Theoretical Aspects of Resting-State Cardiomyocyte Communication for Multi-Nodal Nano-Actuator Pacemakers
Abstract
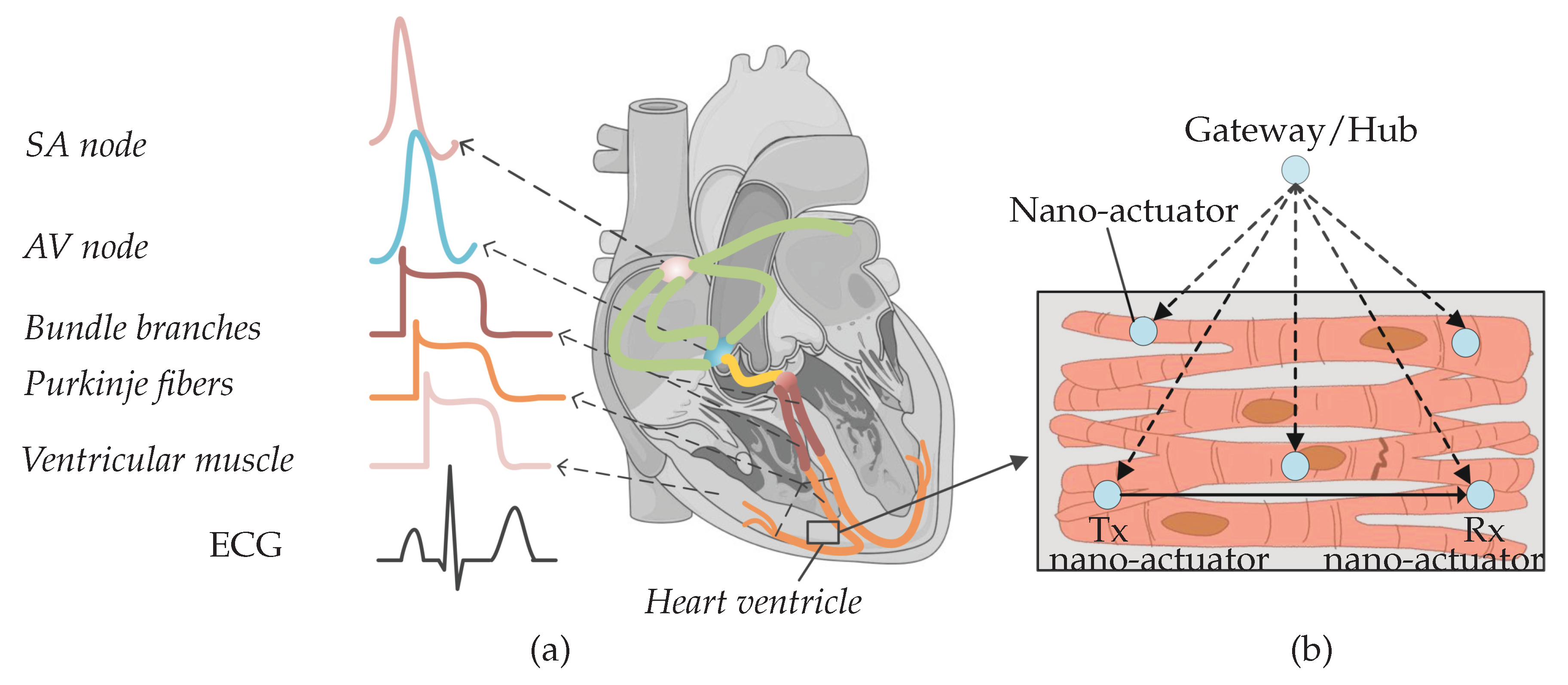
1. Introduction
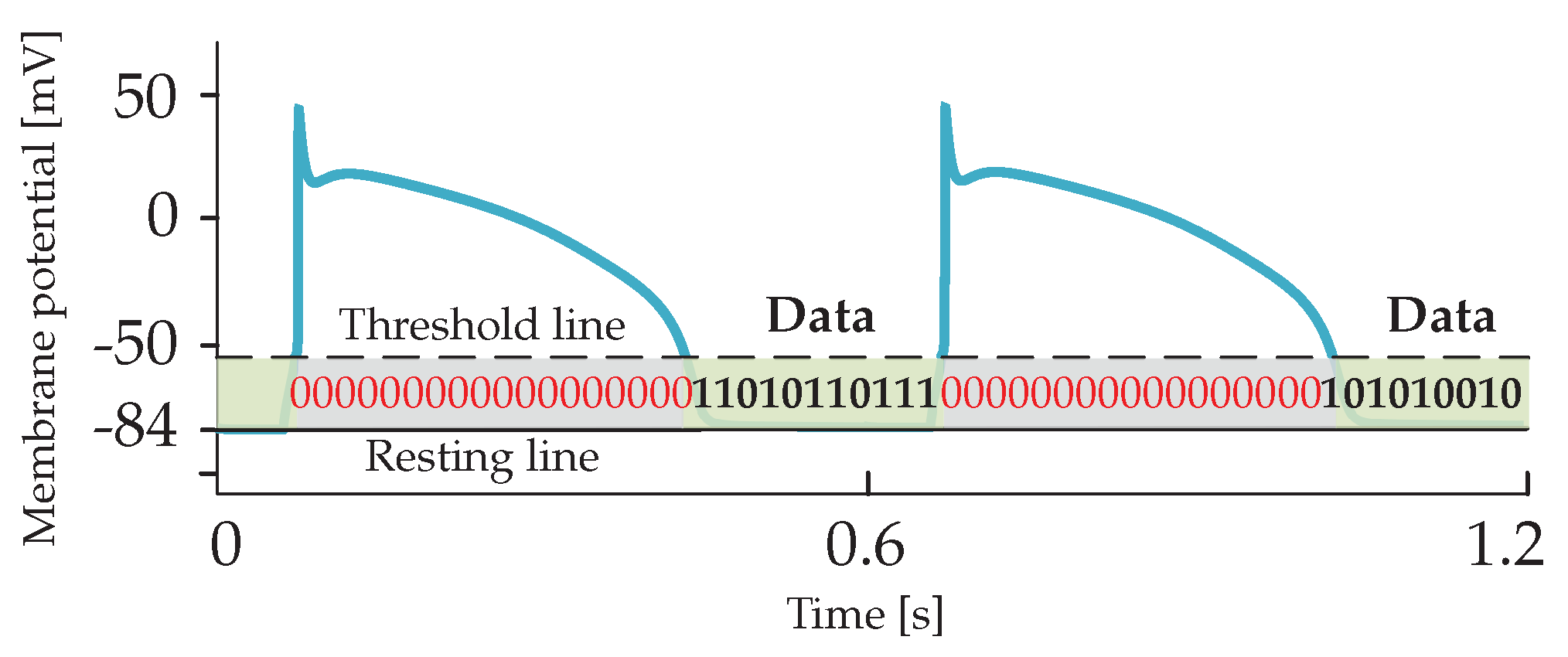
2. Subthreshold Cardiac Communication System
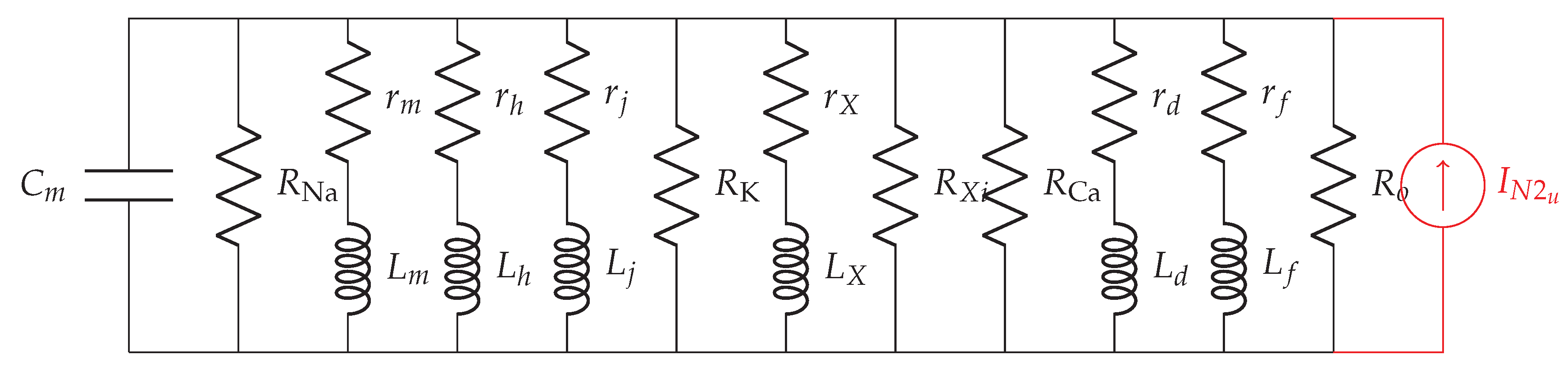
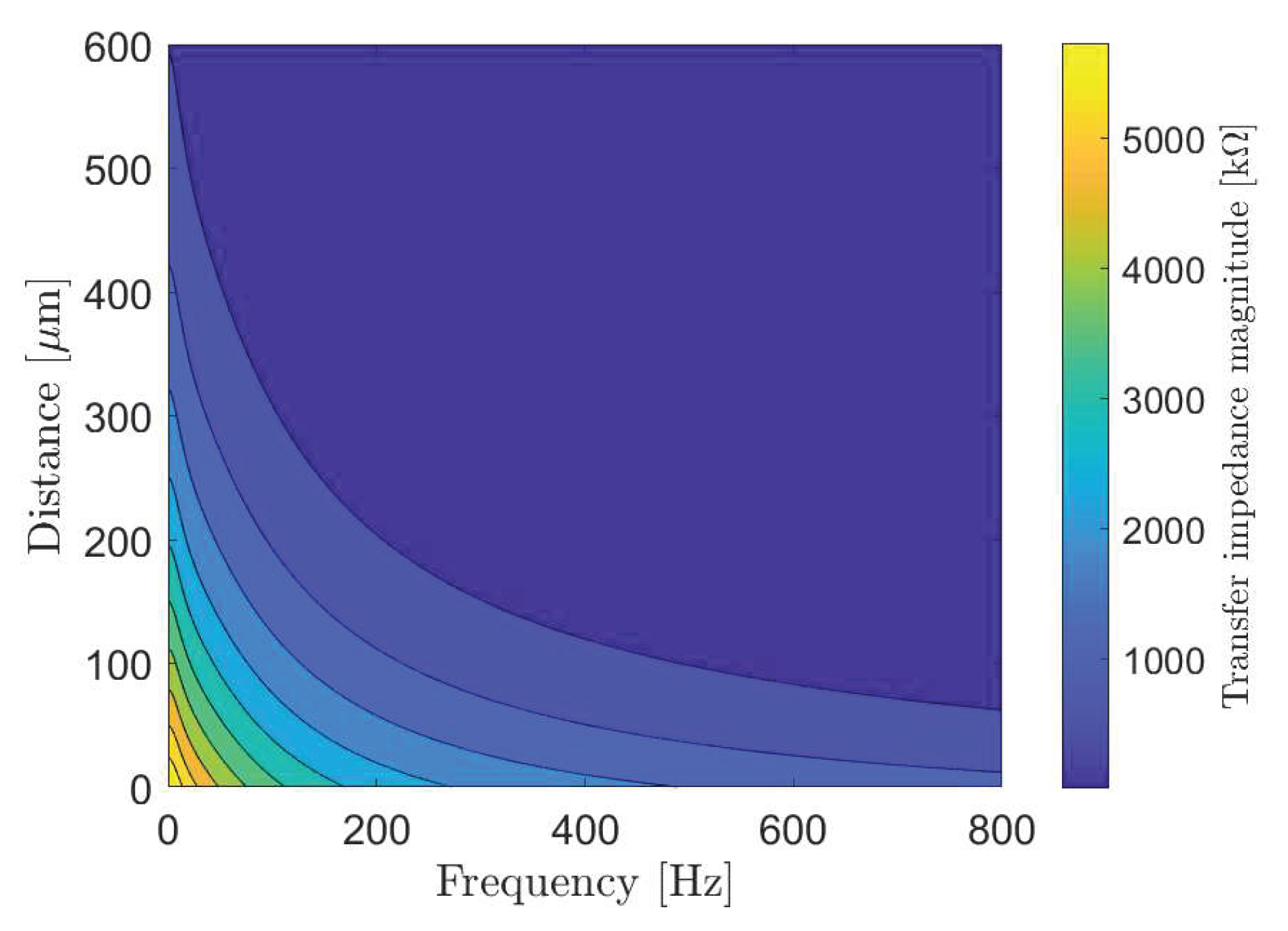
2.1. Transfer Impedance
2.2. Noiseless Input-Output Relation
3. Noise in the Subthreshold Cardiac Communication System
3.1. Encoding Noise
3.2. Membrane-Related Noise
3.2.1. Voltage-Gated Channel Noise
3.2.2. Shot Noise
3.2.3. Thermal Noise
3.3. Noisy Input-Output Relation
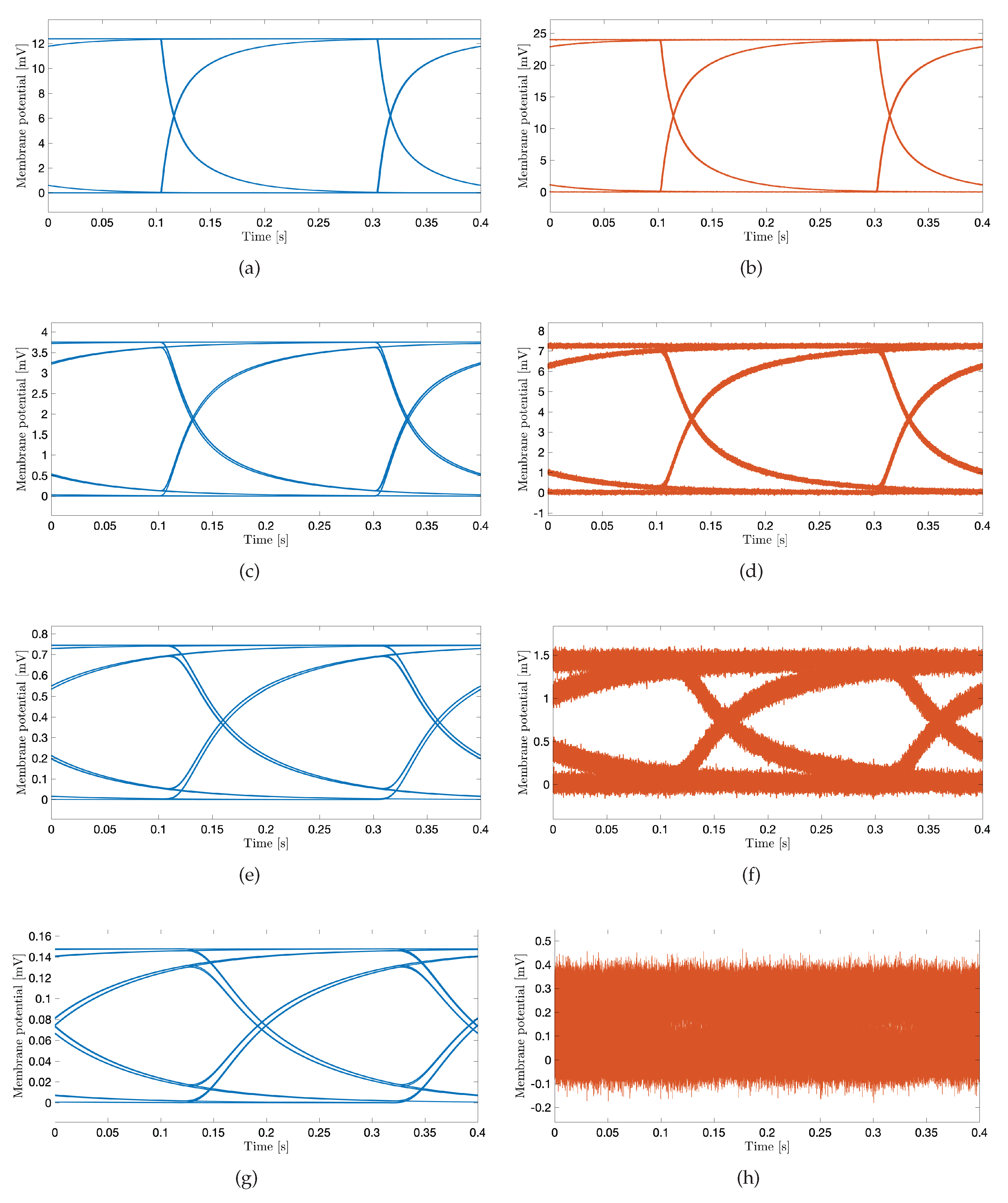
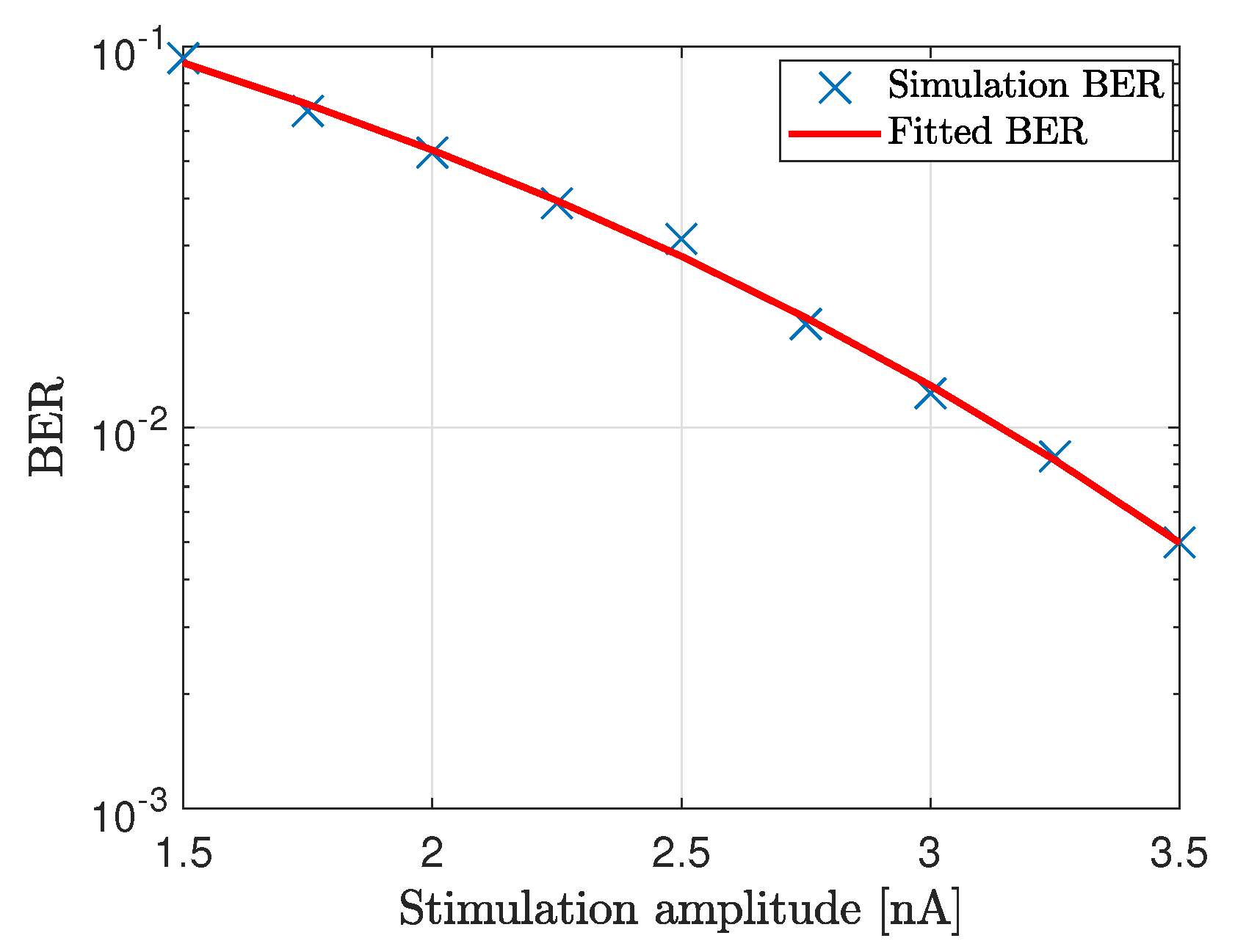
4. Numerical Simulations
5. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
Appendix A
Appendix A.1. Membrane Linearization
Appendix A.2. Derivation of Current Noise PSD
References
- Lu, P.; Veletić, M.; Laasmaa, M.; Vendelin, M.; Louch, W.E.; Halvorsen, P.S.; Bergsland, J.; Balasingham, I. Multi-nodal nano-actuator pacemaker for energy-efficient stimulation of cardiomyocytes. Nano Commun. Netw. 2019, 22, 100270. [Google Scholar] [CrossRef]
- Laasmaa, M.; Lu, P.; Veletić, M.; Louch, W.E.; Bergsland, J.; Balasingham, I.; Vendelin, M. Energy-efficiency of Cardiomyocyte Stimulation with Rectangular Pulses. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yen, H.H.; Wang, X.; Wang, D.; Liaw, H.T. A Node Activation-Based Routing Scheme in Micro/Nanobots Networks. IEEE Access 2019, 7, 144075–144089. [Google Scholar] [CrossRef]
- Atakan, B.; Akan, O.B.; Balasubramaniam, S. Body area nanonetworks with molecular communications in nanomedicine. IEEE Commun. Mag. 2012, 50, 28–34. [Google Scholar] [CrossRef]
