Characteristics of an Implantable Blood Pressure Sensor Packaged by Ultrafast Laser Microwelding
Abstract
1. Introduction
2. Experiments
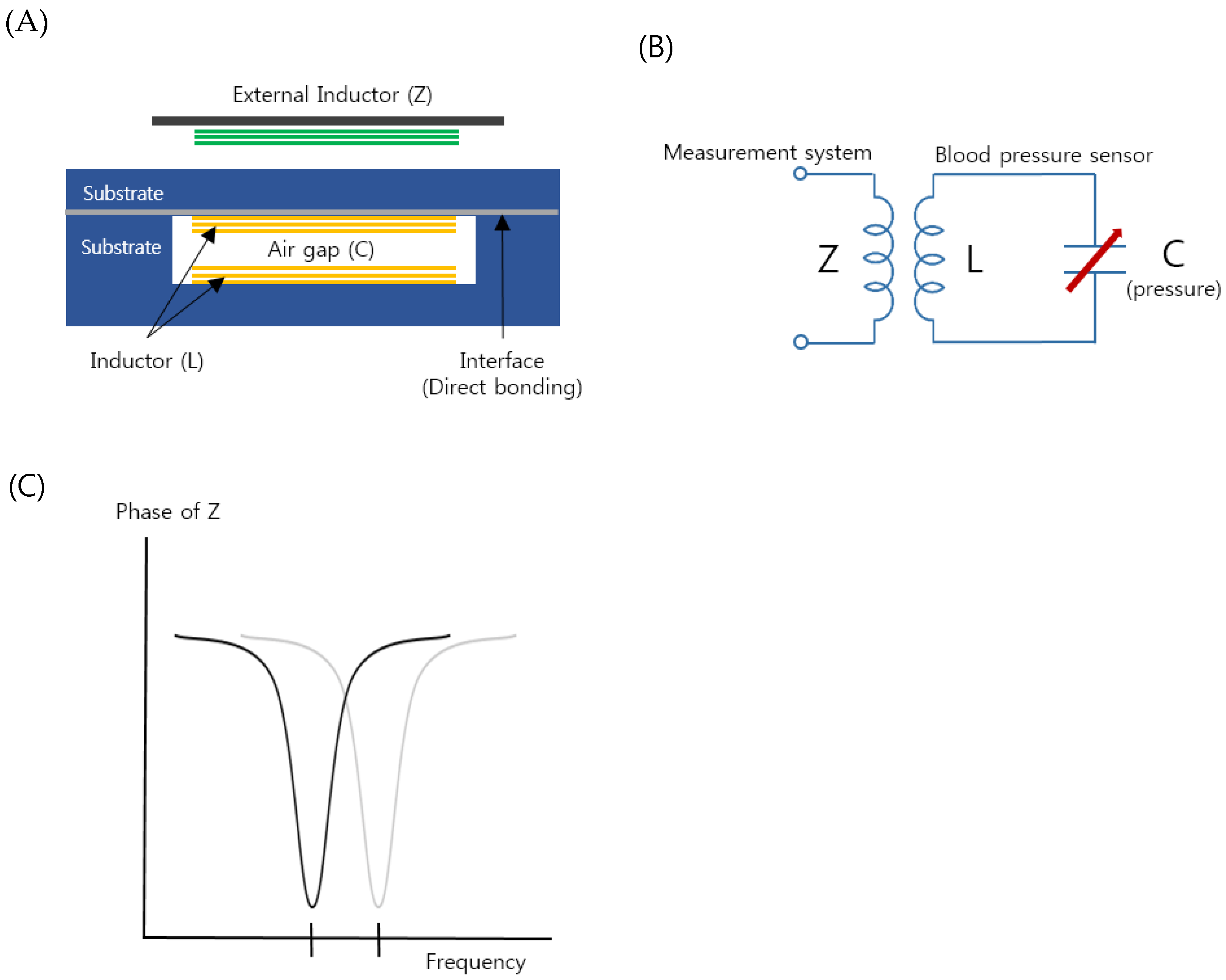
2.1. Principle and Fabrication of Implantable Blood Pressure Sensor
2.2. Optimization of Micro Welding for Implantable Blood Pressure Sensor Packaging
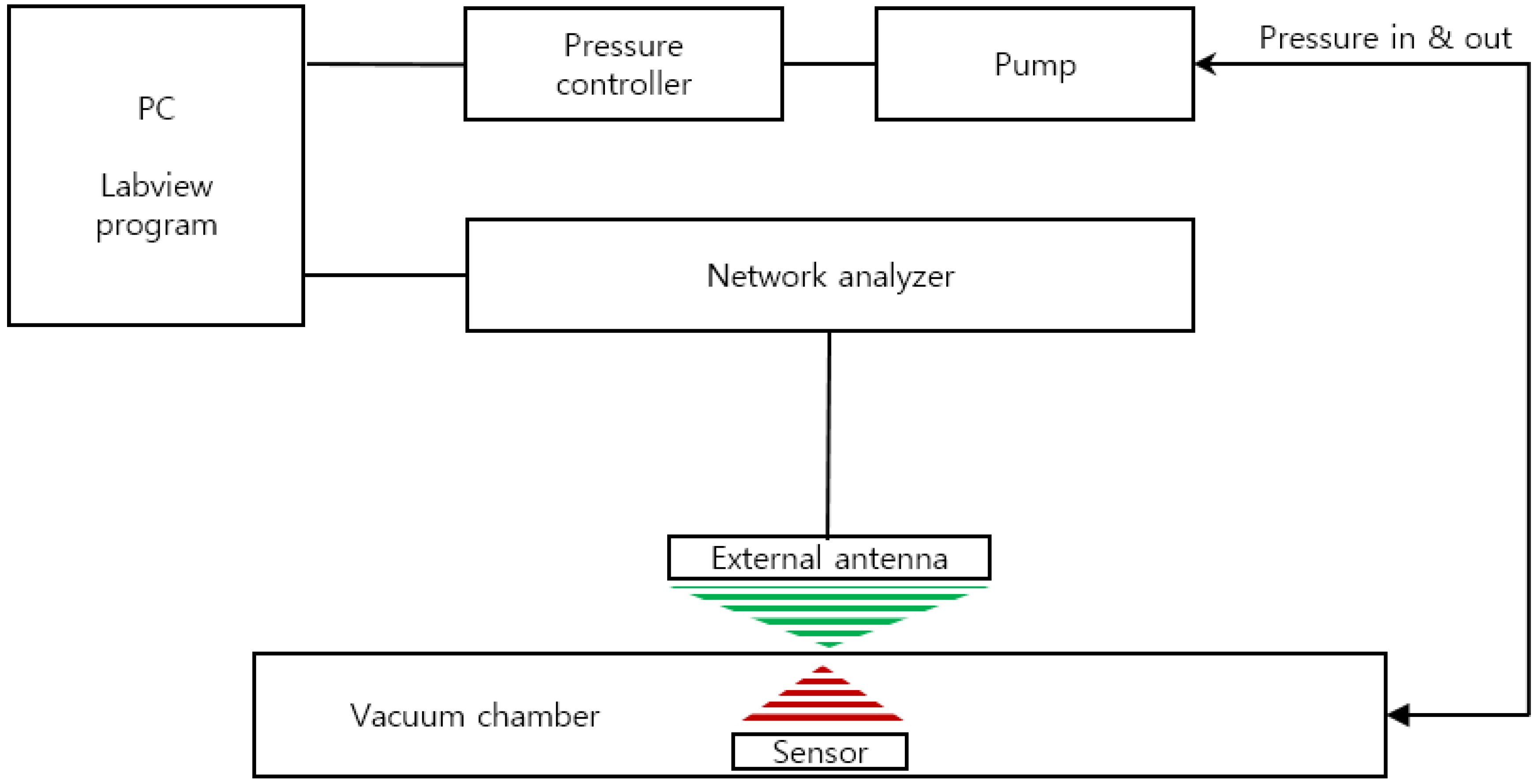
2.3. Evaluation of Improved Packaging
3. Results and Discussion
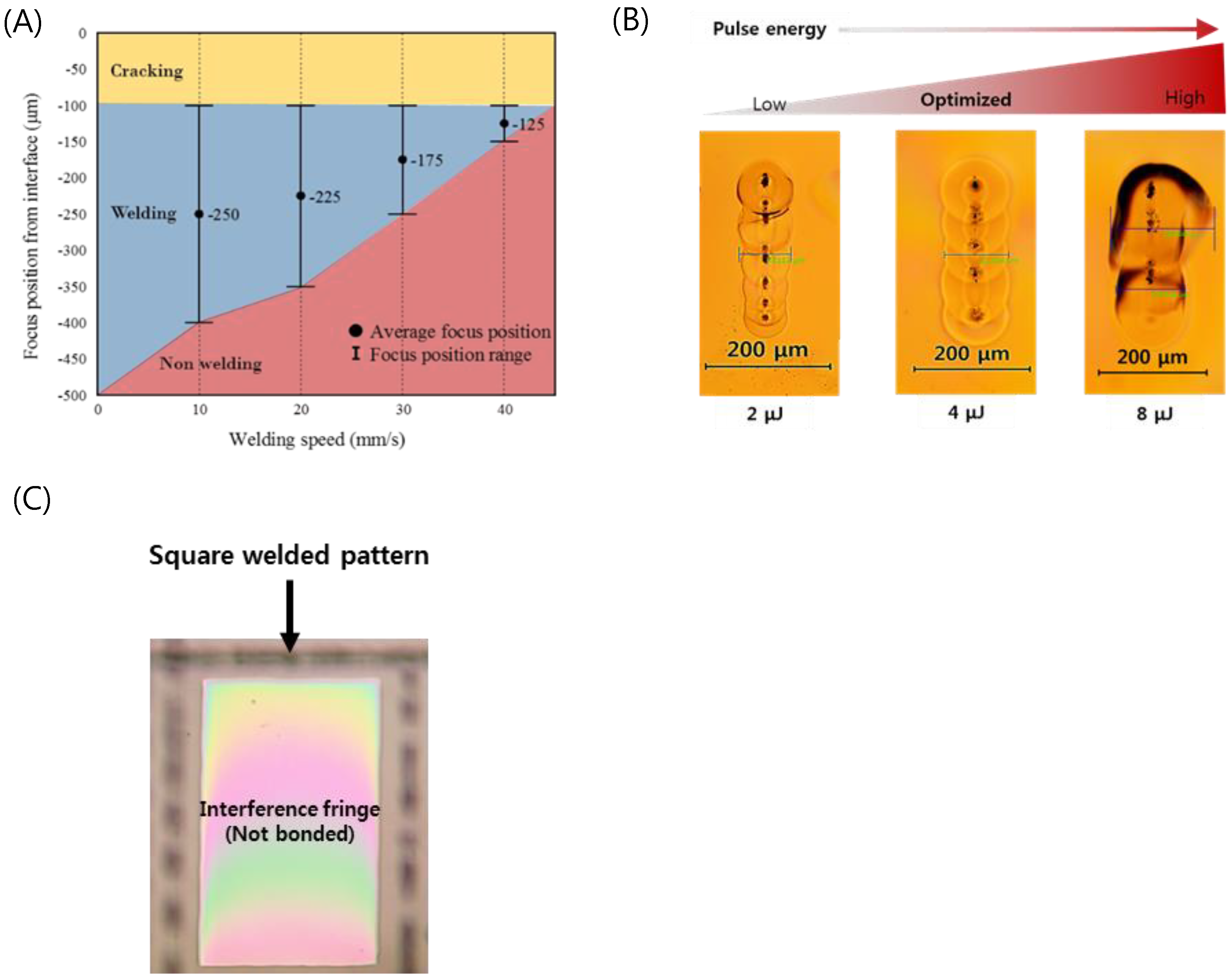
3.1. Optimized Laser Conditions for Quartz Wafer Welding
