Microfluidic Cultivation and Laser Tweezers Raman Spectroscopy of E. coli under Antibiotic Stress
Abstract
:1. Introduction
2. Materials and Methods
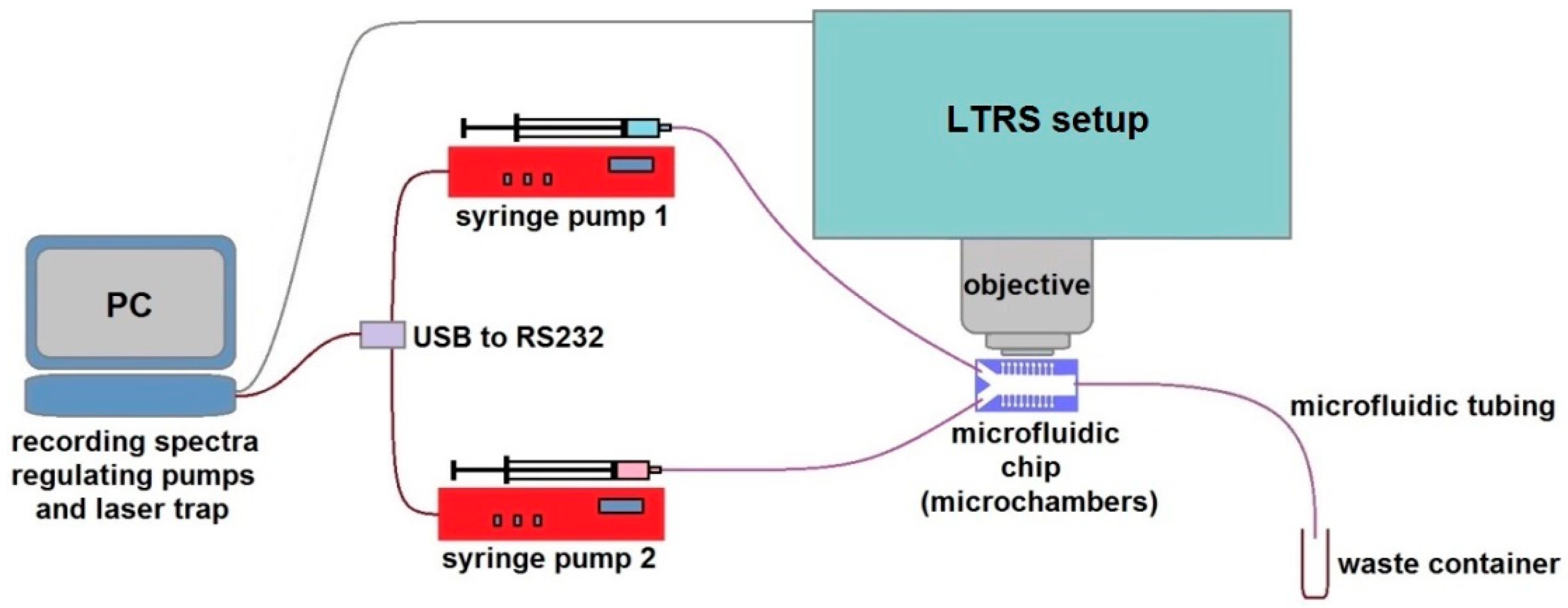
2.1. Optofluidic System
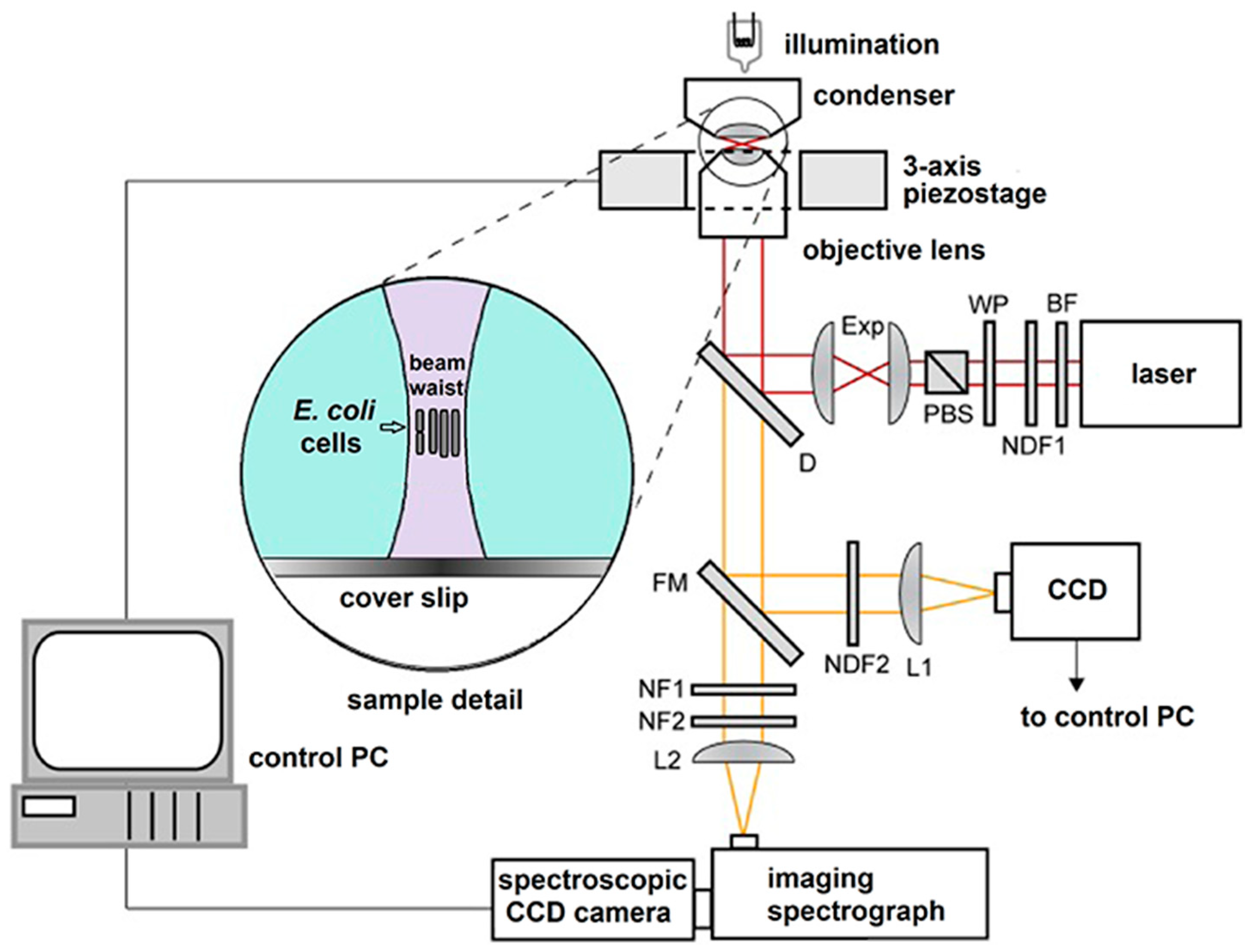
2.2. LTRS System
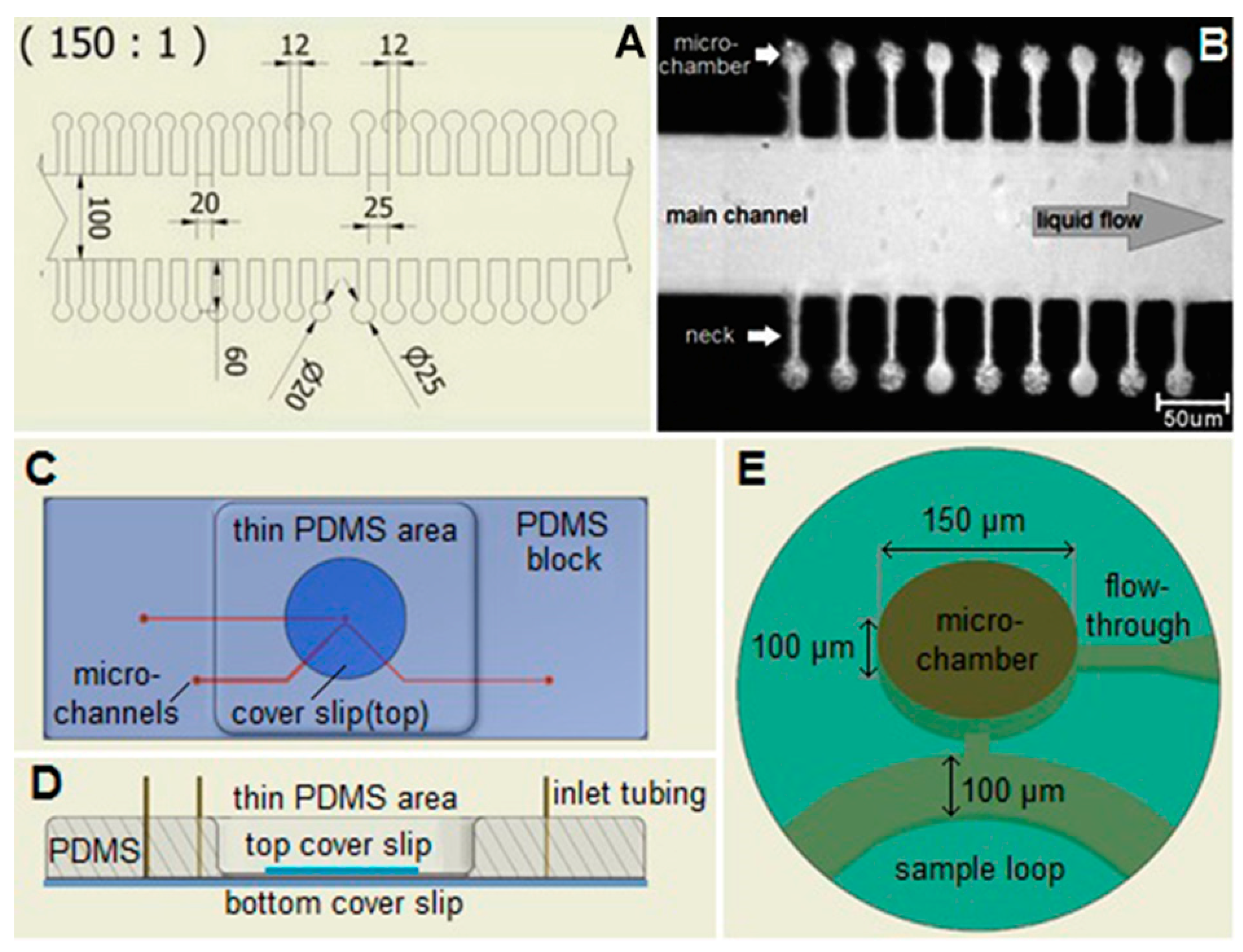
2.3. Microfluidic Chips
2.4. Bacterial Samples: Strain and Growth Condition
2.5. Optical TRAPPING Procedure for Time-Lapse Imaging Experiments
2.6. LTRS Protocol for Raman Characterization of E. coli with 785 nm Excitation
2.7. Raman Spectroscopic Characterization of E. coli in the Bulk with 532 nm Excitation
2.8. Processing and Analysis of Raman Spectral Data
3. Results and Discussion
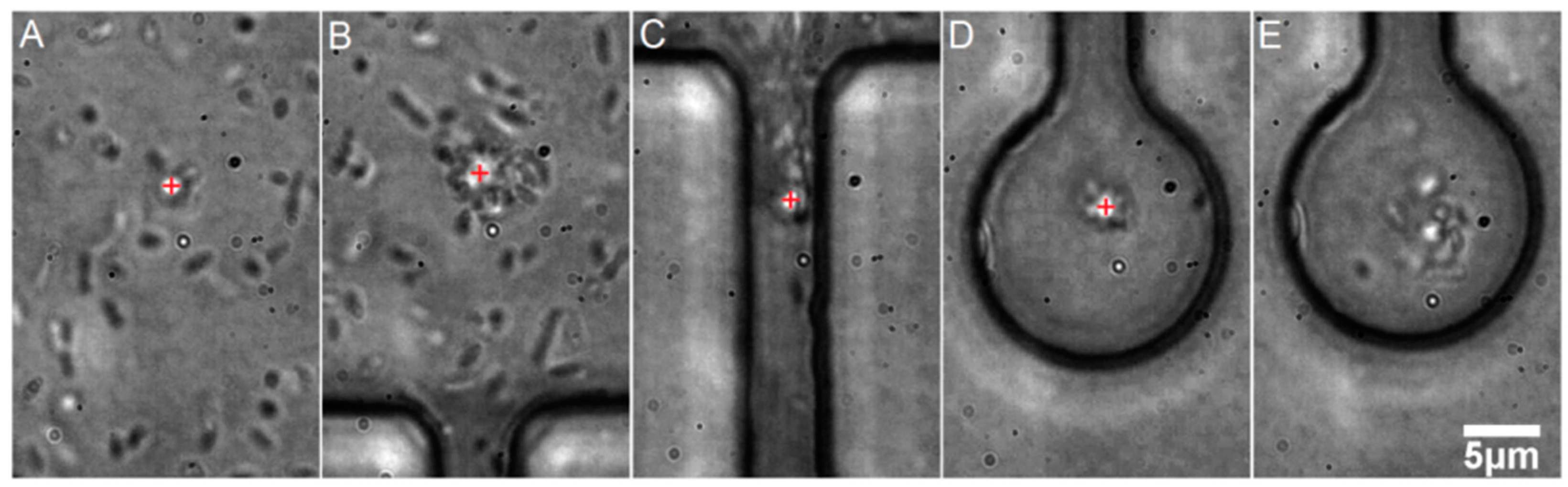
3.1. Optical Trapping in Microfluidic Environment
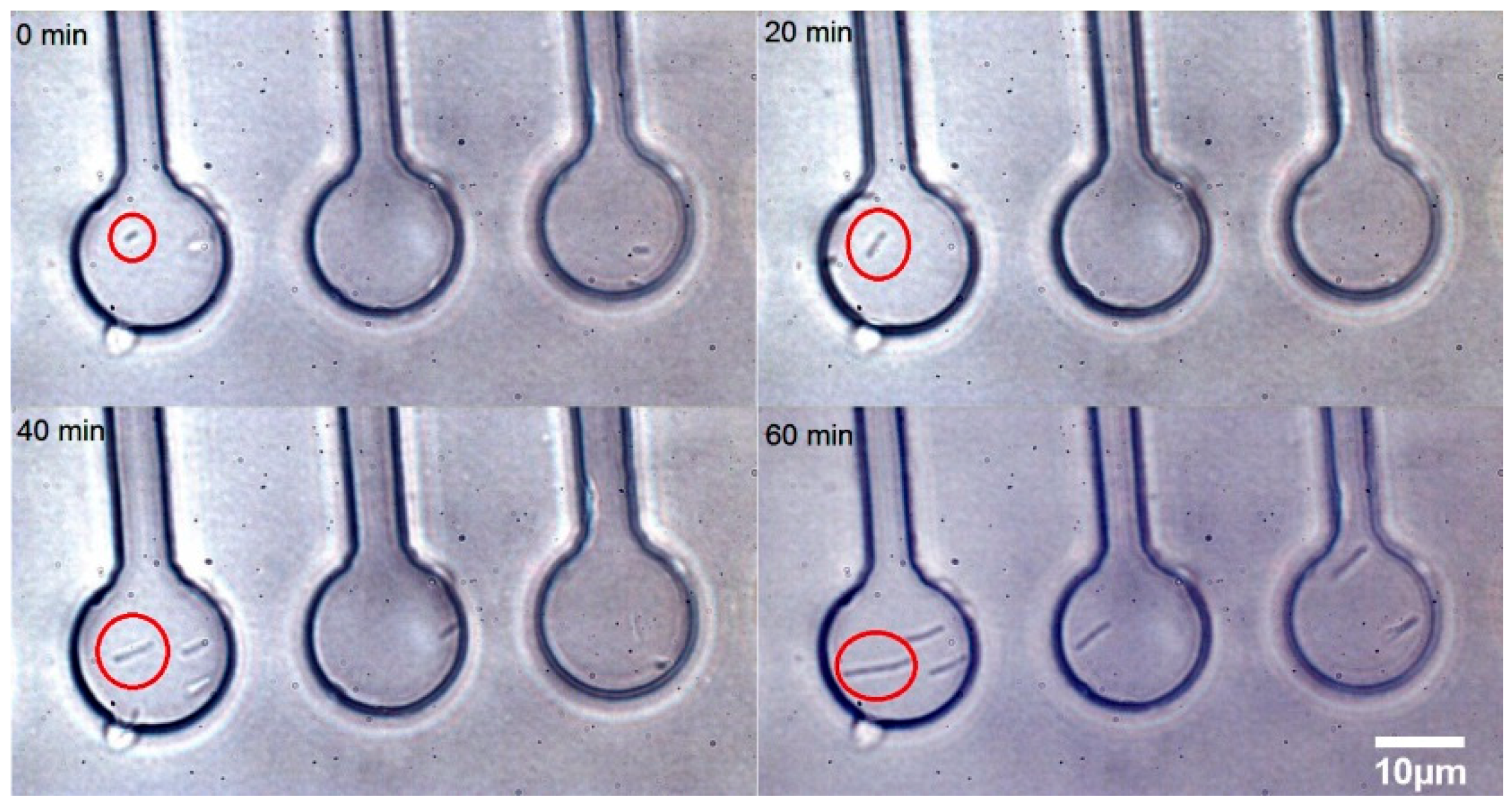
3.2. Time Lapse Observation of E. coli Growth in Microchambers under Antibiotic Stress
3.3. Experiments with LTRS of E. coli Cells with 785 nm Wavelength for Trapping and Raman Excitation
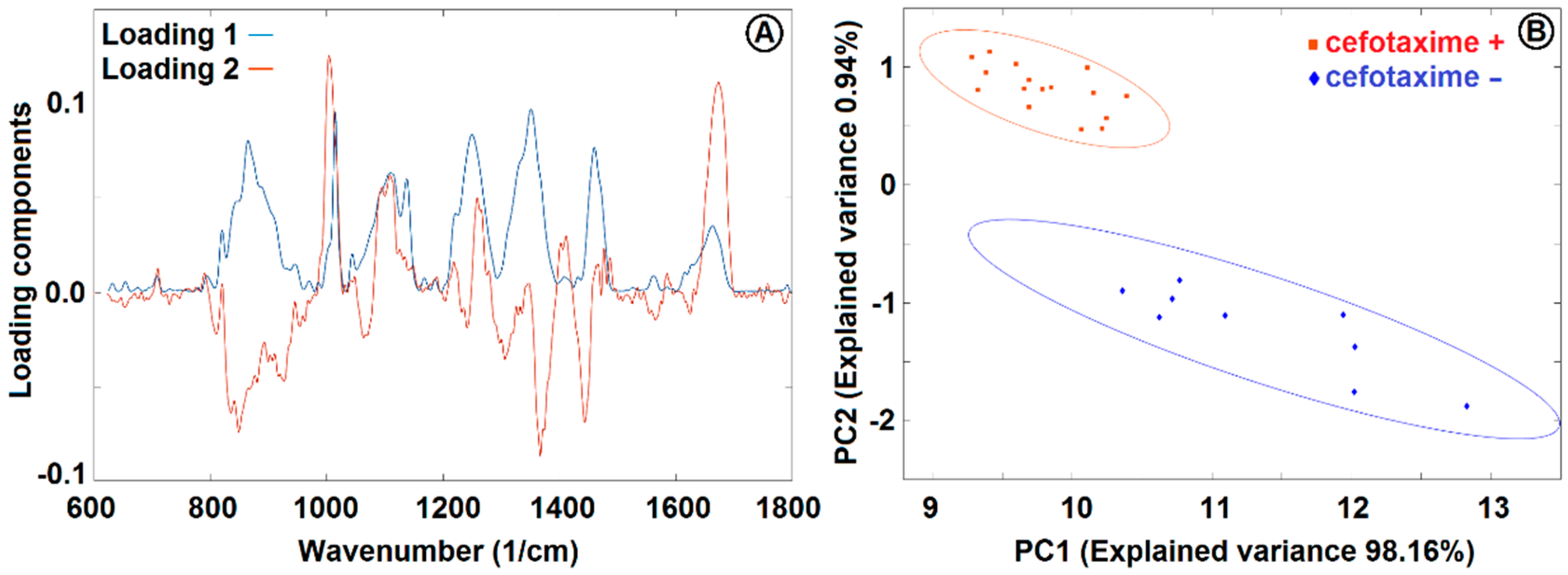
3.4. Raman Microspectroscopy of E. coli Cells with 532 nm Excitation
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Liberale, C.; Cojoc, G.; Bragheri, F.; Minzioni, P.; Perozziello, G.; La Rocca, R.; Ferrara, L.; Rajamanickam, V.; Di Fabrizio, E.; Cristiani, I. Integrated microfluidic device for single-cell trapping and spectroscopy. Sci. Rep. 2013, 3, 1258. [Google Scholar] [CrossRef] [PubMed]
