Does Curved Walking Sharpen the Assessment of Gait Disorders? An Instrumented Approach Based on Wearable Inertial Sensors
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedures
2.3. Equipment
2.4. Data Processing
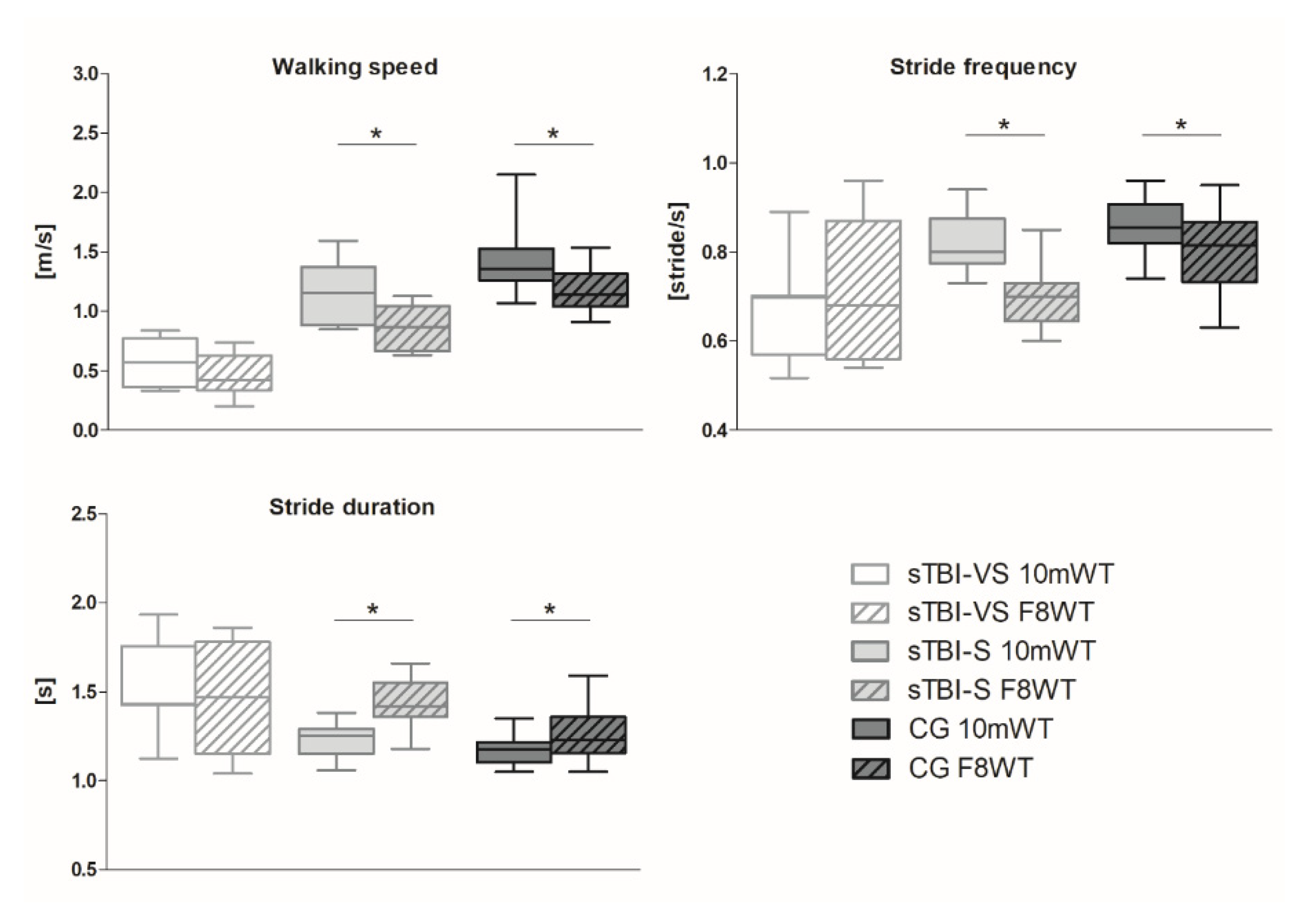
- Average walking speed (WS): total distance/time to complete the test.
