A Diagnostic Device for In-Situ Detection of Swine Viral Diseases: The SWINOSTICS Project
Abstract
1. Introduction
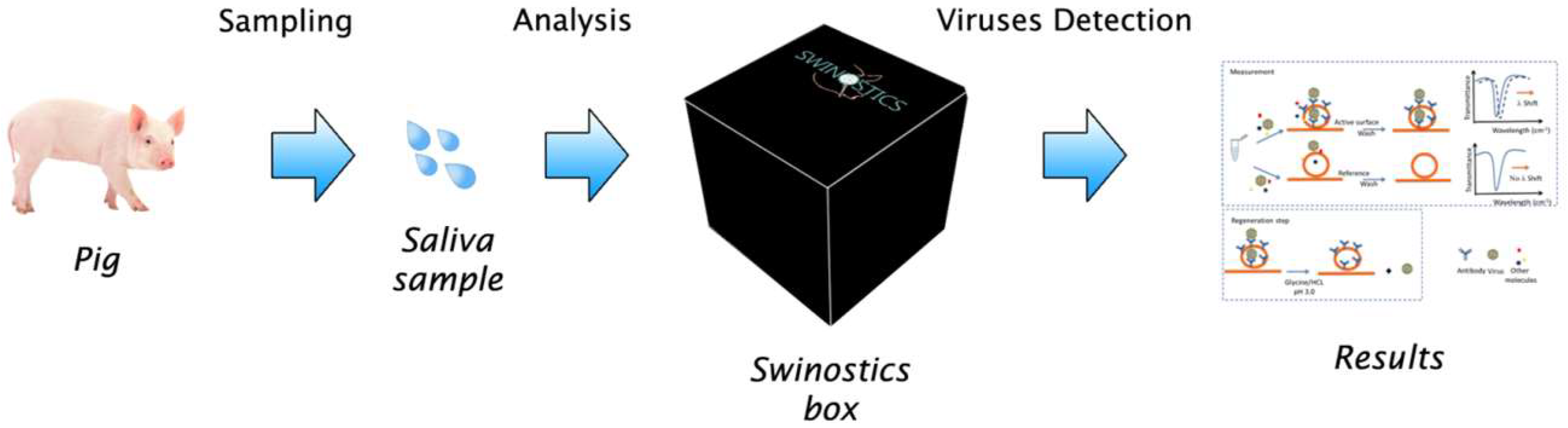
2. SWINOSTICS Overall Concept
- Photonic Integrated Circuit (PIC) technology, as the basis for the development of the biosensors;
- Label-free optical detection, based on refractive index sensing through ring resonators;
SWINOSTICS innovation
- The SWINOSTICS device will be the only field-use device capable of the detection of six important swine viruses of particular interest for the European and global economy.
- The device will use oral fluids as a sample for the analysis. This simplifies the sampling and minimizes the time needed for the test (no sample treatment is needed) allowing also the analysis of wild boar samples collected through adequate lures. The device will anyway be compatible with the use of other types of samples, such as faeces, blood or nasal swabs.
- The sample analysis will require less than 30 min. Additionally, five sensors, working in parallel, will be available within the device, to process up to five samples from five different animals simultaneously.
- The detection of the infectious agents is made using novel biosensors. The patented, nano-deposition technology used to develop the bio-sensing surface allows the specific determination of the presence of the biomarkers of interest.
- The PIC sensors that will be used in the device do not require the use of a fluorescent label for the detection (label-free detection).
- The increased sensitivity of the sensors is supported by the use of commercial monoclonal antibodies against the targeted viruses, which will be immobilized in an oriented manner on the PIC surface.
- The expected sensitivity and specificity levels are about 95% and 90%, respectively.
- Cost-effective and mass production suitable fabrication process of the PIC sensors. The fabrication technology will be CMOS (Complementary metal–oxide–semiconductor)-compatible.
- The device will integrate a biosensor regeneration mechanism, to make each PIC reusable for at least up to 100 times.
