A Multi-Sensor System for Silkworm Cocoon Gender Classification via Image Processing and Support Vector Machine
Abstract
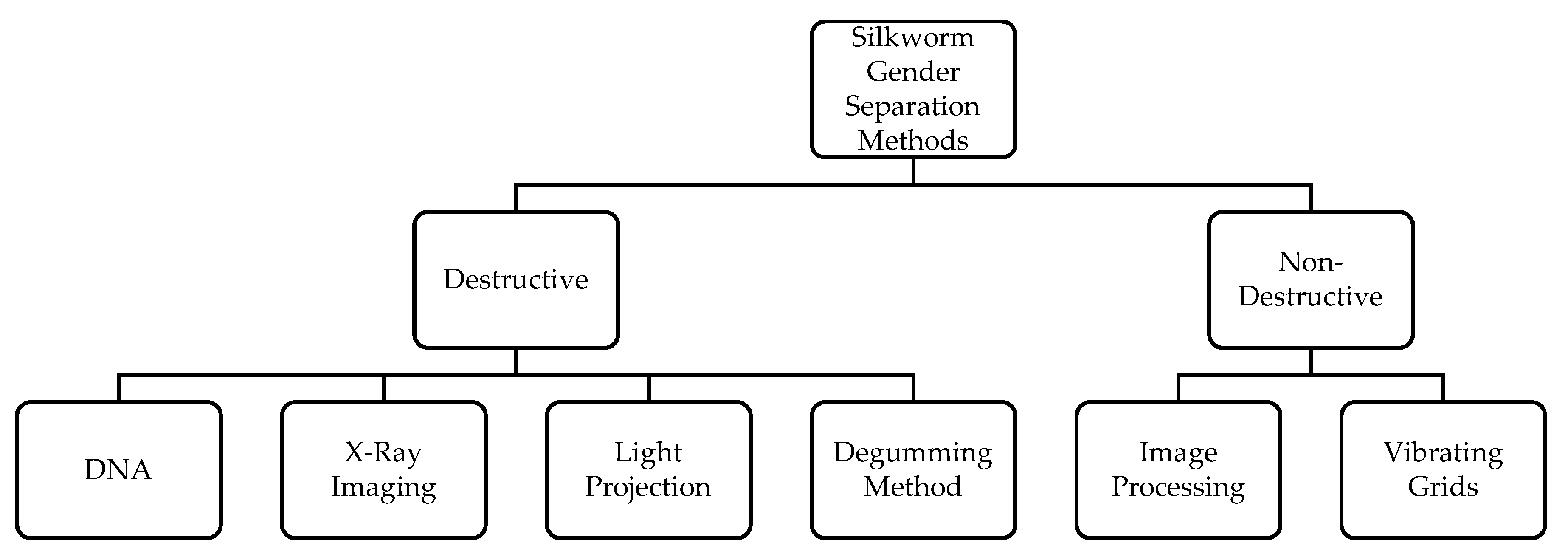
1. Introduction
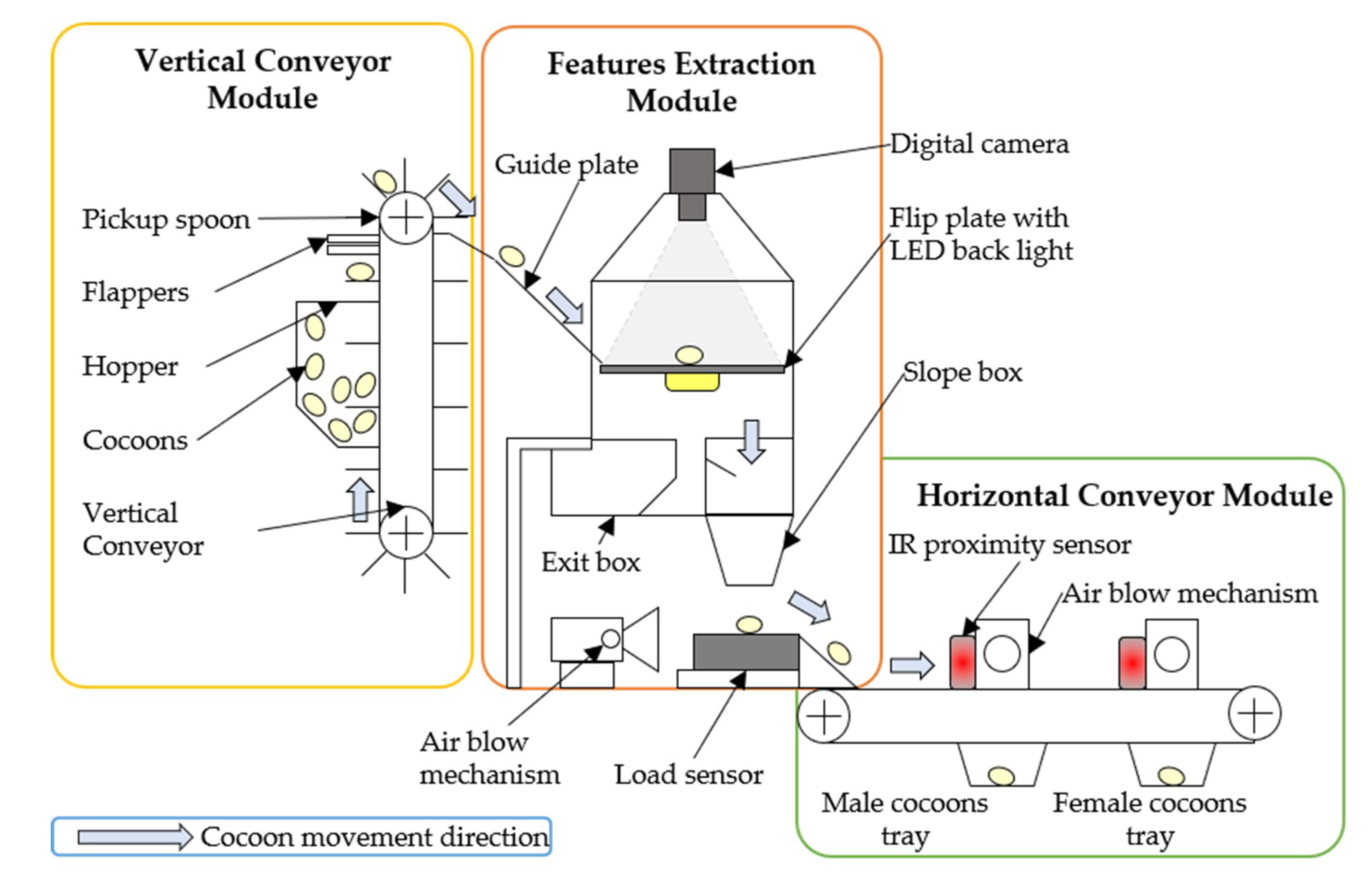
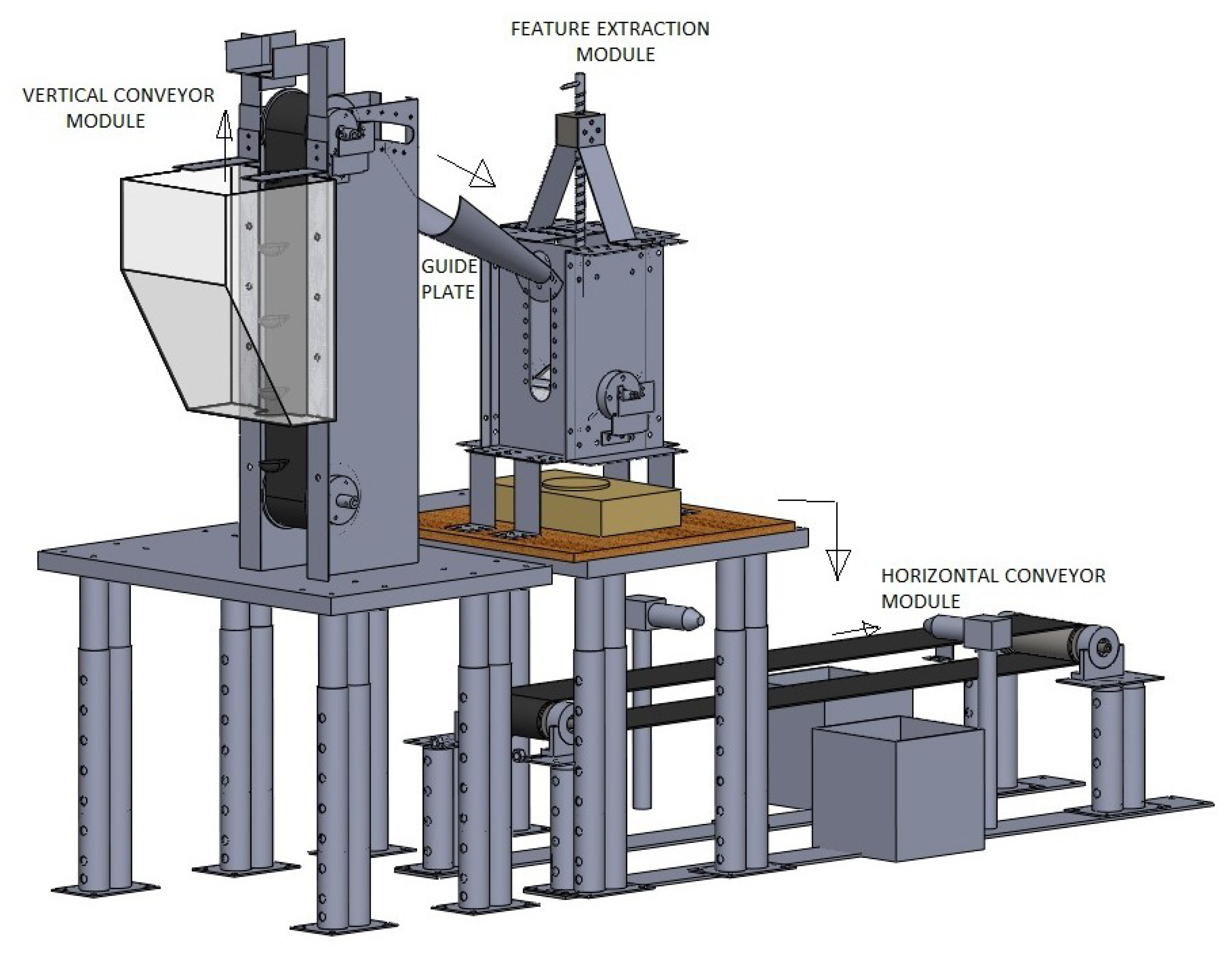
2. Design and Development of Silkworm Cocoon Gender Classification Multi-Sensor System
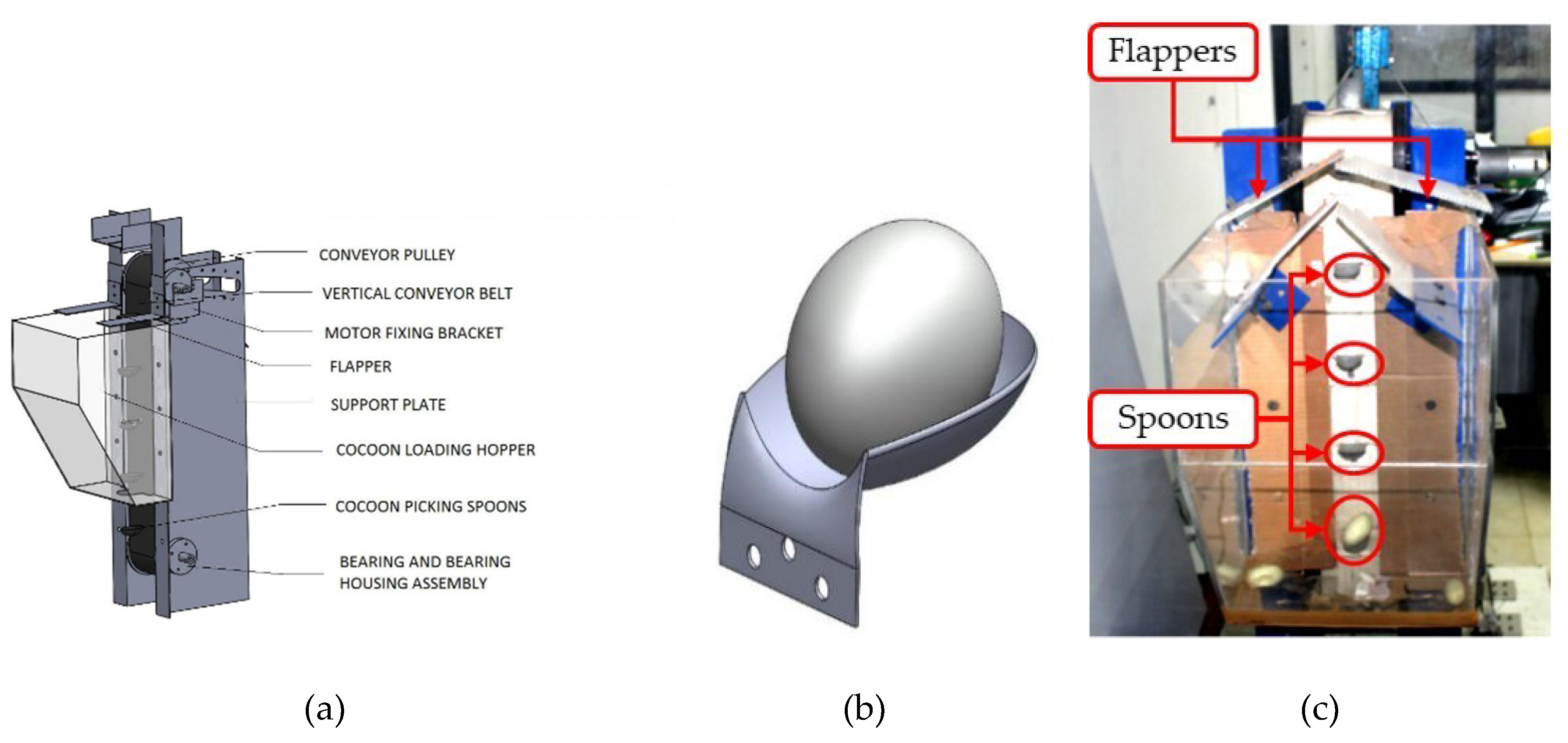
2.1. Vertical Conveyor Module (VCM)
2.2. Features Extraction Module (FEM)
2.3. Horizontal Conveyor Module
2.4. Communication and Synchronization of Modules
