Wearable Sensors for Monitoring of Cigarette Smoking in Free-Living: A Systematic Review
Abstract
1. Introduction
2. Review Methodology
2.1. Identifying Research Question
2.2. Source of Studies
2.3. Search Strategy
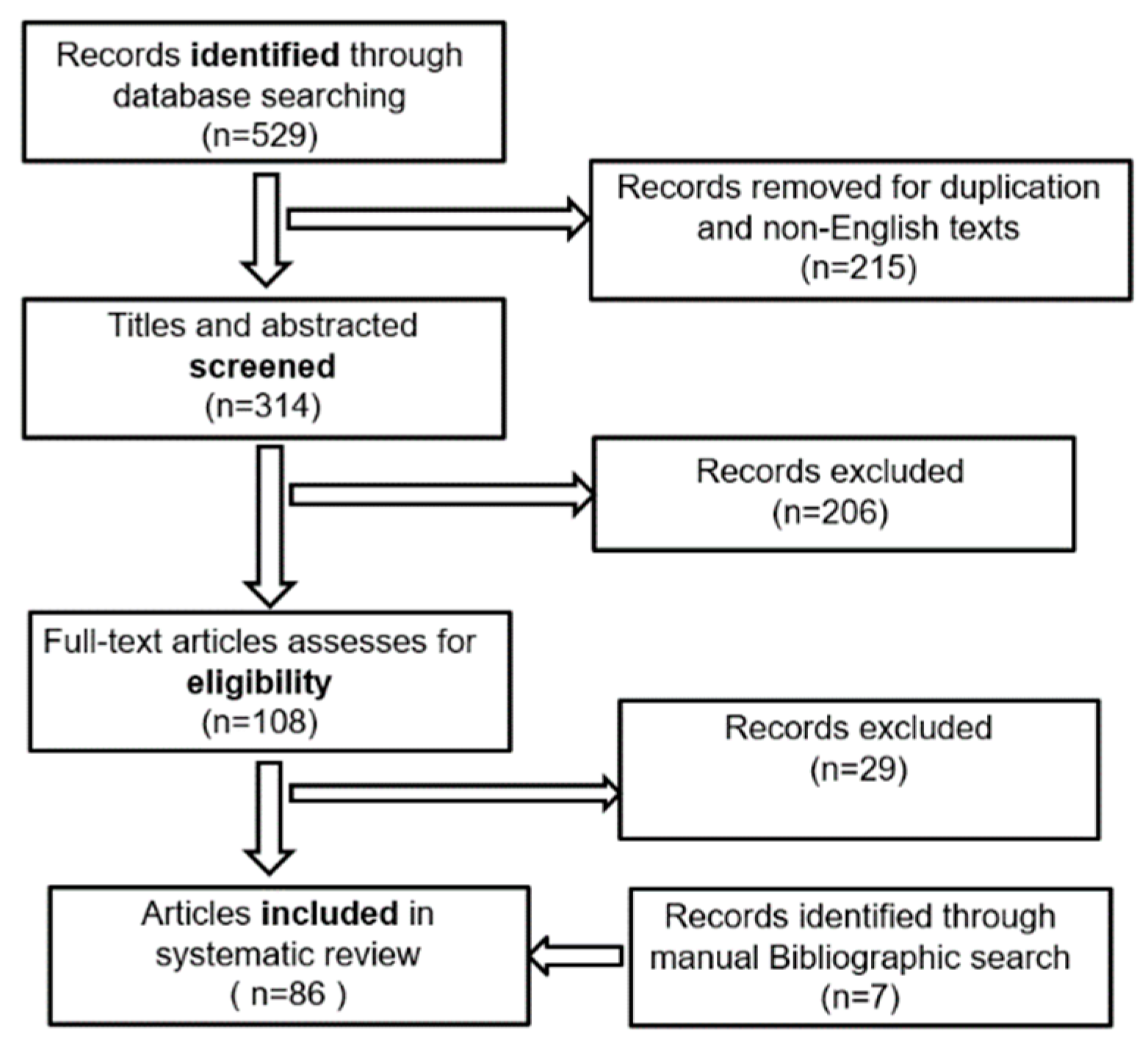
2.4. Results
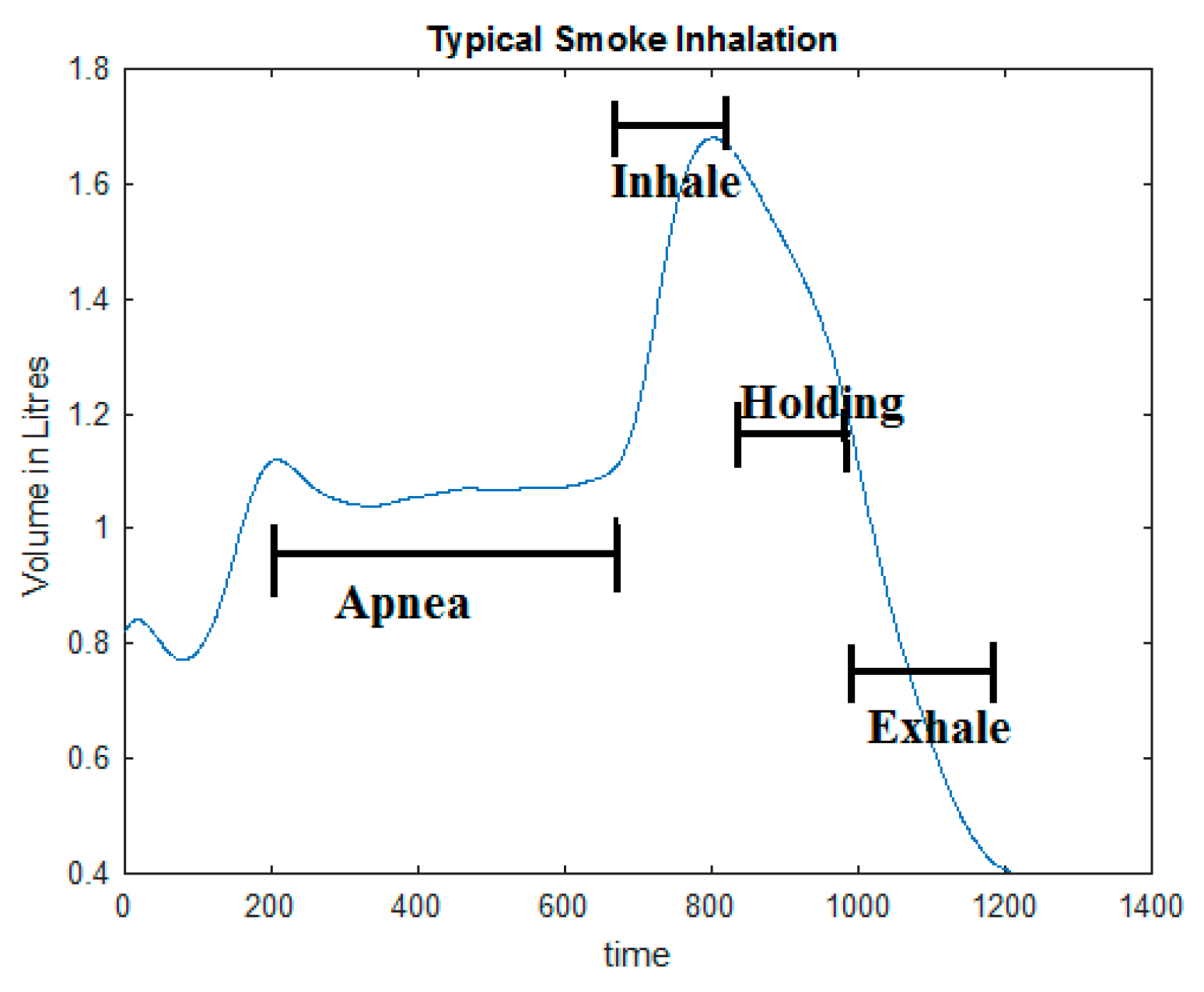
3. Behavioral and Physiological Manifestations of Cigarette Smoking
4. Evaluation of Sensing Methodologies
4.1. Individual Sensor Approach
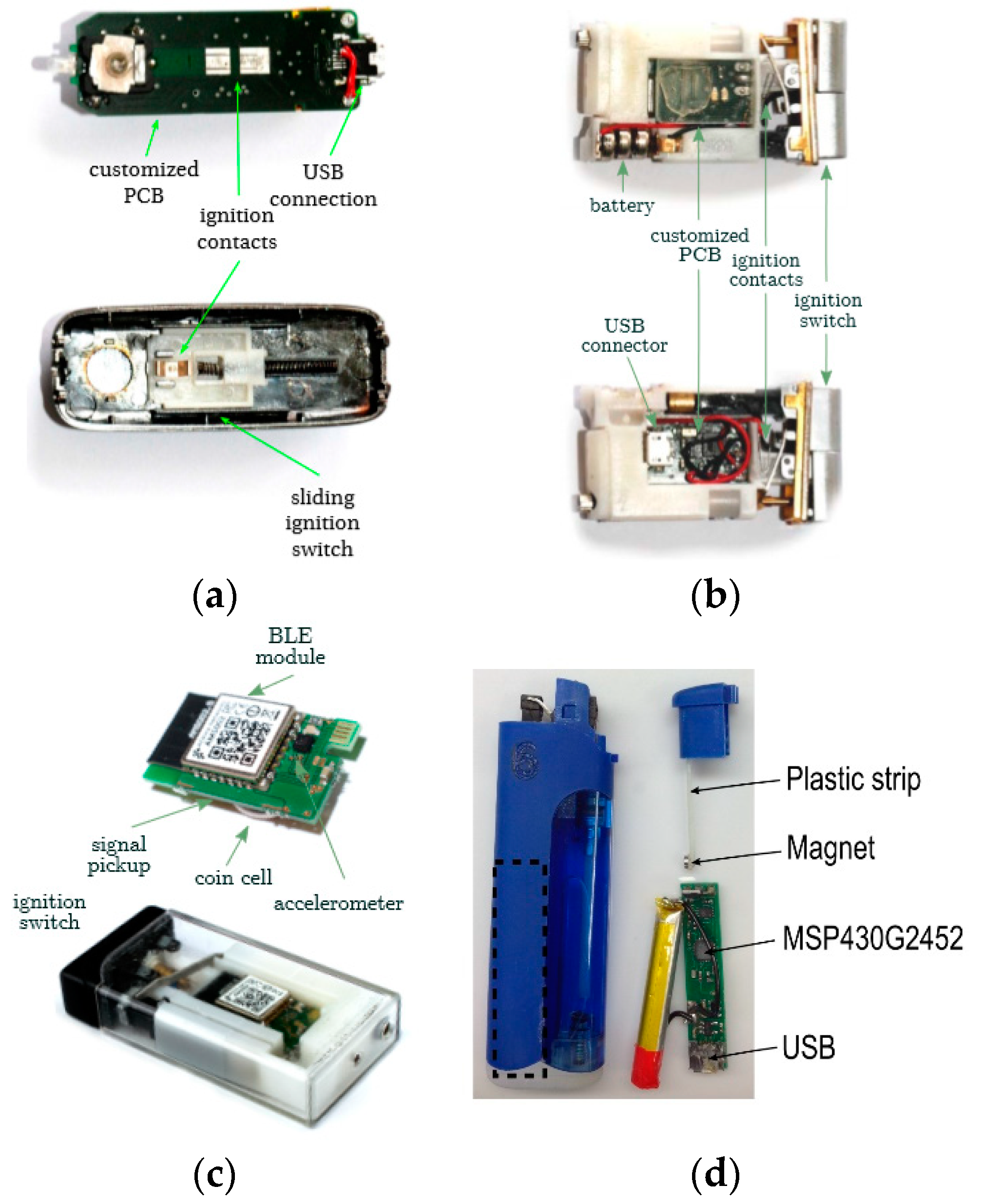
4.1.1. Detection of Smoking Frequency from Cigarette Lighting
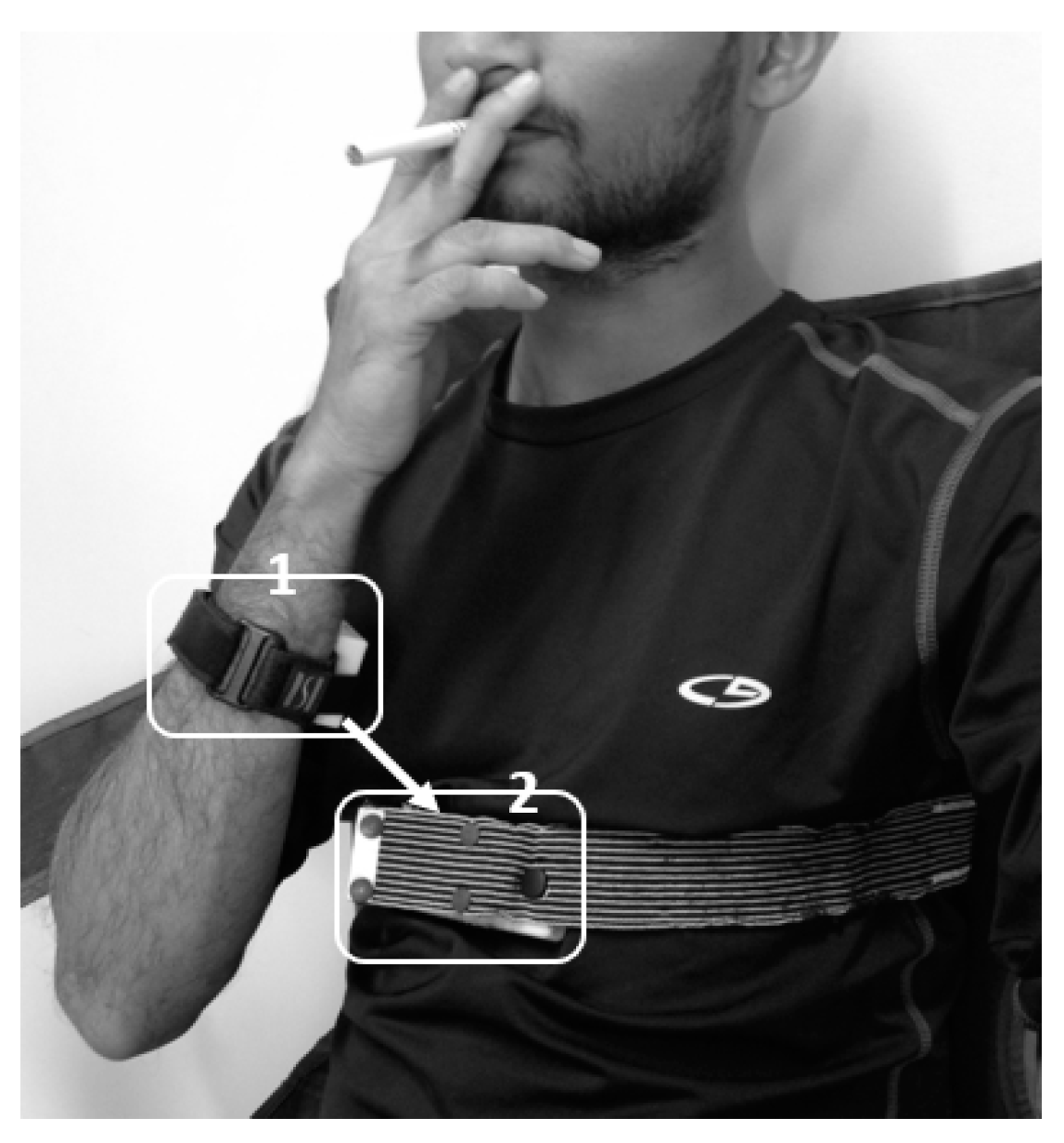
4.1.2. Detection of HMG Preceding Smoking Based on Hand to Mouth Proximity
4.1.3. Detection of Smoking Events and Associated HMGs Based on Linear and Angular Movements of the Hand
4.1.4. Detection of Smoking and Puffs Based on Respiratory Signals
4.1.5. Detection of Smoking Events Based on Acoustic Signals
4.1.6. Detection of Smoking Events Based on Egocentric Camera
4.2. Multi-Sensor Fusion Approach
4.2.1. PACT
4.2.2. AutoSense
4.2.3. PACT2.0
5. Discussion
6. Future Directions
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- WHO. International Classification of Diseases, 11th revision (ICD-11); WHO: Geneva, Switzerland, 2018. Available online: http://www.who.int/classifications/icd/en/ (accessed on 8 July 2018).
- American Cancer Society. Harmful Chemicals in Tobacco Products. Available online: https://www.cancer.org/cancer/cancer-causes/tobacco-and-cancer/carcinogens-found-in-tobacco-products.html (accessed on 8 July 2018).