- Veletić, M.; Balasingham, I. Synaptic Communication Engineering for Future Cognitive Brain–Machine Interfaces. Proc. IEEE 2019, 107, 1425–1441. [Google Scholar] [CrossRef]
- Akyildiz, I.F.; Pierobon, M.; Balasubramaniam, S. An Information Theoretic Framework to Analyze Molecular Communication Systems Based on Statistical Mechanics. Proc. IEEE 2019, 107, 1230–1255. [Google Scholar] [CrossRef]
- Akyildiz, I.F.; Brunetti, F.; Blázquez, C. Nanonetworks: A new communication paradigm. Comput. Netw. 2008, 52, 2260–2279. [Google Scholar] [CrossRef]
- Hiyama, S.; Moritani, Y.; Suda, T.; Egashira, R.; Enomoto, A.; Moore, M.; Nakano, T. Molecular communication. J. Inst. Electron. Inf. Commun. Eng. 2006, 89, 162. [Google Scholar]
- Akyildiz, I.F.; Jornet, J.M.; Han, C. Terahertz band: Next frontier for wireless communications. Phys. Commun. 2014, 12, 16–32. [Google Scholar] [CrossRef]
- Veletić, M.; Floor, P.A.; Babić, Z.; Balasingham, I. Peer-to-peer communication in neuronal nano-network. IEEE Trans. Commun. 2016, 64, 1153–1166. [Google Scholar] [CrossRef]
- Rutherglen, C.; Burke, P. Nanoelectromagnetics: Circuit and electromagnetic properties of carbon nanotubes. Small 2009, 5, 884–906. [Google Scholar] [CrossRef]
- Jornet, J.M.; Akyildiz, I.F. Graphene-based plasmonic nano-antenna for terahertz band communication in nanonetworks. IEEE J. Sel. Areas Commun. 2013, 31, 685–694. [Google Scholar] [CrossRef]
- Malak, D.; Akan, O.B. Molecular communication nanonetworks inside human body. Nano Commun. Netw. 2012, 3, 19–35. [Google Scholar] [CrossRef]
