfish (Pristis pristis). Hydrobiologia 2017, 787, 181–191. [Google Scholar] [CrossRef]
- Lear, K.O.; Whitney, N.M.; Brewster, L.R.; Morris, J.J.; Hueter, R.E.; Gleiss, A.C. Correlations of metabolic rate and body acceleration in three species of coastal sharks under contrasting temperature regimes. J. Exp. Biol. 2017, 220, 397–407. [Google Scholar] [CrossRef]
- Tate, E.E.; Anderson, P.A.; Huber, D.R.; Berzins, I.K. Correlations of Swimming Patterns with Spinal Deformities in the Sand Tiger Shark, Carcharias taurus. Int. J. Comp. Psychol. 2013, 26, 75–82. [Google Scholar]
- Klay, G. Shark dynamics and exhibit design. Drum Croak. 1977, 171, 29–32. [Google Scholar]
- Whitney, N.; Pratt, H.; Pratt, T.; Carrier, J. Identifying shark mating behaviour using three-dimensional acceleration loggers. Endanger. Species Res. 2010, 10, 71–82. [Google Scholar] [CrossRef]
- Rast, W.; Kimmig, S.E.; Giese, L.; Berger, A. Machine learning goes wild: Using data from captive individuals to infer wildlife behaviours. PLoS ONE 2020, 15, e0227317. [Google Scholar] [CrossRef]
- Meese, E.N.; Lowe, C.G. Active acoustic telemetry tracking and tri-axial accelerometers reveal fine-scale movement strategies of a non-obligate ram ventilator. Mov. Ecol. 2020, 8, 8. [Google Scholar] [CrossRef]
- Sakamoto, K.Q.; Sato, K.; Ishizuka, M.; Watanuki, Y.; Takahashi, A.; Daunt, F.; Wanless, S. Can ethograms be automatically generated using body acceleration data from free-ranging birds? PLoS ONE 2009, 4, e5379. [Google Scholar] [CrossRef]
- Nuijten, R.J.M.; Gerrits, T.; Shamoun-Baranes, J.; Nolet, B.A. Less is more: On-board lossy compression of accelerometer data increases biologging capacity. J. Anim. Ecol. 2020, 89, 237–247. [Google Scholar] [CrossRef] [PubMed]

| ID | Sex | Total Length (cm) | Location of Capture |
|---|---|---|---|
| 5541 | F | 127 | Quarantine Point |
| 5542 | F | 124 | Quarantine Point |
| 5544 | F | 117 | Fairlight Beach |
| 5545 | F | 120 | Fairlight Beach |
| Feature | Equation |
|---|---|
| ODBA | |
| VeDBA | |
| Movement variation | |
| Energy | |
| Pitch | |
| Roll |
| Model Category | Model Type | R Package | Model Description |
|---|---|---|---|
| Logic-based | Classification and regression tree (CART) | rpart [27] | - Lightweight and fast decision tree structure that allows for visibility of decisions. - However, they lack the complexity of other methods and may not perform as well as ensemble algorithms. |
| Ensemble | Bagging | ||
| Random forest (RF) | randomForest [28] | - Builds an ensemble of many independent decision trees using different sets of training data that are generated at random and replaced at each selection (known as bagging). - This large number of trees is used to create a consensus and results in the selection of the most common output that will lead to the maximum number of a class in a single node. | |
| Boosting | |||
| Support vector machine (SVM), with radial basis function | e1071 [29] | - Boosting methods fit trees on a modified version of the original data. - By training multiple models additively and in a sequence, these algorithms can identify the errors of weaker, single decision trees. - For example, GBM differs from RF in the order the decision trees are built and the method by which the results are combined. - SVM is an effective tool in datasets with large dimensionality (i.e., a large number of features). | |
| eXtreme gradient boosting (XGB) | xgboost [15] | ||
| C5.0 (C50) | C50 [30] | ||
| Stochastic gradient boosting (GBM) | gbm [31] | ||
