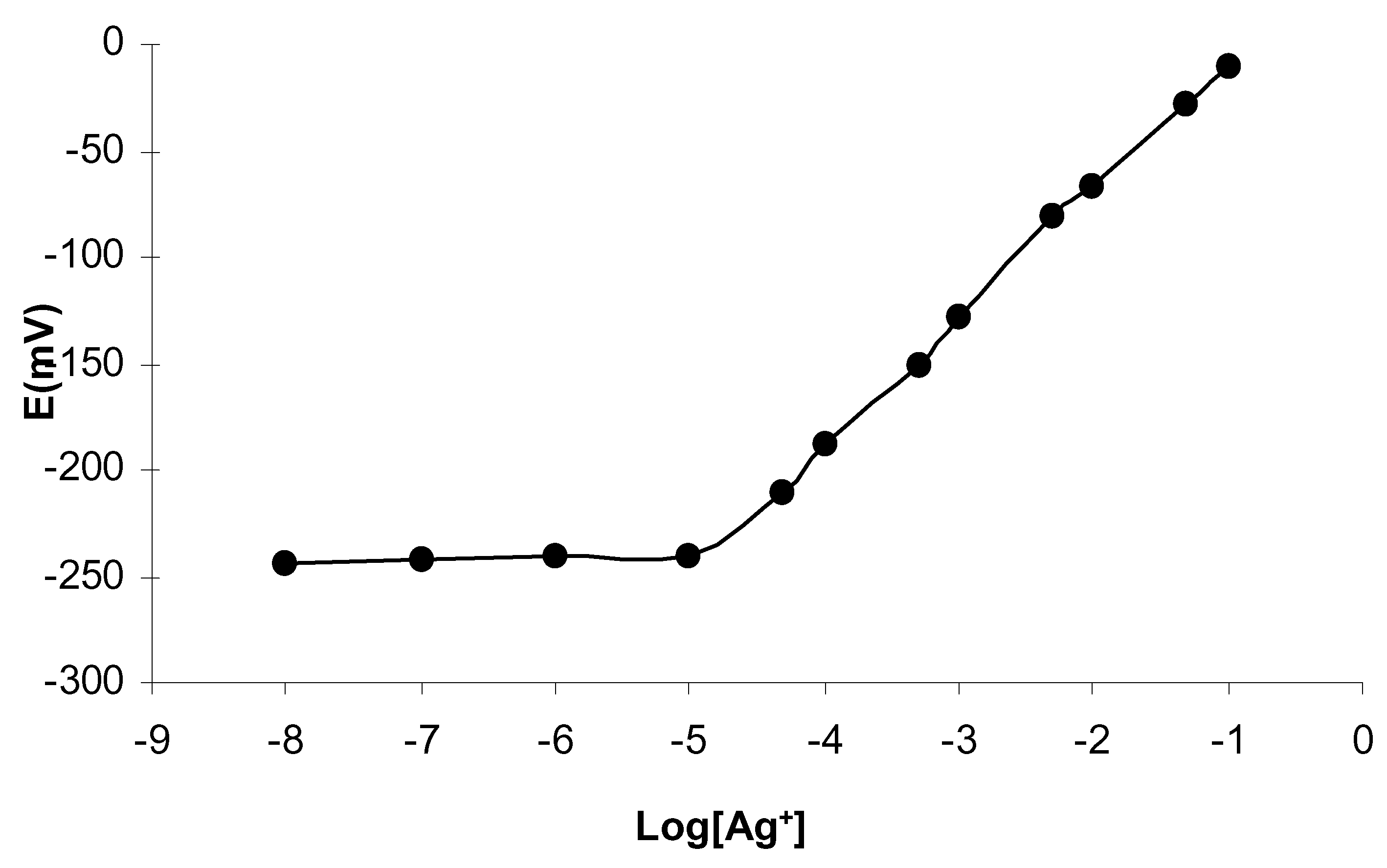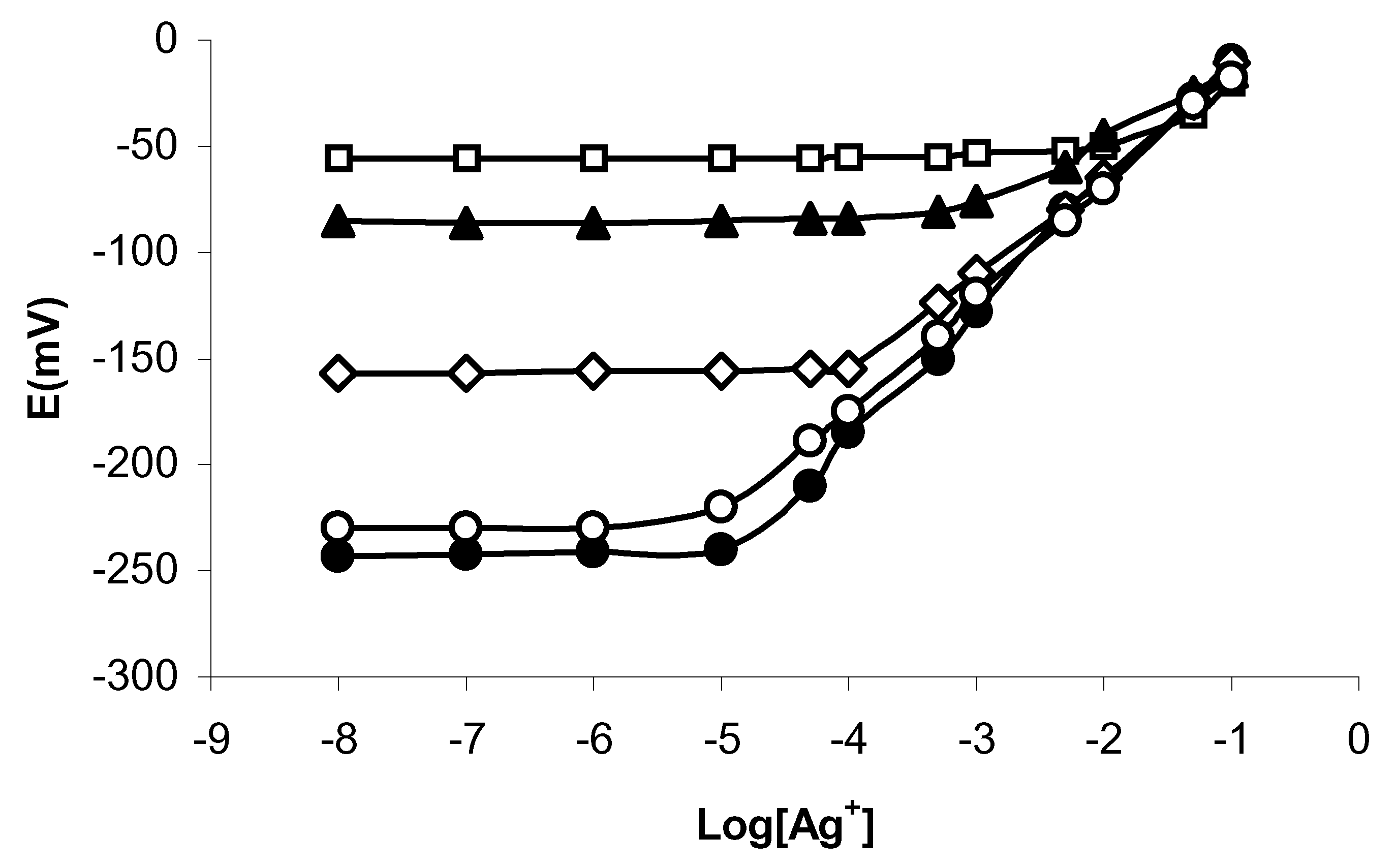Introduction
Amongst the various analytical techniques available, the use of ion-selective membrane electrodes is well establish routine analytical technique. Good ion-selective electrodes(ISEs) possess many advantages over the traditional methods of analysis as they provide accurate, reproducible, fast and often selective determination of various ionic species. Not only this, the ion-selective electrodes(ISEs) allow non-destructive, on line monitoring of particular ion in small volume of sample without any pretreatment. Because of these merits, the use of ISEs is increasing day by day in medicinal, enviornmental, agricultural and industrial field. During last three decades, a number of ion-selective electrodes with polymeric membranes have been reported[
1,
2,
3,
4,
5,
6,
7,
8,
9,
10]. Among the various ligands available for ion-selective electrodes such as crowns, podands, cryptands and spherands, the calixarenes met many of the requirements that an ionophore should satisfy for the use in ion-selective electrodes[