- Farsad, N.; Yilmaz, H.B.; Eckford, A.; Chae, C.B.; Guo, W. A comprehensive survey of recent advancements in molecular communication. IEEE Commun. Surv. Tutor. 2016, 18, 1887–1919. [Google Scholar] [CrossRef]
- Nakano, T.; Suda, T. Molecular communication using dynamic properties of oscillating and propagating patterns in concentration of information molecules. IEEE Trans. Commun. 2017, 65, 3386–3398. [Google Scholar] [CrossRef]
- Akkaya, A.; Yilmaz, H.B.; Chae, C.B.; Tugcu, T. Effect of receptor density and size on signal reception in molecular communication via diffusion with an absorbing receiver. IEEE Commun. Lett. 2015, 19, 155–158. [Google Scholar] [CrossRef]
- Farsad, N.; Guo, W.; Eckford, A.W. Tabletop molecular communication: Text messages through chemical signals. PLoS ONE 2013, 8, e82935. [Google Scholar] [CrossRef] [PubMed]
- Ruhi, N.A.; Bogdan, P. Multiscale modeling of biological communication. In Proceedings of the 2015 IEEE International Conference on Communications (ICC), London, UK, 8–12 June 2015; pp. 1140–1145. [Google Scholar]
- Kadooka, Y.; Iwamura, T.; Nakagawa, M.; Watanabe, M. Developing Biomedical Simulations for Next-generation Healthcare. FUJITSU Sci. Tech. J. 2015, 51, 69–76. [Google Scholar]
- Joyner, R.W. Effects of the discrete pattern of electrical coupling on propagation through an electrical syncytium. Circ. Res. 1982, 50, 192–200. [Google Scholar] [CrossRef]
- Varghese, A. Reciprocal modulation of IK1–INa extends excitability in cardiac ventricular cells. Front. Physiol. 2016, 7, 542. [Google Scholar] [CrossRef]
- Kardami, E.; Doble, B.W. Cardiomyocyte gap junctions: A target of growth-promoting signaling. Trends Cardiovasc. Med. 1998, 8, 180–187. [Google Scholar] [CrossRef]
