Application of the Enzymatic Electrochemical Biosensors for Monitoring Non-Competitive Inhibition of Enzyme Activity by Heavy Metals
Abstract
1. Introduction
2. Results and Discussion
2.1. Amperometric Transducer Design
2.2. Effect of Redox Mediator
2.3. Characterisation of Biosensor Surface
2.4. Effect of Stirring Rate
2.5. Amount of Enzyme Incorporated in Polymer
2.6. Effect of the Applied Potential
2.7. Analytical Performance of Proposed GOx Biosensor
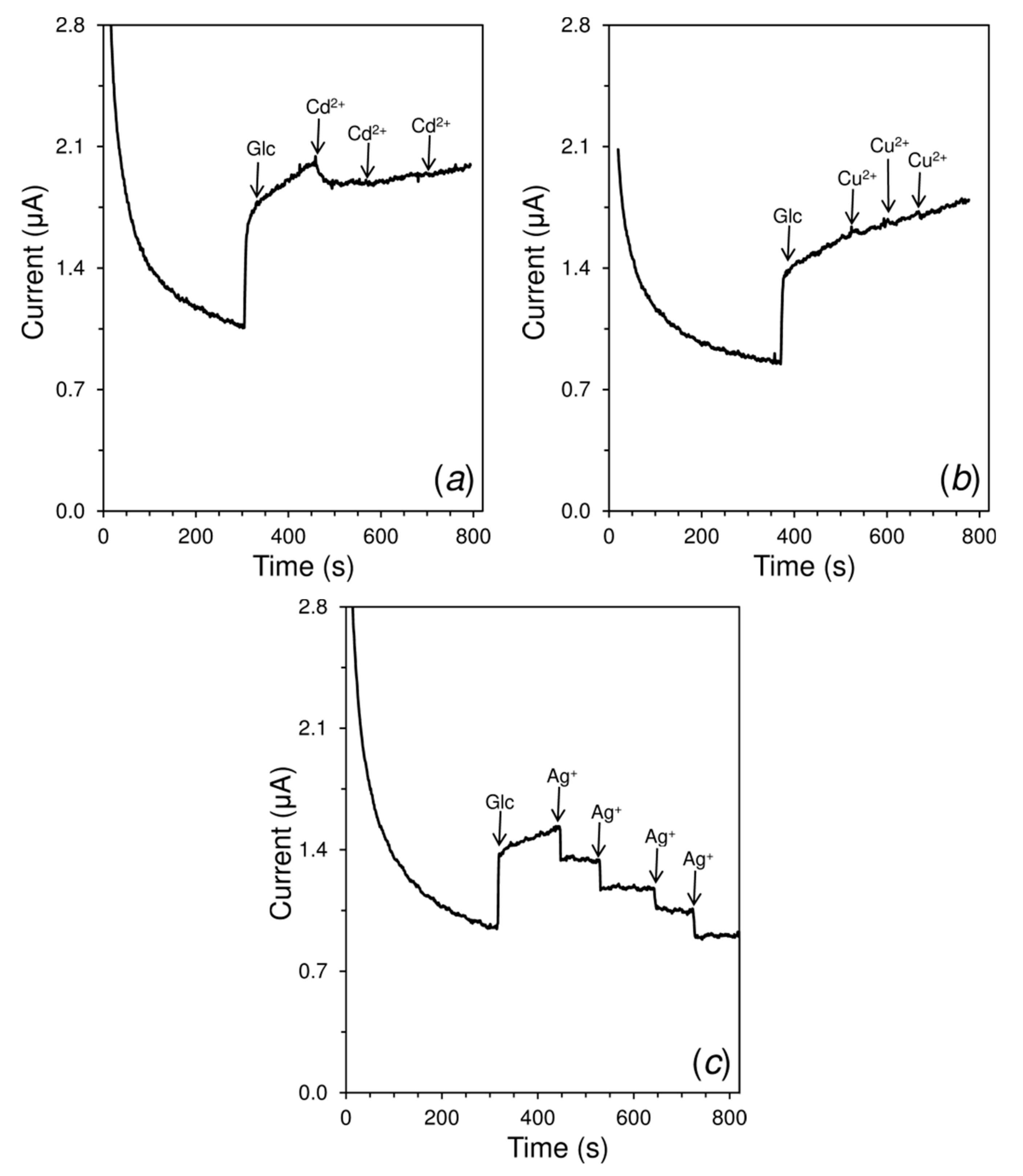
2.8. Study of the Inhibitory Effects of Heavy Metals
3. Materials and Methods
3.1. Chemicals
3.2. Apparatus
3.3. Preparation of Working Electrodes
3.4. Procedure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mäntsälä, P.; Niemi, J. Enzymes: The biological catalysts of life. Physiol. Maintanance 2009, 2, 1–22. [Google Scholar]
- Woodward, J.D.; Trompetter, I.; Sewell, B.T.; Piotrowski, M. Substrate specificity of plant nitrilase complexes is affected by their helical twist. Commun. Biol. 2018, 1, 186. [Google Scholar] [CrossRef] [PubMed]
- Schowen, R.L. How an enzyme surmounts the activation energy barrier. Proc. Natl. Acad. Sci. USA 2003, 100, 11931–11932. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.K. Enzymes: Principles and biotechnological applications. Essays Biochem. 2015, 59, 1–41. [Google Scholar] [CrossRef] [PubMed]
- Aledo, J.C.; Lobo, C.; del Valle, A.E. Energy diagrams for enzyme-catalyzed reactions: Concepts and misconcepts. Biochem. Mol. Biol. Educ. 2003, 31, 234–236. [Google Scholar] [CrossRef]
- Pearce, L.L.; Bominaar, E.L.; Hill, B.C.; Peterson, J. Reversal of cyanide inhibition of cytochrome c oxidase by the auxiliary substrate nitric oxide an endogenous antidote to cyanide poisoning? J. Biol. Chem. 2003, 278, 52139–52145. [Google Scholar] [CrossRef] [PubMed]
- Mizrahi, L.; Achituv, Y. Effect of heavy metals ions on enzyme activity in the mediterranean mussel, donax trunculus. Bull. Environ. Contam. Toxicol. 1989, 42, 854–859. [Google Scholar] [CrossRef]
- Viarengo, A. Biochemical effects of trace metals. Mar. Pollut. Bull. 1985, 16, 153–158. [Google Scholar] [CrossRef]