11]. A number of calixarene derivatives containing pendant ether, amide, ketonic and ester groups have been incorporated as neutral carriers into ion-selective electrodes sensitive to sodium, potassium and cesium ions[
12,
13,
14,
15]. Recently,we reported silver(I) and cesium ion-selective PVC membrane electrode based on bis(pyridine tetramide) macrocycle and calix crown ether ester,respectively[
2,
3,
10].The present paper deals with Schiff-base-p-tert-butylcalix[4]arene derivative [
I] based ISE for selective determination of silver. The silver ion selectivity may be due to electrostatic interaction between the metal ion and aza crown cavity composed of four highly preorganized soft binding units (imine units).
The silver(I) ion selective electrode exhibit a good response for silver ion over wide concentration range of 1.0×10
-5-1.0×10
-1M with a Nernstian slope of 58.9mV per decade. The proposed ion selective electrode incorporates PVC as supporting material to give a pseudo-solid sensing membrane and bis(2-ethylhexyl)sebacate as plasticizing agent. The performance of this electrode which involves linear range, detection limit, response time, slope etc is in agreement and in many respects better than those reported in literature [
8,
9].
Results and Discussion
Schiff-base-p-tert-butylcalix[4]arene derivative
[I] was employed as silver selective ionophore in the preparation of silver ion- selective electrode. The potentiometric response of the electrode is shown in
Fig 1. The measurements were performed in the concentration range of 1.0×10
-8-1.0×10
-1M AgNO
3. The electrode shows linear response in the concentration range from1.0×10
-5to1.0×10
-1M. The detection limit obtained from the intersection of the extrapolated linear regions in the plot, is 6.31×10
-6 M. The sensor shows nearly Nerntian slope 58.7mV per decade. The response time of the sensor is 30 seconds which is quite short. In terms of lifetime of the PVC membrane electrodes, their response stability was observed for over more than six months.
Figure 1.
Potential responses of silver ion sensors based on ionophore[I].
Figure 1.
Potential responses of silver ion sensors based on ionophore[I].
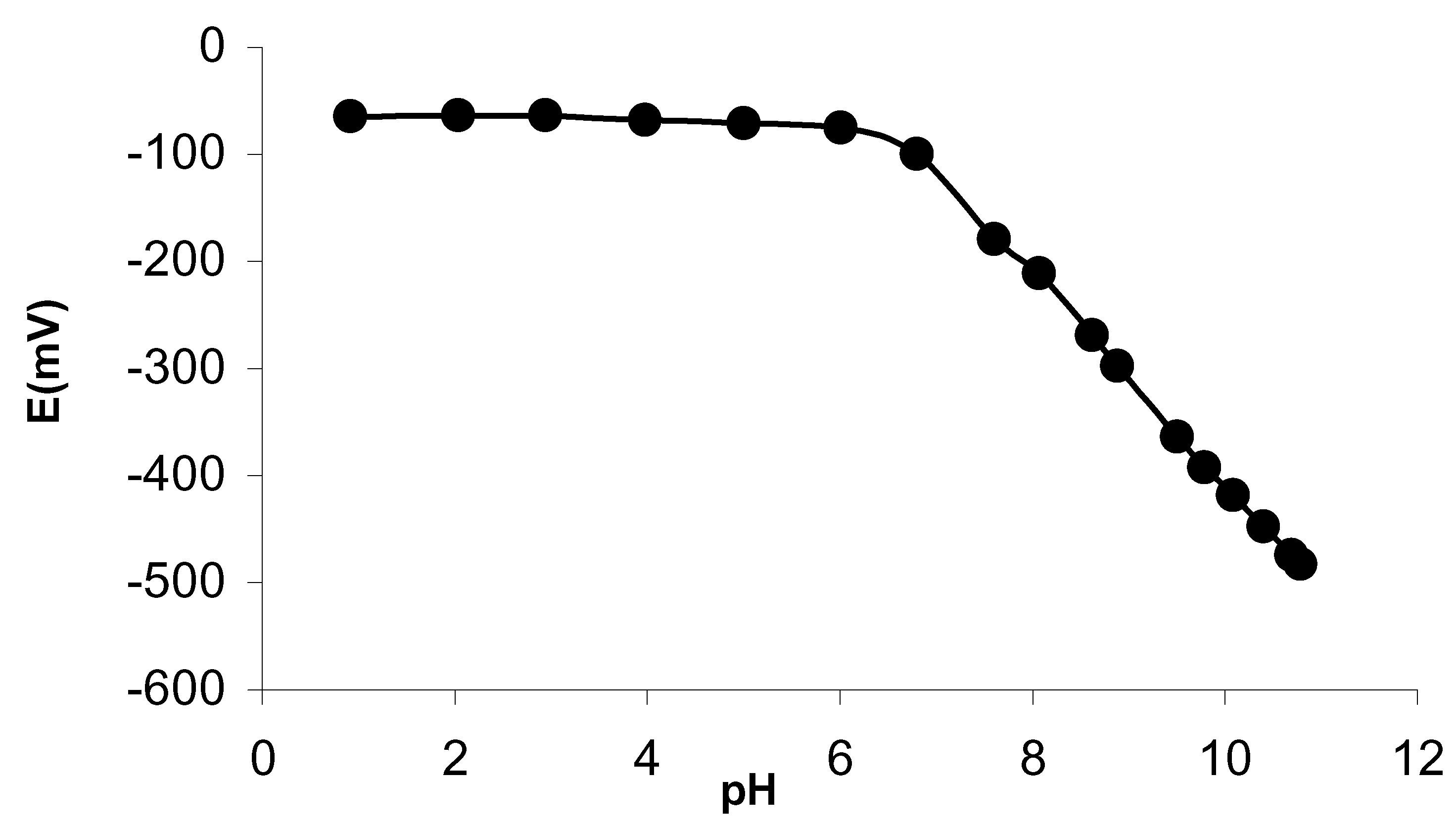
The pH dependance of the membrane potential was also examined at 1.0×10
-2M silver(I) concentration. As shown in
Fig. 2, the membrane electrode shows good stability in relation to pH changes over wide pH range i.e 1.0 -6.0, which may be taken as the functional pH range of the sensor. In this plot, the high variation of the potentiometric response corresponds to pH limit.This points towards the applicability of the developed electrode in the moderately acidic media. As the pH of silver nitrate solutions lies in the functional range of the sensor, do not need any pH adjustment.
Figure 2.
Plot showing the variation of membrane potential with pH at 1.0×10-2 M Ag+ ions.
Figure 2.
Plot showing the variation of membrane potential with pH at 1.0×10-2 M Ag+ ions.
Selectivity is perhaps the single most important characteristic of any sensor which defines the nature of device and the extent to which it may be employed in the determination of a particular ion in the presence of other interfering ions. This is measured in terms of potentiometric selectivity coefficients (log K
PotAg,M) which has been evaluated using fixed interference method at 1.0x10
-2M concentration of interfering ions. The potentiometric selectivity coefficients (logK
PotAg,M) measures the response of the electrode for the primary ion in the presence of foreign ions.
