Explicit and Implicit Responses to Tasting Drinks Associated with Different Tasting Experiences
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Materials
2.2.1. Recording Equipment
2.2.2. Self-Report Rating Scales and Sip Size
2.2.3. Samples
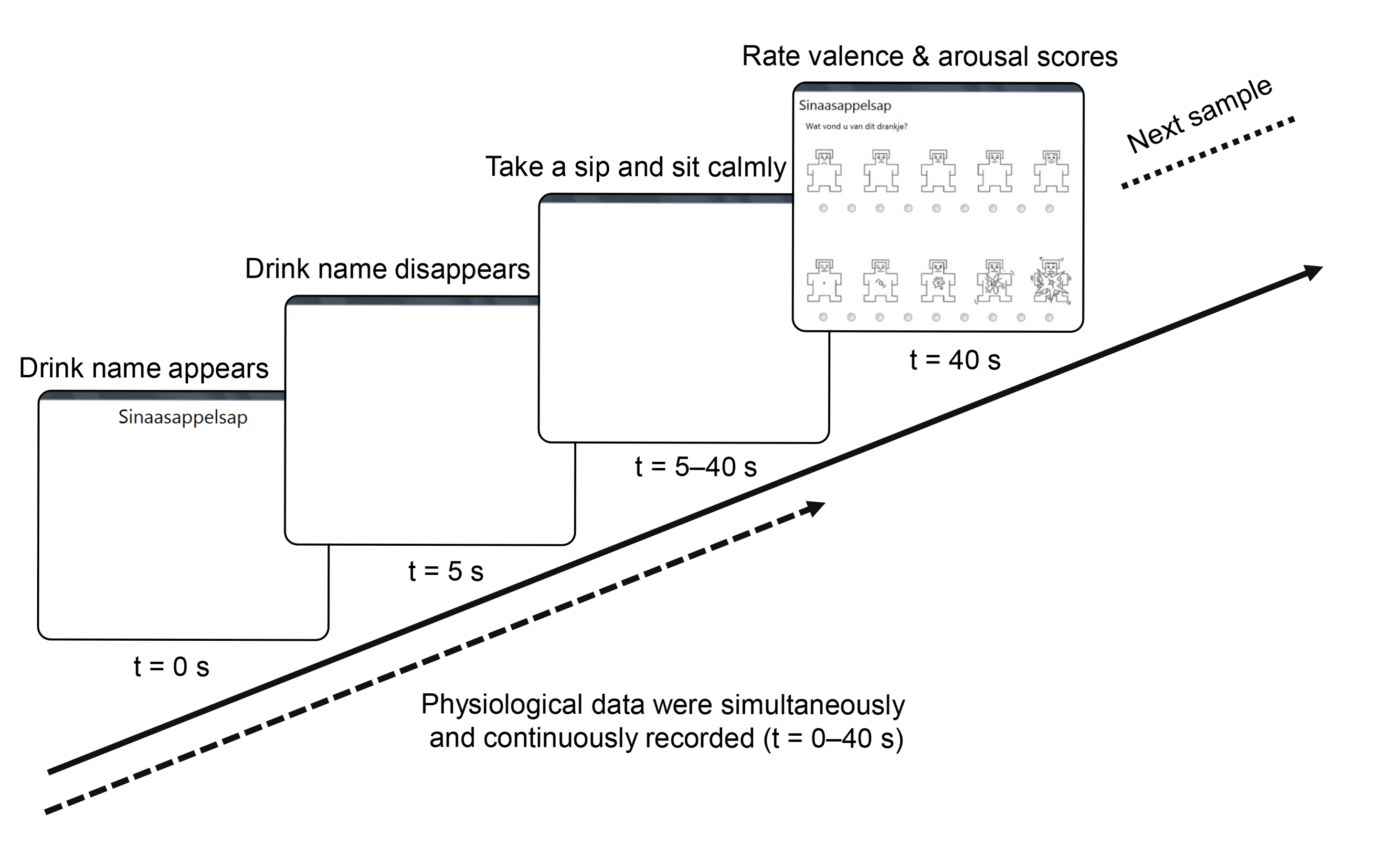
2.3. Design and Procedure
2.4. Data Processing and Analysis
2.4.1. Preprocessing for Facial Expression, EDA, ECG, and EEG
2.4.2. Extraction of Variables
2.4.3. Statistical Analyses
3. Results
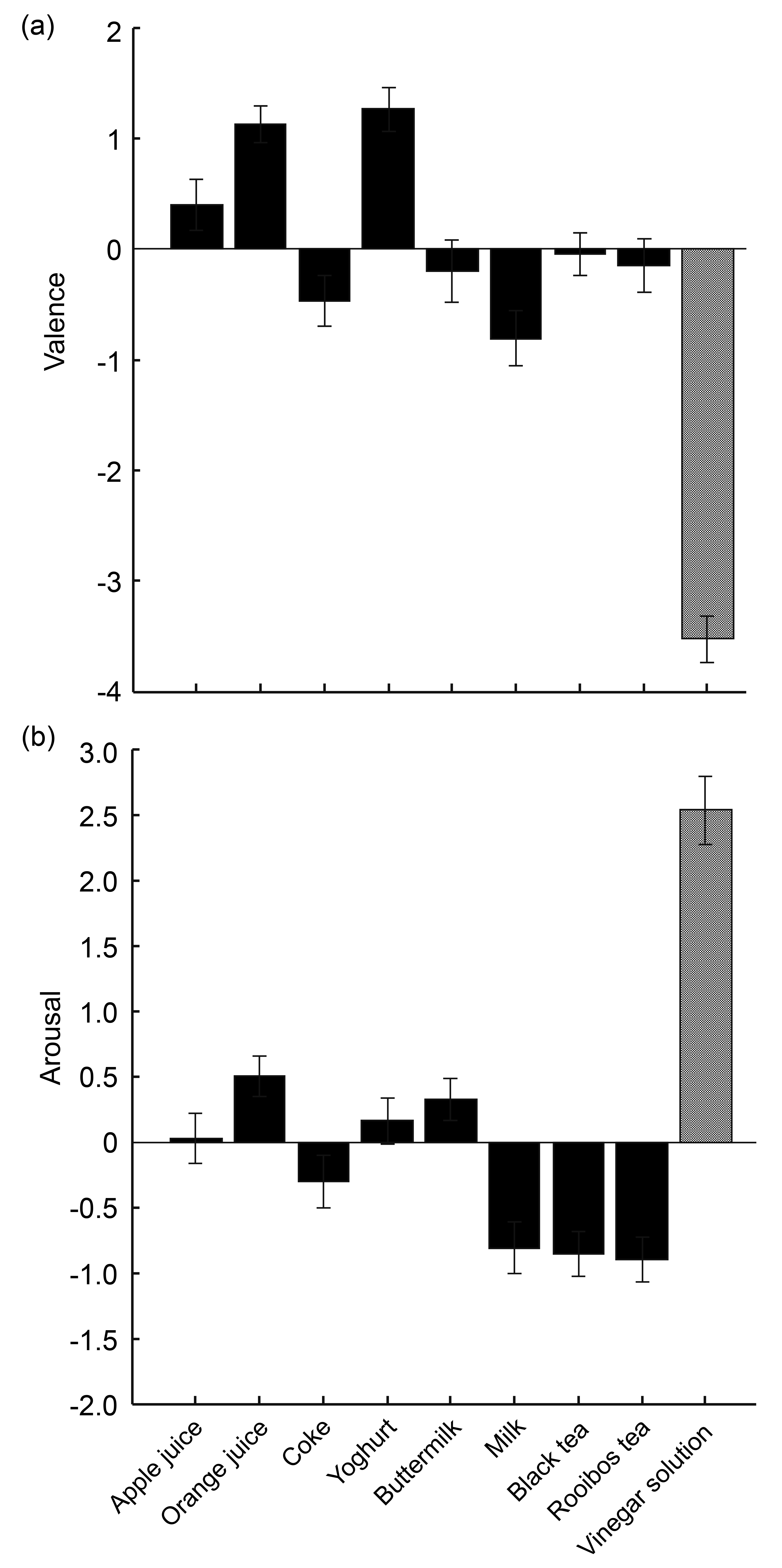
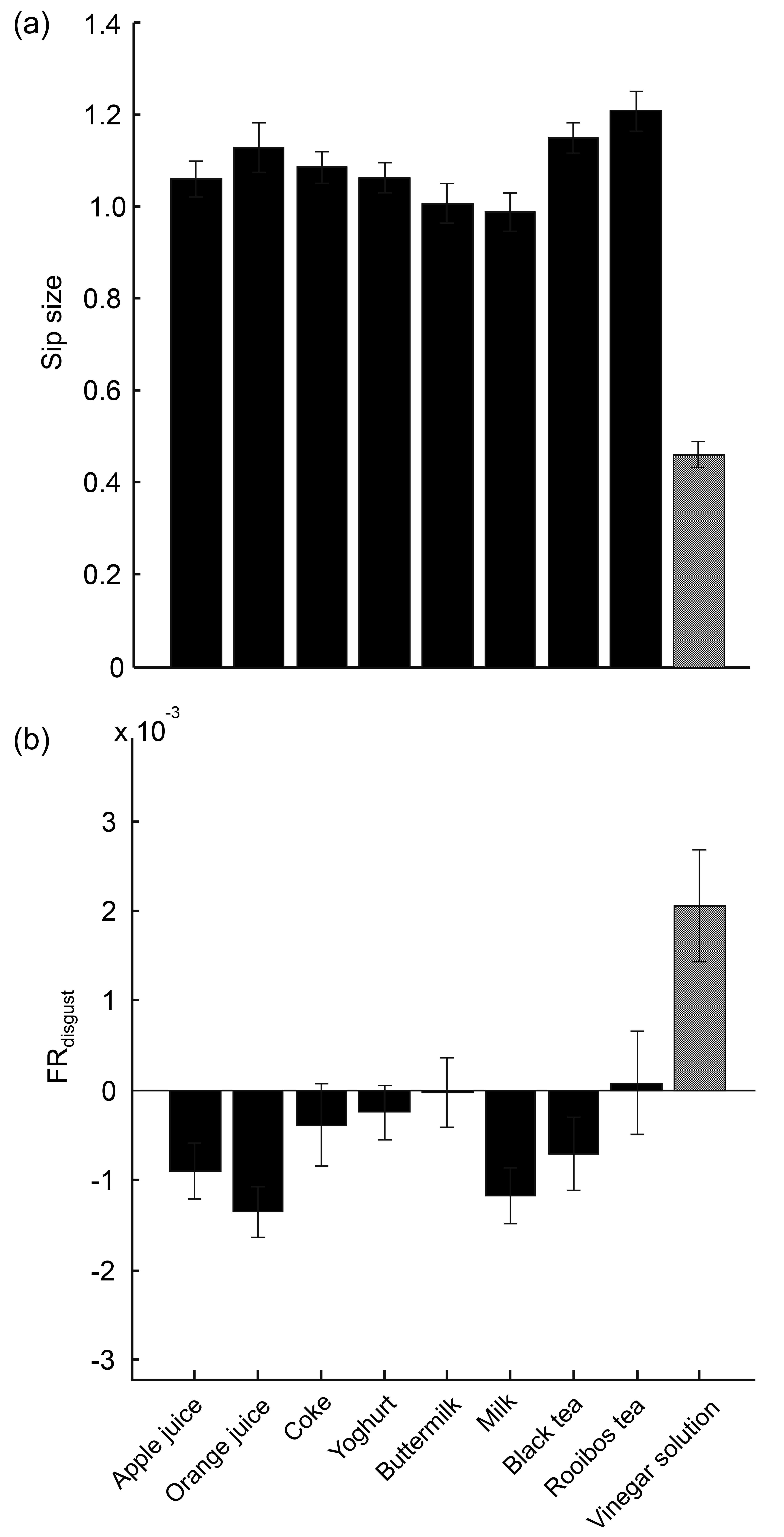
3.1. Sensitivity of Measures to Distinguish between Regular Drinks and Diluted Vinegar
3.2. Sensitivity of Measures to Distinguish between Regular Drinks
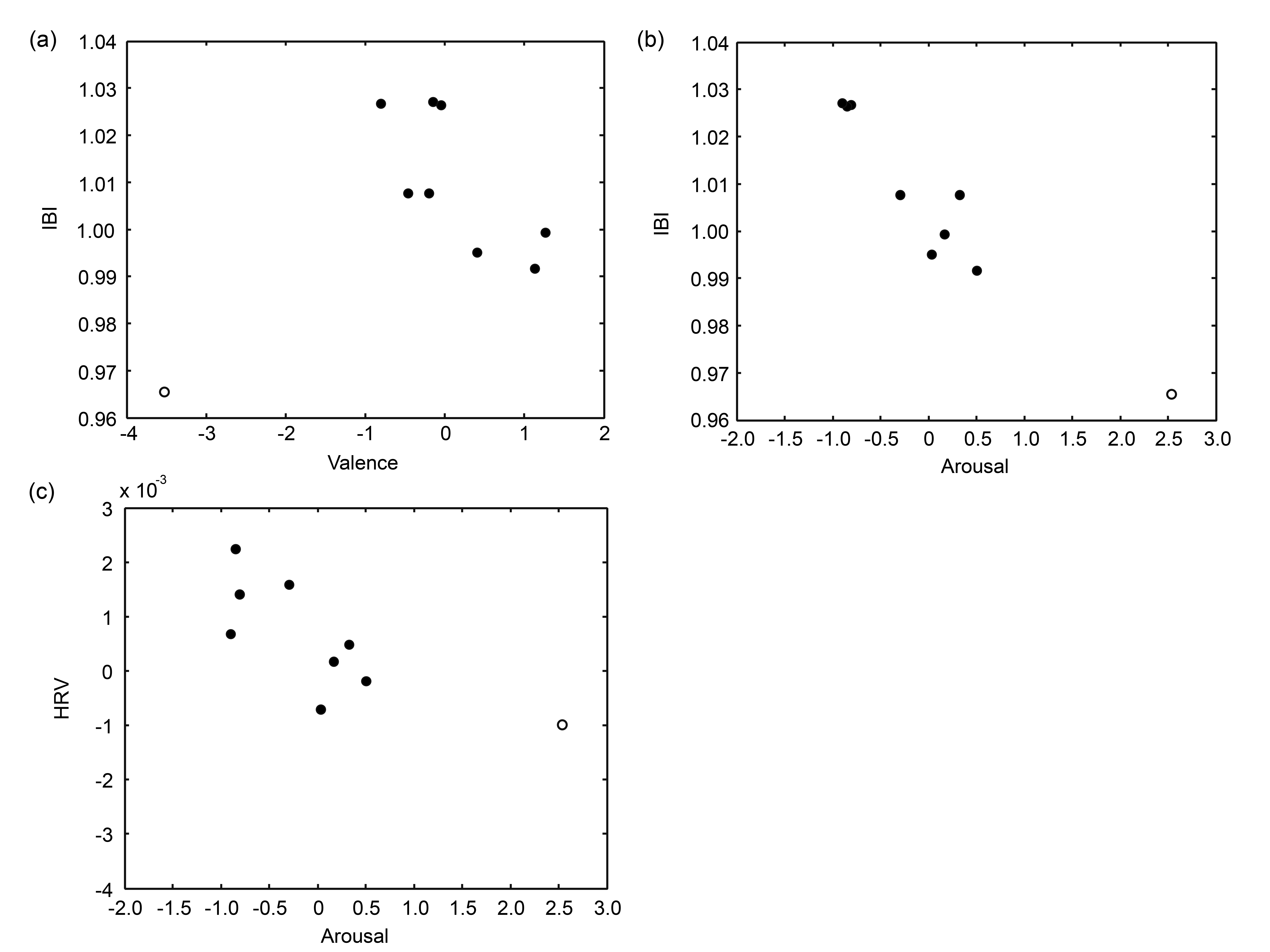
3.3. Association between Implicit Measures and Self-Reported Valence and Arousal for Regular Drinks
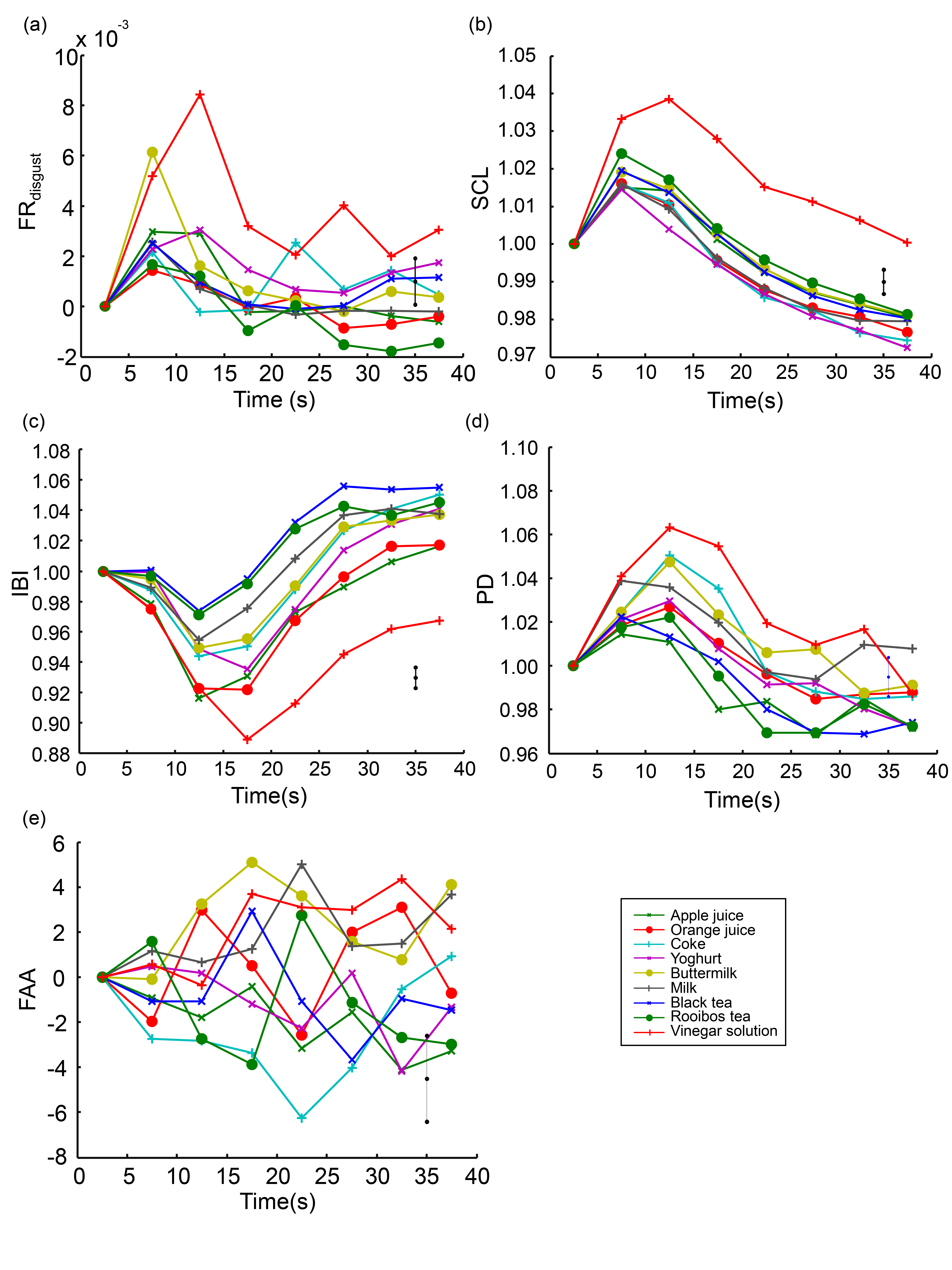
3.4. General Temporal Pattern of Behavioral and Physiological Variables Associated with Taking a Sip
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thomson, D.M.H.; Crocker, C.; Marketo, C.G. Linking sensory characteristics to emotions: An example using dark chocolate. Food Qual. Prefer. 2010, 21, 1117–1125. [Google Scholar] [CrossRef]
- Gutjar, S.; de Graaf, C.; Kooijman, V.; de Wijk, R.A.; Nys, A.; ter Horst, G.J.; Jager, G. The role of emotions in food choice and liking. Food Res. Int. 2015, 76, 216–223. [Google Scholar] [CrossRef]
- Dalenberg, J.R.; Gutjar, S.; Ter Horst, G.J.; de Graaf, K.; Renken, R.J.; Jager, G. Evoked emotions predict food choice. PLoS ONE 2014, 9, e115388. [Google Scholar] [CrossRef]