- Hejri, F.; Veletić, M.; Balasingham, I. On the Cardiac Gap Junctions Channel Modeling. In Proceedings of the Sixth Annual ACM International Conference on Nanoscale Computing and Communication, NANOCOM’19, Dublin, Ireland, 25–27 September 2019; Association for Computing Machinery: New York, NY, USA, 2019; pp. 1–6. [Google Scholar]
- Joyner, R.W.; Picone, J.; Veenstra, R.; Rawling, D. Propagation through electrically coupled cells. Effects of regional changes in membrane properties. Circ. Res. 1983, 53, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Jack, J.J.B.; Noble, D.; Tsien, R.W. Electric Current Flow in Excitable Cells; Clarendon Press: Oxford, UK, 1975. [Google Scholar]
- Mauro, A.; Conti, F.; Dodge, F.; Schor, R. Subthreshold behavior and phenomenological impedance of the squid giant axon. J. Gen. Physiol. 1970, 55, 497–523. [Google Scholar] [CrossRef] [PubMed]
- Clapham, D.E.; DeFelice, L.J. Small signal impedance of heart cell membranes. J. Membr. Biol. 1982, 67, 63–71. [Google Scholar] [CrossRef]
- Sabah, N.; Leibovic, K. Subthreshold oscillatory responses of the Hodgkin-Huxley cable model for the squid giant axon. Biophys. J. 1969, 9, 1206–1222. [Google Scholar] [CrossRef]
- Koch, C. Cable theory in neurons with active, linearized membranes. Biol. Cybern. 1984, 50, 15–33. [Google Scholar] [CrossRef]
- Rudy, Y.; Quan, W. A model study of the effects of the discrete cellular structure on electrical propagation in cardiac tissue. Circ. Res. 1987, 61, 815–823. [Google Scholar] [CrossRef]
- Fear, E.; Stuchly, M. A novel equivalent circuit model for gap-connected cells. Phys. Med. Biol. 1998, 43, 1439–1448. [Google Scholar] [CrossRef]
