6.1. Decision Tree and Hard Rules
Traditionally, fire alarm systems based on smoke detection make use of a single threshold value to define the fire region. This region can be defined more accurately by taking into account readings from other sensors and building a set of thresholds (or rules) that incorporates the multiple sensor signals.
In the early nineties, Ishii et al. presented an approach based on hard rules and a smoke sensor coupled with a thermocouple and a semiconductor CO sensor [
8]. The multi-sensor system was placed in a 6.7 × 4.3 × 2.5 m
3 room in which smoldering fire (wood), flaming fire (n-heptane) and cooking activities (grilled fish) were performed. Based on the instantaneous reading of the three sensors, the authors defined specific regions in the sensor space to limit the fire region.
Figure 8 shows the defined regions and their complexity. Based on the set of rules, fire alarm is only triggered when the acquired point falls outside the volume enclosed by the different planes. As a result, cooking activity did not trigger fire alarm, although smoke density showed response to this activity, which may have reached obscuration threshold limit defined for smoke detectors.
The proposed set of rules, though, is very specific to the experimental setup and tested fire/nuisance scenarios. In order to provide a more general model, the authors proposed a method that uses dynamic features and relies on sensor correlation. In particular, using a similar experimental setup, they found out that heat release and volatile release come together in the performed fire test (metal chair with polyurethane cushion and polyolefin fabric). This sensor correlation was significantly smaller in the tested nuisance scenario (cooking). They proposed, thereby, to use the correlation between heat release and volatile release (and its rate of change) to detect fires. However, unfortunately, authors did not validate this approach with unseen measurements. Moreover, smoldering fires with very slow combustion process may initiate heat release significantly after volatile release and, therefore, the proposed signal correlation may not be a good indicator to predict slow smoldering fires.
In the mid-nineties, research teams from the Department of Fire Protection Engineering and the Department of Chemical Engineering at the University of Maryland (College Park, MD, USA) joined efforts to detect fire situations using a variety of sensors. Initially, researchers performed experiments in a small-scale setup (see
Section 7), in which only chemical sensors were used and samples were introduced using an atomizer. In this section, we will focus their efforts on a continuation work where the sensor system was placed in a larger experimental setup (3.6 × 3.6 × 2.4 m
3), and it included gas sensors and light obscuration sensing.
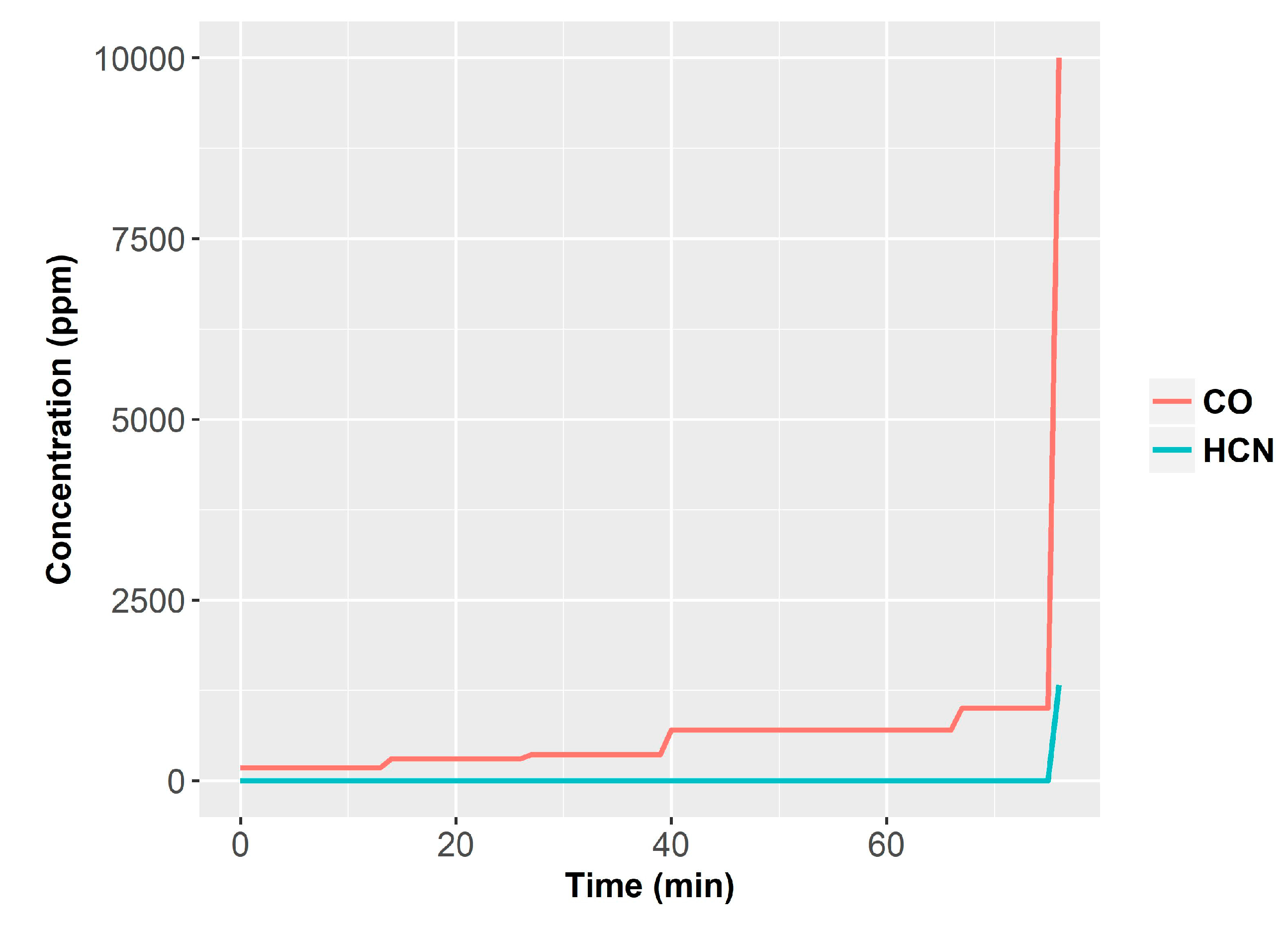
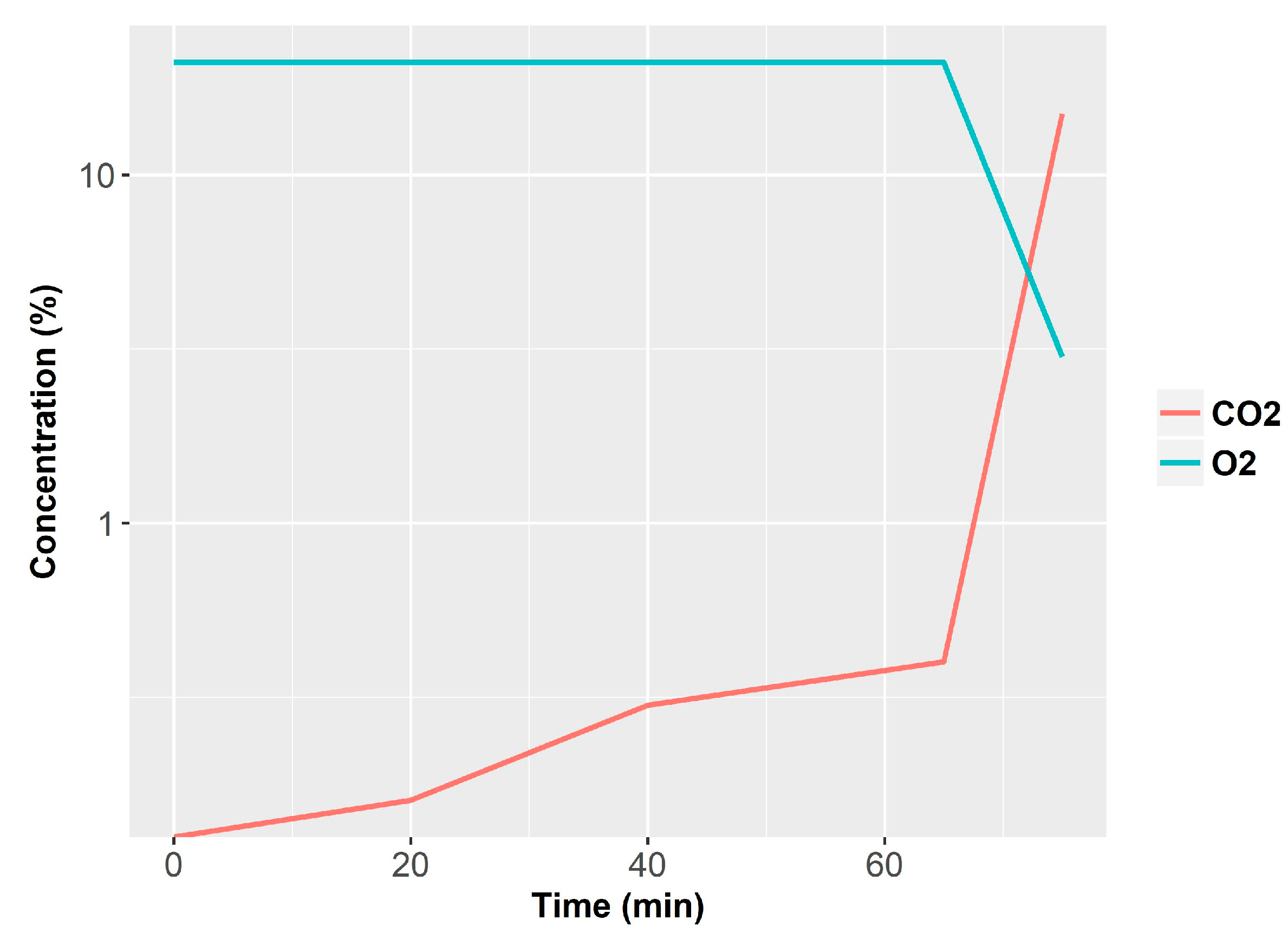
Specifically, the system integrated TGS880 and TGS822 MOX gas sensors (Figaro, Japan), CO (PIR 2000, range 0–1% Horiba, Irvine, CA, USA), CO
2 (Horiba PIR 2000, range 0–5%), O
2 (540A, range 0–20.95%, Servomex, Belgium) sensors, a temperature sensor (thermocouple) and light obscuration detector (OSD-100-5T-BNC, Centronic, UK). Moreover, for comparison purposes, the setup was equipped with two commercial smoke detectors (one photoelectric and one ionization) [
60,
61]. They performed 87 tests, including 34 flame fires, 16 smoldering fires and 37 nuisances.
