Application of a Fluorescent Probe for the Online Measurement of PM-Bound Reactive Oxygen Species in Chamber and Ambient Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Online PINQ System
2.1.1. Flow Switching Assembly
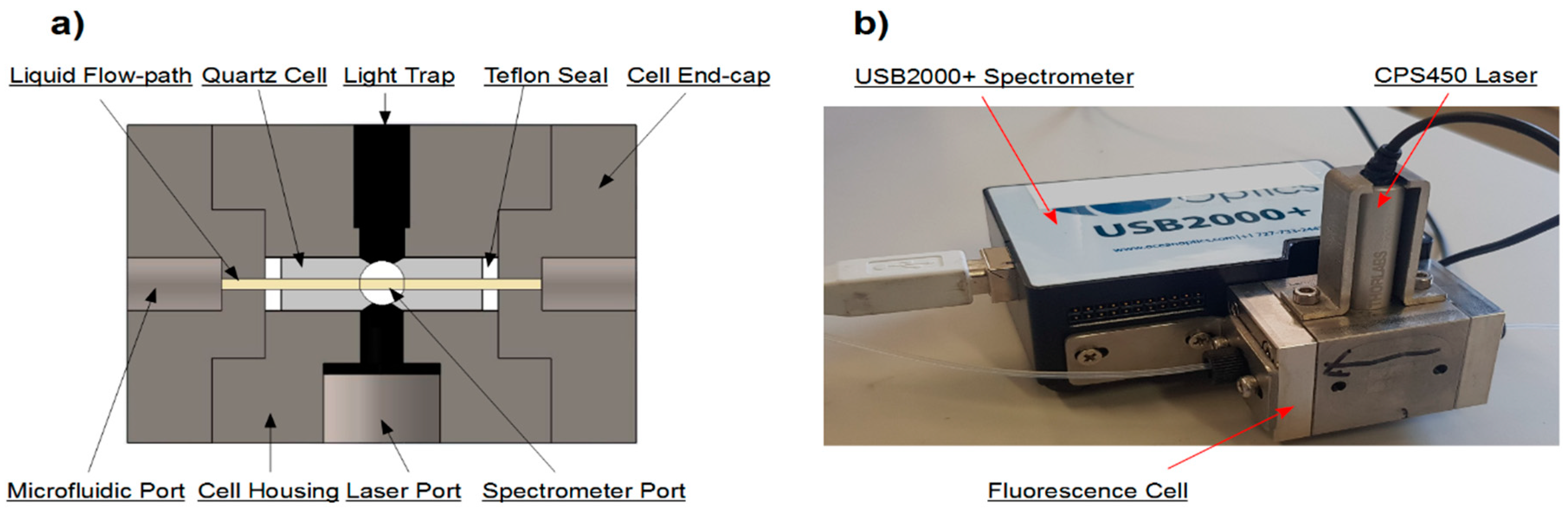
2.1.2. Flow-Through Fluorimeter
2.1.3. Debubbler
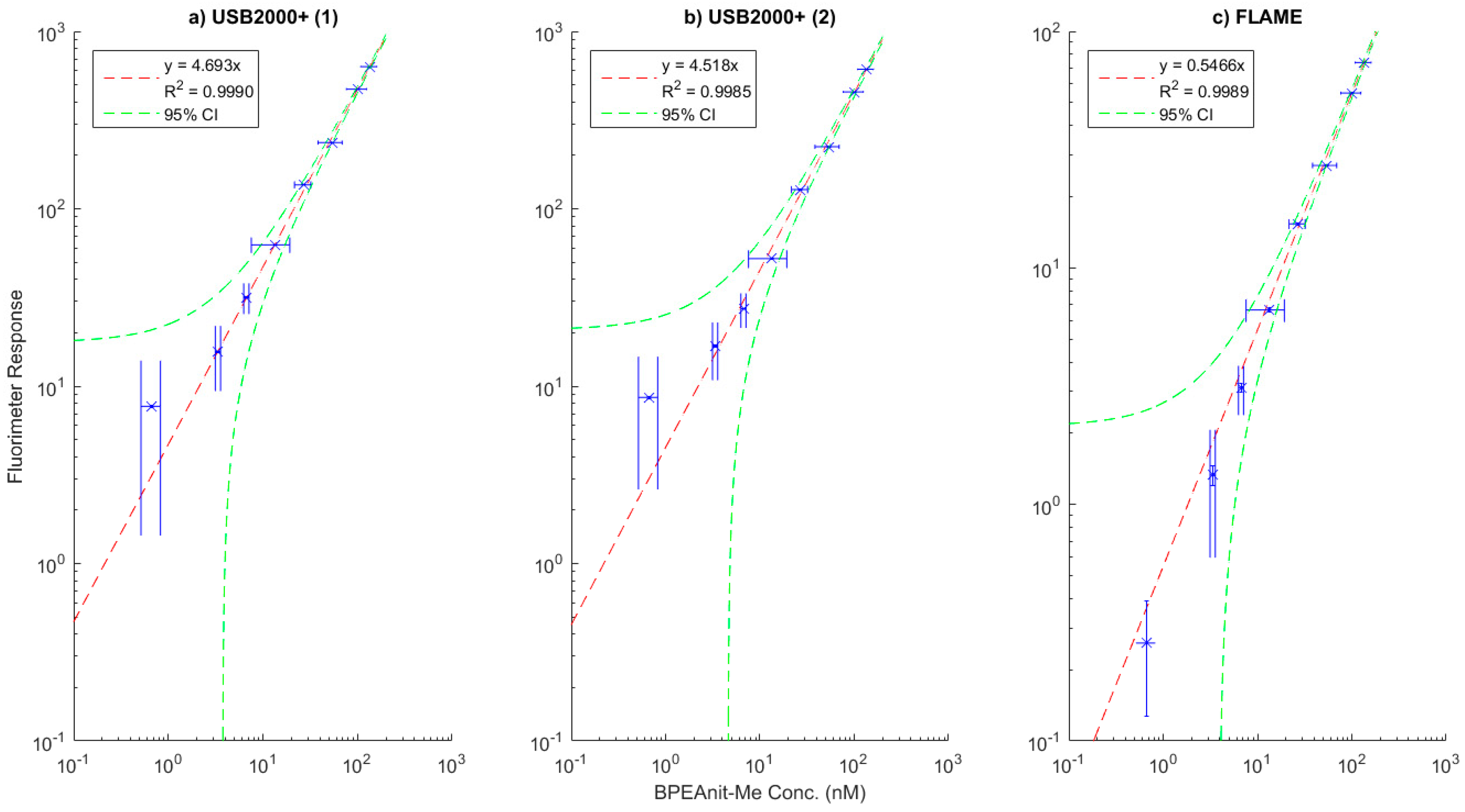
2.2. Fluorimeter Calibration
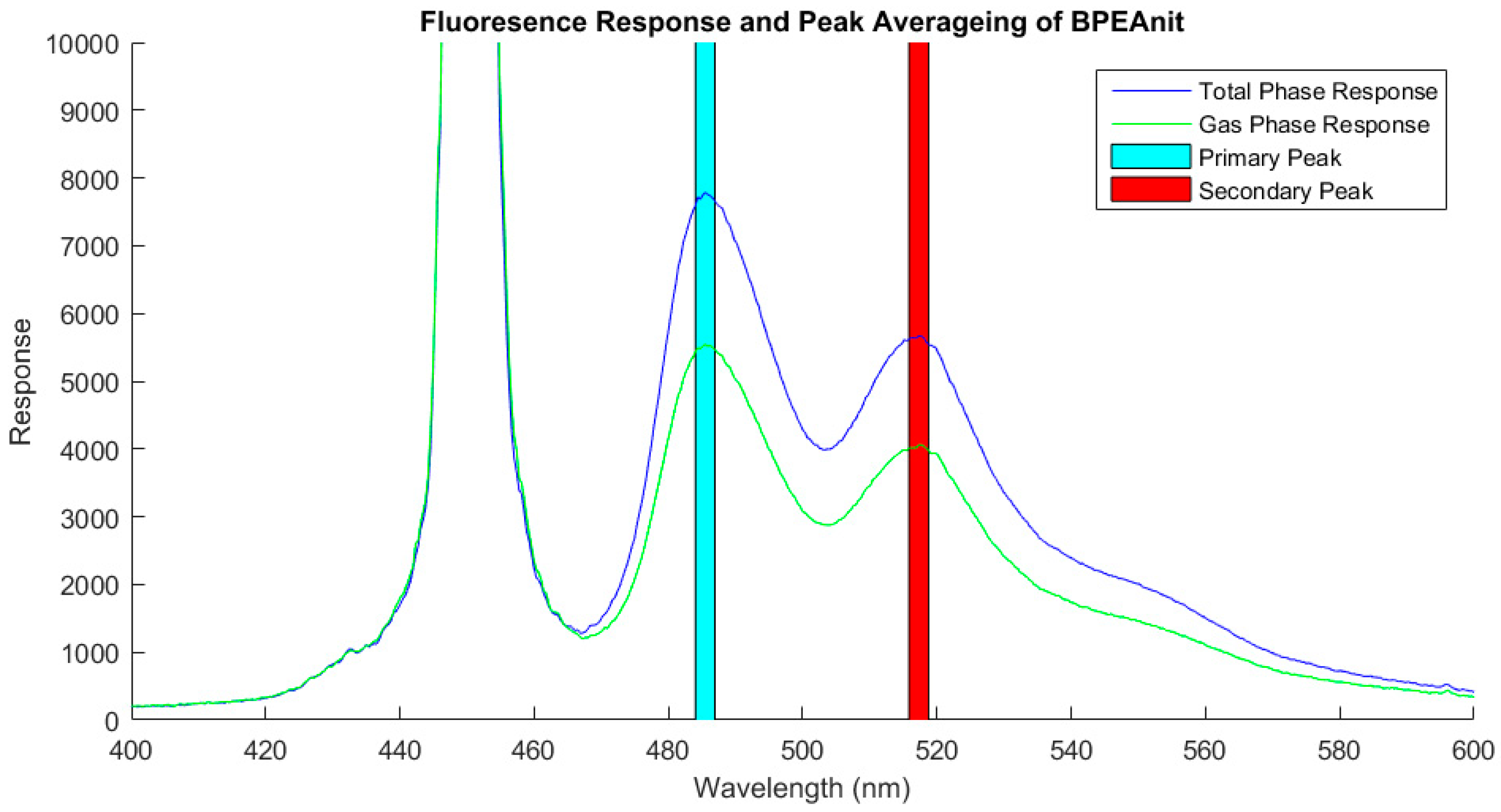
2.3. PINQ Data Analysis
2.4. Side-Stream Cigarette Smoke Chamber Study
2.5. Field Measurements of Background PM in Gaungzhou, China
3. Results
3.1. Calibration Plots of the Flow-Through Fluorimeters
3.2. Data Analysis Methodology
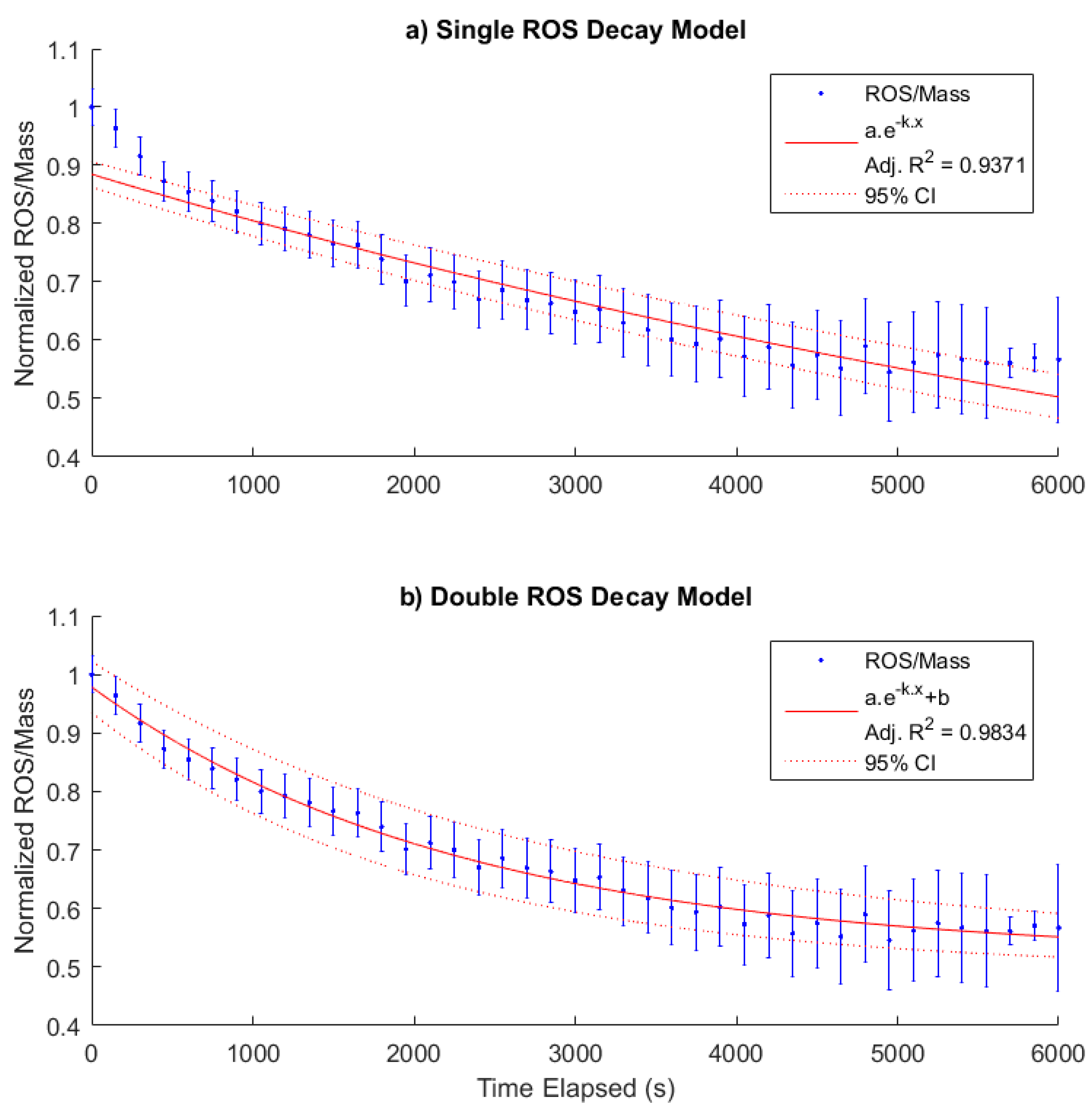
3.3. PM-Bound ROS Half-Lives
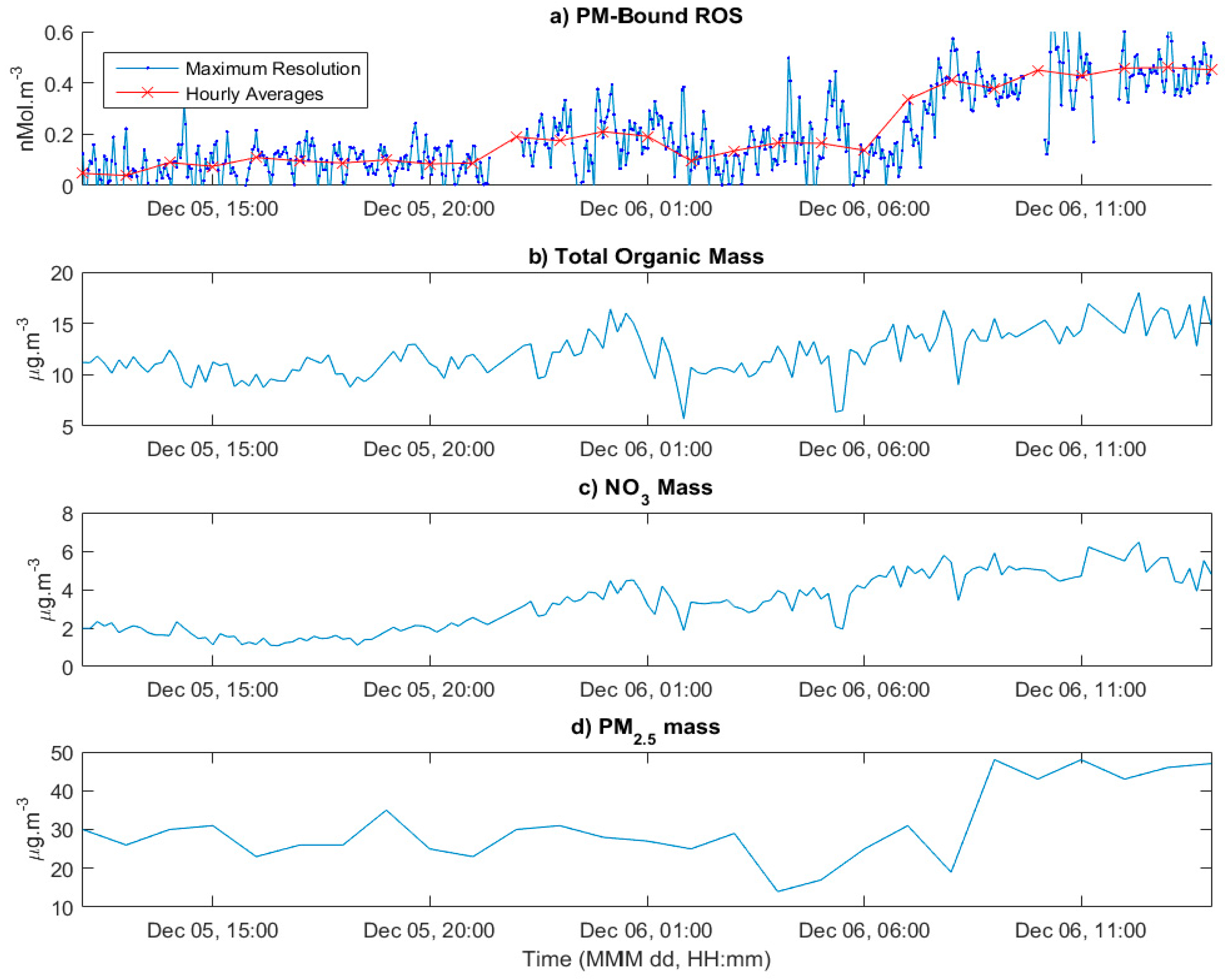
3.4. Application to Ambient Measurements
4. Discussion
4.1. The Online PINQ
4.2. Implications of the Initial Ambient Study
4.3. Half-Lives of PM-Bound ROS
4.4. What Time Resolution is Necessary for the Measurement of PM-Bound ROS?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Englert, N. Fine particles and human health—A review of epidemiological studies. Toxicol. Lett. 2004, 149, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Hoek, G.; Krishnan, R.M.; Beelen, R.; Peters, A.; Ostro, B.; Brunekreef, B.; Kaufman, J.D. Long-term air pollution exposure and cardio-respiratory mortality: A review. Environ. Heal. A Glob. Access Sci. Source 2013, 12, 43. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.; Guo, X.; Cheung, F.M.H.; Yung, K.K.L. The association between PM2.5 exposure and neurological disorders: A systematic review and meta-analysis. Sci. Total Environ. 2019, 655, 1240–1248. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef]
- Hellack, B.; Nickel, C.; Albrecht, C.; Kuhlbusch, T.A.J.; Boland, S.; Baeza-Squiban, A.; Wohlleben, W.; Schins, R.P.F. Analytical methods to assess the oxidative potential of nanoparticles: A review. Environ. Sci. Nano 2017, 4, 1920–1934. [Google Scholar] [CrossRef]
- Li, N.; Sioutas, C.; Cho, A.; Schmitz, D.; Misra, C.; Sempf, J.; Wang, M.; Oberley, T.; Froines, J.; Nel, A. Ultrafine Particulate Pollutants Induce Oxidative Stress and Mitochondrial Damage. Environ. Health Perspect. 2002, 111, 455–460. [Google Scholar] [CrossRef]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative stress. Annu. Rev. Biochem. 2017, 86, 715–748. [Google Scholar] [CrossRef]
- Kelly, F.J. Oxidative stress: Its role in air pollution and adverse health effects. Occup. Environ. Med. 2003, 60, 612–616. [Google Scholar] [CrossRef]
- Halliwell, B.; Whiteman, M. Measuring reactive species and oxidative damage in vivo and in cell culture: How should you do it and what do the results mean? Br. J. Pharmacol. 2004, 142, 231–255. [Google Scholar] [CrossRef]