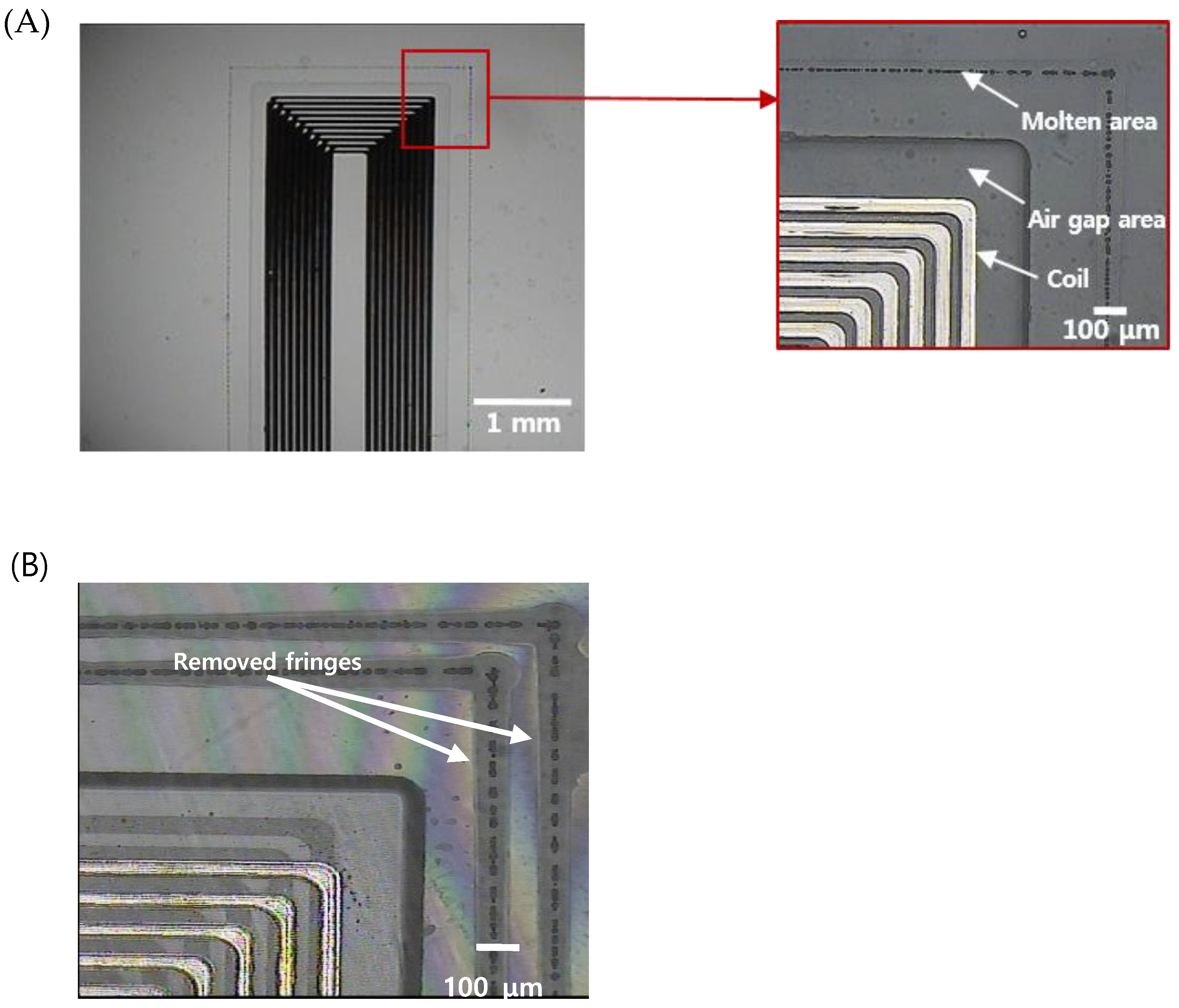
3.2. Implantable Blood Pressure Sensor Hermetic Packaging
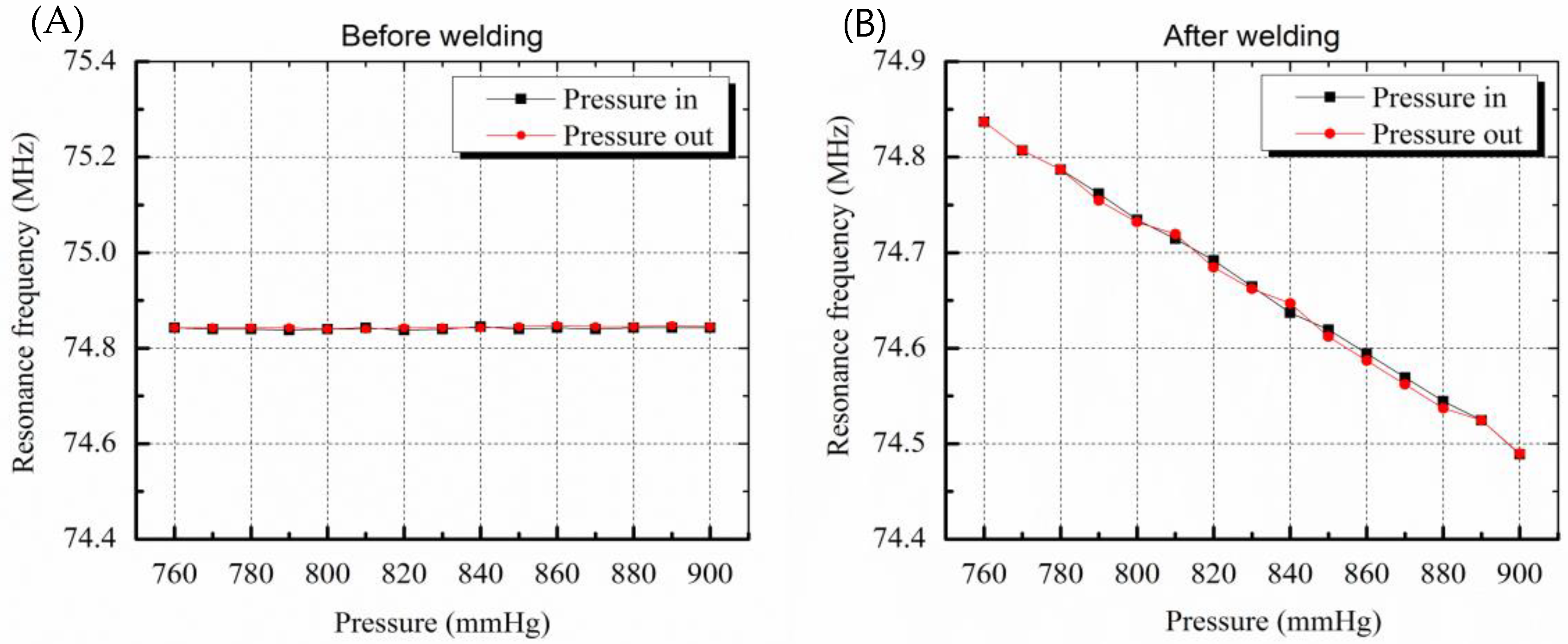
3.3. Chracterization of the Implantable Blood Pressure Sensor after Welding
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Carey, R.M.; Calhoun, D.A.; Bakris, G.L.; Brook, R.D.; Daugherty, S.L.; Dennison-Himmelfarb, C.R.; Lackland, D.T. Resistant hypertension: Detection, evaluation, and management: A scientific statement from the American Heart