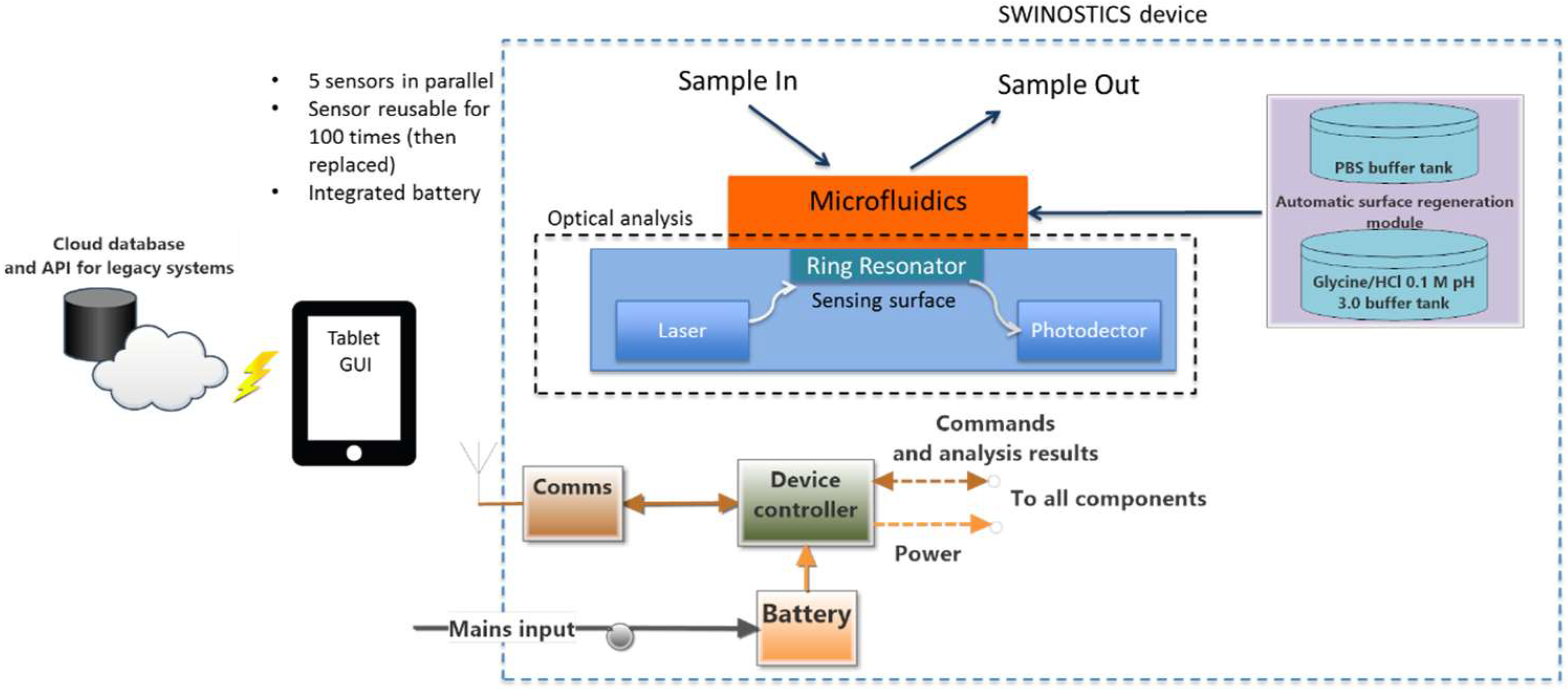
3. SWINOSTICS Device Overall Design and Architecture
3.1. SWINOSTICS Biosensor
3.1.1. Microfluidic Subsystem
3.1.2. Photonic Transducer
3.1.3. Optical Analysis Module
3.1.4. Temperature Conditioning Module
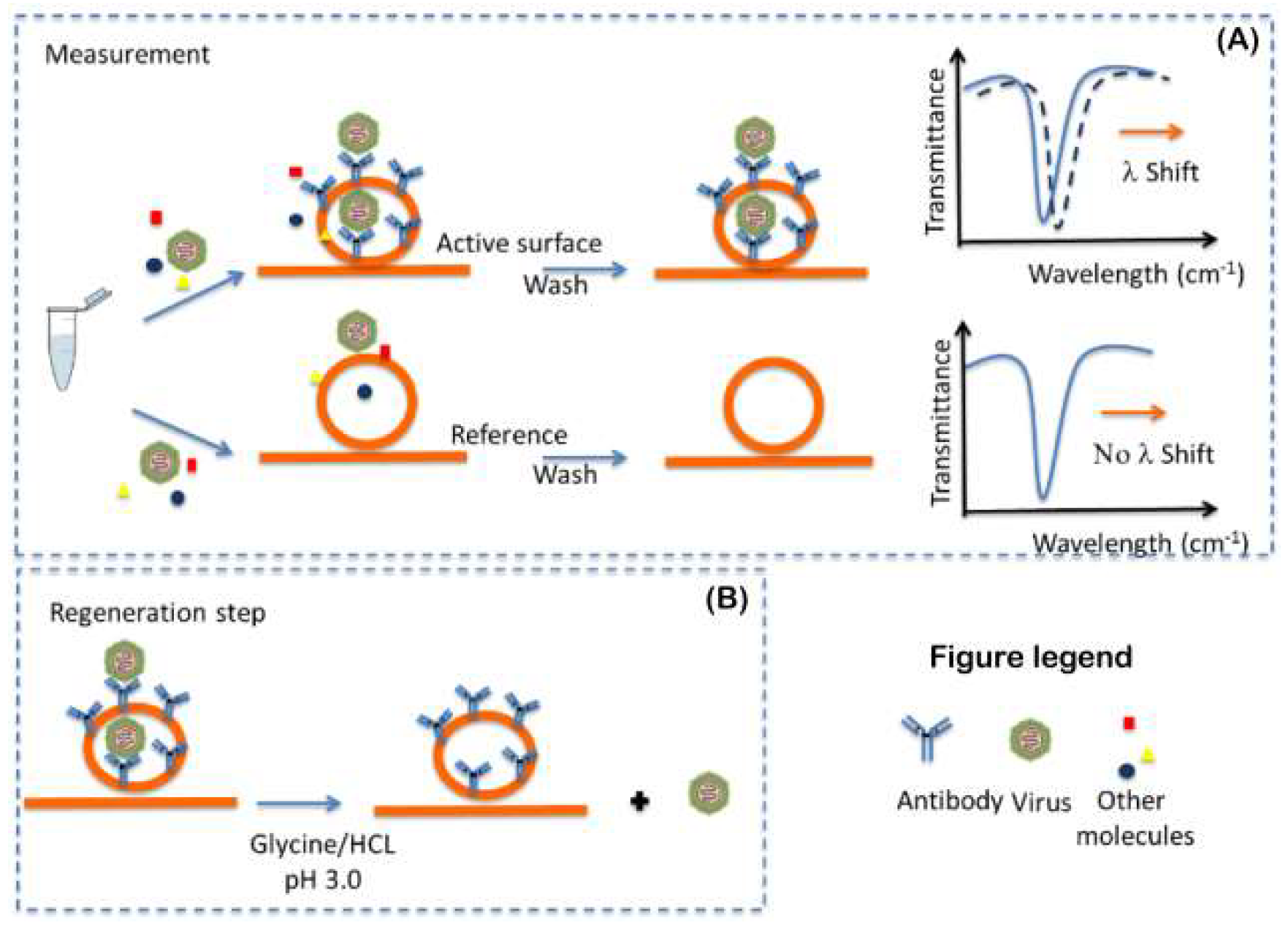
3.2. The Process for Bio-Sensing Surface Regeneration and Preservation
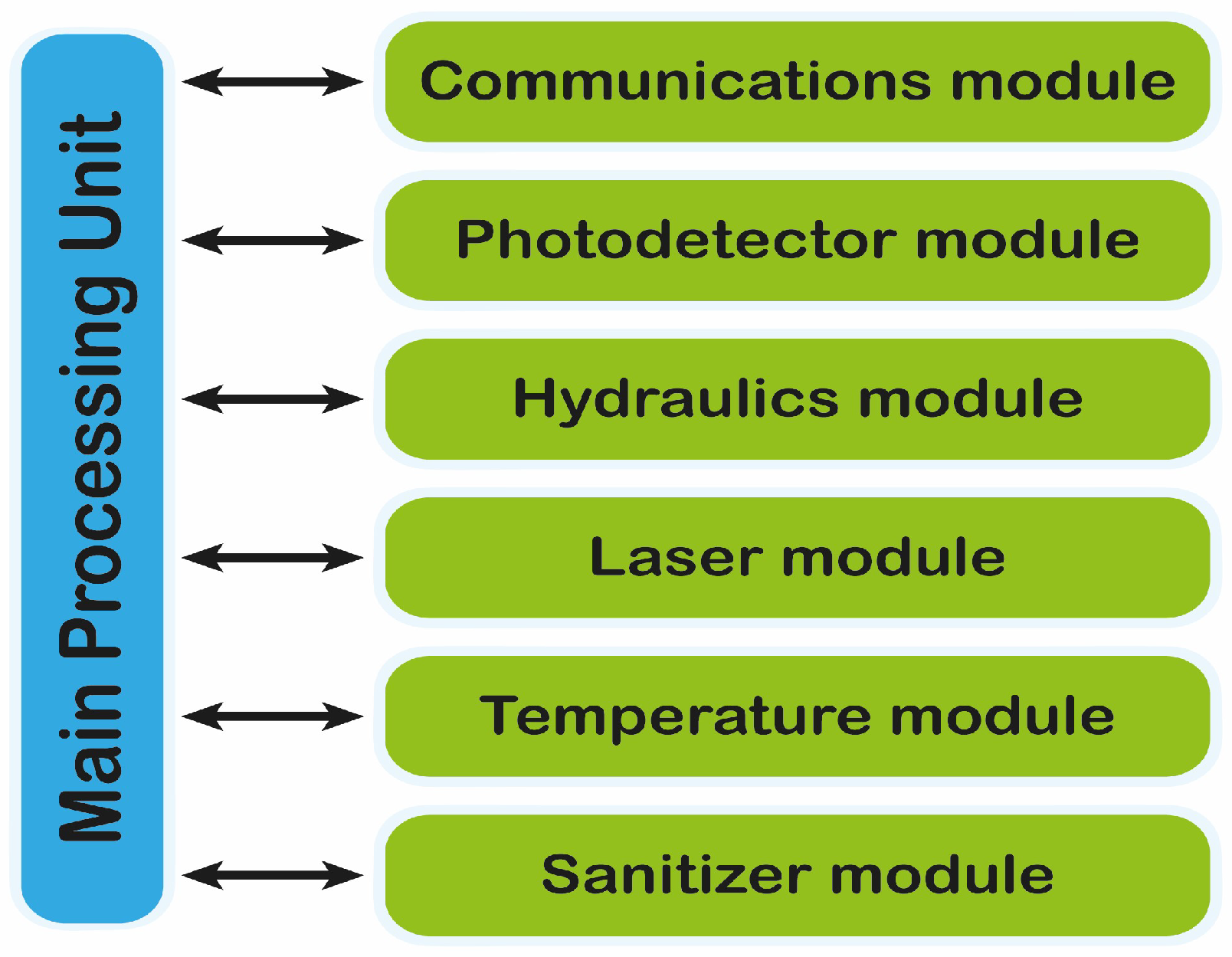
3.3. Processing, Control and Communication Module
4. Field Validation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- United Nations. World Population Prospects. Available online: https://population.un.org/wpp/ (accessed on 9 December 2018).
- FAO. Food and Agriculture Organization of the United Nations Statistical Databases. 2010. Available online: http://www.fao.org/ (accessed on 9 December 2018).