The dimension of signals captured with the two MOX sensors, CO and CO2 sensors and temperature and light sensors was reduced to three dimensions by means of Principal Component Analysis (PCA). Therefore, the dimension of the space was shrunk from six to three, while the three principal components captured 76% of the variance of the original data. They built hard rules on this new space to classify flame fires, smoldering fires or nuisances. The scores were used to define the boundaries of each region as follows:
They compared the performance of the chemical system with a commercially available smoke detector. While commercial detector did not trigger the alarm for 16 of the 50 tested fire conditions, this number was reduced to only two for the multisensory system based on dimension reduction and hard rules. Proposed method also outperformed commercial system in response time, as, by average, flaming fires were detected 45 s faster and smoldering fires were detected 245 s faster, which represented a time reduction of 57% and 30% respectively. However, the system with gas sensors was very sensitive to nuisances as it produced false alarms for 10 out of 37 conditions (10 nuisances were wrongly identified as smoldering fire), while the smoke detector only showed 4 false alarms [
62].
False alarm ratio was improved, at the cost of reducing sensitivity to smoldering fires, when the authors revisited the dataset and considered a new set of sensors. In particular, the system included two MOX sensors, CO and CO
2 sensors and the temperature sensor [
62]. In other words, the photocell was removed from the array of sensors. Using hard rules based on the sensor signals the authors could classify smoldering fires, flame fires, nuisance cases, and background. The rules were defined as follows:
If: CO2 > 210 ppm or T > 105 F: Flaming fire.
Elseif: VTGS822 > 0.9 V and VTGS880 > 0.15 V: Nuisance.
Elseif: CO > 17 ppm and CO2 > 22 ppm and VTGS822 > 0.27 V: Smoldering fire.
Else: Background.
where VTGS8xx denotes acquired voltage from the corresponding MOX sensor conditioning circuit.
Table 6 shows the confusion matrices for the commercial smoke detector, and the two considered multi-sensor arrays with the corresponding decision models. The systems that included chemical sensing outperformed smoke detector in terms of sensitivity to fires. Similarly, the system with the light obscuration sensor showed higher sensitivity to smoldering fires than when the light sensor was removed. However, whether this is due to the information provided by the light sensor or due to the employed decision algorithm remained unexplored. On the other hand, chemical systems showed a higher rate of false alarms than the smoke detector. Actually, as all considered methods rely ultimately on the definition of thresholds, sensitivity and specificity could be adjusted by tuning the corresponding thresholds.
In another work, the same research group explored fire sensitivity and nuisance immunity using another multi-sensor system and different hard rules [
64]. Specifically, they exposed a photoelectric smoke detector, ionization smoke detector, CO sensor and thermocouple to 32 fire tests (smoldering and flaming) and 11 nuisance (cooking tests, smoking and candle) scenarios. Captured signals were filtered to reduce noise and get rid of data spikes. Instantaneous values and rate of rise for each of the sensors were considered.
