Kinetic Exclusion Assay of Biomolecules by Aptamer Capture
Abstract
1. Introduction
2. Materials and Methods
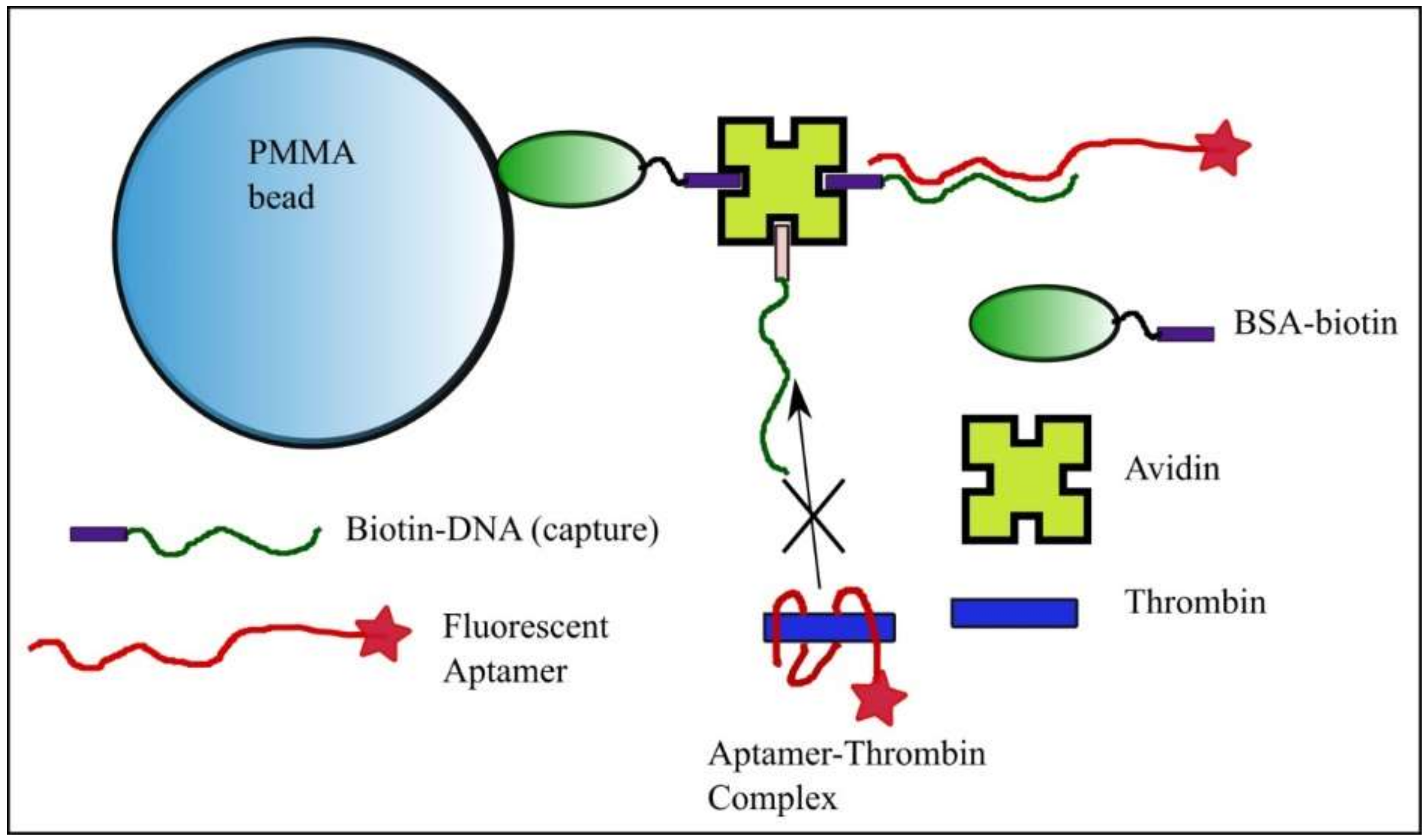
2.1. Solid Phase and Sample Preparation, and Data Collection with the KinExA 3200 Instrument
2.2. Data Analysis
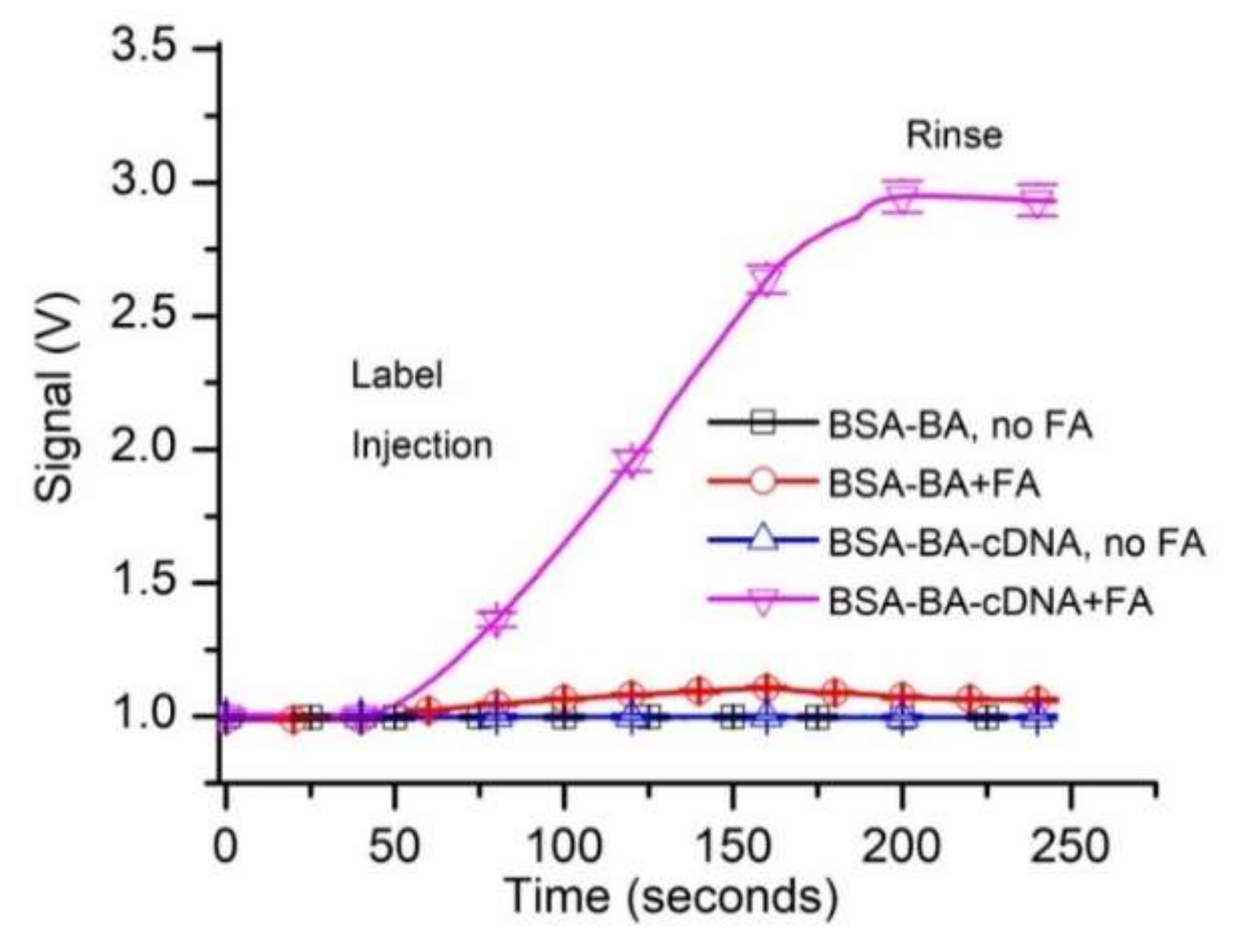
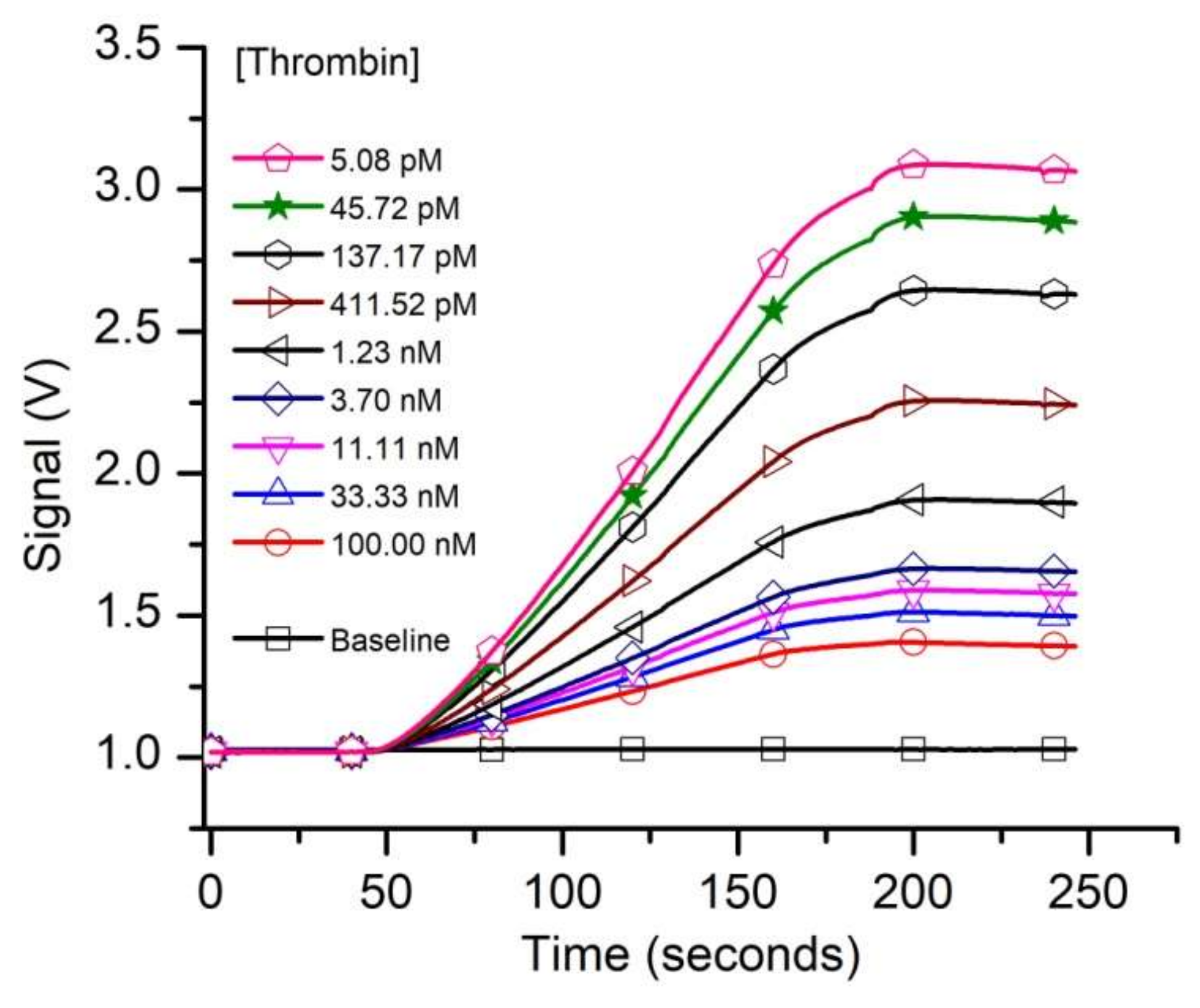
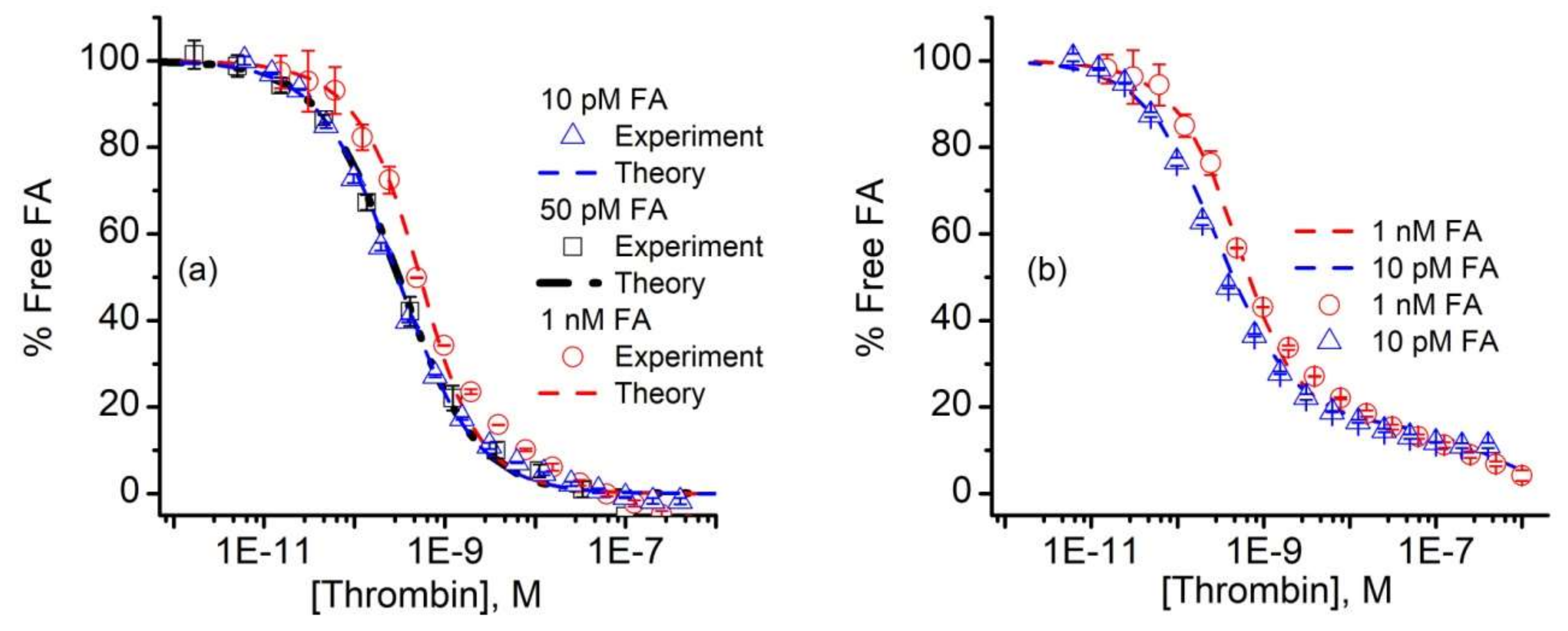
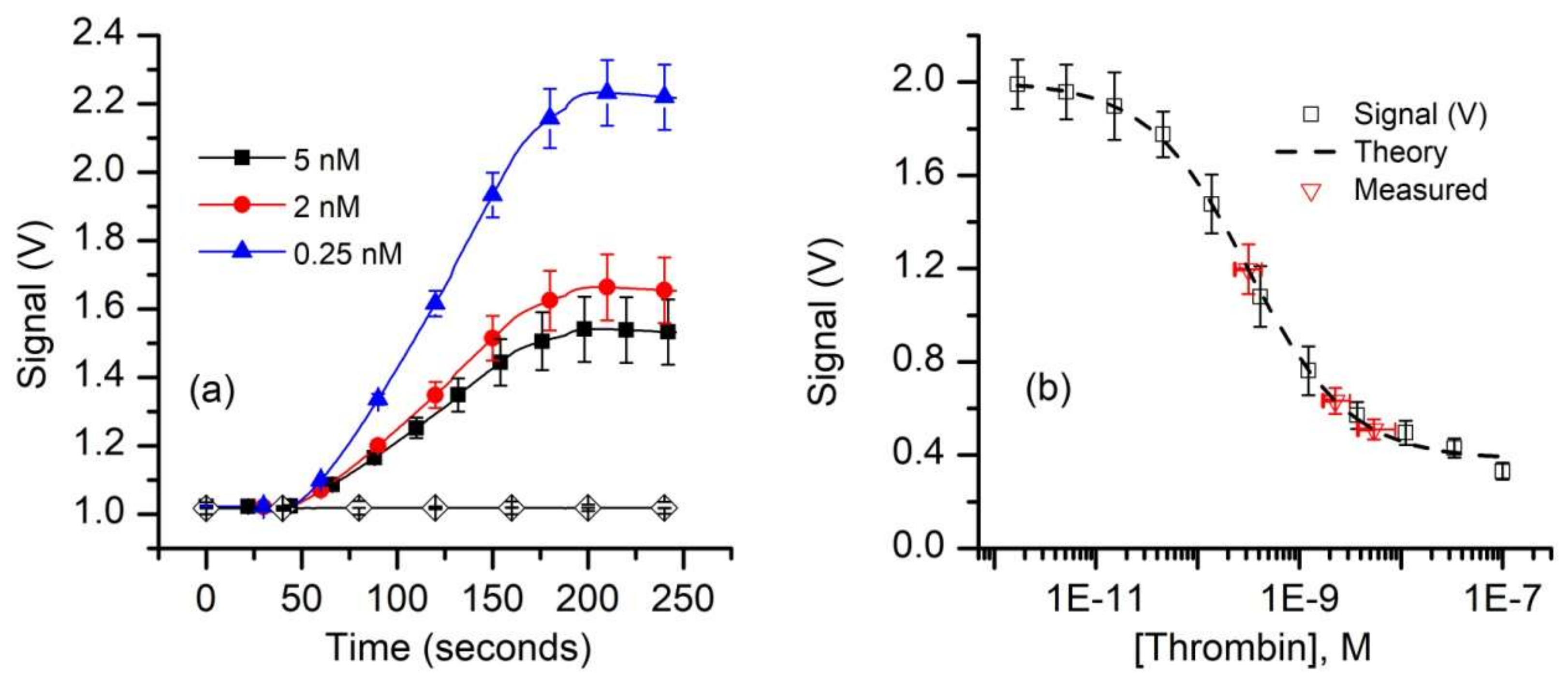
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Kinetics Exclusion Assay Principles
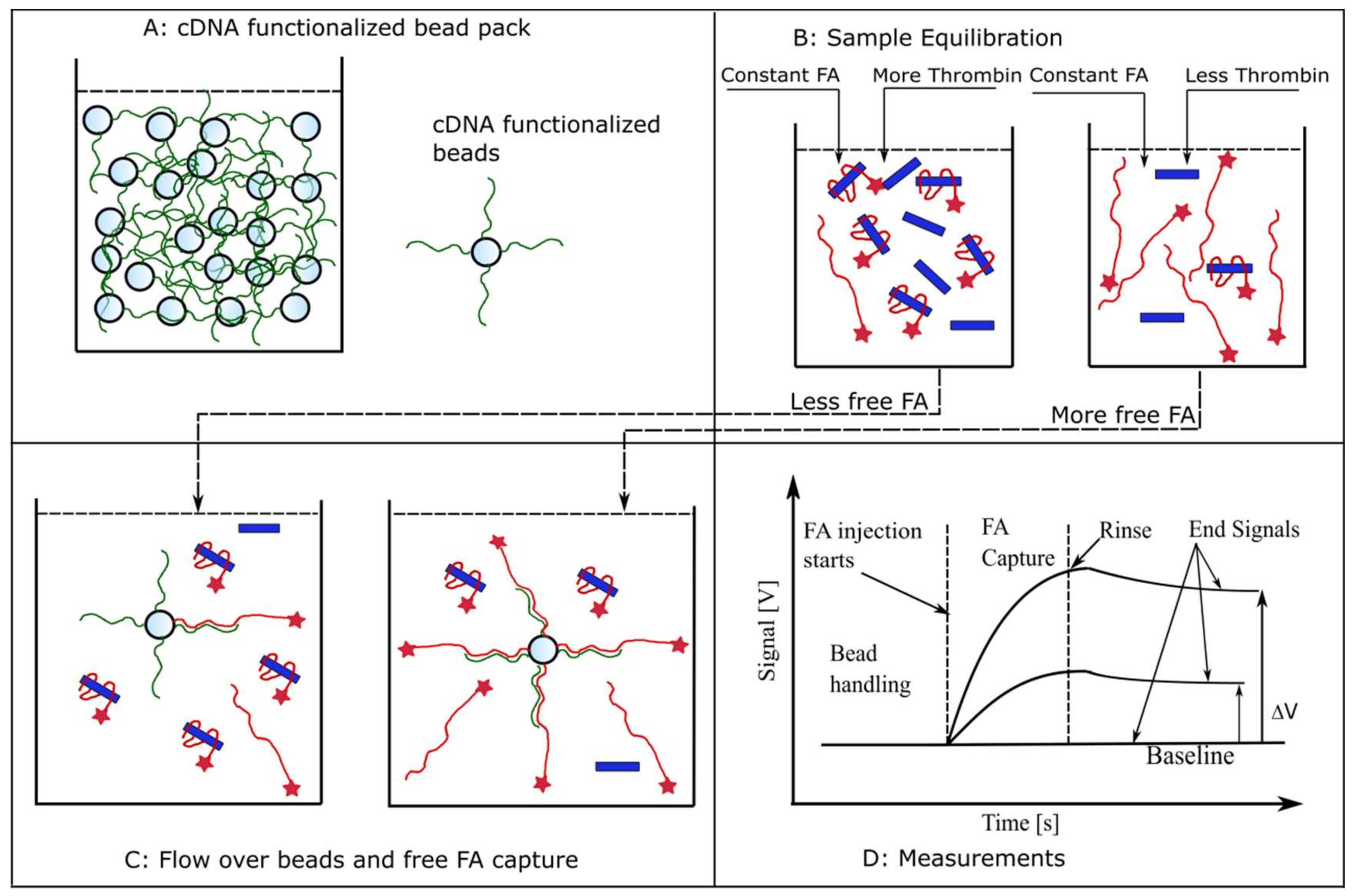
Signal Equations and Modeling
References
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Ellington, A.D.; Szostak, J.W. Selection in vitro of single-stranded DNA molecules that fold into specific ligand-binding structures. Nature 1992, 355, 850–852. [Google Scholar] [CrossRef] [PubMed]
- Stoltenburg, R.; Reinemann, C.; Strehlitz, B. SELEX—A (r)evolutionary method to generate high-affinity nucleic acid ligands. Biomol. Eng. 2007, 24, 381–403. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Dong, S.; Wang, E. Label-free colorimetric detection of aqueous mercury ion (Hg2+) using Hg2+-modulated G-quadruplex-based DNAzymes. Anal. Chem. 2009, 81, 2144–2149. [Google Scholar] [CrossRef]
- Long, F.; Zhu, A.; Shi, H.; Wang, H.; Liu, J. Rapid on-site/in-situ detection of heavy metal ions in environmental water using a structure-switching DNA optical biosensor. Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Chung, C.H.; Kim, J.H.; Jung, J.; Chung, B.H. Nuclease-resistant DNA aptamer on gold nanoparticles for the simultaneous detection of Pb2+ and Hg2+ in human serum. Biosens. Bioelectron. 2013, 41, 827–832. [Google Scholar] [CrossRef]
- Qu, H.; Csordas, A.T.; Wang, J.; Oh, S.S.; Eisenstein, M.S.; Soh, H.T. Rapid and label-free strategy to isolate aptamers for metal ions. ACS Nano 2016, 10, 7558–7565. [Google Scholar] [CrossRef]
- Pfeiffer, F.; Mayer, G. Selection and biosensor application of aptamers for small molecules. Front. Chem. 2016, 4, 1–21. [Google Scholar] [CrossRef]
- Stojanovic, M.N.; de Prada, P.; Landry, D.W. Aptamer-based folding fluorescent sensor for cocaine. J. Am. Chem. Soc. 2001, 123, 4928–4931. [Google Scholar] [CrossRef]
- Robati, R.Y.; Arab, A.; Ramezani, M.; Langroodi, F.A.; Abnous, K.; Taghdisi, S.M. Aptasensors for quantitative detection of kanamycin. Biosens. Bioelectron. 2016, 82, 162–172. [Google Scholar] [CrossRef] [PubMed]
- De-los-Santos-Álvarez, N.; Lobo-Castañón, M.J.; Miranda-Ordieres, A.J.; Tuñón-Blanco, P. Modified-RNA aptamer-based sensor for competitive impedimetric assay of neomycin B. J. Am. Chem. Soc. 2007, 129, 3808–3809. [Google Scholar] [CrossRef]
