Approaches to Enhancing Gas Sensing Properties: A Review
Abstract
:1. Introduction
2. Controlling the Type and Morphology of Nanostructures
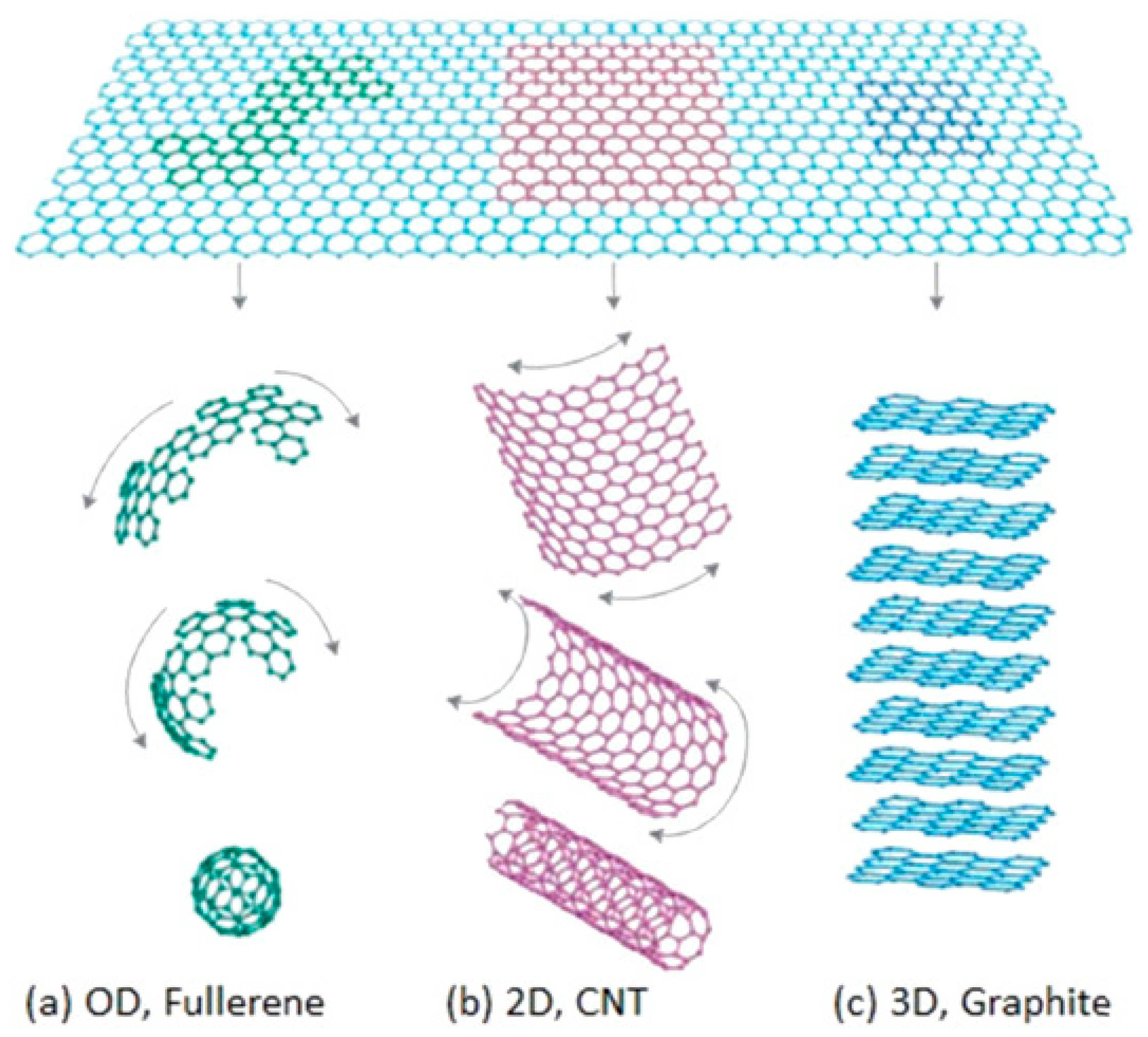
2.1. The Type and Morphology of Nanostructures
2.1.1. The Type of Nanostructures
2.1.2. The Morphology of Nanostructures
2.2. Some Typical Structure and Morphology
2.2.1. Nanorod
2.2.2. Nanosheet
2.2.3. Micro-/Nano-Structured Hollow Spheres
2.2.4. Nanoflower
2.2.5. Core-Shell Structure
3. Doping with Two-Dimensional Nanomaterials
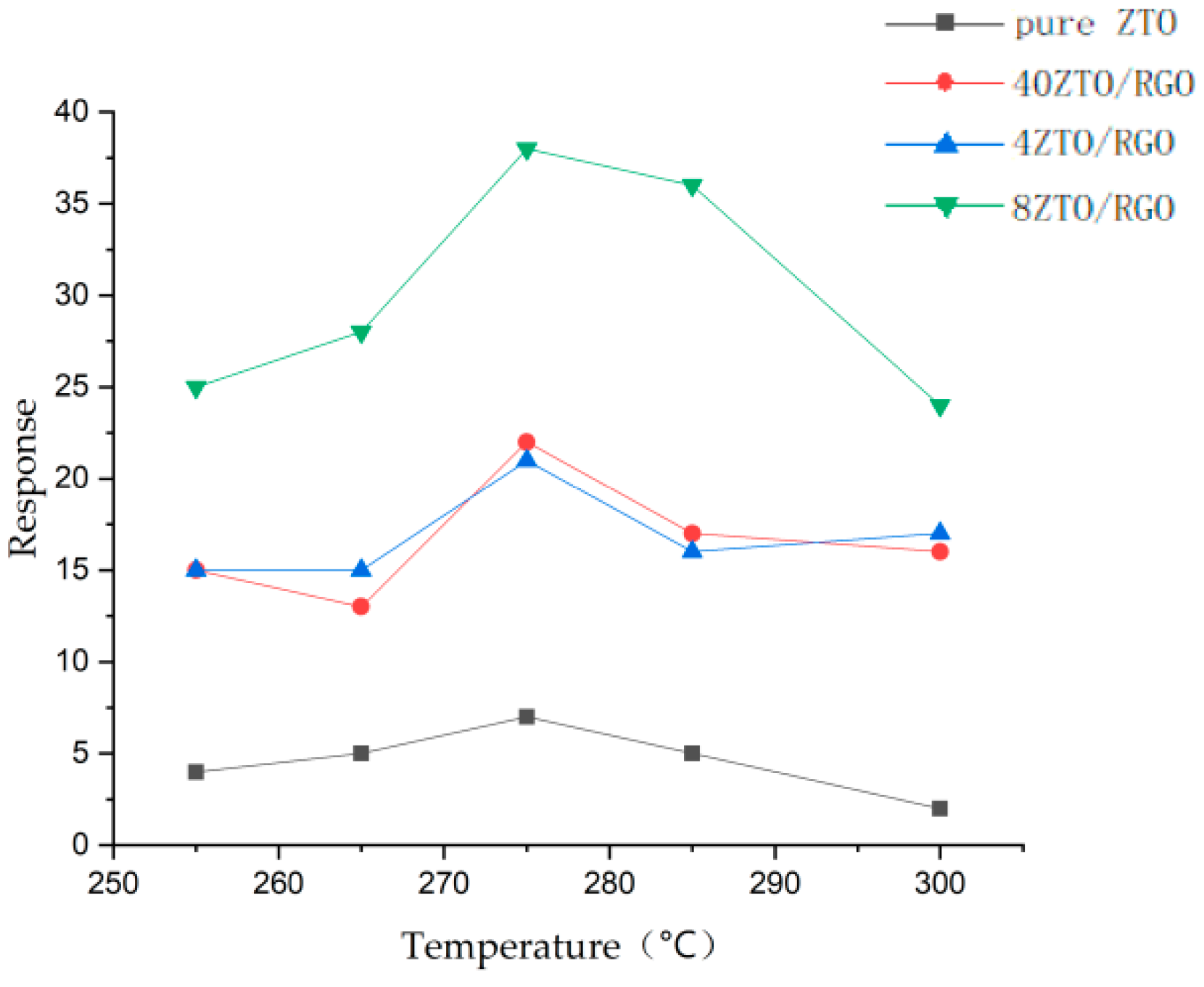
3.1. Graphene
3.2.
