Nanomaterial-Based Sensing and Biosensing of Phenolic Compounds and Related Antioxidant Capacity in Food
Abstract
:1. Introduction
1.1. Phenolic Componds and Health
1.2. Phenolic Compounds and Food Technology
1.3. Analysis of Phenolic Compounds and Antioxidant Capacity
1.4. Nanomaterials
2. Optical Sensing
2.1. Gold Nanoparticles
2.2. Silver Nanoparticles
2.3. Other Metal Nanoparticles
2.4. Quantum Dots
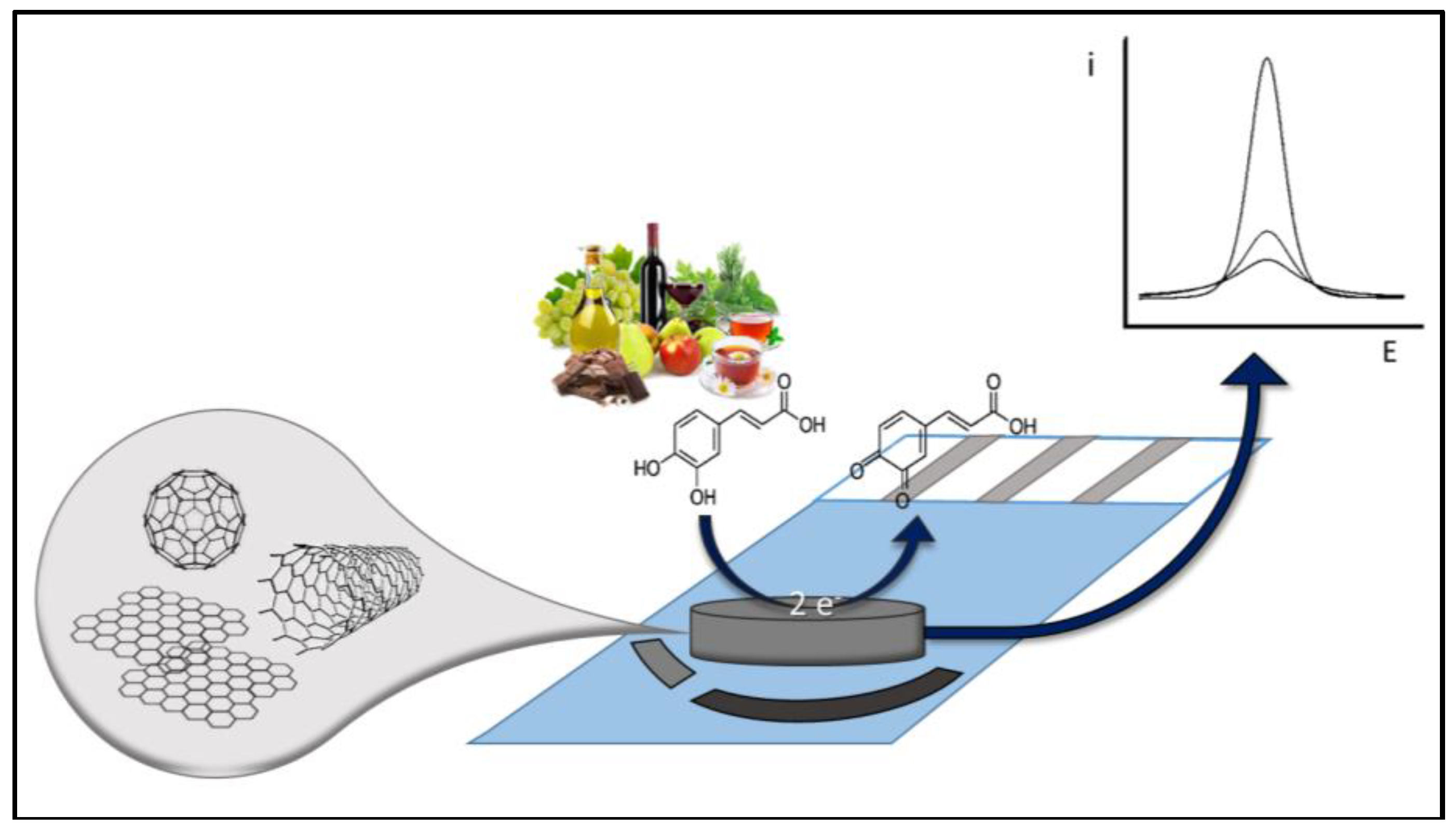
3. Electrochemical Strategies
3.1. CNTs
3.2. Carbon Black
3.3. Graphene
3.4. Alternative Strategies Based on Nanomaterials
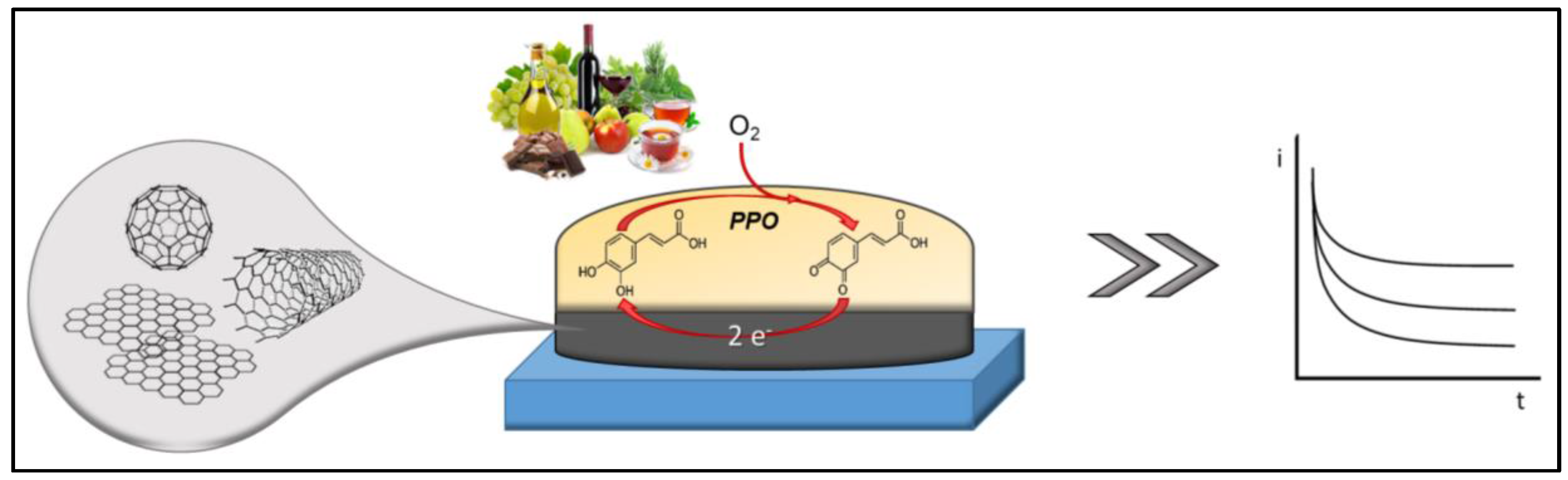
4. Nanomaterial-Based Enzyme Electrodes
5. Conclusions and Perspectives
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS• | 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) |
| AOC | Antioxidant capacity |
| AgNPs | Silver nanoparticles |
| AuNPs | Gold nanoparticles |
| B-DD | Boron doped diamond |
| B-GR | Boron-doped graphene |
| BSA | Bis(trimethylsilyl)acetamide |
| CB | Carbon Black |
| CBNPs | Carbon black nanoparticles |
| CeONPs | Cerium oxide nanoparticle-based |
| CNTs | Carbon nanotubes |
| CPE | Carbon paste electrode |
| CS | Chitosan |
| CTAC | Cetyltrimethylammonium chloride |
| CUPRAC | Cupric ion reducing antioxidant capacity |
| CV | Cyclic Voltammetry |
| DMF | Dimetylformamide |
| DMSO | Dimethyl sulfoxide |
| DPPH• | 2,2-Diphenyl-1-picrylhydrazyl |
| DPV | Differential pulse voltammetry |
| DPV | Voltammetry |
| FIA-AD | Flow-injection analysis with amperometric detection |
| FRAP | Ferric reducing ability of plasma |
| FTIR | Fourier transform infrared spectroscopy |
| GCE | Glassy carbon electrode |
| GEAC | Gallic acid equivalent antioxidant capacity |
| GR | Graphene |
| GNR | Graphene nanoribbon |
| GRO | Graphene oxide |
| GRQDs | Graphene quantum dots |
| H | High |
| IONPs | Iron oxide nanoparticles |
| L | Lower |
| Lacc | Laccase |
| LLE | Liquid–liquid extraction |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| LSPR | Localized surface plasmon resonance |
| MAE | Microwave assisted extraction |
| MNPs | Metal nanoparticles |
| m-phenol | Mono-phenol |
| MWCNTs | Multi-walled carbon nanotubes |
| Nanoceria | Cerium (IV) oxide NPs |
| N-GR | Nitrogen doped graphene |
| N-GR | Nitrogen-doped graphene |
| NM | Nanomaterials |
| NPs | Nanoparticles |
| o-diphenol | Ortho-diphenol |
| ORAC | Oxygen radical absorbance capacity |
| PANI | Polyaniline |
| PCs | Polyphenolic compounds |
| PCA | Principal component analysis |
| PEDOT | Poly (3,4-ethylenedioxythiophene) |
| PEI | Polyethylenimine |
| PLE | Pressurized liquid extraction |
| PMMA | Polymethyl metacrylate |
| Polytyr | Polytyrosine |
| PPO | Polyphenol oxidase |
| PSS | Poly(styrene sulfonate) |
| PtNPs | Platinum nanoparticles |
| PVA | Poly(vinyl alcohol) |
| QDs | Quantum dots |
| RhNPs | Rhodium nanoparticles |
| SCE | Super critical extraction |
| SLE | Solid-liquid extraction |
| SPE | Screen printed electrode |
| SPE | Solid phase extraction |
| SWCNTs | Single-walled carbon nanotubes |
| SWV | Square Wave Voltammetry |
| SWV | Square Wave Voltammetry |
| TA | Total aglycones |
| TEAC | Trolox equivalent antioxidant capacity |
| TEM | Transmission electron microscopy |
| TG | Total glycosides |
| T-GR | Thermally reduced graphene |
| TP | Total Polyphenols |
| TRAP | Total Reactive Antioxidant Potential |
| UAE | Ultrasonic assisted extraction |