- Sun, D.; Lu, J.; Zhang, L.; Chen, Z. Aptamer-based electrochemical cytosensors for tumor cell detection in cancer diagnosis: A review. Anal. Chim. Acta 2019, 1082, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Herr, J.K.; Smith, J.E.; Medley, C.D.; Shangguan, D.; Tan, W. Aptamer-conjugated nanoparticles for selective collection and detection of cancer cells. Anal. Chem. 2006, 78, 2918–2924. [Google Scholar] [CrossRef] [PubMed]
- Shangguan, D.; Li, Y.; Tang, Z.; Cao, Z.C.; Chen, H.W.; Mallikaratchy, P.; Sefah, K.; Yang, C.J.; Tan, W. Aptamers evolved from live cells as effective molecular probes for cancer study. Proc. Natl. Acad. Sci. USA 2006, 103, 11838–11843. [Google Scholar] [CrossRef]
- Zhu, G.; Chen, X. Aptamer-based targeted therapy. Adv. Drug Deliv. Rev. 2018, 134, 65–78. [Google Scholar] [CrossRef]
- Rozenblum, G.T.; Lopez, V.G.; Vitullo, A.D.; Radrizzani, M. Aptamers: Current challenges and future prospects. Expert Opin. Drug Discov. 2016, 11, 127–135. [Google Scholar] [CrossRef]
- Rusconi, C.P.; Roberts, J.D.; Pitoc, G.A.; Nimjee, S.M.; White, R.R.; Quick, G.; Scardino, E.; Fay, W.P.; Sullenger, B.A. Antidote-mediated control of an anticoagulant aptamer in vivo. Nat. Biotechnol. 2004, 22, 1423–1428. [Google Scholar] [CrossRef]
- Ponce, A.T.; Hong, K.L. A Mini-Review: Clinical development and potential of aptamers for thrombotic events treatment and monitoring. Biomedicines 2019, 7, 55. [Google Scholar] [CrossRef]
- Platella, C.; Riccardi, C.; Montesarchio, D.; Roviello, G.N.; Musumeci, D. G-quadruplex-based aptamers against protein targets in therapy and diagnostics. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 1429–1447. [Google Scholar] [CrossRef]
- Kaur, H.; Bruno, J.G.; Kumar, A.; Sharma, T.K. Aptamers in the therapeutics and diagnostics pipelines. Theranostics 2018, 8, 4016–4032. [Google Scholar] [CrossRef] [PubMed]
- Sharma, T.K.; Bruno, J.G.; Dhiman, A. ABCs of DNA aptamer and related assay development. Biotechnol. Adv. 2017, 35, 275–301. [Google Scholar] [CrossRef] [PubMed]
- Rotem, D.; Jayasinghe, L.; Salichou, M.; Bayley, H. Protein detection by nanopores equipped with aptamers. J. Am. Chem. Soc. 2012, 134, 2781–2787. [Google Scholar] [CrossRef] [PubMed]
- Ngundi, M.M.; Shriver-Lake, L.C.; Moore, M.H.; Lassman, M.E.; Ligler, F.S.; Taitt, C.R. Array biosensor for detection of ochratoxin A in cereals and beverages. Anal. Chem. 2005, 77, 148–154. [Google Scholar] [CrossRef]
- Zhang, Y.; Lai, B.; Juhas, M. Recent advances in aptamer discovery and applications. Molecules 2019, 24, 941. [Google Scholar] [CrossRef]