3.3. Black Phosphorus
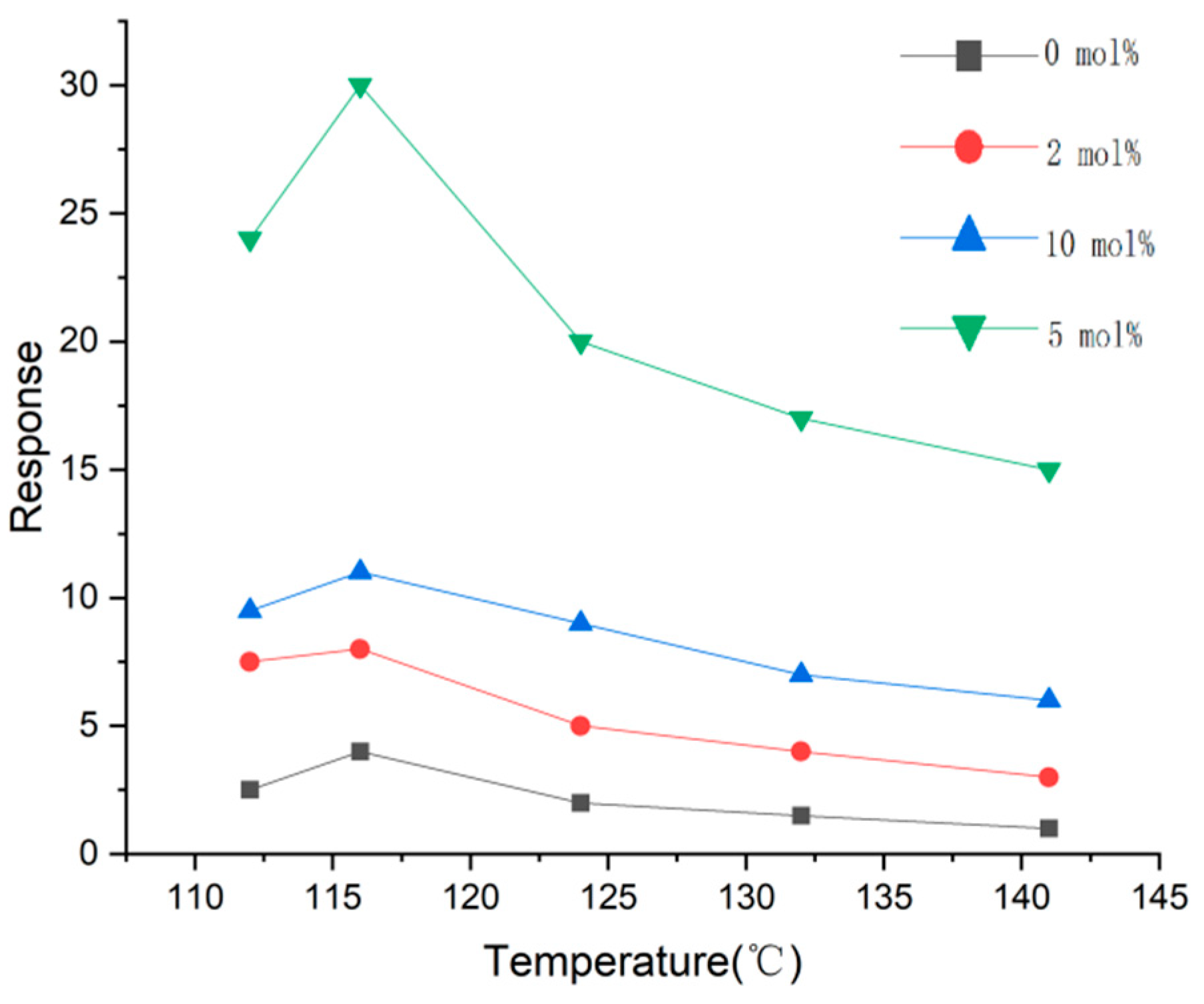
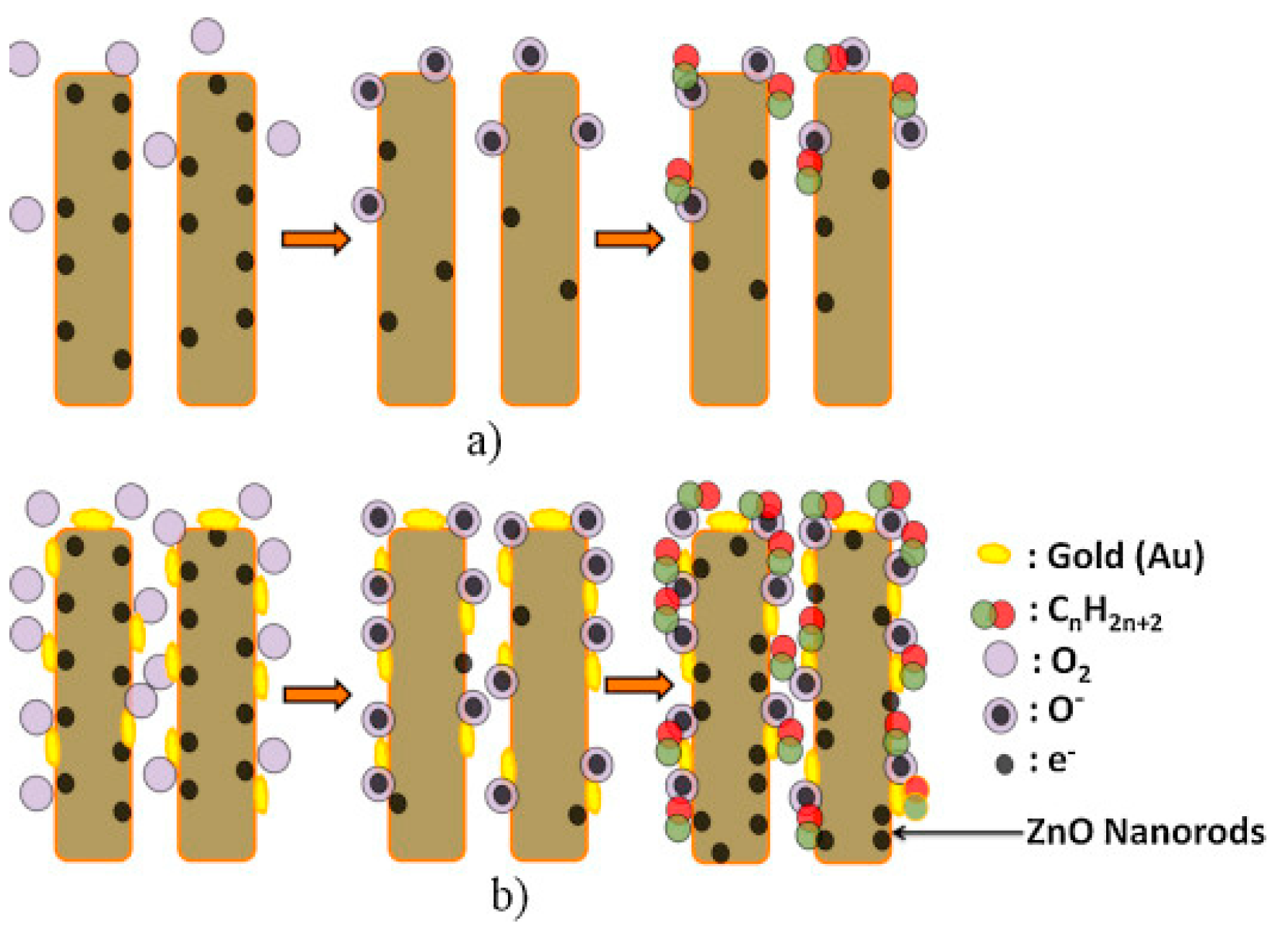
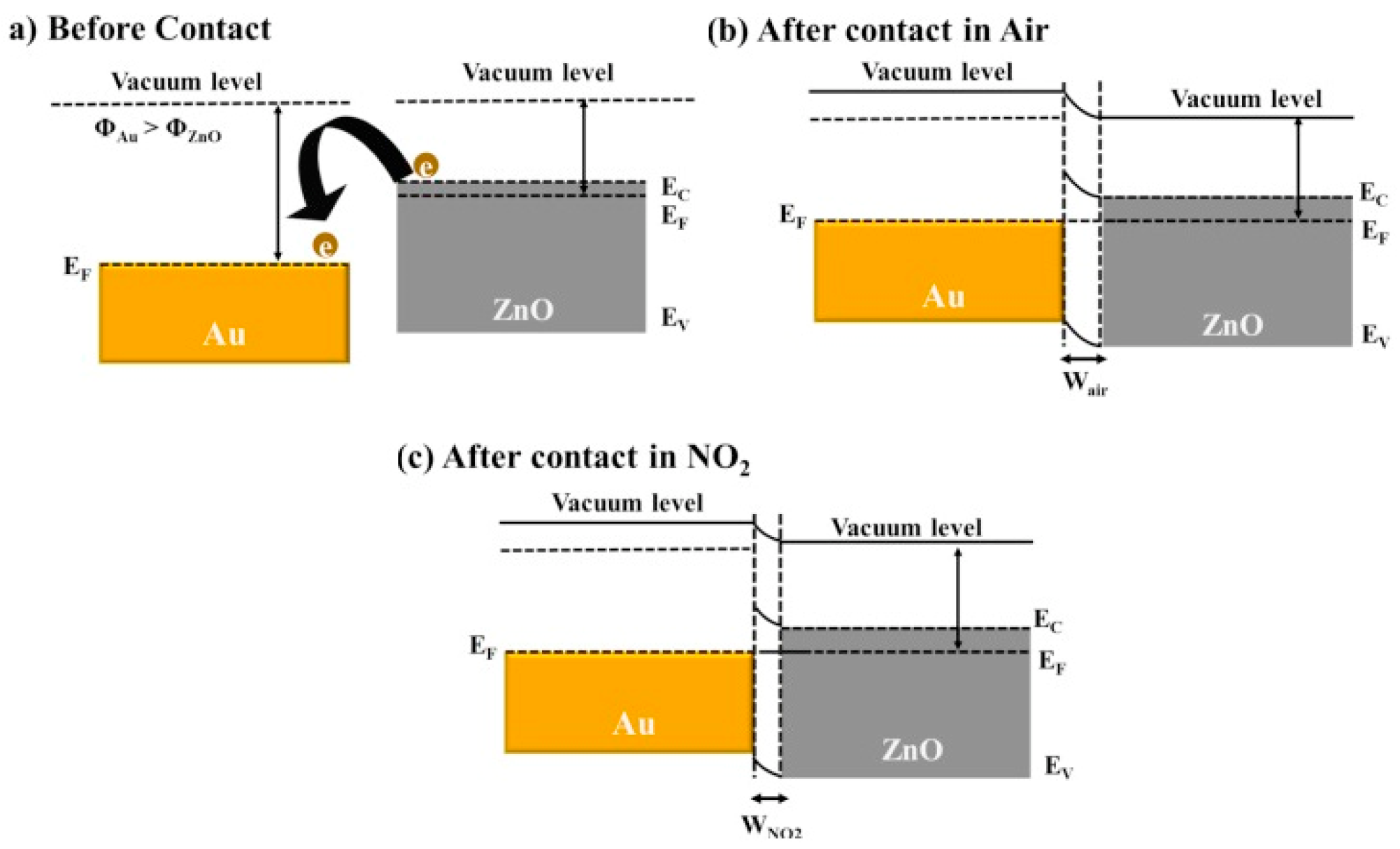
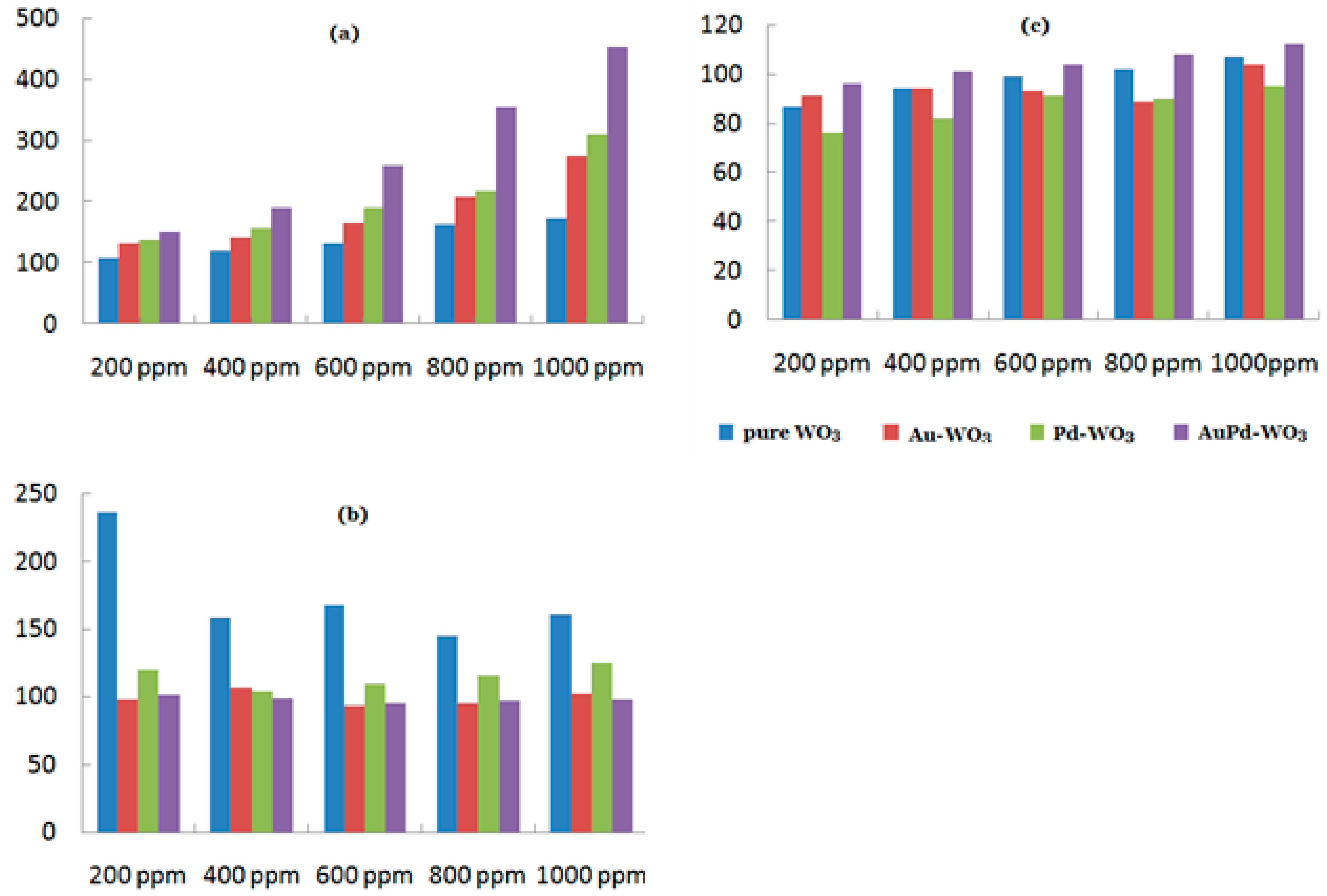
4. Decorating with Noble Metal Nanoparticles
5. Forming the Heterojunction
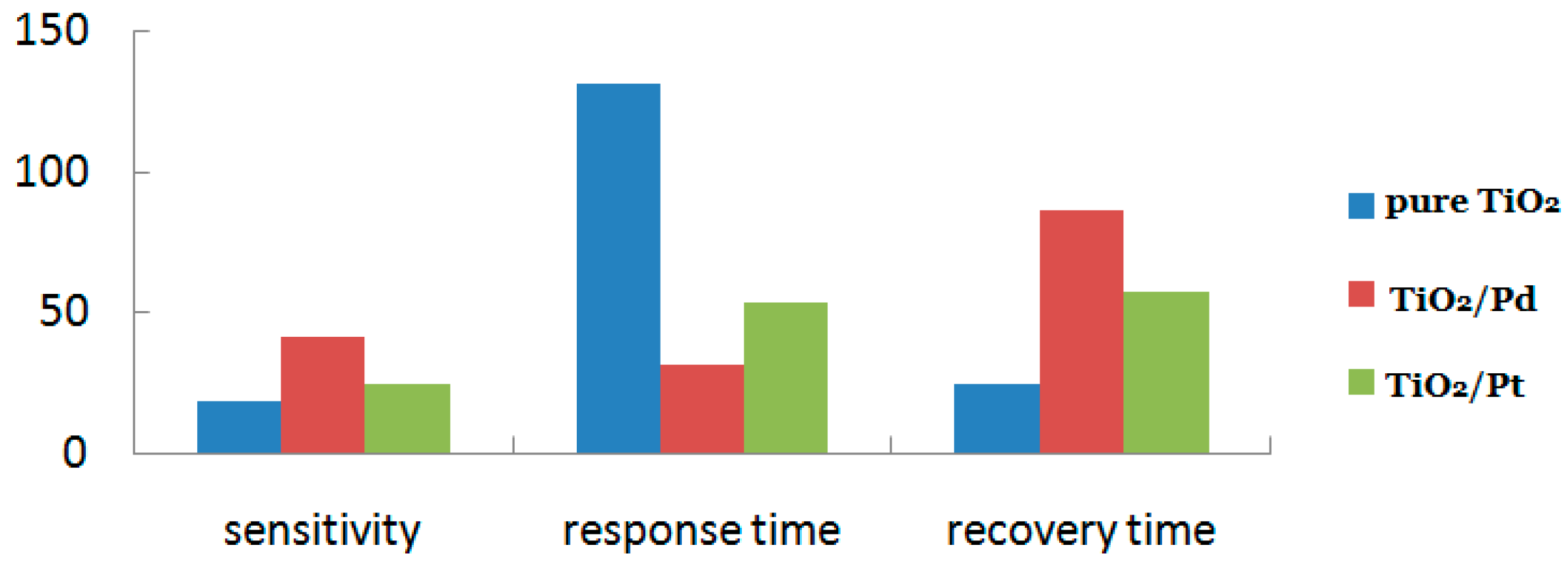
5.1. n-n Heterojunction
5.2. p-p Heterojunction
5.3. p-n Heterojunction
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, Z.; Yao, Z.; Haidry, A.A.; Plecenik, T.; Xie, L.; Sun, L.; Fatima, Q. Resistive-type hydrogen gas sensor based on TiO2: A review. Int. J. Hydrogen Energy 2018, 43, 21114–21132. [Google Scholar] [CrossRef]
- Sertel, B.C.; Sonmez, N.A.; Kaya, M.D.; Ozcelik, S. Development of MgO: TiO2 thin films for gas sensor applications. Ceram. Int. 2019, 45, 2917–2921. [Google Scholar] [CrossRef]
- Meng, F.-L.; Zhang, L.; Jia, Y.; Liu, J.-Y.; Sun, Y.-F.; Luo, T.; Li, M.-Q.; Liu, J.-H.; Huang, X.-J. Electronic chip based on self-oriented carbon nanotube microelectrode array to enhance the sensitivity of indoor air pollutants capacitive detection. Sens. Actuators B Chem. 2011, 153, 103–109. [Google Scholar] [CrossRef]
- Manjula, M.; Karthikeyan, B.; Sastikumar, D. Sensing characteristics of nanocrystalline bismuth oxide clad-modified fiber optic gas sensor. Opt. Lasers Eng. 2017, 95, 78–82. [Google Scholar] [CrossRef]
- Wang, T.-Y.; Li, Y.-Y.; Li, T.-T.; Yu, H.; Yang, Y.; Yang, H.; Dong, X.-T. Enhanced NOx gas sensing properties of Cr2O3 film modified ordered porous ZnO gas sensors. Solid State Ionics 2018, 326, 173–182. [Google Scholar] [CrossRef]
- Laborda, F.; Bolea, E. Nanomaterials Engineered Nanomaterials. In Encyclopedia of Analytical Science, 3rd ed.; Worsfold, P., Poole, C., Townshend, A., Miró, M., Eds.; Academic Press: Oxford, UK, 2018; pp. 108–116. [Google Scholar]
- Heydari, M.; Ghoreishi, S.M.; Khoobi, A. Chemometrics-assisted determination of Sudan dyes using zinc oxide nanoparticle-based electrochemical sensor. Food Chem. 2019, 283, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H. Aptamer Conjugated Quantum Dots for Imaging Cellular Uptake in Cancer Cells. J. Nanosci. Nanotechnol. 2019, 19, 3798–3803. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.; Yao, Y.; Zhang, X.; Yuan, J.; Sun, B.; Gao, X. The Gold Nanocluster Protects Neurons Directly or via Inhibiting Cytotoxic Secretions of Microglia Cell. J. Nanosci. Nanotechnol. 2019, 19, 1986–1995. [Google Scholar] [CrossRef]
- Kolmakov, A.; Zhang, Y.; Cheng, G.; Moskovits, M. Detection of CO and O2 Using Tin Oxide Nanowire Sensors. Adv. Mater. 2003, 15, 997–1000. [Google Scholar] [CrossRef]
- Balaban, O.; Grygorchak, I.; Mitina, N.; Zaichenko, A.; Lukiyanets, B.; Glasunova, V.; Borysiuk, A.; Larkin, M.; Hevus, O.; Pokladok, N.; et al. Fabrication of 1D-Nanofiber/Fe2O3 Composites with Tailored Magnetic Properties. J. Nanosci. Nanotechnol. 2019, 19, 3871–3878. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.; Priya, A.; Arun, A.; Hait, S.; Chowdhury, A. Antibacterial and natural room-light driven photocatalytic activities of CuO nanorods. Mater. Chem. Phys. 2019, 226, 106–112. [Google Scholar] [CrossRef]
- Liu, S.F.; Lin, S.; Swager, T.M. An Organocobalt–Carbon Nanotube Chemiresistive Carbon Monoxide Detector. ACS Sens. 2016, 1, 354–357. [Google Scholar] [CrossRef]
- Choi, H.H.; Lee, J.; Dong, K.-Y.; Ju, B.-K.; Lee, W. Noxious gas detection using carbon nanotubes with Pd nanoparticles. Nanoscale Res. Lett. 2011, 6, 605. [Google Scholar] [CrossRef]
- Wu, L.; Yang, D. Dielectric Properties and Thermal Conductivity of Poly(vinylidene fluoride)-Based Composites with Graphite Nanosheet and Nickel Particle. J. Nanosci. Nanotechnol. 2019, 19, 3591–3596. [Google Scholar] [CrossRef]
- Qiao, Y.; Shi, C.; Wang, X.; Wang, P.; Zhang, Y.; Wang, D.; Qiao, R.; Wang, X.; Zhong, J. Electrospun Nanobelt-Shaped Polymer Membranes for Fast and High-Sensitivity Detection of Metal Ions. ACS Appl. Mater. Interfaces 2019, 11, 5401–5413. [Google Scholar] [CrossRef] [PubMed]
- Sprouster, D.J.; Sun, C.; Zhang, Y.; Chodankar, S.N.; Gan, J.; Ecker, L.E. Irradiation-Dependent Helium Gas Bubble Superlattice in Tungsten. Sci. Rep. 2019, 9, 2277. [Google Scholar] [CrossRef] [PubMed]
- Meng, D.; Liu, D.; Wang, G.; Shen, Y.; San, X.; Li, M.; Meng, F. Low-temperature formaldehyde gas sensors based on NiO-SnO2 heterojunction microflowers assembled by thin porous nanosheets. Sens. Actuators B Chem. 2018, 273, 418–428. [Google Scholar] [CrossRef]
- Ma, S.S.; Li, R.; Lv, C.P.; Xu, W.; Gou, X.L. Facile synthesis of ZnO nanorod arrays and hierarchical nanostructures for photocatalysis and gas sensor applications. J. Hazard. Mater. 2011, 192, 730–740. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Zhou, Q.; Lu, Z.; Xu, L.; Gui, Y.; Tang, C. Morphology Controllable Synthesis of Hierarchical WO3 Nanostructures and C2H2 Sensing Properties. Phys. E Low-Dimens. Syst. Nanostruct. 2019. [Google Scholar] [CrossRef]
- Wu, D.; Li, C.; Kong, Q.; Shi, Z.; Zhang, D.; Wang, L.; Han, L.; Zhang, X.; Lin, Q. Photocatalytic activity of Lu3+/TiO2 prepared by ball milling method. J. Rare Earths 2018, 36, 819–825. [Google Scholar] [CrossRef]
- Antisari, M.V.; Montone, A.; Jovic, N.; Piscopiello, E.; Alvani, C.; Pilloni, L. Low energy pure shear milling: A method for the preparation of graphite nano-sheets. Scr. Mater. 2006, 55, 1047–1050. [Google Scholar] [CrossRef]
- Fireman, M.N.; L’Heureux, G.; Wu, F.; Mates, T.; Young, E.C.; Speck, J.S. High germanium doping of GaN films by ammonia molecular beam epitaxy. J. Cryst. Growth 2019, 508, 19–23. [Google Scholar] [CrossRef]
- Sivajee Ganesh, K.; Purusottamreddy, B.; Jeevan Kumar, P.; Hussain, O.M. Influence of Zr dopant on microstructural and electrochemical properties of LiCoO2 thin film cathodes by RF sputtering. J. Electroanal. Chem. 2018, 828, 71–79. [Google Scholar] [CrossRef]