- Arangio, A.M.; Tong, H.; Socorro, J.; Pöschl, U.; Shiraiwa, M. Quantification of environmentally persistent free radicals and reactive oxygen species in atmospheric aerosol particles. Atmos. Chem. Phys. 2016, 16, 13105–13119. [Google Scholar] [CrossRef]
- Dellinger, B.; Pryor, W.A.; Cueto, R.; Squadrito, G.L.; Hegde, V.; Deutsch, W.A. Role of free radicals in the toxicity of airborne fine particulate matter. Chem. Res. Toxicol. 2001, 14, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Shein, M.; Jeschke, G. Comparison of Free Radical Levels in the Aerosol from Conventional Cigarettes, Electronic Cigarettes, and Heat-Not-Burn Tobacco Products. Chem. Res. Toxicol. 2019, 32, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Fang, T.; Verma, V.; Zeng, L.; Weber, R.J. A method for measuring total aerosol oxidative potential (OP) with the dithiothreitol (DTT) assay and comparisons between an urban and roadside site of water-soluble and total OP. Atmos. Meas. Tech. 2017, 10, 2821–2835. [Google Scholar] [CrossRef]
- Eiguren-Fernandez, A.; Kreisberg, N.; Hering, S. An online monitor of the oxidative capacity of aerosols (o-MOCA). Atmos. Meas. Tech. 2017, 10, 633–644. [Google Scholar] [CrossRef]