- Feng, C.; Dai, S.; Wang, L. Optical aptasensors for quantitative detection of small biomolecules: A review. Biosens. Bioelectron. 2014, 59, 64–74. [Google Scholar] [CrossRef]
- Zhou, J.; Rossi, J. Aptamers as targeted therapeutics: Current potential and challenges. Nat. Rev. Drug Discov. 2017, 16, 181–202. [Google Scholar] [CrossRef]
- Sameiyan, E.; Bagheri, E.; Ramezani, M.; Alibolandi, M.; Abnous, K.; Taghdisi, S.M. DNA origami-based aptasensors. Biosens. Bioelectron. 2019, 143, 111662. [Google Scholar] [CrossRef]
- Deng, B.; Lin, Y.; Wang, C.; Li, F.; Wang, Z.; Zhang, H.; Li, X.-F.; Le, X.C. Aptamer binding assays for proteins: The thrombin example—A review. Anal. Chim. Acta 2014, 837, 1–15. [Google Scholar] [CrossRef]
- Song, S.; Wang, L.; Li, J.; Fan, C.; Zhao, J. Aptamer-based biosensors. TrAC Trends Anal. Chem. 2008, 27, 108–117. [Google Scholar] [CrossRef]
- Jing, M.; Bowser, M.T. Methods for measuring aptamer-protein equilibria: A review. Anal. Chim. Acta 2011, 686, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.L.; McKeague, M.; Liang, J.C.; Smolke, C.D. Kinetic and Equilibrium Binding Characterization of Aptamers to Small Molecules using a Label-Free, Sensitive, and Scalable Platform. Anal. Chem. 2014, 86, 3273–3278. [Google Scholar] [CrossRef] [PubMed]
- Bock, L.C.; Griffin, L.C.; Latham, J.A.; Vermaas, E.H.; Toole, J.J. Selection of single-stranded DNA molecules that bind and inhibit human thrombin. Nature 1992, 355, 564–566. [Google Scholar] [CrossRef] [PubMed]
- Tasset, D.M.; Kubik, M.F.; Steiner, W. Oligonucleotide inhibitors of human thrombin that bind distinct epitopes. J. Mol. Biol. 1997, 272, 688–698. [Google Scholar] [CrossRef]
- Mears, K.S.; Markus, D.L.; Ogunjimi, O.; Whelan, R.J. Experimental and mathematical evidence that thrombin-binding aptamers form a 1 aptamer: 2 protein complex. Aptamers 2018, 2, 64–73. [Google Scholar]
- Lin, P.-H.; Chen, R.-H.; Lee, C.-H.; Chang, Y.; Chen, C.-S.; Chen, W.-Y. Studies of the binding mechanism between aptamers and thrombin by circular dichroism, surface plasmon resonance and isothermal titration calorimetry. Colloids Surf. B 2011, 88, 552–558. [Google Scholar] [CrossRef]
- Li, Y.; Guo, L.; Zhang, F.; Zhang, Z.; Tang, J.; Xie, J. High-sensitive determination of human α-thrombin by its 29-mer aptamer in affinity probe capillary electrophoresis. Electrophoresis 2008, 29, 2570–2577. [Google Scholar] [CrossRef]
- Song, M.; Zhang, Y.; Li, T.; Wang, Z.; Yin, J.; Wang, H. Highly sensitive detection of human thrombin in serum by affinity capillary electrophoresis/laser-induced fluorescence polarization using aptamers as probes. J. Chromatogr. A 2009, 1216, 873–878. [Google Scholar] [CrossRef]