- Hao, S.; Lin, T.; Ning, S.; Qi, Y.; Deng, Z.; Wang, Y. Research on cracking of SiO2 nanofilms prepared by the sol-gel method. Mater. Sci. Semicond. Process. 2019, 91, 181–187. [Google Scholar] [CrossRef]
- Khan, J.; Ullah, H.; Sajjad, M.; Ali, A.; Thebo, K.H. Synthesis, characterization and electrochemical performance of cobalt fluoride nanoparticles by reverse micro-emulsion method. Inorg. Chem. Commun. 2018, 98, 132–140. [Google Scholar] [CrossRef]
- Li, S.; Li, X.; Zou, K.; Huang, Z.; Zhang, L.; Zhou, X.; Guo, D.; Ju, Y. Preparation of single-crystalline BaTi5O11 nanocrystals by hydrothermal method. Mater. Lett. 2019. [Google Scholar] [CrossRef]
- Keller, D.; Henninen, T.R.; Erni, R. Formation of gold nanoparticles in a free-standing ionic liquid triggered by heat and electron irradiation. Micron 2019, 117, 16–21. [Google Scholar] [CrossRef]
- Lu, Z.; Wang, H.; Zeng, J.; Liu, J. Dynamic crystal-growth process observed during hydrothermal coarsening of nanocrystalline hydroxy fluorapatite. J. Cryst. Growth 2009, 311, 4791–4798. [Google Scholar] [CrossRef]
- Kumar, V.; Kumar, P.; Pournara, A.; Vellingiri, K.; Kim, K.-H. Nanomaterials for the sensing of narcotics: Challenges and opportunities. TrAC Trends Anal. Chem. 2018, 106, 84–115. [Google Scholar] [CrossRef]
- Zou, S.; Luo, J.; Lin, Z.D.; Fu, P.; Chen, Z. Acetone gas sensor based on iron molybdate nanoparticles prepared by hydrothermal method with PVP as surfactant. Mater. Res. Express 2018, 5, 7. [Google Scholar] [CrossRef]
- Ren, Y.; Yang, X.; Zhou, X.; Luo, W.; Zhang, Y.; Cheng, X.; Deng, Y. Amphiphilic block copolymers directed synthesis of mesoporous nickel-based oxides with bimodal mesopores and nanocrystal-assembled walls. Chin. Chem. Lett. 2019. [Google Scholar] [CrossRef]
- An, S.; Park, S.; Ko, H.; Lee, C. Enhanced NO2 gas sensing properties of WO3 nanorods encapsulated with ZnO. Appl. Phys. A 2012, 108, 53–58. [Google Scholar] [CrossRef]
- Patil, D.; Patil, P.; Subramanian, V.; Joy, P.A.; Potdar, H.S. Highly sensitive and fast responding CO sensor based on Co3O4 nanorods. Talanta 2010, 81, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, G.N.; Annamalai, K. Development of Gas Sensor and Optical Properties of Zinc Oxide Nanorods by Simple Hydrothermal Approach. Mater. Today Proc. 2017, 4, 12070–12074. [Google Scholar] [CrossRef]
- Narayanan, G.N.; Sankar Ganesh, R.; Karthigeyan, A. Effect of annealing temperature on structural, optical and electrical properties of hydrothermal assisted zinc oxide nanorods. Thin Solid Films 2016, 598, 39–45. [Google Scholar] [CrossRef]
- Oh, E.; Choi, H.-Y.; Jung, S.-H.; Cho, S.; Kim, J.C.; Lee, K.-H.; Kang, S.-W.; Kim, J.; Yun, J.-Y.; Jeong, S.-H. High-performance NO2 gas sensor based on ZnO nanorod grown by ultrasonic irradiation. Sens. Actuators B Chem. 2009, 141, 239–243. [Google Scholar] [CrossRef]
- Long, H.; Li, Y.; Zeng, W. Substrate-free synthesis of WO3 nanorod arrays and their superb NH3-sensing performance. Mater. Lett. 2017, 209, 342–344. [Google Scholar] [CrossRef]
- Choi, P.G.; Izu, N.; Shirahata, N.; Masuda, Y. Fabrication and H2-Sensing Properties of SnO2 Nanosheet Gas Sensors. Acs Omega 2018, 3, 14592–14596. [Google Scholar] [CrossRef]
- Li, Y.; Liu, B.; Wang, H.; Su, X.; Gao, L.; Zhou, F.; Duan, G. Co3O4 nanosheet-built hollow dodecahedrons via a two step self-templated method and their multifunctional applications. Sci. China Mater. 2018, 61, 1575–1586. [Google Scholar] [CrossRef]
- Zhang, Y.-H.; Cai, X.-L.; Song, L.-Z.; Feng, F.-Y.; Ding, J.-Y.; Gong, F.-L. 2D nanosheet-assembled Pd-ZnOmicroflowers for acetone sensor with enhanced performances. J. Phys. Chem. Solids 2019, 124, 330–335. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Zhang, H.W.; Zhu, Y.D.; Dai, Z.F.; Bao, H.M.; Wei, Y.; Cai, W.P. Au-NP-Decorated Crystalline FeOCl Nanosheet: Facile Synthesis by Laser Ablation in Liquid and its Exclusive Gas Sensing Response to HCl at Room Temperature. Adv. Mater. Interfaces 2016, 3, 8. [Google Scholar] [CrossRef]
- Pan, Y.; Liu, B.; Hu, H.; Jiang, H.; Wei, F.; Zhou, M.; Fan, X.; Yu, H.; Niu, G.; Huang, J. Preparation and photocatalytic performance of the rod-shaped Ni-NiO/TiO2 hollow composite structure based on metallization cellulose fibers and TBOT. Vacuum 2019, 159, 1–8. [Google Scholar] [CrossRef]
- Xiao, Y.; Deng, Y.; Huan, W.; Li, J.; Zhang, J.; Xing, M. Hollow-structured Fe2O3/Au/SiO2 nanorods with enhanced and recyclable photo-Fenton oxidation for the remediation of organic pollutants. Mater. Today Chem. 2019, 11, 86–93. [Google Scholar] [CrossRef]
- An, G.S.; Chae, D.H.; Hur, J.U.; Oh, A.H.; Choi, H.-H.; Choi, S.-C.; Oh, Y.-S.; Jung, Y.-G. Hollow-structured Fe3O4@SiO2 nanoparticles: Novel synthesis and enhanced adsorbents for purification of plasmid DNA. Ceram. Int. 2018, 44, 18791–18795. [Google Scholar] [CrossRef]
- Kang, Y.; Li, Z.; Xu, K.; He, X.; Wei, S.; Cao, Y. Hollow SnO2 nanospheres with single-shelled structure and the application for supercapacitors. J. Alloys Compd. 2019, 779, 728–734. [Google Scholar] [CrossRef]
- Lai, X.; Halpert, J.E.; Wang, D. Recent advances in micro-/nano-structured hollow spheres for energy applications: From simple to complex systems. Energy Environ. Sci. 2012, 5, 5604–5618. [Google Scholar] [CrossRef]
- Xie, F.; Qi, M.Z.; Li, W.J.; Wang, K.; Yu, Z.Y.; Liu, B. Classification, Fabrication Methods and Applications of Inorganic Hollow Spheres. Progress Chem. 2011, 23, 2522–2533. [Google Scholar]
- Han, D.; Song, P.; Zhang, H.; Yang, Z.; Wang, Q. Cu2O template-assisted synthesis of porous In2O3 hollow spheres with fast response towards acetone. Mater. Lett. 2014, 124, 93–96. [Google Scholar] [CrossRef]
- Zhai, C.; Zhu, M.; Jiang, L.; Yang, T.; Zhao, Q.; Luo, Y.; Zhang, M. Fast triethylamine gas sensing response properties of nanosheets assembled WO3 hollow microspheres. App. Surf. Sci. 2019, 463, 1078–1084. [Google Scholar] [CrossRef]
