Colorimetric Analysis of Glucose Oxidase-Magnetic Cellulose Nanocrystals (CNCs) for Glucose Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Magnetic CNCs
2.2. Characterization of as-Prepared Nanomaterials
2.3. Immobilization of Glucose Oxidase on Magnetic CNCs
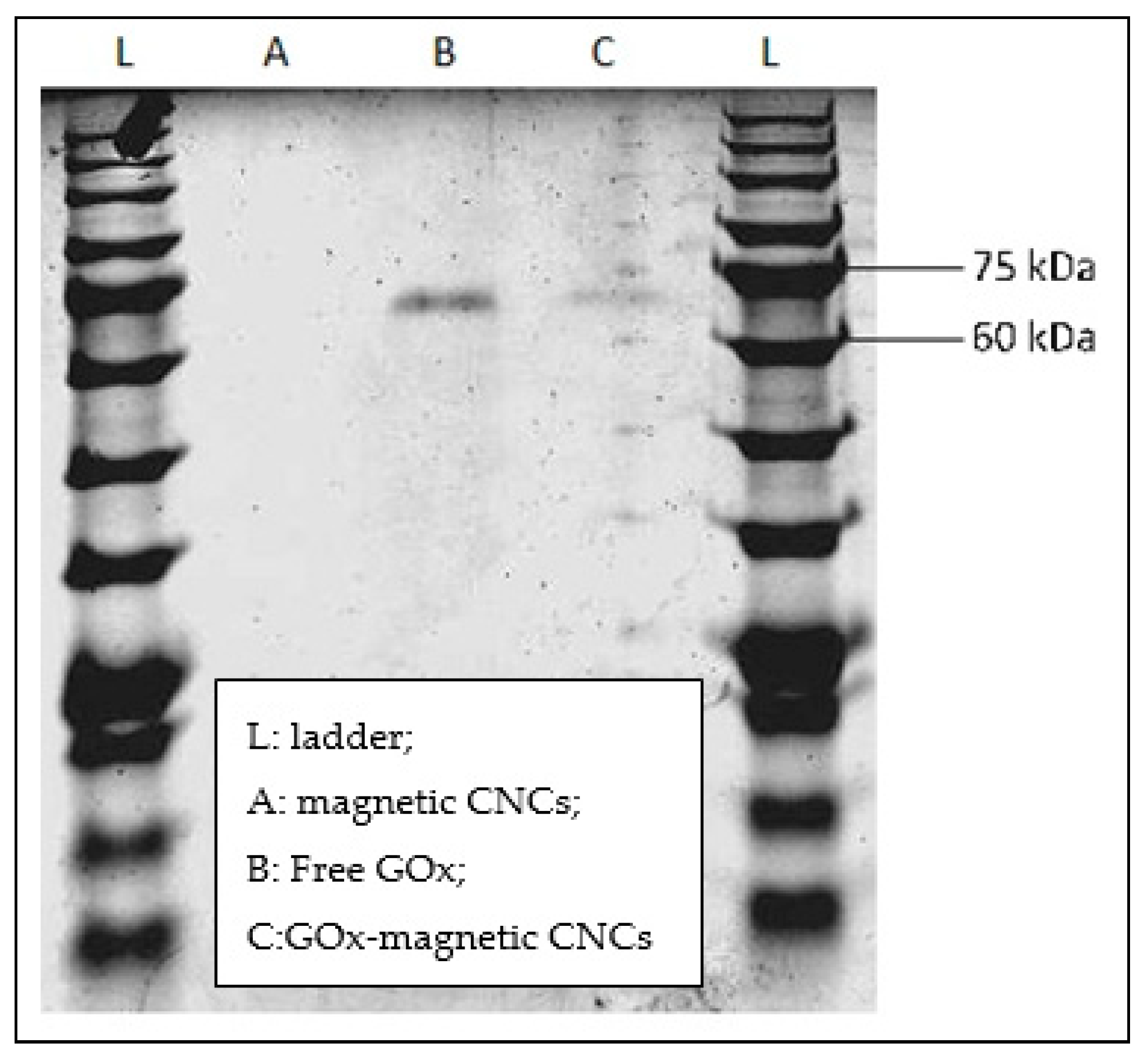
2.3.1. SDS-PAGE
2.3.2. Bradford Assay
2.3.3. Determination of Immobilized GOx Efficiency
2.4. Glucose Detection Using ABTS Assay
2.5. Specificity Test
2.6. Reusability and Storage Stability of Immobilized GOx for Glucose Detection
3. Results and Discussion
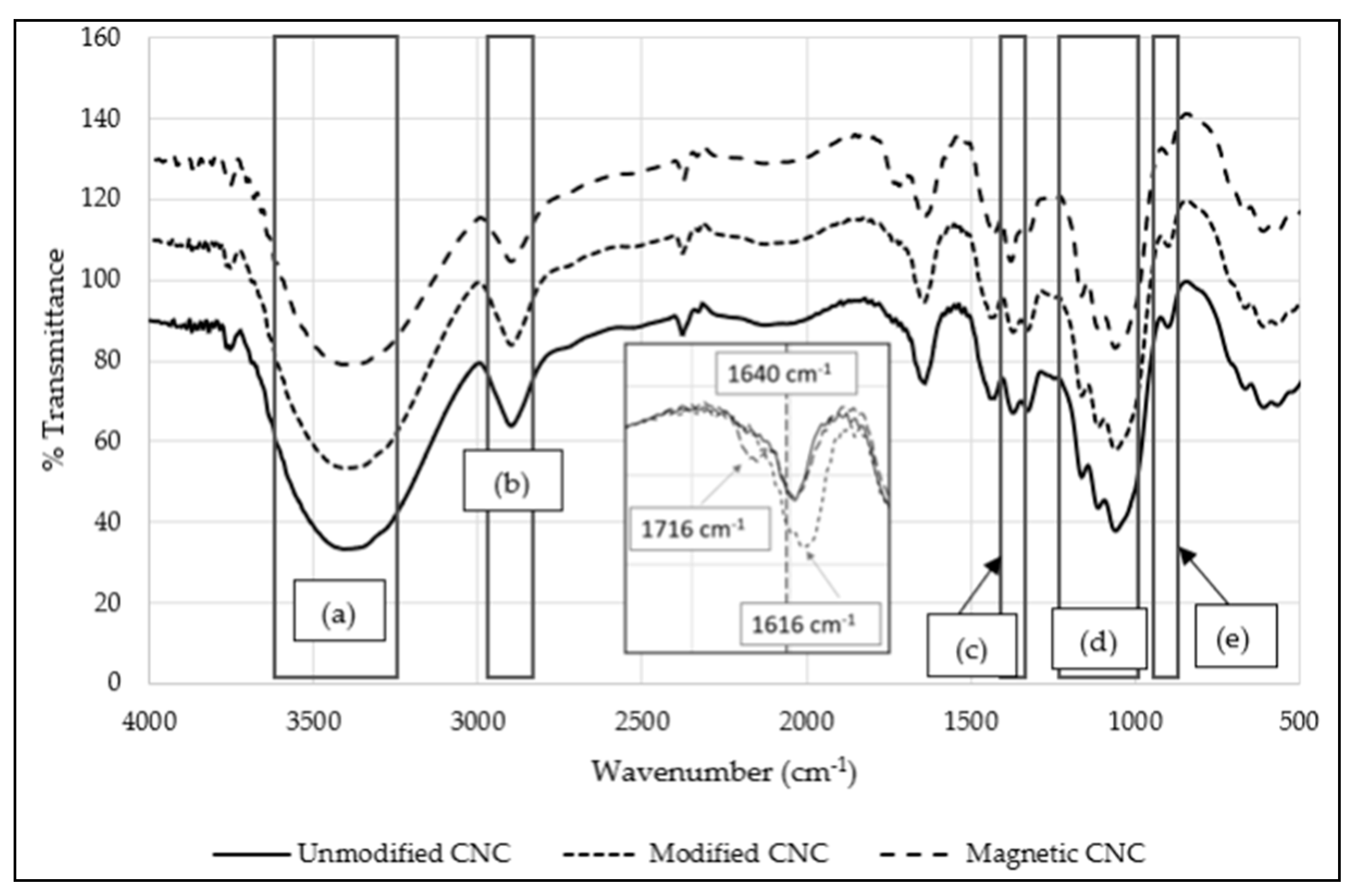
3.1. Characterization
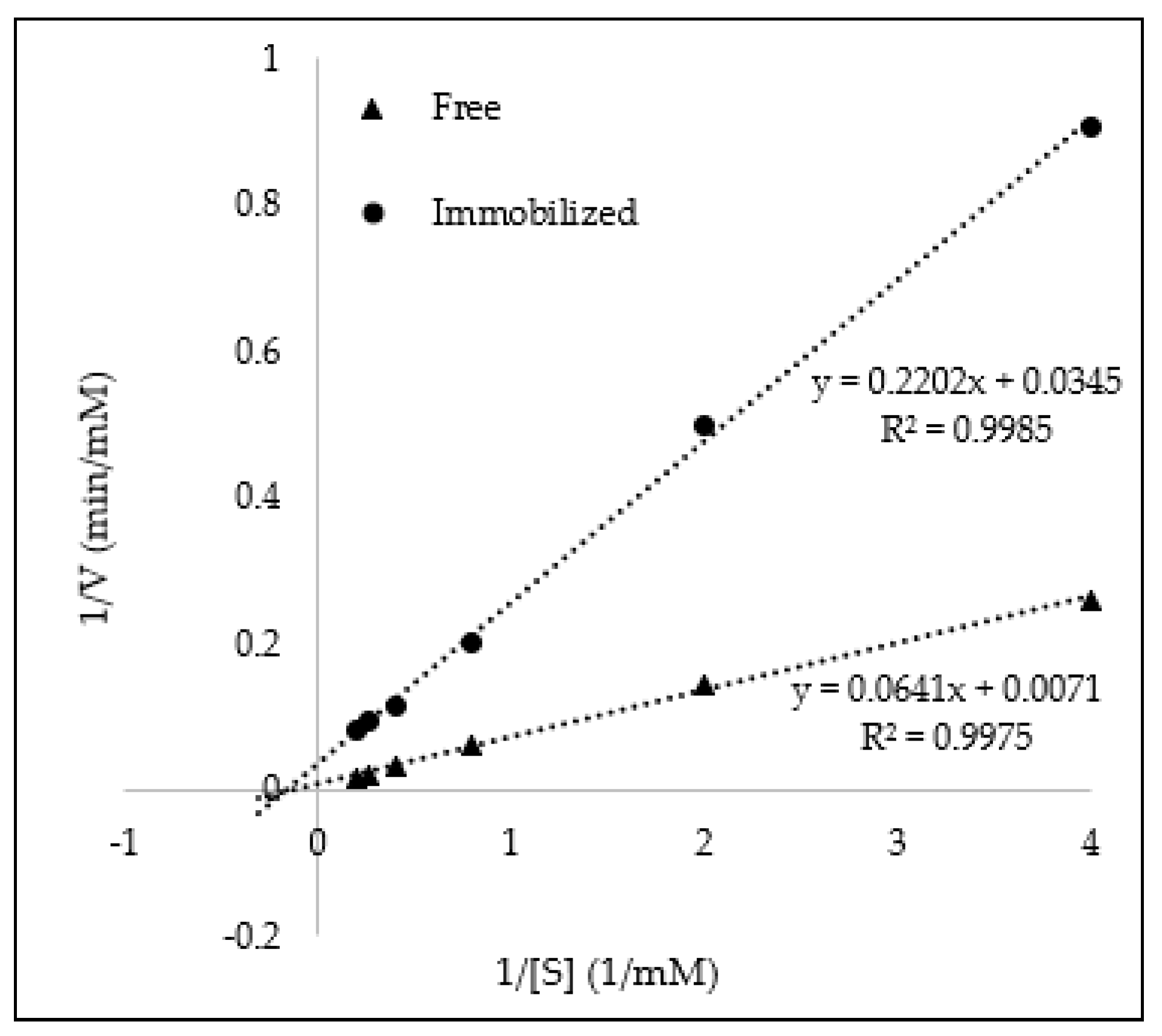
3.2. Immobilization of Glucose Oxidase on Magnetic CNCs
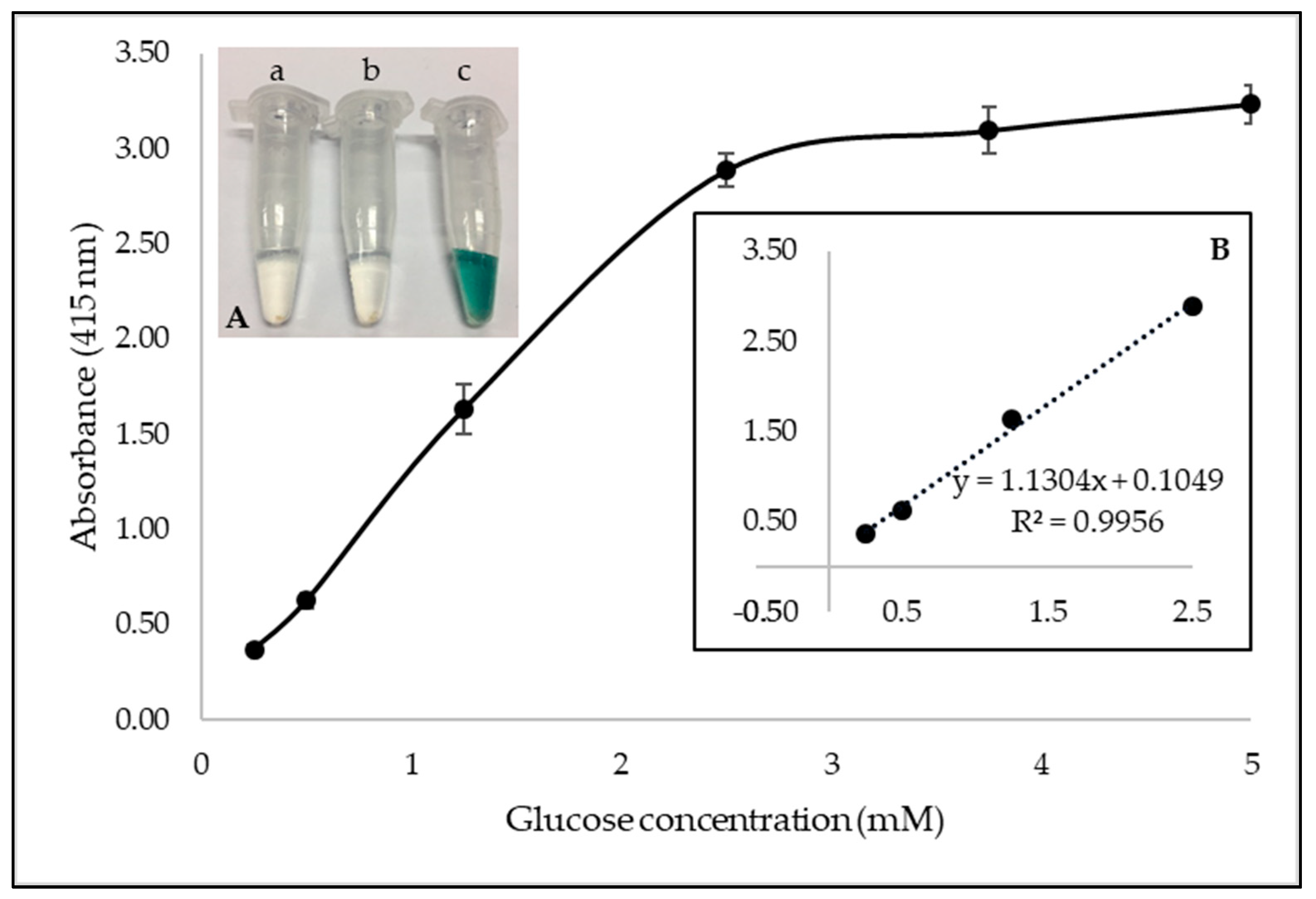
3.3. Glucose Detection
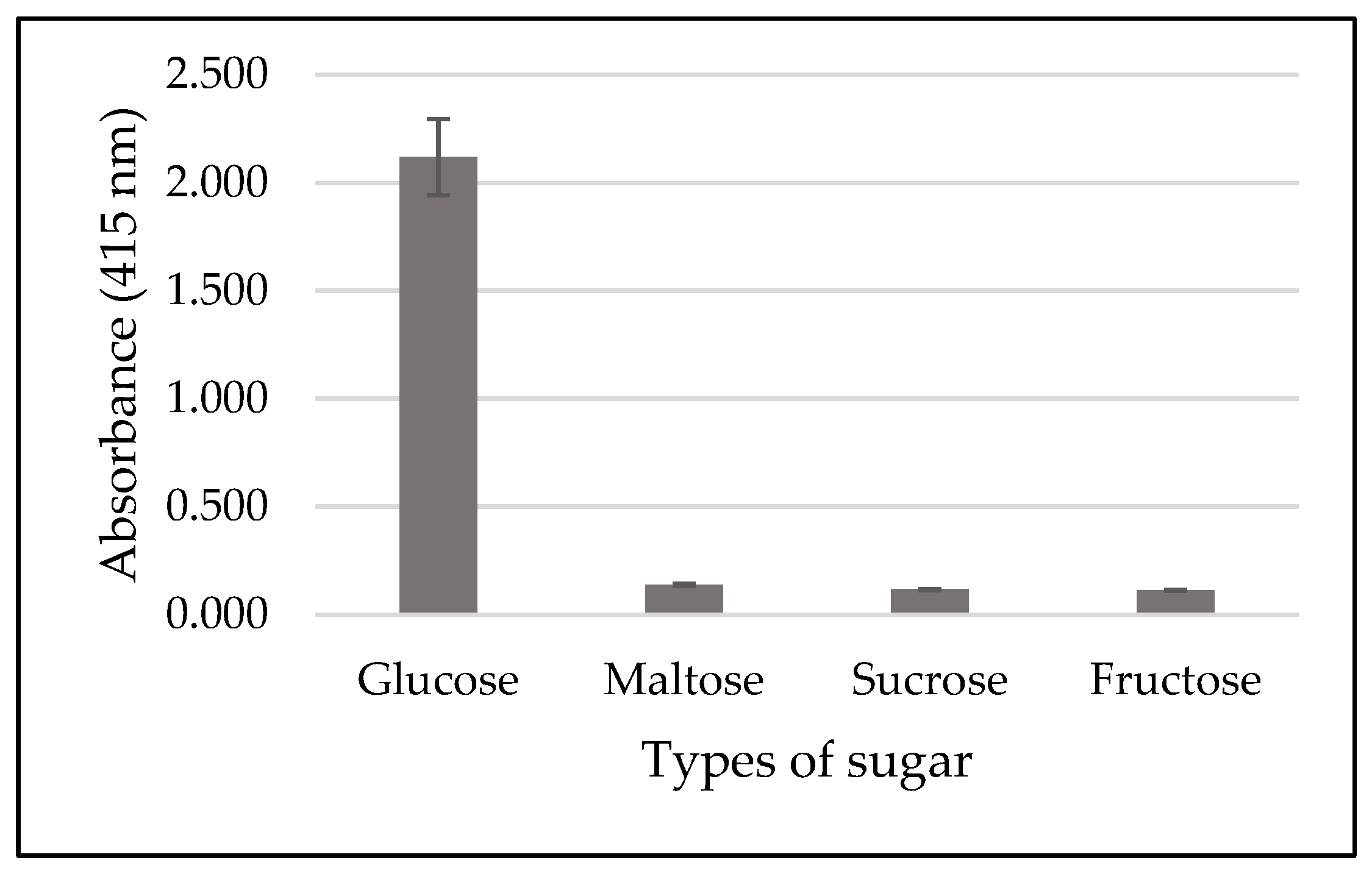
3.4. Specificity Test
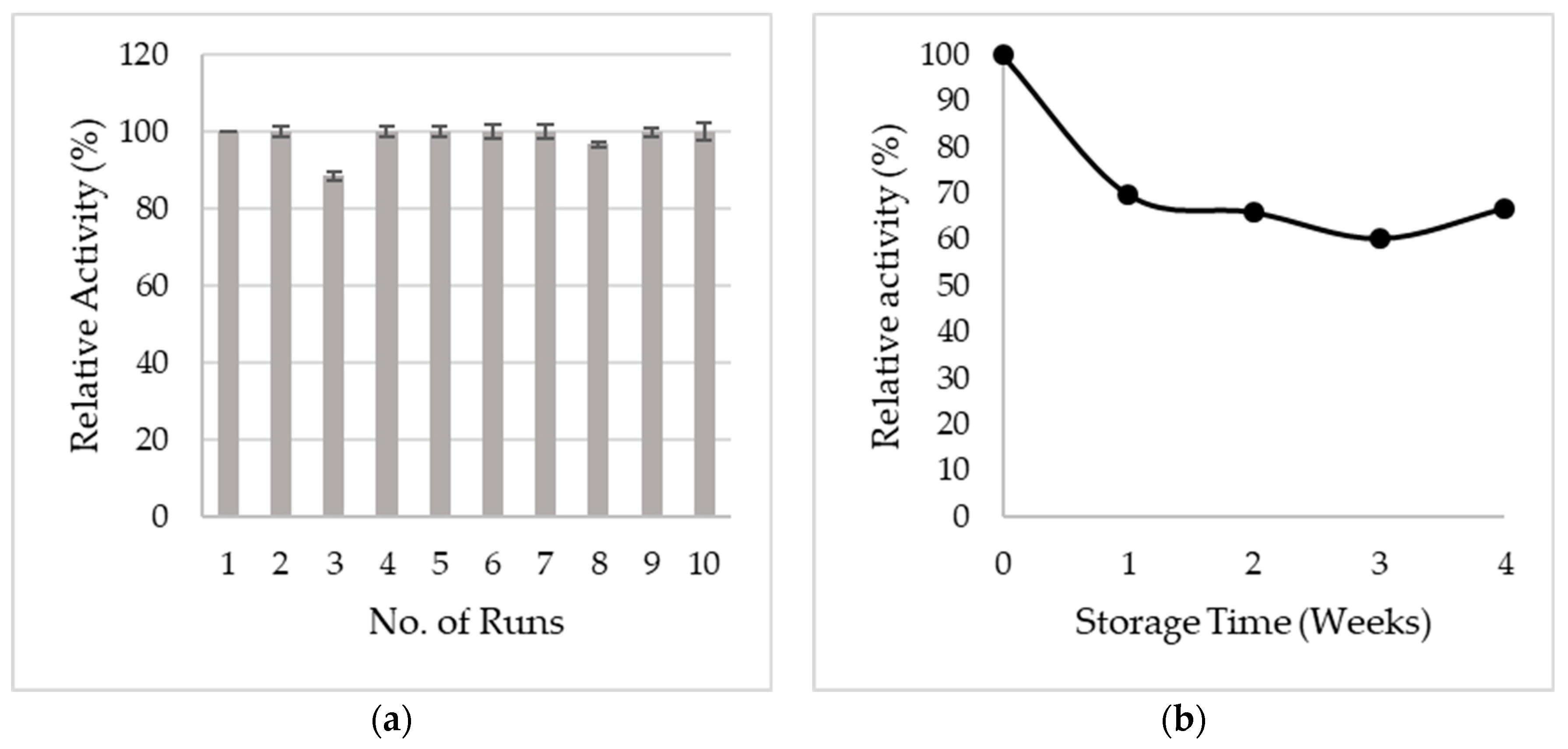
3.5. Reusability and Storage Stability
