Towards Wearable Comprehensive Capture and Analysis of Skeletal Muscle Activity during Human Locomotion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
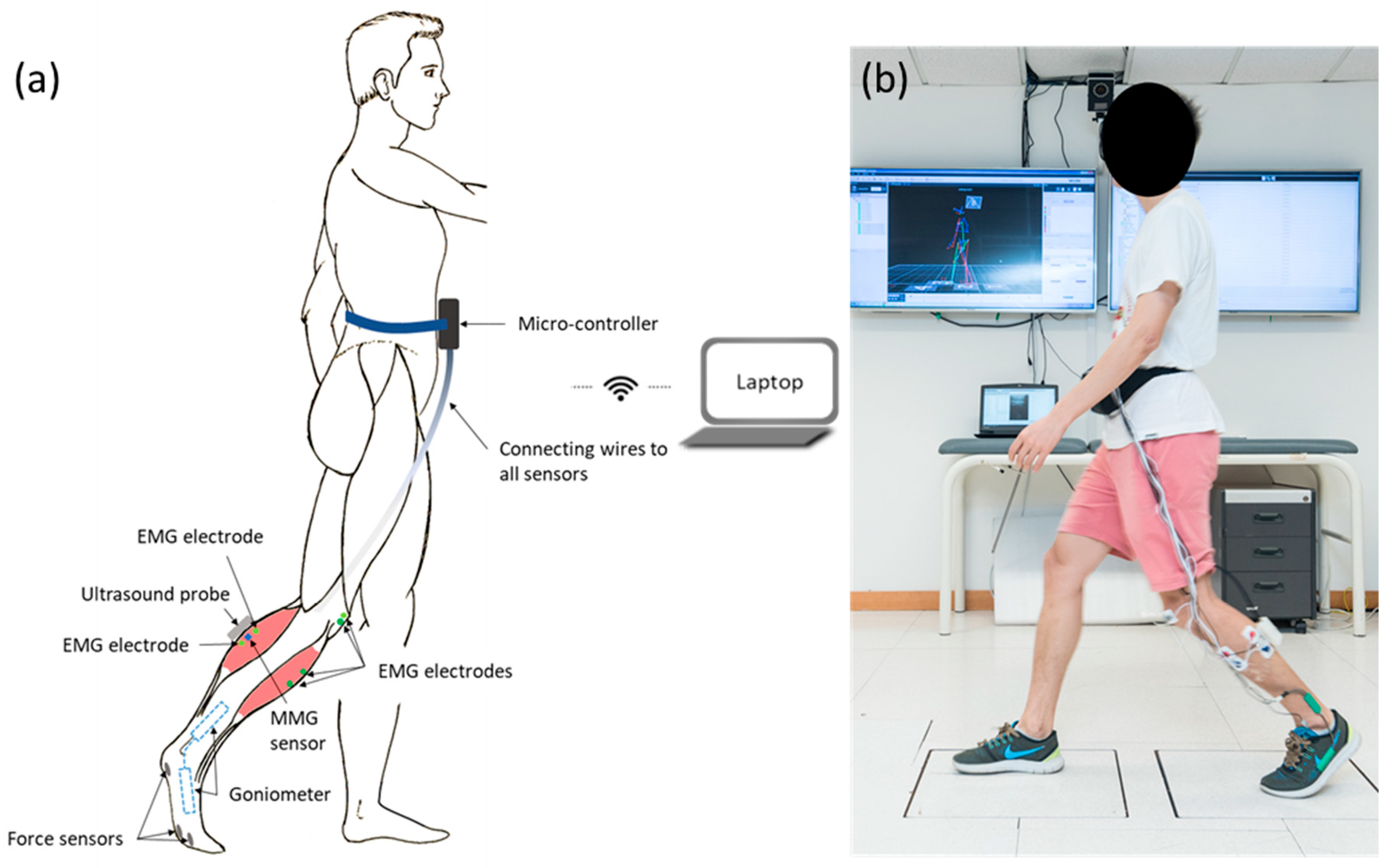
2.2. Wearable Motion Capture and Analysis System with Real-Time SMG (Mobile SMG System)
2.3. Experimental Procedure and Data Acquisition.
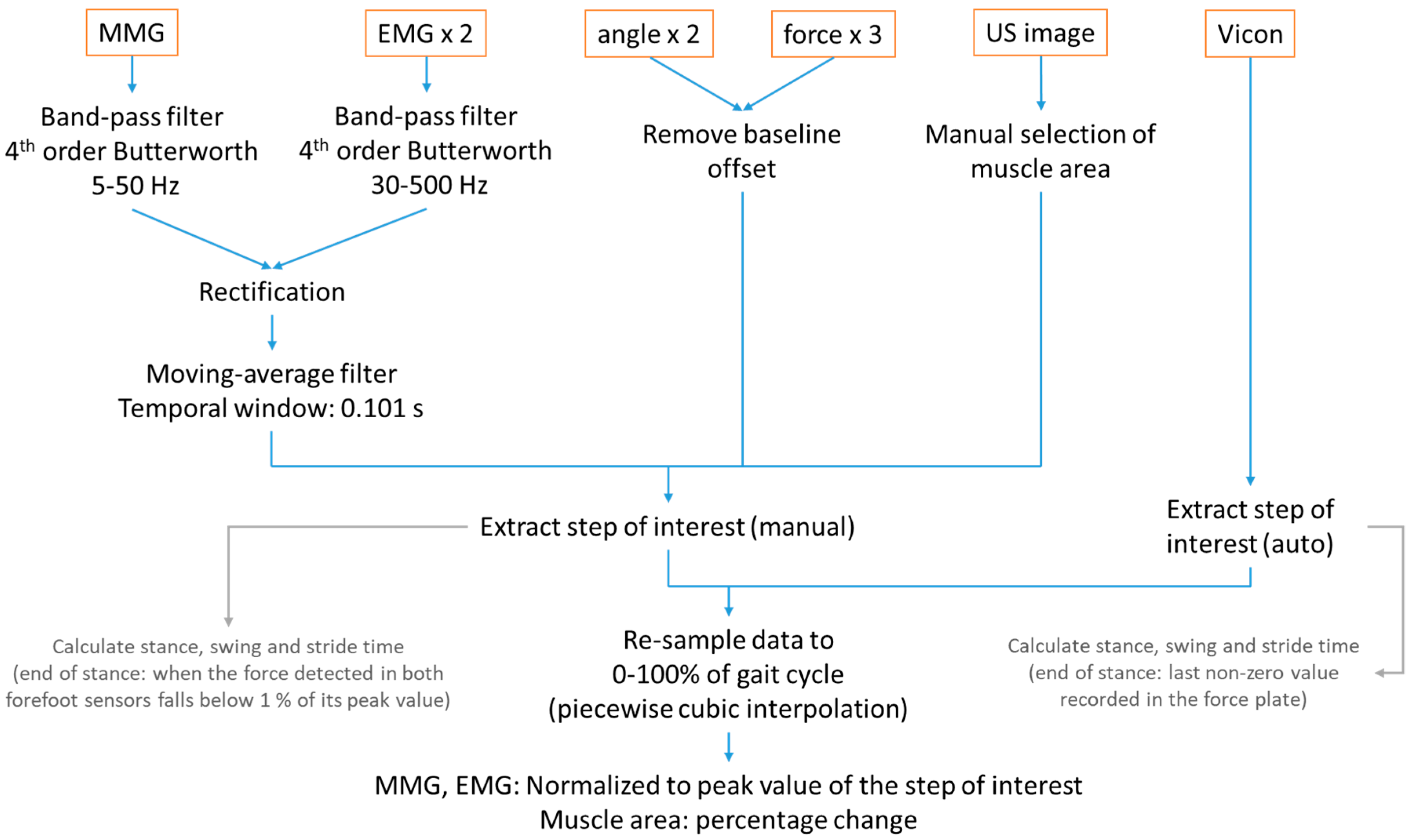
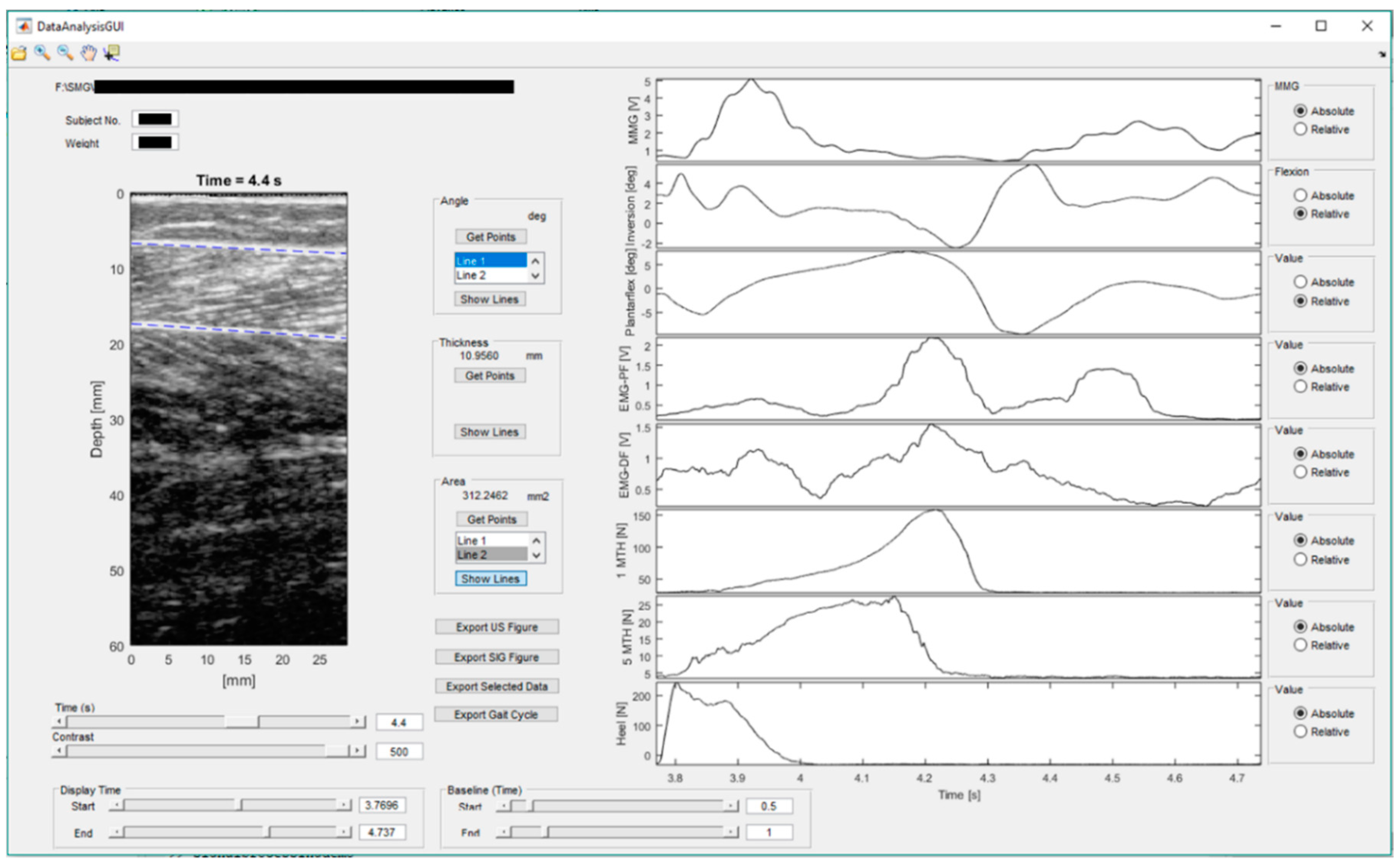
2.4. Signal Processing
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davies, B.L.; Hoffman, R.M.; Healey, K.; Zabad, R.; Kurz, M.J. Errors in the ankle plantarflexor force production are related to the gait deficits of individuals with multiple sclerosis. Hum. Move. Sci. 2017, 51, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Lai, A.K.; Lichtwark, G.A.; Schache, A.G.; Pandy, M.G. Differences in in-vivo muscle fascicle and tendinous tissue behaviour between the ankle plantarflexors during running. Scand. J. Med. Sci. Sports 2018, 28, 1828–1836. [Google Scholar] [CrossRef] [PubMed]
- Norris, E.S.; Wallmann, H.W. Static and dynamic balance after ankle plantarflexor fatigue in older adults. Phys. Occup. Ther. Geriatr. 2016, 34, 57–70. [Google Scholar] [CrossRef]
- Mañago, M.M.; Hebert, J.R.; Kittelson, J.; Schenkman, M. Contributions of Ankle, Knee, Hip, and Trunk Muscle Function to Gait Performance in People with Multiple Sclerosis: A Cross-Sectional Analysis. Phys. Ther. 2018, 98, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Gaudreault, N.; Gravel, D.; Nadeau, S. Evaluation of plantar flexion contracture contribution during the gait of children with Duchenne muscular dystrophy. J. Electromyogr. Kinesiol. 2009, 19, e180–e186. [Google Scholar] [CrossRef] [PubMed]
- Deandrea, S.; Lucenteforte, E.; Bravi, F.; Foschi, R.; La Vecchia, C.; Negri, E. Risk factors for falls in community-dwelling older people: A systematic review and meta-analysis. Epidemiology 2010, 21, 658–668. [Google Scholar] [CrossRef]
- Fujisawa, H.; Suzuki, H.; Nishiyama, T.; Suzuki, M. Comparison of ankle plantar flexor activity between double-leg heel raise and walking. J. Phys. Ther. Sci. 2015, 27, 1523–1526. [Google Scholar] [CrossRef] [Green Version]
- Beck, T.W.; Housh, T.J.; Johnson, G.O.; Cramer, J.T.; Weir, J.P.; Coburn, J.W.; Malek, M.H. Does the frequency content of the surface mechanomyographic signal reflect motor unit firing rates? A brief review. J. Electromyogr. Kinesiol. 2007, 17, 1–13. [Google Scholar] [CrossRef]