- Li, H.-Y.; Deng, Q.-P.; Zhang, D.-W.; Zhou, Y.-L.; Zhang, X.-X. Chemiluminescently labeled aptamers as the affinity probe for interaction analysis by capillary electrophoresis. Electrophoresis 2010, 31, 2452–2460. [Google Scholar] [CrossRef]
- Olmsted, I.R.; Xiao, Y.; Cho, M.; Csordas, A.T.; Sheehan, J.H.; Meiler, J.; Soh, H.T.; Bornhop, D.J. Measurement of aptamer–protein interactions with back-scattering interferometry. Anal. Chem. 2011, 83, 8867–8870. [Google Scholar] [CrossRef]
- Daniel, C.; Roupioz, Y.; Gasparutto, D.; Livache, T.; Buhot, A. Solution-phase vs surface-phase aptamer-protein affinity from a label-free kinetic biosensor. PLoS ONE 2013, 8, e75419. [Google Scholar] [CrossRef] [PubMed]
- Davie, E.W.; Kulman, J.D. An overview of the structure and function of thrombin. Semin. Thromb. Hemost. 2006, 32 (Suppl. S1), 3–15. [Google Scholar] [CrossRef] [PubMed]
- Champagne, K.; Shishido, A.; Root, M.J. Interactions of HIV-1 inhibitory peptide T20 with the gp41 N-HR coiled coil. J. Biol. Chem. 2009, 284, 3619–3627. [Google Scholar] [CrossRef] [PubMed]
- Du, D.; Kato, T.; Suzuki, F.; Park, E.Y. Binding affinity of full-length and extracellular domains of recombinant human (pro)renin receptor to human renin when expressed in the fat body and hemolymph of silkworm larvae. J. Biosci. Bioeng. 2009, 108, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Simón, B.; Miyachi, H.; Karube, I.; Saiki, H. High-sensitive flow-based kinetic exclusion assay for okadaic acid assessment in shellfish samples. Biosens. Bioelectron. 2010, 25, 1395–1401. [Google Scholar] [CrossRef] [PubMed]
- Darwish, I.A.; Wani, T.A.; Alanazi, A.M.; Hamidaddin, M.A.; Zargar, S. Kinetic-exclusion analysis-based immunosensors versus enzyme-linked immunosorbent assays for measurement of cancer markers in biological specimens. Talanta 2013, 111, 13–19. [Google Scholar] [CrossRef]
- Köck, K.; Pan, W.J.; Gow, J.M.; Horner, M.J.; Gibbs, J.P.; Colbert, A.; Goletz, T.J.; Newhall, K.J.; Rees, W.A.; Sun, Y.; et al. Preclinical development of AMG 139, a human antibody specifically targeting IL-23. Br. J. Pharmacol. 2015, 172, 159–172. [Google Scholar] [CrossRef]
- Abdiche, Y.N.; Harriman, R.; Deng, X.; Yeung, Y.A.; Miles, A.; Morishige, W.; Boustany, L.; Zhu, L.; Izquierdo, S.M.; Harriman, W. Assessing kinetic and epitopic diversity across orthogonal monoclonal antibody generation platforms. mAbs 2016, 8, 264–277. [Google Scholar] [CrossRef]
