Automatic Range Adjustment of the Fluorescence Immunochromatographic Assay Based on Image Processing
Abstract
1. Introduction
2. Materials and Methods
2.1. Basic Principles
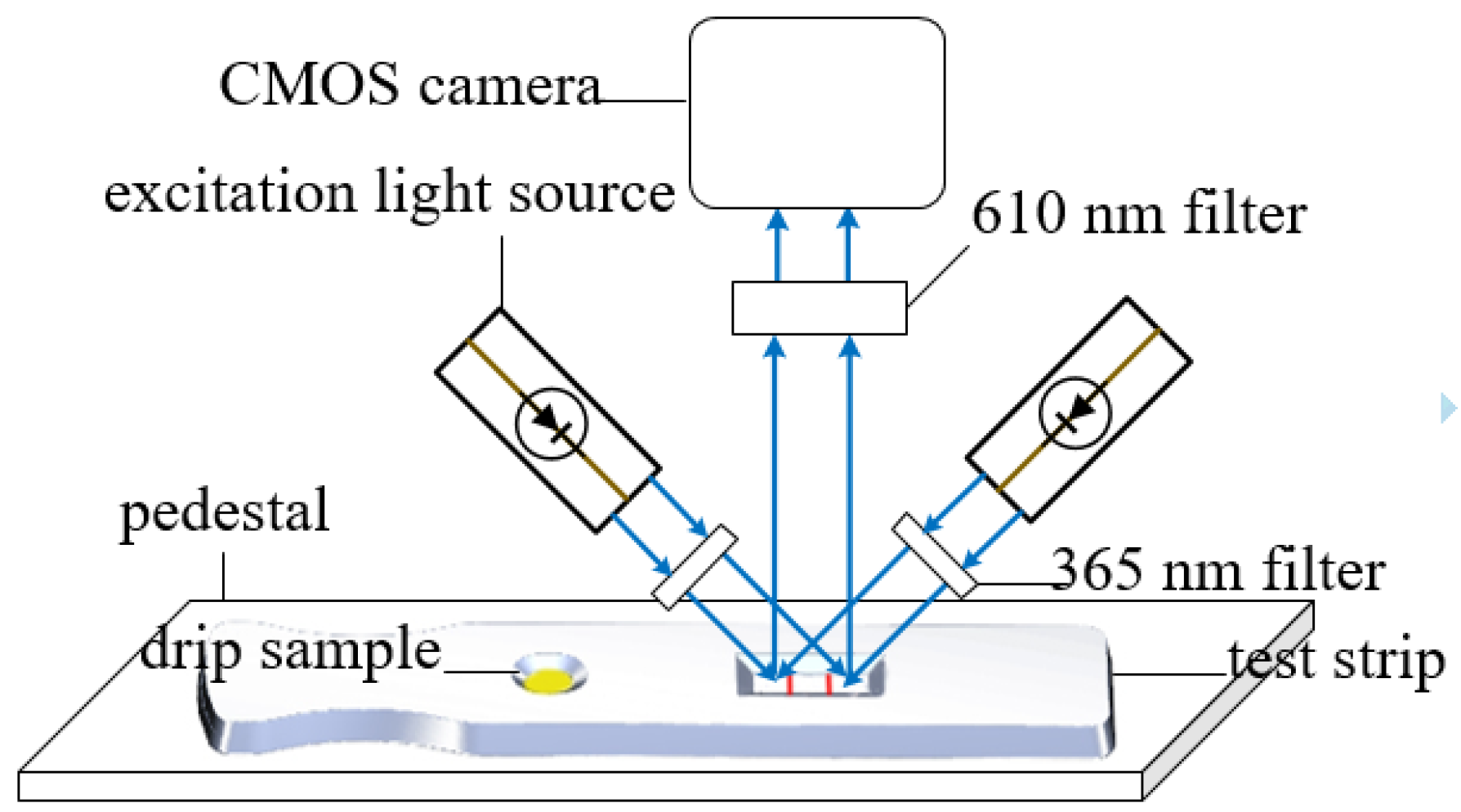
2.1.1. Detection Mechanism of the FICA Based on Image Processing
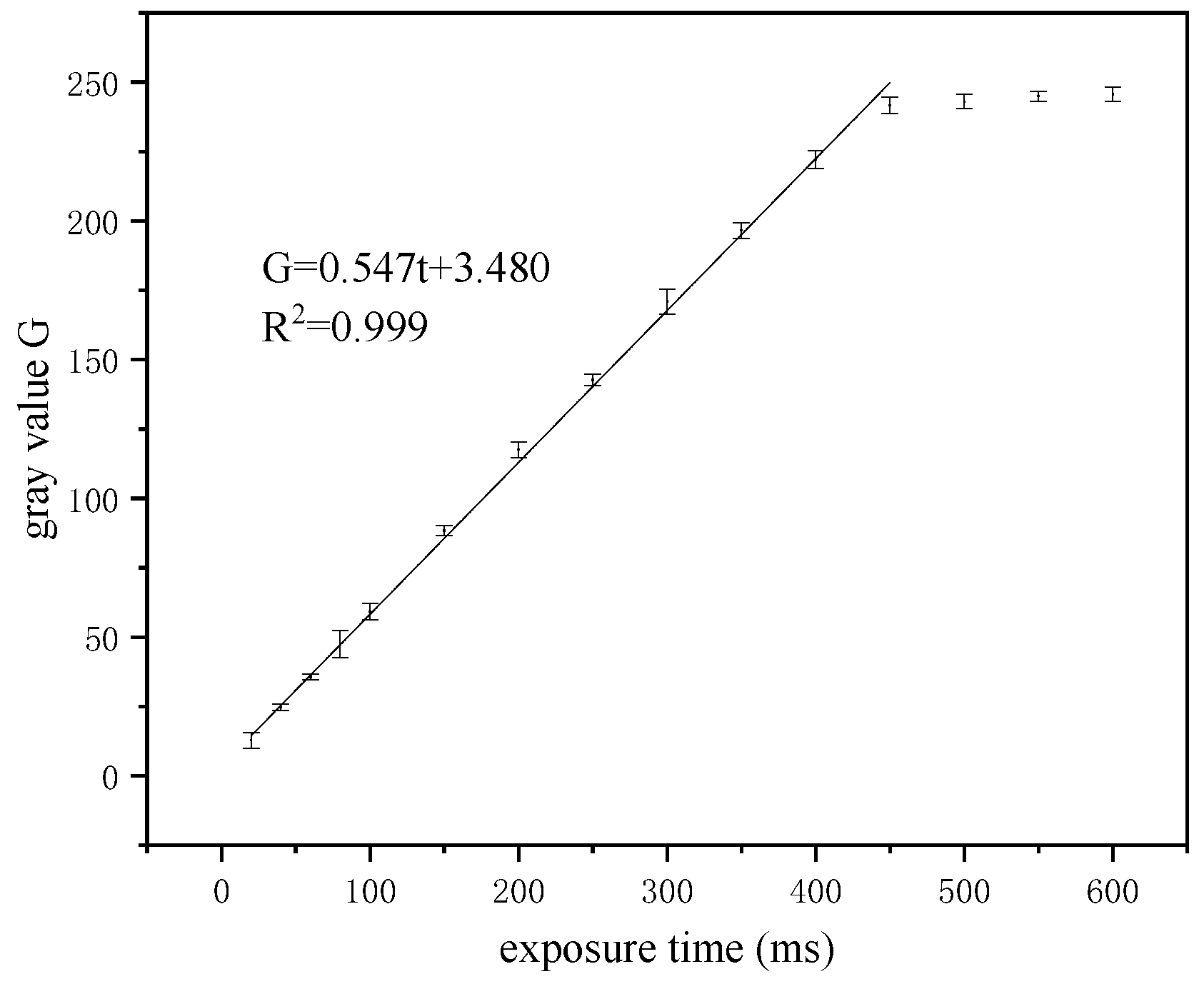
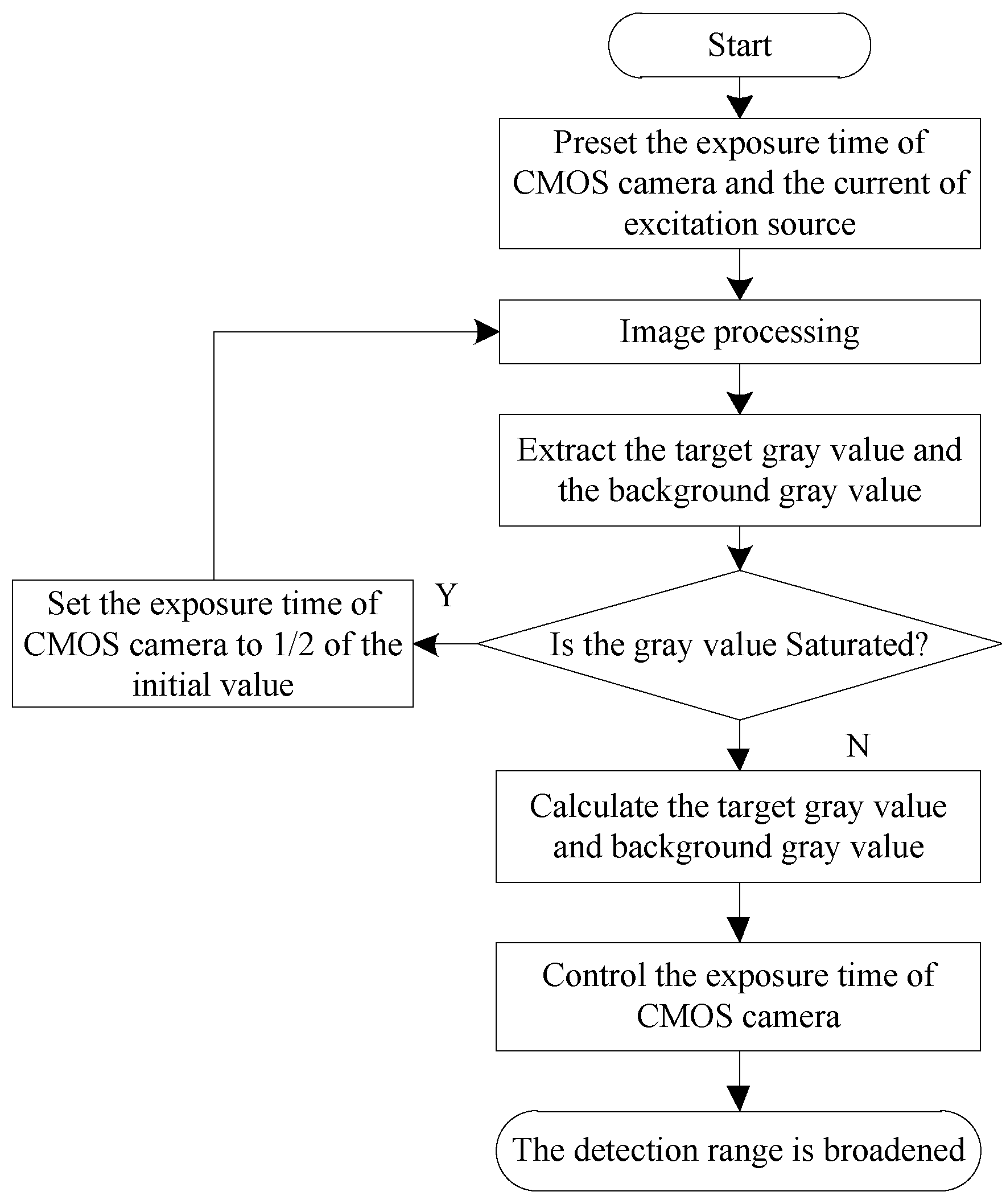
2.1.2. Mechanism of Exposure Time Adjustment
2.2. Methods
2.2.1. Devices
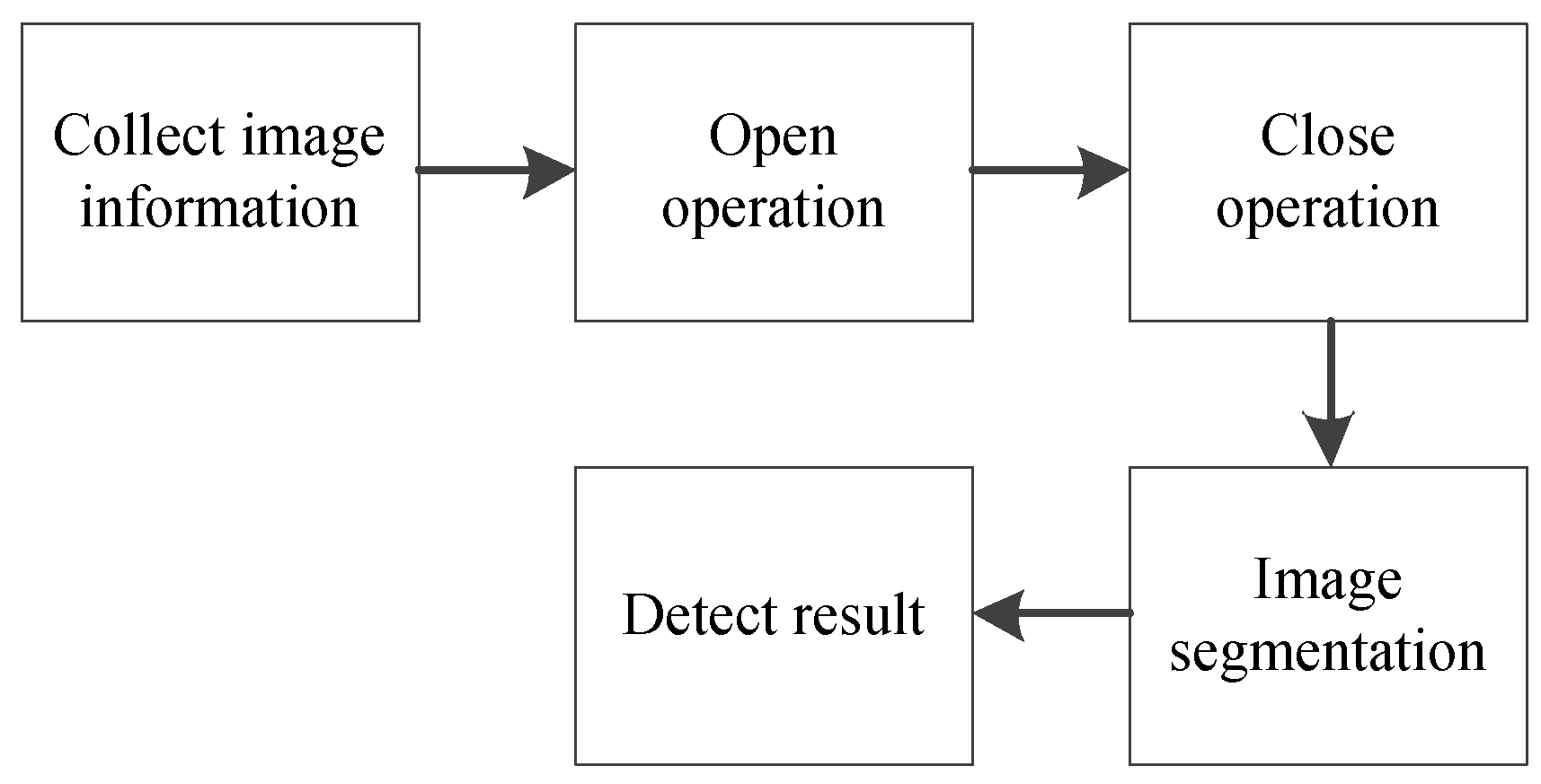
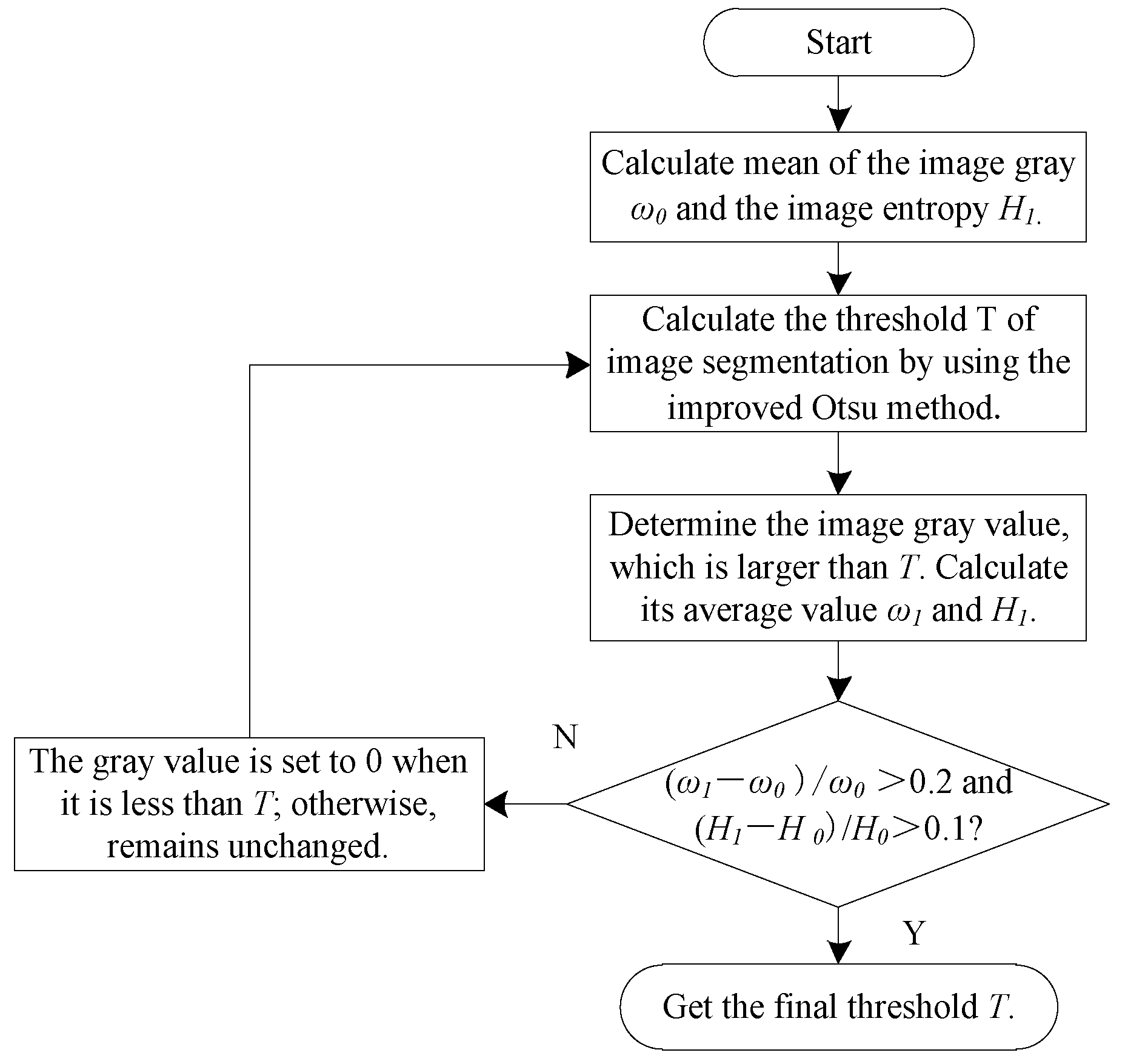
2.2.2. Range Adjustment
3. Results
3.1. Range Adjustment Effect of FICA Strips
3.2. Range Comparison
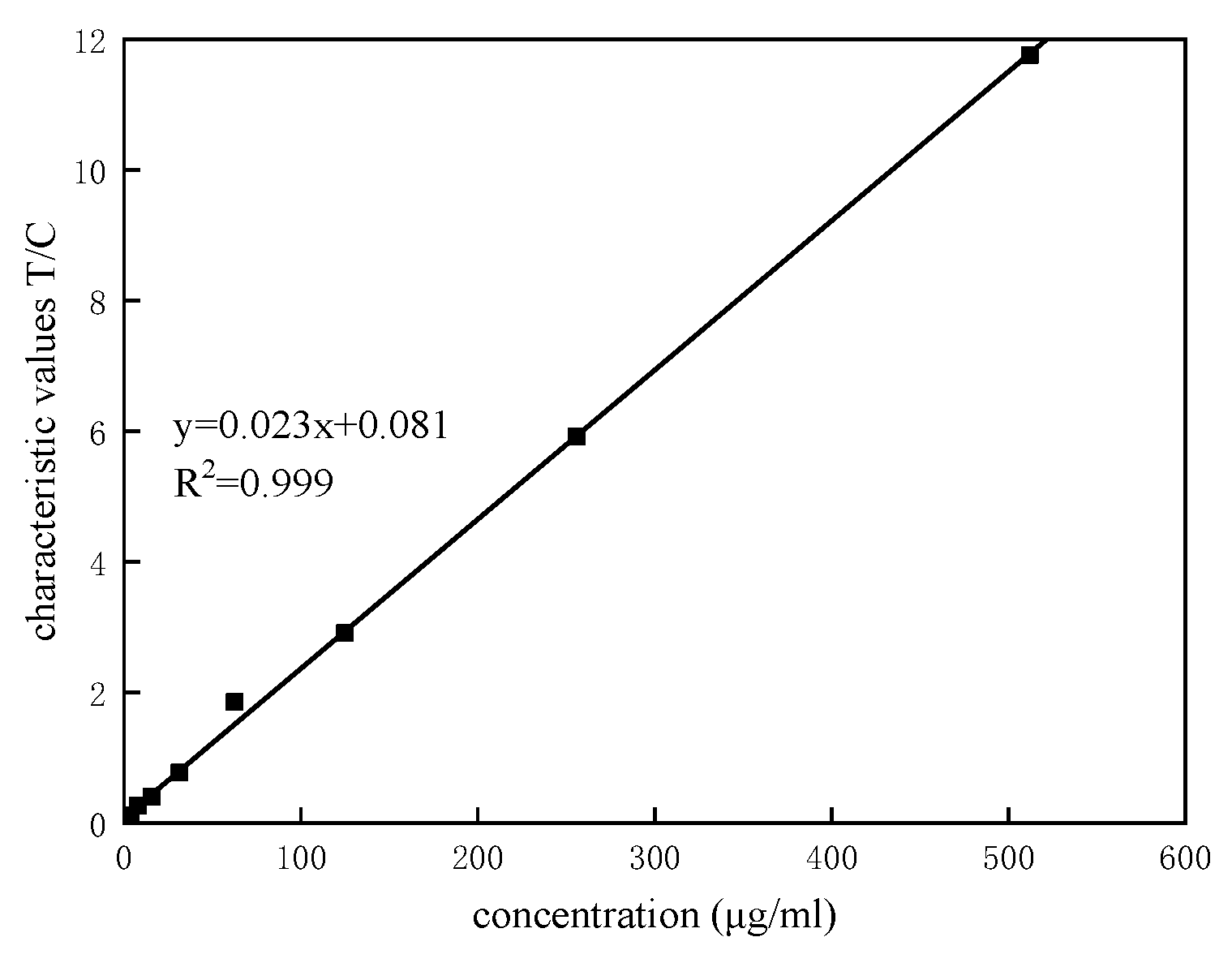
3.3. Linearity
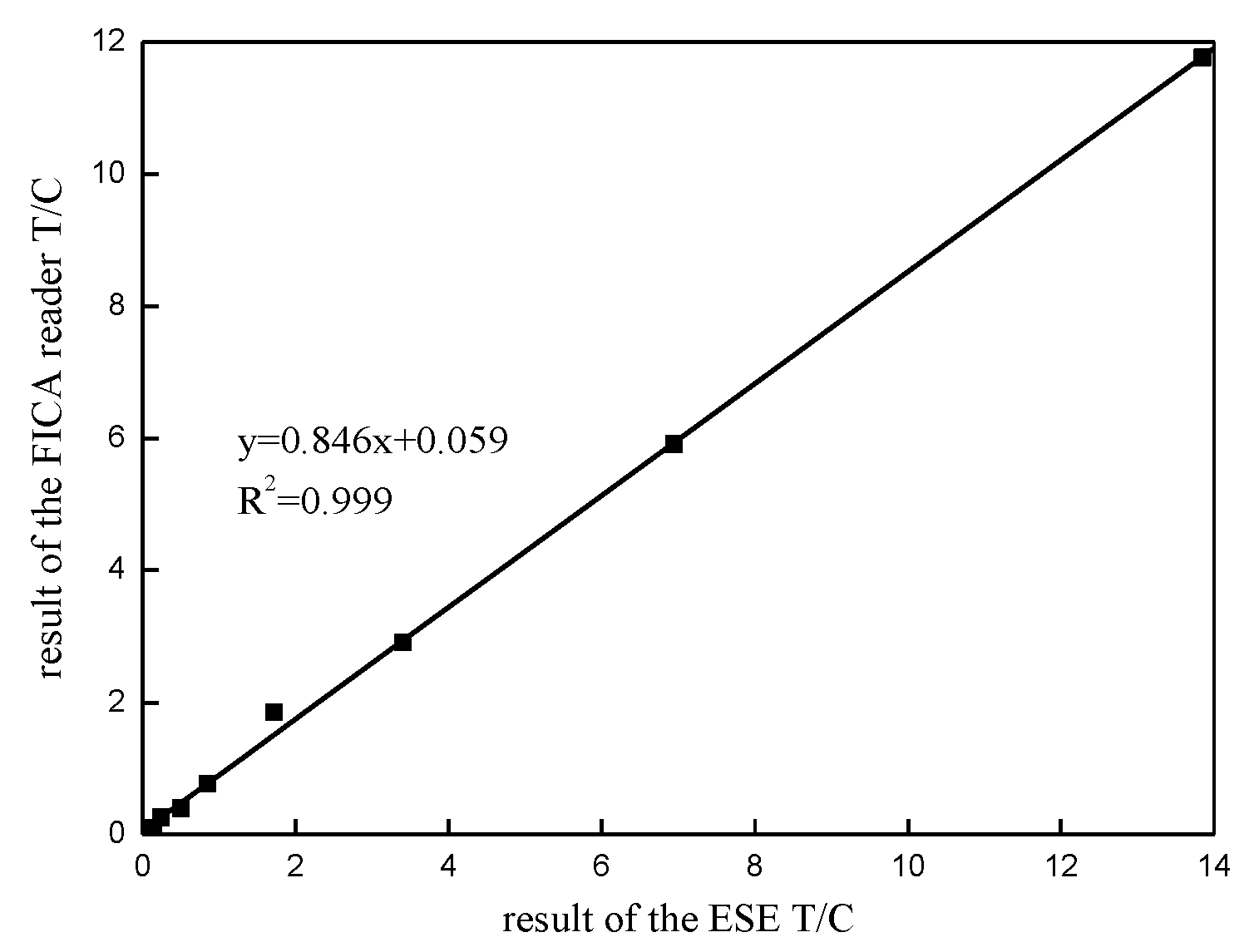
3.4. Accuracy
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Kim, T.K.; Oh, S.W.; Hong, S.C.; Mok, Y.J.; Choi, E.Y. Point-of-care fluorescence immunoassay for cardiac panel biomarkers. J. Clin. Lab. Anal. 2014, 28, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.H.; Zhou, X.H.; Wilkinson, J.S.; Hua, P.; Song, B.D.; Shi, H.C. Integrated optical waveguide-based fluorescent immunosensor for fast and sensitive detection of microcystin-LR in lakes: Optimization and Analysis. Sci. Rep. 2017, 7, 3655. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.W.; Chen, L.Y.; Liang, M.F.; Xu, H.; Tang, S.R.; Yang, H.H.; Song, H.B. Sensitive fluorescence immunoassay of alpha-fetoprotein through copper ions modulated growth of quantum dots in-situ. Sens. Actuator B Chem. 2017, 247, 408–413. [Google Scholar] [CrossRef]
- Seo, S.M.; Kim, S.W.; Park, J.N.; Cho, J.H.; Kim, H.S.; Paek, S.H. A fluorescent immunosensor for high-sensitivity cardiac troponin I using a spatially-controlled polymeric, nano-scale tracer to prevent quenching. Biosens. Bioelectron. 2016, 83, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.L.; Su, J.; Zhang, J.; Wei, X.T.; Guo, A.L. CdTe/CdS quantum dot-labeled fluorescent immunochromatography test strips for rapid detection of Escherichia coli O157: H7. RSC Adv. 2017, 7, 17819–17823. [Google Scholar] [CrossRef]
- Guo, H.L.; Zhou, X.H.; Zhang, Y.; Song, B.D.; Zhang, J.X.; Shi, H.C. Highly sensitive and simultaneous detection of melamine and aflatoxin M1 in milk products by multiplexed planar waveguide fluorescence immunosensor (MPWFI). Food Chem. 2016, 197, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.L.; Zhou, X.H.; Zhang, Y.; Song, B.D.; Liu, L.H.; Zhang, J.X.; Shi, H.C. Highly sensitive and rapid detection of melamine in milk products by planar waveguide fluorescence immunosensor (PWFI). Sens. Actuator B Chem. 2014, 194, 114–119. [Google Scholar] [CrossRef]
- Zhou, X.H.; Liu, L.H.; Xu, W.Q.; Song, B.D.; Sheng, J.W.; He, M.; Shi, H.C. A reusable evanescent wave immunosensor for highly sensitive detection of bisphenol A in water samples. Sci. Rep. 2014, 4, 4572. [Google Scholar]