References
- El Gharras, H. Polyphenols: Food sources, properties and applications—A review. Int. J. Food Sci. Technol. 2009, 44, 2512–2518. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Cimpeanu, C.; Predoi, G. Electrochemical methods for total antioxidant capacity and its main contributors determination: A review. Open Chem. 2015, 13, 824–856. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Nahak, G.; Suar, M.; Sahu, R.K. Antioxidant potential and nutritional values of vegetables: A review. Res. J. Med. Plant 2014, 8, 50–81. [Google Scholar] [CrossRef]
- Trevisan, M.; Browne, R.; Ram, M.; Muti, P.; Freudenheim, J.; Carosella, A.M.; Armstrong, D. Correlates of markers of oxidative status in the general population. Am. J. Epidemiol. 2001, 154, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Lall, R.K.; Syed, D.N.; Adhami, V.M.; Khan, M.I. Dietary polyphenols in prevention and treatment of prostate cancer. Int. J. Mol. Sci. 2015, 16, 3350–3376. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (Poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2012, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Serafini, M.; Peluso, I. Functional foods for health: The interrelated antioxidant and anti-inflammatory role of fruits, vegetables, herbs, spices and cocoa in humans. Curr. Pharm. Des. 2016, 22, 6701–6715. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Fruit polyphenols: A review of anti-inflammatory effects in humans. Crit. Rev. Food Sci. Nutr. 2016, 56, 419–444. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, T.; Kumar Mishra, S. Novel function of polyphenols in human health: A review. Res. J. Res. J. Phytochem. 2015, 9, 116–126. [Google Scholar] [CrossRef]
- Peluso, I.; Miglio, C.; Morabito, G.; Ioannone, F.; Serafini, M. Flavonoids and immune function in human: A systematic review. Crit. Rev. Food Sci. Nutr. 2015, 55, 383–395. [Google Scholar] [CrossRef] [PubMed]
- Peluso, I.; Serafini, M. Antioxidants from black and green tea: From dietary modulation of oxidative stress to pharmacological mechanisms. Br. J. Pharmacol. 2017, 174, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- Di Francesco, A.; Falconi, A.; Di Germanio, C.; Di Bonaventura, M.V.M.; Costa, A.; Caramuta, S.; Del Carlo, M.; Compagnone, D.; Dainese, E.; Cifani, C.; et al. Extravirgin olive oil up-regulates CB1 tumor suppressor gene in human colon cancer cells and in rat colon via epigenetic mechanisms. J. Nutr. Biochem. 2015, 26, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Pasinetti, G.M.; Wang, J.; Ho, L.; Zhao, W.; Dubner, L. Roles of resveratrol and other grape-derived polyphenols in Alzheimer’s disease prevention and treatment. Biochim. Biophys. Acta 2015, 1852, 1202–1208. [Google Scholar] [CrossRef] [PubMed]
- Neveu, V.; Perez-Jimenez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef] [PubMed]
- Frankel, E.N.; Meyer, A.S. The problems of using one-dimensional methods to evaluate multifunctional food and biological antioxidants. J. Sci. Food Agric. 2000, 80, 1925–1941. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Novel antioxidants in food quality preservation and health promotion. Eur. J. Lipid Sci. Technol. 2010, 112, 930–940. [Google Scholar] [CrossRef]
- Perumalla, A.V.S.; Hettiarachchy, N.S. Green tea and grape seed extracts—Potential applications in food safety and quality. Food Res. Int. 2011, 44, 827–839. [Google Scholar] [CrossRef]