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hsueh, C.-J.; Janyasupab, M.; Lee, Y.-H.; Liu, C.-C. Electrochemical glucose sensors. In Encyclopedia of Applied Electrochemistry; Springer: Berlin/Heidelberg, Germany, 2014; pp. 479–485. [Google Scholar]
- Jang, H.; Min, D.-H. Highly precise plasmonic and colorimetric sensor based on enzymatic etching of nanospheres for the detection of blood and urinary glucose. RSC Adv. 2015, 5, 14330–14332. [Google Scholar] [CrossRef]
- Ahmad, R.; Sardar, M. Enzyme immobilization: An overview on nanoparticles as immobilization matrix. Biochem. Anal. Biochem. 2015, 4, 1. [Google Scholar]
- Nguyen, H.H.; Kim, M. An Overview of Techniques in Enzyme Immobilization. Appl. Sci. Converg. Technol. 2017, 26, 157–163. [Google Scholar]
- Lin, N.; Dufresne, A. Nanocellulose in biomedicine: Current status and future prospect. Eur. Polym. J. 2014, 59, 302–325. [Google Scholar] [CrossRef]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Hsieh, Y.-L. Preparation and properties of cellulose nanocrystals: Rods, spheres, and network. Carbohydr. Polym. 2010, 82, 329–336. [Google Scholar] [CrossRef]
- Dong, S.; Hirani, A.A.; Colacino, K.R.; Lee, Y.W.; Roman, M. Cytotoxicity and cellular uptake of cellulose nanocrystals. Nano Life 2012, 2, 1241006. [Google Scholar] [CrossRef]
- Kim, H.J.; Park, S.; Kim, S.H.; Kim, J.H.; Yu, H.; Kim, H.J.; Yang, Y.-H.; Kan, E.; Kim, Y.H.; Lee, S.H. Biocompatible cellulose nanocrystals as supports to immobilize lipase. J. Mol. Catal. B Enzym. 2015, 122, 170–178. [Google Scholar] [CrossRef]
- Edwards, J.V.; Prevost, N.; French, A.; Concha, M.; DeLucca, A.; Wu, Q. Nanocellulose-based biosensors: Design, preparation, and activity of peptide-linked cotton cellulose nanocrystals having fluorimetric and colorimetric elastase detection sensitivity. Engineering 2013, 5, 20. [Google Scholar] [CrossRef]
