Turning Analysis during Standardized Test Using On-Shoe Wearable Sensors in Parkinson’s Disease
Abstract
1. Introduction
2. Methods
2.1. Wearable Measurement System
2.2. Study Population
2.3. Turning Isolation
2.4. Turning Parameters
2.5. Statistical Analysis
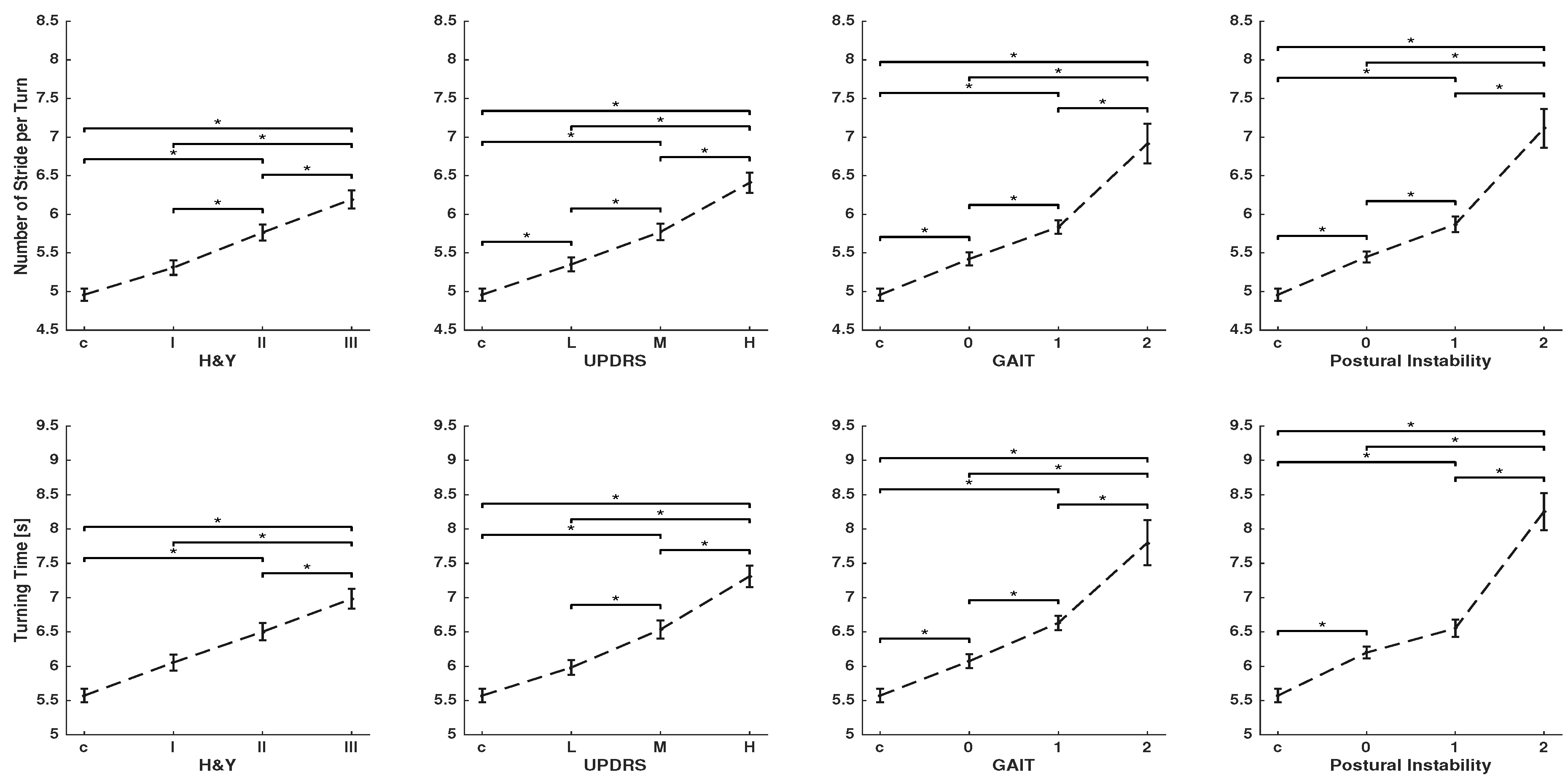
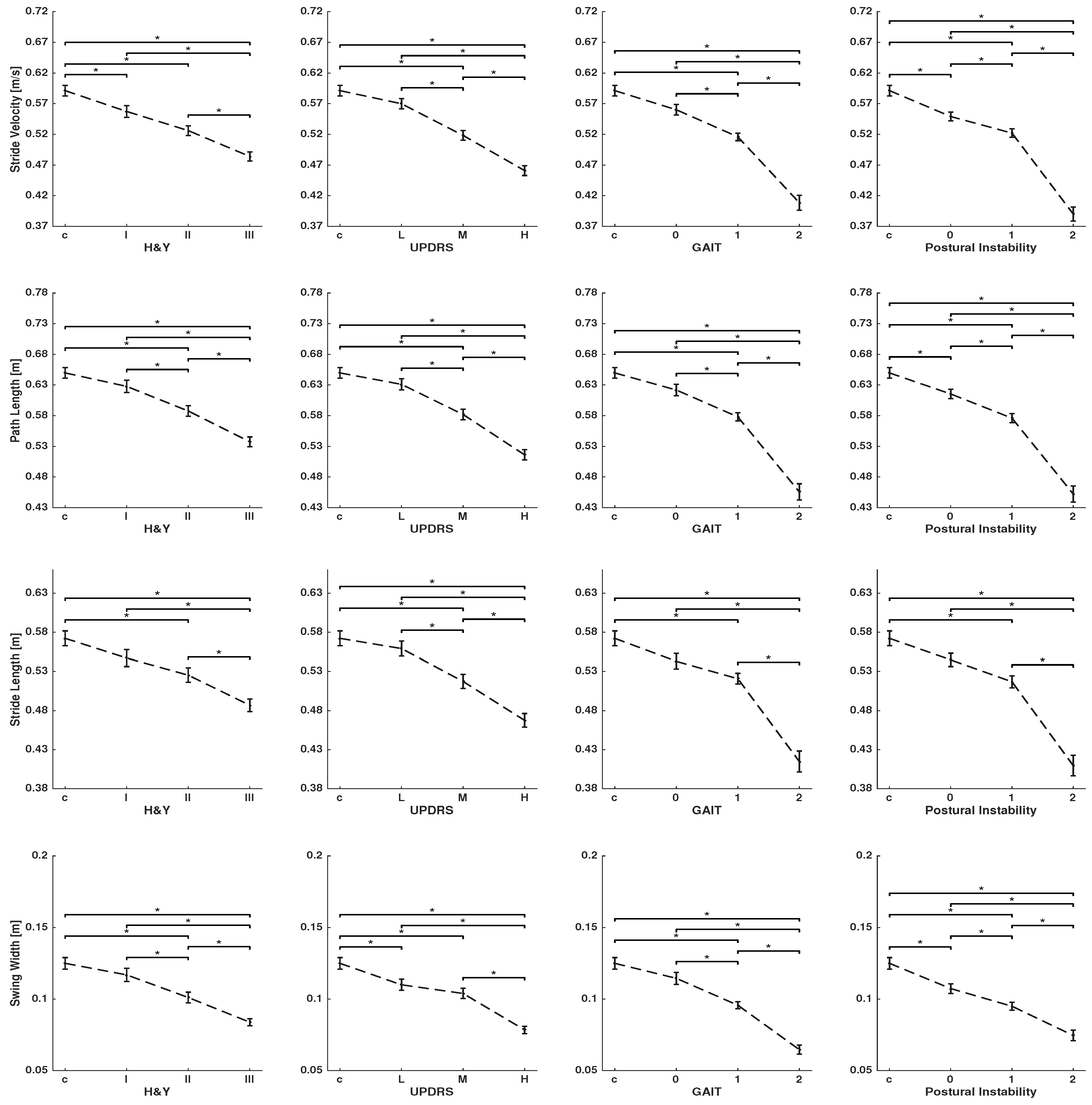
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Stack, E.; Ashburn, A. Dysfunctional turning in Parkinson’s disease. Disabil. Rehabil. 2008, 30, 1222–1229. [Google Scholar] [CrossRef] [PubMed]
- Crenna, P.; Carpinella, I.; Rabuffetti, M.; Calabrese, E.; Mazzoleni, P.; Nemni, R.; Ferrarin, M. The association between impaired turning and normal straight walking in Parkinson’s disease. Gait Posture 2007, 26, 172–178. [Google Scholar] [CrossRef] [PubMed]
- El-Gohary, M.; Pearson, S.; McNames, J.; Mancini, M.; Horak, F.; Mellone, S.; Chiari, L. Continuous monitoring of turning in patients with movement disability. Sensors 2014, 14, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Pickering, R.M.; Grimbergen, Y.A.; Rigney, U.; Ashburn, A.; Mazibrada, G.; Wood, B.; Gray, P.; Kerr, G.; Bloem, B.R. A meta-analysis of six prospective studies of falling in Parkinson’s disease. Mov. Disord. 2007, 22, 1892–1900. [Google Scholar] [CrossRef] [PubMed]
- Stack, E.; Jupp, K.; Ashburn, A. Developing methods to evaluate how people with Parkinson’s disease turn 180∘: An activity frequently associated with falls. Disabil. Rehabil. 2004, 26, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Mancini, M.; Weiss, A.; Herman, T.; Hausdorff, J.M. Turn Around Freezing: Community-Living Turning Behavior in People with Parkinson’s Disease. Front. Neurol. 2018, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Moore, O.; Peretz, C.; Giladi, N. Freezing of gait affects quality of life of peoples with Parkinson’s disease beyond its relationships with mobility and gait. Mov. Disord. 2007, 22, 219–2195. [Google Scholar] [CrossRef]
