Fast Detection of Olive Trees Affected by Xylella Fastidiosa from UAVs Using Multispectral Imaging
Abstract
1. Introduction
- depends on the photosynthetic activity in the VIS region;
- depends on the structure of plants’ leaves and foliage (size, number of leaf layers, etc.) in the NIR region;
- is strongly influenced by the water content in the SWIR region.
2. Materials and Methods
- RGB images (red–green–blue), i.e., common visible images.
- CIR images (color and infrared = Red + Green + NIR) were obtained by substituting the NIR (near-infrared) images into the blue channel on the common RGB images. NIR wavelengths are effective in penetrating atmospheric mist and in determining the health of vegetation. The pigment in the leaves of plants, chlorophyll, strongly absorbs visible light, but the cellular structure of healthy leaves, on the other hand, strongly reflects NIR radiation. Therefore, the stronger the NIR radiation detected by the camera, the healthier the plant is.
- NDVI images. In normalized-difference vegetation index images, each pixel was calculated from the pixels of the same position in the NIR and RED images through the well-known relationship
- -
- San Vito dei Normanni (BR), 40°38′1.12″ N, 17°42′49.10″ E, time 12:45 UTC, with healthy olive trees with a planting layout of about 10 m × 12 m and an age of about 80 years. Sun elevation and azimuth were 51° and 229°, respectively.
- -
- Squinzano (LE), 40°27′35.79″ N, 18° 7′2.69″ E, time 14:30 UTC, with olive trees with symptoms of Xf, with a planting layout of about 8 m × 8 m and an age of about 50 years. Sun elevation and azimuth were 33° and 255°, respectively.
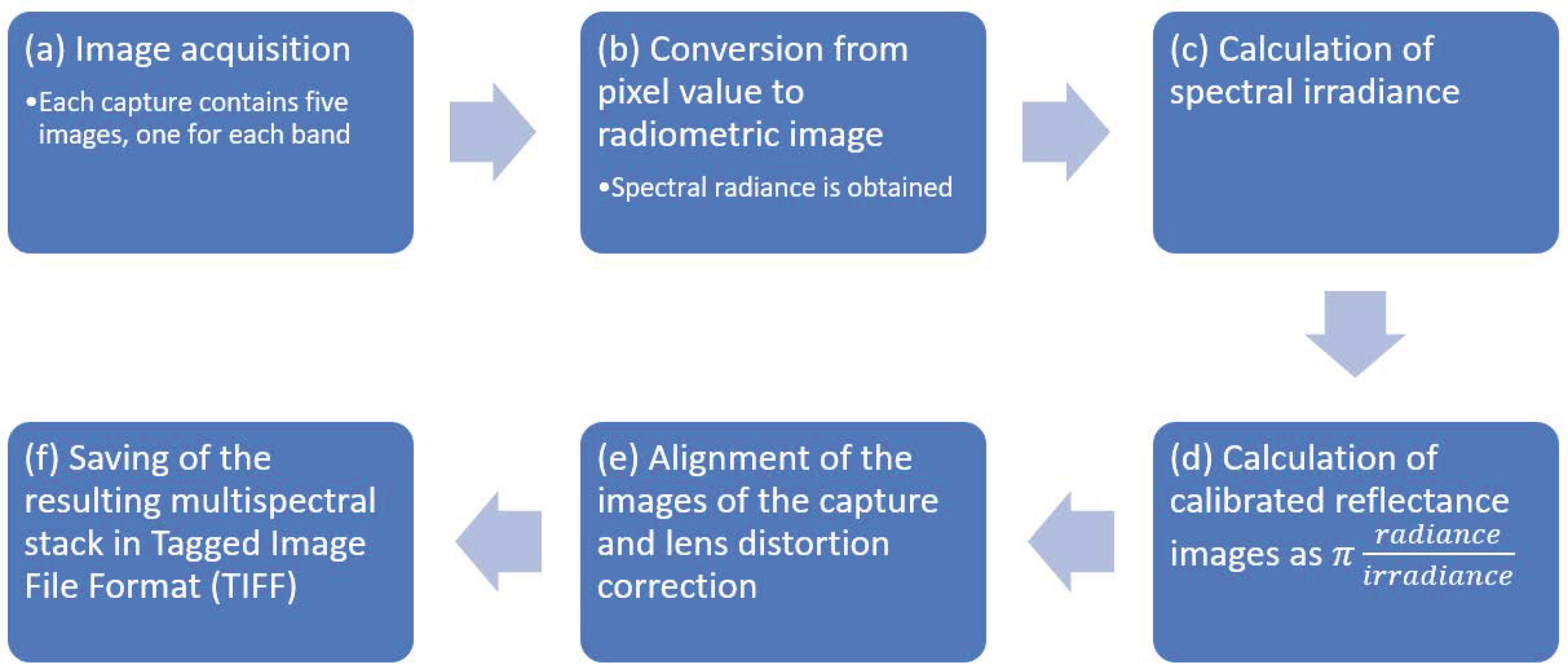
2.1. Image Calibration and Alignment
- Input data were constituted by shots, also named “captures”, where each capture contained five raw digital images in Tagged Image File Format (TIFF), acquired synchronously by the sensors of the five band multispectral camera. One capture was processed at a time.
- The spectral radiance in wavelength, units , was obtained from pixel values in each raw image, taking into account calibration and lens vignette effect parameters provided by the manufacturer, as well as exposure time, black level, and gain of the imaging sensors at the time the images were shot. Spectral radiance images , were obtained, where each is an array.
- Spectral irradiances, , which are the amount of energy per unit area per unit bandwidth () incident on the ground, were calculated from data measured by the downwelling light sensor (DLS) mounted on the drone. Data were acquired by the DLS at the same time as the images were captured by the multispectral camera. When calculating irradiance, the position of the DLS (measured by an onboard sensor) and solar orientation were considered, and clear sky conditions were assumed.