Association. Hypertension 2018, 72, 53–90. [Google Scholar] [CrossRef] [PubMed]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Collins, K.J.; Himmelfarb, C.D.; MacLaughlin, E.J. ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2018, 71, 127–248. [Google Scholar]
- Pickering, T.G.; Hall, J.E.; Appel, L.J.; Falkner, B.E.; Graves, J.; Hill, M.N.; Roccella, E.J. Recommendations for blood pressure measurement in humans and experimental an imals: Part 1: Blood pressure measurement in humans: A statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Circulation 2005, 111, 697–716. [Google Scholar] [PubMed]
- Yu, L.; Kim, B.; Meng, E. Chronically implanted pressure sensors: Challenges and state of the field. Sensors 2014, 14, 20620–20644. [Google Scholar] [CrossRef] [PubMed]
- Springer, F.; Günther, R.W.; Schmitz-Rode, T. Aneurysm sac pressure measurement with minimally invasive implantable pressure sensors: An alternative to current surveillance regimes after EVAR? Cardiovasc. Interv. Radiol. 2008, 31, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Van der Wel, M.C.; Buunk, I.E.; Van Weel, C.; Thien, T.A.; Bakx, J.C. A novel approach to office blood pressure measurement: 30-minute office blood pressure vs daytime ambulatory blood pressure. Ann. Fam. Med. 2011, 9, 128–135. [Google Scholar] [CrossRef] [PubMed]
- We Don’t Just Need Precision Medicine, We Need Precision Health. Available online: https://www.forbes.com/sites/valleyvoices/2016/01/06/we-dont-just-need-precision-medicine-we-need-precision-health/ (accessed on 6 January 2016).