- Dick, T.J.; Nuckols, R.W.; Sawicki, G.S. Tuned or not? Ultrasound measurements of soleus fascicle dynamics during human walking with elastic ankle exoskeletons. In Proceedings of the 41st Annual Meeting of the American Society of Biomechanics, Boulder, CO, USA, 8–11 August 2017. [Google Scholar]
- Miyoshi, T.; Takagi, M.; Yimit, A.; Hagihara, Y.; Komeda, T. Task-Dependent Gastrocnemius Fiber Movement in Humans while Standing as Revealed by Ultrasound Images. In Proceedings of the ICSSE, Morioka, Japan, 6–8 July 2015; pp. 378–388. [Google Scholar]
- Aggeloussis, N.; Giannakou, E.; Albracht, K.; Arampatzis, A. Reproducibility of fascicle length and pennation angle of gastrocnemius medialis in human gait in vivo. Gait Posture 2010, 31, 73–77. [Google Scholar] [CrossRef]
- Lichtwark, G.; Bougoulias, K.; Wilson, A. Muscle fascicle and series elastic element length changes along the length of the human gastrocnemius during walking and running. J. Biomech. 2007, 40, 157–164. [Google Scholar] [CrossRef] [Green Version]
- Ramanathan, A.; Kiran, P.; Arnold, G.; Wang, W.; Abboud, R. Repeatability of the Pedar-X® in-shoe pressure measuring system. Foot Ankle Surg. 2010, 16, 70–73. [Google Scholar] [CrossRef] [PubMed]
- Slyper, R.; Hodgins, J.K. Action capture with accelerometers. In Proceedings of the 2008 ACM SIGGRAPH/Eurographics Symposium on Computer Animation, Dublin, Ireland, 7–9 July 2008; pp. 193–199. [Google Scholar]
- Riaz, Q.; Tao, G.; Krüger, B.; Weber, A. Motion reconstruction using very few accelerometers and ground contacts. Graph. Models 2015, 79, 23–38. [Google Scholar] [CrossRef]
- Vlasic, D.; Adelsberger, R.; Vannucci, G.; Barnwell, J.; Gross, M.; Matusik, W.; Popović, J. Practical motion capture in everyday surroundings. In Proceedings of the ACM Transactions on Graphics (TOG), San Diego, CA, USA, 5–9 August 2007; p. 35. [Google Scholar]
- Stollenwerk, K.; Müllers, J.; Müller, J.; Hinkenjann, A.; Krüger, B. Evaluating an Accelerometer-based System for Spine Shape Monitoring. In Proceedings of the Computational Science and Its Applications—ICCSA, Melbourne, Australia, 2–5 July 2018. [Google Scholar]