- Kusano-Arai, O.; Fukuda, R.; Kamiya, W.; Iwanari, H.; Hamakubo, T. Kinetic exclusion assay of monoclonal antibody affinity to the membrane protein roundabout 1 displayed on baculovirus. Anal. Biochem. 2016, 504, 41–49. [Google Scholar] [CrossRef]
- Li, X.; Kaattari, S.L.; Vogelbein, M.A.; Vadas, G.G.; Unger, M.A. A highly sensitive monoclonal antibody based biosensor for quantifying 3–5 ring polycyclic aromatic hydrocarbons (PAHs) in aqueous environmental samples. Sens. Bio-Sens. Res. 2016, 7, 115–120. [Google Scholar] [CrossRef]
- Wani, T.A.; Zargar, S.; Majid, S.; Darwish, I.A. Analytical application of flow immunosensor in detection of thyroxine and triiodothyronine in serum. ASSAY Drug Dev. Technol. 2016, 14, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Danial, M.; Stauffer, A.N.; Wurm, F.R.; Root, M.J.; Klok, H.-A. Site-specific polymer attachment to HR2 peptide fusion inhibitors against HIV-1 decreases binding association rates and dissociation rates rather than binding affinity. Bioconjug. Chem. 2017, 28, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.K.; Wojciak, J.M. Measuring sphingosine-1-phosphate/protein interactions with the kinetic exclusion assay. In Sphingosine-1-Phosphate: Methods and Protocols; Pébay, A., Turksen, K., Eds.; Methods in Molecular Biology; Springer: New York, NY, USA, 2018; pp. 1–8. ISBN 978-1-4939-7413-9. [Google Scholar]
- Kamat, V.; Rafique, A.; Huang, T.; Olsen, O.; Olson, W. The impact of different human IgG capture molecules on the kinetics analysis of antibody-antigen interaction. Anal. Biochem. 2020, 593, 113580. [Google Scholar] [CrossRef] [PubMed]
- Darling, R.J.; Brault, P.-A. Kinetic exclusion assay technology: Characterization of molecular interactions. ASSAY Drug Dev. Technol. 2004, 2, 647–657. [Google Scholar] [CrossRef]
- Hamidaddin, M.A.; AlRabiah, H.; Darwish, I.A. Development and comparative evaluation of two immunoassay platforms for bioanalysis of crizotinib: A potent drug used for the treatment of non-small cell lung cancer. Talanta 2019, 201, 217–225. [Google Scholar] [CrossRef]
- Darwish, I.A.; AlRabiah, H.; Hamidaddin, M.A. Development of two different formats of heterogeneous fluorescence immunoassay for bioanalysis of afatinib by employing fluorescence plate reader and KinExA 3200 immunosensor. Sci. Rep. 2019, 9, 14742. [Google Scholar] [CrossRef]
- AlRabiah, H.; Hamidaddin, M.A.; Darwish, I.A. Automated flow fluorescent noncompetitive immunoassay for measurement of human plasma levels of monoclonal antibodies used for immunotherapy of cancers with KinExATM 3200 biosensor. Talanta 2019, 192, 331–338. [Google Scholar] [CrossRef]
- Ohmura, N.; Lackie, S.J.; Saiki, H. An immunoassay for small analytes with theoretical detection limits. Anal. Chem. 2001, 73, 3392–3399. [Google Scholar] [CrossRef]