- Spectral reflectance images were obtained from the ratio of reflected and incident light, calculated precisely as
- Reflectance images were corrected for lens distortion using parameters provided by the manufacturer, namely three-element radial distortion and two-element tangential distortion correction parameters. After distortion correction, the five images were aligned by correcting the different points of view of each sensor. For this purpose, the image in the green band was (arbitrarily) selected as a reference, and the other four images were aligned by calculating, for each of them, an eight-parameter homography that maximized the enhanced correlation coefficient (ECC) [71] with the reference. The parameters were obtained using the findTransformECC function of the OpenCV library [72]. Since estimating the parameters of the four homographies was time-consuming, they were calculated for a capture in the middle of the flight and their inverse functions were applied to align the other captures; this procedure was correct because all of them were taken at the same distance from the ground, and at a distance which was greater than the change of depth of the subjects (trees and ground); hence, the images could be aligned in the same way.
- As a result, a properly aligned stack of spectral reflectances was obtained and saved in a five channel TIFF file. Channels in the stack were named . Each channel represented an array of size , for example was the near-infrared reflectance of the pixel in row index and column index , where and .
2.2. 3D Reconstruction
2.3. Tree Segmentation
- (1)
- It contains at least one matched pixel below the elevation threshold. This is justified by the fact that if a segment contains low-elevation points, it is likely it belongs to the ground rather than trees. This condition is expressed as
- (2)
- Its area, i.e., the number of pixels, , divided by whole image area , is larger than the relative threshold . This is justified by the fact that it is unlikely that large segments are part of trees. The relevant condition is
- (3)
- Its mean NIR reflectance is below the threshold . Indeed, a low NIR reflectance can be associated with nonvegetation segments. This is expressed as
- (4)
- Its mean NDVI is below the threshold . Analogously to NIR, a low NDVI can be associated with non-vegetation segments. This is expressed aswhere .
- (1)
- The training set of segments assigned to class , that is , is built with any segment that contains at least one matched pixel below the elevation threshold or that is large—in other words, any segment that satisfies the previously defined condition (3) or the condition (4). For these segments, the condition to satisfy is
- (2)
- The training set of segments of class , for which , is built with any segment that satisfies both of the following conditions:
- It has not been already classified into the training set of class , that is, both conditions (3) and (4) are not satisfied.
- At least one high-elevation matched point of the segment is not on the border of all segments. Here, the border of all segments is defined as the set of any pixel not completely surrounded (considering four-neighborhood connectivity) by pixels of the same segment, further thickened with an additional morphological binary dilation. This condition can be expressed as
- (3)
- Three features are calculated on each segment in the training set: arithmetic mean of NIR values over the pixels of the segment; standard deviation of NIR; arithmetic mean of NDVI. These features are used to train the LDA classifier.
- (4)
- The trained LDA classifier is used to obtain the probability of each segments of class being in class . Reassignment to class is performed if that probability is above the threshold , otherwise the assignment is made to class .
2.4. Classification of Health Status
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Istituto di servizi per il mercato agricolo alimentare (ISMEA). Produzione di olio di Oliva ai Minimi Storici in Italia. Available online: http://www.ismeamercati.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/9224 (accessed on 28 August 2020).
- Elbeaino, T.; Valentini, F.; Abou Kubaa, R.; Moubarak, P.; Yaseen, T.; Digiaro, M. Multilocus sequence typing of Xylella fastidiosa isolated from ulive affected by ‘ulive quick decline syndrome’ in Italy. Phytopathol. Mediterr. 2014, 53, 533–542. [Google Scholar] [CrossRef]
- Haelterman, R.M.; Tolocka, P.A.; Roca, M.E.; Guzmán, F.A.; Fernández, F.D.; Otero, M.L. First presumptive diagnosis of Xylella fastidiosa causing olive scorch in Argentina. Plant Pathol. J. 2015, 97, 393. [Google Scholar] [CrossRef]
- Coletta-Filho, H.D.; Francisco, C.S.; Lopes, J.R.S.; De Oliveira, A.F.; De Oliveira Da Silva, L.F. First report of olive leaf scorch in Brazil associated with Xylella fastidiosa subsp. pauca. Phytopathol. Mediterr. 2016, 55, 130–135. [Google Scholar] [CrossRef]
- Bleve, G.; Marchi, G.; Ranaldi, F.; Gallo, A.; Cimaglia, F.; Logrieco, A.F.; Mita, G.; Ristori, J.; Surico, G. Molecular characteristics of a strain (Salento-1) of Xylella fastidiosa isolated in Apulia (Italy) from an ulive plant with the quick decline syndrome. Phytopathol. Mediterr. 2016, 55, 139–146. [Google Scholar]
- Tolocka, P.A.; Mattio, M.F.; Paccioretti, M.A.; Otero, M.L.; Roca, M.E.; Guzmán, F.A.; Haelterman, R.M. Xylella fastidiosa subsp. pauca ST69 in ulive in Argentina. Plant Pathol. J. 2017, 99, 803. [Google Scholar] [CrossRef]
- Boscia, D.; Altamura, G.; Saponari, M.; Tavano, D.; Zicca, S.; Pollastro, P.; Silletti, M.R.; Savino, V.N.; Martelli, G.P.; Delle Donne, A.; et al. Incidenza di Xylella in uliveti con disseccamento rapido. L’Informatore Agrar. 2017, 27, 47–50. [Google Scholar] [CrossRef]
- Saponari, M.; Boscia, D.; Altamura, G.; Loconsole, G.; Zicca, S.; D’Attoma, G.; Morelli, M.; Palmisano, F.; Saponari, A.; Tavano, D.; et al. Isolation and pathogenicity of Xylella fastidiosa associated to the olive quick decline syndrome in southern Italy. Sci. Rep. 2017, 7, 17723. [Google Scholar] [CrossRef] [PubMed]
- Luvisi, A.; Aprile, A.; Sabella, E.; Vergine, M.; Nicoli, F.; Nutricati, E.; Miceli, A.; Negro, C.; de Bellis, L. Xylella fastidiosa subsp. pauca (CoDiRO strain) infection in four olive (Olea europaea L.) cultivars: Profile of phenolic compounds in leaves and progression of leaf scorch symptoms. Phytopathol. Mediterr. 2017, 56, 259–273. [Google Scholar] [CrossRef]
- Bucci, E.M. Xylella fastidiosa, a new plant pathogen that threatens global farming: Ecology, molecular biology, search for remedies. Biochem. Biophys. Res. Commun. 2018, 502, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Scortichini, M.; Migoni, D.; Angile, F.; Laura, D.E.L.; Girelli, C.R.; Zampella, L.; Mastrobuoni, F.; Fanizzi, F.P. Xylella fastidiosa subsp. pauca on ulive in Salento (Southern Italy): Infected trees have low in planta micronutrient content. Phytopathol. Mediterr. 2019, 58, 39–48. [Google Scholar] [CrossRef]
- EPPO Global Database, Xylella fastidiosa World Distribution Database. Available online: https://gd.eppo.int/taxon/XYLEFA/distribution (accessed on 28 August 2020).