- Ajila, C.M.; Brar, S.K.; Verma, M.; Tyagi, R.D.; Godbout, S.; Valéro, J.R. Extraction and Analysis of Polyphenols: Recent trends. Crit. Rev. Biotechnol. 2011, 31, 227–249. [Google Scholar] [CrossRef] [PubMed]
- Ziyatdinova, G.K.; Budnikov, H.C. Natural phenolic antioxidants in bioanalytical chemistry: State of the art and prospects of development. Russ. Chem. Rev. 2015, 84, 194–224. [Google Scholar] [CrossRef]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef] [PubMed]
- Della Pelle, F.; Di Crescenzo, M.C.; Sergi, M.; Montesano, C.; Di Ottavio, F.; Scarpone, R.; Scortichini, G.; Compagnone, D. Micro-solid-phase extraction (µ-SPE) of organophosphorous pesticides from wheat followed by LC-MS/MS determination. Food Addit. Contam. Part A 2015, 49, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Di Ottavio, F.; Della Pelle, F.; Montesano, C.; Scarpone, R.; Escarpa, A.; Compagnone, D.; Sergi, M. Determination of pesticides in wheat flour using microextraction on packed sorbent coupled to ultra-high performance liquid chromatography and tandem mass spectrometry. Food Anal. Methods 2017, 10, 1699–1708. [Google Scholar] [CrossRef]
- Valls, J.; Millán, S.; Martí, M.P.; Borràs, E.; Arola, L. Advanced separation methods of food anthocyanins, isoflavones and flavanols. J. Chromatogr. A 2009, 1216, 7143–7172. [Google Scholar] [CrossRef] [PubMed]
- Dobes, J.; Zitka, O.; Sochor, J.; Ruttkay-Nedecky, B.; Babula, P.; Beklova, M.; Kynicky, J.; Hubalek, J.; Klejdus, B.; Kizek, R.; et al. Electrochemical tools for determination compounds in plants. A review. Int. J. Electrochem. Sci. 2013, 8, 4520–4542. [Google Scholar]
- Motilva, M.-J.; Serra, A.; Macià, A. Analysis of food polyphenols by ultra high-performance liquid chromatography coupled to mass spectrometry: An overview. J. Chromatogr. A 2013, 1292, 66–82. [Google Scholar] [CrossRef] [PubMed]
- Escarpa, A.; Gonzalez, M.C. Approach to the content of total extractable phenolic compounds from different food samples by comparison of chromatographic and spectrophotometric methods. Anal. Chim. Acta 2001, 427, 119–127. [Google Scholar] [CrossRef]
- Escarpa, A.; Gonzalez, M.C. An overview of analytical chemistry of phenolic compounds in foods. Crit. Rev. Anal. Chem. 2001, 31, 57–139. [Google Scholar] [CrossRef]
- Sánchez-Rangel, J.C.; Benavides, J.; Heredia, J.B.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. The Folin–Ciocalteu assay revisited: Improvement of its specificity for total phenolic content determination. Anal. Methods 2013, 5, 5990. [Google Scholar] [CrossRef]
- López-Alarcón, C.; Denicola, A. Evaluating the antioxidant capacity of natural products: A review on chemical and cellular-based assays. Anal. Chim. Acta 2013, 763, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.N.; Bristi, N.J.; Rafiquzzaman, M. Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm. J. 2013, 21, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Escarpa, A. Lights and shadows on food microfluidics. Lab Chip 2014, 14, 3213–3224. [Google Scholar] [CrossRef] [PubMed]
- Bleeker, E.A.J.; De Jong, W.H.; Geertsma, R.E.; Groenewold, M.; Heugens, E.H.W.; Koers-jacquemijns, M.; Van De Meent, D.; Popma, J.R.; Rietveld, A.G.; Wijnhoven, S.W.P.; et al. Considerations on the EU definition of a nanomaterial: Science to support policy making. Regul. Toxicol. Pharmacol. 2013, 65, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Del Carlo, M.; Compagnone, D. Recent advances in biosensor technology for food safety. Agro Food Ind. Hi-Tech. 2008, 19, 32–35. [Google Scholar]