- Tanaka, R.; Yuhi, T.; Nagatani, N.; Endo, T.; Kerman, K.; Takamura, Y.; Tamiya, E. A novel enhancement assay for immunochromatographic test strips using gold nanoparticles. Anal. Bioanal. Chem. 2006, 385, 1414–1420. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Wang, Y.; Wang, J.; Tang, Z.W.; Pounds, J.G.; Lin, Y.H. Rapid and Sensitive Detection of Protein Biomarker Using a Portable Fluorescence Biosensor Based on Quantum Dots and a Lateral Flow Test Strip. Anal. Chem. 2010, 82, 7008–7014. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.H.; Gong, X.Q.; Song, T.; Yang, J.M.; Zhu, S.J.; Li, Y.H.; Cui, Y.; Li, Y.X.; Zhang, B.B.; Chang, J. Quantum dot-based immunochromatography test strip for rapid, quantitative and sensitive detection of alpha fetoprotein. Biosens. Bioelectron. 2011, 30, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Choi, E.Y.; Kim, D.J.; Kim, J.H.; Kim, T.S.; Oh, S.W. A rapid, simple measurement of human albumin in whole blood using a fluorescence immunoassay (I). Clin. Chim. Acta 2004, 339, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Bai, Z.H.; Luo, Y.B.; Xu, W.T.; Gao, H.F.; Han, P.; Liu, T.; Wang, H.; Chen, A.L.; Huang, K.L. Development of a new fluorescence immunochromatography strip for detection of chloramphenicol residues in chicken muscles. J. Sci. Food Agric. 2013, 93, 3743–3747. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.W.; Zhang, H.; Liu, X.Y.; Trofimchuk, E.; Feng, S.L.; Ma, T.; Gao, X.; Fang, S.B.; Lu, X.N. Advantage of Eu3+-Doped Polystyrene Microspheres Compared with Colloidal Gold Used in Immunochromatographic Assays for the Detection of Melamine in Milk. J. Food Sci. 2017, 82, 694–697. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Huang, H.; Lin, C.; Wei, M.; Du, M.; Pun, S. Research of Range Adjustment for Fluorescent Immuno-Chromatographic Strip Analyzer. Chin. J. Biomed. Eng. 2017, 36, 124–128. [Google Scholar]