- Giampetruzzi, A.; Saponari, M.; Loconsole, G.; Boscia, D.; Savino, V.; Zicca, S.; Landa, B.B.; Chacón-Díaz, C.; Saldarelli, P.; Almeida, R.P.P. Genome-wide analysis provides evidence on the genetic relatedness of the emergent Xylella fastidiosa genotype in Italy to isolates from Central America. Phytopathology 2017, 107, 816–827. [Google Scholar] [CrossRef] [PubMed]
- Acquasanta, F.; Cavalieri, V.; Cioffi, M.; Stefano, C.; D’Accolti, A.; Dal Maso, E.; Diana, F.; Diana, L.; Di Carolo, M.; Facchinetti, D.; et al. Crescenza DongiovanniTradizione e innovazione nel controllo del Philaenus spumarius Linnaeus, 1758 (Hemiptera Aphrophoridae). Atti Giornate Fitopatol. 2018, 1, 181–190. [Google Scholar]
- Cardinale, M.; Luvisi, A.; Meyer, J.B.; Sabella, E.; De Bellis, L.; Cruz, A.C.; Ampatzidis, Y.; Cherubini, P. Specific Fluorescence in Situ Hybridization (FISH) Test to Highlight Colonization of Xylem Vessels by Xylella fastidiosa in Naturally Infected Olive Trees (Olea europaea L.). Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Cornara, D.; Bosco, D.; Fereres, A. Philaenus spumarius: When an old acquaintance becomes a new threat to European agriculture. J. Pest Sci. 2018, 91, 957–972. [Google Scholar] [CrossRef]
- Cruaud, A.; Gonzalez, A.-A.; Godefroid, M.; Nidelet, S.; Streito, J.-C.; Thuillier, J.-M.; Rossi, J.-P.; Santoni, S.; Rasplus, J.-Y. Using insects to detect, monitor and predict the distribution of Xylella fastidiosa: A case study in Corsica. Sci. Rep. 2018, 8, 15628. [Google Scholar] [CrossRef]
- Sabella, E.; Luvisi, A.; Aprile, A.; Negro, C.; Vergine, M.; Miceli, A.; Nicolì, F.; De Bellis, L. Xylella fastidiosa induces differential expression of lignification related-genes and lignin accumulation in tolerant olive trees cv. Leccino. J. Plant Physiol. 2018, 220, 60–68. [Google Scholar] [CrossRef]
- Scortichini, M.; Jianchi, C.; De Caroli, M.; Dalessandro, G.; Pucci, N.; Modesti, V.; L’aurora, A.; Petriccione, M.; Zampella, L.; Mastrobuoni, F.; et al. A zinc, copper and citric acid biocomplex shows promise for control of Xylella fastidiosa subsp. pauca in olive trees in Apulia region (southern Italy). Phytopathol. Mediterr. 2018, 57. [Google Scholar] [CrossRef]
- Ramazzotti, M.; Cimaglia, F.; Gallo, A.; Ranaldi, F.; Surico, G.; Giovanni, M.; Bleve, G.; Marchi, G. Insights on a founder effect: The case of Xylella fastidiosa in the Salento area of Apulia, Italy. Phytopathol. Mediterr. 2018, 57, 8–25. [Google Scholar] [CrossRef]
- Giampetruzzi, A.; Morelli, M.; Saponari, M.; Loconsole, G.; Chiumenti, M.; Boscia, D.; Savino, V.N.; Martelli, G.P.; Saldarelli, P. Xylella fastidiosa strain CoDiRO and olive: A transcriptome view of the interactions, Conference paper. In Proceedings of the First Joint-Effectome-Resistance Network Meeting: Deciphering Plant-Microbe Interactions for Sustainable Crop Production, Banyuls sur Mer, France, 12–15 September 2017. [Google Scholar]
- Castellano, S.; Di Palma, A.M.; Germinara, G.S.; Lippolis, M. Testing of Net Based Protection Systems against the Vector of Xylella Fastidiosa. In Proceedings of the 11th International AIIA Conference: Biosystems Engineering Addressing the Human Challenges of the 21st Century, Bari, Italy, 5–8 July 2017. [Google Scholar]
- Giampetruzzi, A.; Saponari, M.; Almeida, R.P.P.; Essakhi, S.; Boscia, D.; Loconsole, G.; Saldarelli, P. Complete Genome Sequence of the Olive-Infecting Strain Xylella fastidiosa subsp. pauca De Donno. Genome Announc. 2017, 5. [Google Scholar] [CrossRef]
- Cavalieri, V.; Dongiovanni, C.; Altamura, G.; Tauro, D.; Ciniero, A.; Morelli, M.; Bosco, D.; Saponari, M. Evaluation of olive cultivar effect on the efficiency of the acquisition and transmission of Xylella fastidiosa by Philaenus spumarius. In Proceedings of the 3rd Hemipteran-Plant interactions Symposium (HPIS 2017), Madrid, Spain, 4–8 June 2017. [Google Scholar] [CrossRef]