- Average stride duration (SD): time to complete the test/total number of strides.
- Stride frequency (SF) total number of strides/time to complete the test.
- Stability:
- (i)
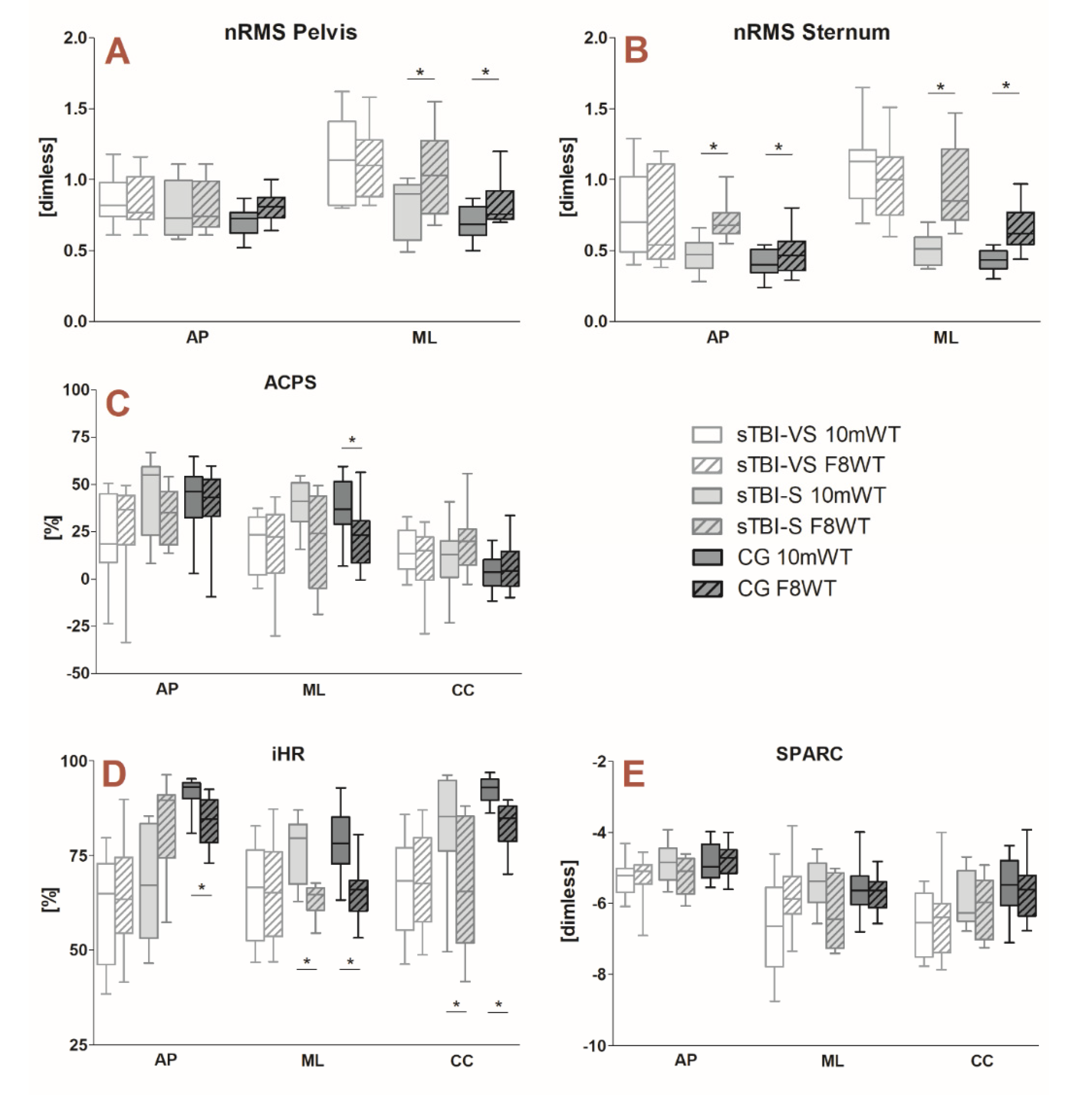
- normalized Root Mean Square (nRMS) values of the measured accelerations at P and S levels, calculated by dividing the RMS obtained for the AP and ML components by the RMS of the CC component. As widely reported in the literature, the greater the nRMS values, the higher the amount of acceleration and, hence, the higher the instability [27];where j is the component (AP and ML), is the upper body level (P and S), N is the number of data sample, and is the acceleration signal;
- (ii)
- attenuation coefficient (AC) [28] between P and S levels, for the acceleration component (j), defined as:A positive coefficient represents an attenuation of the accelerations from lower to upper body levels, whereas a negative coefficient indicates an amplification.