- Hayat, A.; Catanante, G.; Marty, J.L. Current trends in nanomaterial-based amperometric biosensors. Sensors 2014, 14, 23439–23461. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, B.; Merkoçi, A. Nanomaterials based biosensors for food analysis applications. Trends Food Sci. Technol. 2011, 22, 625–639. [Google Scholar] [CrossRef]
- Alarcon-Angeles, G.; Álvarez-Romero, G.A.; Merkoci, A. Emerging nanomaterials for analytical detection. Compr. Anal. Chem. 2016, 74, 195–246. [Google Scholar] [CrossRef]
- Zhu, C.; Yang, G.; Li, H.; Du, D.; Lin, Y. Electrochemical sensors and biosensors based on nanomaterials and nanostructures. Anal. Chem. 2015, 87, 230–249. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Lin, Z.; Su, X.; Tang, Z. Application of Au based nanomaterials in analytical science. Nano Today 2017, 12, 64–97. [Google Scholar] [CrossRef]
- Scarano, S.; Pascale, E.; Minunni, M. The early nucleation stage of gold nanoparticles formation in solution as powerful tool for the colorimetric determination of reducing agents: The case of xylitol and total polyols in oral fluid. Anal. Chim. Acta 2017, 993, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Lopatynskyi, A.M.; Lopatynska, O.G.; Guo, L.J.; Chegel, V.I. Localized surface plasmon resonance biosensor-part I: Theoretical study of sensitivity-extended mie approach. IEEE Sens. J. 2011, 11, 361–369. [Google Scholar] [CrossRef]
- Vasilescu, A.; Sharpe, E.; Andreescu, S. Nanoparticle-based technologies for the detection of food antioxidants. Curr. Anal. Chem. 2012, 8, 495–505. [Google Scholar] [CrossRef]
- Choleva, T.G.; Kappi, F.A.; Giokas, D.L.; Vlessidis, A.G. Paper-based assay of antioxidant activity using analyte-mediated on-paper nucleation of gold nanoparticles as colorimetric probes. Anal. Chim. Acta 2015, 860, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Vilela, D.; González, M.C.; Escarpa, A. Nanoparticles as analytical tools for in-vitro antioxidant-capacity assessment and beyond. TrAC Trends Anal. Chem. 2015, 64, 1–16. [Google Scholar] [CrossRef]
- Della Pelle, F.; Vilela, D.; González, M.C.; Lo Sterzo, C.; Compagnone, D.D.; Del Carlo, M.; Escarpa, A. Antioxidant capacity index based on gold nanoparticles formation. Application to extra virgin olive oil samples. Food Chem. 2015, 178, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Vilela, D.; Castañeda, R.; González, M.C.; Mendoza, S.; Escarpa, A. Fast and reliable determination of antioxidant capacity based on the formation of gold nanoparticles. Microchim. Acta 2015, 182, 105–111. [Google Scholar] [CrossRef]
- Della Pelle, F.; González, M.C.; Sergi, M.; Del Carlo, M.; Compagnone, D.; Escarpa, A. Gold nanoparticles-based extraction-free colorimetric assay in organic media: An optical index for determination of total polyphenols in fat-rich samples. Anal. Chem. 2015, 87, 6905–6911. [Google Scholar] [CrossRef] [PubMed]
- Tułodziecka, A.; Szydłowska-Czerniak, A. Development of a novel gold nanoparticle-based method to determine antioxidant capacity of Brassica oilseeds, white flakes and meal. Food Chem. 2016, 208, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Özyürek, M.; Güngör, N.; Baki, S.; Güçlü, K.; Apak, R. Development of a silver nanoparticle-based method for the antioxidant capacity measurement of polyphenols. Anal. Chem. 2012, 84, 8052–8059. [Google Scholar] [CrossRef] [PubMed]