- Glass, T.R.; Saiki, H.; Blake, D.A.; Blake, R.C.; Lackie, S.J.; Ohmura, N. Use of excess solid-phase capacity in immunoassays: Advantages for semicontinuous, near-real-time measurements and for analysis of matrix effects. Anal. Chem. 2004, 76, 767–772. [Google Scholar] [CrossRef]
- Glass, T.R.; Ohmura, N.; Saiki, H. Least detectable concentration and dynamic range of three immunoassay systems using the same antibody. Anal. Chem. 2007, 79, 1954–1960. [Google Scholar] [CrossRef]
- Glass, T.R.; Winzor, D.J. Confirmation of the validity of the current characterization of immunochemical reactions by kinetic exclusion assay. Anal. Biochem. 2014, 456, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Blake, R.C.; Li, X.; Yu, H.; Blake, D.A. Covalent and noncovalent modifications induce allosteric binding behavior in a monoclonal antibody. Biochemistry 2007, 46, 1573–1586. [Google Scholar] [CrossRef] [PubMed]
- Tigue, N.J.; Bamber, L.; Andrews, J.; Ireland, S.; Hair, J.; Carter, E.; Sridharan, S.; Jovanović, J.; Rees, D.G.; Springall, J.S.; et al. MEDI1873, a potent, stabilized hexameric agonist of human GITR with regulatory T-cell targeting potential. OncoImmunology 2017, 6, e1280645. [Google Scholar] [CrossRef] [PubMed]
- KinExA Pro Software. Available online: https://www.sapidyne.com/kinexapro.html (accessed on 25 February 2020).
- Tabrizi, M.A.; Bornstein, G.G.; Klakamp, S.L.; Drake, A.; Knight, R.; Roskos, L. Translational strategies for development of monoclonal antibodies from discovery to the clinic. Drug Discov. Today 2009, 14, 298–305. [Google Scholar] [CrossRef]
- Varkey, R.; Du, Q.; Karnell, J.L.; Xiao, X.; Casey, K.A.; Woods, R.; Rosenthal, K.; Wilson, S.; Dall’Acqua, W.F.; Wu, H.; et al. Discovery and characterization of potent IL-21 neutralizing antibodies via a novel alternating antigen immunization and humanization strategy. PLoS ONE 2019, 14, e0211236. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.; Wen, W.; Conrad, F.; Meng, Q.; Dong, J.; Sun, Z.; Garcia-Rodriguez, C.; Farr-Jones, S.; Cheng, W.L.; Henderson, D.T.; et al. A single tri-epitopic antibody virtually recapitulates the potency of a combination of three monoclonal antibodies in neutralization of botulinum neurotoxin serotype A. Toxins 2018, 10, 84. [Google Scholar] [CrossRef] [PubMed]
- Szabó, E.; Phillips, D.J.; Droste, M.; Marti, A.; Kretzschmar, T.; Shamshiev, A.; Weller, M. Anti-tumor activity of DLX1008, an anti-VEGFA antibody fragment with low picomolar affinity, in human glioma models. J. Pharmacol. Exp. Ther. 2018, 365, 422–429. [Google Scholar] [CrossRef]