3. Experimental Methodology
4. Results and Discussion
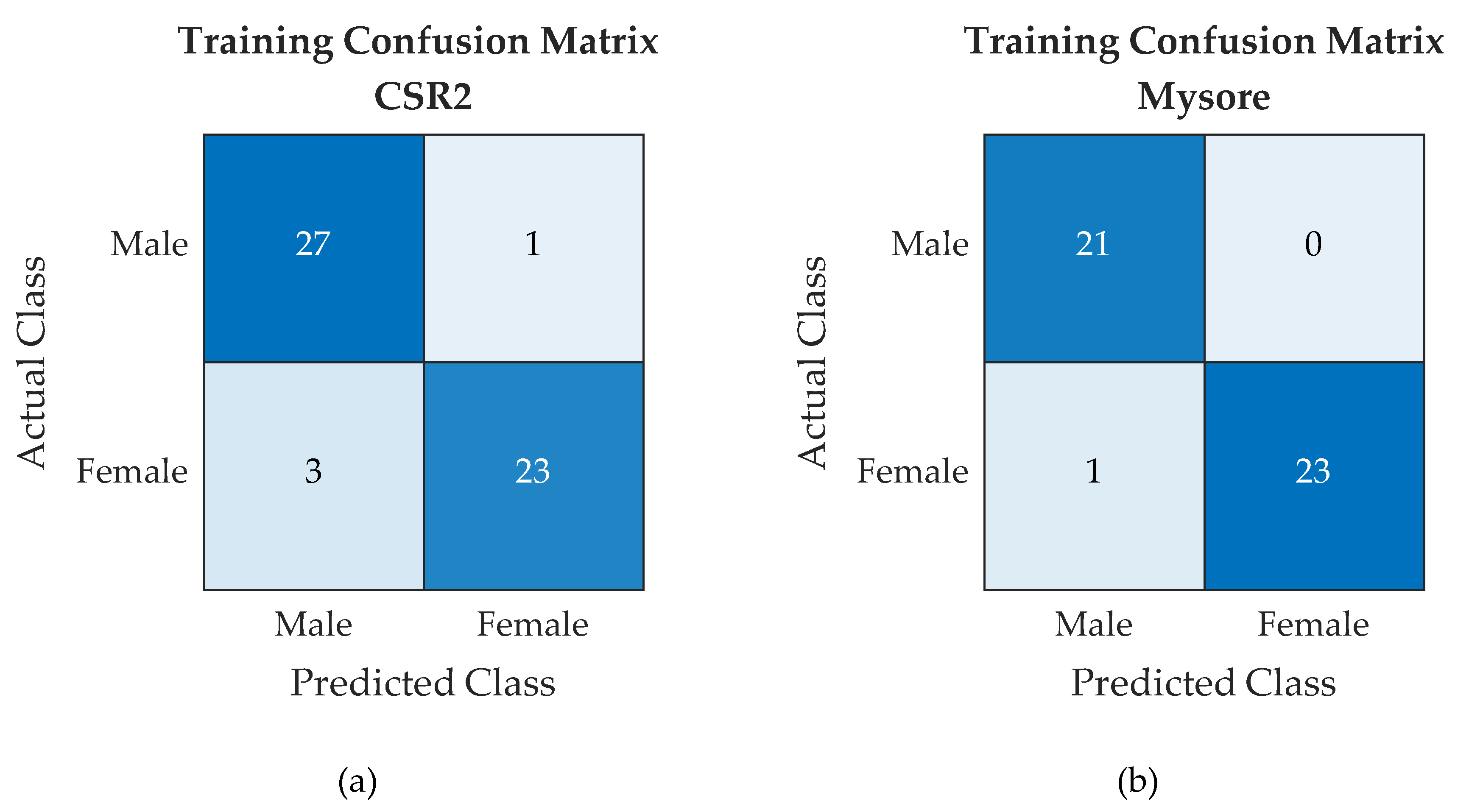
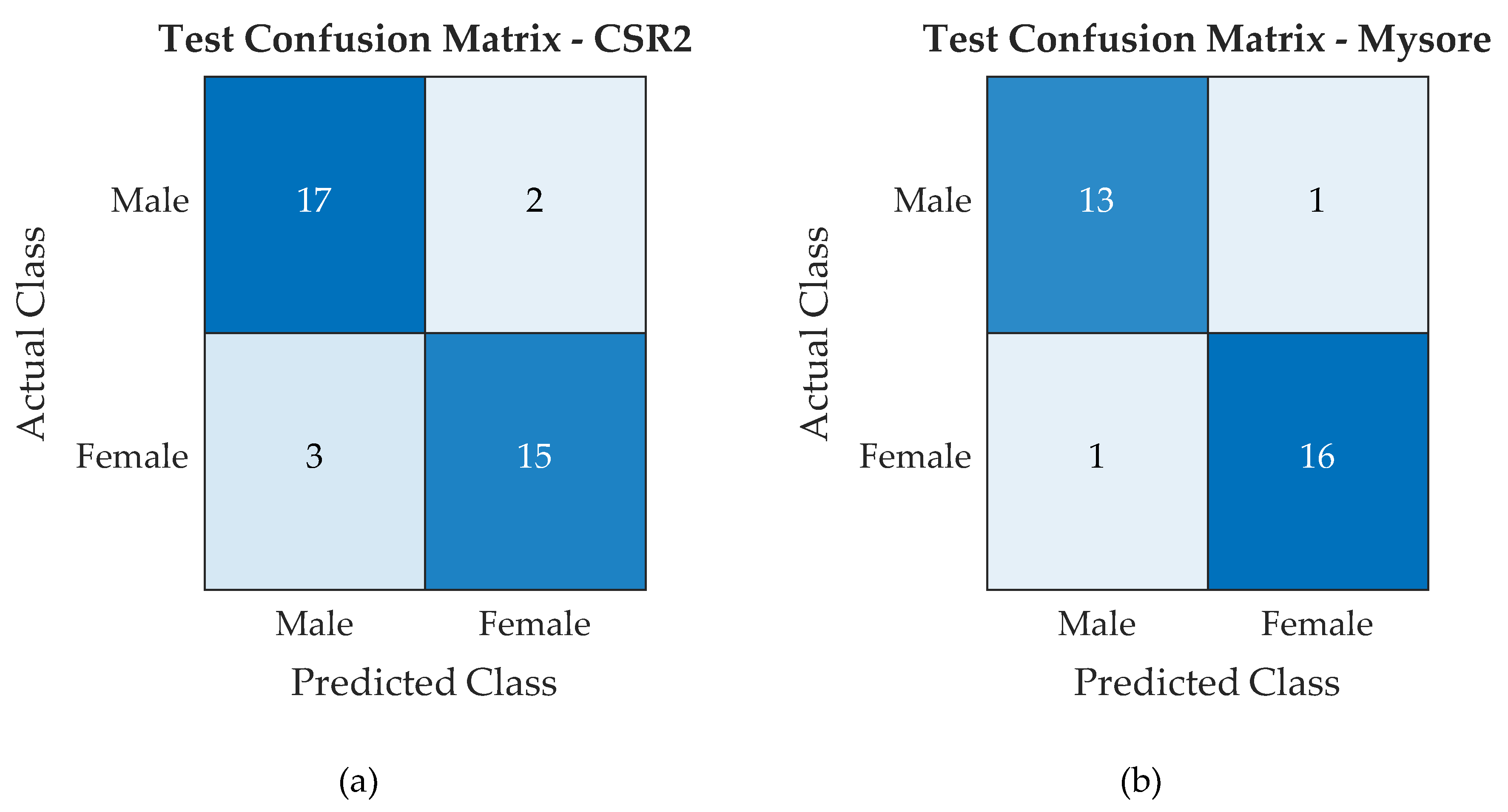
4.1. Classifier Accuracy
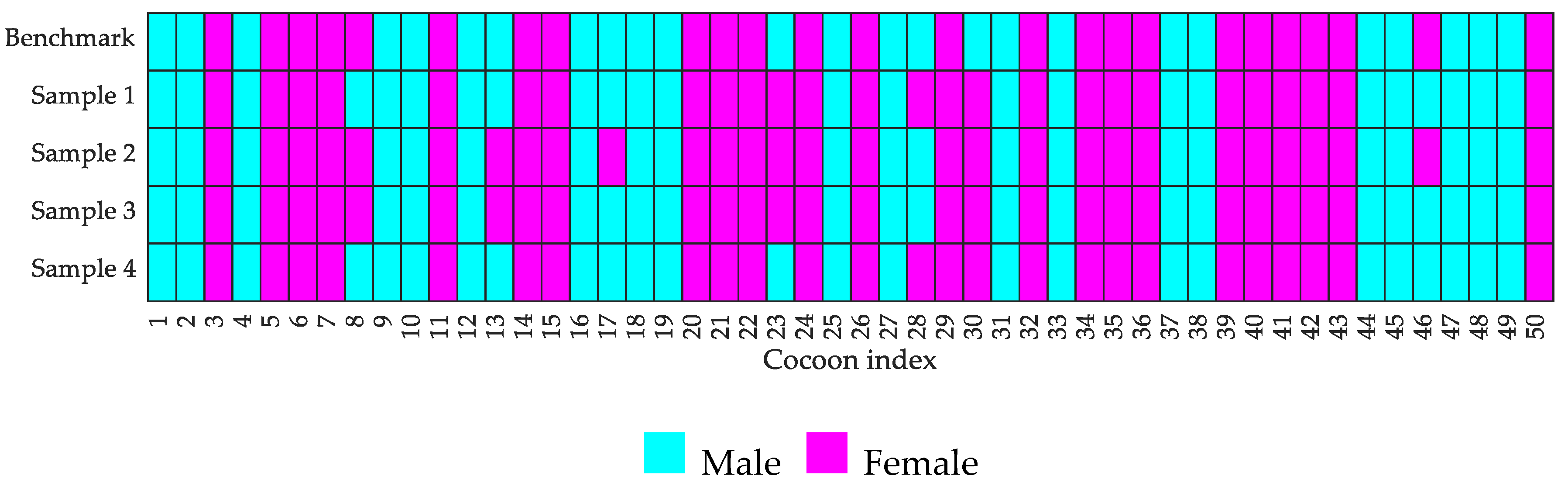
4.2. Robustness and Computation Speed
5. Conclusions
- design optimization to reduce the overall dimensionality and operation speed from a hardware perspective in terms of more powerful workstation and more efficient blower mechanism;
- endowing the VCM with a deflossing unit [5] in order to remove the fibers of the cocoon to avoid clinging phenomena which drastically reduce the system speed;