- National Cancer Institute. Harms of Cigarette Smoking and Health Benefits of Quitting. Available online: https://www.cancer.gov/about-cancer/causes-prevention/risk/tobacco/cessation-fact-sheet (accessed on 8 July 2018).
- C. O. on S. and Health. Smoking and Tobacco Use; Surgeon General’s Reports. 2004. Available online: http://www.cdc.gov/tobacco/data_statistics/sgr/2004/ (accessed on 8 July 2018).
- What Happens to Your Body When You Smoke? Mercola.com. Available online: http://articles.mercola.com/smoking-side-effects.aspx (accessed on 8 July 2018).
- National Survey on Drug Use and Health. Available online: https://nsduhweb.rti.org/respweb/homepage.cfm (accessed on 8 July 2018).
- C. O. on S. and Health. Smoking and Tobacco Use; Fact Sheet; Tobacco-Related Mortality. 9 May 2018. Available online: http://www.cdc.gov/tobacco/data_statistics/fact_sheets/health_effects/tobacco_related_mortality/ (accessed on 8 July 2018).
- C. O. on S. and Health. Smoking and Tobacco Use; Fact Sheet; Health Effects of Secondhand Smoke. 9 May 2018. Available online: http://www.cdc.gov/tobacco/data_statistics/fact_sheets/secondhand_smoke/health_effects/ (accessed on 8 July 2018).
- C. O. on S. and Health. Smoking and Tobacco Use; Fact Sheet. 9 May 2018. Available online: http://www.cdc.gov/tobacco/data_statistics/fact_sheets/fast_facts/ (accessed on 8 July 2018).
