The Investigation of a SAW Oxygen Gas Sensor Operated at Room Temperature, Based on Nanostructured ZnxFeyO Films
Abstract
:1. Introduction
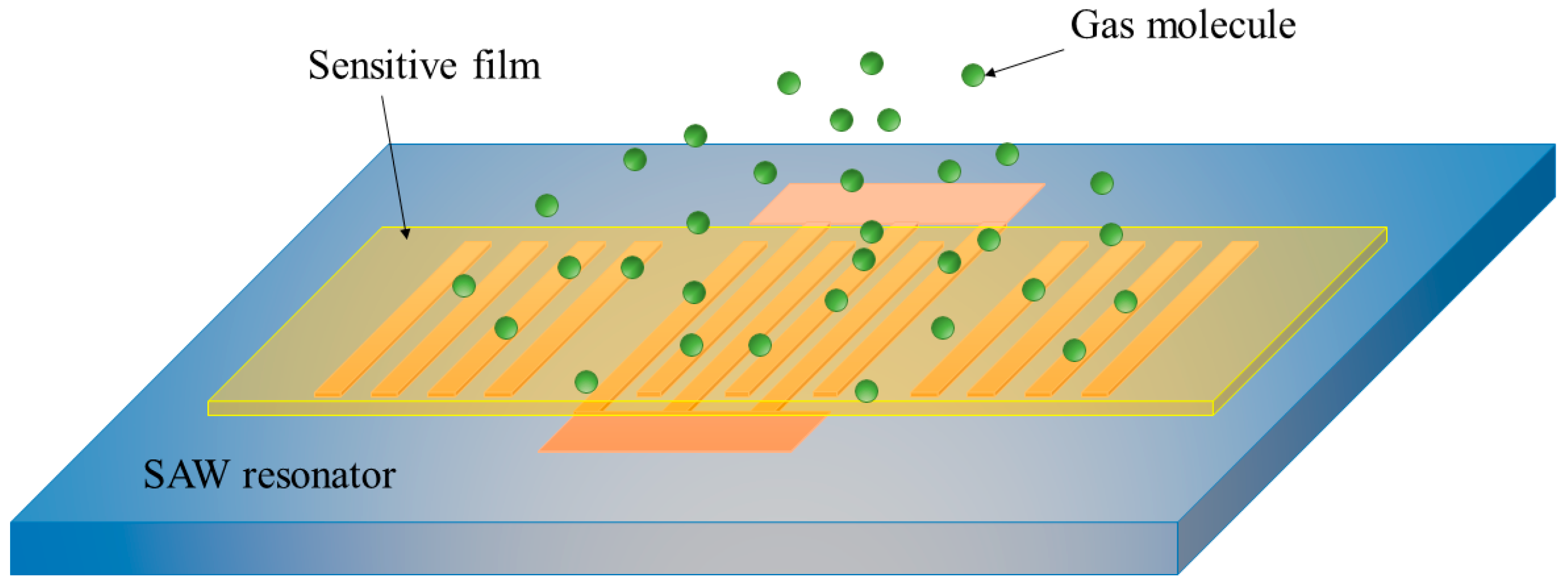
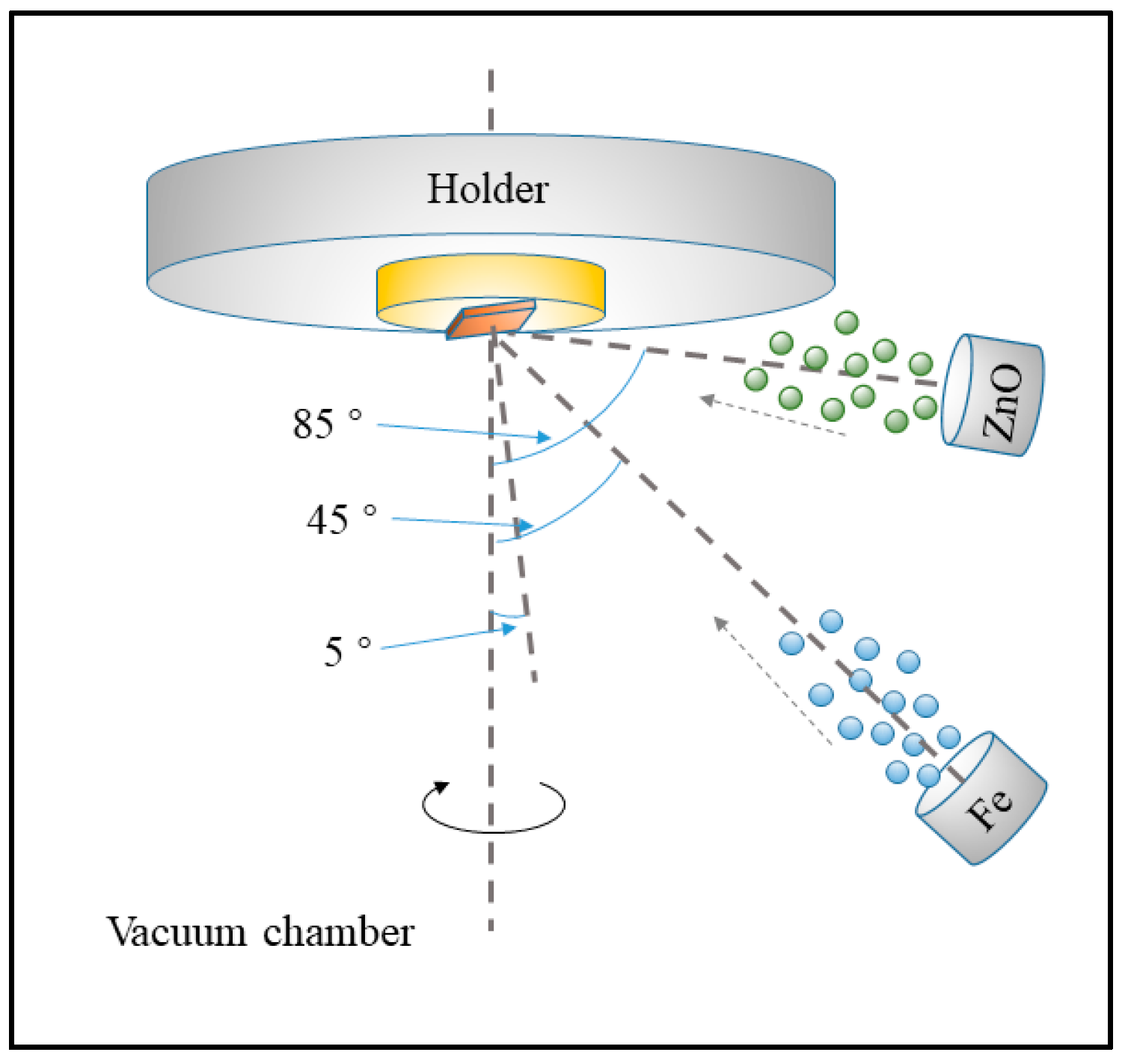
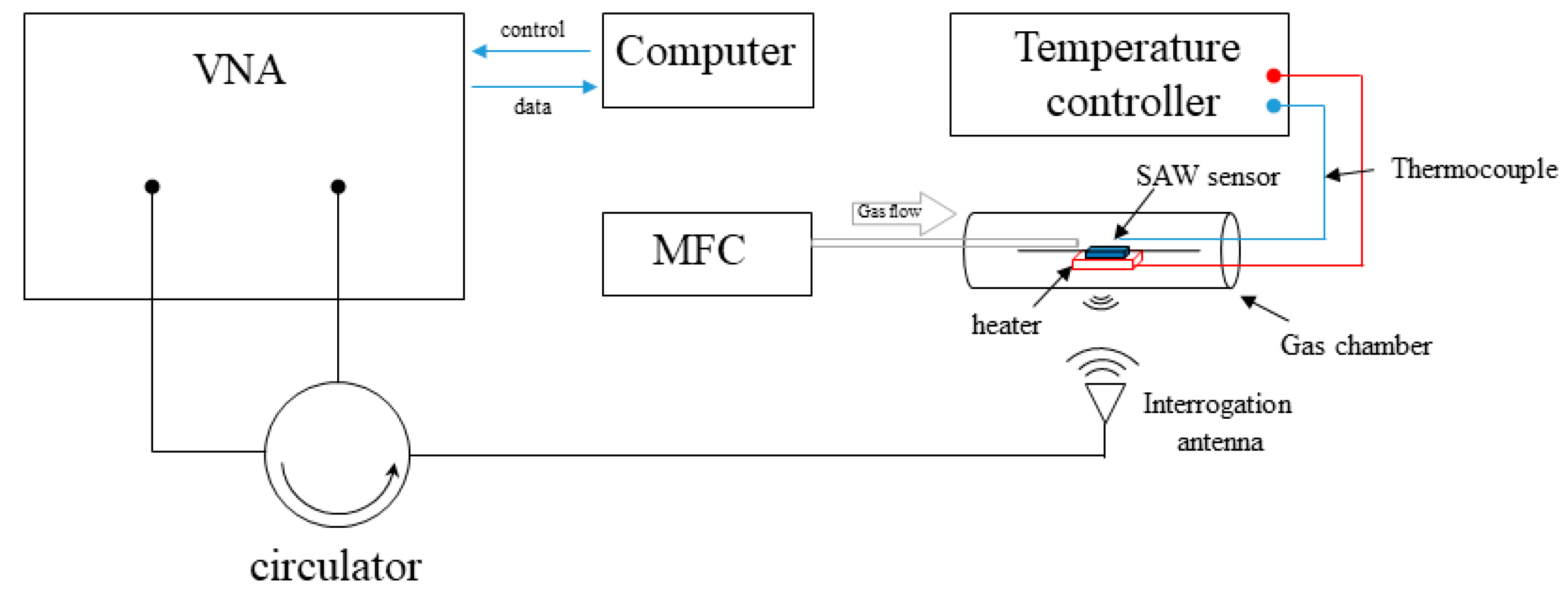
2. Experimental Setup
3. Results and Discussions
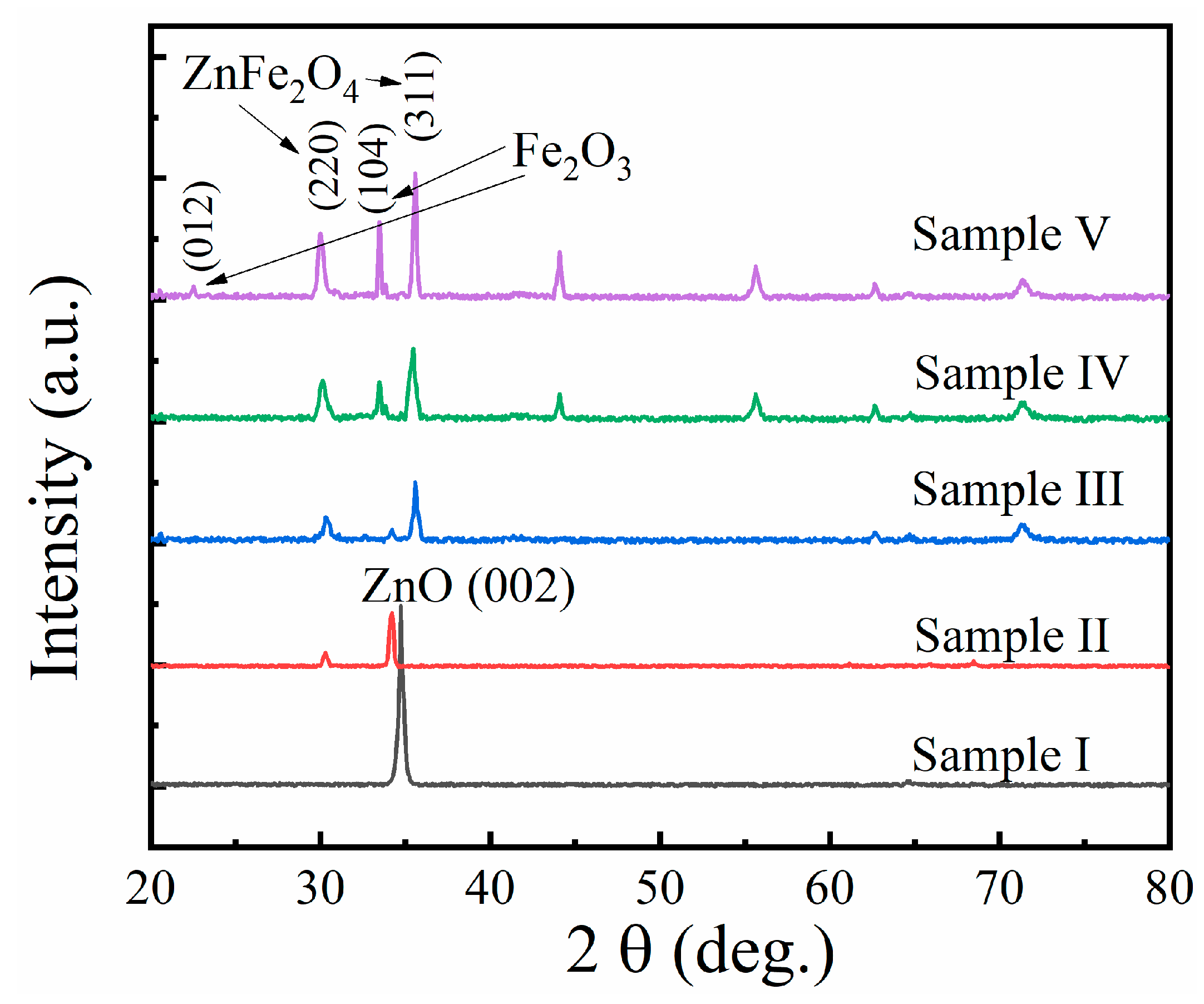
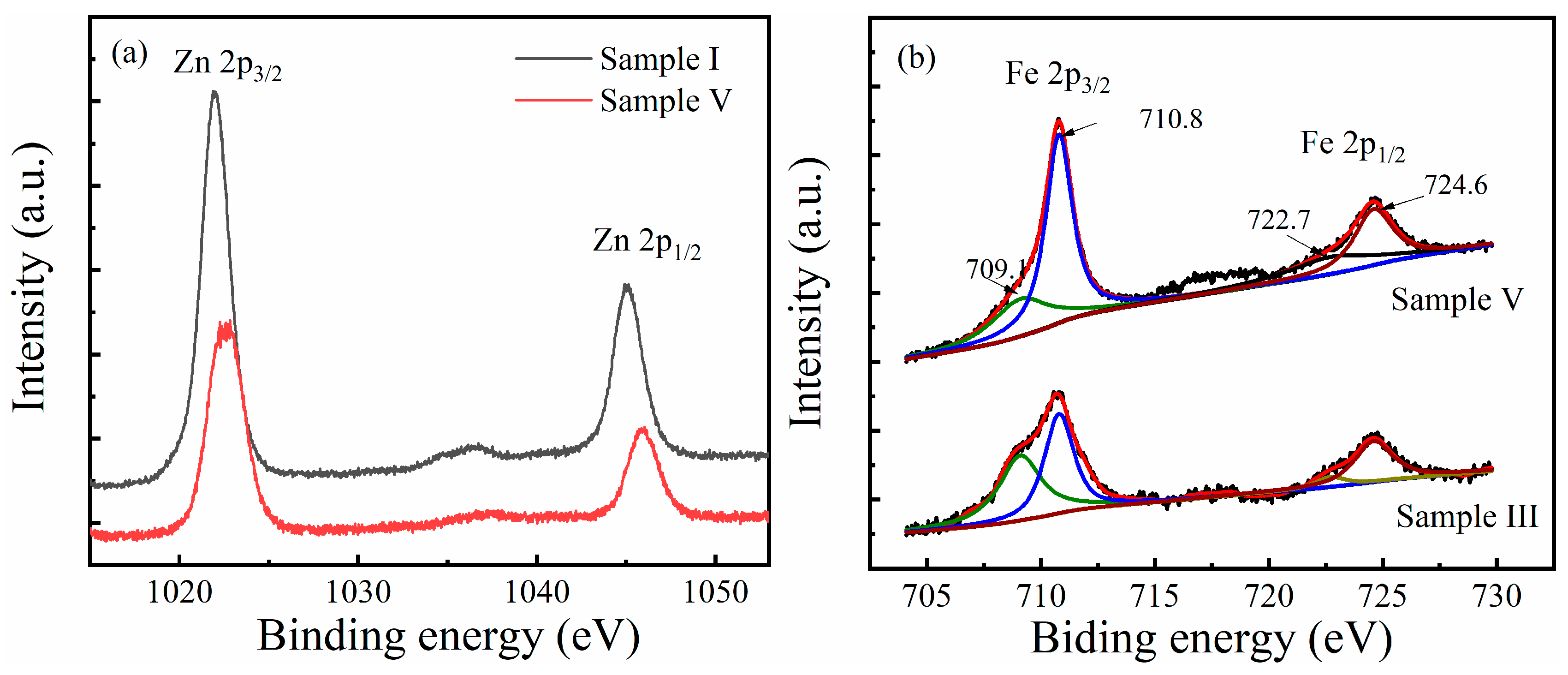
3.1. Characterization of ZnxFeyO Sensitive Films
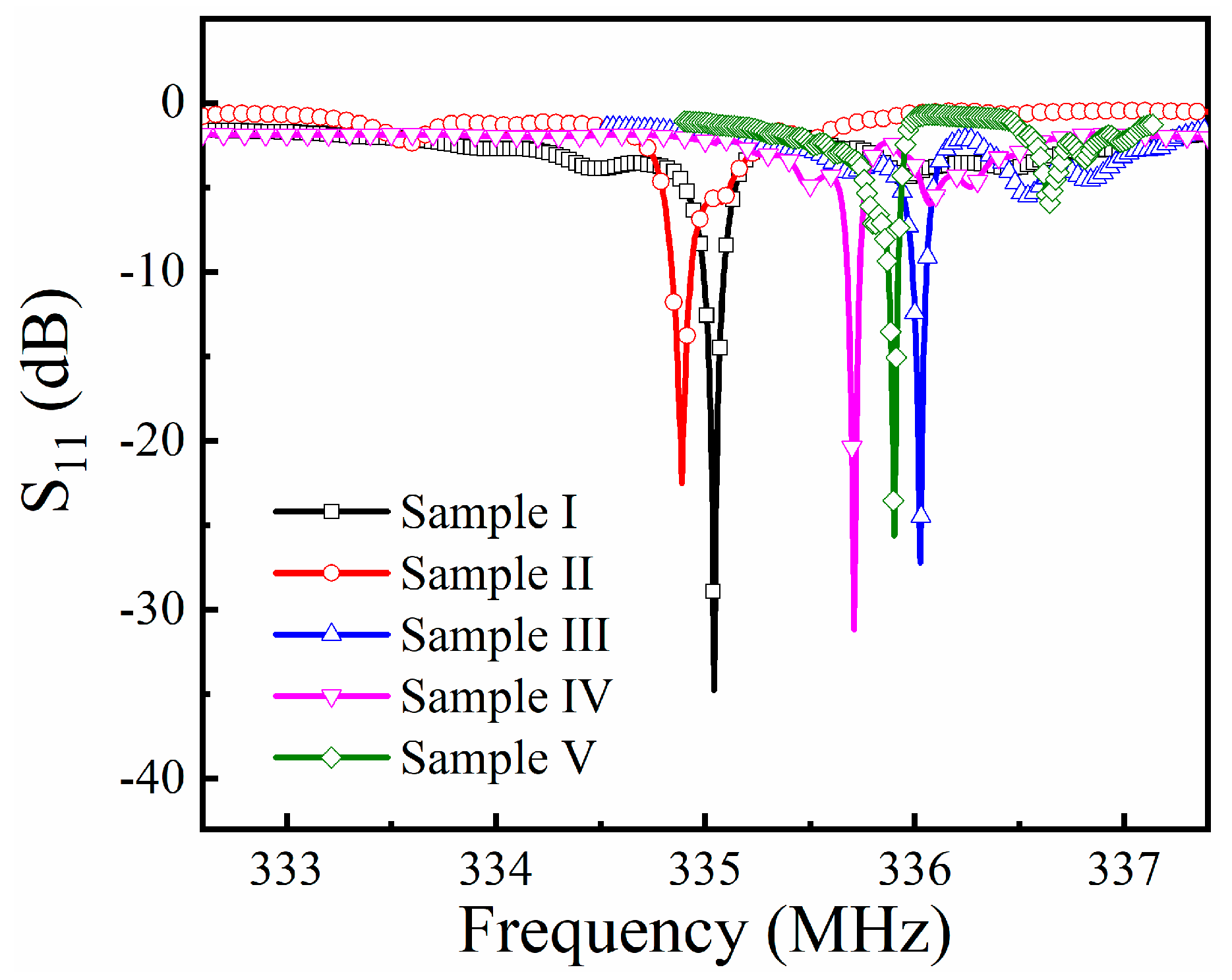
3.2. Characterization of SAW Resonators
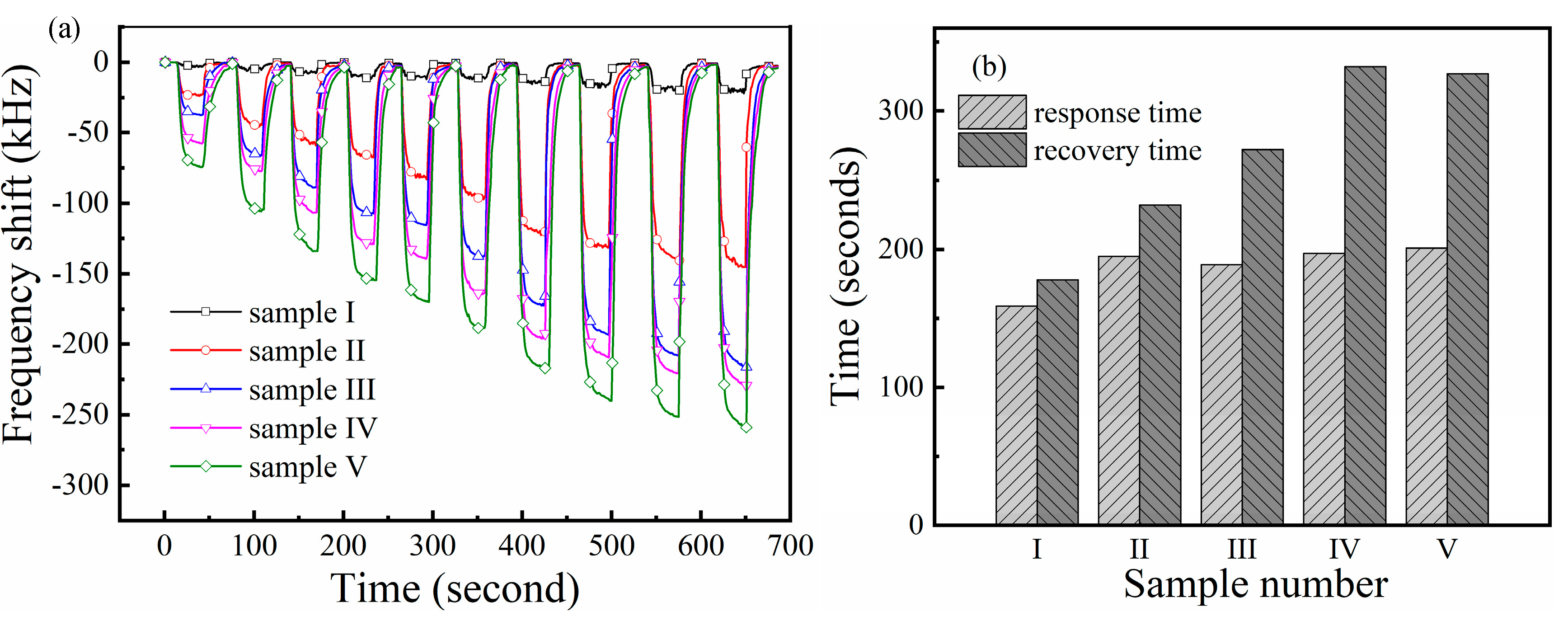
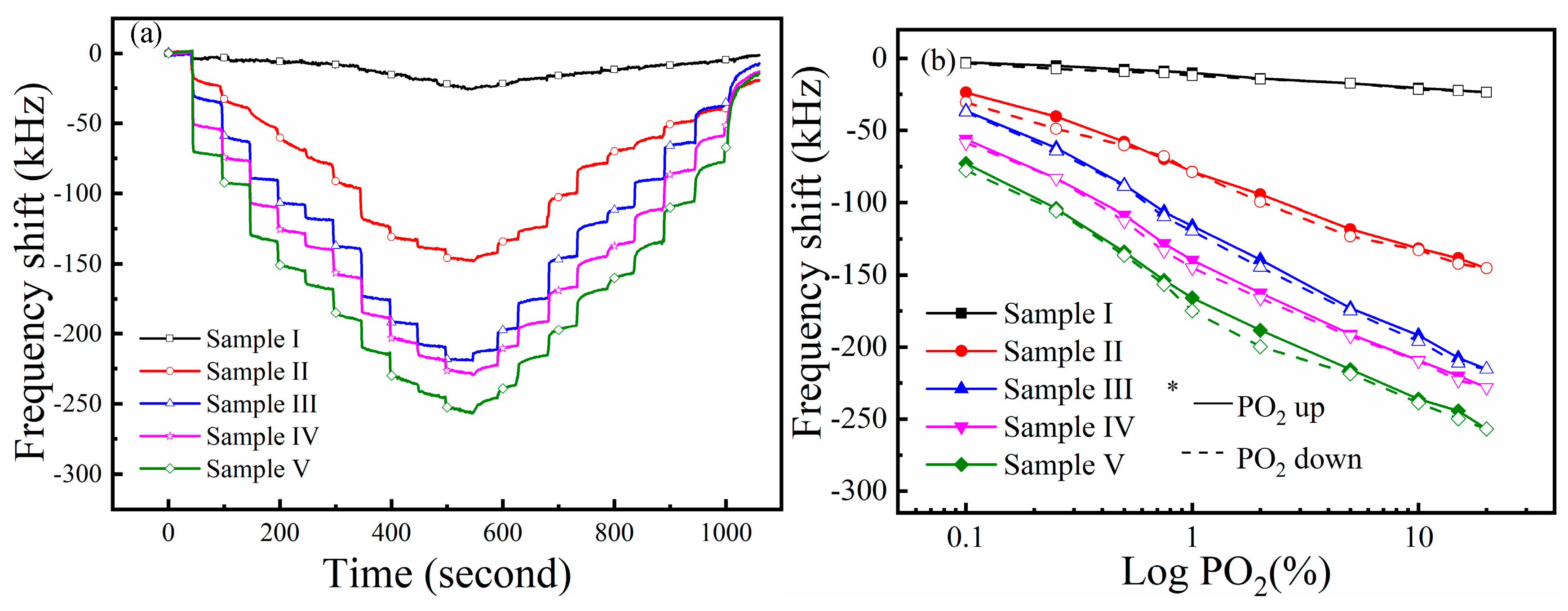
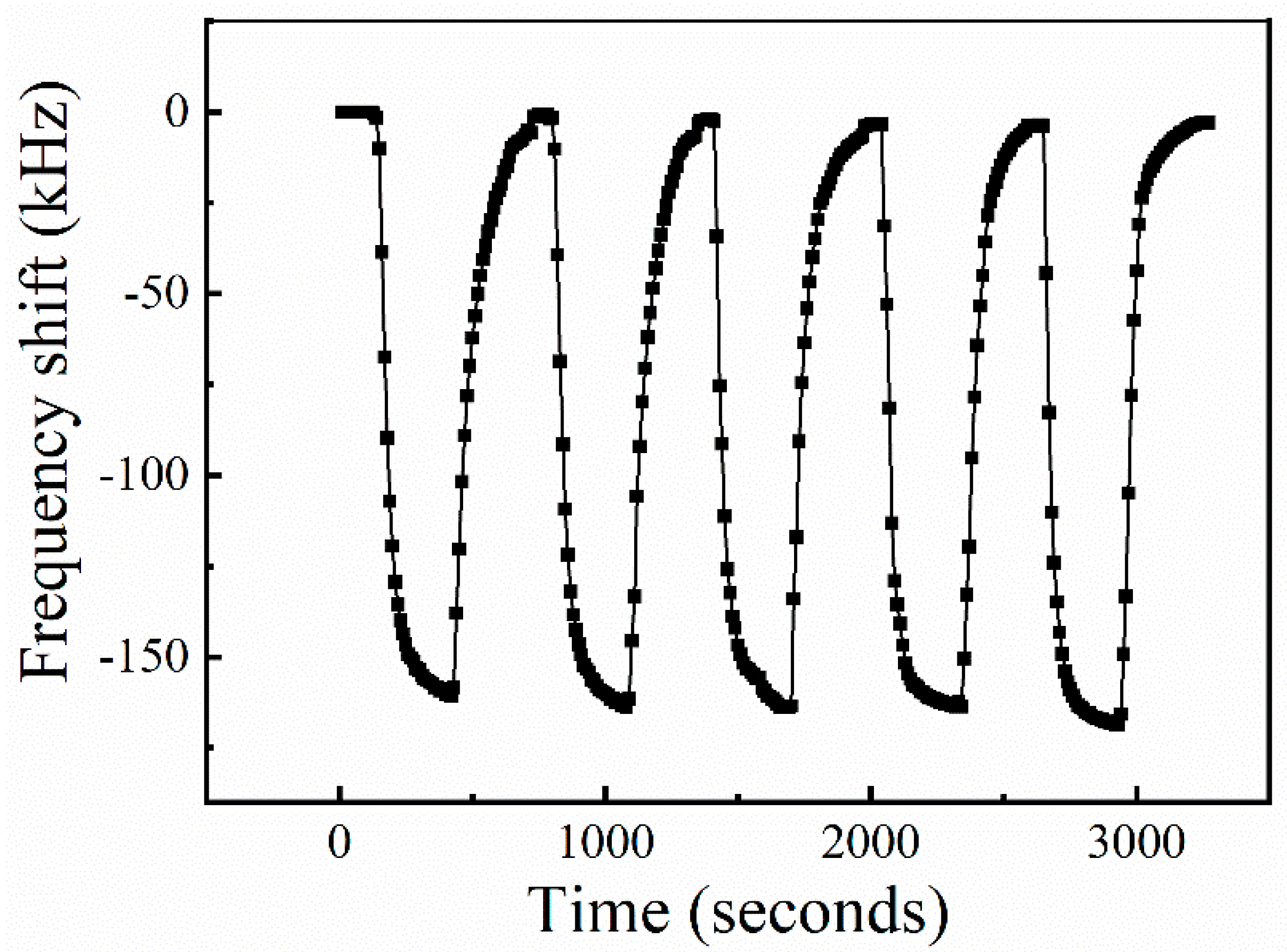
3.3. Gas Sensing Performance of the Sensors
3.4. Sensing Mechanism of ZnxFeyO Sensitive Films on O2 Gas
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhu, R.; Desroches, M.; Yoon, B.; Swager, T.M. Wireless oxygen sensors enabled by Fe (II)-polymer wrapped carbon nanotubes. ACS Sens. 2017, 2, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, J.; Chen, L.; Yao, Y.; Sun, Q.; Zhang, Q. Integrated microoxygen sensor based on nanostructured TiO2 thin films. Micro Nano Lett. 2015, 10, 597–602. [Google Scholar] [CrossRef]
- Chaabouni, F.; Abaab, M.; Rezig, B. Metrological characteristics of ZnO oxygen sensor at room temperature. Sens. Actuators B Chem. 2004, 100, 200–204. [Google Scholar] [CrossRef]
- Liu, B.; Chen, X.; Cai, H.; Mohammad, M.A.; Tian, X.; Tao, L.; Yang, Y.; Ren, T. Surface acoustic wave devices for sensor applications. J. Semicond. 2016, 37, 021001. [Google Scholar] [CrossRef]
- Bhati, V.S.; Ranwa, S.; Fanetti, M.; Valant, M.; Kumar, M. Efficient hydrogen sensor based on Ni-doped ZnO nanostructures by RF sputtering. Sens. Actuators B Chem. 2018, 255, 588–597. [Google Scholar] [CrossRef]
- Phan, T.L.; Zhang, Y.D.; Yang, D.S.; Nghia, N.X.; Thanh, T.D.; Yu, S.C. Defect-induced ferromagnetism in ZnO nanoparticles prepared by mechanical milling. Appl. Phys. Lett. 2013, 102, 072408. [Google Scholar] [CrossRef]