- Redding, B.; Schwab, M.J.; Pan, Y. Review: Raman spectroscopy of optically trapped single biological micro-particles. Sensors 2015, 15, 19021–19046. [Google Scholar] [CrossRef] [PubMed]
- Gilany, K.; Moazeni-Pourasil, R.S.; Jafarzadeh, N.; Savadi-Shiraz, A. Metabolomics fingerprinting of the human seminal plasma of asthenozoospermic patients. Mol. Reprod. Dev. 2014, 81, 84–86. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Hasty, C.E.; Watson, P.A.; Wicksted, J.P.; Stith, R.D.; March, W.F. Analysis of metabolites in aqueous solutions by using laser Raman spectroscopy. Appl. Opt. 1993, 32, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Rösch, P.; Harz, M.; Schmitt, M.; Peschke, K.-D.; Ronneberger, O.; Burkhardt, H.; Motzkus, H.-W.; Lankers, M.; Hofer, S.; Thiele, H.; et al. Chemotaxonomic identification of single bacteria by micro-Raman spectroscopy: Application to clean-room-relevant biological contaminations. Appl. Environ. Microbiol. 2005, 71, 1626–1637. [Google Scholar] [CrossRef] [PubMed]
- Willemse-Erix, D.F.M.; Scholtes-Timmerman, M.J.; Jachtenberg, J.-W.; van Leeuwen, W.B.; Horst-Kreft, D.; Schut, T.C.B.; Deurenberg, R.H.; Puppels, G.J.; van Belkum, A.; Vos, M.C.; et al. Optical fingerprinting in bacterial epidemiology: Raman spectroscopy as a real-time typing method. Clin. Microbiol. 2009, 47, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Stöckel, S.; Kirchhoff, J.; Neugebauer, U.; Rösch, P.; Popp, J. The application of Raman spectroscopy for the detection and identification of microorganisms. J. Raman Spectrosc. 2016, 47, 89–109. [Google Scholar] [CrossRef]