- Riaz, Q.; Vögele, A.; Krüger, B.; Weber, A. One small step for a man: Estimation of gender, age and height from recordings of one step by a single inertial sensor. Sensors 2015, 15, 31999–32019. [Google Scholar] [CrossRef] [PubMed]
- Kobsar, D.; Ferber, R. Wearable Sensor Data to Track Subject-Specific Movement Patterns Related to Clinical Outcomes Using a Machine Learning Approach. Sensors 2018, 18, 2828. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, H.; Claes, K.; De Wolf, C.; Van Damme, X.; Michel, A. A Machine Learning Approach to Automated Gait Analysis for the Noldus Catwalk System. IEEE Trans. Biomed. Eng. 2018, 65, 1133–1139. [Google Scholar] [PubMed]
- Ma, C.Z.-H.; Wong, D.W.-C.; Lam, W.K.; Wan, A.H.-P.; Lee, W.C.-C. Balance improvement effects of biofeedback systems with state-of-the-art wearable sensors: A systematic review. Sensors 2016, 16, 434. [Google Scholar] [CrossRef] [PubMed]
- Wan, A.H.; Wong, D.W.; Ma, C.Z.; Zhang, M.; Lee, W.C. Wearable vibrotactile biofeedback device allowing identification of different floor conditions for lower-limb amputees. Arch. Phys. Med. Rehabil. 2016, 97, 1210–1213. [Google Scholar] [CrossRef]
- Ma, C.Z.-H.; Zheng, Y.-P.; Lee, W.C.-C. Changes in gait and plantar foot loading upon using vibrotactile wearable biofeedback system in patients with stroke. Top. Stroke Rehabil. 2018, 25, 20–27. [Google Scholar] [CrossRef]
- Ma, C.Z.-H.; Wan, A.H.-P.; Wong, D.W.-C.; Zheng, Y.-P.; Lee, W.C.-C. A Vibrotactile and Plantar Force Measurement-Based Biofeedback System: Paving the Way towards Wearable Balance-Improving Devices. Sensors 2015, 15, 31709–31722. [Google Scholar] [CrossRef] [Green Version]
- Ma, C.Z.-H.; Lee, W.C.-C. A wearable vibrotactile biofeedback system improves balance control of healthy young adults following perturbations from quiet stance. Hum. Move. Sci. 2017, 55, 54–60. [Google Scholar] [CrossRef]
- Sienko, K.H.; Balkwill, M.D.; Oddsson, L.I.; Wall, C. The effect of vibrotactile feedback on postural sway during locomotor activities. J. Neuroeng. Rehabil. 2013, 10, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, C.Z.; Wan, A.H.; Wong, D.W.; Zheng, Y.-P.; Lee, W.C. Improving postural control using a portable plantar pressure-based vibrotactile biofeedback system. In Proceedings of the IEEE Conference on Biomedical Engineering and Sciences (IECBES), Kuala Lumpur, Malaysia, 8–10 December 2014; pp. 855–860. [Google Scholar]