| Neural network | Feed-forward neural network (Nnet) | nnet [32] | - Influenced by the function and structure of biological neural networks and can learn highly complex patterns. - By using hidden layers, they create intermediary representations of data that other models cannot reproduce. - AvNnet fits multiple Nnet models and uses the average of the predictions from each constituent model. |
| Model averaged neural network (AvNnet) | avnnet [33] | ||
| Performance Metric | Equation |
|---|---|
| sensitivity | |
| specificity | |
| precision | |
| F-measure | |
| Macro-averaged F-measure |
| Model | Test Accuracy | Macro-Averaged F-measure |
|---|---|---|
| 2 s epoch | ||
| SVM | 89% | 90% |
| RF | 89% | 89.2% |
| XGB | 87.8% | 88.6% |
| GBM | 86.6% | 87.2% |
| C50 | 84.1% | 84.6% |
| CART | 79.3% | 81.7% |
| Nnet | 75.6% | 74.8% |
| AvNnet | 75.6% | 73.2% |
| 1 s epoch | ||
| SVM | 76.8% | 78.2% |
| RF | 85.4% | 84% |
| XGB | 79.3% | 78.1% |
| GBM | 81.7% | 81.5% |
| C50 | 79.3% | 76.4% |
| CART | 72% | 73.3% |
| Nnet | 73.2% | 72.6% |
| AvNnet | 70.7% | 70% |
| Predicted Behaviour | Performance Metric | |||||||
|---|---|---|---|---|---|---|---|---|
| Observed Behaviour | Chew | Rest | Swim | V. Swim | Sensitivity | Specificity | Precision | F-Measure |
| SVM (2 s epoch test) | ||||||||
| Chew | 13 | 0 | 1 | 0 | 68.4% | 98.4% | 92.9% | 78.8% |
| Rest | 2 | 20 | 2 | 0 | 100% | 93.6% | 83.3% | 90.9% |
| Swim | 4 | 0 | 32 | 0 | 91.4% | 91.5% | 88.9% | 90.1% |
| V. Swim | 0 | 0 | 0 | 8 | 100% | 100% | 100% | 100% |
| SVM (1 s epoch test) | ||||||||
| Chew | 8 | 2 | 4 | 0 | 42.1% | 90.5% | 57.1% | 48.5% |
| Rest | 3 | 19 | 2 | 0 | 90.5% | 91.8% | 79.2% | 84.4% |
| Swim | 8 | 0 | 28 | 0 | 82.4% | 83.3% | 77.8% | 80% |
| V. Swim | 0 | 0 | 0 | 8 | 100% | 100% | 100% | 100% |
| RF (2 s epoch test) | ||||||||
| Chew | 10 | 0 | 4 | 0 | 77% | 94.2% | 71.4% | 74.1% |
| Rest | 1 | 21 | 2 | 0 | 100% | 95.1% | 87.5% | 93.3% |
| Swim | 2 | 0 | 34 | 0 | 85% | 95.2% | 94.4% | 89.5% |
| V. Swim | 0 | 0 | 0 | 8 | 100% | 100% | 100% | 100% |
| RF (1 s epoch test) | ||||||||
| Chew | 7 | 2 | 5 | 0 | 70% | 90.3% | 50% | 58.3% |
| Rest | 1 | 21 | 2 | 0 | 91.3% | 95% | 87.5% | 89.4% |
| Swim | 2 | 0 | 34 | 0 | 83% | 95.1% | 94.4% | 88.3% |
| V. Swim | 0 | 0 | 0 | 8 | 100% | 100% | 100% | 100% |
| XGB (2 s epoch test) | ||||||||
| Chew | 11 | 0 | 3 | 0 | 68.8% | 95.5% | 78.6% | 73.3% |
| Rest | 1 | 21 | 2 | 0 | 100% | 95.1% | 87.5% | 93.3% |
| Swim | 4 | 0 | 32 | 0 | 86.5% | 91.1% | 88.9% | 87.7% |
| V. Swim | 0 | 0 | 0 | 8 | 100% | 100% | 100% | 100% |
| XGB (1 s epoch test) | ||||||||
| Chew | 5 | 1 | 8 | 0 | 55.6% | 87.7% | 35.7% | 43.5% |
| Rest | 0 | 21 | 3 | 0 | 91.3% | 95% | 87.5% | 89.4% |
| Swim | 4 | 1 | 31 | 0 | 73.8% | 87.5% | 86.1% | 79.5% |
| V. Swim | 0 | 0 | 0 | 8 | 100% | 100% | 100% | 100% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kadar, J.P.; Ladds, M.A.; Day, J.; Lyall, B.; Brown, C. Assessment of Machine Learning Models to Identify Port Jackson Shark Behaviours Using Tri-Axial Accelerometers. Sensors 2020, 20, 7096. https://doi.org/10.3390/s20247096
Kadar JP, Ladds MA, Day J, Lyall B, Brown C. Assessment of Machine Learning Models to Identify Port Jackson Shark Behaviours Using Tri-Axial Accelerometers. Sensors. 2020; 20(24):7096. https://doi.org/10.3390/s20247096
Chicago/Turabian StyleKadar, Julianna P., Monique A. Ladds, Joanna Day, Brianne Lyall, and Culum Brown. 2020. "Assessment of Machine Learning Models to Identify Port Jackson Shark Behaviours Using Tri-Axial Accelerometers" Sensors 20, no. 24: 7096. https://doi.org/10.3390/s20247096
APA StyleKadar, J. P., Ladds, M. A., Day, J., Lyall, B., & Brown, C. (2020). Assessment of Machine Learning Models to Identify Port Jackson Shark Behaviours Using Tri-Axial Accelerometers. Sensors, 20(24), 7096. https://doi.org/10.3390/s20247096