- Bachlin, M.; Plotnik, M.; Roggen, D.; Maidan, I.; Hausdorff, J.M.; Giladi, N.; Troster, G. Wearable assistant for Parkinson’s disease patients with the freezing of gait symptom. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 436–446. [Google Scholar] [CrossRef]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression and mortality. Neurology 1967, 17, 427–442. [Google Scholar] [CrossRef]
- Stack, E.L.; Ashburn, A.A.; Jupp, K.E. Strategies used by people with Parkinson’s disease who report difficulty turning. Park. Rel. Disord. 2006, 12, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Mak, M.; Patle, A.; Hui-Chan, C. Sudden turn during walking is impaired in people with Parkinson’s disease. Exp. Brain Res. 2008, 190, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Huxham, F.; Baker, R.; Morris, M.E.; Iansek, R. Footstep adjustments used to turn during walking in Parkinson’s disease. Mov. Disord. 2008, 23, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.; Perlmutter, J.; Earhart, G.A. kinematic and electromyographic analysis of turning in people with Parkinson disease. Neurorehabil. Neural Repair 2009, 23, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.; Earhart, G.M. Effects of medication on turning deficits in individuals with Parkinson’s disease. J. Neurol. Phys. Ther. 2010, 34, 11–16. [Google Scholar] [CrossRef]
- King, L.; Mancini, M.; Priest, K.; Salarian, A.; Rodrigues-de Paula, F.; Horak, F. Do clinical scales of balance reflect turning abnormalities in people with Parkinson’s disease? J. Neurol. Phys. Ther. 2012, 36, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Salarian, A.; Zampieri, C.; Horak, F.; Carlson-Kuhta, P.; Nutt, J.; Aminian, K. Analyzing 180 Degrees Turns Using an Inertial System Reveals Early Signs of Progression of Parkinson’s Disease. In Proceedings of the 2009 IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 224–227. [Google Scholar]