- Yu, L.; Guo, F.; Liu, S.; Yang, B.; Jiang, Y.; Qi, L.; Fan, X. Both oxygen vacancies defects and porosity facilitated NO2 gas sensing response in 2D ZnO nanowalls at room temperature. J. Alloy. Compd. 2016, 682, 352–356. [Google Scholar] [CrossRef]
- Kumar, R.; Al-Dossary, O.; Kumar, G.; Umar, A. Zinc oxide nanostructures for NO 2 gas–sensor applications: a review. Nano-Micro Lett. 2015, 7, 97–120. [Google Scholar] [CrossRef]
- Venkatesh, P.S.; Dharmaraj, P.; Purushothaman, V.; Ramakrishnan, V.; Jeganathan, K. Point defects assisted NH3 gas sensing properties in ZnO nanostructures. Sens. Actuators B Chem. 2015, 212, 10–17. [Google Scholar] [CrossRef]
- Marcu, A.; Viespe, C. Surface acoustic wave sensors for Hydrogen and Deuterium detection. Sensors 2017, 17, 1417. [Google Scholar] [CrossRef]
- Motaung, D.E.; Kortidis, I.; Mhlongo, G.H.; Duvenhage, M.M.; Swart, H.C.; Kiriakidis, G.; Ray, S.S. Correlating the magnetism and gas sensing properties of Mn-doped ZnO films enhanced by UV irradiation. RSC Adv. 2016, 6, 26227–26238. [Google Scholar] [CrossRef]
- Al-Hardan, N.H.; Abdullah, M.J.; Aziz, A.A. Performance of Cr-doped ZnO for acetone sensing. Appl. Surf. Sci. 2013, 270, 480–485. [Google Scholar] [CrossRef]
- Miki-Yoshida, M.; Morales, J.; Solis, J. Influence of Al, In, Cu, Fe and Sn dopants on the response of thin film ZnO gas sensor to ethanol vapour. Thin Solid Film. 2000, 373, 137–140. [Google Scholar]
- Si, S.; Li, C.; Wang, X.; Peng, Q.; Li, Y. Fe2O3/ZnO core–shell nanorods for gas sensors. Sens. Actuators B Chem. 2006, 119, 52–56. [Google Scholar] [CrossRef]
- Lu, Y.; Feng, Y.; Wang, F.; Zou, X.; Chen, Z.; Chen, P.; Liu, H.; Su, Y.; Zhang, Q.; Liu, G. Facile hydrothermal synthesis of carbon dots (CDs) doped ZnFe2O4/TiO2 hybrid materials with high photocatalytic activity. J. Photochem. Photobiol. A Chem. 2018, 353, 10–18. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, J.; Chen, F. Preparation and characterization of magnetic TiO 2/ZnFe 2 O 4 photocatalysts by a sol–gel method. Res. Chem. Intermed. 2008, 34, 375–380. [Google Scholar] [CrossRef]
- Wang, C.; Tan, X.; Yan, J.; Chai, B.; Li, J.; Chen, S. Electrospinning direct synthesis of magnetic ZnFe2O4/ZnO multi-porous nanotubes with enhanced photocatalytic activity. Appl. Surf. Sci. 2017, 396, 780–790. [Google Scholar] [CrossRef]
- Driskell, J.D.; Shanmukh, S.; Liu, Y.; Chaney, S.B.; Tang, X.J.; Zhao, Y.P.; Dluhy, R.A. The use of aligned silver nanorod arrays prepared by oblique angle deposition as surface enhanced Raman scattering substrates. J. Phys. Chem. C. 2008, 112, 895–901. [Google Scholar] [CrossRef]
- Ikehara, T.; Lu, J.; Konno, M.; Maeda, R.; Mihara, T. A high quality-factor silicon cantilever for a low detection-limit resonant mass sensor operated in air. J. MicroMech. MicroEng. 2007, 17, 2491. [Google Scholar] [CrossRef]
- Wang, Y.L.; Li, Y.H.; Wang, X.L.; et al. Effects of redox mediators on α-Fe2O3 exposed by {012} and {104} facets for photocatalytic water oxidation. Appl. Catal. B Environ. 2017, 206, 216–220. [Google Scholar] [CrossRef]
- Hashimoto, K.; Hashimoto, K.Y. Surface Acoustic Wave Devices in Telecommunications; Springer: Berlin, Germany, 2000. [Google Scholar]
- Atashbar, M.Z.; Sun, H.T.; Gong, B.; Wlodarski, W.; Lamb, R. XPS study of Nb-doped oxygen sensing TiO2 thin films prepared by sol-gel method. Thin Solid Film. 1998, 326, 238–244. [Google Scholar] [CrossRef]
- Hsieh, P.T.; Chen, Y.; Kao, K.; Wang, C. Luminescence mechanism of ZnO thin film investigated by XPS measurement. Appl. Phys. A 2008, 90, 317–321. [Google Scholar] [CrossRef]
- Bai, S.; Guo, T.; Zhao, Y.; Sun, J.; Li, D.; Chen, A.; Liu, C.C. Sensing performance and mechanism of Fe-doped ZnO microflowers. Sens. Actuators B Chem. 2014, 195, 657–666. [Google Scholar] [CrossRef]