- Larsson, M.; Favilla, R.; Stromberg, T. Assessment of advanced glycated end product accumulation in skin using auto fluorescence multispectral imaging. Comput. Biol. Med. 2017, 85, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Hernot, S.; van Manen, L.; Debie, P.; Mieog, J.S.D.; Vahrmeijer, A.L. Latest developments in molecular tracers for fluorescence image-guided cancer surgery. Lancet Oncol. 2019, 20, E354–E367. [Google Scholar] [CrossRef]
- Oh, S.W.; Moon, J.D.; Park, S.Y.; Jang, H.J.; Kim, J.H.; Nahm, K.B.; Choi, E.Y. Evaluation of fluorescence hs-CRP immunoassay for point-of-care testing. Clin. Chim. Acta 2005, 356, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Yu, W.; Zhu, D. An Improved Image Segmentation Algorithm Based on the Otsu Method. In Proceedings of the 13th ACIS International Conference on Software Engineering, Artificial Intelligence, Networking and Parallel/Distributed Computing, Kyoto, Japan, 8–10 August 2012; pp. 135–139. [Google Scholar]
- Ge, Z.W.; YAO, S.Y.; Xu, J.T.; Su, X.H. A Fast Automatic Exposure Control Method for CMOS Image Sensor. J. Tianjin Univ. 2010, 43, 854–859. [Google Scholar]
- Kuang, C.F.; Luo, D.; Liu, X.; Wang, G.R. Study on factors enhancing photobleaching effect of fluorescent dye. Measurement 2013, 46, 1393–1398. [Google Scholar] [CrossRef]


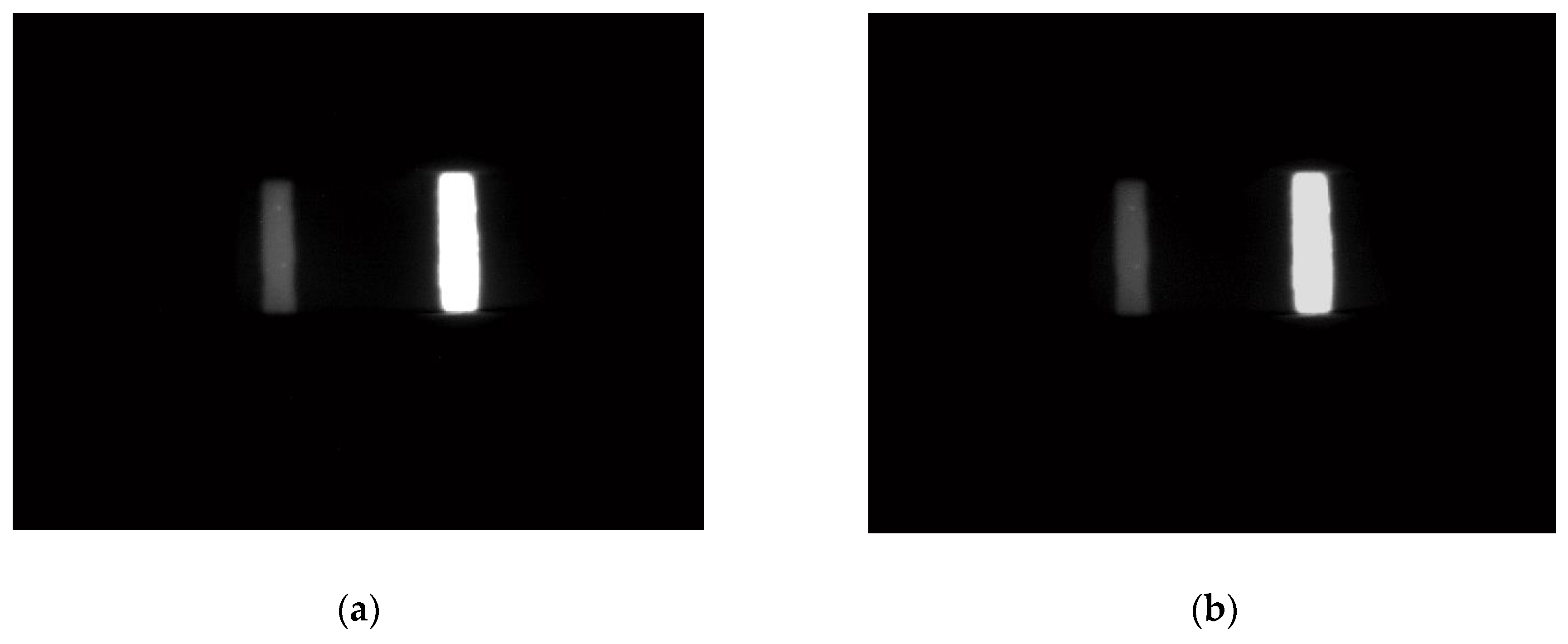
| CRP (μg/mL) | ESEQuant LFR | FICA Reader without Adjustment | FICA Reader with Adjustment |
|---|---|---|---|
| 0.98 | — | — | 0.03 |
| 1.95 | 0.05 | 0.05 | 0.05 |
| 3.91 | 0.14 | 0.10 | 0.11 |
| 7.81 | 0.24 | 0.25 | 0.26 |
| 15.6 | 0.5 | 0.41 | 0.40 |
| 31.25 | 0.85 | 0.75 | 0.77 |
| 62.5 | — | 1.84 | 1.85 |
| 125 | — | 2.92 | 2.91 |
| 256 | — | — | 5.92 |
| 512 | — | — | 11.76 |