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Bishop, E.E.; Kennedy, S.M.; Simpson, S.A.; Pechacek, T.F. Annual Healthcare Spending Attributable to Cigarette Smoking. Am. J. Prev. Med. 2015, 48, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Scholl, P.M.; Kücükyildiz, N.; Laerhoven, K.V. When do you light a fire: Capturing tobacco use with situated, wearable sensors. In Proceedings of the 2013 ACM Conference on Pervasive and Ubiquitous Computing Adjunct Publication, Zurich, Switzerland, 8–12 September 2013; pp. 1295–1304. [Google Scholar]
- Current Cigarette Smoking Among Adults—United States, 2005–2014. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6444a2.htm (accessed on 8 July 2018).
- G. Inc. Most U.S. Smokers Want to Quit, Have Tried Multiple Times. Gallup.com. Available online: https://news.gallup.com/poll/163763/smokers-quit-tried-multiple-times.aspx (accessed on 8 July 2018).
- Gilpin, E.A.; Pierce, J.P.; Farkas, A.J. Duration of smoking abstinence and success in quitting. J. Natl. Cancer Inst. 1997, 89, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Khati, I.; Menvielle, G.; Chollet, A.; Younès, N.; Metadieu, B.; Melchior, M. What distinguishes successful from unsuccessful tobacco smoking cessation? Data from a study of young adults (TEMPO). Prev. Med. Rep. 2015, 2, 679–685. [Google Scholar] [CrossRef]
- Irvin, J.E.; Hendricks, P.S.; Brandon, T.H. The increasing recalcitrance of smokers in clinical trials II: Pharmacotherapy trials. Nicotine Tob. Res. Off. J. Soc. Res. Nicotine Tob. 2003, 5, 27–35. [Google Scholar] [CrossRef]
- Panel, D.G. Treating Tobacco Use and Dependence: 2008 Update; US Department of Health and Human Services: Atlanta, GA, USA, 2008.
- Hartmann-Boyce, J.; Lancaster, T.; Stead, L.F. Print-based self-help interventions for smoking cessation. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef]
- Balanda, K.P.; Lowe, J.B.; O’Connor-Fleming, M.-L. Comparison of two self-help smoking cessation booklets. Tob. Control 1999, 8, 57–61. [Google Scholar] [CrossRef][Green Version]
- Text2Quit: Home. Available online: https://www.text2quit.com/text2quit/Display/display.aspx?CurrentXsltId=5 (accessed on 8 July 2018).
- Michie, S.; Brown, J.; Geraghty, A.W.; Miller, S.; Yardley, L.; Gardner, B.; Shahab, L.; McEwen, A.; Stapleton, J.A.; West, R. Development of StopAdvisor: A theory-based interactive internet-based smoking cessation intervention. Transl. Behav. Med. 2012, 2, 263–275. [Google Scholar] [CrossRef]
- Smoke Free—Quit Smoking Now. Available online: https://smokefreeapp.com/ (accessed on 8 July 2018).
- Smokerlyzer. Available online: https://www.covita.net/smokerlyzer.html (accessed on 8 July 2018).
- Hatziandreu, E.J.; Pierce, J.P.; Fiore, M.C.; Grise, V.; Novotny, T.E.; Davis, R.M. The reliability of self-reported cigarette consumption in the United States. Am. J. Public Health 1989, 79, 1020–1023. [Google Scholar] [CrossRef]
- Hecht, S.S. Similar Uptake of Lung Carcinogens by Smokers of Regular, Light, and Ultralight Cigarettes. Cancer Epidemiol. Biomarkers Prev. 2005, 14, 693–698. [Google Scholar] [CrossRef]
- Velicer, W.F.; Prochaska, J.O.; Rossi, J.S.; Snow, M.G. Assessing outcome in smoking cessation studies. Psychol. Bull. 1992, 111, 23–41. [Google Scholar] [CrossRef] [PubMed]
- Shiffman, S.; Stone, A.A.; Hufford, M.R. Ecological momentary assessment. Annu. Rev. Clin. Psychol. 2008, 4, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Klasnja, P.; Pratt, W. Healthcare in the pocket: Mapping the space of mobile-phone health interventions. J. Biomed. Inform. 2012, 45, 184–198. [Google Scholar] [CrossRef] [PubMed]
- Hatsukami, D.K.; Hertsgaard, L.A.; Vogel, R.I.; Jensen, J.A.; Murphy, S.E.; Hecht, S.S.; Carmella, S.G.; Al’Absi, M.; Joseph, A.M.; Allen, S.S. Reduced Nicotine Content Cigarettes and Nicotine Patch. Cancer Epidemiol. Biomarkers Prev. 2013, 22, 1015–1024. [Google Scholar] [CrossRef]
- Schlam, T.R.; Baker, T.B. Interventions for Tobacco Smoking. Annu. Rev. Clin. Psychol. 2013, 9, 675–702. [Google Scholar] [CrossRef]
- Chamberlain, C.; O’Mara-Eves, A.; Porter, J.; Coleman, T.; Perlen, S.M.; Thomas, J.; McKenzie, J.E. Psychosocial interventions for supporting women to stop smoking in pregnancy. In Cochrane Database of Systematic Reviews; The Cochrane Collaboration, Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2013. [Google Scholar]
- Maciosek, M.V.; LaFrance, A.B.; Dehmer, S.P.; McGree, D.A.; Xu, Z.; Flottemesch, T.J.; Solberg, L.I. Health Benefits and Cost-Effectiveness of Brief Clinician Tobacco Counseling for Youth and Adults. Ann. Fam. Med. 2017, 15, 37–47. [Google Scholar] [CrossRef]
- Schuck, K.; Otten, R.; Kleinjan, M.; Bricker, J.B.; Engels, R.C. Effectiveness of proactive telephone counselling for smoking cessation in parents: Study protocol of a randomized controlled trial. BMC Public Health 2011, 11, 732. [Google Scholar] [CrossRef]
- Patrick, D.L.; Cheadle, A.; Thompson, D.C.; Diehr, P.; Koepsell, T.; Kinne, S. The validity of self-reported smoking: A review and meta-analysis. Am. J. Public Health 1994, 84, 1086–1093. [Google Scholar] [CrossRef]
- Shiffman, S.; Gwaltney, C.J.; Balabanis, M.H.; Liu, K.S.; Paty, J.A.; Kassel, J.D.; Hickcox, M.; Gnys, M. Immediate antecedents of cigarette smoking: An analysis from ecological momentary assessment. J. Abnorm. Psychol. 2002, 111, 531–545. [Google Scholar] [CrossRef]
- Shihadeh, A.; Antonios, C.; Azar, S. A portable, low-resistance puff topography instrument for pulsating, high-flow smoking devices. Behav. Res. Methods 2005, 37, 186–191. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Jacob, P.; Hatsukami, D.; Severson, H.; Hall, S.; Yu, L. Biochemical verification of tobacco use and cessation. Nicotine Tob. Res. 2002, 4, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Höfer, I.; Nil, R.; Bättig, K. Nicotine yield as determinant of smoke exposure indicators and puffing behavior. Pharmacol. Biochem. Behav. 1991, 40, 139–149. [Google Scholar] [CrossRef]
- Gubbi, J.; Marusic, S.; Palaniswami, M. Smoke detection in video using wavelets and support vector machines. Fire Saf. J. 2009, 44, 1110–1115. [Google Scholar] [CrossRef]
- Pavlovic, V.I.; Sharma, R.; Huang, T.S. Visual interpretation of hand gestures for human-computer interaction: A review. IEEE Trans. Pattern Anal. Mach. Intell. 1997, 19, 677–695. [Google Scholar] [CrossRef]
- Stitt, J.P.; Kozlowski, L.T. A System for Automatic Quantification of Cigarette Smoking Behavior. In Proceedings of the 2006 International Conference of the IEEE Engineering in Medicine and Biology Society, Honolulu, HI, USA, 17–21 July 2006; pp. 4771–4774. [Google Scholar]
- CReSS Pocket-Borgwaldt-Hauni GmbH. Available online: https://www.borgwaldt.com/en/products/smoking-machines/smoking-topography-devices/cress-pocket.html (accessed on 8 July 2018).