- Huxham, F.; Baker, R.; Morris, M.E.; Iansek, R. Head and trunk rotation during walking turns in Parkinson’s disease. Mov. Disord. 2008, 23, 1391–1397. [Google Scholar] [CrossRef]
- Mellone, S.; Mancini, M.; King, L.A.; Horak, F.B.; Chiari, L. The quality of turning in Parkinson’s disease: A compensatory strategy to prevent postural instability? J. Neuroeng. Rehabil. 2016, 13, 39. [Google Scholar] [CrossRef]
- Mariani, B.; Jimenez, M.; Vingerhoets, F.; Aminian, K. On-shoe wearable sensors for gait and turning assessment of patients with Parkinson’s disease. IEEE Trans. Biomed. Eng. 2013, 60, 155–158. [Google Scholar] [CrossRef]
- Panebianco, G.P.; Bisi, M.C.; Stagni, R.; Fantozzi, S. Analysis of the performance of 17 algorithms from a systematic review: Influence of sensor position, analysed variable and computational approach in gait timing estimation from IMU measurements. Gait Posture 2018, 66, 76–82. [Google Scholar] [CrossRef]
- Rampp, A.; Barth, J.; Schülein, S.; Gassmann, K.G.; Klucken, J.; Eskofier, B.M. Inertial sensor-based stride parameter calculation from gait sequences in geriatric patients. IEEE Trans. Biomed. Eng. 2014, 62, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Schlachetzki, J.C.; Barth, J.; Marxreiter, F.; Gossler, J.; Kohl, Z.; Reinfelder, S.; Gassner, H.; Aminian, K.; Eskofier, B.M.; Winkler, J.; et al. Wearable sensors objectively measure gait parameters in Parkinson’s disease. PLoS ONE 2017, 12, e0183989. [Google Scholar] [CrossRef] [PubMed]
- Gassner, H.; Raccagni, C.; Eskofier, B.M.; Klucken, J.; Wenning, G.K. The diagnostic scope of sensor-based gait analysis in atypical Parkinsonism: Further observations. Front. Neurol. 2019, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.J.; Daniel, S.E.; Kilford, L.; Lees, A.J. Accuracy of clinical diagnosis of idiopathic parkinson’s disease: A clinico-pathological study of 100 cases. J. Neurol. Neurosurg. Psychiatry 1992, 55, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, J. An Essay on the Shaking Palsy; Neely & Jones: London, UK, 1817. [Google Scholar]