- Xie, C.; Goodman, C.; Dinno, M.A.; Li, Y.Q. Real-time Raman spectroscopy of optically trapped living cells and organelles. Opt. Express 2004, 25, 6208–6214. [Google Scholar] [CrossRef]
- Xie, C.; Chen, D.; Li, Y.-Q. Raman sorting and identification of single living micro-organisms with optical tweezers. Opt. Lett. 2005, 30, 1800–1802. [Google Scholar] [CrossRef] [PubMed]
- Neuman, K.C.; Chadd, E.H.; Liou, G.F.; Bergman, K.; Block, S.M. Characterization of photodamage to Escherichia coli in optical traps. Biophys. J. 1999, 77, 2856–2863. [Google Scholar] [CrossRef]
- Chan, J.W.; Winhold, H.; Corzett, M.H.; Ulloa, J.; Cosman, M.; Balhorn, R.; Huser, T. Monitoring dynamic protein expression in living E. coli bacterial cells by laser tweezers Raman spectroscopy. Cytom. Part A 2007, 71A, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Hamon, M.; Jambovane, S. Microfluidics for antibiotic susceptibility and toxicity testing. Bioengineering 2016, 3, 25. [Google Scholar] [CrossRef] [PubMed]
- Assmann, C.; Kirchhoff, J.; Beleites, C.; Hey, J.; Kostudis, S.; Pfister, W.; Schlattmann, P.; Popp, J.; Neugebauer, U. Identification of vancomycin interaction with Enterococcus faecalis within 30 min of interaction time using Raman spectroscopy. Anal. Bioanal. Chem. 2015, 407, 8343–8352. [Google Scholar] [CrossRef] [PubMed]