- Cardello, A.V.; Meiselman, H.L.; Schutz, H.G.; Craig, C.; Given, Z.; Lesher, L.L.; Eicher, S. Measuring emotional responses to foods and food names using questionnaires. Food Qual. Prefer. 2012, 24, 243–250. [Google Scholar] [CrossRef]
- Kuenzel, J.; Zandstra, E.H.; Lion, R.; Blanchette, I.; Thomas, A.; El-Deredy, W. Conditioning unfamiliar and familiar flavours to specific positive emotions. Food Qual. Prefer. 2010, 21, 1105–1107. [Google Scholar] [CrossRef]
- Kenney, E.; Adhikari, K. Recent developments in identifying and quantifying emotions during food consumption. J. Sci. Food Agric. 2016, 96, 3627–3630. [Google Scholar] [CrossRef] [PubMed]
- Schifferstein, H.N. Employing consumer research for creating new and engaging food experiences in a changing world. Curr. Opin. Food Sci. 2015, 3, 27–32. [Google Scholar] [CrossRef]
- Köster, E.P.; Mojet, J. From mood to food and from food to mood: A psychological perspective on the measurement of food-related emotions in consumer research. Food Res. Int. 2015, 76, 180–191. [Google Scholar] [CrossRef]
- Kaneko, D.; Toet, A.; Brouwer, A.-M.; Kallen, V.; Van Erp, J.B. Methods for evaluating emotions evoked by food experiences: A literature review. Front. Psychol. 2018, 9, 911. [Google Scholar] [CrossRef]
- Venkatraman, V.; Dimoka, A.; Pavlou, P.A.; Vo, K.; Hampton, W.; Bollinger, B.; Hershfield, H.E.; Ishihara, M.; Winer, R.S. Predicting advertising success beyond traditional measures: New insights from neurophysiological methods and market response modeling. J. Mark. Res. 2015, 52, 436–452. [Google Scholar] [CrossRef]
- Winkielman, P.; Berridge, K.; Sher, S. Emotion, consciousness, and social behavior. In Handbook of Social Neuroscience; Oxford University Press: Oxford, UK, 2011; pp. 195–211. [Google Scholar]