- Teerasong, S.; Jinnarak, A.; Chaneam, S.; Wilairat, P.; Nacapricha, D. Poly(vinyl alcohol) capped silver nanoparticles for antioxidant assay based on seed-mediated nanoparticle growth. Talanta 2017, 170, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Szydlowska-Czerniak, A.; Tulodziecka, A.; Szlyk, E. A silver nanoparticle-based method for determination of antioxidant capacity of rapeseed and its products. Analyst 2012, 137, 3750–3759. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, E.; Frasco, T.; Andreescu, D.; Andreescu, S. Portable ceria nanoparticle-based assay for rapid detection of foodantioxidants (NanoCerac). Analyst 2013, 138, 249–262. [Google Scholar] [CrossRef] [PubMed]
- TułodzieckaSzyd, A.; Szydłowska-Czerniak, A. Determination of total antioxidant capacity of rapeseed and its by-products by a novel cerium oxide nanoparticle-based spectrophotometric method. Food Anal. Methods 2016, 9, 3053–3062. [Google Scholar] [CrossRef]
- Szydłowska-Czerniak, A.; Łaszewska, A.; Tułodziecka, A. A novel iron oxide nanoparticle-based method for the determination of the antioxidant capacity of rapeseed oils at various stages of the refining process. Anal. Methods 2015, 7, 4650–4660. [Google Scholar] [CrossRef]
- Gatselou, V.; Christodouleas, D.C.; Kouloumpis, A.; Gournis, D.; Giokas, D.L. Determination of phenolic compounds using dpectral and color transitions of rhodium nanoparticles. Anal. Chim. Acta 2016, 932, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, E.; Bradley, R.; Frasco, T.; Jayathilaka, D.; Marsh, A.; Andreescu, S. Metal oxide based multisensor array and portable database for field analysis of antioxidants. Sens. Actuators B Chem. 2014, 193, 552–562. [Google Scholar] [CrossRef] [PubMed]
- Hemmateenejad, B.; Shamsipur, M.; Khosousi, T.; Shanehsaz, M.; Firuzi, O. Antioxidant activity assay based on the inhibition of oxidation and photobleaching of l-cysteine-capped CdTe quantum dots. Analyst 2012, 137, 4029. [Google Scholar] [CrossRef] [PubMed]
- Akshath, U.S.; Shubha, L.R.; Bhatt, P.; Thakur, M.S. Quantum dots as optical labels for ultrasensitive detection of polyphenols. Biosens. Bioelectron. 2014, 57, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Dwiecki, K.; Nogala-Kałucka, M.; Polewski, K. Determination of total phenolic compounds in common beverages using CdTe quantum dots. J. Food Process. Preserv. 2017, 41, 1–7. [Google Scholar] [CrossRef]
- Benítez-Martínez, S.; Valcárcel, M. Chemical Graphene quantum dots as sensor for phenols in olive oil. Sens. Actuators B Chem. 2014, 197, 350–357. [Google Scholar] [CrossRef]
- Álvarez-Diduk, R.; Orozco, J.; Merkoçi, A. Paper strip-embedded graphene quantum dots: A screening device with a smartphone readout. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nery, E.W.; Kubota, L.T. Sensing approaches on paper-based devices: A review. Anal. Bioanal. Chem. 2013, 405, 7573–7595. [Google Scholar] [CrossRef] [PubMed]
- Del Carlo, M.; Amine, A.; Haddam, M.; Della Pelle, F.; Fusella, G.C.; Compagnone, D. Selective voltammetric analysis of o-diphenols from olive oil using Na2MoO4 as electrochemical mediator. Electroanalysis 2012, 24, 889–896. [Google Scholar] [CrossRef]