- Perry, B.; Sones, K. Global Livestock Disease Dynamics over the Last Quarter Century: Drivers, Impacts and Implications; Background paper for the SOFA 2009; FAO: Rome, Italy, 2009. [Google Scholar]
- Niemz, A.; Ferguson, T.M. Point-of-care nucleic acid testing for infectious diseases. Trends Biotechnol. 2011, 29, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Holland, C.A.; Kiechle, F.L. Point-of-care molecular diagnostic systems—Past, present and future. Curr. Opin. Microbiol. 2005, 8, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Zhang, Y. Advances in microfluidic PCR for point-of-care infectious disease diagnostics. Biotechnol. Adv. 2011, 29, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Craw, P.; Balachandran, W. Isothermal nucleic acid amplification technologies for point-of-care diagnostics: A critical review. Lab Chip 2012, 21, 2469–2486. [Google Scholar] [CrossRef] [PubMed]
- Asiello, P.J.; Baeumner, A.J. Miniaturized isothermal nucleic acid amplification, a review. Lab Chip 2011, 11, 1420–1430. [Google Scholar] [CrossRef]
- Jahns, S.; Bräu, M. Handheld imaging photonic crystal biosensor for multiplexed, label-free protein detection. Biomed. Opt. Express 2015, 6, 3724–3736. [Google Scholar] [CrossRef] [PubMed]
- Ksendzov, A.; Lin, Y. Integrated optics ring-resonator sensors for protein detection. Opt. Lett. 2005, 30, 3344–3346. [Google Scholar] [CrossRef]
- Lopez, G.; Estevez, M. Recent advances in nanoplasmonic biosensors: Applications and lab-on-a-chip integration. Nanophotonics 2017, 6, 1–14. [Google Scholar] [CrossRef]
- Swinostics. Available online: http://swinostics.eu/ (accessed on 9 December 2018).