- De Benedictis, M.; De Caroli, M.; Baccelli, I.; Marchi, G.; Bleve, G.; Gallo, A.; Ranaldi, F.; Falco, V.; Pasquali, V.; Piro, G.; et al. Vessel occlusion in three cultivars of Olea europaeanaturally exposed to Xylella fastidiosain open field. J. Phytopathol. 2017, 165, 589–594. [Google Scholar] [CrossRef]
- Boscia, D.; Altamura, G.; Di Carolo, M.; Dongiovanni, C.; Fumarola, G.; Giampetruzzi, A.; Greco, P.; La Notte, P.; Loconsole, G.; Manni, F.; et al. Resistenza a Xylella fastidiosa in olivo: Stato dell’arte e prospettive. Inf. Agrar. 2017, 11, 59–63. [Google Scholar]
- Strona, G.; Carstens, C.J.; Beck, P.S.A. Network analysis reveals why Xylella fastidiosa will persist in Europe. Nat. Sci. Rep. 2017, 7, 71. [Google Scholar] [CrossRef] [PubMed]
- White, S.; Bullock, J.M.; Hooftman, D.A.P.; Chapman, D.S. Modelling the spread and control of Xylella fastidiosa in the early stages of invasion in Apulia, Italy. Biol. Invasions 2017, 19, 1825–1837. [Google Scholar] [CrossRef] [PubMed]
- Landa, B.B.; Montes Borrego, M.; Navas Cortés, J.A. Situación actual de Xylella fastidiosa en Europa y riesgo para la sostenibilidad del olivar. In Proceedings of the I Congreso Ibérico de Olivicultura, Badajoz, Spain, 13–15 April 2016. [Google Scholar]
- The Italian Society of Plant Pathology. In Proceedings of the XXII Congress of Italian Society of Plant Pathology, Rome, Italy, 19–22 September 2016.
- Farias, P.R.; Roberto, S.R.; Lopes, J.R.S.; Perecin, D. Geostatistical characterization of the spatial distribution of Xylella fastidiosa sharpshooter vectors on citrus. Neotrop. Entomol. 2004, 33, 13–20. [Google Scholar] [CrossRef]
- Andria, G.; Cavone, G.; Lanzolla, A.M. Modelling study for assessment and forecasting variation of urban air pollution. Measurement 2008, 41, 222–229. [Google Scholar] [CrossRef]
- Karcher, S.J. Polymerase Chain Reaction, in Molecular Biology—A Project Approach; Academic Press: Cambridge, MA, USA, 1995; pp. 215–227. [Google Scholar]
- Delong, R.K. Experiment 6—Polymerase Chain Reaction (PCR), in Book Introductory Experiments on Biomolecules and Their Interactions; Academic Press: Cambridge, MA, USA, 2015; pp. 59–66. ISBN 978-0-12-800969-7. [Google Scholar] [CrossRef]
- Engvall, E.; Perlmann, P. Enzyme-linked immunosorbent assay (ELISA). In Protides of the Biological Fluids; Academic Press: Cambridge, MA, USA, 1971; pp. 553–556. [Google Scholar]
- Lequin, R.M. Enzyme Immunoassay (EIA)/Enzyme-Linked Immunosorbent Assay (ELISA). Clin. Chem. 2005, 51, 2415–2418. [Google Scholar] [CrossRef]
- Cataldo, A.; Tarricone, L.; Attivissimo, F.; Trotta, A. Simultaneous measurement of dielectric properties and levels of liquids using a TDR method. Measurement 2008, 41, 307–319. [Google Scholar] [CrossRef]
- Giaquinto, N.; Cataldo, A.; D’Aucelli, G.M.; De Benedetto, E.; Cannazza, G. Water Detection Using Bi-Wires as Sensing Elements: Comparison Between Capacimetry-Based and Time-of-Flight-Based Techniques. IEEE Sens. J. 2016, 16, 4309–4317. [Google Scholar] [CrossRef]
- Giaquinto, N.; D’Aucelli, G.M.; De Benedetto, E.; Cannazza, G.; Cataldo, A.; Piuzzi, E.; Masciullo, A. Criteria for Automated Estimation of Time of Flight in TDR Analysis. IEEE Trans. Instrum. Meas. 2016, 65, 1215–1224. [Google Scholar] [CrossRef]
- Attivissimo, F.; Carducci, C.G.C.; Lanzolla, A.M.L.; Spadavecchia, M. An Extensive Unified Thermo-Electric Module Characterization Method. Sensors 2016, 16, 2114. [Google Scholar] [CrossRef]
- Casagli, N.; Frodella, W.; Morelli, S.; Tofani, V.; Ciampalini, A.; Intrieri, E.; Raspini, F.; Rossi, G.; Tanteri, L.; Lu, P. Spaceborne, UAV and ground-based remote sensing techniques for landslide mapping, monitoring and early warning. Geoenvironmental Disasters 2017, 4, 136. [Google Scholar] [CrossRef]
- White, J.C.; Coops, N.C.; Wulder, M.A.; Vastaranta, M.; Hilker, T.; Tompalski, P. Remote Sensing Technologies for Enhancing Forest Inventories: A Review. Can. J. Remote Sens. 2016, 42, 619–641. [Google Scholar] [CrossRef]