- Weber, M.J.; Yoshihara, Y.; Sawaby, A.; Charthad, J.; Chang, T.C.; Arbabian, A. A Miniaturized Single-Transducer Implantable Pressure Sensor with Time-Multiplexed Ultrasonic Data and Power Links. IEEE J. Solid-State Circuits 2018, 53, 1089–1101. [Google Scholar] [CrossRef]
- Bartholome, W.G. A revolution in understanding: how ethics has transformed health care decision making. QRB. Qual. Rev. Bull. 1992, 18, 6–11. [Google Scholar] [CrossRef]
- Allen, M.G. Micromachined endovascularly-implantable wireless aneurysm pressure sensors: From concept to clinic. In Proceedings of the 13th International Conference on Solid-state Sensors, Actuators and Microsystems, Seoul, Korea, 5–9 June 2005. [Google Scholar]
- Fonseca, M.A.; Allen, M.G.; Kroh, J.; White, J. Flexible wireless passive pressure sensors for biomedical applications. In Proceedings of the Solid-State Sensor, Actuator, and Microsystems Workshop, Hilton Head Island, SC, USA, 4–8 June 2006; pp. 37–42. [Google Scholar]
- Fonseca, M.A.; English, J.M.; Von Arx, M.; Allen, M.G. Wireless micromachined ceramic pressure sensor for high-temperature applications. J. Microelectromech. Syst. 2002, 11, 337–343. [Google Scholar] [CrossRef]
- Fassbender, H.; Mokwa, W.; Gortz, M.; Trieu, K.; Urban, U.; Schmitz-Rode, T.; Osypka, P. Fully implantable blood pressure sensor for hypertonic patients. In Proceedings of the IEEE Sensors, Lecce, Italy, 26–29 October 2008; pp. 1226–1229. [Google Scholar]
- Murphy, O.H.; Bahmanyar, M.R.; Borghi, A.; McLeod, C.N.; Navaratnarajah, M.; Yacoub, M.H.; Toumazou, C. Continuous in vivo blood pressure measurements using a fully implantable wireless SAW sensor. Biomed. Microdevices 2013, 15, 737–749. [Google Scholar] [CrossRef]
- Cong, P.; Chaimanonart, N.; Ko, W.H.; Young, D.J. A wireless and batteryless 10-bit implantable blood pressure sensing microsystem with adaptive RF powering for real-time laboratory mice monitoring. IEEE J. Solid-State Circuits 2009, 44, 3631–3644. [Google Scholar] [CrossRef]
- Chow, E.Y.; Chlebowski, A.L.; Chakraborty, S.; Chappell, W.J.; Irazoqui, P.P. Fully wireless implantable cardiovascular pressure monitor integrated with a medical stent. IEEE Trans. Biomed. Eng. 2010, 57, 1487–1496. [Google Scholar] [CrossRef]
- Joung, Y.H. Development of implantable medical devices: From an engineering perspective. Int. Neurourol. J. 2013, 17, 98. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.T.; Kim, S.I.; Joung, Y.H. Design and Fabrication of Implantable LC Resonant Blood Pressure Sensor. J. Korean Inst. Electr. Electron. Mater. Eng. 2013, 26, 171–176. [Google Scholar] [CrossRef][Green Version]
- Kim, S.I.; Kim, E.B.; So, S.K.; Choi, J.; Joung, Y.H. Development of Implantable Blood Pressure Sensor Using Quartz Wafer Direct Bonding and Ultrafast Laser Cutting. J. Biome. Eng. Res. 2016, 37, 168–177. [Google Scholar] [CrossRef]
- Jacobson, B.; Nordberg, L. Endoradiosondes for pressure telemetering. IRE Trans. Bio-Med. Electron. 1961, 8, 192–196. [Google Scholar] [CrossRef]
- Olsen, E.R.; Collins, C.C.; Loughborough, W.F.; Richards, V.; Adams, J.E.; Pinto, D.W. Intracranial pressure measurement with a miniature passive implanted pressure transensor. Am. J. Surg. 1967, 113, 727–729. [Google Scholar] [CrossRef]
- Yang, C.; Zhao, C.; Wold, L.; Kaufman, K.R. Biocompatibility of a physiological pressure sensor. Biosens. Bioelectron. 2003, 19, 51–58. [Google Scholar] [CrossRef]