- Huang, Q.-H.; Zheng, Y.-P.; Chena, X.; He, J.F.; Shi, J. A system for the synchronized recording of sonomyography, electromyography and joint angle. Open Biomed. Eng. J. 2007, 1, 77. [Google Scholar] [PubMed]
- Shi, J.; Zheng, Y.-P.; Huang, Q.-H.; Chen, X. Continuous monitoring of sonomyography, electromyography and torque generated by normal upper arm muscles during isometric contraction: Sonomyography assessment for arm muscles. IEEE Trans. Biomed. Eng. 2008, 55, 1191–1198. [Google Scholar] [PubMed]
- Zhou, G.-Q.; Zheng, Y.-P.; Zhou, P. Measurement of Gender Differences of Gastrocnemius Muscle and Tendon Using Sonomyography during Calf Raises: A Pilot Study. BioMed Res. Int. 2017, 2017, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, G.-Q.; Chan, P.; Zheng, Y.-P. Automatic measurement of pennation angle and fascicle length of gastrocnemius muscles using real-time ultrasound imaging. Ultrasonics 2015, 57, 72–83. [Google Scholar] [CrossRef]
- Wettenschwiler, P.D.; Stämpfli, R.; Lorenzetti, S.; Ferguson, S.J.; Rossi, R.M.; Annaheim, S. How reliable are pressure measurements with Tekscan sensors on the body surface of human subjects wearing load carriage systems? Int. J. Ind. Ergon. 2015, 49, 60–67. [Google Scholar] [CrossRef]
- Lempereur, M.; Brochard, S.; Leboeuf, F.; Rémy-Néris, O. Validity and reliability of 3D marker based scapular motion analysis: A systematic review. J. Biomech. 2014, 47, 2219–2230. [Google Scholar] [CrossRef] [PubMed]
- Barker, S.; Craik, R.; Freedman, W.; Herrmann, N.; Hillstrom, H. Accuracy, reliability, and validity of a spatiotemporal gait analysis system. Med. Eng. Phys. 2006, 28, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Plewa, K.; Samadani, A.; Chau, T. Comparing electro- and mechano-myographic muscle activation patterns in self-paced pediatric gait. J. Electromyogr. Kinesiol. 2017, 36, 73–80. [Google Scholar] [CrossRef]
- Richards, J. The Comprehensive Textbook of Clinical Biomechanics; Elsevier Health Sciences: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Plewa, K. Analysis and Interpretation of Lower Limb Mechanomyographic Signals in Human Gait. Ph.D. Thesis, University of Toronto, Toronto, ON, Canada, 2018. [Google Scholar]