- Schröder, U.; Kirchhoff, J.; Hübner, U.; Mayer, G.; Glaser, U.; Henkel, T.; Pfister, W.; Fritzsche, W.; Popp, J.; Neugebauer, U. On-Chip spectroscopic assessment of microbial susceptibility to antibiotics within 3½ hours. J. Biophotonic 2017, 10, 1547–1557. [Google Scholar] [CrossRef] [PubMed]
- Schröder, U.-C.; Beleites, C.; Assmann, C.; Glaser, U.; Hübner, U.; Pfister, W.; Fritzsche, W.; Popp, J.; Neugebauer, U. Detection of vancomycin resistances in enterococci within 3½ hours. Sci. Rep. 2015, 5, 8271. [Google Scholar] [CrossRef] [PubMed]
- Kirchhoff, J.; Glaser, U.; Bohnert, J.A.; Pletz, M.; Popp, J.; Neugebauer, U. Simple ciprofloxacin resistance test and determination of minimal inhibitory concentration (MIC) within two hours using Raman spectroscopy. Anal. Chem. 2018, 90, 1811–1818. [Google Scholar] [CrossRef] [PubMed]
- Schröder, U.-C.; Ramoji, A.; Glaser, U.; Sachse, S.; Leiterer, C.; Cszaki, A.; Huebner, U.; Fritzsche, W.; Pfister, W.; Bauer, M.; et al. Combined dielectrophoresis-Raman setup for the classification of pathogens recovered from the urinary tract. Anal. Chem. 2013, 85, 10717–10724. [Google Scholar] [CrossRef] [PubMed]
- Schröder, U.-C.; Bokeloh, F.; O’Sullivan, M.; Glaser, U.; Wolf, K.; Pfister, W.; Popp, J.; Ducreé, J.; Neugebauer, U. Rapid, culture-independent, optical diagnostics of centrifugally captured bacteria from urine samples. Biomicrofluidics 2015, 9, 044118. [Google Scholar] [CrossRef] [PubMed]