- Verastegui-Tena, L.; Schulte-Holierhoek, A.; Van Trijp, H.; Piqueras-Fiszman, B. Beyond expectations: The responses of the autonomic nervous system to visual food cues. Physiol. Behav. 2017, 179, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Beyts, C.; Chaya, C.; Dehrmann, F.; James, S.; Smart, K.; Hort, J. A comparison of self-reported emotional and implicit responses to aromas in beer. Food Qual. Prefer. 2017, 59, 68–80. [Google Scholar] [CrossRef]
- Steiner, J.E.; Glaser, D.; Hawilo, M.E.; Berridge, K.C. Comparative expression of hedonic impact: Affective reactions to taste by human infants and other primates. Neurosci. Biobehav. Rev. 2001, 25, 53–74. [Google Scholar]
- Danner, L.; Sidorkina, L.; Joechl, M.; Duerrschmid, K. Make a face! Implicit and explicit measurement of facial expressions elicited by orange juices using face reading technology. Food Qual. Prefer. 2014, 32, 167–172. [Google Scholar] [CrossRef]
- De Wijk, R.A.; He, W.; Mensink, M.G.J.; Verhoeven, R.H.G.; de Graaf, C. ANS responses and facial expressions differentiate between the taste of commercial breakfast drinks. PLoS ONE 2014, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rousmans, S.; Robin, O.; Dittmar, A.; Vernet-Maury, E. Autonomic nervous system responses associated with primary tastes. Chem. Senses 2000, 25, 709–718. [Google Scholar] [CrossRef] [PubMed]
- De Wijk, R.A.; Kooijman, V.; Verhoeven, R.H.G.; Holthuysen, N.T.E.; de Graaf, C. Autonomic nervous system responses on and facial expressions to the sight, smell, and taste of liked and disliked foods. Food Qual. Prefer. 2012, 26, 196–203. [Google Scholar] [CrossRef]