- Incani, V.; Danumah, C.; Boluk, Y. Nanocomposites of nanocrystalline cellulose for enzyme immobilization. Cellulose 2013, 20, 191–200. [Google Scholar] [CrossRef]
- Yang, R.; Tan, H.; Wei, F.; Wang, S. Peroxidase conjugate of cellulose nanocrystals for the removal of chlorinated phenolic compounds in aqueous solution. Biotechnology 2008, 7, 233–241. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, R.; Zhen, C.; Li, B. Magnetic cellulose nanocrystals: Synthesis by electrostatic self-assembly approach and efficient use for immobilization of papain. J. Mol. Catal. B Enzym. 2016, 134, 164–171. [Google Scholar] [CrossRef]
- Cao, S.-L.; Li, X.-H.; Lou, W.-Y.; Zong, M.-H. Preparation of a novel magnetic cellulose nanocrystal and its efficient use for enzyme immobilization. J. Mater. Chem. B 2014, 2, 5522–5530. [Google Scholar] [CrossRef]
- Dhar, P.; Kumar, A.; Katiyar, V. Magnetic cellulose nanocrystal based anisotropic polylactic acid nanocomposite films: Influence on electrical, magnetic, thermal, and mechanical properties. ACS Appl. Mater. Interfaces 2016, 8, 18393–18409. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Filpponen, I.; Johansson, L.-S.; Mohammadi, P.; Latikka, M.; Linder, M.B.; Ras, R.H.; Rojas, O.J. Complexes of magnetic nanoparticles with cellulose nanocrystals as regenerable, highly efficient, and selective platform for protein separation. Biomacromolecules 2017, 18, 898–905. [Google Scholar] [CrossRef]
- Sadasivuni, K.K.; Ponnamma, D.; Ko, H.-U.; Kim, H.C.; Zhai, L.; Kim, J. Flexible NO2 sensors from renewable cellulose nanocrystals/iron oxide composites. Sens. Actuators B Chem. 2016, 233, 633–638. [Google Scholar] [CrossRef]
- Lamaming, J.; Hashim, R.; Sulaiman, O.; Leh, C.P.; Sugimoto, T.; Nordin, N.A. Cellulose nanocrystals isolated from oil palm trunk. Carbohydr. Polym. 2015, 127, 202–208. [Google Scholar] [CrossRef]