- Hodson, P.V. The effect of metal metabolism on uptake, disposition and toxicity in fish. Aquat. Toxicol. 1988, 11, 3–18. [Google Scholar] [CrossRef]
- Martin, T.; Holdich, D. The acute lethal toxicity of heavy metals to peracarid crustaceans (with particular reference to fresh-water asellids and gammarids). Water Res. 1986, 20, 1137–1147. [Google Scholar] [CrossRef]
- Lu, H.P.; Xun, L.; Xie, X.S. Single-molecule enzymatic dynamics. Science 1998, 282, 1877–1882. [Google Scholar] [CrossRef] [PubMed]
- Blackmond, D.G. Reaction progress kinetic analysis: A powerful methodology for mechanistic studies of complex catalytic reactions. Angew. Chem. Int. Ed. 2005, 44, 4302–4320. [Google Scholar] [CrossRef] [PubMed]
- Kirk, O.; Borchert, T.V.; Fuglsang, C.C. Industrial enzyme applications. Curr. Opin. Biotechnol. 2002, 13, 345–351. [Google Scholar] [CrossRef]
- Hasan, F.; Shah, A.A.; Hameed, A. Industrial applications of microbial lipases. Enzym. Microb. Technol. 2006, 39, 235–251. [Google Scholar] [CrossRef]
- Vellard, M. The enzyme as drug: Application of enzymes as pharmaceuticals. Curr. Opin. Biotechnol. 2003, 14, 444–450. [Google Scholar] [CrossRef]
- Bothner, B.; Chavez, R.; Wei, J.; Strupp, C.; Phung, Q.; Schneemann, A.; Siuzdak, G. Monitoring enzyme catalysis with mass spectrometry. J. Biol. Chem. 2000, 275, 13455–13459. [Google Scholar] [CrossRef]
- Cheng, S.; Wu, Q.; Xiao, H.; Chen, H. Online monitoring of enzymatic reactions using time-resolved desorption electrospray ionization mass spectrometry. Anal. Chem. 2017, 89, 2338–2344. [Google Scholar] [CrossRef]
- Xu, Z.; Yao, S.; Wei, Y.; Zhou, J.; Zhang, L.; Wang, C.; Guo, Y. Monitoring enzyme reaction and screening of inhibitors of acetylcholinesterase by quantitative matrix-assisted laser desorption/ionization fourier transform mass spectrometry. J. Am. Soc. Mass Spectrom. 2008, 19, 1849–1855. [Google Scholar] [CrossRef]
- Westley, C.; Fisk, H.; Xu, Y.; Hollywood, K.A.; Carnell, A.J.; Micklefield, J.; Turner, N.J.; Goodacre, R. Real-time monitoring of enzyme-catalysed reactions using deep UV resonance raman spectroscopy. Chem. A Eur. J. 2017, 23, 6983–6987. [Google Scholar] [CrossRef]