- STAR—IDAZ. Available online: https://www.star-idaz.net/organisation/ (accessed on 9 December 2018).
- ASF Regionalization in the EU. Available online: https://ec.europa.eu/food/sites/food/files/animals/docs/ad_control-measures_asf_pl-lt-regionalisation.pdf (accessed on 9 December 2018).
- Classical Swine Fever (Hog Cholera). 2009. Available online: http://www.oie.int/fileadmin/Home/eng/Animal_Health_in_the_World/docs/pdf/Disease_cards/CLASSICAL_SWINE_FEVER.pdf (accessed on 9 December 2018).
- Kuhn, J.H.; Lauck, M. Reorganization and expansion of the nidoviral family Arteriviridae. Arch. Virol. 2016, 161, 755–768. [Google Scholar] [CrossRef]
- Cui, J.; Biernacka, K. Circulation of Porcine Parvovirus Types 1 through 6 in Serum Samples Obtained from Six Commercial Polish Pig Farms. Transbound. Emerg. Dis. 2017, 64, 1945–1952. [Google Scholar] [CrossRef]
- Oliver-Ferrando, S.; Segalés, J. Evaluation of natural porcine circovirus type 2 (PCV2) subclinical infection and seroconversion dynamics in piglets vaccinated at different ages. Vet. Res. 2016, 47, 121. [Google Scholar] [CrossRef]
- Castrucci, M.R.; Donatelli, I. Genetic reassortment between avian and human influenza A viruses in Italian pigs. Virology 1993, 193, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Leland, D.S.; Ginocchio, C.C. Role of Cell Culture for Virus Detection in the Age of Technology. Clin. Microbiol. Rev. 2007, 20, 49–78. [Google Scholar] [CrossRef] [PubMed]
- Wongsrichanalai, C.; Barcus, M.J. A review of malaria diagnostic tools: Microscopy and rapid diagnostic test (RDT). Am. J. Trop. Med. Hyg. 2007, 77, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Baron, E.J.; Miller, J.M. A guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2013 recommendations by the Infectious Diseases Society of America (IDSA) and the American Society for Microbiology (ASM). Clin. Infect. Dis. 2013, 57, 22–121. [Google Scholar] [CrossRef] [PubMed]
- Reichel, M.P.; Lanyon, S.R. Moving past serology: Diagnostic options without serum. Vet. J. 2016, 215, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Josko, D. Molecular virology in the clinical laboratory. Clin. Lab. Sci. 2010, 23, 231–236. [Google Scholar]
- Scagnolari, C.; Turriziani, O. Consolidation of molecular testing in clinical virology. Expert Rev. Anti-Infect. Ther. 2017, 15, 387–400. [Google Scholar] [CrossRef]
- Watzinger, F.; Ebner, K. Detection and monitoring of virus infections by real-time PCR. Mol. Asp. Med. 2006, 27, 254–298. [Google Scholar] [CrossRef]
- Peccoud, J.; Jacob, C. Theoretical uncertainty of measurements using quantitative polymerase chain reaction. Biophys. J. 1996, 71, 101–108. [Google Scholar] [CrossRef]
- Broude, N.E.; Zhang, L. Multiplex allele-specific target amplification based on PCR suppression. Proc. Natl. Acad. Sci. USA 2001, 98, 206–211. [Google Scholar] [CrossRef]
- Syvänen, A.C. Toward genome-wide SNP genotyping. Nat. Genet. 2005, 37, S5–S10. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Dineva, M.A. Simple amplification-based assay: A nucleic acid-based point-of-care platform for HIV-1 testing. J. Infect. Dis. 2010, 15, S65–S72. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.J.; Park, K. Isothermal DNA amplification in vitro: The helicase-dependent amplification system. Cell. Mol. Life Sci. 2006, 66, 3325–3336. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Notomi, T. Loop-mediated isothermal amplification (LAMP): A rapid, accurate, and cost-effective diagnostic method for infectious diseases. J. Infect. Chemother. 2009, 15, 62–69. [Google Scholar] [CrossRef]
- Fang, X.; Liu, Y. Loop-mediated isothermal amplification integrated on microfluidic chips for point-of-care quantitative detection of pathogens. Anal. Chem. 2010, 82, 3002–3006. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Yamamoto, T. Quantification of Virus Particles Using Nanopore-Based Resistive-Pulse Sensing Techniques. Front. Microbiol. 2016, 7, 1500. [Google Scholar] [CrossRef]