- Fang, L.; Zu, X.; Li, Z.; Zhu, S.; Liu, C.; Zhou, W.; Wang, L. Synthesis and characteristics of Fe3+-doped SnO2 nanoparticles via sol–gel-calcination or sol–gel-hydrothermal route. J. Alloy. Compd. 2008, 454, 261–267. [Google Scholar] [CrossRef]

| Parameters | Value |
|---|---|
| Acoustic wavelength (μm) | 1.5–3.0 |
| Strip width (μm) | 2 |
| Aperture (μm) | 800 |
| Strip numbers in IDT | 101 |
| Strip numbers in reflectors | 400 |
| Thickness of electrodes | 100 nm/10 nm (Au/Ti) |
| Substrate material | LGS (La3Ga5SiO14) |
| Sample Number | Zn | Fe | ||||
|---|---|---|---|---|---|---|
| Sputtering Method | Power (W) | Composition (at%) | Sputtering Method | Current (A) | Composition (at%) | |
| I | RF | 100 | 100 | DC | 0 | 0 |
| II | 96.5 | 0.02 | 3.5 | |||
| III | 41 | 0.05 | 59 | |||
| IV | 29.9 | 0.08 | 70.1 | |||
| V | 24.5 | 0.1 | 75.5 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shu, L.; Jiang, T.; Xia, Y.; Wang, X.; Yan, D.; Wu, W. The Investigation of a SAW Oxygen Gas Sensor Operated at Room Temperature, Based on Nanostructured ZnxFeyO Films. Sensors 2019, 19, 3025. https://doi.org/10.3390/s19133025
Shu L, Jiang T, Xia Y, Wang X, Yan D, Wu W. The Investigation of a SAW Oxygen Gas Sensor Operated at Room Temperature, Based on Nanostructured ZnxFeyO Films. Sensors. 2019; 19(13):3025. https://doi.org/10.3390/s19133025
Chicago/Turabian StyleShu, Lin, Tao Jiang, Yudong Xia, Xuemin Wang, Dawei Yan, and Weidong Wu. 2019. "The Investigation of a SAW Oxygen Gas Sensor Operated at Room Temperature, Based on Nanostructured ZnxFeyO Films" Sensors 19, no. 13: 3025. https://doi.org/10.3390/s19133025
APA StyleShu, L., Jiang, T., Xia, Y., Wang, X., Yan, D., & Wu, W. (2019). The Investigation of a SAW Oxygen Gas Sensor Operated at Room Temperature, Based on Nanostructured ZnxFeyO Films. Sensors, 19(13), 3025. https://doi.org/10.3390/s19133025