- Miller, R.B.; Karn, R.C. A rapid spectrophotometric method for the determination of esterase activity. J. Biochem. Biophys. Methods 1980, 3, 345–354. [Google Scholar] [CrossRef]
- German, N.; Voronovic, J.; Ramanavicius, A.; Ramanaviciene, A. Gold nanoparticles and polypyrrole for glucose biosensor design. Procedia Eng. 2012, 47, 482–485. [Google Scholar] [CrossRef]
- Kim, J.-H.; Lee, D.; Bae, T.-S.; Lee, Y.-S. The electrochemical enzymatic glucose biosensor based on mesoporous carbon fibers activated by potassium carbonate. J. Ind. Eng. Chem. 2015, 25, 192–198. [Google Scholar] [CrossRef]
- Kotzian, P.; Brázdilová, P.; Kalcher, K.; Vytřas, K. Determination of hydrogen peroxide, glucose and hypoxanthine using (bio) sensors based on ruthenium dioxide-modified screen-printed electrodes. Anal. Lett. 2005, 38, 1099–1113. [Google Scholar] [CrossRef]
- Kotzian, P.; Brázdilová, P.; Řezková, S.; Kalcher, K.; Vytřas, K. Amperometric glucose biosensor based on rhodium dioxide-modified carbon ink. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 2006, 18, 1499–1504. [Google Scholar] [CrossRef]
- Wei, A.; Sun, X.W.; Wang, J.; Lei, Y.; Cai, X.; Li, C.M.; Dong, Z.; Huang, W. Enzymatic glucose biosensor based on ZnO nanorod array grown by hydrothermal decomposition. Appl. Phys. Lett. 2006, 89, 123902. [Google Scholar] [CrossRef]
- Weng, W.-H.; Wang, C.-W.; Pang, S.-T.; Pan, T.-M. Enzymatic glucose biosensor based on TbYxOy electrolyte-insulator-semiconductor. J. Electrochem. Soc. 2016, 163, B445–B452. [Google Scholar] [CrossRef]
- Ashrafi, A.M.; Koudelkova, Z.; Sedlackova, E.; Richtera, L.; Adam, V. Electrochemical sensors and biosensors for determination of mercury ions. J. Electrochem. Soc. 2018, 165, B824–B834. [Google Scholar] [CrossRef]
- Wang, J. Stripping analysis at bismuth electrodes: A review. Electroanal. Int. J. Devot. Fundam. Pract. Asp. Electroanal. 2005, 17, 1341–1346. [Google Scholar] [CrossRef]
- Liang, M.; Jin, F.; Liu, R.; Su, R.; Qi, W.; Yu, Y.; Wang, L.; He, Z. Enhanced electrochemical detection performance of multiwall carbon nanotubes functionalized by aspartame. J. Mater. Sci. 2013, 48, 5624–5632. [Google Scholar] [CrossRef]