- Adão, T.; Hruška, J.; Pádua, L.; Bessa, J.; Peres, E.; Morais, R.; Sousa, J.J. Hyperspectral Imaging: A Review on UAV-Based Sensors, Data Processing and Applications for Agriculture and Forestry. Remote Sens. 2017, 9, 1110. [Google Scholar] [CrossRef]
- Turner, I.; Harley, M.D.; Drummond, C.D. UAVs for coastal surveying. Coast. Eng. 2016, 114, 19–24. [Google Scholar] [CrossRef]
- Nagesh Kumar, D.; Reshmidevi, T.V. Review Remote Sensing Applications in Water Resources. J. Indian Inst. Sci. 2013, 93, 163–187. [Google Scholar]
- Adamo, F.; Andria, G.; Di Nisio, A.; Carducci, C.G.C.; Lay-Ekuakille, A.; Mattencini, G.; Spadavecchia, M. Designing and prototyping a sensors head for test and certification of UAV components. Int. J. Smart Sens. Intell. Syst. 2017, 10, 646–672. [Google Scholar] [CrossRef]
- Petritoli, E.; Leccese, F.; Ciani, L. Reliability and Maintenance Analysis of Unmanned Aerial Vehicles. Sensors 2018, 18, 3171. [Google Scholar] [CrossRef]
- Ruffa, F.; De Capua, C.; Morello, R.; Liu, Z. Temperature Sensing and Evaluation of Thermal Effects on Battery Packs for Automotive Applications. IEEE Sens. J. 2019, 19, 11634–11645. [Google Scholar] [CrossRef]
- Andria, G.; Attivissimo, F.; Lanzolla, A. Interpolation techniques for non-stationary signal analysis. Measurement 1997, 21, 1–16. [Google Scholar] [CrossRef]
- D’Aucelli, G.M.; Giaquinto, N.; Carducci, C.G.C.; Spadavecchia, M.; Trotta, A. Uncertainty evaluation of the Unified Method for thermo-electric module characterization. Measurement 2019, 131, 751–763. [Google Scholar] [CrossRef]
- Zini, E.; Briotti, V.; Morra di Cella, U.; Menin, A.; Marletto, V. Rapporto Earth Observation: Analisi Delle Attività Osservabili da Satelliti e a Mezzo di APR. 2016. Available online: http://www.assoarpa.it/wp-content/uploads/2019/01/Rapporto-GdL-E1c-Ver-1.2.pdf (accessed on 28 August 2020).
- Radoglou-Grammatikis, P.; Sarigiannidis, P.; Lagkas, T.; Moscholios, I. A compilation of UAV applications for precision agriculture. Comput. Netw. 2020, 172, 107148. [Google Scholar] [CrossRef]
- Dash, J.P.; Pearse, G.D.; Watt, M.S. UAV Multispectral Imagery Can Complement Satellite Data for Monitoring Forest Health. Remote Sens. 2018, 10, 1216. [Google Scholar] [CrossRef]
- Shin, P.; Sankey, T.T.; Moore, M.; Thode, A.E. Evaluating Unmanned Aerial Vehicle Images for Estimating Forest Canopy Fuels in a Ponderosa Pine Stand. Remote Sens. 2018, 10, 1266. [Google Scholar] [CrossRef]
- Deng, L.; Mao, Z.; Li, X.; Hu, Z.; Duan, F.; Yan, Y. UAV-based multispectral remote sensing for precision agriculture: A comparison between different cameras. ISPRS J. Photogramm. Remote Sens. 2018, 146, 124–136. [Google Scholar] [CrossRef]
- Rey, B.; Aleixos, N.; Cubero, S.; Blasco, J. Xf-Rovim. A Field Robot to Detect Olive Trees Infected by Xylella fastidiosa Using Proximal Sensing. Remote Sens. 2019, 11, 221. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Camino, C.; Beck, P.S.A.; Calderon, R.; Hornero, A.; Hernández-Clemente, R.; Kattenborn, T.; Montes-Borrego, M.; Susca, L.; Morelli, M.; et al. Previsual symptoms of Xylella fastidiosa infection revealed in spectral plant-trait alterations. Nat. Plants 2018, 4, 432–439. [Google Scholar] [CrossRef]
- Hornero, A.; Hernández-Clemente, R.; North, P.; Beck, P.; Boscia, D.; Navas-Cortes, J.; Zarco-Tejada, P. Monitoring the incidence of Xylella fastidiosa infection in olive orchards using ground-based evaluations, airborne imaging spectroscopy and Sentinel-2 time series through 3-D radiative transfer modelling. Remote Sens. Environ. 2020, 236, 111480. [Google Scholar] [CrossRef]
- Hornero, A.; Hernández-Clemente, R.; Beck, P.S.; Navas-Cortés, J.A.; Zarco-Tejada, P.J. Using Sentinel-2 Imagery to Track Changes Produced by Xylella fastidiosa in Olive Trees; IEEE: Piscataway Township, NJ, USA, 2018; pp. 9060–9062. [Google Scholar]
- Distretto Tecnologico Pugliese (DTA). Remote Early Detection of Xylella (REDOX). Available online: https://www.dtascarl.org/en/progettidta/redox/ (accessed on 28 August 2020).