- Berridge, K.C. Food reward: Brain substrates of wanting and liking. Neurosci. Biobehav. Rev. 1996, 20, 1–25. [Google Scholar]
- Berridge, K.C.; Ho, C.-Y.; Richard, J.M.; DiFeliceantonio, A.G. The tempted brain eats: Pleasure and desire circuits in obesity and eating disorders. Brain Res. 2010, 1350, 43–64. [Google Scholar] [CrossRef]
- Lang, P.J. The emotion probe: Studies of motivation and attention. Am. Psychol. 1995, 50, 372. [Google Scholar] [CrossRef]
- Piqueras-Fiszman, B.; Kraus, A.A.; Spence, C. “Yummy” versus “Yucky”! Explicit and implicit approach–avoidance motivations towards appealing and disgusting foods. Appetite 2014, 78, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Russell, J.A.; Pratt, G. A description of the affective quality attributed to environments. J. Pers. Soc. Psychol. 1980, 38, 311. [Google Scholar] [CrossRef]
- Vansteelandt, K.; Claes, L.; Muehlenkamp, J.; De Cuyper, K.; Lemmens, J.; Probst, M.; Vanderlinden, J.; Pieters, G. Variability in affective activation predicts non-suicidal self-injury in eating disorders. Eur. Eating Disorders Rev. 2013, 21, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Roth, W.T. 8 A Comparison of P300 and Skin Conductance Response. In Advances in Psychology; Elsevier: Amsterdam, The Netherlands, 1983; Volume 10, pp. 177–199. [Google Scholar]
- Brouwer, A.; Zander, T.; van Erp, J.; Korteling, J.; Bronkhorst, A. Using neurophysiological signals that reflect cognitive or affective state: Six recommendations to avoid common pitfalls. Front. Neurosci. 2015, 9, 136. [Google Scholar] [CrossRef] [PubMed]
- Cacioppo, J.T.; Gardner, W.L. Emotion. Annu. Rev. Psychol. 1999, 50, 191–214. [Google Scholar] [CrossRef]