- Scheller, F.W.; Schubert, F.; Neumann, B.; Pfeiffer, D.; Hintsche, R.; Dransfeld, I.; Wollenberger, U.; Renneberg, R.; Warsinke, A.; Johansson, G. Second generation biosensors. Biosens. Bioelectron. 1991, 6, 245–253. [Google Scholar] [CrossRef]
- Peigney, A.; Laurent, C.; Flahaut, E.; Bacsa, R.; Rousset, A. Specific surface area of carbon nanotubes and bundles of carbon nanotubes. Carbon 2001, 39, 507–514. [Google Scholar] [CrossRef]
- Sýs, M.; Žabčíková, S.; Červenka, L.; Vytřas, K. Comparison of adsorptive with extractive stripping voltammetry in electrochemical determination of retinol. Potr. SJF Sci. 2017, 11, 96–105. [Google Scholar] [CrossRef]
- Nguyen, H.; Park, J.; Kang, S.; Kim, M. Surface plasmon resonance: A versatile technique for biosensor applications. Sensors 2015, 15, 10481–10510. [Google Scholar] [CrossRef] [PubMed]
- Wolfschmidt, H.; Baier, C.; Gsell, S.; Fischer, M.; Schreck, M.; Stimming, U. STM, SECPM, AFM and electrochemistry on single crystalline surfaces. Materials 2010, 3, 4196–4213. [Google Scholar] [CrossRef] [PubMed]
- Vang, R.T.; Lauritsen, J.V.; Laegsgaard, E.; Besenbacher, F. Scanning tunneling microscopy as a tool to study catalytically relevant model systems. Chem. Soc. Rev. 2008, 37, 2191–2203. [Google Scholar] [CrossRef]
- Bard, A.J.; Fan, F.R.F.; Kwak, J.; Lev, O. Scanning electrochemical microscopy. Introduction and principles. Anal. Chem. 1989, 61, 132–138. [Google Scholar] [CrossRef]
- Vahabi, S.; Salman, B.N.; Javanmard, A. Atomic force microscopy application in biological research: A review study. Iran. J. Med Sci. 2013, 38, 76. [Google Scholar]
- Sýs, M.; Pekec, B.; Kalcher, K.; Vytřas, K. Amperometric enzyme carbon paste-based biosensor for quantification of hydroquinone and polyphenolic antioxidant capacity. Int. J. Electrochem. Sci. 2013, 8, 9030–9040. [Google Scholar]
- Pan, B.; Xing, B. Adsorption mechanisms of organic chemicals on carbon nanotubes. Environ. Sci. Technol. 2008, 42, 9005–9013. [Google Scholar] [CrossRef]
- Rasouli, H.; Naji, L.; Hosseini, M.G. Electrochemical and electromechanical behavior of Nafion-based soft actuators with PPy/CB/MWCNT nanocomposite electrodes. RSC Adv. 2017, 7, 3190–3203. [Google Scholar] [CrossRef]