- MicaSense Inc. MicaSense RedEdge-M Multispectral Camera User Manual. Rev. 01, November 2017. Available online: https://support.micasense.com/hc/en-us/articles/215261448-RedEdge-User-Manual-PDF-Download (accessed on 28 August 2020).
- FLIR Systems Inc. FLIR Vue Pro Web Product Page. Available online: https://www.flir.com/products/vue-pro/ (accessed on 28 August 2020).
- FLIR Systems Inc. FLIR Vue Pro and Vue Pro R User Guide, Document Number: 436-0013-10—Version 110. Available online: https://flir.netx.net/file/asset/11422/original (accessed on 28 August 2020).
- Sixfab GmbH. Raspberry Pi 3G-4G/LTE Base Shield V2. Available online: https://sixfab.com/product/raspberry-pi-3g-4glte-base-shield-v2, (accessed on 28 August 2020).
- Sixfab GmbH. Quectel EC25 Mini PCle 4G/LTE Module. Available online: https://sixfab.com/product/quectel-ec25-mini-pcle-4glte-module/ (accessed on 28 August 2020).
- Sixfab GmbH. LTE Main & Diversity & GNSS Triple Port u.FL Antenna—100mm. Available online: https://sixfab.com/product/lte-main-diversity-gnss-triple-port-u-fl-antenna-100mm/ (accessed on 28 August 2020).
- Xue, J.; Su, B. Significant Remote Sensing Vegetation Indices: A Review of Developments and Applications. J. Sens. 2017, 2017, 1353691. [Google Scholar] [CrossRef]
- Antognelli, S. NDVI and NDMI Vegetation Indices: Instructions for Use. Available online: https://www.agricolus.com/en/indici-vegetazione-ndvi-ndmi-istruzioni-luso/ (accessed on 28 August 2020).
- Regione Puglia. Map of the Xylella fastidiosa Diffusion on Apulia. Available online: http://webapps.sit.puglia.it/freewebapps/MonitoraggioXFSintesi/ (accessed on 28 August 2020).
- MicaSense Knowledge Base. RedEdge Camera Radiometric Calibration Model. Available online: https://support.micasense.com/hc/en-us/articles/115000351194-RedEdge-Camera-Radiometric-Calibration-Model (accessed on 28 August 2020).
- Evangelidis, G.; Psarakis, E. Parametric Image Alignment Using Enhanced Correlation Coefficient Maximization. IEEE Trans. Pattern Anal. Mach. Intell. 2008, 30, 1858–1865. [Google Scholar] [CrossRef]
- Bradski, G. The OpenCV Library. Dr. Dobb’s J. Softw. Tools. Available online: https://www.drdobbs.com/open-source/the-opencv-library/1844043192000 (accessed on 28 August 2020).
- Petritoli, E.; Leccese, F.; Leccisi, M. Inertial Navigation Systems for UAV: Uncertainty and Error Measurements. In Proceedings of the 2019 IEEE 5th International Workshop on Metrology for AeroSpace (MetroAeroSpace), Torino, Italy, 19–21 June 2019; pp. 1–5. [Google Scholar] [CrossRef]
- Wu, C. Towards linear-time incremental structure from motion. In Proceedings of the 2013 International Conference on 3D Vision, 3DV 2013, Seattle, WA, USA, 29 June–1 July 2013; pp. 127–134. [Google Scholar] [CrossRef]