- Kreibig, S.D. Autonomic nervous system activity in emotion: A review. Biol. Psychol. 2010, 84, 394–421. [Google Scholar] [CrossRef] [PubMed]
- Gomes, L.M.; Silva, R.G.; Melo, M.; Silva, N.N.; Vanderlei, F.M.; Garner, D.M.; de Abreu, L.C.; Valenti, V.E. Effects of Effortful Swallow on Cardiac Autonomic Regulation. Dysphagia 2016, 31, 188–194. [Google Scholar] [CrossRef]
- Bradley, M.M.; Greenwald, M.K.; Petry, M.C.; Lang, P.J. Remembering pictures: Pleasure and arousal in memory. J. Exp. Psychol. Learn. Mem. Cogn. 1992, 18, 379–390. [Google Scholar] [CrossRef]
- Grossman, P.; Taylor, E.W. Toward understanding respiratory sinus arrhythmia: Relations to cardiac vagal tone, evolution and biobehavioral functions. Biol. Psychol. 2007, 74, 263–285. [Google Scholar] [CrossRef]
- Berntson, G.G.; Thomas Bigger, J.; Eckberg, D.L.; Grossman, P.; Kaufmann, P.G.; Malik, M.; Nagaraja, H.N.; Porges, S.W.; Saul, J.P.; Stone, P.H. Heart rate variability: Origins, methods, and interpretive caveats. Psychophysiology 1997, 34, 623–648. [Google Scholar] [CrossRef]
- Brouwer, A.; Hogervorst, M.; Grootjen, M.; van Erp, J.; Zandstra, E. Neurophysiological responses during cooking food associated with different emotions. Food Qual. Prefer. 2017. [Google Scholar] [CrossRef]
- Ohgami, Y.; Kotani, Y.; Tsukamoto, T.; Omura, K.; Inoue, Y.; Aihara, Y.; Nakayama, M. Effects of monetary reward and punishment on stimulus-preceding negativity. Psychophysiology 2006, 43, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Harmon-Jones, E.; Gable, P.A.; Peterson, C.K. The role of asymmetric frontal cortical activity in emotion-related phenomena: A review and update. Biol. Psychol. 2010, 84, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Harmon-Jones, E.; Gable, P.A. Neural activity underlying the effect of approach-motivated positive affect on narrowed attention. Psychol. Sci. 2009, 20, 406–409. [Google Scholar] [CrossRef] [PubMed]