Authors proposed nine different hard rules using different combinations of sensors and features. Resulting sensitivity and specificity were evaluated individually for each set of rules, and they were compared to thresholded smoke detectors. Results indicated that the rule involving the rate of temperature rise, CO concentration, and smoke detection (using ionization detector) provided the best immunity to false alarms and fire sensitivity. In particular, the selected rule was as follows:
The authors concluded that rules that included CO measurements resulted in faster detection of smoldering fires than smoke detectors. Similarly, the rate of temperature rise resulted in faster fire detection, or at least, similar, than smoke detectors. Authors also proposed several rules to define fire/non-fire regions after PCA was applied to data. However, authors did not find any significant improvement after defining ellipses in the lower-dimension space. Authors attributed the similar performance of the rules defined directly in the sensor space with the rules defined after the PCA to the limited number of sensors which is not large enough to flourish the benefits of dimensionality reduction.
In summary, the research efforts carried out by the Department of Fire Protection Engineering and the University of Maryland showed that simple hard rules could be defined such that fire and nuisance situations can be discriminated. They also showed that dimensionality reduction could be performed before the definition of the decision rules. When compared to smoke detectors, chemical-based fire detectors showed improved sensitivity, although it came at expenses of higher false positive rate. The remaining challenge is keeping high sensitivity while specificity remains at acceptable levels.
Chen et al. proposed a system that combined smoke detector with carbon monoxide and carbon dioxide measurements [
65]. They compared the performance of the multi-sensor system with the performance of only the smoke detector. The smoke detector was based on light scattering and, when operating alone, it triggered a fire alarm when the threshold of 15% obs/m was reached. CO and CO
2 detection were performed by means of a diode laser-based absorption spectrometer, which was composed of a laser, InGaAs diodes and reference and measurement cells.
The proposed algorithm for the multi-sensor system was based on dynamic features, specifically, the rate of change of the smoke, CO, and CO2 signals. Then, a decision tree was built to output, continuously, fire/non-fire prediction. Briefly, fire was only predicted when smoke rate of rise was higher than a threshold and the rate of rise of CO or (non-exclusive) the rate of rise of CO2 were higher than the corresponding thresholds. The authors explored two methods to estimate the signals’ rate of increase. First, the rate of rise was estimated fitting a linear function to the captured data points using 10-s time windows. The second method included a moving average filter before the linear fit was computed. The thresholds were adjusted for each volatile and method, resulting in the following rules for the first and the second methods respectively:
where V
smoke represents the voltage captured from the output of the smoke detector. The mentioned algorithm was patented by the authors [
66].
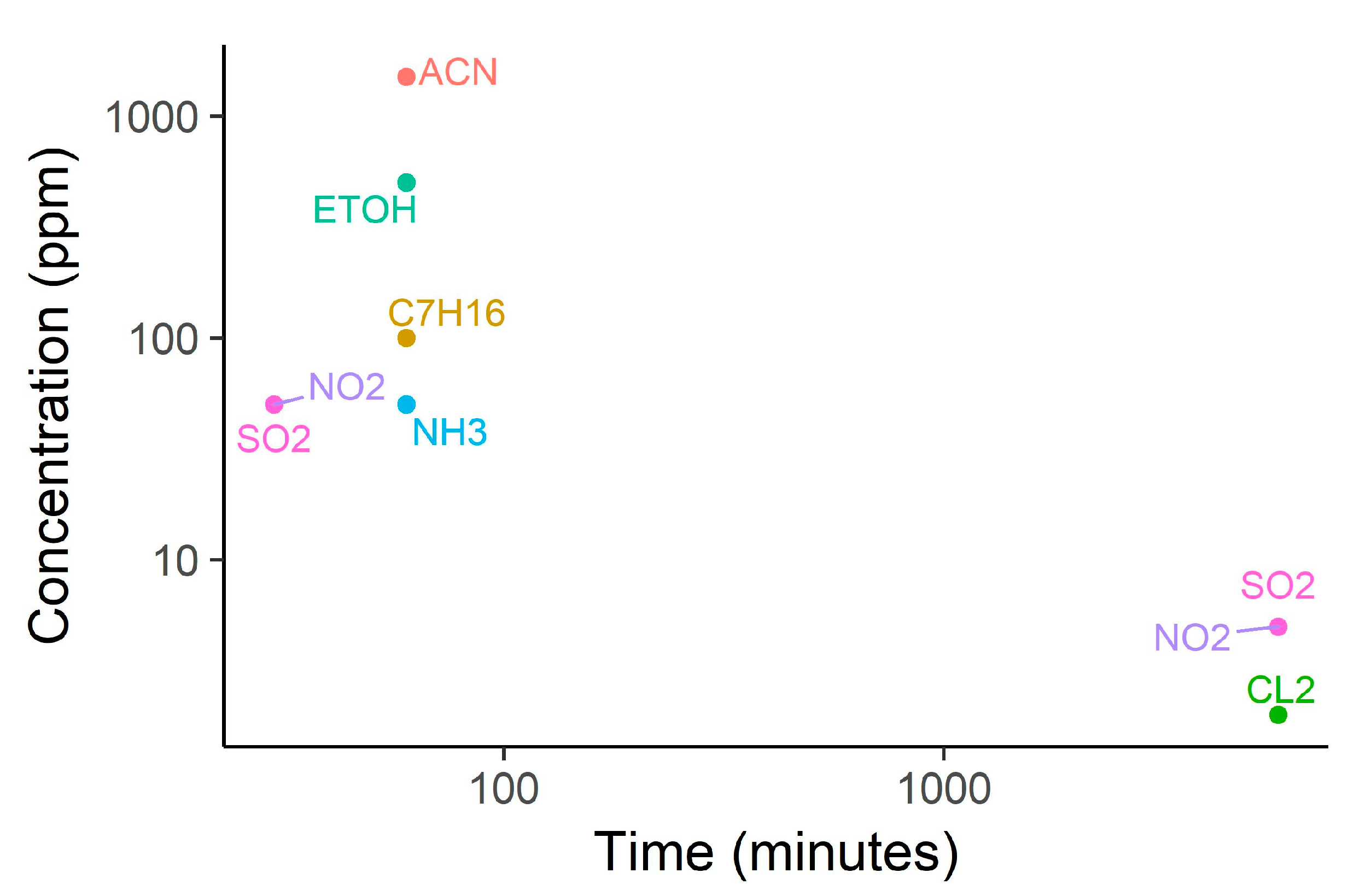
The authors tested their approach using a collected dataset that included a total of 30 fires (smoldering and flame) performed in a 2.2 × 1.4 × 4 m3 unventilated room. Smoldering fires included HDPE beads, PVC clad wire, mixed fabrics (with different ignition methods) and green canvas. Flame fires included heptane, toluene, methanol and mixed plastics. Two or three repetitions were carried out for each fire type. Authors also tested immunity to false alarms. In particular, they tested nuisances that may be present in aircrafts. Specifically, tested nuisances included dry ice, insecticide bomb (aerosol), halon, water, methanol, ethanol, acetone, and ammonia.