- Elhadi, M.M.O.; Ma, C.Z.; Wong, D.W.C.; Wan, A.H.P.; Lee, W.C.C. Comprehensive gait analysis of healthy older adults who have undergone long-distance walking. J. Aging Phys. Act. 2017, 25, 367–377. [Google Scholar] [CrossRef]
- Elhadi, M.M.O.; Ma, C.Z.-H.; Lam, W.K.; Lee, W.C.-C. Biomechanical Approach in Facilitating Long-Distance Walking of Elderly People Using Footwear Modifications. Gait Posture 2018, 54, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Jaskólski, A.; Andrzejewska, R.; Marusiak, J.; Kisiel-Sajewicz, K.; Jaskólska, A. Similar response of agonist and antagonist muscles after eccentric exercise revealed by electromyography and mechanomyography. J. Electromyogr. Kinesiol. 2007, 17, 568–577. [Google Scholar] [CrossRef] [PubMed]
 : significantly high correlation between the Vicon and mobile SMG systems;
: significantly high correlation between the Vicon and mobile SMG systems;  : significant changes in trend in consecutive 5% intervals;
: significant changes in trend in consecutive 5% intervals;  : significantly high correlation in intraclass correlation coefficient (ICC) among three trials; The bolded orange line illustrates the averaged data measured by the mobile SMG system; The bolded blue line illustrates the averaged data measured by the Vicon system; The thin dashed line illustrates the standard deviation (SD) of each corresponding bold line.
: significantly high correlation in intraclass correlation coefficient (ICC) among three trials; The bolded orange line illustrates the averaged data measured by the mobile SMG system; The bolded blue line illustrates the averaged data measured by the Vicon system; The thin dashed line illustrates the standard deviation (SD) of each corresponding bold line.
 : significantly high correlation between the Vicon and mobile SMG systems;
: significantly high correlation between the Vicon and mobile SMG systems;  : significant changes in trend in consecutive 5% intervals;
: significant changes in trend in consecutive 5% intervals;  : significantly high correlation in intraclass correlation coefficient (ICC) among three trials; The bolded orange line illustrates the averaged data measured by the mobile SMG system; The bolded blue line illustrates the averaged data measured by the Vicon system; The thin dashed line illustrates the standard deviation (SD) of each corresponding bold line.