- Lin, P.-H.; Huang, S.-C.; Chen, K.-P.; Li, B.-R.; Li, Y.-K. Effective Construction of a High-Capacity Boronic Acid Layer on a Quartz Crystal Microbalance Chip for High-Density Antibody Immobilization. Sensors 2019, 19, 28. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, J.; Joung, Y.-H.; Choi, J.; Koo, C. Bonding Strength of a Glass Microfluidic Device Fabricated by Femtosecond Laser Micromachining and Direct Welding. Micromachines 2018, 9, 639. [Google Scholar] [CrossRef] [PubMed]
- Suratwala, T.I.; Miller, P.E.; Bude, J.D.; Steele, W.A.; Shen, N.; Monticelli, M.V.; Wong, L.L. HF-based etching processes for improving laser damage resistance of fused silica optical surfaces. J. Am. Ceram. Soc. 2011, 94, 416–428. [Google Scholar] [CrossRef]
- Kim, S.I.; Kim, J.; Koo, C.; Joung, Y.; Choi, J. Rapid prototyping of 2D glass microfluidic devices based on femtosecond laser assisted selective etching process. In Proceedings of the SPIE 10522, Frontiers in Ultrafast Optics: Biomedical, Scientific, and Industrial Applications XVIII, San Francisco, CA, USA, 19 February 2018. [Google Scholar]
- Wang, C.; Wang, Y.; Tian, Y.; Wang, C.; Suga, T. Room-temperature direct bonding of silicon and quartz glass wafers. Appl. Phys. Lett. 2017, 110, 221602. [Google Scholar] [CrossRef]
- Gong, Y.; Park, J.M.; Lim, J. An interference-assisted thermal bonding method for the fabrication of thermoplastic microfluidic devices. Micromachines 2016, 7, 211. [Google Scholar] [CrossRef] [PubMed]
- Heptonstall, A.; Barton, M.; Cantley, C.; Cumming, A.; Cagnoli, G.; Hough, J.; Torrie, C. Investigation of mechanical dissipation in CO2 laser-drawn fused silica fibres and welds. Classical Quantum Gravity 2010, 27, 035013. [Google Scholar] [CrossRef]
- Sugioka, K.; Cheng, Y. Ultrafast lasers—Reliable tools for advanced materials processing. Light Sci. Appl. 2014, 3, e149. [Google Scholar] [CrossRef]
- Shah, L.; Tawney, J.; Richardson, M.; Richardson, K. Femtosecond laser deep hole drilling of silicate glasses in air. Appl. Surf. Sci. 2001, 183, 151–164. [Google Scholar] [CrossRef]
- Tamaki, T.; Watanabe, W.; Nishii, J.; Itoh, K. Welding of transparent materials using femtosecond laser pulses. Jpn. J. Appl. Phys. 2005, 44, L687. [Google Scholar] [CrossRef]
- Tamaki, T.; Watanabe, W.; Itoh, K. Laser micro-welding of transparent materials by a localized heat accumulation effect using a femtosecond fiber laser at 1558 nm. Opt. Express 2006, 14, 10460–10468. [Google Scholar] [CrossRef]
- Miyamoto, I.; Cvecek, K.; Okamoto, Y.; Schmidt, M. Novel fusion welding technology of glass using ultrashort pulse lasers. Phys. Procedia 2010, 5, 483–493. [Google Scholar] [CrossRef][Green Version]
- Tan, H.; Duan, J.A. Welding of glasses in optical and partial-optical contact via focal position adjustment of femtosecond-laser pulses at moderately high repetition rate. Appl. Phys. A 2017, 123, 481. [Google Scholar] [CrossRef]
- Gstalter, M.; Chabrol, G.; Bahouka, A.; Dorkenoo, K.D.; Rehspringer, J.L.; Lecler, S. Optimum parameters for high-repetition rate femtosecond laser glass welding using an optical head with long focal length. In Proceedings of the SPIE 10683, Fiber Lasers and Glass Photonics: Materials through Applications, Strasbourg, France, 23 May 2018. [Google Scholar]
- Huang, H.; Yang, L.M.; Liu, J. Direct welding of fused silica with femtosecond fiber laser. In Proceedings of the SPIE 8244, Laser-based Micro- and Nanopackaging and Assembly VI, San Francisco, CA, USA, 17 February 2012. [Google Scholar]
- Miyamoto, I.; Cvecek, K.; Schmidt, M. Crack-free conditions in welding of glass by ultrashort laser pulse. Opt. Express 2013, 21, 14291–14302. [Google Scholar] [CrossRef] [PubMed]