- DeFelice, L.J.; Firth, D.R. Spontaneous Voltage Fluctuations in Glass Microelectrodes. IEEE Trans. Biomed. Eng. 1971, BME-18, 339–351. [Google Scholar] [CrossRef]
- Kavcic, A.; Patapoutian, A. A signal-dependent autoregressive channel model. IEEE Trans. Magn. 1999, 35, 2316–2318. [Google Scholar] [CrossRef]
- Jabbari, A.; Balasingham, I. Noise characterization in a stochastic neural communication network. Nano Commun. Netw. 2013, 4, 65–72. [Google Scholar] [CrossRef]
- Clay, J.R.; DeFelice, L.; DeHaan, R.L. Current noise parameters derived from voltage noise and impedance in embryonic heart cell aggregates. Biophys. J. 1979, 28, 169–184. [Google Scholar] [CrossRef]
- Brunetti, R.; Affinito, F.; Jacoboni, C.; Piccinini, E.; Rudan, M. Shot noise in single open ion channels: A computational approach based on atomistic simulations. J. Comput. Electron. 2007, 6, 391–394. [Google Scholar] [CrossRef]
- Manwani, A.; Koch, C. Detecting and estimating signals in noisy cable structures, I: Neuronal noise sources. Neural Comput. 1999, 11, 1797–1829. [Google Scholar] [CrossRef]
- Manwani, A.; Steinmetz, P.N.; Koch, C. Channel noise in excitable neural membranes. In Proceedings of the Advances in Neural Information Processing Systems, Denver, CO, USA, 20 June 2000; pp. 143–149. [Google Scholar]
- Steinmetz, P.N.; Manwani, A.; Koch, C.; London, M.; Segev, I. Subthreshold voltage noise due to channel fluctuations in active neuronal membranes. J. Comput. Neurosci. 2000, 9, 133–148. [Google Scholar] [CrossRef]
- Manwani, A.; Koch, C. Detecting and estimating signals in noisy cable structures, II: Information theoretical analysis. Neural Comput. 1999, 11, 1831–1873. [Google Scholar] [CrossRef]