- Bhalla, N.; Jolly, P.; Formisano, N.; Estrela, P. Introduction to biosensors. Essays Biochem. 2016, 60, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Anojčić, J.; Guzsvány, V.; Vajdle, O.; Madarász, D.; Rónavári, A.; Kónya, Z.; Kalcher, K. Hydrodynamic chronoamperometric determination of hydrogen peroxide using carbon paste electrodes coated by multiwalled carbon nanotubes decorated with MnO2 or Pt particles. Sens. Actuators B: Chem. 2016, 233, 83–92. [Google Scholar] [CrossRef]
- Cueni-Villoz, N.; Devigili, A.; Delodder, F.; Cianferoni, S.; Feihl, F.; Rossetti, A.O.; Eggimann, P.; Vincent, J.-L.; Taccone, F.S.; Oddo, M. Increased blood glucose variability during therapeutic hypothermia and outcome after cardiac arrest. Crit. Care Med. 2011, 39, 2225–2231. [Google Scholar] [CrossRef] [PubMed]
- Blanco, A.; Blanco, G. Chapter 8—Enzymes. In Medical Biochemistry; Blanco, A., Blanco, G., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 153–175. [Google Scholar]
- Ghica, M.E.; Brett, C.M. Glucose oxidase inhibition in poly (neutral red) mediated enzyme biosensors for heavy metal determination. Microchim. Acta 2008, 163, 185–193. [Google Scholar] [CrossRef]
- Malitesta, C.; Guascito, M. Heavy metal determination by biosensors based on enzyme immobilised by electropolymerisation. Biosens. Bioelectron. 2005, 20, 1643–1647. [Google Scholar] [CrossRef] [PubMed]
- Mugheri, A.Q.; Tahira, A.; Sherazi, S.T.H.; Abro, M.I.; Willander, M.; Ibupoto, Z.H. An amperometric indirect determination of heavy metal ions through inhibition of glucose oxidase immobilized on cobalt oxide nanostructures. Sens. Lett. 2016, 14, 1178–1186. [Google Scholar] [CrossRef]
- Varjovi, M.J.; Sabzi, R.E.; Borghei, S.M. Determination of heavy metal ions by an amperometric biosensor based on glucose oxidase immobilized onto single-walled carbon nanotubes/Nile blue nanocomposite. J. Iran. Chem. Soc. 2018, 15, 1765–1774. [Google Scholar] [CrossRef]
- Ashrafi, A.M.; Cerovac, S.; Mudrić, S.; Guzsvány, V.; Husáková, L.; Urbanová, I.; Vytřas, K. Antimony nanoparticle-multiwalled carbon nanotubes composite immobilized at carbon paste electrode for determination of trace heavy metals. Sens. Actuators B: Chem. 2014, 191, 320–325. [Google Scholar] [CrossRef]