- Wang, L.; Wang, S.; Wang, Y.; Zhang, H.; Kang, Y.; Huang, W. Synthesis of hierarchical SnO2 nanostructures assembled with nanosheets and their improved gas sensing properties. Sens. Actuators B Chem. 2013, 188, 85–93. [Google Scholar] [CrossRef]
- Song, Y.; Chen, F.; Zhang, Y.; Zhang, S.; Liu, F.; Sun, P.; Yan, X.; Lu, G. Fabrication of highly sensitive and selective room-temperature nitrogen dioxide sensors based on the ZnO nanoflowers. Sens. Actuators B Chem. 2019, 287, 191–198. [Google Scholar] [CrossRef]
- Zhu, L.; Zeng, W.; Li, Y. A non-oxygen adsorption mechanism for hydrogen detection of nanostructured SnO2 based sensors. Mater. Res. Bull. 2019, 109, 108–116. [Google Scholar] [CrossRef]
- Guan, C.; Xia, X.; Meng, N.; Zeng, Z.; Cao, X.; Soci, C.; Zhang, H.; Fan, H.J. Hollow core-shell nanostructure supercapacitor electrodes: Gap matters. Energy Environ. Sci. 2012, 5, 9085–9090. [Google Scholar] [CrossRef]
- Ma, Z.; Shao, G.; Fan, Y.; Wang, G.; Song, J.; Shen, D. Construction of Hierarchical alpha-MnO2 Nanowires@Ultrathin delta-MnO2 Nanosheets Core-Shell Nanostructure with Excellent Cycling Stability for High-Power Asymmetric Supercapacitor Electrodes. ACS Appl. Mater. Interfaces 2016, 8, 9050–9058. [Google Scholar] [CrossRef]
- Santra, S.; Liesenfeld, B.; Bertolino, C.; Dutta, D.; Cao, Z.H.; Tan, W.H.; Moudgil, B.M.; Mericle, R.A. Fluorescence lifetime measurements to determine the core-shell nanostructure of FITC-doped silica nanoparticles: An optical approach to evaluate nanoparticle photostability. J. Lumin. 2006, 117, 75–82. [Google Scholar] [CrossRef]
- Kong, J.; Liu, Z.; Yang, Z.; Tan, H.R.; Xiong, S.; Wong, S.Y.; Li, X.; Lu, X. Carbon/SnO2/carbon core/shell/shell hybrid nanofibers: Tailored nanostructure for the anode of lithium ion batteries with high reversibility and rate capacity. Nanoscale 2012, 4, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Feng, J.; Bian, X.; Liu, J.; Xu, H.; An, Y. A controlled red phosphorus@Ni-Pcore@shell nanostructure as an ultralong cycle-life and superior high-rate anode for sodium-ion batteries. Energy Environ. Sci. 2017, 10, 1222–1233. [Google Scholar] [CrossRef]
- Misra, M.; Kapur, P.; Singla, M.L. Surface plasmon quenched of near band edge emission and enhanced visible photocatalytic activity of Au@ZnO core-shell nanostructure. Appl. Catal. B Environ. 2014, 150, 605–611. [Google Scholar] [CrossRef]
- Senapati, S.; Srivastava, S.K.; Singh, S.B.; Mishra, H.N. Magnetic Ni/Ag core-shell nanostructure from prickly Ni nanowire precursor and its catalytic and antibacterial activity. J. Mater. Chem. 2012, 22, 6899–6906. [Google Scholar] [CrossRef]
- Veisi, H.; Taheri, S.; Hemmati, S. Preparation of polydopamine sulfamic acid-functionalized magnetic Fe3O4 nanoparticles with a core/shell nanostructure as heterogeneous and recyclable nanocatalysts for the acetylation of alcohols, phenols, amines and thiols under solvent-free conditions. Green Chem. 2016, 18, 6337–6348. [Google Scholar] [CrossRef]
- Xu, M.; Chen, D.; Huang, P.; Wan, Z.; Zhou, Y.; Ji, Z. A dual-functional upconversion core@shell nanostructure for white-light-emission and temperature sensing. J. Mater. Chem. C 2016, 4, 6516–6524. [Google Scholar] [CrossRef]
- Yu, Y.-T.; Majhi, S.M.; Song, H.-G. Synthesis and Gas Sensing Properties of Au@In2O3 Core-shell Nanoparticles. Procedia Eng. 2016, 168, 227–230. [Google Scholar] [CrossRef]
- Jin, C.; Park, S.; Kim, H.; Lee, C. Ultrasensitive multiple networked Ga2O3-core/ZnO-shell nanorod gas sensors. Sens. Actuators B Chem. 2012, 161, 223–228. [Google Scholar] [CrossRef]
- Si, S.; Li, C.; Wang, X.; Peng, Q.; Li, Y. Fe2O3/ZnO core–shell nanorods for gas sensors. Sens. Actuators B Chem. 2006, 119, 52–56. [Google Scholar] [CrossRef]
- Li, F.; Zhang, T.; Gao, X.; Wang, R.; Li, B. Coaxial electrospinning heterojunction SnO2/Au-doped In2O3 core-shell nanofibers for acetone gas sensor. Sens. Actuators B Chem. 2017, 252, 822–830. [Google Scholar] [CrossRef]
- Park, S.; Ko, H.; Lee, S.; Kim, H.; Lee, C. Light-activated gas sensing of Bi2O3-core/ZnO-shell nanobelt gas sensors. Thin Solid Films 2014, 570, 298–302. [Google Scholar] [CrossRef]
- Thanh Le, D.T.; Trung, D.D.; Chinh, N.D.; Thanh Binh, B.T.; Hong, H.S.; Van Duy, N.; Hoa, N.D.; Van Hieu, N. Facile synthesis of SnO2–ZnO core–shell nanowires for enhanced ethanol-sensing performance. Curr. Appl. Phys. 2013, 13, 1637–1642. [Google Scholar] [CrossRef]
- Xu, H.; Li, W.; Han, R.; Zhai, T.; Yu, H.; Chen, Z.; Wu, X.; Wang, J.; Cao, B. Enhanced triethylamine sensing properties by fabricating Au@SnO2/α-Fe2O3 core-shell nanoneedles directly on alumina tubes. Sens. Actuators B Chem. 2018, 262, 70–78. [Google Scholar] [CrossRef]
- Wang, Y.; Qu, F.; Liu, J.; Wang, Y.; Zhou, J.; Ruan, S. Enhanced H2S sensing characteristics of CuO-NiO core-shell microspheres sensors. Sens. Actuators B Chem. 2015, 209, 515–523. [Google Scholar] [CrossRef]
- Yao, Y.; Yin, M.; Yan, J.; Yang, D.; Liu, S. Controllable synthesis of Ag-WO3 core-shell nanospheres for light-enhanced gas sensors. Sens. Actuators B Chem. 2017, 251, 583–589. [Google Scholar] [CrossRef]
- Yu, Q.; Zhu, J.; Xu, Z.; Huang, X. Facile synthesis of α-Fe2O3@SnO2 core–shell heterostructure nanotubes for high performance gas sensors. Sens. Actuators B Chem. 2015, 213, 27–34. [Google Scholar] [CrossRef]
- Zhu, Z.; Kao, C.-T.; Wu, R.-J. A highly sensitive ethanol sensor based on Ag@TiO2 nanoparticles at room temperature. Appl. Surf. Sci. 2014, 320, 348–355. [Google Scholar] [CrossRef]
- Li, C.; Su, Y.; Lv, X.; Zuo, Y.; Yang, X.; Wang, Y. Au@Pd core–shell nanoparticles: A highly active electrocatalyst for amperometric gaseous ethanol sensors. Sens. Actuators B Chem. 2012, 171–172, 1192–1198. [Google Scholar] [CrossRef]