- Moulton, M.C.; Braydich-Stolle, L.K.; Nadagouda, M.N.; Kunzelman, S.; Hussain, S.M.; Varma, R.S. Synthesis, characterization and biocompatibility of “green” synthesized silver nanoparticles using tea polyphenols. Nanoscale 2010, 2, 763. [Google Scholar] [CrossRef] [PubMed]
- Marambio-Jones, C.; Hoek, E.M.V. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J. Nanoparticle Res. 2010, 12, 1531–1551. [Google Scholar] [CrossRef]
- Sharma, V.K.; Yngard, R.A.; Lin, Y. Silver nanoparticles: Green synthesis and their antimicrobial activities. Adv. Colloid Interface Sci. 2009, 145, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Jiong, M.; Cnen, J.Y.; Zhang, Y.; Wang, P.N.; Guo, J.; Yang, W.L.; Wang, C.C. Photochemical instability of thiol-capped CdTe quantum dots in aqueous solution and living cells: Process and mechanism. J. Phys. Chem. B 2007, 111, 12012–12016. [Google Scholar] [CrossRef]
- Petryayeva, E.; Algar, W.R.; Medintz, I.L. Quantum dots in bioanalysis: A review of applications across various platforms for fluorescence spectroscopy and imaging. Appl. Spectrosc. 2013, 67, 215–252. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Ge, Y.; Chen, Q.; Xu, S.; Wang, N.; Zhang, X. Hydrothermal synthesis of highly luminescent CdTe quantum dots by adjusting precursors’ concentration and their conjunction with BSA as biological fluorescent probes. Talanta 2007, 72, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Sochor, J.; Dobes, J.; Krystofova, O.; Ruttkay-Nedecky, B.; Babula, P.; Pohanka, M.; Jurikova, T.; Zitka, O.; Adam, V.; Klejdus, B.; et al. Electrochemistry as a tool for studying antioxidant properties. Int. J. Electrochem. Sci. 2013, 8, 8464–8489. [Google Scholar]
- Barroso, M.F.; De-Los-Santos-Álvarez, N.; Delerue-Matos, C.; Oliveira, M.B.P.P. Towards a reliable technology for antioxidant capacity and oxidative damage evaluation: Electrochemical (bio)sensors. Biosens. Bioelectron. 2011, 30, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hoyos-Arbeláez, J.; Vázquez, M.; Contreras-Calderón, J. Electrochemical methods as a tool for determining the antioxidant capacity of food and beverages: A review. Food Chem. 2017, 221, 1371–1381. [Google Scholar] [CrossRef] [PubMed]
- Enache, T.A.; Oliveira-Brett, A.M. Phenol and para-substituted phenols electrochemical oxidation pathways. J. Electroanal. Chem. 2011, 655, 9–16. [Google Scholar] [CrossRef]
- Ligler, F.S.; White, H.S. Nanomaterials in analytical chemistry. Anal. Chem. 2013, 85, 11161–11162. [Google Scholar] [CrossRef] [PubMed]
- Pumera, M.; Escarpa, A. Nanomaterials as electrochemical detectors in microfluidics and CE: Fundamentals, designs, and applications. Electrophoresis 2009, 30, 3315–3323. [Google Scholar] [CrossRef] [PubMed]
- Blandón-Naranjo, L.; Della Pelle, F.; Vázquez, M.V.; Gallego, J.; Santamaría, A.; Alzate-Tobón, M.; Compagnone, D. Electrochemical behaviour of microwave-assisted oxidized MWCNTs based disposable electrodes: proposal of a NADH electrochemical sensor. Electroanalysis 2018. [Google Scholar] [CrossRef]
- Blandón-Naranjo, L.; Hoyos-Arbeláez, J.; Vázquez, M.V.; Della Pelle, F.; Compagnone, D. NADH oxidation onto different carbon-based sensors: Effect of structure and surface-oxygenated Groups. J. Sens. 2018, in press. [Google Scholar]
- Blasco, A.J.; González Crevillén, A.; González, M.C.; Escarpa, A. Direct electrochemical sensing and detection of natural antioxidants and antioxidant capacity in vitro systems. Electroanalysis 2007, 19, 2275–2286. [Google Scholar] [CrossRef]