Table 1 shows potentiometric selectivity coefficient data of Schiff-base–p-tert-butyl calix[4]arene based silver ion- selective electrode for interfering cations relative to Ag
+. The selectivity coefficients data indicate that K
PotAg,M values are of the order of 10
-3 for monovalent and for divalent metal ions (except for the mercury(II)ions) the values are of the order of 10
-4 and 10
-5. Therefore, the electrode can be used for the determination of silver ions in the presence of interfering ions.
These results demonstrated that Schiff-base-p-tert-butylcalix[4]arene[
I] acted primarily as a Ag
+ selective ionophore and avoided interference from other cations when present at high concentration of 1.0×10
-2M. Even the strongest interferent Hg
2+ does not disturb the performance of sensor when present at the concentration less than 1.0×10
-5M (
Fig. 3).
In a comparison with PVC based Ag
+ ion sensor having calix[4]arene derivatives with functional hetrocyclic group such as pyridyl[
9] as ionophore, it is interesting to note that the interference from Hg
2+ is much prominent in normal performance of the sensor, Log K
PotAg,Hg = -1.80. Further, the slope and linear range, detection limit of the proposed sensor are comparable and better in many respects than that sensor. From another comparison with Ag
+ ion selective electrode incorporating bis(3- pyridinecarboxylate)calix[4]arene as an ionophore[
8] in which the Hg
2+ act as an strongest interferent with Log K
PotAg,Hg >+1.0.,we find the sensitivity and selectivity of the Ag
+ selective electrode develops in this study are better than those of previous electrodes.
Table 1.
The selectivity coefficients of diverse ions.
Table 1.
The selectivity coefficients of diverse ions.
| Diverse ions | KPotAg,M | Log KPotAg,M |
|---|
| K+ | 2.24×10-3 | -2.65 |
| Na+ | 4.47×10-3 | -2.35 |
| NH4+ | 4.47×10-3 | -2.35 |
| Mg2+ | 2.69×10-5 | -4.57 |
| Ca2+ | 3.80×10-4 | -3.42 |
| Ba2+ | 7.94×10-5 | -4.10 |
| Ni2+ | 5.01×10-5 | -4.30 |
| Co2+ | 2.51×10-4 | -3.60 |
| Cu2+ | 1.59×10-4 | -3.80 |
| Pb2+ | 3.55×10-4 | -3.54 |
| Hg2+ | 1.0×10-2 | -2.00 |
| Zn2+ | 1.51×10-4 | -3.82 |
| Cd2+ | 3.80×10-4 | -3.42 |
Figure 3.
Plots showing E vs Log [Ag+] in the presence of Hg2+ ions at varying level of interference ( )1.0×10-2 , (∏)1.0×10-3, (⊄) 1.0×10-4 (;)1.0×10-5 and (•) 0.0 M.
Figure 3.
Plots showing E vs Log [Ag+] in the presence of Hg2+ ions at varying level of interference ( )1.0×10-2 , (∏)1.0×10-3, (⊄) 1.0×10-4 (;)1.0×10-5 and (•) 0.0 M.