- Symmetry: improved Harmonic Ratio (iHR) calculated for each acceleration component (j) measured at the pelvis level, as proposed by [29]. It was calculated as:where an iHR of 0% is considered as total asymmetry and 100% as perfect symmetry. According to [29], the number of considered harmonics was 20 and the number of considered strides for each participant was greater than 20.
- Smoothness: SPectral ARC length (SPARC) [30] calculated for each acceleration component (j) measured at the P level. It estimates smoothness by calculating the arc length of the Fourier magnitude spectrum of a given signal profile within a defined frequency range. The calculation of SPARC has been performed, as follows:where A () is the Fourier magnitude spectrum of the acceleration signal and () is the normalized magnitude spectrum. is adaptively selected based on a given threshold and is upper bound by . The higher the SPARC value, the smoother the movement.
2.5. Statistical Analysis
3. Results
3.1. Spatiotemporal Parameters
3.2. Gait Quality Indices
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Segal, A.D.; Orendurff, M.S.; Czerniecki, J.M.; Shofer, J.B.; Klute, G.K. Local dynamic stability in turning and straight-line gait. J. Biomech. 2008, 41, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- Courtine, G.; Schieppati, M. Human walking along a curved path I. Body trajectory, segment orientation and the effect of vision. Eur. J. Neurosci. 2003, 18, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Courtine, G.; Schieppati, M. Tuning of a basic coordination pattern constructs straight-ahead and curved walking in humans. J. Neurophysiol. 2004, 91, 1524–1535. [Google Scholar] [CrossRef] [PubMed]
- Hase, K.; Stein, R.B. Turning strategies during human walking. J. Neurophysiol. 1999, 81, 2914–2922. [Google Scholar] [CrossRef] [PubMed]
- Vieilledent, S.; Kerlirzin, Y.; Dalbera, S.; Berthoz, A. Relationship between velocity and curvature of a human locomotor trajectory. Neurosci. Lett. 2001, 305, 65–69. [Google Scholar] [CrossRef]
- Dite, W.; Temple, V.A. Development of a clinical measure of turning for older adults. Am. J. Phys. Med. Rehabil. 2002, 81, 857–866. [Google Scholar] [CrossRef]
- King, L.; Mancini, M.; Salarian, A.; Holmstrom, L.; McNames, J.; Horak, F.B. Mobility lab to assess balance and gait with synchronized body-worn sensors. J. Bioeng. Biomed. Sci. 2013, 1–15. [Google Scholar] [CrossRef]
- Turcato, A.M.; Godi, M.; Giardini, M.; Arcolin, I.; Nardone, A.; Giordano, A.; Schieppati, M. Abnormal gait pattern emerges during curved trajectories in high-functioning Parkinsonian patients walking in line at normal speed. PLoS ONE 2018, 13, 1–26. [Google Scholar] [CrossRef]
- Guglielmetti, S.; Nardone, A.; de Nunzio, A.M.; Godi, M.; Schieppati, M. Walking along circular trajectories in Parkinson’s disease. Mov. Disord. 2009, 24, 598–604. [Google Scholar] [CrossRef]
- Godi, M.; Nardone, A.; Schieppati, M. Curved walking in hemiparetic patients. J. Rehabil. Med. 2010, 42, 858–865. [Google Scholar] [CrossRef]
- Fino, P.C.; Nussbaum, M.A.; Brolinson, P.G. Locomotor deficits in recently concussed athletes and matched controls during single and dual-task turning gait: Preliminary results. J. Neuroeng. Rehabil. 2016, 13, 1–15. [Google Scholar] [CrossRef] [PubMed]