- Gable, P.; Harmon-Jones, E. Relative left frontal activation to appetitive stimuli: Considering the role of individual differences. Psychophysiology 2008, 45, 275–278. [Google Scholar] [CrossRef] [PubMed]
- WorldMedicalAssociation. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. J. Korean Med. Assoc. 2014, 81, 14. [Google Scholar]
- Bradley, M.M.; Lang, P.J. Measuring emotion: The self-assessment manikin and the semantic differential. J. Behav. Ther. Exp. Psychiatry 1994, 25, 49–59. [Google Scholar] [CrossRef]
- Brouwer, A.; Hogervorst, M.; Reuderink, B.; van der Werf, Y.; van Erp, J. Physiological signals distinguish between reading emotional and non-emotional sections in a novel. Brain-Comput. Interfaces 2015, 2, 76–89. [Google Scholar] [CrossRef]
- Hogervorst, M.A.; Brouwer, A.-M.; Van Erp, J.B. Combining and comparing EEG, peripheral physiology and eye-related measures for the assessment of mental workload. Front. Neurosci. 2014, 8, 322. [Google Scholar] [CrossRef]
- Appelhans, B.M.; Luecken, L.J. Heart rate variability as an index of regulated emotional responding. Rev. Gen. Psychol. 2006, 10, 229. [Google Scholar] [CrossRef]
- Oostenveld, R.; Fries, P.; Maris, E.; Schoffelen, J.-M. FieldTrip: Open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput. Intell. Neurosci. 2011, 2011, 1. [Google Scholar] [CrossRef] [PubMed]