- Valdés, M.G.; González, A.C.V.; Calzón, J.A.G.; Díaz-García, M.E. Analytical nanotechnology for food analysis. Microchim. Acta 2009, 166, 1–19. [Google Scholar] [CrossRef]
- Gao, C.; Guo, Z.; Liu, J.-H.; Huang, X.-J. The new age of carbon nanotubes: An updated review of functionalized carbon nanotubes in electrochemical sensors. Nanoscale 2012, 4, 1948. [Google Scholar] [CrossRef] [PubMed]
- Yáñez-Sedeño, P.; Pingarrón, J.M.; Riu, J.; Rius, F.X. Electrochemical sensing based on carbon nanotubes. TrAC Trends Anal. Chem. 2010, 29, 939–953. [Google Scholar] [CrossRef]
- Yang, C.; Denno, M.E.; Pyakurel, P.; Venton, B.J. Recent trends in carbon nanomaterial-based electrochemical sensors for biomolecules: A review. Anal. Chim. Acta 2015, 887, 17–37. [Google Scholar] [CrossRef] [PubMed]
- Souza, C.P.; Legari, F.; Zarbin, A.J.G.; Marcolino-Júnior, L.H.; Bergamini, M.F. Voltammetric determination of the antioxidant capacity in wine samples using a carbon nanotube modified electrode. J. Agric. Food Chem. 2011, 59, 7620–7625. [Google Scholar] [CrossRef] [PubMed]
- Sanchez Arribas, A.; Martinez-Fernandez, M.; Moreno, M.; Bermejo, E.; Zapardiel, A.; Chicharro, M. Analysis of total polyphenols in wines by FIA with highly stable amperometric detection using carbon nanotube-modified electrodes. Food Chem. 2013, 136, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Ziyatdinova, G.; Salikhova, I.; Budnikov, H. Chronoamperometric estimation of cognac and brandy antioxidant capacity using MWNT modified glassy carbon electrode. Talanta 2014, 125, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Ziyatdinova, G.; Aytuganova, I.; Nizamova, A.; Budnikov, H. Differential Pulse Voltammetric Assay of Coffee Antioxidant Capacity with MWNT-Modified Electrode. Food Anal. Methods 2013, 6, 1629–1638. [Google Scholar] [CrossRef]
- Ziyatdinova, G.; Kozlova, E.; Budnikov, H. Chronocoulometry of wine on multi-walled carbon nanotube modified electrode: Antioxidant capacity assay. Food Chem. 2016, 196, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Eguílaz, M.; Gutierrez, A.; Gutierrez, F.; Gonzalez-Domínguez, J.M.; Ansón-Casaos, A.; Hernández-Ferrer, J.; Ferreyra, N.F.; Martínez, M.T.; Rivas, G. Covalent functionalization of single-walled carbon nanotubes with polytyrosine: Characterization and analytical applications for the sensitive quantification of polyphenols. Anal. Chim. Acta 2016, 909, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Crevillén, A.G.; Pumera, M.; González, M.C.; Escarpa, A. Towards lab-on-a-chip approaches in real analytical domains based on microfluidic chips/electrochemical multi-walled carbon nanotube platforms. Lab Chip 2009, 9, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Vilela, D.; Martín, A.; González, M.C.; Escarpa, A. Fast and reliable class-selective isoflavone index determination on carbon nanotube press-transferred electrodes using microfluidic chips. Analyst 2014, 139, 2342–2347. [Google Scholar] [CrossRef] [PubMed]
- Talarico, D.; Arduini, F.; Constantino, A.; Del Carlo, M.; Compagnone, D.; Moscone, D.; Palleschi, G. Electrochemistry communications carbon black as successful screen-printed electrode modifier for phenolic compound detection. Electrochem. Commun. 2015, 60, 78–82. [Google Scholar] [CrossRef]