- Zhou, J.; Bruns, E.A.; Zotter, P.; Stefenelli, G.; Prévôt, A.S.H.; Baltensperger, U.; EI-Haddad, I.; Dommen, J. Development, characterization and first deployment of an improved online reactive oxygen species analyzer. Atmos. Meas. Tech. Discuss. 2017, 1–27. [Google Scholar] [CrossRef]
- Wragg, F.P.H.; Fuller, S.J.; Freshwater, R.; Green, D.C.; Kelly, F.J.; Kalberer, M. An automated online instrument to quantify aerosol-bound reactive oxygen species (ROS) for ambient measurement and health-relevant aerosol studies. Atmos. Meas. Tech. 2016, 9, 4891–4900. [Google Scholar] [CrossRef]
- Venkatachari, P.; Hopke, P.K. Development and Laboratory Testing of an Automated Monitor for the Measurement of Atmospheric Particle-Bound Reactive Oxygen Species (ROS). Aerosol Sci. Technol. 2008, 42, 629–635. [Google Scholar] [CrossRef]
- Hung, H.-F.; Wang, C.-S. Experimental determination of reactive oxygen species in Taipei aerosols. J. Aerosol Sci. 2001, 32, 1201–1211. [Google Scholar] [CrossRef]
- Fang, T.; Verma, V.; Guo, H.; King, L.E.; Edgerton, E.S.; Weber, R.J. A semi-automated system for quantifying the oxidative potential of ambient particles in aqueous extracts using the dithiothreitol (DTT) assay: Results from the Southeastern Center for Air Pollution and Epidemiology (SCAPE). Atmos. Meas. Tech. Discuss. 2014, 7, 7245–7279. [Google Scholar] [CrossRef]