- Pilát, Z.; Jonáš, A.; Ježek, J.; Zemánek, P. Effects of infrared optical trapping on Saccharomyces cerevisiae in a microfluidic system. Sensors 2017, 17, 2640. [Google Scholar] [CrossRef] [PubMed]
- Jonáš, A.; Pilát, Z.; Ježek, J.; Bernatová, S.; Fořt, T.; Zemánek, P.; Aas, M.; Kiraz, A. Thermal tuning of spectral emission from optically trapped liquid-crystal droplet resonators. JOSA B 2017, 34, 1855–1864. [Google Scholar] [CrossRef]
- Samek, O.; Jonáš, A.; Pilát, Z.; Zemánek, P.; Nedbal, L.; Tříska, J.; Kotas, P.; Trtílek, M. Raman Microspectroscopy of individual algal Cells: Sensing unsaturation of storage lipids in vivo. Sensors 2010, 10, 8635–8651. [Google Scholar] [CrossRef] [PubMed]
- Pilát, Z.; Bernatová, S.; Ježek, J.; Šerý, M.; Samek, O.; Zemánek, P.; Nedbal, L.; Trtílek, M. Raman microspectroscopy of algal lipid Bodies: Beta-carotene as a sensor. SPIE Proc. 2011, 8306, 83060L. [Google Scholar] [CrossRef]
- Pilát, Z.; Bernatová, S.; Ježek, J.; Šerý, M.; Samek, O.; Zemánek, P.; Nedbal, L.; Trtílek, M. Raman microspectroscopy of algal lipid Bodies: Beta-carotene quantification. J. Appl. Phycol. 2012, 24, 541–546. [Google Scholar] [CrossRef]
- Bernatová, S.; Samek, O.; Pilát, Z.; Šerý, M.; Ježek, J.; Jákl, P.; Šiler, M.; Krzyžánek, V.; Zemánek, P.; Holá, V.; et al. Following the mechanisms of bacteriostatic versus bactericidal action using Raman spectroscopy. Molecules 2013, 18, 13188–13199. [Google Scholar] [CrossRef] [PubMed]