- Ashrafi, A.M.; Vytřas, K. Stripping voltammetric determination of mercury (II) at antimony-coated carbon paste electrode. Talanta 2011, 85, 2700–2702. [Google Scholar] [CrossRef]
- Ashrafi, A.M.; Vytřas, K. New procedures for voltammetric determination of copper (II) using antimony film-coated carbon paste electrodes. Electrochimica Acta 2012, 73, 112–117. [Google Scholar] [CrossRef]
- Ashrafi, A.M.; Vytřas, K. Determination of trace bismuth (III) by stripping voltammetry at antimony-coated carbon paste electrode. Int. J. Electrochem. Sci. 2012, 7, 68–76. [Google Scholar]
- Ashrafi, A.M.; Vytřas, K. Codeposited antimony-bismuth film carbon paste electrodes for electrochemical stripping determination of trace heavy metals. Int. J. Electrochem. Sci. 2013, 8, 2095–2103. [Google Scholar]
- Bonfil, Y.; Brand, M.; Kirowa-Eisner, E. Trace determination of mercury by anodic stripping voltammetry at the rotating gold electrode. Analytica Chimica Acta 2000, 424, 65–76. [Google Scholar] [CrossRef]
- Hocevar, S.B.; Švancara, I.; Ogorevc, B.; Vytřas, K. Antimony film electrode for electrochemical stripping analysis. Analytical chemistry 2007, 79, 8639–8643. [Google Scholar] [CrossRef] [PubMed]
- Perone, S. The Application of Stripping Analysis to the Determination of Silver (I) Using Graphite Electrodes. Anal. Chem. 1963, 35, 2091–2094. [Google Scholar] [CrossRef]
- Švancara, I.; Vytřas, K.; Hua, C.; Smyth, M.R. Voltammetric determination of mercury (II) at a carbon paste electrode in aqueous solutions containing tetraphenylborate ion. Talanta 1992, 39, 391–396. [Google Scholar] [CrossRef]
- Svobodová, E.; Baldrianová, L.; Hocevar, S.B.; Svancara, I. Electrochemical stripping analysis of selected heavy metals at antimony trioxide-modified carbon paste electrode. Int. J. Electrochem. Sci. 2012, 7, 197–210. [Google Scholar]
- Yang, Y.; Wang, Z.; Yang, M.; Guo, M.; Wu, Z.; Shen, G.; Yu, R. Inhibitive determination of mercury ion using a renewable urea biosensor based on self-assembled gold nanoparticles. Sens. Actuators B Chem. 2006, 114, 1–8. [Google Scholar] [CrossRef]
- D’Souza, S.F. Microbial biosensors. Biosens. Bioelectron. 2001, 16, 337–353. [Google Scholar] [CrossRef]