| 1024 | — | — | — |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, R.; Wu, H.; Yang, J.; Jiang, H.; Du, M.; Vai, M.; Pun, S.; Gao, Y. Automatic Range Adjustment of the Fluorescence Immunochromatographic Assay Based on Image Processing. Sensors 2020, 20, 209. https://doi.org/10.3390/s20010209
Jiang R, Wu H, Yang J, Jiang H, Du M, Vai M, Pun S, Gao Y. Automatic Range Adjustment of the Fluorescence Immunochromatographic Assay Based on Image Processing. Sensors. 2020; 20(1):209. https://doi.org/10.3390/s20010209
Chicago/Turabian StyleJiang, Ruixin, Huihuang Wu, Jianpeng Yang, Haiyan Jiang, Min Du, Mangi Vai, Siohang Pun, and Yueming Gao. 2020. "Automatic Range Adjustment of the Fluorescence Immunochromatographic Assay Based on Image Processing" Sensors 20, no. 1: 209. https://doi.org/10.3390/s20010209
APA StyleJiang, R., Wu, H., Yang, J., Jiang, H., Du, M., Vai, M., Pun, S., & Gao, Y. (2020). Automatic Range Adjustment of the Fluorescence Immunochromatographic Assay Based on Image Processing. Sensors, 20(1), 209. https://doi.org/10.3390/s20010209