- Samek, O.; Mlynáriková, K.; Bernatová, S.; Ježek, J.; Krzyžánek, V.; Šiler, M.; Zemánek, P.; Růžička, F.; Holá, V.; Mahelová, M. Candida parapsilosis biofilm identification by Raman spectroscopy. Int. J. Mol. Sci. 2014, 15, 23924–23935. [Google Scholar] [CrossRef] [PubMed]
- Samek, O.; Bernatová, S.; Ježek, J.; Šiler, M.; Šerý, M.; Krzyžánek, V.; Hrubanová, K.; Zemánek, P.; Holá, V.; Růžička, F. Identification of individual biofilm-forming bacterial cells using Raman tweezers. J. Biomed. Opt. 2015, 20, 051038. [Google Scholar] [CrossRef] [PubMed]
- Mlynáriková, K.; Samek, O.; Bernatová, S.; Růžička, F.; Ježek, J.; Hároniková, A.; Šiler, M.; Zemánek, P.; Holá, V. Influence of culture media on microbial fingerprints using Raman spectroscopy. Sensors 2015, 15, 29635–29647. [Google Scholar] [CrossRef] [PubMed]
- Petrov, D.V. Raman spectroscopy of optically trapped particles. J. Opt. A Pure Appl. Opt. 2007, 9, 139–156. [Google Scholar] [CrossRef]
- Jonáš, A.; Zemánek, P. Light at work: The use of optical forces for particle manipulation, sorting, and analysis. Electrophoresis 2008, 29, 4813–4851. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Whitesides, G.M. Soft lithography. Ann. Rev. Mater. Sci. 1998, 28, 153–184. [Google Scholar] [CrossRef]
- Pilát, Z.; Ježek, J.; Šerý, M.; Trtílek, M.; Nedbal, L.; Zemánek, P. Optical trapping of microalgae at 735–1064 nm: Photodamage assessment. J. Photochem. Photobiol. B Biol. 2013, 121, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Brandt, N.N.; Brovko, O.O.; Chikishev, A.Y.; Paraschuk, O.D. Optimization of the Rolling-Circle Filter for Raman Background Subtraction. Appl. Spectrosc. 