- Chey, C.; Ibupoto, Z.; Khun, K.; Nur, O.; Willander, M.J.S. Indirect determination of mercury ion by inhibition of a glucose biosensor based on ZnO nanorods. Sensors 2012, 12, 15063–15077. [Google Scholar] [CrossRef]
- Guascito, M.R.; Malitesta, C.; Mazzotta, E.; Turco, A. Inhibitive determination of metal ions by an amperometric glucose oxidase biosensor: Study of the effect of hydrogen peroxide decomposition. Sens. Actuators B Chem. 2008, 131, 394–402. [Google Scholar] [CrossRef]
- Samphao, A.; Rerkchai, H.; Jitcharoen, J.; Nacapricha, D.; Kalcher, K. Indirect determination of mercury by inhibition of glucose oxidase immobilized on a carbon paste electrode. Int. J. Electrochem. Sci. 2012, 7, 1001–1010. [Google Scholar]
- Jung, W.K.; Koo, H.C.; Kim, K.W.; Shin, S.; Kim, S.H.; Park, Y.H. Antibacterial activity and mechanism of action of the silver ion in Staphylococcus aureus and Escherichia coli. Appl. Environ. Microbiol. 2008, 74, 2171–2178. [Google Scholar] [CrossRef] [PubMed]
- Colowick, S.P.; Kaplan, N.O.; McCormick, D.B.; Wright, L.D. Methods in Enzymology; Academic Press: New York, NY, USA, 1955; Volume 1. [Google Scholar]
- Nečas, D.; Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Open Phys. 2012, 10, 181–188. [Google Scholar] [CrossRef]
- Bass, M.; Berman, A.; Singh, A.; Konovalov, O.; Freger, V. Surface structure of Nafion in vapor and liquid. J. Phys. Chem. B 2010, 114, 3784–3790. [Google Scholar] [CrossRef] [PubMed]
- Schachl, K.; Turkušić, E.; Komersová, A.; Bartoš, M.; Moderegger, H.; Švancara, I.; Alemu, H.; Vytřas, K.; Jimenez-Castro, M.; Kalcher, K. Amperometric determination of glucose with a carbon paste biosensor. Collect. Czechoslov. Chem. Commun. 2002, 67, 302–313. [Google Scholar] [CrossRef]
- Švancara, I.; Metelka, R.; Vytřas, K. Piston-driven carbon paste electrode holders for electrochemical measurements. Sens. Electroanal. 2005, 1, 7–18. [Google Scholar]
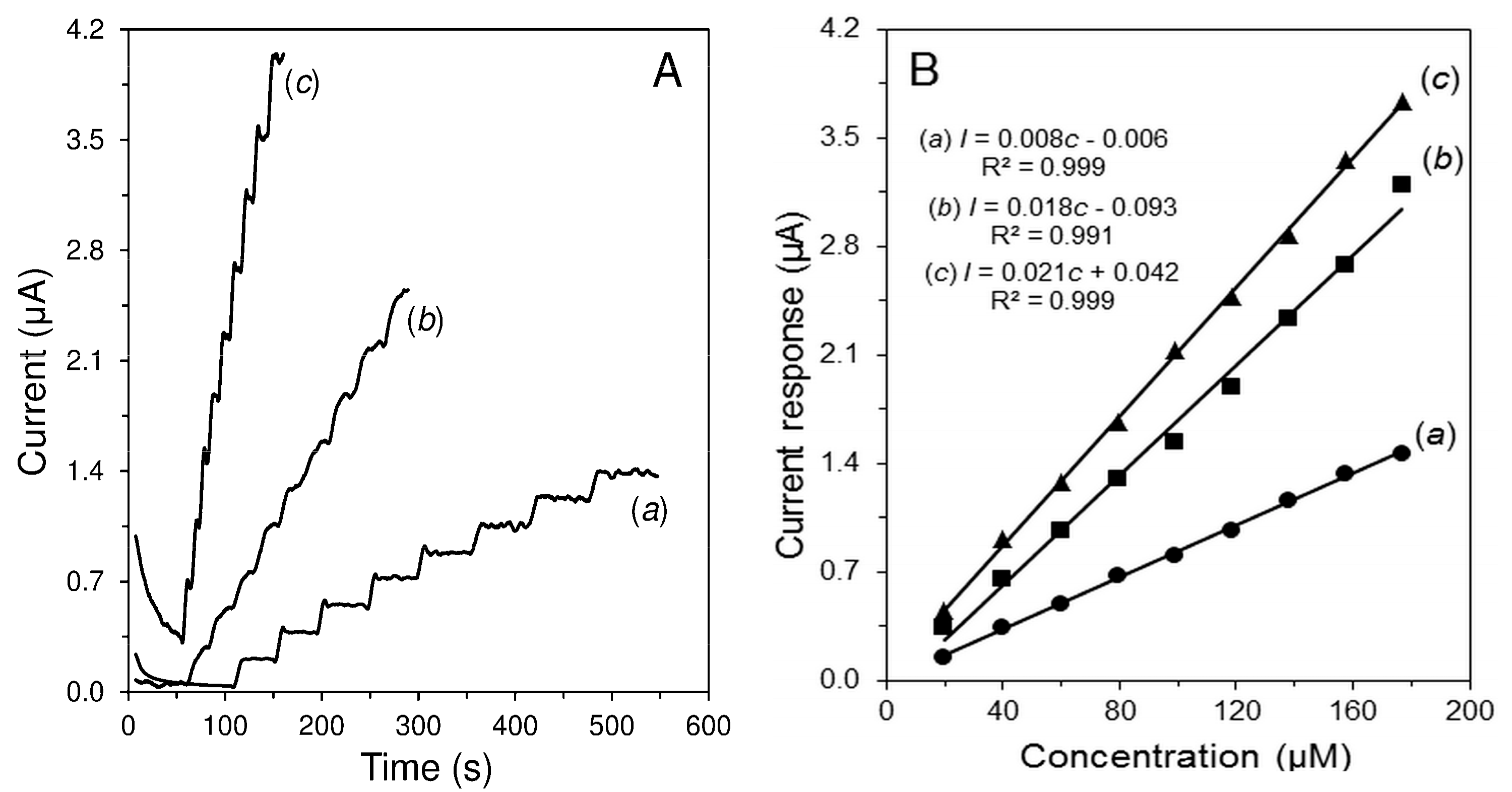
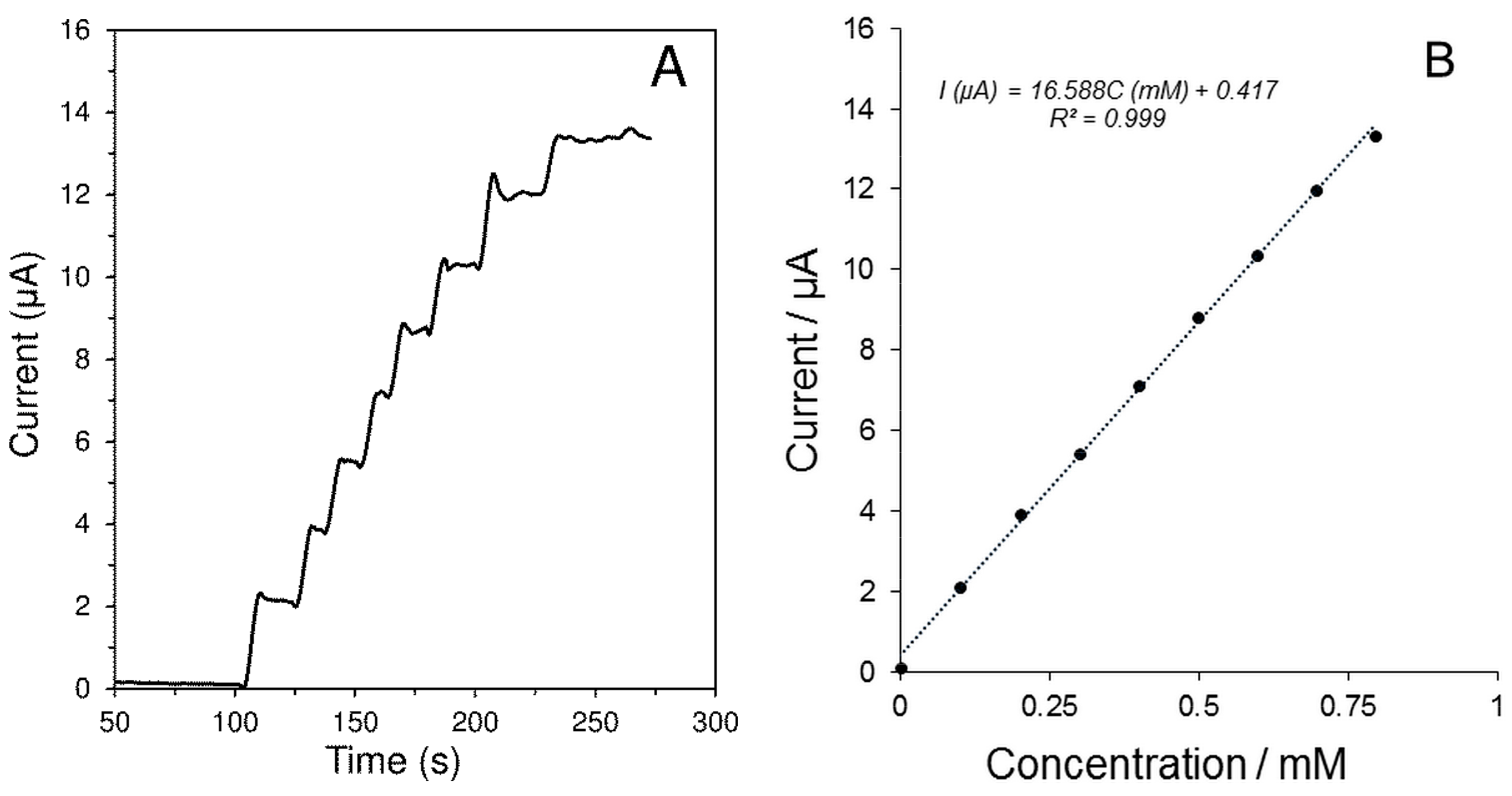


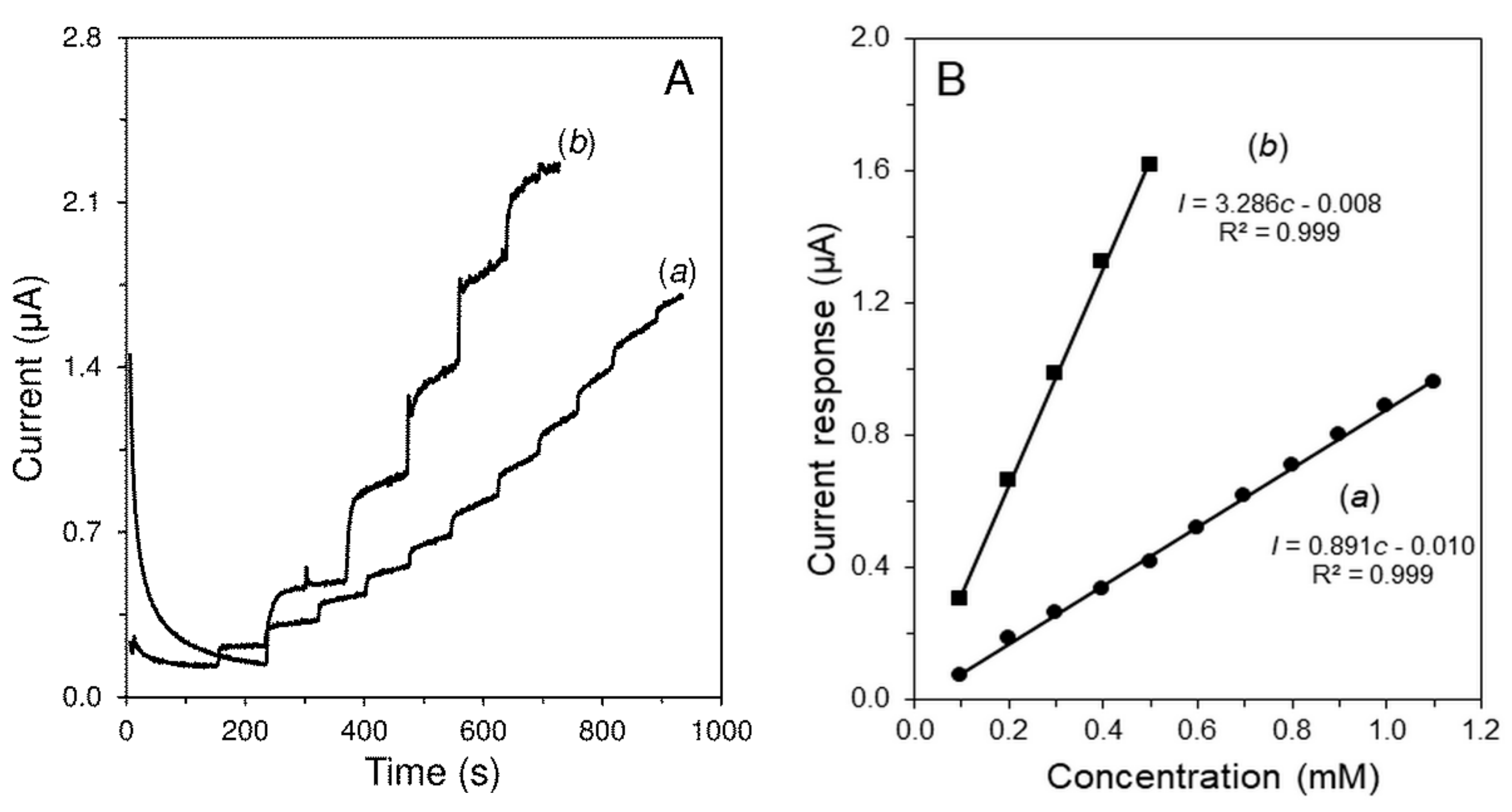
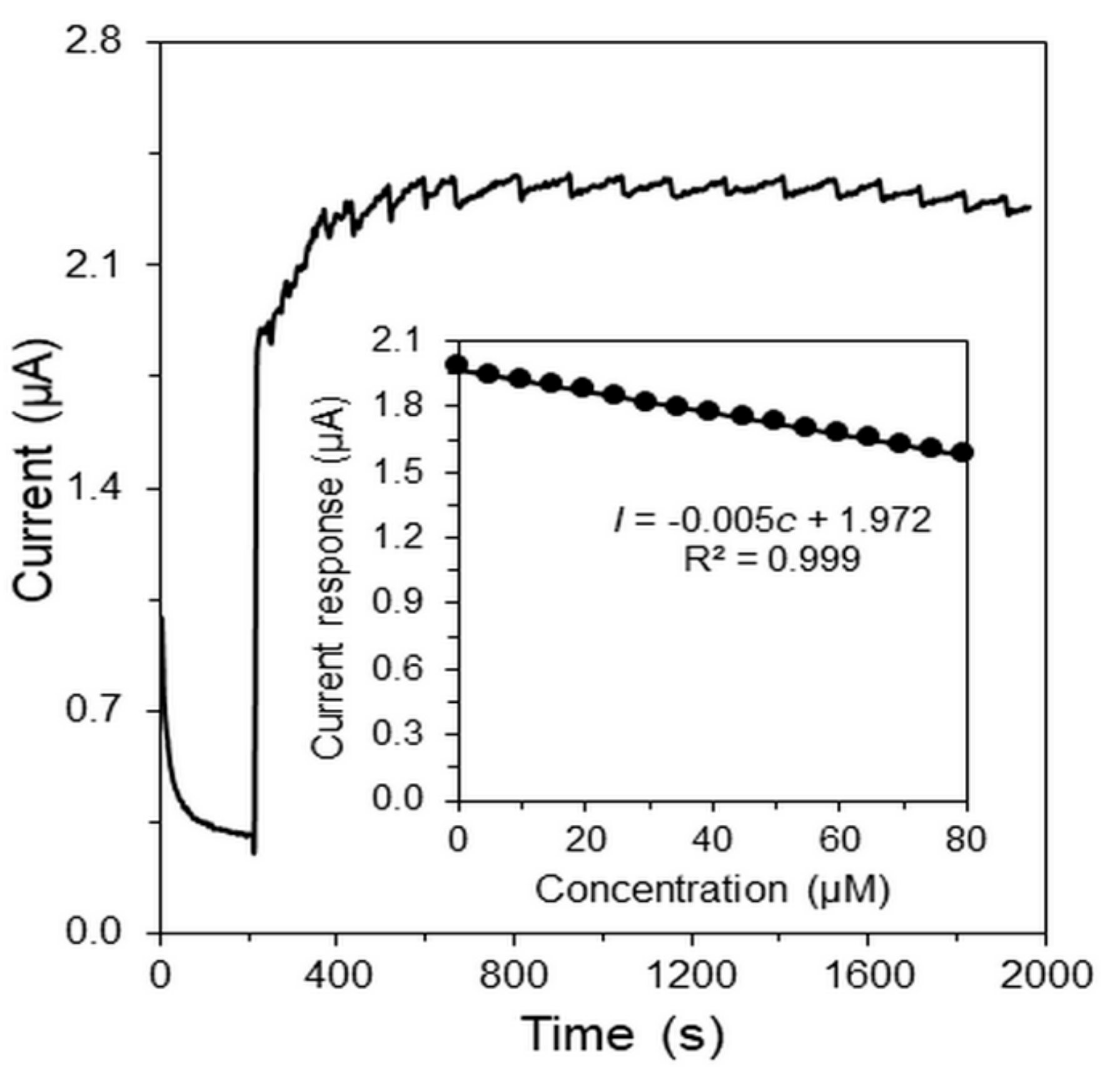
| Parameter | H2O2 | Glc |
|---|---|---|
| Peak potential (V) | +0.40 | +0.40 |
| Linearity range (µM) | 10–800 | 100–800 |