- Brinkman, M.C.; Kim, H.; Chuang, J.C.; Kroeger, R.R.; Deojay, D.; Clark, P.I.; Gordon, S.M. Comparison of True and Smoothed Puff Profile Replication on Smoking Behavior and Mainstream Smoke Emissions. Chem. Res. Toxicol. 2015, 28, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Hsieh, J.; Cheng, J.; Cheng, S.; Tseng, S. Human Smoking Event Detection Using Visual Interaction Clues. In Proceedings of the 2010 20th International Conference on Pattern Recognition, Istanbul, Turkey, 3–26 August 2010; pp. 4344–4347. [Google Scholar]
- Mukhopadhyay, S.C. Wearable Sensors for Human Activity Monitoring: A Review. IEEE Sens. J. 2015, 15, 1321–1330. [Google Scholar] [CrossRef]
- Li, I.; Dey, A.K.; Forlizzi, J. Understanding My Data, Myself: Supporting Self-reflection with Ubicomp Technologies. In Proceedings of the 13th International Conference on Ubiquitous Computing, New York, NY, USA, 8 November–1 December 2011; pp. 405–414. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- West, R. Tobacco smoking: Health impact, prevalence, correlates and interventions. Psychol. Health 2017, 32, 1018–1036. [Google Scholar] [CrossRef]
- Shiffman, S.; Rathbun, S.L. Point process analyses of variations in smoking rate by setting, mood, gender, and dependence. Psychol. Addict. Behav. 2011, 25, 501–510. [Google Scholar] [CrossRef]
- Peretti-Watel, P.; Constance, J.; Guilbert, P.; Gautier, A.; Beck, F.; Moatti, J.-P. Smoking too few cigarettes to be at risk? Smokers’ perceptions of risk and risk denial, a French survey. Tob. Control 2007, 16, 351–356. [Google Scholar] [CrossRef]
- Griffiths, R.R.; Henningfield, J.E.; Bigelow, G.E. Human cigarette smoking: Manipulation of number of puffs per bout, interbout interval and nicotine dose. J. Pharmacol. Exp. Ther. 1982, 220, 256–265. [Google Scholar] [PubMed]
- Sazonov, E.; Metcalfe, K.; Lopez-Meyer, P.; Tiffany, S. RF hand gesture sensor for monitoring of cigarette smoking. In Proceedings of the 2011 Fifth International Conference on Sensing Technology, Palmerston North, New Zealand, 28 November–1 December 2011; pp. 426–430. [Google Scholar]
- Scholl, P.M.; van Laerhoven, K. A Feasibility Study of Wrist-Worn Accelerometer Based Detection of Smoking Habits. In Proceedings of the 2012 Sixth International Conference on Innovative Mobile and Internet Services in Ubiquitous Computing, Palermo, Italy, 4–6 July 2012; pp. 886–891. [Google Scholar]
- How to Smoke a Cigarette. Wikihow. Available online: https://www.wikihow.com/Smoke-a-Cigarette (accessed on 8 July 2018).
- Rodenstein, D.O.; Stănescu, D.C. Pattern of inhalation of tobacco smoke in pipe, cigarette, and never smokers. Am. Rev. Respir. Dis. 1985, 132, 628–632. [Google Scholar] [PubMed]
- Echebarria, I.T.U.; Imtiaz, S.A.; Peng, M.; Rodriguez-Villegas, E. Monitoring smoking behaviour using a wearable acoustic sensor. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Seogwipo, Korea, 11–15 July 2017; pp. 4459–4462. [Google Scholar]
- Omvik, P. How smoking affects blood pressure. Blood Press. 1996, 5, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, S.; Bhatt, K.; Dubey, A.K.; Roy, A.; Singh, S.; Naik, N.; Seth, S.; Bhargava, B. Acute electrocardiographic changes during smoking: An observational study. BMJ Open. 2013, 3, e002486. [Google Scholar] [CrossRef] [PubMed]
- Scholl, P.M.; van Laerhoven, K. Lessons learned from designing an instrumented lighter for assessing smoking status. In Proceedings of the 2017 ACM International Joint Conference on Pervasive and Ubiquitous Computing and Proceedings of the 2017 ACM International Symposium on Wearable Computers on-UbiComp ’17, Maui, HI, USA, 11–15 September 2017; pp. 1016–1021. [Google Scholar]
- Imtiaz, M.; Ramos-Garcia, R.; Senyurek, V.; Tiffany, S.; Sazonov, E. Development of a Multisensory Wearable System for Monitoring Cigarette Smoking Behavior in Free-Living Conditions. Electronics 2017, 6, 104. [Google Scholar] [CrossRef] [PubMed]
- Quitbit|Smart Lighters to Help Track, Reduce & Quit Smoking. Available online: http://www.quitbitlighter.com/ (accessed on 8 July 2018).
- Lopez-Meyer, P. Detection of Hand-to-Mouth Gestures Using a RF Operated Proximity Sensor for Monitoring Cigarette Smoking. Open Biomed. Eng. J. 2013, 7, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Vidrine, D.; Crowder, E.; Intille, S. Automated Detection of Puffing and Smoking with Wrist Accelerometers. In Proceedings of the 8th International Conference on Pervasive Computing Technologies for Healthcare, Oldenburg, Germany, 20–23 May 2014. [Google Scholar]
- Cole, C.A.; Thrasher, J.F.; Strayer, S.M.; Valafar, H. Resolving ambiguities in accelerometer data due to location of sensor on wrist in application to detection of smoking gesture. In Proceedings of the 2017 IEEE EMBS International Conference on Biomedical Health Informatics (BHI), Orlando, FL, USA, 16–19 February 2017; pp. 489–492. [Google Scholar]
- Cole, C.A.; Anshari, D.; Lambert, V.; Thrasher, J.F.; Valafar, H. Detecting Smoking Events Using Accelerometer Data Collected Via Smartwatch Technology: Validation Study. JMIR mHealth uHealth 2017, 5, e189. [Google Scholar] [CrossRef]
- Shoaib, M.; Scholten, H.; Havinga, P.J.M.; Incel, O.D. A hierarchical lazy smoking detection algorithm using smartwatch sensors. In Proceedings of the 2016 IEEE 18th International Conference on e-Health Networking, Applications and Services (Healthcom), Munich, Germany, 14–17 September 2016; pp. 1–6. [Google Scholar]
- Añazco, E.V.; Lopez, P.R.; Lee, S.; Byun, K.; Kim, T.-S. Smoking Activity Recognition Using a Single Wrist IMU and Deep Learning Light. In Proceedings of the 2nd International Conference on Digital Signal Processing—ICDSP 2018, Tokyo, Japan, 25–27 February 2018; pp. 48–51. [Google Scholar]
- SmokeBeat—Apps on Google Play. Available online: https://play.google.com/store/apps/details?id=com.somatix.smokebeat&hl=en_US (accessed on 18 August 2019).
- Skinner, A.L.; Stone, C.J.; Doughty, H.; Munafò, M.R. StopWatch: The Preliminary Evaluation of a Smartwatch-Based System for Passive Detection of Cigarette Smoking. Nicotine Tob. Res. 2018, 21, 257–261. [Google Scholar] [CrossRef]
- Bhandari, B.; Lu, J.; Zheng, X.; Rajasegarar, S.; Karmakar, C. Non-invasive sensor based automated smoking activity detection. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Seogwipo, Korea, 11–15 July 2017; pp. 845–848. [Google Scholar]
- Raiff, B.; Karataş, Ç.; McClure, E.; Pompili, D.; Walls, T. Laboratory Validation of Inertial Body Sensors to Detect Cigarette Smoking Arm Movements. Electronics 2014, 3, 87–110. [Google Scholar] [CrossRef]
- Lu, J.; Wang, J.; Zheng, X.; Karmakar, C.; Rajasegarar, S. Detection of Smoking Events from Confounding Activities of Daily Living. In Proceedings of the Australasian Computer Science Week Multiconference on ACSW 2019, Sydney, Australia, 29–31 January 2019; pp. 1–9. [Google Scholar]
- Parate, A.; Chiu, M.-C.; Chadowitz, C.; Ganesan, D.; Kalogerakis, E. RisQ: Recognizing smoking gestures with inertial sensors on a wristband. In Proceedings of the 12th annual international conference on Mobile systems, applications, and services—MobiSys ’14, Bretton Woods, NH, USA, 16–19 June 2014; pp. 149–161. [Google Scholar]
- Kim, A.; Golnaraghi, M.F. A quaternion-based orientation estimation algorithm using an inertial measurement unit. In Proceedings of the PLANS 2004. Position Location and Navigation Symposium (IEEE Cat. No. 04CH37556), Monterey, CA, USA, 26–29 April 2004; pp. 268–272. [Google Scholar]
- Akyazi, O.; Batmaz, S.; Kosucu, B.; Arnrich, B. SmokeWatch: A smartwatch smoking cessation assistant. In Proceedings of the 2017 25th Signal Processing and Communications Applications Conference (SIU), Antalya, Turkey, 15–18 May 2017; pp. 1–4. [Google Scholar]
- Parate, A.; Ganesan, D. Detecting Eating and Smoking Behaviors Using Smartwatches. In Mobile Health; Rehg, J.M., Murphy, S.A., Kumar, S., Eds.; Springer International Publishing: Cham, Germany, 2017; pp. 175–201. [Google Scholar]