Results indicated that there is no significant difference between the two methods proposed to compute the signal derivatives, and no false alarms were detected throughout the tests. However, the multi-sensor system showed better sensitivity to fire than the smoke detector. Due to the small amount of smoke released by heptane, methanol, PVC wire and mixed fabrics, smoke detector did not trigger fire alarm for these four types of fire. The multi-sensor system only missed methanol fire. However, the authors adapted the rules such that fire is predicted when two of the three rate of rise features exceeded the corresponding threshold. With the new formulation, the multi-sensor system was able to detect methanol fire as well. Moreover, multi-sensor system also showed improved detection time, reducing, for example, detection time of HDPE bead fire from 616 s to 320 s.
The authors showed that defining rules based on the rate of change of the signals may be beneficial, as these dynamic features overcome issues with baseline shifts and may detect changes faster. Finally, the outstanding sensitivity and robustness to false alarms of the multi-sensor system may be due to the specificity of the employed chemical gas sensors. The immunity to false alarms may not be found when using less-costly, broad-response gas sensors.
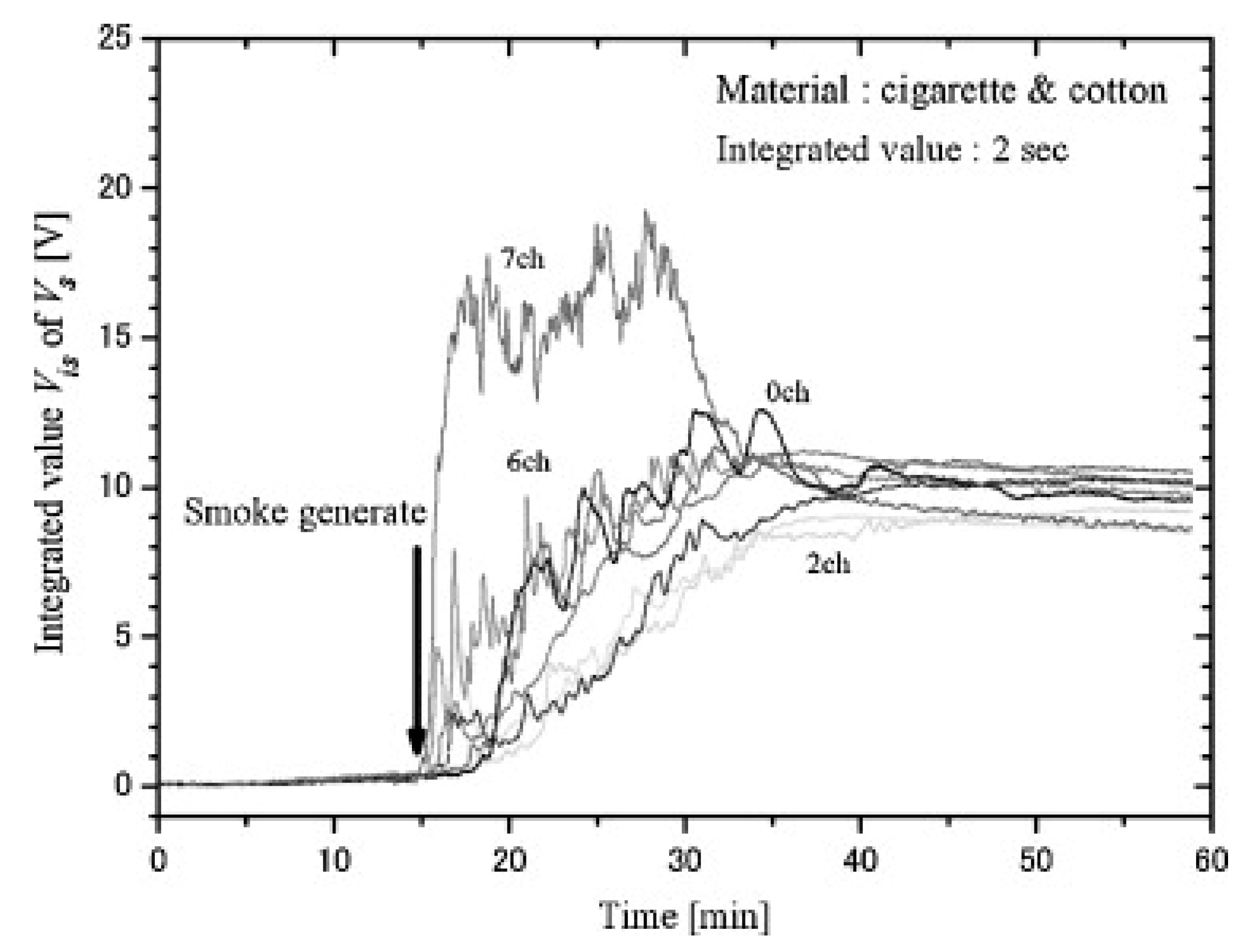
Gottuk et al. presented a system that combined smoke detectors with CO detection using an electrochemical gas cell [
67]. The authors performed fires and nuisances in a 49 m
3 room. Large variety of flame and smoldering fires (heptane, alcohol, gasoline, flaming polyurethane, smoldering polyurethane, cardboard, cotton fabric, flaming cotton wick, smoldering cotton wick, cotton batting, upholstery fabric, PVC cable, smoldering wood at different temperatures) and nuisances (Wesson oil, toast, melting cheese, bacon, propane burner, kerosene heater, cigarette smoke, people smoking, water steam) were induced in the room, with different number of repetitions each scenario. Two smoke detector systems (ionization and photoelectric), along with gas sensors were installed in the test room.