- Hedayat, F.; Stevanovic, S.; Miljevic, B.; Bottle, S.; Ristovski, Z.D.D. Review-evaluating the molecular assays for measuring the oxidative potential of particulate matter. Chem. Ind. Chem. Eng. Q. 2015, 21, 201–210. [Google Scholar] [CrossRef]
- Pal, A.K.; Bello, D.; Budhlall, B.; Rogers, E.; Milton, D.K. Screening for Oxidative Stress Elicited by Engineered Nanomaterials: Evaluation of Acellular DCFH Assay. Dose. Response. 2012, 10, 308–330. [Google Scholar] [CrossRef] [PubMed]
- Fuller, S.J.J.; Wragg, F.P.H.P.H.; Nutter, J.; Kalberer, M. Comparison of on-line and off-line methods to quantify reactive oxygen species (ROS) in atmospheric aerosols. Atmos. Environ. 2014, 92, 97–103. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, Y.; Zhang, Y.; Fang, D.; Schauer, J.J. Optimization of the Measurement of Particle-Bound Reactive Oxygen Species with 2′,7′-dichlorofluorescin (DCFH). Water. Air. Soil Pollut. 2016, 227, 164. [Google Scholar] [CrossRef]
- King, L.E.; Weber, R.J. Development and testing of an online method to measure ambient fine particulate reactive oxygen species (ROS) based on the 2′,7′-dichlorofluorescin (DCFH) assay. Atmos. Meas. Tech. 2013, 6, 1647–1658. [Google Scholar] [CrossRef]
- Wang, Y.; Hopke, P.K.; Sun, L.; Chalupa, D.C.; Utell, M.J. Laboratory and field testing of an automated atmospheric particle-bound reactive oxygen species sampling-analysis system. J. Toxicol. 2011, 2011, 419476–419479. [Google Scholar] [CrossRef]