- Bell, A.J.; Sejnowski, T.J. An information-maximization approach to blind separation and blind deconvolution. Neural Comput. 1995, 7, 1129–1159. [Google Scholar] [CrossRef] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Papousek, I.; Weiss, E.M.; Schulter, G.; Fink, A.; Reiser, E.M.; Lackner, H.K. Prefrontal EEG alpha asymmetry changes while observing disaster happening to other people: Cardiac correlates and prediction of emotional impact. Biol. Psychol. 2014, 103, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Goedhart, A.D.; Van der Sluis, S.; Houtveen, J.H.; Willemsen, G.; De Geus, E.J. Comparison of time and frequency domain measures of RSA in ambulatory recordings. Psychophysiology 2007, 44, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Camm, A.J.; Malik, M.; Bigger, J.; Breithardt, G.; Cerutti, S.; Cohen, R.; Coumel, P.; Fallen, E.; Kennedy, H.; Kleiger, R. Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996, 93, 1043–1065. [Google Scholar]

| Measures | Discrimination Power: Vinegar vs. Regular Drinks (z) |
|---|---|
| Valence | 14.84 |
| Arousal | 13.42 |
| Sip size | 19.50 |
| FRdisgust | 5.97 |
| SCL | 7.97 |
| IBI | 10.99 |
| HRV | 1.94 |
| PD | 4.33 |
| FAA | 2.63 |
| Measures | Regular Drink Sensitivity (F) | Regular Drink Sensitivity (p) |
|---|---|---|
| Valence | 10.6 | <0.001 |
| Arousal | 10.1 | <0.001 |
| Sip size | 3.10 | 0.003 |
| FRdisgust | 1.83 | 0.080 |
| SCL | 1.51 | 0.162 |
| IBI | 17.0 | <0.001 |
| HRV | 1.31 | 0.244 |
| PD | 0.87 | 0.532 |
| FAA | 1.16 | 0.327 |
| Apple Juice | Orange Juice | Milk | Buttermilk | Yogurt Drink | Coke | Rooibos Tea | Black Tea | |
|---|---|---|---|---|---|---|---|---|
| Apple Juice | - | ns | V, A, I | ns | ns | ns | A, I | A, I |
| Orange Juice | - | V, A, I | V, I | ns | V, A, I | V, A, I | V, A, I | |