- Petrović, M.; Petrovic, J.; Simić, G.; Ilić, I.; Daničić, A.; Vukčević, M.; Bojović, B.; Hadžievski, L.; Allsop, T.; Webb, D.J. A new method for respiratory-volume monitoring based on long-period fibre gratings. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 2660–2663. [Google Scholar]
- Chen, G.; de la Cruz, I.; Rodriguez-Villegas, E. Automatic lung tidal volumes estimation from tracheal sounds. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 1497–1500. [Google Scholar]
- Goto, E.; Noshiro, M.; Hosaka, H.; Minamitani, H. Ambulatory non-calibration measurement of tidal volume using optical fiber sensors. In Proceedings of the 17th International Conference of the Engineering in Medicine and Biology Society, Montreal, QC, Canada, 20–24 September 1995; pp. 1037–1038. [Google Scholar]
- Retory, Y.; Niedzialkowski, P.; de Picciotto, C.; Bonay, M.; Petitjean, M. New Respiratory Inductive Plethysmography (RIP) Method for Evaluating Ventilatory Adaptation during Mild Physical Activities. PLoS ONE 2016, 11, e0151983. [Google Scholar] [CrossRef]
- Zhang, Z.; Zheng, J.; Wu, H.; Wang, W.; Wang, B.; Liu, H. Development of a Respiratory Inductive Plethysmography Module Supporting Multiple Sensors for Wearable Systems. Sensors 2012, 12, 13167–13184. [Google Scholar] [CrossRef] [PubMed]
- Cohn, M.A.; Rao, A.S.; Broudy, M.; Birch, S.; Watson, H.; Atkins, N.; Davis, B.; Stott, F.D.; Sackner, M.A. The respiratory inductive plethysmograph: A new non-invasive monitor of respiration. Bull. Eur. Physiopathol. Respir. 1982, 18, 643–658. [Google Scholar] [PubMed]
- Fiamma, M.-N.; Samara, Z.; Baconnier, P.; Similowski, T.; Straus, C. Respiratory inductive plethysmography to assess respiratory variability and complexity in humans. Respir. Physiol. Neurobiol. 2007, 156, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Rymut, R.; Slotty, E.; Kini, N. Method and Apparatus for Monitoring Respiration Using Signals from a Piezoelectric Sensor Mounted on a Substrate. U.S. Patent 6517497B2, 11 February 2003. [Google Scholar]
- Tobin, M.J.; Jenouri, G.; Sackner, M.A. Subjective and objective measurement of cigarette smoke inhalation. Chest 1982, 82, 696–700. [Google Scholar] [CrossRef]
- Taylor, D.R.; Reid, W.D.; Pare, P.D.; Fleetham, J.A. Cigarette smoke inhalation patterns and bronchial reactivity. Thorax 1988, 43, 65–70. [Google Scholar] [CrossRef]
- Charles, F.K.; Krautter, G.R.; Mariner, D.C. Post-puff respiration measures on smokers of different tar yield cigarettes. Inhal. Toxicol. 2009, 21, 712–718. [Google Scholar] [CrossRef]
- Duffty, P.; Spriet, L.; Bryan, M.H.; Bryan, A.C. Respiratory induction plethysmography (Respitrace): An evaluation of its use in the infant. Am. Rev. Respir. Dis. 1981, 123, 542–546. [Google Scholar]
- Sazonov, E.; Lopez-Meyer, P.; Tiffany, S. A wearable sensor system for monitoring cigarette smoking. J. Stud. Alcohol Drugs 2013, 74, 956–964. [Google Scholar] [CrossRef]
- Ali, A.A.; Hossain, S.M.; Hovsepian, K.; Rahman, M.M.; Plarre, K.; Kumar, S. mPuff: Automated detection of cigarette smoking puffs from respiration measurements. In Proceedings of the 2012 ACM/IEEE 11th International Conference on Information Processing in Sensor Networks (IPSN), Beijing, China, 16–19 April 2012; pp. 269–280. [Google Scholar]
- Nongpoh, B.; Ray, R.; Dutta, S.; Banerjee, A. AutoSense: A Framework for Automated Sensitivity Analysis of Program Data. IEEE Trans. Softw. Eng. 2017, 43, 1110–1124. [Google Scholar] [CrossRef]
- Ramos-Garcia, R.I.; Imtiaz, M.H.; Sazonov, E.; Tiffany, S.T. Evaluation of RIP sensor calibration stability for daily estimation of lung volume. In Proceedings of the 2017 Eleventh International Conference on Sensing Technology (ICST), Sydney, Australia, 4–6 December 2017; pp. 1–5. [Google Scholar]
- Wang, H.; Yen, C.; Liang, J.; Wang, Q.; Liu, G.; Song, R. A Robust Electrode Configuration for Bioimpedance Measurement of Respiration. J. Healthc. Eng. 2014, 5, 313–328. [Google Scholar] [CrossRef]
- Lopez-Meyer, P.; Sazonov, E. Automatic breathing segmentation from wearable respiration sensors. In Proceedings of the 2011 Fifth International Conference on Sensing Technology, Palmerston North, New Zealand, 28 November–1 December 2011; pp. 156–160. [Google Scholar]
- Ramos-Garcia, R.I.; Tiffany, S.; Sazonov, E. Using respiratory signals for the recognition of human activities. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; pp. 173–176. [Google Scholar]
- Senyurek, V.Y.; Imtiaz, M.H.; Belsare, P.; Tiffany, S.; Sazonov, E. A Comparison of SVM and CNN-LSTM Based Approach for Detecting Smoke Inhalations from Respiratory signal. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 3262–3265. [Google Scholar]
- Belsare, P.; Senyurek, V.Y.; Imtiaz, M.H.; Ti_any, S.; Sazonov, E. Computation of Cigarette Smoke Exposure Metrics from Breathing. IEEE Trans. Biomed. Eng. submitted July 2019.
- Cui, J.; Wang, L.; Gu, T.; Tao, X.; Lu, J. An Audio-based Hierarchical Smoking Behavior Detection System Based on A Smart Neckband Platform. In Proceedings of the 13th International Conference on Mobile and Ubiquitous Systems: Computing, Networking and Services, New York, NY, USA, 28 November–1 December 2016; pp. 190–199. [Google Scholar]
- Imtiaz, M.H.; Hossain, D.; Senyurek, V.Y.; Belsare, P.; Ti_any, S.; Sazonov, E. Validation of a Wearable Egocentric Camera as a Monitoring Tool of Free-living Cigarette Smoking. J. Nicotine Tob. Res. Accepted October 2019.
- Imtiaz, M.H.; Hossain, D.; Senyurek, V.Y.; Ti_any, S.; Sazonov, E. Estimation of Behavioural Metrics of Cigarette Smoking from a Wearable Egocentric Camera. IEEE J. Biomed. Health Inform. submitted August 2019.
- Lopez-Meyer, P.; Tiffany, S.; Patil, Y.; Sazonov, E. Monitoring of Cigarette Smoking Using Wearable Sensors and Support Vector Machines. IEEE Trans. Biomed. Eng. 2013, 60, 1867–1872. [Google Scholar] [CrossRef] [PubMed]
- Patil, Y.; Lopez-Meyer, P.; Tiffany, S.; Sazonov, E. Detection of cigarette smoke inhalations from respiratory signals using reduced feature set. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 6031–6034. [Google Scholar]
- Patil, Y.; Tiffany, S.; Sazonov, E. Understanding smoking behavior using wearable sensors: Relative importance of various sensor modalities. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 6899–6902. [Google Scholar]
- Berry, D.; Bell, J.; Sazonov, E. Detection of cigarette smoke inhalations from respiratory signals using decision tree ensembles. SoutheastCon 2015, 2015, 1–4. [Google Scholar]
- Lopez-Meyer, P.; Tiffany, S.; Sazonov, E. Identification of cigarette smoke inhalations from wearable sensor data using a Support Vector Machine classifier. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 4050–4053. [Google Scholar]
- Saleheen, N.; Ali, A.A.; Hossain, S.M.; Sarker, H.; Chatterjee, S.; Marlin, B.; Ertin, E.; Al’Absi, M.; Kumar, S. puffMarker: A Multi-Sensor Approach for Pinpointing the Timing of First Lapse in Smoking Cessation. In Proceedings of the 2015 ACM International Joint Conference on Pervasive and Ubiquitous Computing, Osaka, Japan, 7–11 September 2015; pp. 999–1010. [Google Scholar]
- Senyurek, V.; Imtiaz, M.; Belsare, P.; Tiffany, S.; Sazonov, E. Cigarette Smoking Detection with an Inertial Sensor and A Smart Lighter. Sensors 2019, 19, 570. [Google Scholar] [CrossRef]
- Senyurek, V.Y.; Imtiaz, M.H.; Belsare, P.; Tiffany, S.; Sazonov, E. Smoking detection based on regularity analysis of hand to mouth gestures. Biomed. Signal Process. Control 2019, 51, 106–112. [Google Scholar] [CrossRef]
- Senyurek, V.Y.; Imtiaz, M.H.; Belsare, P.; Ti_any, S.; Sazonov, E. A CNN-LSTM Neural Network for Recognition Cigarette Smoking Inhalation in Smoking Events Using Wearable Sensors. Submitted to the Biomedical Engineering Letters. Springer.