- Fairfull-Smith, K.E.; Bottle, S.E. The synthesis and physical properties of novel polyaromatic profluorescent isoindoline nitroxide probes. European J. Org. Chem. 2008, 2008, 5391–5400. [Google Scholar] [CrossRef]
- Brown, R.A.; Stevanovic, S.; Bottle, S.; Ristovski, Z.D. An instrument for the rapid quantification of PM-bound ROS: The Particle Into Nitroxide Quencher (PINQ). Atmos. Meas. Tech. 2019, 12, 2387–2401. [Google Scholar] [CrossRef]
- Stevanovic, S.; Vaughan, A.; Hedayat, F.; Salimi, F.; Rahman, M.M.; Zare, A.; Brown, R.A.J.; Brown, R.A.J.; Wang, H.; Zhang, Z.; et al. Oxidative potential of gas phase combustion emissions—An underestimated and potentially harmful component of air pollution from combustion processes. Atmos. Environ. 2017, 158, 227–235. [Google Scholar] [CrossRef]
- Stevanovic, S.; Miljevic, B.; Madl, P.; Clifford, S.; Ristovski, Z. Characterisation of a Commercially Available Thermodenuder and Diffusion Drier for Ultrafine Particles Losses. AEROSOL AIR Qual. Res. 2015, 15, 357. [Google Scholar] [CrossRef]
- Stevanovic, S.; Gali, N.K.; Salimi, F.; Brown, R.A.; Ning, Z.; Cravigan, L.; Brimblecombe, P.; Bottle, S.; Ristovski, Z.D. Diurnal profiles of particle-bound ROS of PM2.5 in urban environment of Hong Kong and their association with PM2.5, black carbon, ozone and PAHs. Atmos. Environ. 2019, 117023. [Google Scholar] [CrossRef]