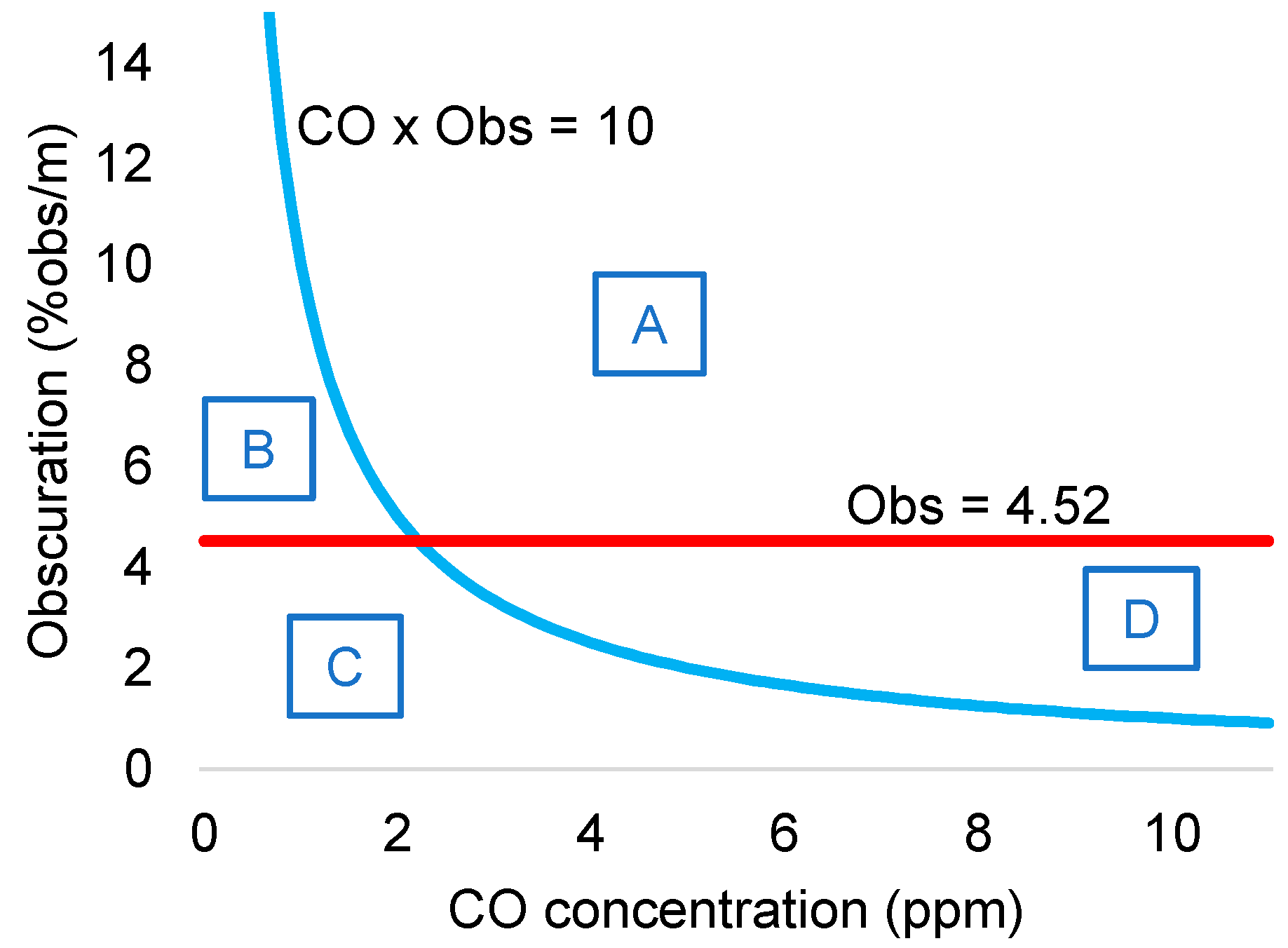
The authors set the detection threshold of smoke detectors to 4.52% obs/m for the ionization detector and 6.72% obs/m for the photoelectric smoke detector. Results confirmed that ionization detectors show better sensitivity to flaming fires, whereas photoelectric detectors show better performance for smoldering fire detection.
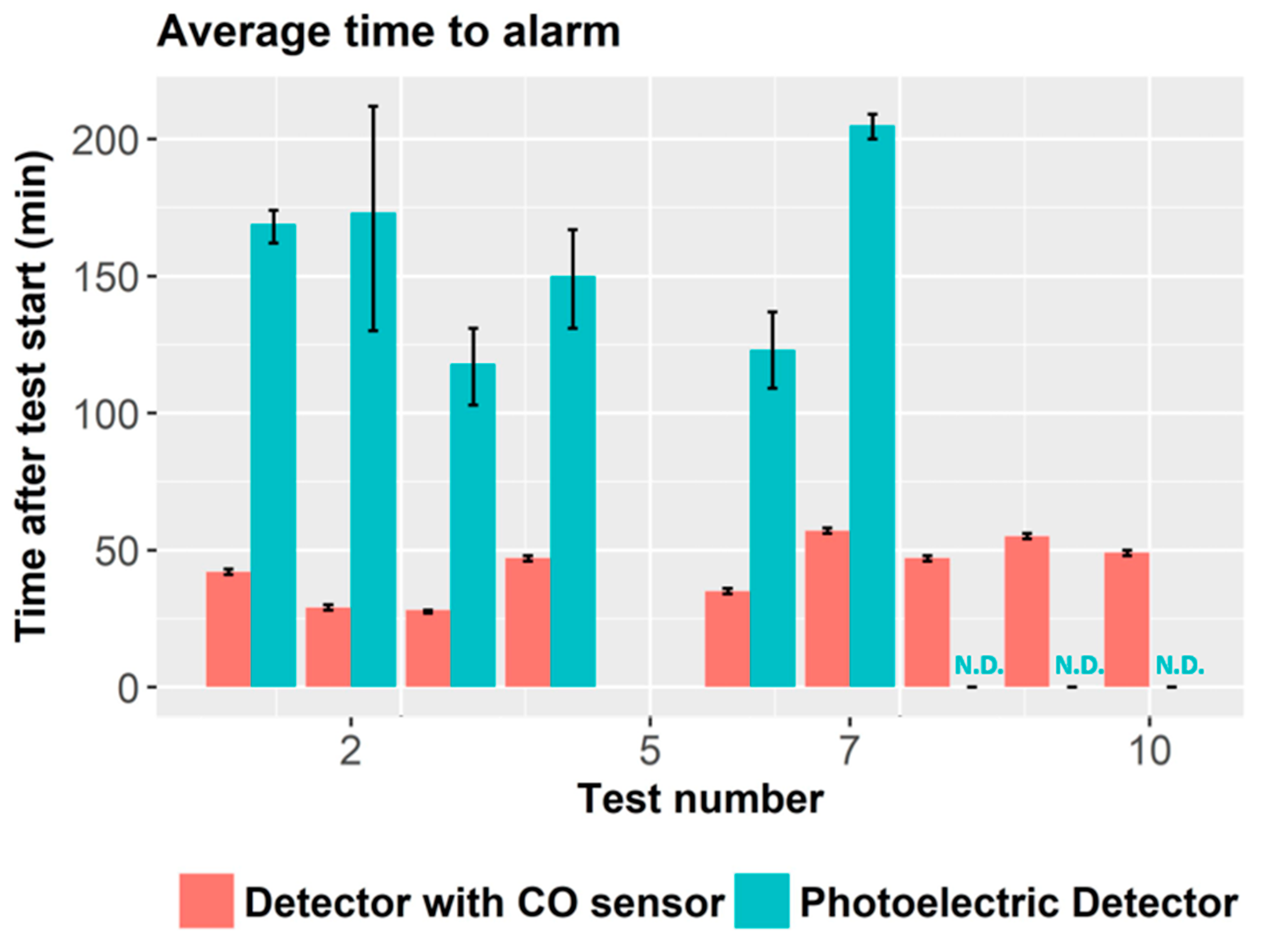
The proposed multi-sensor algorithm for fire detection was based on the readings from the ionization fire detector and the CO sensor. The authors developed a simple rule that takes into account the readings from both sensors such that high concentrations of CO also triggered fire alarm. In particular, the criteria was as follows: the alarm was triggered when the product of the ionization detector output (% obs/m) times the CO sensor reading (in ppm) was greater than 10 (% obs/m)(ppm). By coupling the CO sensor to the ionization smoke detector, boundaries of fire/non-fire regions could be defined, as shown in
Figure 9.