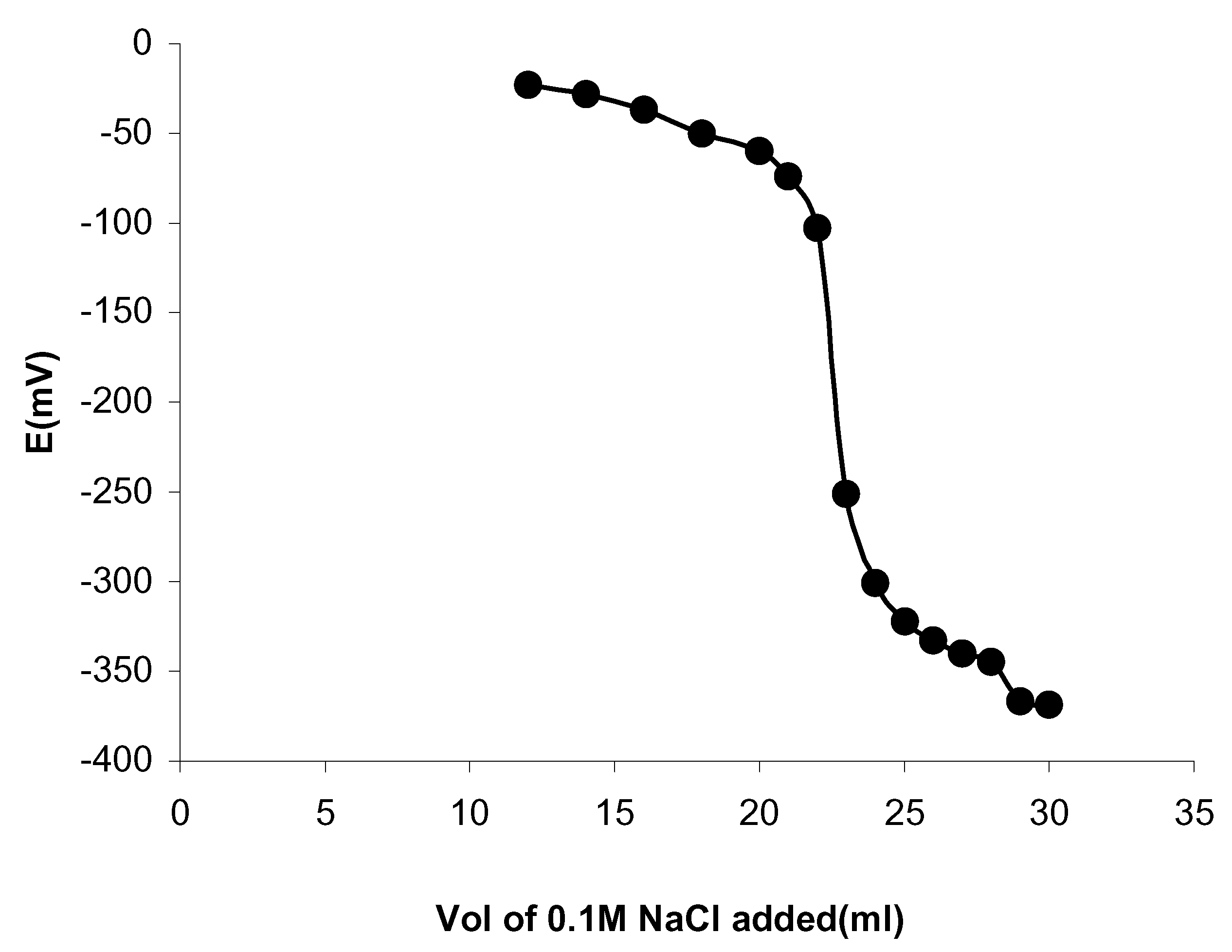
The proposed sensor works well under laboratory conditions and can be successfully employed in the potentiometric tritrations of Ag
+ solutions with sodium chloride and the resulting titration curve is shown in
Fig. 4. The electrode assembly can also used to determine chloride ion concentration in the tap water by potentiometric titration.
Figure 4.
Plot showing the potentiometric titration of 1.0×10-1M of AgNO3 with 1.0×10-1M NaCl solution.
Figure 4.
Plot showing the potentiometric titration of 1.0×10-1M of AgNO3 with 1.0×10-1M NaCl solution.
Table 2.
Analysis of water sample spiked with silver(I).
Table 2.
Analysis of water sample spiked with silver(I).
| Sample No. | [Ag+]AAS(ppm)/ SD(n=4) | [Ag+]ISE(ppm)/ SD(n= 4) |
|---|
| 1 | 40/ 0.3 | 39.5/ 0.2 |
| 2 | 29/ 0.4 | 29.1/ 0.3 |
| 3 | 19.5/ 0.3 | 19.3/ 0.3 |
The applicability of the sensor is illustrated by measuring the silver(I) ion potentiometrically in Doubly distilled deionised water (DDW) spiked with 40, 30 and 20ppm silver(I). The results obtained were compared with those obtained from atomic absorption spectrometric (AAS) analysis and were found in good agreement (
Table 2).