- Guo, L.; Yang, Z.; Li, Y.; Zu, B.; Dou, X. Sensitive, real-time and anti-interfering detection of nitro-explosive vapors realized by ZnO/rGO core/shell micro-Schottky junction. Sens. Actuators B Chem. 2017, 239, 286–294. [Google Scholar] [CrossRef]
- Li, S.; Diao, Y.; Yang, Z.; He, J.; Wang, J.; Liu, C.; Liu, F.; Lu, H.; Yan, X.; Sun, P.; et al. Enhanced room temperature gas sensor based on Au-loaded mesoporous In2O3 nanospheres@polyaniline core-shell nanohybrid assembled on flexible PET substrate for NH3 detection. Sens. Actuators B Chem. 2018, 276, 526–533. [Google Scholar] [CrossRef]
- Saboor, F.H.; Khodadadi, A.A.; Mortazavi, Y.; Asgari, M. Microemulsion synthesized silica/ZnO stable core/shell sensors highly selective to ethanol with minimum sensitivity to humidity. Sens. Actuators B Chem. 2017, 238, 1070–1083. [Google Scholar] [CrossRef]
- Park, S.; Kim, S.; Sun, G.-J.; Lee, C. Synthesis, structure and ethanol sensing properties of Ga2O3-core/WO3-shell nanostructures. Thin Solid Films 2015, 591, 341–345. [Google Scholar] [CrossRef]
- Runa, A.; Zhang, X.; Wen, G.; Zhang, B.; Fu, W.; Yang, H. Actinomorphic flower-like n-ZnO/p-ZnFe2O4 composite and its improved NO2 gas-sensing property. Mater. Lett. 2018, 225, 73–76. [Google Scholar] [CrossRef]
- Uddin, A.S.M.I.; Yaqoob, U.; Hassan, K.; Chung, G.-S. Effects of Pt shell thickness on self-assembly monolayer Pd@Pt core-shell nanocrystals based hydrogen sensing. Int. J. Hydrogen Energy 2016, 41, 15399–15410. [Google Scholar] [CrossRef]
- Gangu, K.K.; Maddila, S.; Mukkamala, S.B.; Jonnalagadda, S.B. Characteristics of MOF, MWCNT and graphene containing materials for hydrogen storage: A review. J. Energy Chem. 2019, 30, 132–144. [Google Scholar] [CrossRef]
- Pumera, M. Electrochemistry of graphene, graphene oxide and other graphenoids: Review. Electrochem. Commun. 2013, 36, 14–18. [Google Scholar] [CrossRef]
- Ren, S.; Rong, P.; Yu, Q. Preparations, properties and applications of graphene in functional devices: A concise review. Ceram. Int. 2018, 44, 11940–11955. [Google Scholar] [CrossRef]
- Al Hassan, M.R.; Sen, A.; Zaman, T.; Mostari, M.S. Emergence of graphene as a promising anode material for rechargeable batteries: A review. Mater. Today Chem. 2019, 11, 225–243. [Google Scholar] [CrossRef]
- Vasut, F.; Oubraham, A.; Soare, A.M.; Marinoiu, A.; Ion-Ebrasu, D.; Dragan, M. Platinum supported on graphene—PTFE as catalysts for isotopic exchange in a detritiation plant. Fusion Eng. Des. 2018. [Google Scholar] [CrossRef]
- Nikbakht, H.; Latifi, H.; Pak, M.; Behroodi, E.; Oraie, M.; Zibaii, M.I. Sensitivity enhancement of cylindrically-symmetric optical fiber refractive index sensors by utilizing graphene. Opt. Commun. 2018, 429, 1–4. [Google Scholar] [CrossRef]
- Justh, N.; Berke, B.; Laszlo, K.; Bakos, L.P.; Szabo, A.; Hernadi, K.; Szilagyi, I.M. Preparation of graphene oxide/semiconductor oxide composites by using atomic layer deposition. Appl. Surf. Sci. 2018, 453, 245–251. [Google Scholar] [CrossRef]
- Wu, C.-H.; Pu, N.-W.; Liu, Y.-M.; Chen, C.-Y.; Peng, Y.-Y.; Cheng, T.-Y.; Lin, M.-H.; Ger, M.-D. Improving rate capability of lithium-ion batteries using holey graphene as the anode material. J. Taiwan Inst. Chem. Eng. 2017, 80, 511–517. [Google Scholar] [CrossRef]
- Bera, S.; Kundu, S.; Khan, H.; Jana, S. Polyaniline coated graphene hybridized SnO2 nanocomposite: Low temperature solution synthesis, structural property and room temperature ammonia gas sensing. J. Alloys Compd. 2018, 744, 260–270. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Zhang, B.; Wang, Y.; Gao, Y.; Liang, X.; Wang, Y.; Lu, G. Flower-like In2O3 modified by reduced graphene oxide sheets serving as a highly sensitive gas sensor for trace NO2 detection. J. Colloid Interface Sci. 2017, 504, 206–213. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, J.; Wang, X.; Zeng, D.; Xie, C. Enhancing room-temperature NO2 sensing properties via forming heterojunction for NiO-rGO composited with SnO2 nanoplates. Sens. Actuators B Chem. 2017, 243, 1010–1019. [Google Scholar] [CrossRef]
- Li, Y.; Luo, N.; Sun, G.; Zhang, B.; Lin, L.; Jin, H.; Wang, Y.; Bala, H.; Cao, J.; Zhang, Z. In situ decoration of Zn2SnO4 nanoparticles on reduced graphene oxide for high performance ethanol sensor. Ceram. Int. 2018, 44, 6836–6842. [Google Scholar] [CrossRef]
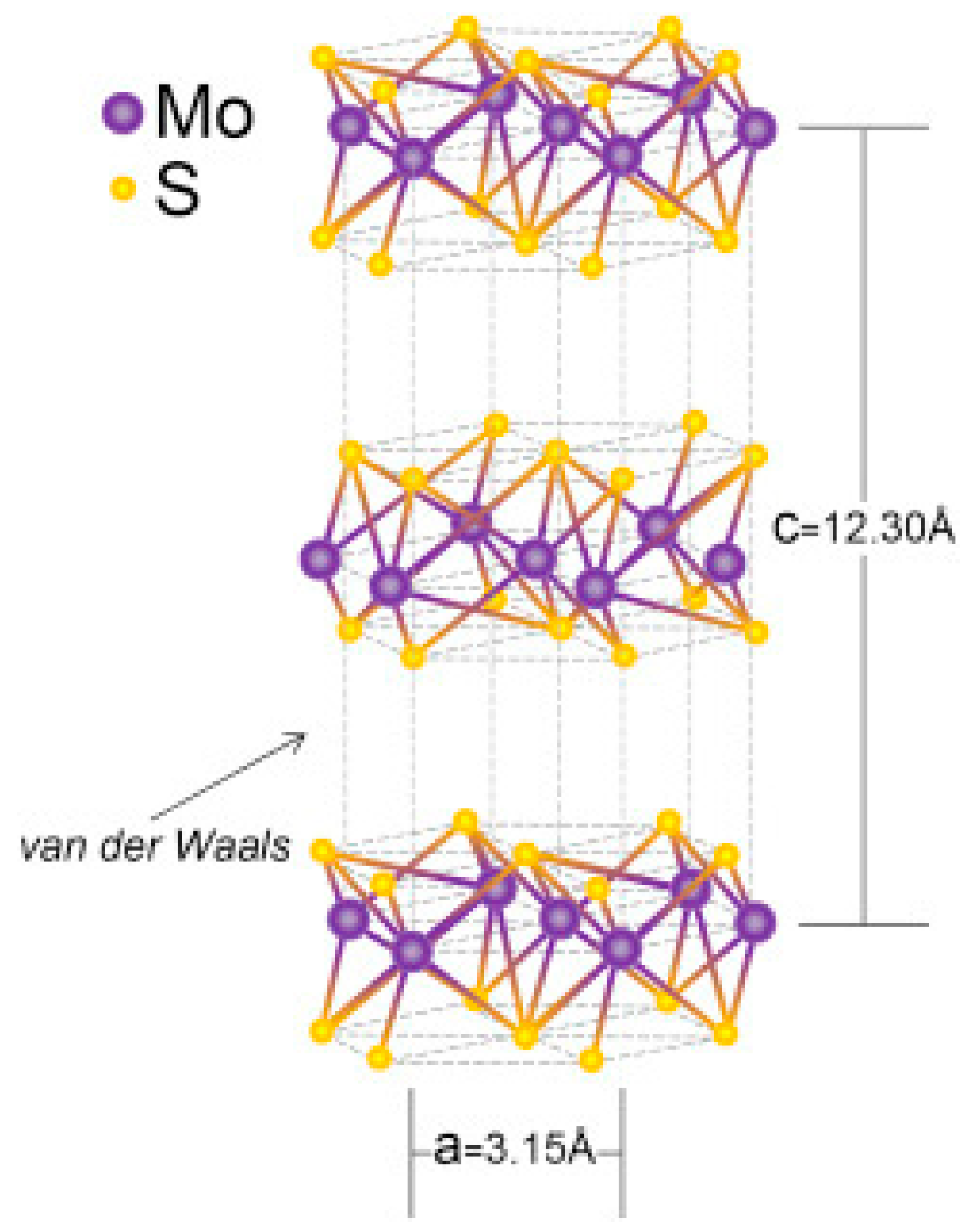
- Furlan, K.P.; de Mello, J.D.B.; Klein, A.N. Self-lubricating composites containing MoS2: A review. Tribol. Int. 2018, 120, 280–298. [Google Scholar] [CrossRef]
- Krishnan, U.; Kaur, M.; Singh, K.; Kumar, M.; Kumar, A. A synoptic review of MoS2: Synthesis to applications. Superlattices Microstruct. 2019, 128, 274–297. [Google Scholar] [CrossRef]
- Sinha, A.; Dhanjai; Tan, B.; Huang, Y.; Zhao, H.; Dang, X.; Chen, J.; Jain, R. MoS2 nanostructures for electrochemical sensing of multidisciplinary targets: A review. TrAC Trends Anal. Chem. 2018, 102, 75–90. [Google Scholar] [CrossRef]
- Jinxiao, W.; Jianfeng, Y.; Jun, Y.; Guanjun, Q.; Hang, W.; WangRui, H. Superior MoS2-decorated CNT composite materials for photoelectric detectors. Opt. Mater. 2018, 86, 113–118. [Google Scholar] [CrossRef]
- Kathiravan, D.; Huang, B.-R.; Saravanan, A.; Prasannan, A.; Hong, P.-D. Highly enhanced hydrogen sensing properties of sericin-induced exfoliated MoS2 nanosheets at room temperature. Sens. Actuators B Chem. 2019, 279, 138–147. [Google Scholar] [CrossRef]
- Benavente, E.; Durán, F.; Sotomayor-Torres, C.; González, G. Heterostructured layered hybrid ZnO/MoS2 nanosheets with enhanced visible light photocatalytic activity. J. Phys. Chem. Solids 2018, 113, 119–124. [Google Scholar] [CrossRef]
- Luxa, J.; Mazánek, V.; Mackova, A.; Malinsky, P.; Akhmadaliev, S.; Sofer, Z. Tuning of electrocatalytic properties of MoS2 by chalcogenide ion implantation. Appl. Mater. Today 2019, 14, 216–223. [Google Scholar] [CrossRef]