- El-Gohary, M.; Pearson, S.; McNames, J.; Mancini, M.; Horak, F.; Mellone, S.; Chiari, L. Continuous monitoring of turning in patients with movement disability. Sensors 2014, 14, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Tinetti, M.E. Performance-oriented assessment of mobility problems in elderly patients. J. Am. Geriatr. Soc. 1986, 34, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Higashi, Y.; Yamakoshi, K.; Fujimoto, T.; Sekine, M.; Tamura, T. Quantitative evaluation of movement using the timed up-and-go test. IEEE Eng. Med. Biol. Mag. 2008, 27, 38–46. [Google Scholar] [CrossRef]
- Hess, R.J.; Brach, J.S.; Piva, S.R.; vanSwearingen, J.M. Walking skill can be assessed in older adults: Validity of the Figure-of-8 Walk Test. Phys. Ther. 2010, 90, 89–99. [Google Scholar] [CrossRef]
- Wong, S.S.T.; Yam, M.S.; Ng, S.S.M. The Figure-of-Eight Walk test: Reliability and associations with stroke-specific impairments. Disabil. Rehabil. 2013, 35, 1896–1902. [Google Scholar] [CrossRef]
- Song, H.S.; Kim, J.Y. The effects of complex exercise on walking ability during direction change and falls efficacy in the elderly. J. Phys. Ther. Sci. 2015, 27, 1365–1367. [Google Scholar] [CrossRef]
- Belluscio, V.; Bergamini, E.; Tramontano, M.; Bustos, A.O.; Allevi, G.; Formisano, R.; Vannozzi, G.; Buzzi, M.G. Gait quality assessment in survivors from severe traumatic brain injury: An instrumented approach based on inertial sensors. Sensors 2019, 19, 5315. [Google Scholar] [CrossRef]
- Bergamini, E.; Iosa, M.; Belluscio, V.; Morone, G.; Tramontano, M.; Vannozzi, G. Multi-sensor assessment of dynamic balance during gait in patients with subacute stroke. J. Biomech. 2017, 61, 208–215. [Google Scholar] [CrossRef]
- Stuart, S.; Parrington, L.; Martini, D.N.; Kreter, N.; Chesnutt, J.C.; Fino, P.C.; King, L.A. Analysis of free-living mobility in people with mild traumatic brain injury and healthy controls: Quality over quantity. J. Neurotrauma 2020, 37, 139–145. [Google Scholar] [CrossRef]
- Teasdale, G.M. Head injury. J. Neurol. Neurosurg. Psychiatry 1995, 58, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Ciurli, P.; Bivona, U.; Barba, C.; Onder, G.; Silvestro, D.; Azicnuda, E.; Rigon, J.; Formisano, R. Metacognitive unawareness correlates with executive function impairment after severe traumatic brain injury. J. Int. Neuropsychol. Soc. 2010, 16, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical power analysis. Curr. Dir. Psychol. Sci. 1992, 1, 98–101. [Google Scholar] [CrossRef]
- Herman, T.; Inbar-Borovsky, N.; Brozgol, M.; Giladi, N.; Hausdorff, J.M. Dynamic Gait index in healthy older adults: The role of stair climbing, fear of falling and gender. Gait Posture 2010, 29, 237–241. [Google Scholar] [CrossRef]
- Shumway-Cook, A.; Baldwin, M.; Polissar, N.L.; Gruber, W. Predicting the probability for falls in community-dwelling older adults. Phys. Ther. 1997, 77, 812–819. [Google Scholar] [CrossRef]
- Bergamini, E.; Ligorio, G.; Summa, A.; Vannozzi, G.; Cappozzo, A.; Sabatini, A.M. Estimating orientation using magnetic and inertial sensors and different sensor fusion approaches: Accuracy assessment in manual and locomotion tasks. Sensors 2014, 14, 18625–18649. [Google Scholar] [CrossRef]
- Kavanagh, J.J.; Menz, H.B. Accelerometry: A technique for quantifying movement patterns during walking. Gait Posture 2008, 28, 1–15. [Google Scholar] [CrossRef]