- Miljevic, B.; Fairfull-Smith, K.E.; Bottle, S.E.; Ristovski, Z.D. The application of profluorescent nitroxides to detect reactive oxygen species derived from combustion-generated particulate matter: Cigarette smoke— A case study. Atmos. Environ. 2010, 44, 2224–2230. [Google Scholar] [CrossRef]
- Fröhlich, R.; Cubison, M.; Slowik, J.; Bukowiecki, N.; Prévôt, A.; Baltensperger, U.; Schneider, J.; Kimmel, J.; Gonin, M.; Rohner, U.; et al. The ToF-ACSM: A portable aerosol chemical speciation monitor with TOFMS detection. Atmos. Meas. Tech. 2013, 6, 3225. [Google Scholar] [CrossRef]
- Stevanovic, S.; Miljevic, B.; Eaglesham, G.K.; Bottle, S.E.; Ristovski, Z.D.; Fairfull-Smith, K.E. The use of a nitroxide probe in DMSO to capture free radicals in particulate pollution. European J. Org. Chem. 2012, 5908–5912. [Google Scholar] [CrossRef]
- Vesna, O.; Sax, M.; Kalberer, M.; Gaschen, A.; Ammann, M. Product study of oleic acid ozonolysis as function of humidity. Atmos. Environ. 2009, 43, 3662–3669. [Google Scholar] [CrossRef]

| Cl− | NO3− | SO42− | NH4+ | Org | |
|---|---|---|---|---|---|
| Corr. Fac. | 0.39 | 0.65 | 0.28 | 0.50 | 0.54 |