The multi-sensor system was compared to traditional smoke detectors. Despite the simplicity of the proposed rule, the multi-sensor system detected 42 out of 53 fire tests, while ionization and photoelectric detectors detected 25 and 29 of the tested fires, respectively. Briefly, the multi-sensor system detects the union of the set of fires that are detected by the ionization and the photoelectric detectors, except for some smoldering wood (at lower temperature) and PVC cable, which can be detected by photoelectric detector and did not trigger alarm for the ionization smoke + CO detector.
Immunity to false alarms was also improved with CO measurement. For example, water steam increased obscuration measure and triggered smoke detector alarms, but it did not increase CO sensor readings, which prevented triggering fire alarm for the multi-sensor system. Photoelectric and ionization showed false alarm to 17 and nine of the 27 tested nuisances. Multi-sensor system only triggered false alarm in six of the nuisance scenarios. Moreover, time response was also improved. Ionization detector coupled to CO sensor showed faster response time than ionization detector alone, except for heptane and polyurethane fires.
The authors showed that adding CO measurements to light obscuration sensor can improve both fire sensitivity and false alarm immunity. Simple hard rules can successfully process sensor signals. However, the authors already discussed a limitation of the proposed rule as its asymptotic nature makes it necessary very high levels of CO concentration (or smoke) if smoke (or CO concentration) levels are very low. This rule will delay the detection of fires that, for instance, generate small CO concentration. The authors proposed adding additional rules to cut the asymptotic behavior in its limits.
All in all, hard decision rules have been explored recurrently over the years. The popularity of this choice is probably due to the classic operation of smoke-detectors that rely on signal thresholds. The natural path is, hence, reshaping fire regions defined with light obscuration thresholds to obtain more accurate fire regions that incorporate additional information from chemical gas sensors. On the bright side, hard rules are considered as “white boxes” as they are easy to interpret [
68]. Acquired knowledge of the system behavior is translated to a readable set of rules.
On the downside, decision rules may become too complex when many different nuisances are considered, as each scenario may require its own set of conditions to be excluded from the fire region. Also, and most significantly, hard rules depend heavily on the presented dataset. This is usually not-desired as one aims at building models robust to noise and able to generalize to new data or new experimental conditions (room size and geometry, fire types, nuisances, etc.). One limitation that we found in the literature is the fact that generalization to other experimental conditions is not explored. To what extent defined rules are valid when the system is placed in a different room, under different ventilation conditions or when the sensors are at different distances from the fire source remained, mostly, unexplored.
Dynamic features were also proposed to improve the accuracy and the generalization ability of the models. For example, it was found that rate of rise of CO and CO
2 concentration levels can improve the ability of the system to discriminate between fire and nuisance scenarios. In reference [
69], only one nuisance showed CO
2 increase rate higher than 0.1 ppm/s, and only two nuisances induced CO increase rate higher than 0.025 ppm/s. Although CO
2 was found to increase at high rates during fire, it also does so when the room is occupied by individuals (the presence of people in a non-ventilated room can induce CO
2 increase rates as high as 0.5 ppm/s). Therefore, CO rate of rise was suggested over CO
2 rate of rise to discriminate fire from nuisances.
Also, using dynamic features, such as rate of rise, becomes beneficial as these features are insensitive to baseline shifts and may provide faster responses. For example, derivative features were shown to change faster than the mean of the signal computed in the same time window [
65].
Similar to static features, thresholds for dynamic features may be also specific to room size or geometry. However, experiments in two test rooms suggested that room effects can be incorporated to the model by including (and adjusting) rate of rise thresholds in the algorithms [
63].
Finally, approaches based on linear data transformation (PCA) have been proposed to define hard rules in the transformed sensor space. These rules may be intricate and complex in the original space, but they may become simple in the new space. Moreover, if enough repetitions are included in the original data matrix, the new data projection can find the
mean direction for each fire/nuisance type and reject inherent variability for each scenario [
64].
Hard decision rules have been proved to provide good prediction ability when tested under the same conditions than the calibration conditions. However, other classification algorithms that usually show lower generalization error [
68] have also been explored for reliable fire detection.
6.2. Neural Networks
In the early 1990’s, Okayama studied the use of neural networks to assess the risk of fire using a variety of sensors [
70]. He adapted the configuration of the neural network to address three different tasks, using different sensor ensembles and sensor features for each task.
First, a three-layer neural network with three input neurons, five hidden neurons and three output neurons was used to output three fire indicators. Three sensors (temperature, carbon monoxide sensor, photoelectric smoke sensor) were considered to feed the input layer of the network. Static features for CO and smoke sensors were extracted, whereas dynamic feature (rate of rise) was extracted from the temperature sensor. Additionally, to extract the corresponding features sensor readings were normalized such that the ranges 0–20% obs/m, 1–100 ppm of CO, and 0–10 °C/min were mapped to the interval 0–1. The output of the network was associated with three indicators (fire probability, fire risk and smoldering fire probability), which were also set in the range of 0–1. The neural network was trained using 12 fire patterns.
In the second task, only the photoelectric smoke sensor was used. Two features were extracted from the sensor signal: instant value and rate of rise. The features were also normalized to the range 0–1, corresponding to 0–20% obs/m and 0–20% obs/m per minute, respectively. The architecture of the network consisted of two input, four hidden, and two output neurons. The relevant output neuron was associated with fire probability and 18 fire patterns were presented to train the network weights.