- Cvecek, K.; Miyamoto, I.; Strauss, J.; Wolf, M.; Frick, T.; Schmidt, M. Sample preparation method for glass welding by ultrashort laser pulses yields higher seam strength. Appl. Opt. 2011, 50, 1941–1944. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.; Zimmermann, F.; Eberhardt, R.; Tunnermann, A.; Nolte, S. Toward laser welding of glasses without optical contacting. Appl. Phys. A 2015, 121, 1–9. [Google Scholar] [CrossRef]
- Okamoto, Y.; Miyamoto, I.; Cvecek, K.; Okada, A.; Takahashi, K.; Schmidt, M. Evaluation of Molten Zone in Micro-welding of Glass by Picosecond Pulsed Laser. J. Laser Micro/Nanoeng. 2013, 8, 65–69. [Google Scholar] [CrossRef]
- Kaiser, E. Laser Welding of Glass Replaces Glueing Procedure: Glass welding with a femtosecond laser brings economic advantages and new design options. Laser Tech. J. 2016, 13, 22–25. [Google Scholar] [CrossRef]
- Nopper, R.; Niekrawietz, R.; Reindl, L. Wireless readout of passive LC sensors. IEEE Trans. Instrum. Meas. 2010, 59, 2450–2457. [Google Scholar] [CrossRef]
- Chen, P.J.; Saati, S.; Varma, R.; Humayun, M.S.; Tai, Y.C. Wireless intraocular pressure sensing using microfabricated minimally invasive flexible-coiled LC sensor implant. J. Microelectromech. Syst. 2010, 19, 721–734. [Google Scholar] [CrossRef]
- Rangsten, P.; Vallin, O.; Hermansson, K.; Backlund, Y. Quartz-to-Quartz Direct Bonding. J. Electrochem. Soc. 1999, 146, 1104–1105. [Google Scholar] [CrossRef]
- Cvecek, K.; Odato, R.; Dehmel, S.; Miyamoto, I.; Schmidt, M. Gap bridging in joining of glass using ultra short laser pulses. Opt. Express 2015, 23, 5681–5693. [Google Scholar] [CrossRef]
- Chen, J.; Carter, R.M.; Thomson, R.R.; Hand, D.P. Avoiding the requirement for pre-existing optical contact during picosecond laser glass-to-glass welding. Opt. Express 2015, 23, 18645–18657. [Google Scholar] [CrossRef] [PubMed]
- Malvindi, M.A.; Brunetti, V.; Vecchio, G.; Galeone, A.; Cingolani, R.; Pompa, P.P. SiO2 nanoparticles biocompatibility and their potential for gene delivery and silencing. Nanoscale 2012, 4, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Cvecek, K.; Miyamoto, I.; Schmidt, M. Gas bubble formation in fused silica generated by ultra-short laser pulses. Opt. Express 2014, 22, 15877–15893. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.; Döring, S.; Tünnermann, A.; Nolte, S. Bonding of glass with femtosecond laser pulses at high repetition rates. Appl. Phys. A 2010, 103, 257–261. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.; Park, J.; So, S.; Ahn, S.; Choi, J.; Koo, C.; Joung, Y.-H. Characteristics of an Implantable Blood Pressure Sensor Packaged by Ultrafast Laser Microwelding. Sensors 2019, 19, 1801. https://doi.org/10.3390/s19081801
Kim S, Park J, So S, Ahn S, Choi J, Koo C, Joung Y-H. Characteristics of an Implantable Blood Pressure Sensor Packaged by Ultrafast Laser Microwelding. Sensors. 2019; 19(8):1801. https://doi.org/10.3390/s19081801
Chicago/Turabian StyleKim, Sungil, Jaesoon Park, Sangkyun So, Sanghoon Ahn, Jiyeon Choi, Chiwan Koo, and Yeun-Ho Joung. 2019. "Characteristics of an Implantable Blood Pressure Sensor Packaged by Ultrafast Laser Microwelding" Sensors 19, no. 8: 1801. https://doi.org/10.3390/s19081801
APA StyleKim, S., Park, J., So, S., Ahn, S., Choi, J., Koo, C., & Joung, Y.-H. (2019). Characteristics of an Implantable Blood Pressure Sensor Packaged by Ultrafast Laser Microwelding. Sensors, 19(8), 1801. https://doi.org/10.3390/s19081801