| p-Value | 8 × 10–7 | 2 × 10–19 | 6 × 10–4 | 7 × 10–11 | 5 × 10–13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brown, R.; Stevanovic, S.; Brown, Z.; Cai, M.; Zhou, S.; Song, W.; Wang, X.; Miljevic, B.; Zhao, J.; Bottle, S.; et al. Application of a Fluorescent Probe for the Online Measurement of PM-Bound Reactive Oxygen Species in Chamber and Ambient Studies. Sensors 2019, 19, 4564. https://doi.org/10.3390/s19204564
Brown R, Stevanovic S, Brown Z, Cai M, Zhou S, Song W, Wang X, Miljevic B, Zhao J, Bottle S, et al. Application of a Fluorescent Probe for the Online Measurement of PM-Bound Reactive Oxygen Species in Chamber and Ambient Studies. Sensors. 2019; 19(20):4564. https://doi.org/10.3390/s19204564
Chicago/Turabian StyleBrown, Reece, Svetlana Stevanovic, Zachary Brown, Mingfu Cai, Shengzhen Zhou, Wei Song, Xinming Wang, Branka Miljevic, Jun Zhao, Steven Bottle, and et al. 2019. "Application of a Fluorescent Probe for the Online Measurement of PM-Bound Reactive Oxygen Species in Chamber and Ambient Studies" Sensors 19, no. 20: 4564. https://doi.org/10.3390/s19204564
APA StyleBrown, R., Stevanovic, S., Brown, Z., Cai, M., Zhou, S., Song, W., Wang, X., Miljevic, B., Zhao, J., Bottle, S., & Ristovski, Z. (2019). Application of a Fluorescent Probe for the Online Measurement of PM-Bound Reactive Oxygen Species in Chamber and Ambient Studies. Sensors, 19(20), 4564. https://doi.org/10.3390/s19204564