- O’Donnell, C.; van Rossum, M.C. Systematic analysis of the contributions of stochastic voltage gated channels to neuronal noise. Front. Comput. Neurosci. 2014, 8, 105. [Google Scholar] [CrossRef]
- Ab-Rahman, M.S.; Ibrahim, M.F.; Rahni, A.A.A. Thermal Noise Effect in FTTH Communication Systems. In Proceedings of the 2008 Fourth Advanced International Conference on Telecommunications, Athens, Greece, 8–13 June 2008; pp. 364–370. [Google Scholar]
- Luo, C.h.; Rudy, Y. A model of the ventricular cardiac action potential. Depolarization, repolarization, and their interaction. Circ. Res. 1991, 68, 1501–1526. [Google Scholar] [CrossRef]
- Hodgkin, A.L.; Huxley, A.F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Phys. 1952, 117, 500–544. [Google Scholar] [CrossRef]
- Harald, R. Ion channels in cardiac cell membranes. Annu. Rev. Phys. 1984, 46, 473–484. [Google Scholar]
- Reuter, H.; Stevens, C.F.; Tsien, R.W.; Yellen, G. Properties of single calcium channels in cardiac cell culture. Nature 1982, 297, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Cachelin, A.B.; De Peyer, J.E.; Kokubun, S.; Reuter, H. Sodium channels in cultured cardiac cells. J. Phys. 1983, 340, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, T. Conductance and kinetics of delayed rectifier potassium channels in nodal cells of the rabbit heart. J. Phys. 1987, 387, 227–250. [Google Scholar] [CrossRef] [PubMed]
- Vitale, F.; Summerson, S.R.; Aazhang, B.; Kemere, C.; Pasquali, M. Neural stimulation and recording with bidirectional, soft carbon nanotube fiber microelectrodes. ACS Nano 2015, 9, 4465–4474. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.G.; DeSaix, P.; Johnson, E.; Johnson, J.E.; Korol, O.; Kruse, D.H.; Poe, B.; Wise, J.A.; Young, K.A. Anatomy and Physiology; OpenStax College: Houston, TX, USA, 2014. [Google Scholar]