- Jia, Y.; Lin, Y.; Ma, Y.; Shi, W. Hierarchical MnS2-MoS2 nanotubes with efficient electrochemical performance for energy storage. Mater. Des. 2018, 160, 1071–1079. [Google Scholar] [CrossRef]
- Charoo, M.S.; Wani, M.F.; Hanief, M.; Rather, M.A. Tribological Properties of MoS2 Particles as Lubricant Additive on EN31 Alloy Steel and AISI 52100 Steel Ball. Mater. Today Proc. 2017, 4, 9967–9971. [Google Scholar] [CrossRef]
- Luo, Y.; Zhang, C. Pt-activated TiO2-MoS2 nanocomposites for H2 detection at low temperature. J. Alloys Compd. 2018, 747, 550–557. [Google Scholar] [CrossRef]
- Baek, D.-H.; Kim, J. MoS2 gas sensor functionalized by Pd for the detection of hydrogen. Sens. Actuators B Chem. 2017, 250, 686–691. [Google Scholar] [CrossRef]
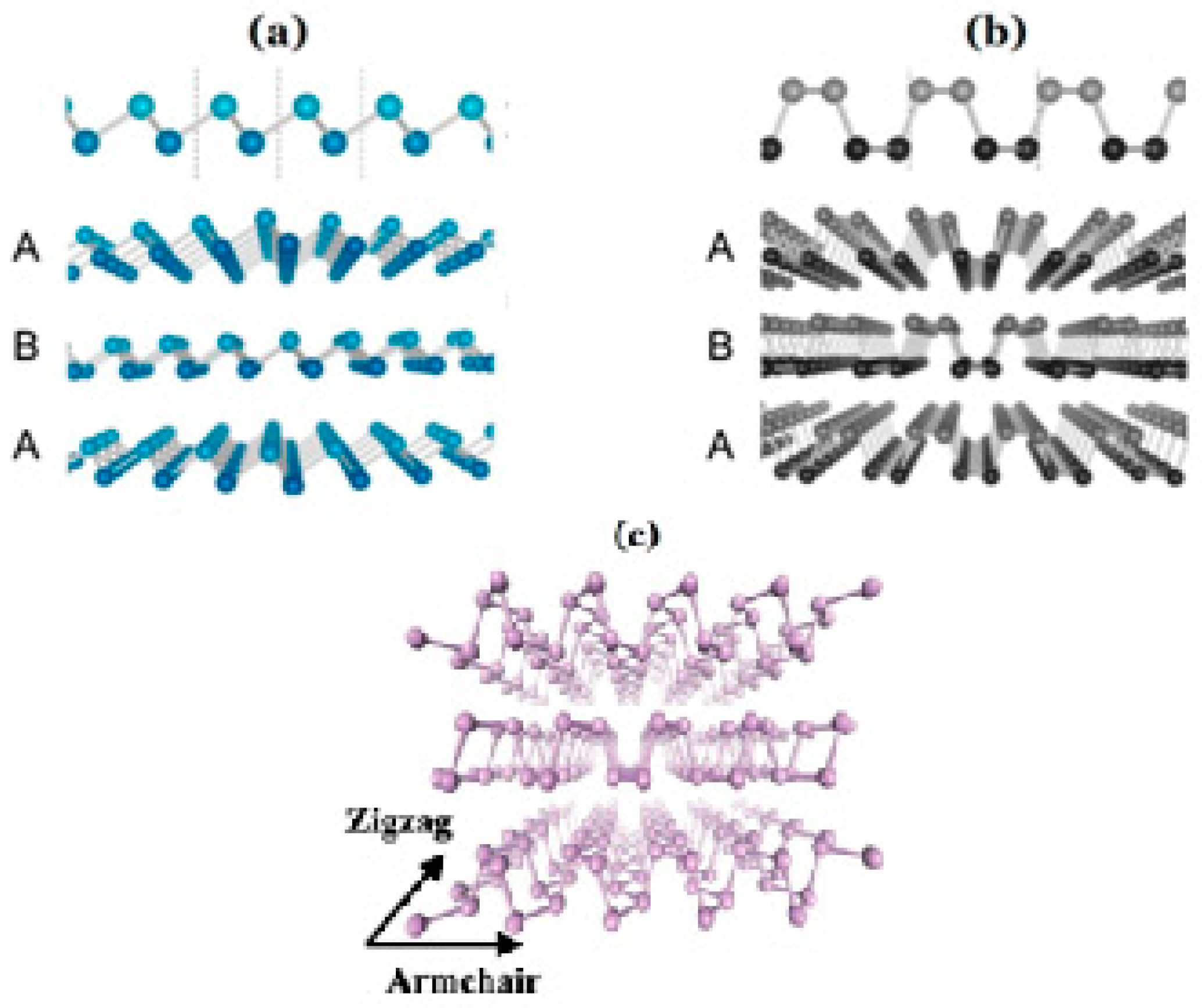
- Yoo, S.; Kim, S.; Song, Y.-W. Lithography-free fabrication of field effect transistor channels with randomly contact-printed black phosphorus flakes. Mater. Sci. Semicond. Process. 2018, 86, 58–62. [Google Scholar] [CrossRef]
- Zhang, R.; Chen, W. Recent advances in graphene-based nanomaterials for fabricating electrochemical hydrogen peroxide sensors. Biosens. Bioelectron. 2017, 89, 249–268. [Google Scholar] [CrossRef]
- Yi, Y.; Yu, X.-F.; Zhou, W.; Wang, J.; Chu, P.K. Two-dimensional black phosphorus: Synthesis, modification, properties, and applications. Mater. Sci. Eng. R Rep. 2017, 120, 1–33. [Google Scholar] [CrossRef]
- Yang, A.J.; Wang, D.W.; Wang, X.H.; Zhang, D.Z.; Koratkar, N.; Rong, M.Z. Recent advances in phosphorene as a sensing material. Nano Today 2018, 20, 13–32. [Google Scholar] [CrossRef]
- Anju, S.; Ashtami, J.; Mohanan, P.V. Black phosphorus, a prospective graphene substitute for biomedical applications. Mater. Sci. Eng. C 2019. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.C.; Kim, C.M.; Jang, H.K.; Lee, J.W.; Joo, M.-K.; Kim, G.-T. Degradation pattern of black phosphorus multilayer field-effect transistors in ambient conditions: Strategy for contact resistance engineering in BP transistors. Appl. Surf. Sci. 2017, 419, 637–641. [Google Scholar] [CrossRef]
- Seo, S.; Park, B.; Kim, Y.; Lee, H.U.; Kim, H.; Lee, S.Y.; Kim, Y.; Won, J.; Kim, Y.J.; Lee, J. Black phosphorus quantum dot-based field-effect transistors with ambipolar characteristics. App. Surf. Sci. 2018, 448, 576–582. [Google Scholar] [CrossRef]
- Zhou, G.; Pu, H.; Chang, J.; Sui, X.; Mao, S.; Chen, J. Real-time electronic sensor based on black phosphorus/Au NPs/DTT hybrid structure: Application in arsenic detection. Sens. Actuators B Chem. 2018, 257, 214–219. [Google Scholar] [CrossRef]
- Guo, Z.; Ding, W.; Liu, X.; Sun, Z.; Wei, L. Two-dimensional black phosphorus: A new star in energy applications and the barrier to stability. Appl. Mater. Today 2019, 14, 51–58. [Google Scholar] [CrossRef]
- Lin, S.; Li, Y.; Qian, J.; Lau, S.P. Emerging opportunities for black phosphorus in energy applications. Mater. Today Energy 2019, 12, 1–25. [Google Scholar] [CrossRef]
- Xu, J.; Song, Y.J.; Park, J.H.; Lee, S. Graphene/black phosphorus heterostructured photodetector. Solid-State Electron. 2018, 144, 86–89. [Google Scholar] [CrossRef]
- Lee, G.; Kim, S.; Jung, S.; Jang, S.; Kim, J. Suspended black phosphorus nanosheet gas sensors. Sens. Actuators B Chem. 2017, 250, 569–573. [Google Scholar] [CrossRef]
- Hu, Z.; Li, Y.; Hussain, E.; Huang, X.; Zhang, Y.; Niu, N.; Shahzad, S.A.; Yu, C. Black phosphorus nanosheets based sensitive protease detection and inhibitor screening. Talanta 2019, 197, 270–276. [Google Scholar] [CrossRef]
- Han, Z.; Wang, J.; Liao, L.; Pan, H.; Shen, S.; Chen, J. Phosphorus doped TiO2 as oxygen sensor with low operating temperature and sensing mechanism. Appl. Surf. Sci. 2013, 273, 349–356. [Google Scholar] [CrossRef]
- Meng, F.; Zheng, H.; Sun, Y.; Li, M.; Liu, J. Trimethylamine Sensors Based on Au-Modified Hierarchical Porous Single-Crystalline ZnO Nanosheets. Sensors 2017, 17, 1478. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Zhang, J.; Meng, F.; Li, Y.; Li, R.; Chang, Y.; Zhao, J.; Han, E.; Wang, S. Highly Sensitive Ammonia Sensors Based on Ag-Decorated WO3Nanorods. IEEE Trans. Nanotechnol. 2018, 17, 1252–1258. [Google Scholar] [CrossRef]
- Deng, X.; Zhang, L.; Guo, J.; Chen, Q.; Ma, J. ZnO enhanced NiO-based gas sensors towards ethanol. Mater. Res. Bull. 2017, 90, 170–174. [Google Scholar] [CrossRef]
- Wang, P.; Dong, T.; Jia, C.; Yang, P. Ultraselective acetone-gas sensor based ZnO flowers functionalized by Au nanoparticle loading on certain facet. Sens. Actuators B Chem. 2019, 288, 1–11. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Zhou, L.; Liu, D.; Liu, F.; Liu, F.; Liang, X.; Yan, X.; Gao, Y.; Lu, G. The role of Ce doping in enhancing sensing performance of ZnO-based gas sensor by adjusting the proportion of oxygen species. Sens. Actuators B Chem. 2018, 273, 991–998. [Google Scholar] [CrossRef]
- Arunkumar, S.; Hou, T.; Kim, Y.-B.; Choi, B.; Park, S.H.; Jung, S.; Lee, D.-W. Au Decorated ZnO hierarchical architectures: Facile synthesis, tunable morphology and enhanced CO detection at room temperature. Sens. Actuators B Chem. 2017, 243, 990–1001. [Google Scholar] [CrossRef]
- Lin, L.; Liu, T.; Zhang, Y.; Liang, X.; Sun, R.; Zeng, W.; Wang, Z. Enhancing ethanol detection by heterostructural silver nanoparticles decorated polycrystalline zinc oxide nanosheets. Ceram. Int. 2016, 42, 3138–3144. [Google Scholar] [CrossRef]
- Liu, X.; Sun, Y.; Yu, M.; Yin, Y.; Du, B.; Tang, W.; Jiang, T.; Yang, B.; Cao, W.; Ashfold, M.N.R. Enhanced ethanol sensing properties of ultrathin ZnO nanosheets decorated with CuO nanoparticles. Sens. Actuators B Chem. 2018, 255, 3384–3390. [Google Scholar] [CrossRef]
- Barbosa, M.S.; Suman, P.H.; Kim, J.J.; Tuller, H.L.; Varela, J.A.; Orlandi, M.O. Gas sensor properties of Ag- and Pd-decorated SnO micro-disks to NO2, H2 and CO: Catalyst enhanced sensor response and selectivity. Sens. Actuators B Chem. 2017, 239, 253–261. [Google Scholar] [CrossRef]