- Felzenszwalb, P.F.; Huttenlocher, D.P. Efficient Graph-Based Image Segmentation. Int. J. Comput. Vis. 2004, 59, 167–181. [Google Scholar] [CrossRef]
- Van Der Walt, S.J.; Schonberger, J.L.; Nunez-Iglesias, J.; Boulogne, F.; Warner, J.; Yager, N.; Gouillart, E.; Yu, T. scikit-image: Image processing in Python. PeerJ 2014, 2, e453. [Google Scholar] [CrossRef] [PubMed]
- Izenman, A.J. Linear Discriminant Analysis, in Book Modern Multivariate Statistical Techniques; Springer: New York, NY, USA, 2013; pp. 237–280. ISBN 978-0-387-78189-1. [Google Scholar] [CrossRef]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Wu, X.; Shen, X.; Cao, L.; Wang, G.; Cao, F. Assessment of Individual Tree Detection and Canopy Cover Estimation using Unmanned Aerial Vehicle based Light Detection and Ranging (UAV-LiDAR) Data in Planted Forests. Remote Sens. 2019, 11, 908. [Google Scholar] [CrossRef]
- Åkerblom, M.; Raumonen, P.; Mãkipãã, R.; Kaasalainen, M. Automatic tree species recognition with quantitative structure models. Remote Sens. Environ. 2017, 191, 1–12. [Google Scholar] [CrossRef]
- Kuzmin, A. Automatic Segment-Level Tree Species Recognition Using High Resolution Aerial Winter Imagery. Eur. J. Remote Sens. 2016, 49, 239–259. [Google Scholar] [CrossRef]











| Sensor | Central Wavelength (nm) | Filter Bandwidth (FWHM) (nm) |
|---|---|---|
| Blue | 475 | 20 |
| Green | 560 | 20 |
| Red | 668 | 10 |
| Near-IR | 840 | 40 |
| Red-Edge | 717 | 10 |
| Symbol | Description |
|---|---|
| Number of pixel rows and pixel columns in each band of the multispectral reflectance stack | |
| Row and column indexes of a pixel in a band, and . | |
| Reflectances corresponding to pixel index of the five bands. | |
| Number of pixels in a given image that match with other images for 3D reconstruction with photogrammetry | |
| Set of matched pixels | |
| Pixel indexes of matched pixels, | |
| 3D coordinates corresponding to matched pixels, | |
| Standardized difference between and interpolating plane | |
| Standardized elevation threshold | |
| Parameters for Felsenszwalb’s oversegmentation, respectively: scale; standard deviation of Gaussian kernel for preprocessing of image; minimum component size. | |
| Number of Felsenszwalb’s segments | |
| Set of pixels indexes ( in the Felsenszwalb’s segment, . It is used to indicate that segment. | |
| Number of pixels in | |
| Results of the first, second, and final segment-classification method, respectively | |
| Classes defined for the segment-classification methods:(“not part of a tree”), (“part of a tree”) and (“unknown”) | |
| Relative area threshold for the first segment-classification method | |
| Mean NIR reflectance threshold for the first segment-classification method | |
| Mean NDVI reflectance threshold for the first segment-classification method | |