- Fu, E.; Chinowsky, T. SPR imaging-based salivary diagnostics system for the detection of small molecule analytes. Ann. N. Y. Acad. Sci. 2007, 1098, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Ly, N.; Foley, K. Integrated label-free protein detection and separation in real time using confined surface plasmon resonance imaging. Anal. Chem. 2007, 79, 2546–2551. [Google Scholar] [CrossRef]
- AFRICAN_SWINE_FEVER. 2013. Available online: https://www.oie.int/fileadmin/Home/eng/Animal_Health_in_the_World/docs/pdf/Disease_cards/AFRICAN_SWINE_FEVER.pdf (accessed on 9 December 2018).
- Grau, F.R.; Schroeder, M.E. Detection of African swine fever, classical swine fever, and foot-and-mouth disease viruses in swine oral fluids by multiplex reverse transcription real-time polymerase chain reaction. J. Vet. Diagn. Investig. 2015, 27, 140–149. [Google Scholar] [CrossRef]
- Davies, K.; Goatley, L.C. Survival of African Swine Fever Virus in Excretions from Pigs Experimentally Infected with the Georgia 2007/1 Isolate. Transbound. Emerg. Dis. 2017, 64, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Petrov, A.; Schotte, U. Alternative sampling strategies for passive classical and African swine fever surveillance in wild boar. Vet. Microbiol. 2014, 173, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Kittawornrat, A.; Prickett, J. Porcine reproductive and respiratory syndrome virus (PRRSV) in serum and oral fluid samples from individual boars: Will oral fluid replace serum for PRRSV surveillance? Virus Res. 2010, 154, 170–176. [Google Scholar] [CrossRef]
- Pepin, B.J.; Kittawornrat, A. Comparison of specimens for detection of porcine reproductive and respiratory syndrome virus infection in boar studs. Transbound. Emerg. Dis. 2015, 62, 295–304. [Google Scholar] [CrossRef]
- Detmer, S.E.; Patnayak, D.P. Detection of Influenza A virus in porcine oral fluid samples. J. Vet. Diagn. Investig. 2011, 23, 241–247. [Google Scholar] [CrossRef]
- Decorte, I.; Steensels, M. Detection and Isolation of Swine Influenza A Virus in Spiked Oral Fluid and Samples from Individually Housed, Experimentally Infected Pigs: Potential Role of Porcine Oral Fluid in Active Influenza A Virus Surveillance in Swine. PLoS ONE 2015, 10, e0139586. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.T.; Gerber, P.F. Characterization of porcine parvovirus type 2 (PPV2) which is highly prevalent in the USA. Vet. Microbiol. 2012, 161, 325–330. [Google Scholar] [CrossRef]
- Truyen, U. Porcine Parvovirus in Diseases of Swine, 10th ed.; Zimmerman, J.J., Karriker, L.A., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2012; ISBN 9780813822679. [Google Scholar]
- Ogawa, H.; Taira, O. Multiplex PCR and multiplex RT-PCR for inclusive detection of major swine DNA and RNA viruses in pigs with multiple infections. J. Virol. Methods 2009, 160, 210–214. [Google Scholar] [CrossRef]
- Yang, Y.; Qin, X. Rapid and specific detection of porcine parvovirus by isothermal recombinase polymerase amplification assays. Mol. Cell. Probes 2016, 30, 300–305. [Google Scholar] [CrossRef]
- Prickett, J.R.; Zimmerman, J.J. The development of oral fluid-based diagnostics and applications in veterinary medicine. Anim. Health Res. Rev. 2010, 11, 207–216. [Google Scholar] [CrossRef]
- D’Auria, S.; Borini, S.M.; Rossi, A.M.; Rossi, M. Process of Immobilizing Biomolecules in Porous Supports by Using an Electronic Beam. U.S. Patent 20090036326, 5 February 2009. [Google Scholar]
- D’Auria, S.; Borini, S.M.; Rossi, A.M; Rossi, M. Process of Immobilizing Biomolecules in Porous Supports by Using an Electronic Beam. EP Patent 1819434 A1, 22 August 2007. [Google Scholar]
- Varriale, A.; Bonnot, K. Self-oriented monolayer immobilization of ovalbumin and B. cereus antibody molecules on a chemically modified surface of silicon nitride fosters the enhancement of capture of bio-agents. Colloids Surf. B Biointerfaces 2016, 148, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Thermoelectric Modules. Available online: https://thermal.ferrotec.com/technology/ (accessed on 9 December 2018).