- Nakate, U.T.; Bulakhe, R.N.; Lokhande, C.D.; Kale, S.N. Au sensitized ZnO nanorods for enhanced liquefied petroleum gas sensing properties. Appl. Surf. Sci. 2016, 371, 224–230. [Google Scholar] [CrossRef]
- Kim, J.-H.; Mirzaei, A.; Kim, H.W.; Kim, S.S. Improving the hydrogen sensing properties of SnO2 nanowire-based conductometric sensors by Pd-decoration. Sens. Actuators B Chem. 2019, 285, 358–367. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, B.; Liu, J.; Yang, Q.; Cui, X.; Gao, Y.; Chuai, X.; Liu, F.; Sun, P.; Liang, X.; et al. Au-loaded mesoporous WO3: Preparation and n-butanol sensing performances. Sens. Actuators B Chem. 2016, 236, 67–76. [Google Scholar] [CrossRef]
- Zhang, S.; Song, P.; Zhang, J.; Yan, H.; Li, J.; Yang, Z.; Wang, Q. Highly sensitive detection of acetone using mesoporous In2O3 nanospheres decorated with Au nanoparticles. Sens. Actuators B Chem. 2017, 242, 983–993. [Google Scholar] [CrossRef]
- Choi, M.S.; Bang, J.H.; Mirzaei, A.; Oum, W.; Na, H.G.; Jin, C.; Kim, S.S.; Kim, H.W. Promotional effects of ZnO-branching and Au-functionalization on the surface of SnO2 nanowires for NO2 sensing. J. Alloys Compd. 2019, 786, 27–39. [Google Scholar] [CrossRef]
- Esfandiar, A.; Ghasemi, S.; Irajizad, A.; Akhavan, O.; Gholami, M.R. The decoration of TiO2/reduced graphene oxide by Pd and Pt nanoparticles for hydrogen gas sensing. Int. J. Hydrogen Energy 2012, 3, 15423–15432. [Google Scholar] [CrossRef]
- Fan, F.Y.; Zhang, J.J.; Li, J.; Zhang, N.; Hong, R.R.; Deng, X.C.; Tang, P.G.; Li, D.Q. Hydrogen sensing properties of Pt-Au bimetallic nanoparticles loaded on ZnO nanorods. Sens. Actuators B Chem. 2017, 241, 895–903. [Google Scholar] [CrossRef]
- Ma, R.J.; Li, G.D.; Zou, X.X.; Gao, R.Q.; Chen, H.; Zhao, X. Bimetallic Pt-Au nanocatalysts decorated In2O3 nests composed of ultrathin nanosheets for Type 1 diabetes diagnosis. Sens. Actuators B Chem. 2018, 270, 247–255. [Google Scholar] [CrossRef]
- Pajooheshpour, N.; Rezaei, M.; Hajian, A.; Afkhami, A.; Sillanpaa, M.; Arduini, F.; Bagheri, H. Protein templated Au-Pt nanoclusters-graphene nanoribbons as a high performance sensing layer for the electrochemical determination of diazinon. Sens. Actuators B Chem. 2018, 275, 180–189. [Google Scholar] [CrossRef]
- Hassan, K.; Chung, G.S. Catalytically activated quantum-size Pt/Pd bimetallic core-shell nanoparticles decorated on ZnO nanorod clusters for accelerated hydrogen gas detection. Sens. Actuators B Chem. 2017, 239, 824–833. [Google Scholar] [CrossRef]
- Kutukov, P.; Rumyantseva, M.; Krivetskiy, V.; Filatova, D.; Batuk, M.; Hadermann, J.; Khmelevsky, N.; Aksenenko, A.; Gaskov, A. Influence of Mono- and Bimetallic PtOx, PdOx, PtPdOx Clusters on CO Sensing by SnO2 Based Gas Sensors. Nanomaterials 2018, 8, 917. [Google Scholar] [CrossRef] [PubMed]
- Malkov, I.V.; Krivetskii, V.V.; Potemkin, D.I.; Zadesenets, A.V.; Batuk, M.M.; Hadermann, J.; Marikutsa, A.V.; Rumyantseva, M.N.; Gas’kov, A.M. Effect of Bimetallic Pd/Pt Clusters on the Sensing Properties of Nanocrystalline SnO2 in the Detection of CO. Russ. J. Inorg. Chem. 2018, 63, 1007–1011. [Google Scholar] [CrossRef]
- Nie, Q.; Zhang, W.; Wang, L.R.; Guo, Z.; Li, C.Y.; Yao, J.; Li, M.; Wu, D.M.; Zhou, L.Q. Sensitivity enhanced, stability improved ethanol gas sensor based on multi-wall carbon nanotubes functionalized with Pt-Pd nanoparticles. Sens. Actuators B Chem. 2018, 270, 140–148. [Google Scholar] [CrossRef]
- Rahaman, M.H.; Hassan, K.; Chung, G.-S.; Kim, H.C. Catalytic Behaviors of Pt/Pd Bimetallic Core-shell Nanoparticles Decorated on Different Basal Podium for Fast Response Hydrogen Sensing. IEEE Sens. 2017, 1323–1325. [Google Scholar] [CrossRef]
- Majumdar, S.; Nandi, A.; Saha, H. Synergistic Effects of Dual-Metal Catalysts for Selective Butane Detection by SnO2/Graphene Nanocomposite Sensor. IEEE Sens. J. 2018, 18, 6517–6526. [Google Scholar] [CrossRef]
- Ng, K.C.; Lin, F.C.; Yang, P.W.; Chuang, Y.C.; Chang, C.K.; Yeh, A.H.; Kuo, C.S.; Kao, C.R.; Liu, C.C.; Jeng, U.S.; et al. Fabrication of Bimetallic Au-Pd-Au Nanobricks as an Archetype of Robust Nanoplasmonic Sensors. Chem. Mater. 2018, 30, 204–213. [Google Scholar] [CrossRef]
- Misra, M.; Singh, N.; Gupta, R.K. Enhanced visible-light-driven photocatalytic activity of Au@Ag core-shell bimetallic nanoparticles immobilized on electrospun TiO2 nanofibers for degradation of organic compounds. Catal. Sci. Technol. 2017, 7, 570–580. [Google Scholar] [CrossRef]
- Kim, S.; Park, S.; Park, S.; Lee, C. Acetone sensing of Au and Pd-decorated WO3 nanorod sensors. Sens. Actuators B Chem. 2015, 209, 180–185. [Google Scholar] [CrossRef]
- Motaung, D.E.; Mhlongo, G.H.; Makgwane, P.R.; Dhonge, B.P.; Cummings, F.R.; Swart, H.C.; Ray, S.S. Ultra-high sensitive and selective H2 gas sensor manifested by interface of n-n heterostructure of CeO2-SnO2 nanoparticles. Sens. Actuators B Chem. 2018, 254, 984–995. [Google Scholar] [CrossRef]
- Xu, H.; Ju, J.; Li, W.; Zhang, J.; Wang, J.; Cao, B. Superior triethylamine-sensing properties based on TiO2/SnO2 n-n heterojunction nanosheets directly grown on ceramic tubes. Sens. Actuators B Chem. 2016, 228, 634–642. [Google Scholar] [CrossRef]
- Wang, T.; Kou, X.; Zhao, L.; Sun, P.; Liu, C.; Wang, Y.; Shimanoe, K.; Yarnazoe, N.; Lu, G. Flower-like ZnO hollow microspheres loaded with CdO nanoparticles as high performance sensing material for gas sensors. Sens. Actuators B Chem. 2017, 250, 692–702. [Google Scholar] [CrossRef]
- Li, W.; Ma, S.; Li, Y.; Yang, G.; Mao, Y.; Luo, J.; Gengzang, D.; Xu, X.; Yan, S. Enhanced ethanol sensing performance of hollow ZnO-SnO2 core-shell nanofibers. Sens. Actuators B Chem. 2015, 211, 392–402. [Google Scholar] [CrossRef]
- Ju, D.-X.; Xu, H.-Y.; Qiu, Z.-W.; Zhang, Z.-C.; Xu, O.; Zhang, J.; Wang, J.-Q.; Cao, B.-Q. Near Room Temperature, Fast-Response, and Highly Sensitive Triethylamine Sensor Assembled with Au-Loaded ZnO/SnO2 Core Shell Nanorods on Flat Alumina Substrates. ACS Appl. Mater. Interfaces 2015, 7, 19163–19171. [Google Scholar] [CrossRef]