- Liddament, M.; Husten, J.; Estephan, T.; Laine, D.; Mabon, D.; Pukac, L.; Lyons, J.; Clarke, A.W.; Doyle, A. Higher Binding Affinity and in vitro Potency of Reslizumab for Interleukin-5 Compared with Mepolizumab. Allergy Asthma Immunol. Res. 2019, 11, 291. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, L.; Zhang, H.; Li, Y.; Liu, L.; Chen, Y.; Qiu, X.; Yu, D. Development of a Portable SPR Sensor for Nucleic Acid Detection. Micromachines 2020, 11, 526. [Google Scholar] [CrossRef]
- Delport, F.; Pollet, J.; Janssen, K.; Verbruggen, B.; Knez, K.; Spasic, D.; Lammertyn, J. Real-time monitoring of DNA hybridization and melting processes using a fiber optic sensor. Nanotechnology 2012, 23, 065503. [Google Scholar] [CrossRef]
- Zagorodko, O.; Spadavecchia, J.; Serrano, A.Y.; Larroulet, I.; Pesquera, A.; Zurutuza, A.; Boukherroub, R.; Szunerits, S. Highly Sensitive Detection of DNA Hybridization on Commercialized Graphene-Coated Surface Plasmon Resonance Interfaces. Anal. Chem. 2014, 86, 11211–11216. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhan, J.; Man, B.; Jiang, S.; Yue, W.; Gao, S.; Guo, C.; Liu, H.; Li, Z.; Wang, J.; et al. Real-time reliable determination of binding kinetics of DNA hybridization using a multi-channel graphene biosensor. Nat. Commun. 2017, 8, 14902. [Google Scholar] [CrossRef] [PubMed]
- Ohmura, N.; Tsukidate, Y.; Shinozaki, H.; Lackie, S.J.; Saiki, H. Combinational use of antibody affinities in an immunoassay for extension of dynamic range and detection of multiple analytes. Anal. Chem. 2003, 75, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Jones, R.M.; Glass, T.R.; Navoa, R.; Wang, Y.; Grace, M.J. Measurement of the functional affinity constant of a monoclonal antibody for cell surface receptors using kinetic exclusion fluorescence immunoassay. J. Immunol. Methods 2005, 304, 1–14. [Google Scholar] [CrossRef]
- Glass, T.R.; Ohmura, N.; Morita, K.; Sasaki, K.; Saiki, H.; Takagi, Y.; Kataoka, C.; Ando, A. Improving an immunoassay response to related polychlorinated biphenyl analytes by mixing antibodies. Anal. Chem. 2006, 78, 7240–7247. [Google Scholar] [CrossRef]
- Gubler, H.; Schopfer, U.; Jacoby, E. Theoretical and experimental relationships between percent inhibition and IC50 data observed in high-throughput screening. J. Biomol. Screen. 2013, 18, 1–13. [Google Scholar] [CrossRef]
- Drake, A.W.; Tang, M.L.; Papalia, G.A.; Landes, G.; Haak-Frendscho, M.; Klakamp, S.L. Biacore surface matrix effects on the binding kinetics and affinity of an antigen/antibody complex. Anal. Biochem. 2012, 429, 58–69. [Google Scholar] [CrossRef]