- DeFelice, L.J. Introduction to Membrane Noise; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Johnston, D.; Wu, S.M.S. Foundations of Cellular Neurophysiology; MIT Press: Cambridge, MA, USA, 1994. [Google Scholar]



| Parameter | Description | Unit |
|---|---|---|
| Input current PSD | A/Hz | |
| Membrane impedance (per unit length) | k cm | |
| Resistivity of membrane | k cm | |
| Equivalent longitudinal impedance (per unit length) | k/cm | |
| Equivalent longitudinal resistivity | k cm | |
| Transfer impedance | k | |
| Output voltage PSD | mV/Hz | |
| Input current | A | |
| Input current corrupted with input-dependent noise | A | |
| Input-dependent noise current | A | |
| Current PSD of input-dependent noise current | A/Hz | |
| Current PSD of the input corrupted with input-dependent noise | A/Hz | |
| Current PSD of voltage-gated channel noise | A/Hz/cm | |
| Current PSD of shot noise | A/Hz/cm | |
| Current PSD of thermal noise | A/Hz/cm | |
| Current PSD of potassium ions | A/Hz/cm | |
| Current PSD of sodium ions | A/Hz/cm | |
| Current PSD of calcium ions | A/Hz/cm | |
| Current PSD of membrane-related noise | A/Hz/cm | |
| Voltage PSD of membrane-related noise | mV/Hz | |
| Output noisy voltage PSD | mV/Hz |
| Parameter | Description | Value | Unit |
|---|---|---|---|
| Specific membrane capacitance | 1 | F/cm | |
| Reversal potential | 54.4 | mV | |
| Reversal potential | −77 | mV | |
| Reversal potential | 40 | mV | |
| Channel conductance | 15 | pS | |
| Channel density | 1–16 | /m | |