- Imtiaz, M.H.; Senyurek, V.Y.; Belsare, P.; Tiffany, S.; Sazonov, E. Objective Detection of Cigarette Smoking from Physiological Sensor Signals. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 3563–3566. [Google Scholar]
- Wattal, S.; Spear, S.K.; Imtiaz, M.H.; Sazonov, E. A polypyrrole-coated textile electrode and connector for wearable ECG monitoring. In Proceedings of the 2018 IEEE 15th International Conference on Wearable and Implantable Body Sensor Networks (BSN), Las Vegas, NV, USA, 4–7 March 2018; pp. 54–57. [Google Scholar]
- Chen, T.; Zhang, X.; Jiang, H.; Asaeikheybari, G.; Goel, N.; Hooper, M.W.; Huang, M.C. Are you smoking? Automatic alert system helping people keep away from cigarettes. Smart Health 2018, 9–10, 158–169. [Google Scholar]
- McClernon, F.J.; Choudhury, R.R. I Am Your Smartphone, and I Know You Are About to Smoke: The Application of Mobile Sensing and Computing Approaches to Smoking Research and Treatment. Nicotine Tob. Res. 2013, 15, 1651–1654. [Google Scholar] [CrossRef]
- Qin, Y.; Qian, W.; Shojaati, N.; Osgood, N. Identifying Smoking from Smartphone Sensor Data and Multivariate Hidden Markov Models. In Social, Cultural, and Behavioral Modeling; Lee, D., Lin, Y.-R., Osgood, N., Thomson, R., Eds.; Springer International Publishing: Cham, Germany, 2017; Volume 10354, pp. 230–235. [Google Scholar]
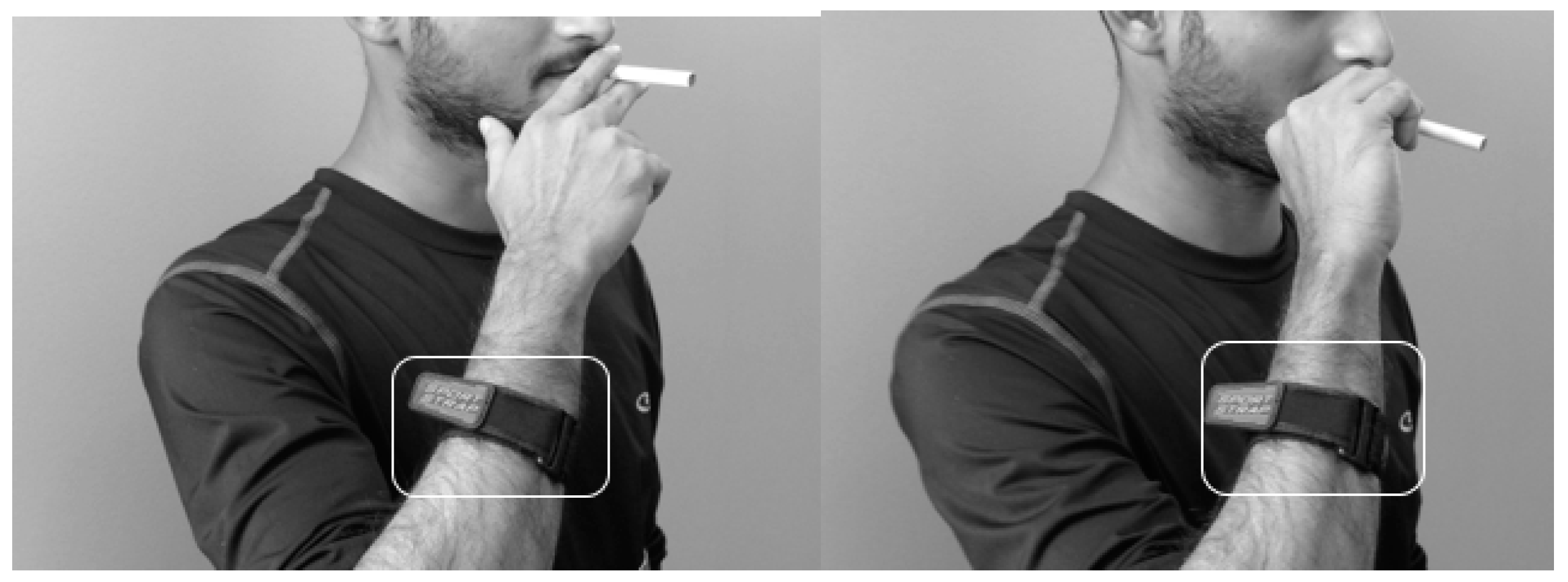

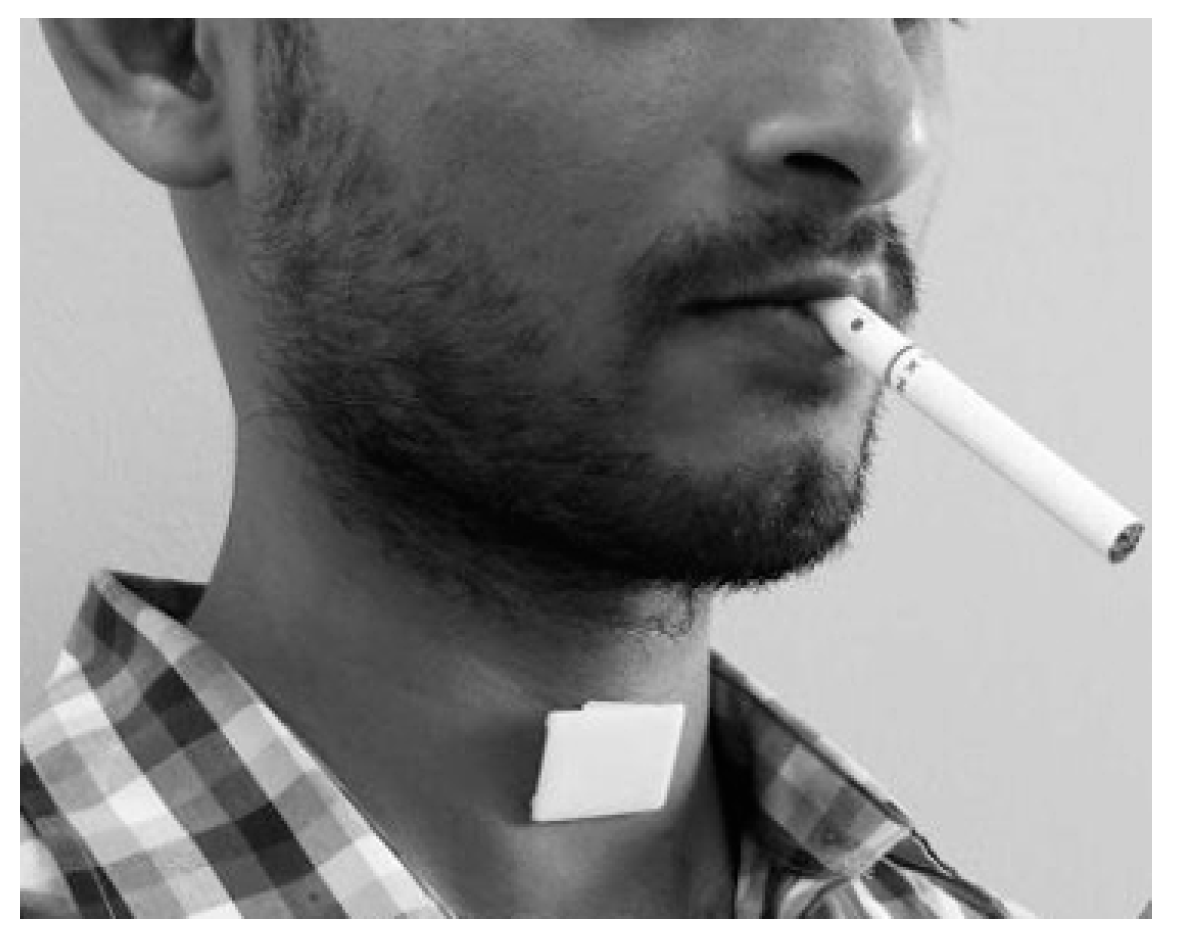
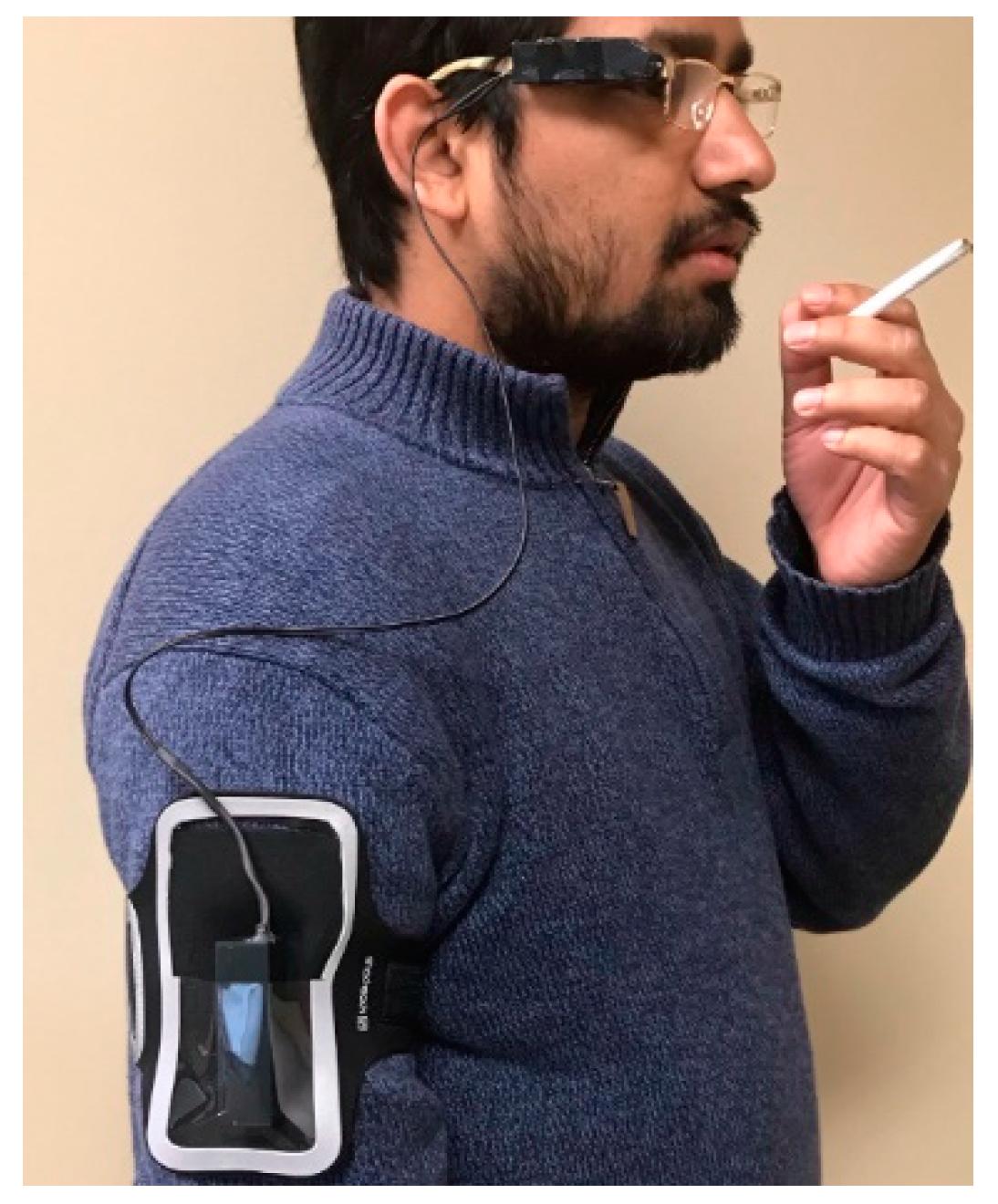
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| 1. Articles published in peer-reviewed venues. | 1. Articles that considered tobacco smoking, other than using cigarettes. |
| 2. Articles published since 1990. | 2. Papers not written in English. |
| 3. Articles must address a certain combination of words, i.e., (cigarette smoking/ smoking detection) + (sensor/ wearable) + (validation/ participant/ subject / human study). | 3. Detection system other than first smoke. |
| 4. Portable systems with embedded wearable sensors. | 4. Subjects under the age of 18 years. |
| Article Types | Total Articles |
|---|---|
| Articles describe the self-reporting of cigarette smoking | 16 |
| Articles describe CO-measurement and biomarker-based approaches | 10 |
| Articles describe wearable and surveillance-video camera-based approaches | 5 |
| Phenomena Used for Smoking Detection | Number of Published Papers | ||||||
|---|---|---|---|---|---|---|---|
| <2007 | 2007–2009 | 2010–2011 | 2012–2013 | 2014–2015 | 2016–2019 | Total | |
| Cigarette Packet | - | - | - | - | - | - | 0 |
| Lighting Event | - | - | - | 1 | 1 | 2 | 4 |
| Hand to Mouth Proximity | - | - | 1 | 5 | 1 | 1 | 8 |
| Smoking Hand Gestures | - | - | - | 4 | 4 | 11 | 19 |
| Smoking-specific respiration pattern | 3 | - | 2 | 5 | 2 | 4 | 16 |
| Breathing Sound | - | - | - | - | - | 2 | 2 |
| Egocentric Vision | - | - | - | - | - | 2 | 2 |
| Total Publications (By year) | 3 | - | 3 | 15 | 8 | 22 | 51 |
| Type | Versions | Type | Lighting Mechanism | Features/Limitation | Microcontroller | Interface | Battery | Validation Study |
|---|---|---|---|---|---|---|---|---|
| Ubi-Lighter [60] | V1 | Electric Coil | Slide Down Switch | Often hard to light up | Atmega32U2 | Universal Serial Bus (USB) | 200 mAh | 3 subjects (11.36 ± 8.15 days), Free-living |
| V2 | Gas Lighter | Push Switch | One-time usage device | Atmega32U2 | USB | 30 mAh | 8 subjects (11.36 ± 8.15 days), Free-living | |
| V3 | Piezo-Ignition based | Push Switch | Contact-less data transmission via Bluetooth Low Energy (BLE) | Atmega32U2 | USB, BLE | 48 mAh | - | |
| PACT [61] | Gas Lighter | Push Switch | Hall sensor based | MSP430G2452 | USB | 210 mAh | 40 subjects (24 h each), Free-living |
| Ref | Transmitter Circuit | Receiver | Transmitter Antenna | Receiver Antenna | Data Storage | Validation Study |
| [53] | Simple sine wave oscillator with a rectangular loop antenna | Large receiver module | 40 × 15 × 5 mm, 860 uH ± 10%, 13 ohms (Sonmicro) | 100 × 110 × 5 mm, 1080 uH, and 8.3 ohms (Sonmicro) | Logomatic V2.0, Sparkfun Electronics (commercial data-logger) | 20 subjects in the lab |
| [61] | Tank circuit, opposite ends of the series antenna are connected to an MCU, two 180° phase shifted PWM outputs (50% duty cycle) | Compa-ratively small receiver module | 7.2 mH ± 2%, 91-ohm transponder coil (Coilcraft) | 7.2 mH ± 2%, 91-ohm transponder coil (Coilcraft) | Embedded data logger with STM32 MCU | 40 subjects both in the lab and free-living |
| Ref | IMU Type | Sensor Chip | Employed IMU Range | MCU | Sampling Frequency | Data Access | Validation Study |
|---|---|---|---|---|---|---|---|
| [54] | 3D | ADXL345 on the ‘Hedgehog’ platform | ± 2g | PIC18F | 20 Hz | Embedded SD card | 4 subjects |
| [69] | 6D | MMA7260Q on the ShimmerTM Platform | ± 6g and ± 500 degree/s | ShimmerTM Platform | 50 Hz | Wirelessly Transmitted | 6 subjects |
| [70] | 9D | MPU-9150 | - | - | 50 Hz | Wirelessly Transmitted | 19 subjects |
| [61] | 6D | LSM6DS3 | ± 8g and 2000 dps | STM32L151RD | 100 Hz | Embedded SD card | 40 subjects |