- Schuck, P.; Zhao, H. The role of mass transport limitation and surface heterogeneity in the biophysical characterization of macromolecular binding processes by SPR biosensing. Methods Mol. Biol. 2010, 627, 15–54. [Google Scholar] [CrossRef]
- Bromage, E.S.; Vadas, G.G.; Harvey, E.; Unger, M.A.; Kaattari, S.L. Validation of an antibody-based biosensor for rapid quantification of 2,4,6-trinitrotoluene (TNT) contamination in ground water and river water. Environ. Sci. Technol. 2007, 41, 7067–7072. [Google Scholar] [CrossRef]
- Blake, D.A.; Jones, R.M.; Blake, R.C., II; Pavlov, A.R.; Darwish, I.A.; Yu, H. Antibody-based sensors for heavy metal ions. Biosens. Bioelectron. 2001, 16, 799–809. [Google Scholar] [CrossRef]
- Sasaki, K.; Oguma, S.; Namiki, Y.; Ohmura, N. Monoclonal antibody to trivalent chromium chelate complex and its application to measurement of the total chromium concentration. Anal. Chem. 2009, 81, 4005–4009. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Glass, T.R.; Ohmura, N. Validation of accuracy of enzyme-linked immunosorbent assay in hybridoma screening and proposal of an improved screening method. Anal. Chem. 2005, 77, 1933–1939. [Google Scholar] [CrossRef] [PubMed]
- Bedinger, D.H.; Goldfine, I.D.; Corbin, J.A.; Roell, M.K.; Adams, S.H. Differential pathway coupling of the activated insulin receptor drives signaling selectivity by XMetA, an allosteric partial agonist antibody. J. Pharmacol. Exp. Ther. 2015. [Google Scholar] [CrossRef]
- Kariolis, M.S.; Miao, Y.R.; Diep, A.; Nash, S.E.; Olcina, M.M.; Jiang, D.; Jones, D.S., II; Kapur, S.; Mathews, I.I.; Koong, A.C.; et al. Inhibition of the GAS6/AXL pathway augments the efficacy of chemotherapies. J. Clin. Investig. 2017, 127, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Rathanaswami, P.; Babcook, J.; Gallo, M. High-affinity binding measurements of antibodies to cell-surface-expressed antigens. Anal. Biochem. 2008, 373, 52–60. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, M.H.; Fologea, D. Kinetic Exclusion Assay of Biomolecules by Aptamer Capture. Sensors 2020, 20, 3442. https://doi.org/10.3390/s20123442
Smith MH, Fologea D. Kinetic Exclusion Assay of Biomolecules by Aptamer Capture. Sensors. 2020; 20(12):3442. https://doi.org/10.3390/s20123442
Chicago/Turabian StyleSmith, Mark H., and Daniel Fologea. 2020. "Kinetic Exclusion Assay of Biomolecules by Aptamer Capture" Sensors 20, no. 12: 3442. https://doi.org/10.3390/s20123442
APA StyleSmith, M. H., & Fologea, D. (2020). Kinetic Exclusion Assay of Biomolecules by Aptamer Capture. Sensors, 20(12), 3442. https://doi.org/10.3390/s20123442