- Haji Ghassemi, N.; Hannink, J.; Martindale, C.F.; Gassner, H.; Müller, M.; Klucken, J.; Eskofier, B.M. Segmentation of Gait Sequences in Sensor-Based Movement Analysis: A Comparison of Methods in Parkinson’s Disease. Sensors 2018, 18, 145. [Google Scholar] [CrossRef]
- Barth, J.; Oberndorfer, C.; Pasluosta, C.; Schülein, S.; Gassner, H.; Reinfelder, S.; Kugler, P.; Schuldhaus, D.; Winkler, J.; Klucken, J.; et al. Stride segmentation during free walk movements using multi-dimensional subsequence dynamic time warping on inertial sensor data. Sensors 2015, 15, 6419–6440. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Kanzler, C.M.; Barth, J.; Klucken, J.; Eskofier, B.M. Inertial sensor based gait analysis discriminates subjects with and without visual impairment caused by simulated macular degeneration. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2016, 2016, 4979–4982. [Google Scholar]
- Salarian, A.; Horak, F.B.; Carlson-Kuhta, P.; Nutt, J.; Zampieri, C.; Aminian, K. iTUG, a Sensitive and Reliable Measure of Mobility. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 18, 303–310. [Google Scholar] [CrossRef]
- Herman, T.; Giladi, N.; Hausdorff, J.M. Properties of the ‘timed up and go’ test: More than meets the eye. Gerontology 2011, 57, 203–210. [Google Scholar] [CrossRef]
- Klucken, J.; Barth, J.; Kugler, P.; Schlachetzki, J.; Henze, T.; Marxreiter, F.; Kohl, Z.; Steidl, R.; Hornegger, J.; Eskofier, B.; et al. Unbiased and Mobile Gait Analysis Detects Motor Impairment in Parkinson’s Disease. PLoS ONE 2013, 8, e56956. [Google Scholar] [CrossRef]

| PD (N = 108) | Control (N = 42) | |
|---|---|---|
| Age (years) | 57.61 ± 10.42 [36–85] | 58.78 ± 11.14 [41–84] |
| Sex (Male/Female) | 74/34 | 25/17 |
| Height (m) | 1.74 ± 0.1 | 1.73 ± 0.07 |
| BMI | 25.81 ± 3.71 | 26.48 ± 3.76 |
| Hoehn and Yahr stage | 2.06 ± 0.84 | |
| I (<1) | 28 | |
| II (1-2] | 34 | |
| III (2<) | 46 | |
| UPDRS-III total | 18.24 ± 9.8 [2–50] | |