| Slope (µA∙µM−1) | 0.017 ± 0.005 | 0.003 ± 0.0002 |
| Correlation coefficient (r) | 0.999 ± 0.001 | 0.999 ± 0.001 |
| 1 LOD (µM) | 7.5 | 17.4 |
| LOQ (µM) | 25.0 | 52.7 |
| RSD% (n = 3) | 4.2 | 3.5 |
| 2 Confidence interval (µM) | 1.3 | 1.1 |
| Electrode Material | Electrochemical Technique | Enzyme | Linear Range | LOD | Ref. |
|---|---|---|---|---|---|
| SPCEs | Amperometry | Urs | 0.37–4.99 µM | 0.31 µM | [59] |
| ISFET | Potentiometry, Conductometry | AChE, BCHE | 10–50 µM | 10 µM | [60] |
| ZnO-NRs | Potentiometry | GOx | 50 nM–20 mM | 0.5 nM | [61] |
| PtE | Amperometry | GOx | 5–180 µM | 2.5 µM | [62] |
| CPE | Amperometry | GOx | 10–160 µM | 2.5 µM | [63] |
| GCE/MWCNT-RuO2/Nafion® | Amperometry | GOx | 5–80 µM | 1.0 µM | This work |
| Heavy Metal | 1 Relative Inhibition (%) | Response Time (s) |
|---|---|---|
| Cadmium(II) | 34.2 | 37 |
| Copper(II) | 8.4 | 16 |
| Mercury(II) | 100 | 10 |
| Silver(I) | 48.6 | 9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashrafi, A.M.; Sýs, M.; Sedláčková, E.; Shaaban Farag, A.; Adam, V.; Přibyl, J.; Richtera, L. Application of the Enzymatic Electrochemical Biosensors for Monitoring Non-Competitive Inhibition of Enzyme Activity by Heavy Metals. Sensors 2019, 19, 2939. https://doi.org/10.3390/s19132939
Ashrafi AM, Sýs M, Sedláčková E, Shaaban Farag A, Adam V, Přibyl J, Richtera L. Application of the Enzymatic Electrochemical Biosensors for Monitoring Non-Competitive Inhibition of Enzyme Activity by Heavy Metals. Sensors. 2019; 19(13):2939. https://doi.org/10.3390/s19132939
Chicago/Turabian StyleAshrafi, Amir M., Milan Sýs, Eliška Sedláčková, Amir Shaaban Farag, Vojtěch Adam, Jan Přibyl, and Lukáš Richtera. 2019. "Application of the Enzymatic Electrochemical Biosensors for Monitoring Non-Competitive Inhibition of Enzyme Activity by Heavy Metals" Sensors 19, no. 13: 2939. https://doi.org/10.3390/s19132939
APA StyleAshrafi, A. M., Sýs, M., Sedláčková, E., Shaaban Farag, A., Adam, V., Přibyl, J., & Richtera, L. (2019). Application of the Enzymatic Electrochemical Biosensors for Monitoring Non-Competitive Inhibition of Enzyme Activity by Heavy Metals. Sensors, 19(13), 2939. https://doi.org/10.3390/s19132939