- extend the experimental campaign to a wider variety of cocoon breeds to improve the system generalization and to increase the system versatility.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Asakura, T.; Kametani, S.; Suzuki, Y. Encyclopedia of Polymer Science and Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 1–19. [Google Scholar]
- Rockwood, D.N.; Preda, R.C.; Yücel, T.; Wang, X.; Lovett, M.L.; Kaplan, D.L. Materials fabrication from bombyx mori silk fibroin. Nat. Protoc. 2011, 6, 1612. [Google Scholar] [CrossRef] [PubMed]
- Sprague, K.U. Bombyx mori silk proteins: Characterization of large polypeptides. Biochemistry 1975, 14, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, S.; Morimoto, T.; Nagata, S.; Tashiro, Y. Studies on the posterior silk gland of the silkworm, bombyx mori. II. Cytolytic processes in posterior silk gland cells during metamorphosis from larva to pupa. J. Cell Biol. 1968, 38, 589–603. [Google Scholar] [CrossRef] [PubMed]
- Murugesh Babu, K. Introduction to silk and sericulture. In Silk; Elsevier: Amsterdam, The Netherlands, 2013; pp. 1–32. [Google Scholar]
- Lee, Y.-W.; Food and Agriculture Organization of the United Nations. Silk Reeling and Testing Manual; Food and Agriculture Organization of the United Nations: Rome, Italy, 1999; ISBN 9251042934. [Google Scholar]
- Govt. of India Central Silk Board—Ministry of Textiles Registered Seed Producers. Available online: http://csb.gov.in/wp-content/uploads/2019/04/FRESH-STATE-WISE-LIST-OF-RSPs-AS-ON-MARCH-2019.pdf (accessed on 4 April 2019).