: significantly high correlation in intraclass correlation coefficient (ICC) among three trials; The bolded orange line illustrates the averaged data measured by the mobile SMG system; The bolded blue line illustrates the averaged data measured by the Vicon system; The thin dashed line illustrates the standard deviation (SD) of each corresponding bold line.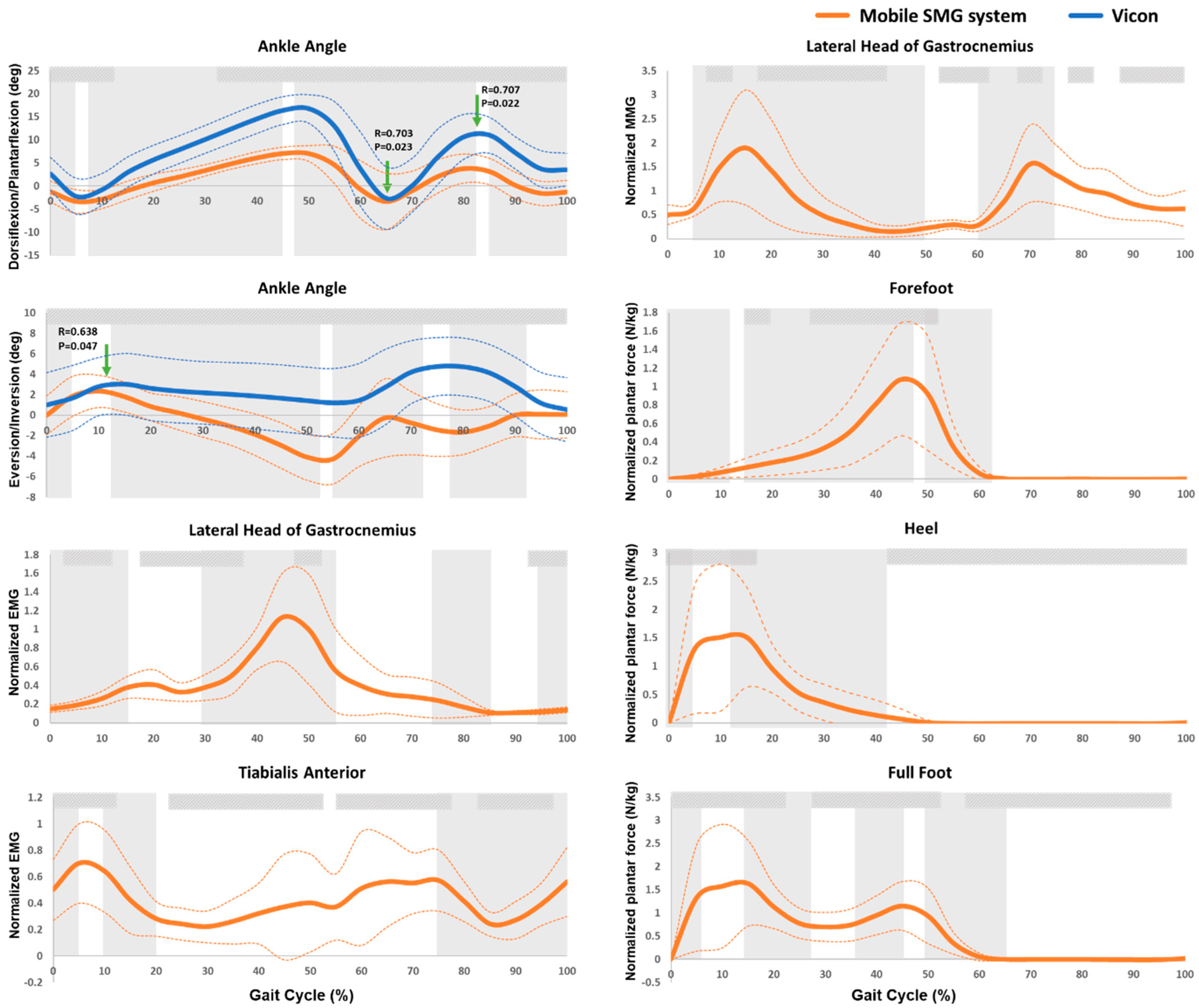
 : significant changes in trend in consecutive 5% intervals;
: significant changes in trend in consecutive 5% intervals;  : significantly high correlation in the intraclass correlation coefficient (ICC) among three trials.
: significantly high correlation in the intraclass correlation coefficient (ICC) among three trials.
 : significant changes in trend in consecutive 5% intervals;
: significant changes in trend in consecutive 5% intervals;  : significantly high correlation in the intraclass correlation coefficient (ICC) among three trials.