| Probability of a segment of being in class calculated by the LDA classifier of the second segment-classification method | |
| Probability threshold of for putting in class | |
| Classes defined for health status classification of trees: (“negative”) when they are in good health status, and (“positive”) for bad health status | |
| Classes defined for pixels of negative and positive trees, respectively | |
| Probability that a pixel of coordinates belongs to the class of pixels of positive trees | |
| Radius in pixels of the disk for morphological binary erosion of segmented trees | |
| Labeled image of connected components of segmented trees | |
| Set of pixels of a given component in , representing a segmented tree | |
| Number of pixels in | |
| Set of probabilities associated with pixels in , relevant to a given tree segment | |
| Sequence of probabilities calculated by the LDA classifier for pixels in a given set , sorted from higher to lower probability | |
| Number of highest-probability pixels used for tree classification | |
| Mean probability value over pixels for a given set | |
| Probability threshold for classifying a segmented tree into |
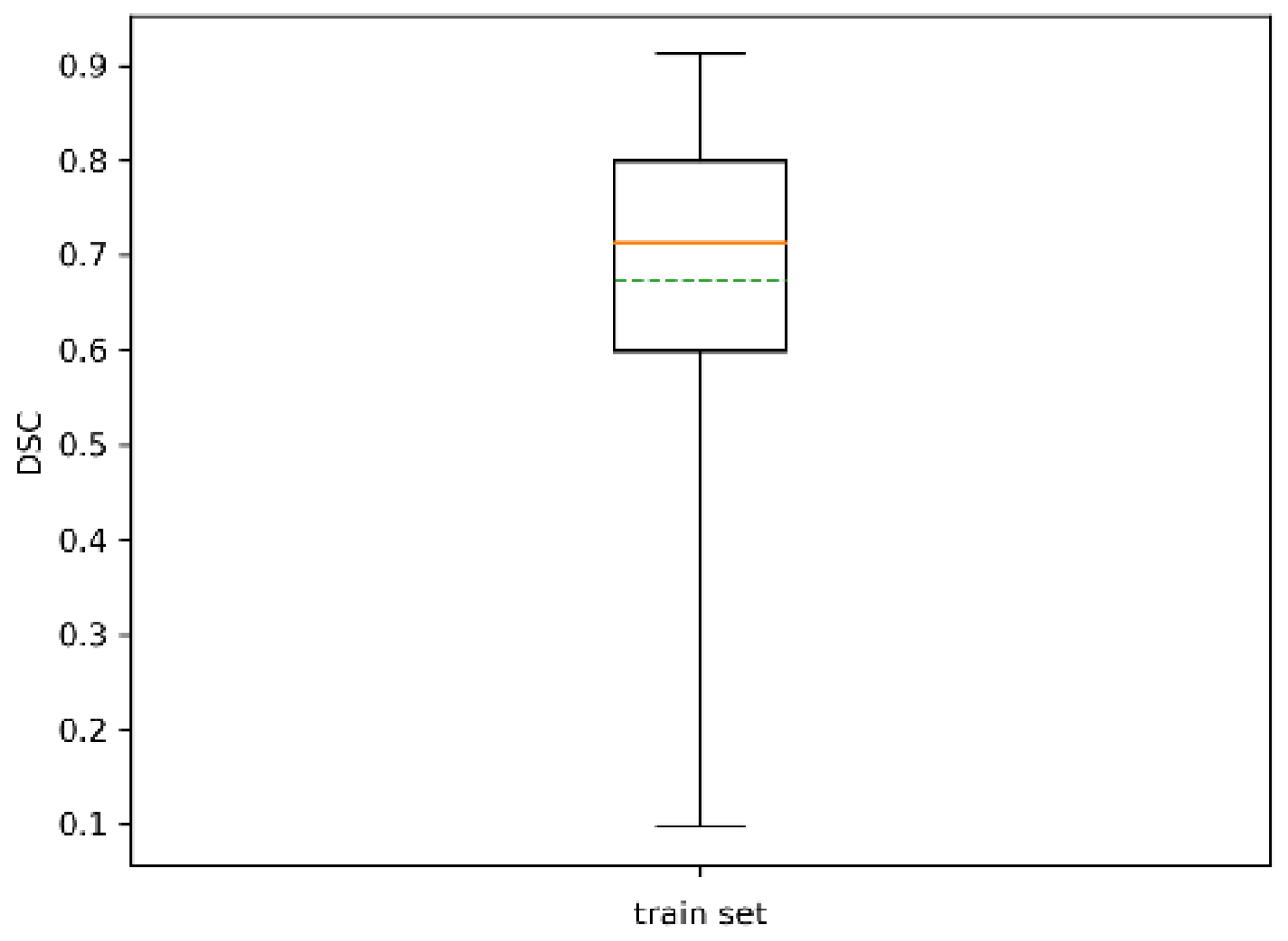
| # of Trees | 71 |
|---|---|
| Mean | 0.68 |
| Std | 0.16 |
| # of trees with | 10 |
| # of trees with | 1 |
| # of trees with | 0 |
| Min | 0.12 |
| Ground Truth | Predicted Negative | Predicted Positive |
|---|---|---|
| Negative | 15 | 4 |
| Positive | 0 | 52 |
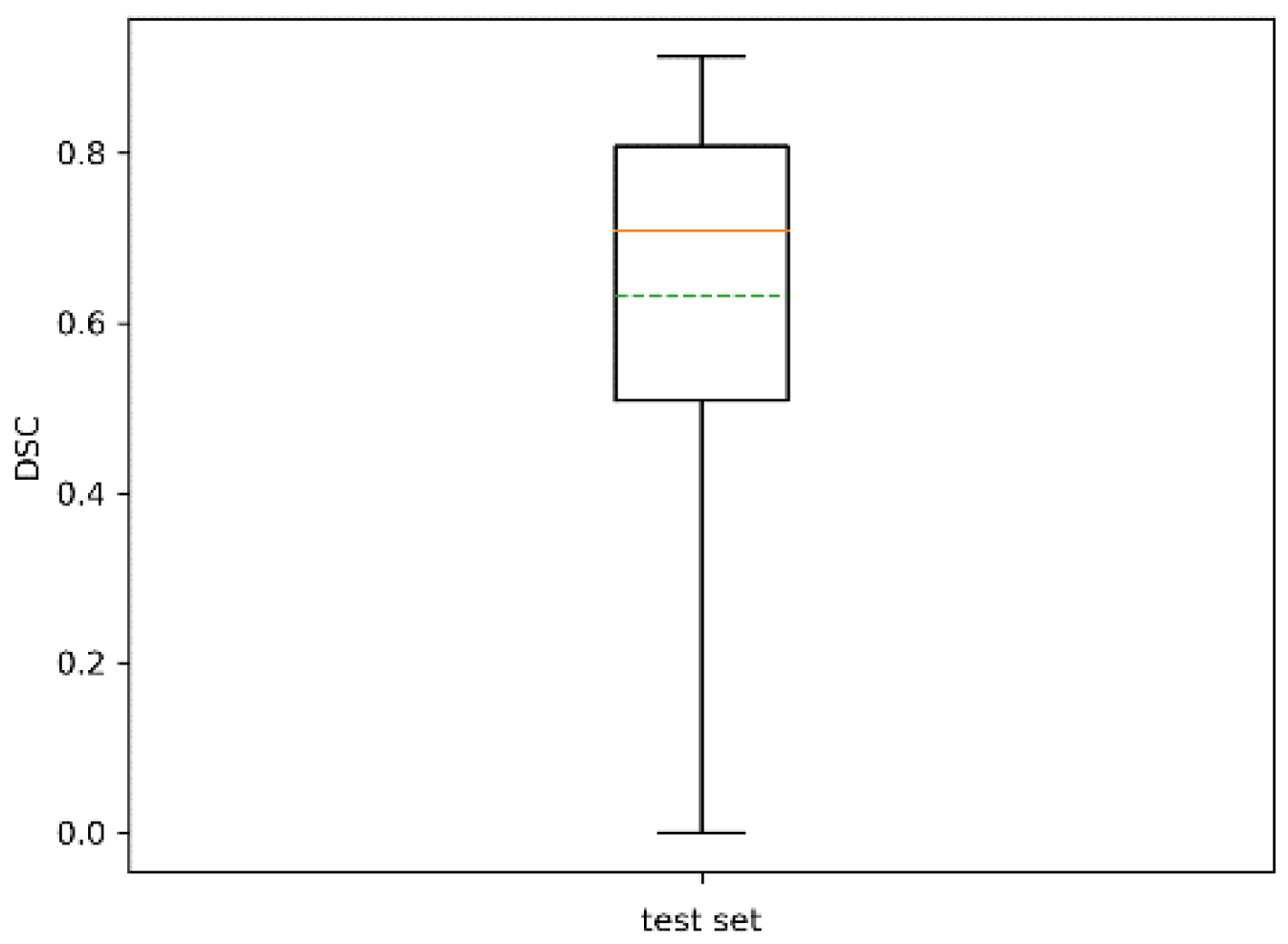
| # of Trees | 71 |
|---|---|
| Mean | 0.66 |
| Std | 0.21 |
| # of trees with | 16 |
| # of trees with | 4 |
| # of trees with | 0 |
| Min | 0.02 |
| Ground Truth | Predicted Negative | Predicted Positive |
|---|---|---|
| Negative | 18 | 0 |
| Positive | 1 | 52 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Nisio, A.; Adamo, F.; Acciani, G.; Attivissimo, F. Fast Detection of Olive Trees Affected by Xylella Fastidiosa from UAVs Using Multispectral Imaging. Sensors 2020, 20, 4915. https://doi.org/10.3390/s20174915
Di Nisio A, Adamo F, Acciani G, Attivissimo F. Fast Detection of Olive Trees Affected by Xylella Fastidiosa from UAVs Using Multispectral Imaging. Sensors. 2020; 20(17):4915. https://doi.org/10.3390/s20174915
Chicago/Turabian StyleDi Nisio, Attilio, Francesco Adamo, Giuseppe Acciani, and Filippo Attivissimo. 2020. "Fast Detection of Olive Trees Affected by Xylella Fastidiosa from UAVs Using Multispectral Imaging" Sensors 20, no. 17: 4915. https://doi.org/10.3390/s20174915
APA StyleDi Nisio, A., Adamo, F., Acciani, G., & Attivissimo, F. (2020). Fast Detection of Olive Trees Affected by Xylella Fastidiosa from UAVs Using Multispectral Imaging. Sensors, 20(17), 4915. https://doi.org/10.3390/s20174915