| Milk | - | A, I | V, A, I | I | S | ns | ||
| Buttermilk | - | V | ns | A, S, I | A, I | |||
| Yogurt Drink | - | V | V, A, I | V, A, I | ||||
| Coke | - | I | I | |||||
| Rooibos Tea | - | ns | ||||||
| Black Tea | - |
| Valence | Arousal | |||
|---|---|---|---|---|
| ρ | p-Value | ρ | p-Value | |
| Sip size | 0.2290 | 0.585 | −0.3051 | 0.462 |
| FRdisgust | −0.2285 | 0.586 | −0.0720 | 0.866 |
| SCL | −0.3488 | 0.397 | −0.3718 | 0.365 |
| IBI | −0.7326 | 0.039 | −0.9225 | 0.001 |
| HRV | −0.6413 | 0.087 | −0.7093 | 0.049 |
| PD | 0.0189 | 0.965 | 0.2210 | 0.599 |
| FAA | 0.3570 | 0.385 | 0.6180 | 0.102 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaneko, D.; Hogervorst, M.; Toet, A.; van Erp, J.B.F.; Kallen, V.; Brouwer, A.-M. Explicit and Implicit Responses to Tasting Drinks Associated with Different Tasting Experiences. Sensors 2019, 19, 4397. https://doi.org/10.3390/s19204397
Kaneko D, Hogervorst M, Toet A, van Erp JBF, Kallen V, Brouwer A-M. Explicit and Implicit Responses to Tasting Drinks Associated with Different Tasting Experiences. Sensors. 2019; 19(20):4397. https://doi.org/10.3390/s19204397
Chicago/Turabian StyleKaneko, Daisuke, Maarten Hogervorst, Alexander Toet, Jan B. F. van Erp, Victor Kallen, and Anne-Marie Brouwer. 2019. "Explicit and Implicit Responses to Tasting Drinks Associated with Different Tasting Experiences" Sensors 19, no. 20: 4397. https://doi.org/10.3390/s19204397
APA StyleKaneko, D., Hogervorst, M., Toet, A., van Erp, J. B. F., Kallen, V., & Brouwer, A.-M. (2019). Explicit and Implicit Responses to Tasting Drinks Associated with Different Tasting Experiences. Sensors, 19(20), 4397. https://doi.org/10.3390/s19204397