- Saito, T.; Isogai, A. TEMPO-mediated oxidation of native cellulose. The effect of oxidation conditions on chemical and crystal structures of the water-insoluble fractions. Biomacromolecules 2004, 5, 1983–1989. [Google Scholar] [CrossRef]
- Bustami, Y.; Moo-Young, M.; Anderson, W. Analysis of the heterogeneous structure of iron oxide/gold nanoparticles and their application in a nanosensor. Sens. Actuators B Chem. 2017, 245, 753–764. [Google Scholar] [CrossRef]
- George, J.; Sabapathi, S. Cellulose nanocrystals: Synthesis, functional properties, and applications. Nanotechnol. Sci. Appl. 2015, 8, 45. [Google Scholar] [CrossRef]
- Cui, L.; Liu, Z.; Si, C.; Hui, L.; Kang, N.; Zhao, T. Influence of steam explosion pretreatment on the composition and structure of wheat straw. BioResources 2012, 7, 4202–4213. [Google Scholar]
- Mohan, J. Organic Spectroscopy: Principles and Applications; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Ruan, D.; Zhang, L.; Mao, Y.; Zeng, M.; Li, X. Microporous membranes prepared from cellulose in NaOH/thiourea aqueous solution. J. Membr. Sci. 2004, 241, 265–274. [Google Scholar] [CrossRef]
- Cao, Y.; Tan, H. Structural characterization of cellulose with enzymatic treatment. J. Mol. Struct. 2004, 705, 189–193. [Google Scholar] [CrossRef]
- Zhang, Y.; Tsitkov, S.; Hess, H. Proximity does not contribute to activity enhancement in the glucose oxidase–horseradish peroxidase cascade. Nat. Commun. 2016, 7, 13982. [Google Scholar] [CrossRef] [PubMed]
- Uygun, D.A.; Öztürk, N.; Akgöl, S.; Denizli, A. Novel magnetic nanoparticles for the hydrolysis of starch with Bacillus licheniformis α-amylase. J. Appl. Polym. Sci. 2012, 123, 2574–2581. [Google Scholar] [CrossRef]
- Ahmad, R.; Sardar, M. Immobilization of Cellulase on TiO2 Nanoparticles by Physical and Covalent Methods: A Comparative Study. 2014. Available online: http://nopr.niscair.res.in/handle/123456789/29326 (accessed on 14 March 2019).