2006, 60, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Yoo, J.; Lee, M.; Kim, E.-G.; Lee, J.S.; Lee, S.; Joo, S.; Song, S.H.; Kim, E.-C.; Lee, J.C.; et al. A rapid antimicrobial susceptibility test based on single-cell morphological analysis. Sci. Transl. Med. 2014, 6, 267ra174. [Google Scholar] [CrossRef] [PubMed]
- Long, D.A. The Raman Effect: A Unified Treatment of the Theory of Raman Scattering by Molecules; John Wiley & Sons Ltd.: Chichester, UK, 2002; pp. 21–22, ISBNs 0-471-49028-8 (Hardback), 0-470-84576-7 (Electronic). [Google Scholar]
- Harz, M.; Rosch, P.; Peschke, K.-D.; Ronneberger, O.; Burkhardt, H.; Popp, J. Micro-Raman spectroscopic identification of bacterial cells of the genus Staphylococcus and dependence on their cultivation conditions. Analyst 2005, 130, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Dutta, R.K.; Sharma, P.K.; Pandey, A.C. Surface enhanced Raman spectra of Escherichia coli cells using ZnO nanoparticles. Dig. J. Nanomater. Biostruct. 2009, 4, 83–87. [Google Scholar]
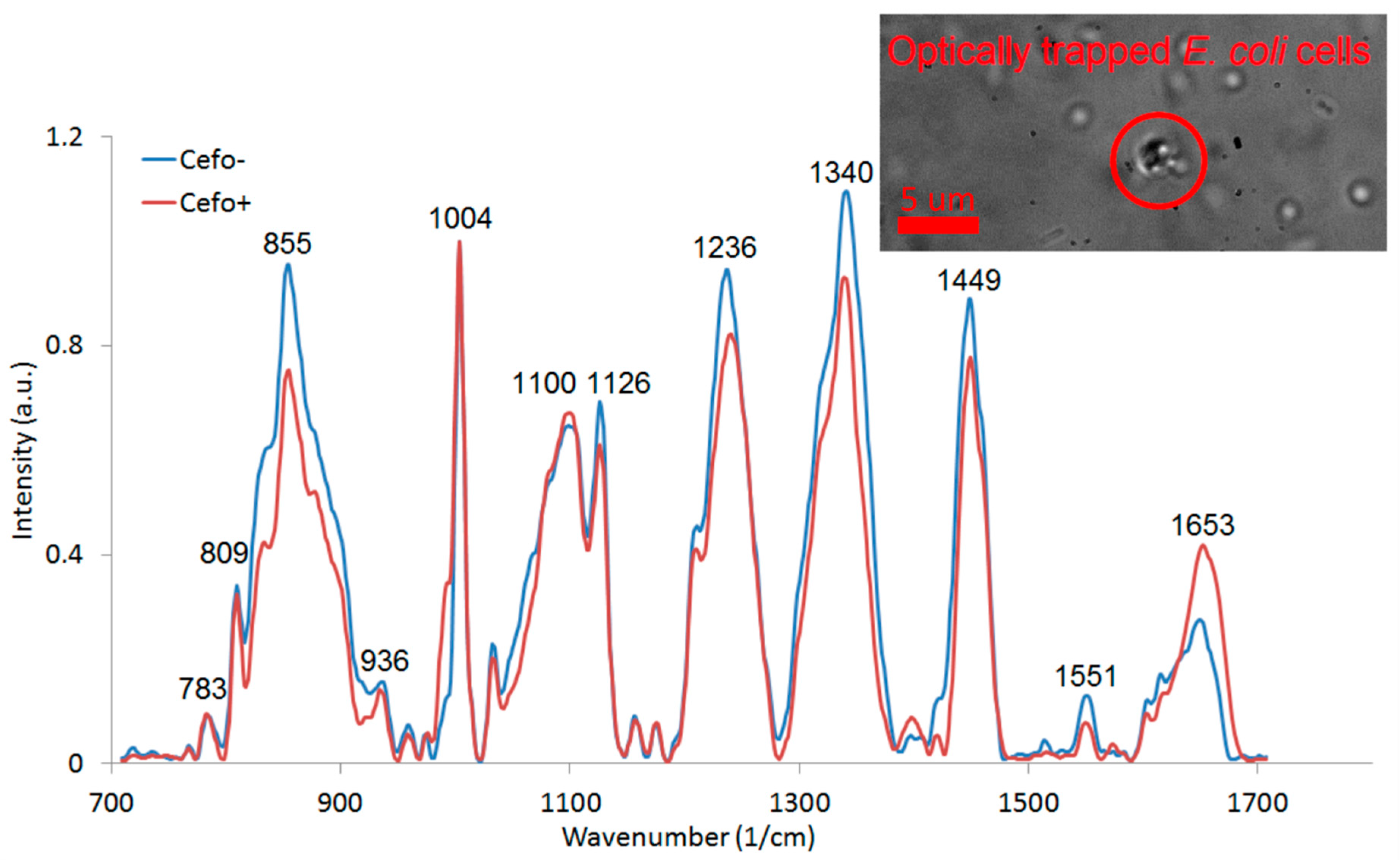
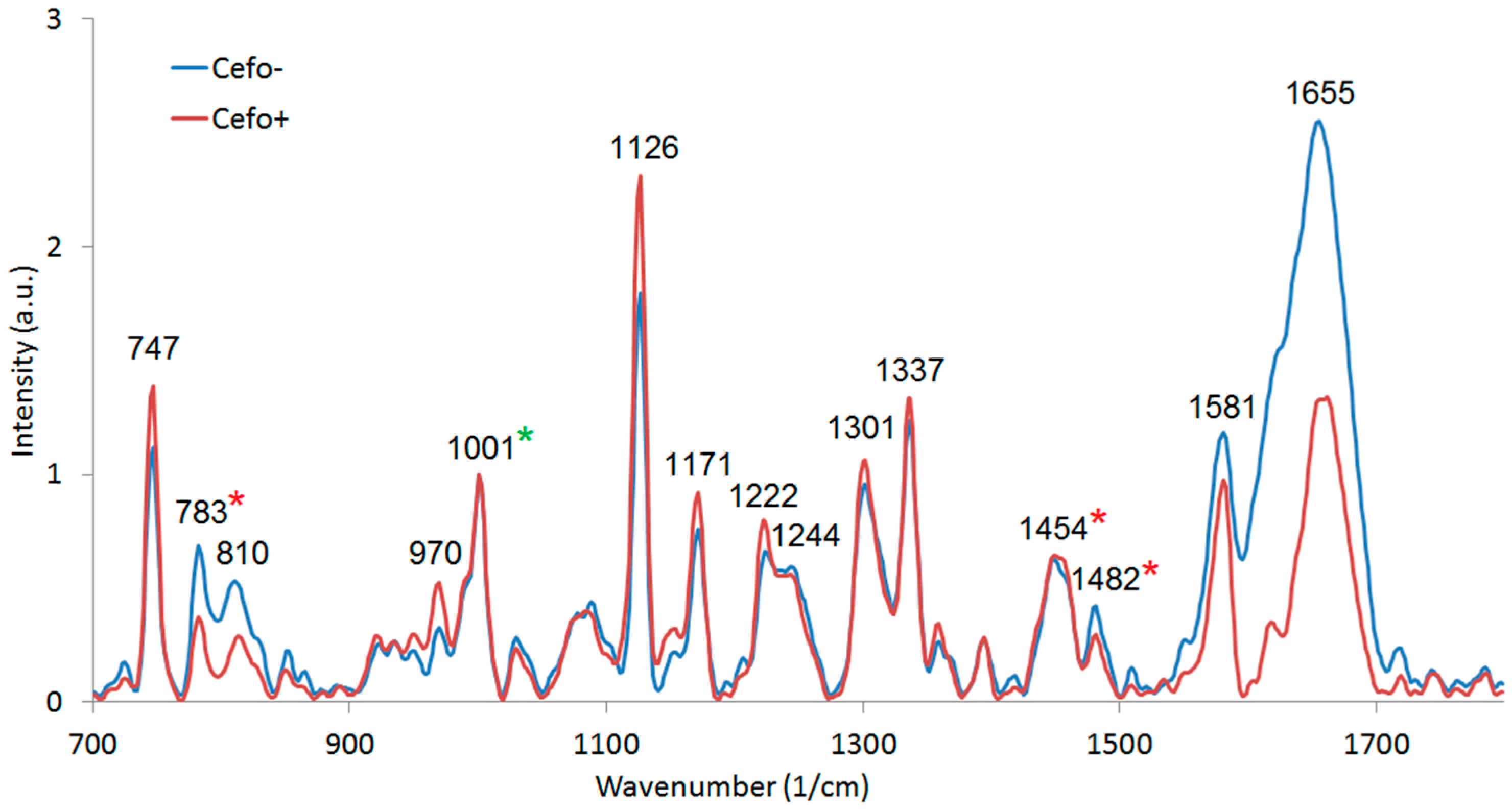
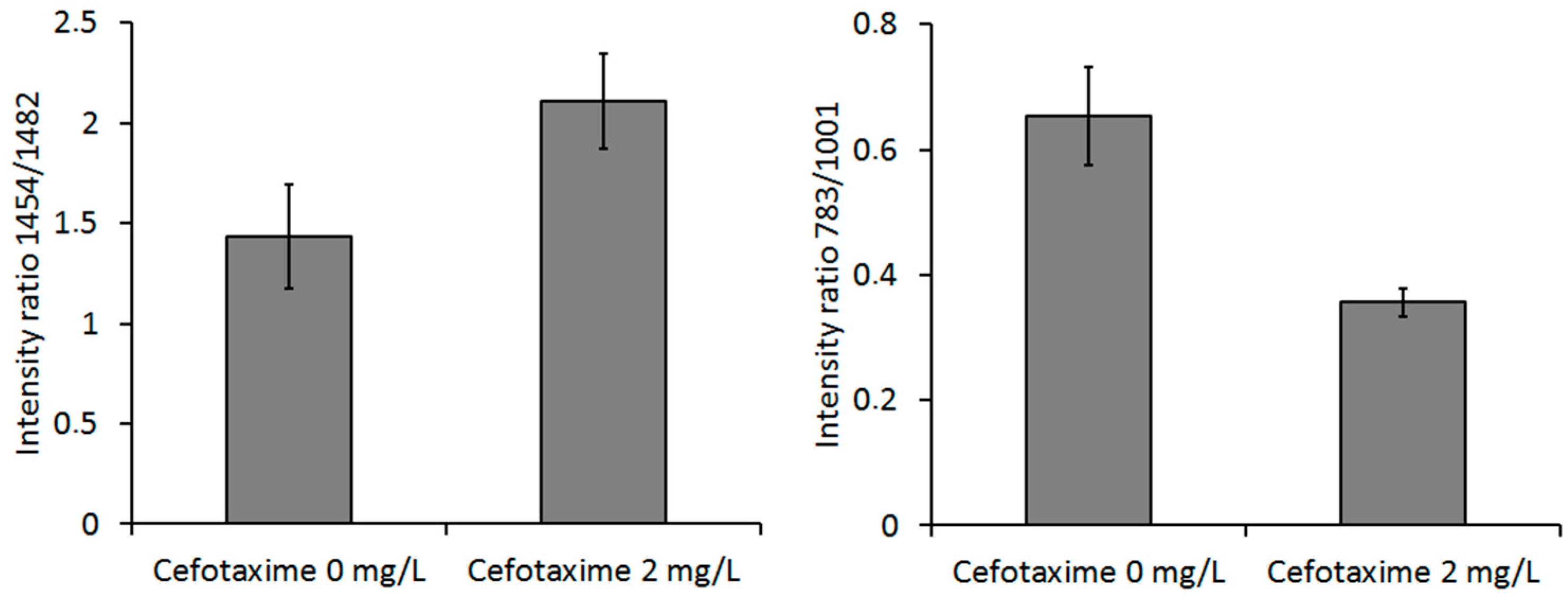
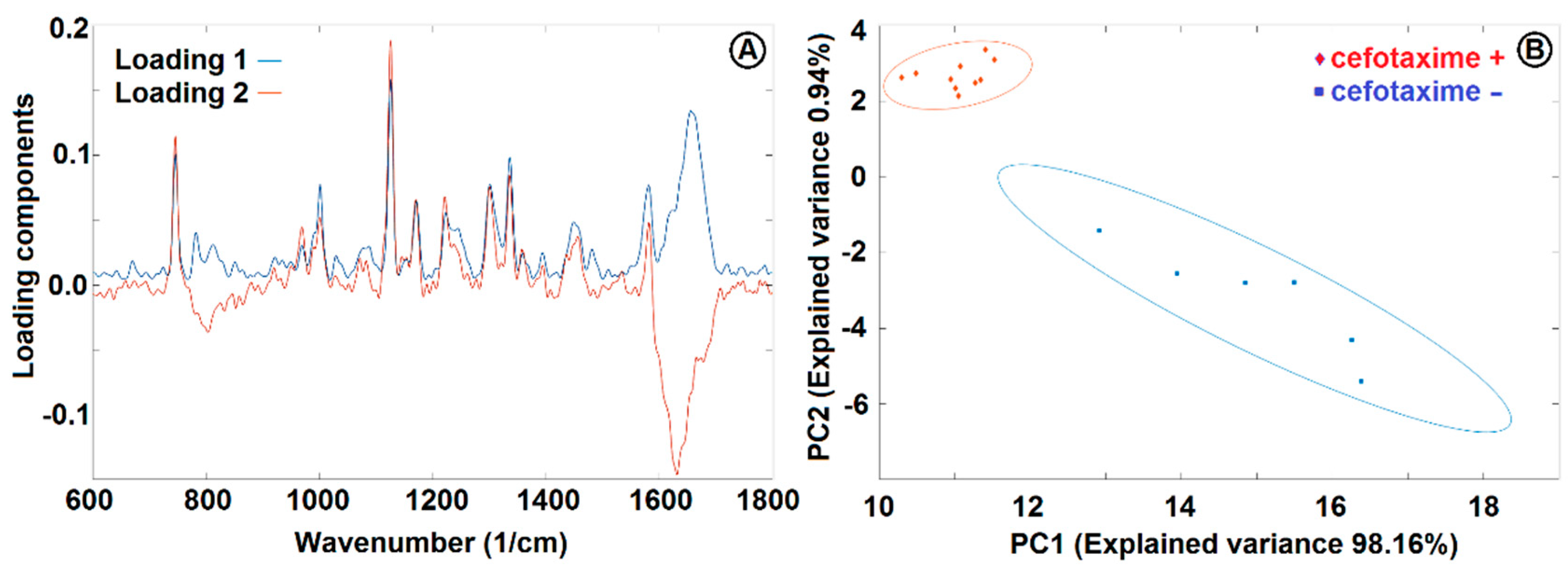
| Wavenumber (1/cm) 1 | Assignment | Wavenumber (1/cm) | Assignment |
|---|---|---|---|
| 728 (719, 723) | Adenine | 1095 (1100, 1094) | DNA: OPO− |