| Targeted Virus | Sample usually Used | SWINOSTICS Approach |
|---|---|---|
| African Swine Fever (ASFV) | Whole blood or blood serum in live animals. Tissues from post-mortem animals such as tonsil, spleen and lymph nodes [37] | Oral fluid [38,39,40] in live animals, blood serum from post-mortem animals [41] |
| Porcine Reproductive and Respiratory Syndrome (PRRSV) | Whole blood and blood serum in live animals. Tissues such as lung, respiratory tract, spleen and tonsils in post-mortem animals [42]) | Oral fluid and blood serum [43,44] |
| Swine Influenza A (SIV) | Nasal swabs and lung tissues [45] | Oral fluid and nasal swabs [46,47] |
| Porcine Parvovirus (PPV) | Whole mummified small foetuses/lung tissue from aborted foetuses [48,49] | Oral fluid and faeces [49] |
| Porcine Circovirus (PCV2) | Blood serum, bronchiolar lavage fluid, tissue homogenates | Oral fluid [18] |
| Classical Swine Fever (CSF) | Whole blood in live animals. Tissues such as tonsils, pharyngeal or mesenteric lymph nodes, spleen, kidney, and distal ileum in post-mortem animals [15] | Oral fluid [40,49] in live animals, blood serum from post-mortem animals [49] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montagnese, C.; Barattini, P.; Giusti, A.; Balka, G.; Bruno, U.; Bossis, I.; Gelasakis, A.; Bonasso, M.; Philmis, P.; Dénes, L.; et al. A Diagnostic Device for In-Situ Detection of Swine Viral Diseases: The SWINOSTICS Project. Sensors 2019, 19, 407. https://doi.org/10.3390/s19020407
Montagnese C, Barattini P, Giusti A, Balka G, Bruno U, Bossis I, Gelasakis A, Bonasso M, Philmis P, Dénes L, et al. A Diagnostic Device for In-Situ Detection of Swine Viral Diseases: The SWINOSTICS Project. Sensors. 2019; 19(2):407. https://doi.org/10.3390/s19020407
Chicago/Turabian StyleMontagnese, Concetta, Paolo Barattini, Alessandro Giusti, Gyula Balka, Ugo Bruno, Ioannis Bossis, Athanasios Gelasakis, Matteo Bonasso, Panayiotis Philmis, Lilla Dénes, and et al. 2019. "A Diagnostic Device for In-Situ Detection of Swine Viral Diseases: The SWINOSTICS Project" Sensors 19, no. 2: 407. https://doi.org/10.3390/s19020407
APA StyleMontagnese, C., Barattini, P., Giusti, A., Balka, G., Bruno, U., Bossis, I., Gelasakis, A., Bonasso, M., Philmis, P., Dénes, L., Peransi, S., Rodrigo, M., Simón, S., Griol, A., Wozniakowski, G., Podgorska, K., Pugliese, C., Nannucci, L., D’Auria, S., & Varriale, A. (2019). A Diagnostic Device for In-Situ Detection of Swine Viral Diseases: The SWINOSTICS Project. Sensors, 19(2), 407. https://doi.org/10.3390/s19020407