- Sreekumar, S.; Kadono-Okuda, K.; Nagayasu, K.; Hara, W. Identification of 2nd chromosome region translocated onto the W chromosome by RFLP with EST-cDNA clones in the gensei-kouken strains of the mulberry silkworm, bombyx mori L. Genet. Mol. Biol. 2009, 33, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Bindroo, B.B.; Manthira Moorthy, S. Genetic divergence, implication of diversity, and conservation of silkworm, bombyx mori. Int. J. Biodivers. 2014. [Google Scholar] [CrossRef]
- Narasimhanna, M.N. Manual on Silkworm Egg Production; Central Silk Board: Karnataka, India, 1988. [Google Scholar]
- Sakai, H.; Aoki, F.; Suzuki, M.G. Identification of the key stages for sex determination in the silkworm, bombyx mori. Dev. Genes Evol. 2014, 224, 119–123. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Suzuki, M.G. Sex determination: Insights from the silkworm. J. Genet. 2010, 89, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Ohbayashi, F.; Suzuki, M.G.; Shimada, T. Sex determination in bombyx mori. Curr. Sci. 2002, 83, 466–471. [Google Scholar]
- Calvin, J.; Witdouck, C.J. System and Method for Sorting Larvae Cocoons. U.S. Patent 7,737,379, 15 June 2010. [Google Scholar]
- Tang, P. Sex identification method for silkworm larva. CN Patent CN1990881A, 4 July 2007. [Google Scholar]
- Cai, J.R.; Yuan, L.M.; Liu, B.; Sun, L. Nondestructive gender identification of silkworm cocoons using X-ray imaging with multivariate data analysis. Anal. Methods 2014, 6, 7224–7233. [Google Scholar] [CrossRef]
- Wang, G.-Z.; Li, J.; Hu, Y.-T.; Li, Y.; Du, Z.-Y. Fault identification of chemical processes based on k-NN variable contribution and CNN data reconstruction methods. Sensors 2019, 19, 929. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Liang, G.; Tian, H.; Sun, J.; Wan, C. Electronic nose-based technique for rapid detection and recognition of moldy apples. Sensors 2019, 19, 1526. [Google Scholar] [CrossRef] [PubMed]
- Segreto, T.; Simeone, A.; Teti, R. Principal component analysis for feature extraction and NN pattern recognition in sensor monitoring of chip form during turning. CIRP J. Manuf. Sci. Technol. 2014, 7, 202–209. [Google Scholar] [CrossRef]
- Mahesh, V.G.V.; Raj, A.N.J.; Celik, T. Silkworm cocoon classification using fusion of zernike moments-based shape descriptors and physical parameters for quality egg production. Int. J. Intell. Syst. Technol. Appl. 2017, 16, 246–248. [Google Scholar] [CrossRef]
- Subramanya, G. Effect of X-ray irradiation on the male moths of two tropical races of the silkworm bombyx mori and inheritance of induced Sterility in the progenies. Int. J. Insect Sci. 2010, 2, IJIS-S3655. [Google Scholar] [CrossRef]
- Hayashizaki, S.; Machida, J.; Maeda, H. Method and device for determining sex of silkworm pupa. JP Patent JP2007071620A, 22 March 2007. [Google Scholar]
- El-Raie, A.E.; El-Rahman, A.A.; Saker, H.E.; Hassan, H.E. Comparative light inspection to sort bombyx mori l. cocoons. Misr J. Agric. Eng. 2006, 23, 651–668. [Google Scholar]
- Yang, B.; Lu, F.; Ren, J.; Zeng, W.; Tang, X.; Shi, X.; Liang, Y.; Li, Y.; Ding, Z.; Jia, Z. Automatic male and female sorting apparatus for cocoons judged by fluorescence color. CN Patent CN101403142B, 18 August 2010. [Google Scholar]
- Zhang, Y.; Yu, X.; Shen, W.; Ma, Y.; Zhou, L.; Xu, N.; Yi, S. Mechanism of fluorescent cocoon sex identification for silkworms bombyx mori. Sci. China Life Sci. 2010, 53, 1330–1339. [Google Scholar] [CrossRef] [PubMed]
- Yu, X. Method for identifying male silk of mulberry silkworm. CN Patent CN102590482A, 18 July 2012. [Google Scholar]