: significantly high correlation in the intraclass correlation coefficient (ICC) among three trials.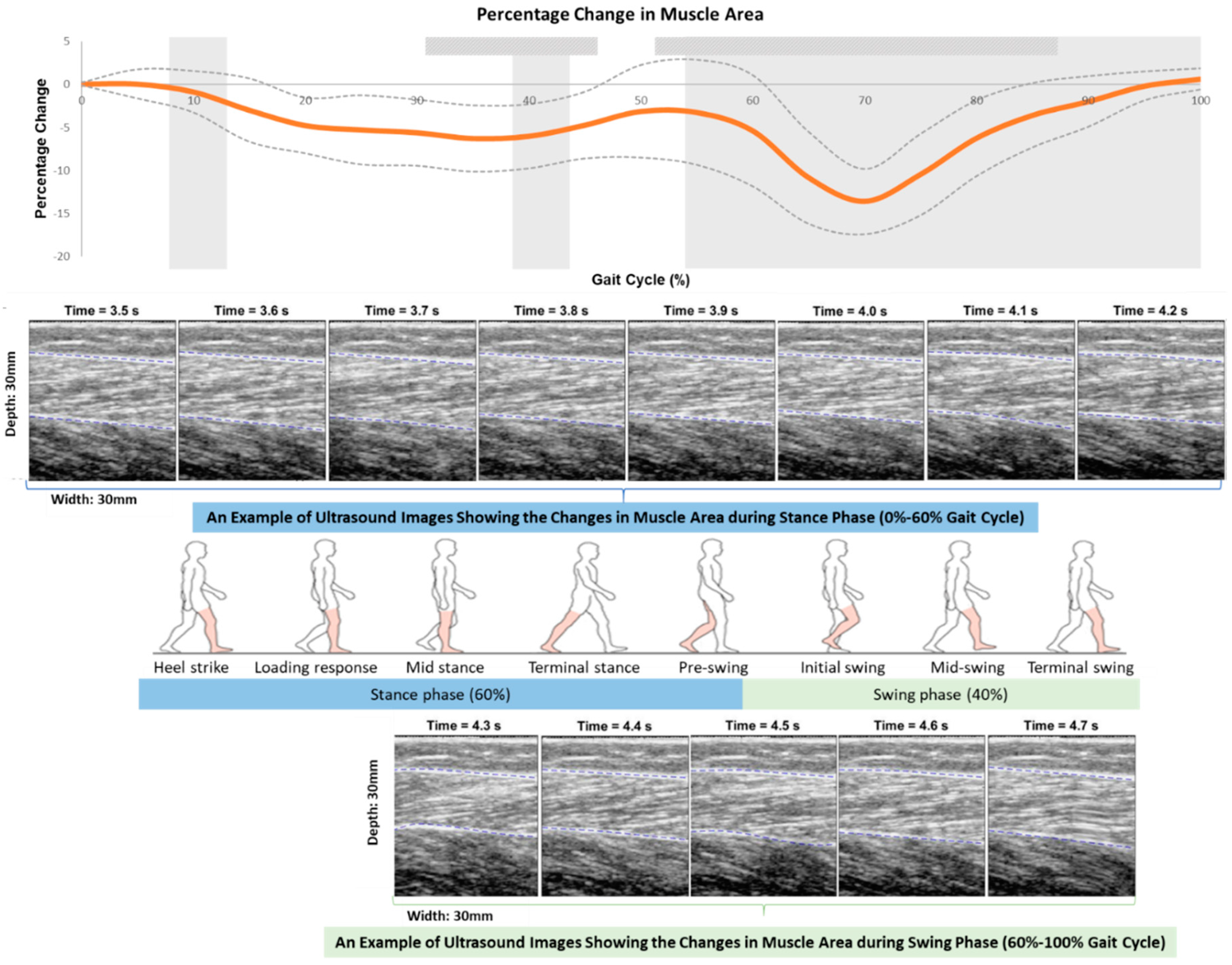
| Gait Cycle | % Muscle Area | MMG | In-/E-Version | Plantar-/Dorsi-Flexion | EMG_ Plantarflexor | EMG_ Dorsiflexor | Plantar Force | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % | ICC | p Value | ICC | p Value | ICC | p Value | ICC | p Value | ICC | p Value | ICC | p Value | ICC | p Value |
| 0 | NA | NA | −0.017 | 0.484 | 0.761 | 0.003 | 0.647 | 0.013 | 0.427 | 0.074 | 0.712 | 0.006 | NA | NA |
| 5 | 0.041 | 0.415 | 0.321 | 0.134 | 0.711 | 0.006 | 0.524 | 0.038 | 0.662 | 0.011 | 0.84 | 0.001 | 0.919 | <0.001 |
| 10 | −0.496 | 0.983 | 0.803 | 0.001 | 0.655 | 0.011 | 0.522 | 0.038 | 0.754 | 0.003 | 0.623 | 0.016 | 0.988 | <0.001 |
| 15 | 0.195 | 0.221 | 0.456 | 0.061 | 0.709 | 0.006 | 0.432 | 0.072 | 0.408 | 0.083 | 0.347 | 0.117 | 0.913 | <0.001 |
| 20 | −0.243 | 0.747 | 0.81 | 0.001 | 0.561 | 0.028 | 0.258 | 0.181 | 0.744 | 0.004 | 0.386 | 0.094 | 0.524 | 0.038 |
| 25 | −0.448 | 0.999 | 0.667 | 0.01 | 0.501 | 0.045 | 0.4 | 0.087 | 0.831 | 0.001 | 0.889 | <0.001 | 0.471 | 0.056 |
| 30 | −0.249 | 0.806 | 0.589 | 0.022 | 0.54 | 0.033 | 0.487 | 0.05 | 0.877 | <0.001 | 0.695 | 0.007 | 0.768 | 0.002 |
| 35 | 0.512 | 0.027 | 0.731 | 0.004 | 0.653 | 0.012 | 0.621 | 0.016 | 0.978 | <0.001 | 0.737 | 0.004 | 0.915 | <0.001 |
| 40 | 0.521 | 0.041 | 0.529 | 0.036 | 0.828 | 0.001 | 0.775 | 0.002 | 0.413 | 0.081 | 0.592 | 0.021 | 0.799 | 0.001 |
| 45 | 0.499 | 0.042 | 0.316 | 0.138 | 0.885 | <0.001 | 0.741 | 0.004 | −0.204 | 0.742 | 0.816 | 0.001 | 0.803 | 0.001 |
| 50 | 0.378 | 0.087 | 0.359 | 0.11 | 0.868 | <0.001 | 0.548 | 0.031 | 0.705 | 0.006 | 0.79 | 0.002 | 0.766 | 0.003 |
| 55 | 0.607 | 0.016 | 0.593 | 0.021 | 0.857 | <0.001 | 0.734 | 0.004 | −0.105 | 0.605 | 0.448 | 0.065 | 0.105 | 0.331 |
| 60 | 0.644 | 0.018 | 0.547 | 0.032 | 0.909 | <0.001 | 0.95 | <0.001 | −0.018 | 0.485 | 0.943 | <0.001 | 0.646 | 0.013 |
| 65 | 0.416 | 0.045 | 0.01 | 0.448 | 0.914 | <0.001 | 0.914 | <0.001 | 0.353 | 0.113 | 0.872 | <0.001 | 0.813 | 0.001 |