| Low [0–12] | 36 | |
| [13–22] | 38 | |
| High [23<) | 34 | |
| Laterality based on Rigidity item (upper and lower extremity) | ||
| No rigidity or both sides | 22% | |
| Right side | 42% | |
| Left side | 36% | |
| Gait item | ||
| 0 [0] | 34 | |
| 1 (0–1] | 62 | |
| 2 (1–2] | 12 | |
| Postural stability item | ||
| 0 [0] | 46 | |
| 1 (0–1] | 49 | |
| 2 (1–2] | 13 |
| Parameters | H&Y | UPDRS | Gait | Postural Instability |
|---|---|---|---|---|
| Number of Strides per-Turn | 0.172 * | 0.2 * | 0.202 * | 0.232 * |
| Turning Time | 0.149 * | 0.199 * | 0.187 * | 0.228 * |
| Stride Velocity | 0.054 * | 0.057 * | 0.06 * | 0.069 * |
| Path Length | 0.054 * | 0.054 * | 0.06 * | 0.063 * |
| Stride Length | 0.03 * | 0.03 * | 0.034 * | 0.038 * |
| Mid Swing | 0.034 * | 0.035 * | 0.039 * | 0.029 * |
| Stride Time | 0.003 | 0.003 | 0.002 | 0.007 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haji Ghassemi, N.; Hannink, J.; Roth, N.; Gaßner, H.; Marxreiter, F.; Klucken, J.; Eskofier, B.M. Turning Analysis during Standardized Test Using On-Shoe Wearable Sensors in Parkinson’s Disease. Sensors 2019, 19, 3103. https://doi.org/10.3390/s19143103
Haji Ghassemi N, Hannink J, Roth N, Gaßner H, Marxreiter F, Klucken J, Eskofier BM. Turning Analysis during Standardized Test Using On-Shoe Wearable Sensors in Parkinson’s Disease. Sensors. 2019; 19(14):3103. https://doi.org/10.3390/s19143103
Chicago/Turabian StyleHaji Ghassemi, Nooshin, Julius Hannink, Nils Roth, Heiko Gaßner, Franz Marxreiter, Jochen Klucken, and Björn M. Eskofier. 2019. "Turning Analysis during Standardized Test Using On-Shoe Wearable Sensors in Parkinson’s Disease" Sensors 19, no. 14: 3103. https://doi.org/10.3390/s19143103
APA StyleHaji Ghassemi, N., Hannink, J., Roth, N., Gaßner, H., Marxreiter, F., Klucken, J., & Eskofier, B. M. (2019). Turning Analysis during Standardized Test Using On-Shoe Wearable Sensors in Parkinson’s Disease. Sensors, 19(14), 3103. https://doi.org/10.3390/s19143103