Similar to the second task, the third task considered only the photoelectric sensor, but the dynamic feature was changed. In particular, the two extracted features were the instantaneous sensor reading and the time duration (normalized to 0–1) since the sensor signal exceeded a defined threshold. The network consisted of two input neurons, four hidden neurons, and one output neuron (that accounted for fire probability). The network was trained with 10 patterns. Finally, task 3 was extended to consider ventilation conditions. Ventilation was incorporated to the neural network as a third digital input that took 0/1 for ventilation on/off.
After the mentioned neural networks were trained, output values provided by the model showed acceptable correlations with the defined values, also when chemical sensors were combined with smoke detectors. Unfortunately, different measurements were used to train the different models, making not possible the comparison between the considered tasks. Moreover, very few details on the experimental protocol are presented in the original work, the time at which the vector of features was extracted to feed the neural network was not specified, or details on the criteria to quantify the output indicators were omitted, which represent the alarm signals. Nevertheless, results presented by Okayama were encouraging as, although the simplicity of the neural network, the model could assign a probability to the presented measurements. He also considered dynamic features, showing that there is relevant information in the temporal response of the signals. Actually, he envisioned that further work should consider models that are able to process time-series directly.
In a following work, Okayama and Sasaki considered nuisance scenarios, which were omitted in Okayama’s previous work. They coupled a MOX gas sensor to a smoke detector to discriminate fire from nuisances using a neural network (four input neurons, four hidden neurons, one output neuron) [
71]. The sensors were exposed to sixteen measurements that included fire repetitions (beechwood smoldering fire at 2 m or 3 m from the sensors) and non-fire situations (smoking, cooking, coffee aroma, background). Four features were considered to feed the three-layer neural network: normalized sensor level and normalized rate of change per minute, for each sensor. The output neuron was associated to fire probability. For training the network, fire probability was manually assigned in the range 0–1 according to the distance of the sensors to the fire source or the type of nuisance.
Unlike their initial work, the neural network processed all the captured signals continuously. As a result, fire probability was provided as a function of time. Results showed that the system was able to output fire probability continuously, providing reasonable values as smoldering fires were being developed. However, the model showed difficulties to reject nuisances (mainly cooking activities). This shortcoming was attributed to air turbulence that took place in the test room (270 m
3) that limited the accuracy of the classifier [
71,
72].
In a similar work also using neural network, Okayama studied the feasibility of fire detection using only chemical sensors (see
Section 7, reference [
73]).
In order to reduce fire detection time and increase the reliability of fire detectors, Derbel integrated three metal-oxide gas sensors with a commercially available optical (light-scattering) fire detector and a temperature sensor [
4]. Specifically, the gas sensors were selected for carbon monoxide, hydrogen, and ammonia detection.
The system was exposed to flaming fires (TF1, TF4, TF5) and smoldering fires (TF2, TF3) inspired by the EN54 standard, a non-standard fire (cable fire) and two nuisance scenarios (disco-fog generated with a commercial fog machine, and cigarette-smoke using a force pump that regulated the burning process).
In order to build a model to detect fires, different dynamic features were tested. First, a moving window and FFT transformation provided features from the sensors’ signals. Second, feature extraction was performed by means of scaling the quadratic mean value of the signals, and then a back-propagation neural network was used to output the prediction. In both cases, results indicated that incorporating chemical and temperature sensors to the optical fire detector provided faster alarm signals in a more reliable manner (unlike the optical fire detector, the multisensory system did not show false alarms for cigarette smoke and disco-fog).
However, since no repetitions were acquired, models were trained and tested using features of the same measurements. Features of sensor signals corresponding to the same measurement were distributed in train and test. TF1, TF2, TF3, TF5, cable fire and cigarette smoke appear both in training and test, and only disco-fog and TF4 were left completely for test. Hence, training and test vectors are not completely independent. This questionable dataset partitioning yielded, most likely, to overfitting and overoptimistic results.
Finally, to what extent the performance increase of the system is due to the integration of the temperature sensor or the chemical sensors was not explored. This would provide very meaningful insights for the design of chemical-based fire detection systems.
Neural networks have shown good performance for fire prediction. However, more elaborate networks have been presented to account for the prior probability distribution function, such as Probabilistic Neural networks. Taking into account prior probability seems critical in fire prediction, as one expects the system in rest state for most of the time.
6.3. Probabilistic Neural Network
A remarkable work in fire detection was published by Rose-Pehrsson et al. (Naval Research Laboratory) in 2000 [
74]. They studied the response of different sensor technologies to 24 different types of fire and 12 nuisances (see
Table 7 for the complete set of fire/nuisance scenarios considered in the study). Several repetitions of the scenarios were performed in a 96 m
3 test compartment, for a total of 240 events (120 background recordings, 82 fires and 38 nuisance sources). To the best of our knowledge, the considered dataset represents the largest dataset, with the largest variety of fire types and nuisance scenarios, collected for fire detection with chemical gas sensors. The large variety of fire and nuisance sources enabled a thorough study on fire detection sensitivity and system reliability. Moreover, the authors also placed a large number of sensors in the measuring compartment. The variety of sensing technologies and the benchmark measurements performed with commercial smoke detectors allowed to achieve another relevant goal of their work: the study of sensor similarities and the selection of an optimal subset of sensors for reliable fire detection.
In particular, the authors placed 20 sensors of different types in the measuring compartment. A variety of chemical gas sensors was installed to target various combustion products. Chemical sensors included carbon monoxide (at two concentration ranges), oxygen, hydrogen, hydrogen chloride, hydrogen cyanide, hydrogen sulfide, sulfur dioxide, nitric oxide and nitrogen dioxide electrochemical cells, a NDIR for CO2 and a MOX for hydrocarbon detection. Commercially available smoke detection systems (ionization and photoelectric) and an optical density meter system were also included to obtain reference measurements, and temperature and humidity were monitored during the measurements as well.
The sensitivity to fire detection and the immunity to nuisance sources of photoelectric and ionization fire detectors were used to benchmark the system that incorporated gas sensors. Conventional alarms were triggered when signals reached different obscuration thresholds. In particular, three thresholds were tested for each smoke detector. First, alarm thresholds were set to 4.2% obs/m for ionization and 11.0% obs/m for photoelectric detectors, which correspond to typical alarm thresholds. Minimum alarm level allowed by the UL 268 Standard (1.63% obs/m) and half of it (0.82% obs/m) were also tested as alarm thresholds. Using a total of 120 events (82 fires and 38 nuisances), confusion matrices for each detector type and threshold values were computed.
Results with smoke detectors showed that, at lower alarm levels, systems showed high sensitivity to fires, but low immunity to nuisances. At lower alarm levels, 73% of the fires were correctly detected by the photoelectric detector, but false alarm ratio was as high as 47%. Oppositely, at the higher alarm level, the system could detect only 38% of the fires, while 82% of the nuisances were rejected. Similar behavior was observed with the ionization detector. When background measurements were also included, best overall classification ratio were obtained at lower threshold alarm levels (83% and 88% for photoelectric and ionization detectors respectively).