- Mazzà, C.; Iosa, M.; Pecoraro, F.; Cappozzo, A. Control of the upper body accelerations in young and elderly women during level walking. J. Neuroeng. Rehabil. 2008, 5, 1–10. [Google Scholar] [CrossRef]
- Pasciuto, I.; Bergamini, E.; Iosa, M.; Vannozzi, G.; Cappozzo, A. Overcoming the limitations of the Harmonic Ratio for the reliable assessment of gait symmetry. J. Biomech. 2017, 53, 84–89. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Melendez-Calderon, A.; Roby-Brami, A.; Burdet, E. On the analysis of movement smoothness. J. Neuroeng. Rehabil. 2015, 12, 1–11. [Google Scholar] [CrossRef]
- Godi, M.; Giardini, M.; Schieppati, M. Walking along curved trajectories. Changes with age and Parkinson’s disease. Hints to rehabilitation. Front. Neurol. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tramontano, M.; Bergamini, E.; Iosa, M.; Belluscio, V.; Vannozzi, G.; Morone, G. Vestibular rehabilitation training in patients with subacute stroke: A preliminary randomized controlled trial. NeuroRehabilitation 2018, 43, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Formisano, R.; Zasler, N.D. Posttraumatic parkinsonism. J. Head Trauma Rehabil. 2014, 29, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Turcato, A.M.; Godi, M.; Giordano, A.; Schieppati, M.; Nardone, A. The generation of centripetal force when walking in a circle: Insight from the distribution of ground reaction forces recorded by plantar insoles. J. Neuroeng. Rehabil. 2015, 12, 1–12. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Melendez-Calderon, A.; Burdet, E. A robust and sensitive metric for quantifying movement smoothness. IEEE Trans. Biomed. Eng. 2012, 59, 2126–2136. [Google Scholar] [CrossRef]

| sTBI-VS | sTBI-S | CG | |
|---|---|---|---|
| Nr. of participants | 11 | 9 | 20 |
| Nr. of males | 7 | 5 | 12 |
| Age (ears) | 32.0 (9.7) | 34.1 (9.1) | 33.6 (10.8) |
| Time since trauma (days) | 504 (456) | 302 (178) | - |
| Body mass (kg) | 72.0 (13.7) | 75.9 (16.2) | 78.3 (14.9) |
| Body height (m) | 1.74 (0.02) | 1.73 (0.04) | 1.78 (0.05) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belluscio, V.; Bergamini, E.; Tramontano, M.; Formisano, R.; Buzzi, M.G.; Vannozzi, G. Does Curved Walking Sharpen the Assessment of Gait Disorders? An Instrumented Approach Based on Wearable Inertial Sensors. Sensors 2020, 20, 5244. https://doi.org/10.3390/s20185244
Belluscio V, Bergamini E, Tramontano M, Formisano R, Buzzi MG, Vannozzi G. Does Curved Walking Sharpen the Assessment of Gait Disorders? An Instrumented Approach Based on Wearable Inertial Sensors. Sensors. 2020; 20(18):5244. https://doi.org/10.3390/s20185244
Chicago/Turabian StyleBelluscio, Valeria, Elena Bergamini, Marco Tramontano, Rita Formisano, Maria Gabriella Buzzi, and Giuseppe Vannozzi. 2020. "Does Curved Walking Sharpen the Assessment of Gait Disorders? An Instrumented Approach Based on Wearable Inertial Sensors" Sensors 20, no. 18: 5244. https://doi.org/10.3390/s20185244
APA StyleBelluscio, V., Bergamini, E., Tramontano, M., Formisano, R., Buzzi, M. G., & Vannozzi, G. (2020). Does Curved Walking Sharpen the Assessment of Gait Disorders? An Instrumented Approach Based on Wearable Inertial Sensors. Sensors, 20(18), 5244. https://doi.org/10.3390/s20185244