| Ref | IMU Type | Pre-processing | Candidate Selection | Window Size | No of Extracted Feature | No of Selected Feature | Classifier | Detection | Validation |
|---|---|---|---|---|---|---|---|---|---|
| [54] | 3D | equalized ripple (equi- ripple) FIR low-pass filter (fc = 1 Hz) | Y-axis accelerometer | 5.4 sec | 4 | 4 | Gaussian Mixture | Smoking | K-fold validation |
| [64] | 3D | - | RF threshold | 25 sec 50% overlap | 5 | 5 | Random Forest (RF), Thresholding | Hand-to-mouth gesture (HMG), Smoking | 5-fold |
| [69] | 6D | low-pass filter (fc = 5 Hz) | Moving window | 10 sec | 10 | 10 | Support- vector machine (SVM), Edge detector | HMG, Smoking | - |
| [70] | 9D | - | Distance calculation Moving window | - | 34 | 34 | Conditional Random Forest | HMG, Smoking | 10-fold & leave one out cross validation (LOOS) |
| [66] | 6D in smartwatch | - | Moving window | 30 sec | 6 | 4 (Empirically chosen) | Hierarchical 2 layer | Smoking | LOOS |
| [65] | 3D in smartwatch | - | Euler transformation | - | 3 | 3 | Artificial Neural Network | Smoking | K-fold validation |
| [68] | 6D in smartwatch | - | Hand movement | - | 3 | 3 | 3 stage analytical pipeline using Decision Tree | Smoking | LOOS |
| [72] | 3D in smartwatch | - | sliding window x-axis accelerometer | 10 s | 1 | 1 | Dynamic Time wrapping algorithm (CWRT) | Smoking | LOOS |
| Ref | No of IMU | IMU Placement | Dataset | Subject | Activities | Study Type | Detection | Performance |
|---|---|---|---|---|---|---|---|---|
| [54] | 1 (3D) | wrist | Data of 23 days | 4 | Smoking-standing | Free- living | Smoking | Precision 0.51, Recall 0.70 |
| [64] | 4 (3D) | Dominant wrist and upper arm, non-domin-ant wrist, ankle | 11.8 Hour (34 smoking, 481 puff) | 6 | Smoking-eating, walk, Talk, Drink, Stand | Lab | HMG, Smoking | F1-score 0.70 for HMG, 0.79 for smoking |
| [69] | 4 (6D) | Wrist, upper arm near the shoulder, upper arm near elbow, elbow | 21 Hour | 6 | Smoking-sitting, walk, Smoking-resting, cellphone use | Lab | HMG, Smoking | False Positive Rate 0.07–0.2 |
| [70] | 1 (9D) | Wrist, elbow | 28 Hour, 369 puffs (48 h for wild) | 15-lab, 4-free-living | Smoking-stand, Smoking-talking, Smoking-walking, eat, drink | Lab, Free-living | HMG, Smoking | F1-score 0.85, Precision 0.95, Recall 0.81 |
| [66] | 1 (6D) in Smart watch | wrist | 45 Hour, 17 h smoking of 230 cigarettes | 11 | Smoking-stand, Smoking-sitting, Eat, Drink, Group conversation, Sitting, | Lab | Smoking | F1-score 0.83–0.94 (person-independent) F1-score 0.90–0.97 (person-dependent) |
| [65] | 1 (3D) in smartwatch | wrist | 35 smoking, 155 non-smoking sessions, | 2 | Not mentioned | Lab | Smoking | Accuracy 0.85–0.95 |
| [67] | 1 (6D) in band | wrist | 1584 epochs of hand gestures | 1 | Sitting, Walking, Eating | Lab | Smoking | Accuracy 0.94 Recall 0.91 |
| [68] | 1 (6D) in smartwatch | wrist | - | 38 | Smoking-sitting, Drink, Eat | Lab, Free-living | Smoking | Precision 0.86, Recall 0.71 |
| [72] | 1 (3D) in smartwatch | wrist | - | 26 | Smoking-stand, Eat, Drink | Lab | Smoking | F1-score 0.96 |
| Ref | Belt Placement | RIP Belt/ Module | Signal Output | Data storage | Validation Study |
|---|---|---|---|---|---|
| [85] | Thoracic and abdominal | DuraBelt Pro-Tech Inc. connected to zRIP, Philips Respironics, Murrysville, PA | Analog Data | Commercial data logger: Logomatic V2.0, Sparkfun Electronics | 20 subjects in the lab |
| [86] | Thoracic | AutoSense RIP belts | Analog Data | Wireless transmission to smartphone | 35 in lab and free-living |
| [61] | Thoracic | SleepSense Inductive Plethysmography, S.L.P Inc. | Pulse Data | Embedded data logger with STM32 MCU | 40 both in the lab and free-living |
| Ref | No of RIPBand | Pre-processing | De-noising | Artifact Removal | Feature Extracted | Classifier Employed | Signal Classification | Validation | Study Type | Performance Matrices |
|---|---|---|---|---|---|---|---|---|---|---|
| [89] | 2 (Thoracic TC, Abdominal AB) | 1. Tidal Volume and Airflow measurement from TC, AB signals 2. Signal Normalization to the range of -1 and 1 | - | An ideal band pass filter, fc = 0.0001–10 Hz | - | Simple Peak-Valley Detection | 4 activities (resting, reading aloud, food intake and smoking) | Train- 5 fold cross-val; Test-LOOS | Lab, 20 subject | Accuracy: Resting-0.96, Reading-0.89, Food intake-0.91, Smoking-0.89 |
| [90] | Average Gaussian filter of 25 points | Z-norm 16 features Using Window 0.5s, 50% overlap | Left-to-right hidden Markov models | 5 activities (sedentary, walking, eating, talking, and cigarette smoking) | LOOS | Lab, 20 subject | Precision 0.60, Recall 0.67 F1-score 0.62 | |||
| [86] | 1 (Thoracic TC) | - | - | 17 features from each 30s window | Supervised and semi-supervised support vector | Puff or non-puff | LOOS | Lab, 10 subject | Accuracy 0.91 |
| Ref | Sensor Details | Subject Involved | Study Details | Total Smoking Events |
|---|---|---|---|---|
| [57] | WADD 3,74 × 2.4 × 2.1 cm, 17 g | 2 | In lab (1 session) | 6 |
| [91] | Smart neckband: dual-core 1.5 GHz CPU, 1 GB RAM, Android 4.2 OS | 16 | Free-living (1 week) | 143 |