The obtained classification ratio values served as a benchmark to compare the performance of gas sensor-based fire detection systems. The authors developed a pattern recognition algorithm for fire detection based on probabilistic neural networks (PNN). All gas sensor signals were filtered with Savitzky-Golay routine to reduce noise. Only steady-state features were considered, which were extracted at discrete times defined by reference photoelectric detector. Finally, before training PNN, matrices were scaled to zero-mean and unit-variance. The authors followed a leave-one-out cross-validation strategy, i.e., they sequentially trained all but one observation and predicted the class of the sample that was left out. This procedure was repeated until all the measurements were set aside for test.
Best results were obtained with a subset of five sensors: O2 (model 6C, City Technology, Portsmouth, UK), H2S (model TC4A-1A, City Technology), RH (model HX93, Omega, Stamford, CT, USA) ionization smoke detector (model 4098-9716, Simplex, Westminster, MA, USA) and photoelectric smoke detector (model 4098-9701, Simplex). With this array, 98% of correct classification was achieved.
The authors concluded that smoke detectors are important for the detection of fires. Results showed that systems including at least one smoke detector had higher sensitivity to fire. However, results indicated that gas sensors provide additional useful information for the discrimination of nuisances and early fire detection. Actually, nuisance rejection could be improved up to 25% when CO2, O2, CO, hydrocarbons, temperature and NO sensors were combined with smoke detectors at the lower threshold level.
In a continuation of their work, the authors demonstrated the flexibility of the PNN algorithm [
75]. Using a subset of sensors (photoelectric smoke detector, ionization smoke detector, CO and CO
2 sensors), they adjusted probability density function for each class. As a result, they could define the boundaries for each class. When the threshold was set to 100%, no false alarms were found, but only 60% of fires were successfully detected. As the threshold was lowered, fire detection ratio increased, at the cost of increasing false alarm ratio as well. By plotting the sensitivity and false alarm rate in a Receiver Operator Curve (ROC), the authors could select the threshold (85%) that provided similar detection rates than reference smoke detectors. However, false alarm rate was greatly reduced. At the selected threshold, the system detected 78% of the fires and less than 20% of the nuisances produced false alarm. This result clearly improved performance of reference smoke detection systems, as they showed 66.7% and 74.1% of sensitivity and 66.3% and 41.7% of false alarm ratios for ionization and photoelectric systems, respectively. Results, therefore, confirmed their previous findings that suggested that combining gas sensors with smoke detectors helps to reduce false alarm rates.
All in all, work from Rose-Pehrsson et al. confirmed the feasibility of chemical gas sensors for fire detection and that gas sensors can improve false alarm immunity. The work is particularly valuable as it relies on an extensive dataset that included 24 fire types and 12 nuisances. By collecting such dataset, the authors ensured the generalization of their approach, which sometimes is overlooked by other works due to the cost of the experimental setups and data acquisition. They also explored different sensors targeting various combustion products and proposed a reduced set of sensors for fire detection. A final decision on the sensing technologies should be taken according to target specifications and also other considerations such as system cost, time stability, calibration cost, power requirements, size, and others.
Finally, the authors also remarked that future developments need to consider temporal sensor responses. Since fires are dynamic events, authors expected that considering dynamic features would help in the detection of fires, in particular, capturing the dynamic change of oxygen and carbon monoxide [
32].
6.4. Hierarchical LDA
In another very interesting work performed at Saarland University, researchers developed a system based on a single MOX sensor to reduce false alarms in underground fires, specifically, in coal mines [
76]. Although their approach relies on a single MOX sensor, the authors benefit from the fact that MOX sensors exhibit different sensitivity and selectivity when operating at different temperatures, behaving therefore like different virtual sensors. Sensor’s operating temperature can be controlled by applying certain power on a built-in heater placed next to the sensing layer. Briefly, the authors modulated the sensor’s operating temperature and extracted multiple features using a single sensor.
The gas sensor operated in temperature modulation cycle (65-s period function) to increase the sensitivity and selectivity to the target compounds. The temperature profile included temperature ramps and high temperature operation steps. The authors considered several features from the acquired sensor signal. They extracted sensor values at defined temperatures (at discrete times) and slopes of the signal when transitioning between operating temperatures. Extracted features were selected such that, according to previous studies, they are suitable for the discrimination of relevant compounds.
The authors studied thoroughly the scenario of underground fires and identified the volatiles that result from fire (CO and ethane), its ratio (100/1), and the interfering gases (relative humidity, methane, CO, NOX or hydrogen). Based on previous investigations, the researchers designed a measurement profile that simulated, in a laboratory setting, fire and non-fire situations in underground atmosphere. Different concentration levels of CO, C2H4, NO2, H2 were presented to the sensor at different humidity (30%, 50%, 70%) background levels.
Next, they performed a 4-step hierarchical strategy based on Linear Discriminant Analysis (LDA). At each step, the captured data was sequentially classified according to the three levels of humidity (first layer), three levels of methane (second layer), presence of H2 or presence of CO or NO2 (third classifier), fire/non-fire condition (final classifier). This methodology is equivalent to a decision tree that leads to different final classifiers, the output of which predicts fire, non-fire, or warning situation. The proposed method may be overfitted to the used empirical data since it considers only discrete values of interferences, while in real scenarios, those values will take a continuous distribution.
In a more recent study [
2], data acquired in laboratory conditions was compared to field test data. Authors showed that field test data resemble data generated in lab conditions, validating their approach. However, all data was classified as normal operational situation since data only represented “non-fire situations”, i.e., CH
4, CO and NOx were found at standard concentration levels. The system was operating over several months, which revealed sensor drift. Changing sensor sensitivity in time may make system predictions unreliable as system calibration becomes obsolete. To counteract drift effects, however, authors proposed self-monitoring and self-diagnosis strategies [
77].
In our view, the above-discussed work provides a very valuable example of using a temperature-modulated sensor to extract various informative features from a single sensor. Using a single sensor, rather than an array of sensors, results in smaller and cost-efficient systems. All in all, the authors performed a very detailed analysis of the scenario and exposed the monitoring system to the relevant volatiles at different humidity levels. The authors developed a 4-step hierarchical classification algorithm that, according to the atmosphere composition, selects the final classifier to predict the presence of fire. This approach seems unpractical when the number of conditions of the environment (the number of interfering gases and concentration levels) increases, for example beyond the restricted scenario of underground mines. The proposed model is not defined when, for instance, the sensors are exposed to 60% RH (which path should the decision model follow? 50% or 70% RH?). In more complex environments, with a larger number of interfering volatiles, it seems more reasonable to build an integral model that considers all the conditions simultaneously and is defined for continuous variables.