| 70 | 0.421 | 0.049 | 0.615 | 0.017 | 0.88 | <0.001 | 0.929 | <0.001 | 0.075 | 0.366 | 0.744 | 0.004 | 0.825 | 0.001 |
| 75 | 0.658 | 0.008 | 0.05 | 0.397 | 0.926 | <0.001 | 0.938 | <0.001 | 0.001 | 0.459 | 0.919 | <0.001 | 0.521 | 0.039 |
| 80 | 0.688 | 0.009 | 0.589 | 0.022 | 0.898 | <0.001 | 0.933 | <0.001 | −0.032 | 0.504 | 0.263 | 0.177 | 0.714 | 0.006 |
| 85 | 0.306 | 0.169 | −0.072 | 0.559 | 0.821 | 0.001 | 0.898 | <0.001 | 0.019 | 0.436 | 0.685 | 0.008 | 0.717 | 0.005 |
| 90 | 0.317 | 0.147 | 0.554 | 0.03 | 0.803 | 0.001 | 0.743 | 0.004 | 0.266 | 0.175 | 0.844 | 0.001 | 0.8 | 0.001 |
| 95 | 0.048 | 0.399 | 0.658 | 0.011 | 0.853 | <0.001 | 0.593 | 0.021 | 0.569 | 0.026 | 0.659 | 0.011 | 0.843 | 0.001 |
| 100 | −0.253 | 0.835 | 0.799 | 0.001 | 0.88 | <0.001 | 0.665 | 0.01 | 0.583 | 0.023 | 0.463 | 0.059 | 0.461 | 0.059 |
| Sig. ICC (mean ± SD) | 0.552 ± 0.101 | 0.653 ± 0.102 | 0.782 ± 0.132 | 0.746 ± 0.157 | 0.745 ± 0.135 | 0.773 ± 0.107 | 0.781 ± 0.129 | |||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, C.Z.-H.; Ling, Y.T.; Shea, Q.T.K.; Wang, L.-K.; Wang, X.-Y.; Zheng, Y.-P. Towards Wearable Comprehensive Capture and Analysis of Skeletal Muscle Activity during Human Locomotion. Sensors 2019, 19, 195. https://doi.org/10.3390/s19010195
Ma CZ-H, Ling YT, Shea QTK, Wang L-K, Wang X-Y, Zheng Y-P. Towards Wearable Comprehensive Capture and Analysis of Skeletal Muscle Activity during Human Locomotion. Sensors. 2019; 19(1):195. https://doi.org/10.3390/s19010195
Chicago/Turabian StyleMa, Christina Zong-Hao, Yan To Ling, Queenie Tsung Kwan Shea, Li-Ke Wang, Xiao-Yun Wang, and Yong-Ping Zheng. 2019. "Towards Wearable Comprehensive Capture and Analysis of Skeletal Muscle Activity during Human Locomotion" Sensors 19, no. 1: 195. https://doi.org/10.3390/s19010195
APA StyleMa, C. Z.-H., Ling, Y. T., Shea, Q. T. K., Wang, L.-K., Wang, X.-Y., & Zheng, Y.-P. (2019). Towards Wearable Comprehensive Capture and Analysis of Skeletal Muscle Activity during Human Locomotion. Sensors, 19(1), 195. https://doi.org/10.3390/s19010195