- Tang, W.; Wang, J. Methanol sensing micro-gas sensors of SnO2-ZnO nanofibers on Si/SiO2/Ti/Pt substrate via stepwise-heating electrospinning. J. Mater. Sci. 2015, 50, 4209–4220. [Google Scholar] [CrossRef]
- Wu, Q.; Yang, H.; Zhu, H.; Gao, Z. Construction of CNCs-TiO2 heterojunctions with enhanced photocatalytic activity for crystal violet removal. Optik 2019, 179, 195–206. [Google Scholar] [CrossRef]
- Wu, X.; Ng, Y.H.; Wen, X.; Chung, H.Y.; Wong, R.J.; Du, Y.; Dou, S.X.; Amal, R.; Scott, J. Construction of a Bi2MoO6:Bi2Mo3O12 heterojunction for efficient photocatalytic oxygen evolution. Chem. Eng. J. 2018, 353, 636–644. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, J.; Zeng, J.; Shi, Y.; Lin, S.; Huang, G.; Wang, H.; Kong, Z.; Xi, J.; Ji, Z. MnS coupled with ultrathin MoS2 nanolayers as heterojunction photocatalyst for high photocatalytic and photoelectrochemical activities. J. Alloys Compd. 2019, 771, 364–372. [Google Scholar] [CrossRef]
- Dolai, S.; Dey, R.; Hussain, S.; Bhar, R.; Kumar Pal, A. Photovoltaic properties of F:SnO2/CdS/CuO/Ag heterojunction solar cell. Mater. Res. Bull. 2019, 109, 1–9. [Google Scholar] [CrossRef]
- Rosas-Laverde, N.M.; Pruna, A.; Busquets-Mataix, D.; Marí, B.; Cembrero, J.; Salas Vicente, F.; Orozco-Messana, J. Improving the properties of Cu2O/ZnO heterojunction for photovoltaic application by graphene oxide. Ceram. Int. 2018, 44, 23045–23051. [Google Scholar] [CrossRef]
- Liu, C.; Zhou, J.; Su, J.; Guo, L. Turning the unwanted surface bismuth enrichment to favourable BiVO4/BiOCl heterojunction for enhanced photoelectrochemical performance. Appl. Catal. B Environ. 2019, 241, 506–513. [Google Scholar] [CrossRef]
- Mao, L.; Ji, K.; Yao, L.; Xue, X.; Wen, W.; Zhang, X.; Wang, S. Molecularly imprinted photoelectrochemical sensor for fumonisin B1 based on GO-CdS heterojunction. Biosens. Bioelectron. 2019, 127, 57–63. [Google Scholar] [CrossRef]
- Zhou, W.; Jiang, T.; Zhao, Y.; Xu, C.; Pei, C.; Xue, H. Ultrathin Ti/TiO2/BiVO4 nanosheet heterojunction arrays for photoelectrochemical water oxidation. J. Alloys Compd. 2019, 777, 1152–1158. [Google Scholar] [CrossRef]
- Martínez-Saucedo, G.; Castanedo-Pérez, R.; Torres-Delgado, G.; Márquez-Marín, J.; Zelaya-Ángel, O. Cuprous oxide/cadmium stannate heterojunction diodes obtained by dip-coating method. J. Alloys Compd. 2019, 774, 153–159. [Google Scholar] [CrossRef]
- Sharma, A.; Tomar, M.; Gupta, V. Low temperature operating SnO2 thin film sensor loaded with WO3 micro-discs with enhanced response for NO2 gas. Sens. Actuators B Chem. 2012, 161, 1114–1118. [Google Scholar] [CrossRef]
- Alali, K.T.; Lu, Z.; Zhang, H.; Liu, J.; Liu, Q.; Li, R.; Aljebawi, K.; Wang, J. P-p heterojunction CuO/CuCo2O4 nanotubes synthesized via electrospinning technology for detecting n-propanol gas at room temperature. Inorg. Chem. Front. 2017, 4, 1219–1230. [Google Scholar] [CrossRef]
- Tai, H.; Xu, X.; Ye, Z.; Liu, C.; Xie, G.; Jiang, Y. P-P heterojunction sensor of self-assembled polyaniline nano-thin film/microstructure silicon array for NH3 detection. Chem. Phys. Lett. 2015, 621, 58–64. [Google Scholar] [CrossRef]
- Gong, J.; Li, Y.; Hu, Z.; Zhou, Z.; Deng, Y. Ultrasensitive NH3 Gas Sensor from Polyaniline Nanograin Enchased TiO2 Fibers. J. Phys. Chem. C 2010, 114, 9970–9974. [Google Scholar] [CrossRef]
- Huang, H.; Gong, H.; Chow, C.L.; Guo, J.; White, T.J.; Tse, M.S.; Tan, O.K. Low-Temperature Growth of SnO2 Nanorod Arrays and Tunable n-p-n Sensing Response of a ZnO/SnO2 Heterojunction for Exclusive Hydrogen Sensors. Adv. Funct. Mater. 2011, 21, 2680–2686. [Google Scholar] [CrossRef]









| Type of Sensitive Material | Type of Aimed Gas | Response(S) |
|---|---|---|
| n-type | oxidizing | |
| reducing | ||
| p-type | oxidizing | |
| reducing |
| The Character of Structure | Typical Morphology | |
|---|---|---|
| 0D structure | three dimensions are in the nanoscale | Nanoparticle [7], quantum dot [8], nanocluster [9] |
| 1D structure | two dimensions are in the nanoscale | Nanowire [10], nanofiber [11], nanorod [12], nanotube [13,14] |
| 2D structure | one dimension is in the nanoscale | Nanosheet [15], nanobelt [16], superlattice [17] |
| 3D structure | assembled by one kind or more low dimensional materials | Nanoflowers [18] |
| Method | Advantages | Disadvantages | Examples |
|---|---|---|---|
| solid phase method | simple synthesis process, high yield, less pollution | uneven distribution of particle size, high agglomeration | ball milling method [21], shear milling method [22] |
| vapor phase method | high purity powder, small particle size, less agglomeration | high cost, high requirements for instruments | molecular beam epitaxy [23], cathode sputtering [24] |
| liquid phase method | simple synthesis process, controllable particle size | low distribution, low uniformity | sol-gel method [25], micro-emulsion method [26], hydrothermal method [27] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, Z.; Li, R.; Meng, F.; Zhang, J.; Zuo, K.; Han, E. Approaches to Enhancing Gas Sensing Properties: A Review. Sensors 2019, 19, 1495. https://doi.org/10.3390/s19071495
Yuan Z, Li R, Meng F, Zhang J, Zuo K, Han E. Approaches to Enhancing Gas Sensing Properties: A Review. Sensors. 2019; 19(7):1495. https://doi.org/10.3390/s19071495
Chicago/Turabian StyleYuan, Zhenyu, Rui Li, Fanli Meng, Junjie Zhang, Kaiyuan Zuo, and Erchou Han. 2019. "Approaches to Enhancing Gas Sensing Properties: A Review" Sensors 19, no. 7: 1495. https://doi.org/10.3390/s19071495
APA StyleYuan, Z., Li, R., Meng, F., Zhang, J., Zuo, K., & Han, E. (2019). Approaches to Enhancing Gas Sensing Properties: A Review. Sensors, 19(7), 1495. https://doi.org/10.3390/s19071495