| Features of Wearable Systems | Respiratory Inductance Plethysmography | Electrical Proximity Sensing | Inertial Approach | Egocentric Camera |
|---|---|---|---|---|
| Body Positions | Abdominal or Thoracic area | Transmitter on wrist and Receiver on the chest surface | Mostly on wrist or lower elbow | Eye, chest or Wrist. Eye-level camera was explored. |
| Comfort | Moderate, worn as a belt | Moderate | High, flexible to implement in body locations | High, however a privacy concern exists |
| Applications | Characteristic breathing pattern detection | Characteristic hand to mouth proximity | Characteristic hand gesture of smoking | Smoking puff, environment, context detection |
| Highest Performance | Accuracy of 0.81 in detecting puff events [86] | Recall of 0.90 in detecting hand to mouth gestures preceding smoking [63] | Precision 0.95 and F1-score 0.85 in detecting smoking events [70] | Recall of 1 in detecting smoking events (manual image review) |
| Advantages | Indirect monitoring | Good tolerance to electromagnetic interference | Able to be embedded in a highly wearable wristband or smartwatch | Direct monitoring |
| Challenges | Accuracy needs improvement | Combination of other sensors is necessary to improve applicability | Detected gestures often confused with eating; limited by concurrent activity and confounding gestures | Privacy concern for both wearer and people in surroundings |
| Applicable to free-living settings | Thoroughly tested | Moderately tested | Thoroughly tested | Feasibility tested |
| Obtrusiveness | Unobtrusive | Unobtrusive | Unobtrusive | Unobtrusive |
| Contact with Skin | Not mandatory, can be worn over clothing | Not required | Not required if wristband employed | Not required. |
| Fusion Platform | AutoSense | PACT | PACT v2 |
|---|---|---|---|
| Sensing element employed in smoking research | RIP sensing, 6-axis IMU (other sensors not utilized yet in smoking research) | RIP, Proximity | RIP, Bioimpedance sensor, ECG, 6-axis IMU and Instrument lighter (other sensors not utilized yet in smoking research) |
| Sampling Frequency | 21.3 Hz for RIP, 16 Hz for Inertial Sensor | 100 Hz | 100 Hz for IMU, RIP, Proximity; 1 KHz for Physiological sensor |
| Device Storage | N/A | Portable Datalogger (Logomatic V2, Sparkfun Electronics, Boulder, CO) | On Board 4-GB Micro SD card |
| Sensor data Transmission Method | To smartphone via ANT Radio. | N/A | N/A |
| Data analysis/processing method | Published | Published | Published |
| Clinical or Validation Survey | Performed over more than 100 subjects in different studies | Performed over 20 regular smokers. | Performed over 40 regular smokers. |
| Tested in Free-living | Tested over 61 regular smokers in different studies | Not tested | Tested over 40 regular smokers. |
| Gold Standard Comparison | Manual annotation by an observer. | Push Button based manual annotation | Manual Video Annotation and cellphone registration |
| System longevity (Battery Life) | More than a day | More than a day | More than a day |
| Ref | De-noising and Artifact Removal | Pre-processing | Approach | Key Points | Performance Matrices | Validation | |
|---|---|---|---|---|---|---|---|
| Subject-Independent | Subject-Dependent | ||||||
| [92] | 1. Gaussian Average Filter of 25-point sliding window 2. Ideal Band Pass Filter: fc = 0.0001–10 Hz | Normalization on both Proximity Signal and tidal Volume | SVM | - | Precision 0.87, Recall 0.80 | Precision 0.90, Recall 0.90 | LOOS |
| [93] | SVM | 1503 Feature Vectors | F1-score: 0.81 | F1-score: 0.90 | LOOS | ||
| 27 Empirical Feature Vectors | F1-score: 0.65 | F1-score: 0.68 | |||||
| 16 Forward Feature Selected Feature Vectors | F1-score: 0.67 | F1-score: 0.94 | |||||
| [94] | SVM | Employing Thoracic Signal (TC) | F1-score: 0.41 | F1-score: 0.85 | LOOS | ||
| Employing Abdominal Signal (AB) | F1-score: 0.46 | F1-score: 0.88 | |||||
| Employing Proximity Signal (PS) | F1-score: 0.59 | F1-score: 0.90 | |||||
| [93] | Ensemble | Adaboost | F1-score: 0.71 | F1-score: 0.77 | LOOS | ||
| Bagging | F1-score: 0.70 | F1-score: 0.82 | |||||
| Random Forest | F1-score: 0.69 | F1-score: 0.84 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imtiaz, M.H.; Ramos-Garcia, R.I.; Wattal, S.; Tiffany, S.; Sazonov, E. Wearable Sensors for Monitoring of Cigarette Smoking in Free-Living: A Systematic Review. Sensors 2019, 19, 4678. https://doi.org/10.3390/s19214678
Imtiaz MH, Ramos-Garcia RI, Wattal S, Tiffany S, Sazonov E. Wearable Sensors for Monitoring of Cigarette Smoking in Free-Living: A Systematic Review. Sensors. 2019; 19(21):4678. https://doi.org/10.3390/s19214678
Chicago/Turabian StyleImtiaz, Masudul H., Raul I. Ramos-Garcia, Shashank Wattal, Stephen Tiffany, and Edward Sazonov. 2019. "Wearable Sensors for Monitoring of Cigarette Smoking in Free-Living: A Systematic Review" Sensors 19, no. 21: 4678. https://doi.org/10.3390/s19214678
APA StyleImtiaz, M. H., Ramos-Garcia, R. I., Wattal, S., Tiffany, S., & Sazonov, E. (2019). Wearable Sensors for Monitoring of Cigarette Smoking in Free-Living: A Systematic Review. Sensors, 19(21), 4678. https://doi.org/10.3390/s19214678