| 783 (783, 783) | Nucleic acids (C, T) | 1126 (1126, 1126) | C–N, C, T |
| 813 (809, 810) | Tyrosine | 1257 (1236, 1244) | Amide III |
| 857 (855, 853) | Tyrosine | 1340 (1340, 1337) | Nucleic acids (A, G) |
| 936 (936, 934) | DNA backbone | 1453 (1449, 1454) | C–H2 def., lipids |
| 1004 (1004, 1001) | Phenylalanine | 1660 (1653, 1655) | Amide I |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilát, Z.; Bernatová, S.; Ježek, J.; Kirchhoff, J.; Tannert, A.; Neugebauer, U.; Samek, O.; Zemánek, P. Microfluidic Cultivation and Laser Tweezers Raman Spectroscopy of E. coli under Antibiotic Stress. Sensors 2018, 18, 1623. https://doi.org/10.3390/s18051623
Pilát Z, Bernatová S, Ježek J, Kirchhoff J, Tannert A, Neugebauer U, Samek O, Zemánek P. Microfluidic Cultivation and Laser Tweezers Raman Spectroscopy of E. coli under Antibiotic Stress. Sensors. 2018; 18(5):1623. https://doi.org/10.3390/s18051623
Chicago/Turabian StylePilát, Zdeněk, Silvie Bernatová, Jan Ježek, Johanna Kirchhoff, Astrid Tannert, Ute Neugebauer, Ota Samek, and Pavel Zemánek. 2018. "Microfluidic Cultivation and Laser Tweezers Raman Spectroscopy of E. coli under Antibiotic Stress" Sensors 18, no. 5: 1623. https://doi.org/10.3390/s18051623
APA StylePilát, Z., Bernatová, S., Ježek, J., Kirchhoff, J., Tannert, A., Neugebauer, U., Samek, O., & Zemánek, P. (2018). Microfluidic Cultivation and Laser Tweezers Raman Spectroscopy of E. coli under Antibiotic Stress. Sensors, 18(5), 1623. https://doi.org/10.3390/s18051623