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2007, 2, 577. [Google Scholar] [CrossRef] [PubMed]
- Adams, E.C., Jr.; Mast, R.L.; Free, A.H. Specificity of glucose oxidase. Arch. Biochem. Biophys. 1960, 91, 230–234. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, R.; Wang, H.; Zhao, W.; Ding, L. Immobilization of glucose oxidase on Fe3O4/SiO2 magnetic nanoparticles. Biotechnol. Lett. 2010, 32, 817–821. [Google Scholar] [CrossRef]
- Chang, Q.; Tang, H. Optical determination of glucose and hydrogen peroxide using a nanocomposite prepared from glucose oxidase and magnetite nanoparticles immobilized on graphene oxide. Microchim. Acta 2014, 181, 527–534. [Google Scholar] [CrossRef]
- Wang, H.; Huang, J.; Wang, C.; Li, D.; Ding, L.; Han, Y. Immobilization of glucose oxidase using CoFe2O4/SiO2 nanoparticles as carrier. Appl. Surf. Sci. 2011, 257, 5739–5745. [Google Scholar] [CrossRef]
- Rossi, L.M.; Quach, A.D.; Rosenzweig, Z. Glucose oxidase-magnetite nanoparticle bioconjugate for glucose sensing. Anal. Bioanal. Chem. 2004, 380, 606–613. [Google Scholar] [CrossRef] [PubMed]

| Sample | Zeta Potential (mV) |
|---|---|
| CNCs | −42.53 ± 1.595 1 |
| IONPs | +28.37 ± 1.002 1 |
| Magnetic CNCs | −22.97 ± 0.306 1 |
| Immobilization Support | Reusability | References |
|---|---|---|
| Magnetic CNCs | 100% of activity retained after 10 cycles. | This study |
| Fe3O4 nanoparticles | 75% of activity retained after 4 cycles. | [32] |
| Fe3O4/SiO2 nanoparticles | 60% of activity retained after 6 cycles. | [31] |
| CoFe2O4/SiO2 nanoparticles | 57% of activity retained after 7 cycles. | [33] |
| Fe3O4 nanoparticles | 50% of activity retained after 5 cycles. | [34] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yee, Y.C.; Hashim, R.; Mohd Yahya, A.R.; Bustami, Y. Colorimetric Analysis of Glucose Oxidase-Magnetic Cellulose Nanocrystals (CNCs) for Glucose Detection. Sensors 2019, 19, 2511. https://doi.org/10.3390/s19112511
Yee YC, Hashim R, Mohd Yahya AR, Bustami Y. Colorimetric Analysis of Glucose Oxidase-Magnetic Cellulose Nanocrystals (CNCs) for Glucose Detection. Sensors. 2019; 19(11):2511. https://doi.org/10.3390/s19112511
Chicago/Turabian StyleYee, Ying Chuin, Rokiah Hashim, Ahmad Ramli Mohd Yahya, and Yazmin Bustami. 2019. "Colorimetric Analysis of Glucose Oxidase-Magnetic Cellulose Nanocrystals (CNCs) for Glucose Detection" Sensors 19, no. 11: 2511. https://doi.org/10.3390/s19112511
APA StyleYee, Y. C., Hashim, R., Mohd Yahya, A. R., & Bustami, Y. (2019). Colorimetric Analysis of Glucose Oxidase-Magnetic Cellulose Nanocrystals (CNCs) for Glucose Detection. Sensors, 19(11), 2511. https://doi.org/10.3390/s19112511