- Liu, C.; Ren, Z.H.; Wang, H.Z.; Yang, P.Q.; Zhang, X.L. Analysis on gender of silkworms by MRI technology. In Proceedings of the 2008 International Conference on BioMedical Engineering and Informatics, Sanya, China, 28–30 May 2008. [Google Scholar]
- Udaya, C.J.; Subrata, R.; Garageswari, N.R.; Kariyappa. An automatic Tasar cocoon separating machine. Indian Patent 2374/CHE/2008 A, 7 October 2011. [Google Scholar]
- Sumriddetchkajorn, S.; Kamtongdee, C. Optical penetration-based silkworm pupa gender sensor structure. Appl. Opt. 2012, 51, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Abutaleb, A.S. Automatic thresholding of gray-level pictures using two-dimensional entropy. Comput. Vis. Graph. Image Process. 1989, 47, 22–32. [Google Scholar] [CrossRef]
- Daniel Ratna Raju, P.; Neelima, G. Image segmentation by using histogram thresholding. Int. J. Comput. Sci. Eng. Technol. 2012, 2, 776–779. [Google Scholar]
- Otsu, N. A Threshold selection method from gray-level histograms. IEEE Trans. Syst. Man. Cybern. 1979, 9, 62–66. [Google Scholar] [CrossRef]
- Ruppert, D. The elements of statistical learning: Data mining, inference, and prediction. J. Am. Stat. Assoc. 2004. [Google Scholar] [CrossRef]
- Cortes, C.; Vapnik, V. Support-vector networks. Mach. Learn. 1995, 20, 273–297. [Google Scholar] [CrossRef]
- Pal, M.; Mather, P.M. Support vector machines for classification in remote sensing. Int. J. Remote Sens. 2005, 26, 1007–1011. [Google Scholar] [CrossRef]
- Boser, B.E.; Guyon, I.M.; Vapnik, V.N. A training algorithm for optimal margin classifiers. In Proceedings of the Fifth Annual Workshop on Computational Learning Theory, Pittsburgh, PA, USA, 27–29 July 1992. [Google Scholar]
- Nawi, N.M.; Atomi, W.H.; Rehman, M.Z. The effect of data pre-processing on optimized training of artificial neural networks. Procedia Technol. 2013, 11, 32–39. [Google Scholar] [CrossRef]
- Ting, K.M. Confusion matrix. In Encyclopedia of Machine Learning and Data Mining; Springer: Boston, MA, USA, 2017; p. 260. ISBN 978-1-4899-7687-1. [Google Scholar]
- Huddleston, S.H.; Brown, G.G. Machine learning. In Informs Analytics Body of Knowledge; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 231–274. ISBN 9781119505914. [Google Scholar]
- Bishop, C.M. Pattern Recognition and Machine Learning (Information Science and Statistics); Springer: New York, NY, USA, 2006; ISBN 9780387310732. [Google Scholar]
- Araujo, L.M.L.; Filho, N.A.R.; Riedi, C.A. Respiratory allergy to moth: The importance of sensitization to bombyx mori in children with asthma and rhinitis. J. Pediatr. 2014, 90, 176–181. [Google Scholar] [CrossRef]
- Reshma, S.; Adhikrao, D.J. Occupational health and physiological profile of sericulture industry workers with respect to Workplace environment. Int. J. Pharm. Bio Sci. 2016, 7, 137–143. [Google Scholar]
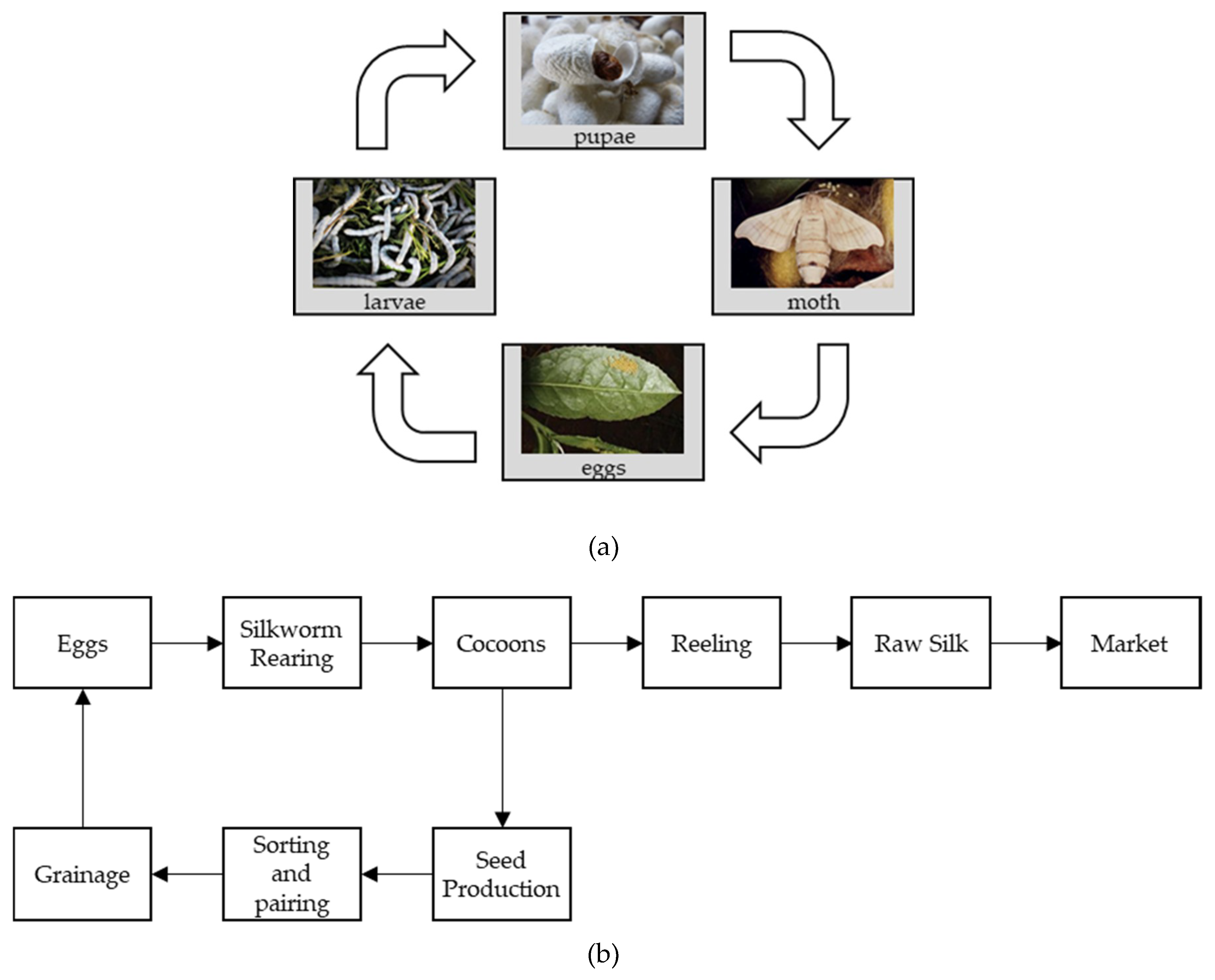







| Pre-Cocoon Stage | Post-Cocoon Stage |
|---|---|
| Machine for crushing shoots | Cocoon de-flossing machine |
| Mulberry pruning machine | Denier detecting device in silk reeling |
| Litter separation machine | Long skein silk book making machine |
| Pedal-operated reeling twisting machine for muga and tasar silk Reeling | |
| Reeling and twisting machine | |
| Solar-operated spinning machine | |
| Motorized pedal-operated spinning machine | |
| Wet reeling machine |
| Stages | Methods | Remarks |
|---|---|---|
| Chromosome | Presence or absence of the “W” chromosome Female—ZW chromosome Male—ZZ chromosome | Not practical—high cost |
| Egg | Color of the egg Males are usually light yellow Females are dark brown in color | Not practical—need for skilled workers |