| Channel conductance | 9–25 | pS | |
| Channel density | 0.5–5 | /m | |
| Channel conductance | 1.6 | pS | |
| Channel density | 0.7 | /m | |
| Equivalent longitudinal resistivity | 600 | cm | |
| Surface-to-volume ratio | 4440 | cm | |
| Cell length | 100 | m | |
| a | Cell radius | 10 | m |
| Parameter | Value | Unit |
|---|---|---|
| 1.13 × 10 | kcm | |
| −1.64 × 10 | kcm | |
| −0.98 | ks·cm | |
| 1.92 × 10 | kcm | |
| 7.74 × 10 | ks·cm | |
| 3.13 × 10 | kcm | |
| 5.32 × 10 | ks·cm | |
| 2.13 × 10 | kcm | |
| −3.11 × 10 | kcm | |
| −711.64 | ks·cm | |
| 2.32 × 10 | kcm | |
| 160.35 | kcm | |
| −15.29 | kcm | |
| −0.13 | ks·cm | |
| 3.31 × 10 | kcm | |
| 1.76 × 10 | ks·cm | |
| 6.11 × 10 | kcm |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, P.; Veletić, M.; Bergsland, J.; Balasingham, I. Theoretical Aspects of Resting-State Cardiomyocyte Communication for Multi-Nodal Nano-Actuator Pacemakers. Sensors 2020, 20, 2792. https://doi.org/10.3390/s20102792
Lu P, Veletić M, Bergsland J, Balasingham I. Theoretical Aspects of Resting-State Cardiomyocyte Communication for Multi-Nodal Nano-Actuator Pacemakers. Sensors. 2020; 20(10):2792. https://doi.org/10.3390/s20102792
Chicago/Turabian StyleLu, Pengfei, Mladen Veletić, Jacob Bergsland, and Ilangko Balasingham. 2020. "Theoretical Aspects of Resting-State Cardiomyocyte Communication for Multi-Nodal Nano-Actuator Pacemakers" Sensors 20, no. 10: 2792. https://doi.org/10.3390/s20102792
APA StyleLu, P., Veletić, M., Bergsland, J., & Balasingham, I. (2020). Theoretical Aspects of Resting-State Cardiomyocyte Communication for Multi-Nodal Nano-Actuator Pacemakers. Sensors, 20(10), 2792. https://doi.org/10.3390/s20102792