| Larval | Markings are exhibited on the larval body Female—crescent marking Males—plain | Sex separation is possible only on the 1st day of 5th instar. Process is laborious and too slow operation, larvae may get injured |
| Cocoon | Color and weight Color—females are golden yellow/ Males white—CSR2 Weight—females are heavier than males | Color depends on various silkworm breeds Weight—each cocoon is weighed individually and sorted—presently followed in grainages—non-destructive |
| Pupa | Males are smaller in size whereas females are plumper | Reliable/low error—cocoons have to be cut open to remove the pupae, which may cause injury to pupae |
| Moth | Males are small, slender active moving in semi-circles with bent abdomen/females are bigger with bloated abdomen and rather lethargic | Males and females are easily separated. Selfing takes place affecting the quality of the eggs, health hazards from moth dust |
| Training Set | Testing Set | |||
|---|---|---|---|---|
| M | F | M | F | |
| CSR2 | 28 | 26 | 19 | 18 |
| Pure Mysore | 21 | 24 | 14 | 17 |
| Parameter | Description |
|---|---|
| Area () | Describes the number of pixels in the region of the shape |
| Perimeter () | Provides the number of pixels in the boundary of the shape |
| Major axis length () | Specifies the length (in pixels) of the major axis of the ellipse that has the same normalized second central moments as the region |
| Minor axis length () | Specifies the length (in pixels) of the minor axis of the ellipse that has the same normalized second central moments as the region |
| Eccentricity () | Measure of the aspect ratio. Computed using minimum bounding box (smallest rectangle containing every point in the shape) method.
|
| Circularity/Roundness () | Circularity ratio represents how a shape is similar to a circle. It is given by the ratio of the area of a shape to the shape’s perimeter square. |
| Rectangularity () | Represents how rectangular a shape is, i.e. how much it fills its minimum bounding rectangle. It is given by: |
| Solidity () | Describes the extent to which the shape is convex or concave. It is given by:
|
| Convex area | Specifies the number of pixels in convex image. It is given by:
|
| Training | Testing | |||
|---|---|---|---|---|
| PM | CSR2 | Pure Mysore | CSR2 | Pure Mysore |
| Accuracy: | 0.9259 | 0.9778 | 0.8649 | 0.9355 |
| True Male Rate | 0.9642 | 1.0000 | 0.8947 | 0.9286 |
| True Female Rate | 0.8846 | 0.9583 | 0.8333 | 0.9412 |
| Male Predictive Value | 0.9000 | 0.9545 | 0.85 | 0.9286 |
| Female Predictive Value | 0.9583 | 1.0000 | 0.8824 | 0.9412 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joseph Raj, A.N.; Sundaram, R.; Mahesh, V.G.V.; Zhuang, Z.; Simeone, A. A Multi-Sensor System for Silkworm Cocoon Gender Classification via Image Processing and Support Vector Machine. Sensors 2019, 19, 2656. https://doi.org/10.3390/s19122656
Joseph Raj AN, Sundaram R, Mahesh VGV, Zhuang Z, Simeone A. A Multi-Sensor System for Silkworm Cocoon Gender Classification via Image Processing and Support Vector Machine. Sensors. 2019; 19(12):2656. https://doi.org/10.3390/s19122656
Chicago/Turabian StyleJoseph Raj, Alex Noel, Rahul Sundaram, Vijayalakshmi G.V. Mahesh, Zhemin Zhuang, and Alessandro Simeone. 2019. "A Multi-Sensor System for Silkworm Cocoon Gender Classification via Image Processing and Support Vector Machine" Sensors 19, no. 12: 2656. https://doi.org/10.3390/s19122656
APA StyleJoseph Raj, A. N., Sundaram, R., Mahesh, V. G. V., Zhuang, Z., & Simeone, A. (2019). A Multi-Sensor System for Silkworm Cocoon Gender Classification via Image Processing and Support Vector Machine. Sensors, 19(12), 2656. https://doi.org/10.3390/s19122656