- Della Pelle, F.; Di Battista, R.; Vázquez, L.; Palomares, F.J.; Del Carlo, M.; Sergi, M.; Escarpa, A. Press-transferred carbon black nanoparticles for class-selective antioxidant electrochemical detection. Appl. Mater. Today 2017, 9, 29–36. [Google Scholar] [CrossRef]
- Raymundo-Pereira, P.A.; Campos, A.M.; Prado, T.M.; Furini, L.N.; Boas, N.V.; Calegaro, M.L.; Machado, S.A.S. Synergy between Printex nano-carbons and silver nanoparticles for sensitive estimation of antioxidant activity. Anal. Chim. Acta 2016, 926, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Hui, K.H.; Ambrosi, A.; Pumera, M.; Bonanni, A. Improving the analytical performance of graphene oxide towards the assessment of polyphenols. Chem. A Eur. J. 2016, 22, 3830–3834. [Google Scholar] [CrossRef] [PubMed]
- Chng, C.E.; Sofer, Z.; Pumera, M.; Bonanni, A. Doped and undoped graphene platforms: The influence of structural properties on the detection of polyphenols. Sci. Rep. 2016, 6, 20673. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Zheng, D.; Tanaka, H.; Zhan, F.; Yuan, X.; Gao, F.; Wang, Q. An electrochemical sensor for gallic acid based on Fe2O3/electro-reduced graphene oxide composite: Estimation for the antioxidant capacity index of wines. Mater. Sci. Eng. C 2015, 57, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Tirawattanakoson, R.; Rattanarat, P.; Ngamrojanavanich, N. Free radical scavenger screening of total antioxidant capacity in herb and beverage using graphene/PEDOT: PSS-modified electrochemical sensor. JEAC 2016, 767, 68–75. [Google Scholar] [CrossRef]
- Magro, M.; Bonaiuto, E.; Baratella, D.; de Almeida Roger, J.; Jakubec, P.; Corraducci, V.; Tuček, J.; Malina, O.; Zbořil, R.; Vianello, F. Electrocatalytic nanostructured ferric tannates: Characterization and application of a polyphenol nanosensor. ChemPhysChem 2016, 17, 3196–3203. [Google Scholar] [CrossRef] [PubMed]
- Andrei, V.; Sharpe, E.; Vasilescu, A.; Andreescu, S. A single use electrochemical sensor based on biomimetic nanoceria for the detection of wine antioxidants. Talanta 2016, 156–157, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.S.; Shanmugam, R.; Nellaiappan, S. Chemical Tea quality assessment by analyzing key polyphenolic functional groups using flow injection analysis coupled with a dual electrochemical detector. Sens. Actuators B Chem. 2016, 227, 352–361. [Google Scholar] [CrossRef]
- Cetó, X.; Céspedes, F.; Pividori, M.I.; Gutiérrez, J.M.; del Valle, M. Resolution of phenolic antioxidant mixtures employing a voltammetric bio-electronic tongue. Analyst 2012, 137, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Arduini, F.; Amine, A.; Majorani, C.; Di Giorgio, F.; De Felicis, D.; Cataldo, F.; Moscone, D.; Palleschi, G. High performance electrochemical sensor based on modified screen-printed electrodes with cost-effective dispersion of nanostructured carbon black. Electrochem. Commun. 2010, 12, 346–350. [Google Scholar] [CrossRef]
- Silva, T.A.; Moraes, F.C.; Janegitz, B.C.; Fatibello-Filho, O.; Lu, R.W. Electrochemical biosensors based on nanostructured carbon black: A review. J. Nanomater. 2017, 2017, 4571614. [Google Scholar] [CrossRef]
- Arduini, F.; Cinti, S.; Scognamiglio, V.; Moscone, D. Nanomaterials in electrochemical biosensors for pesticide detection: Advances and challenges in food analysis. Microchim. Acta 2016, 183, 2063–2083. [Google Scholar] [CrossRef]
- Della Pelle, F.; Vázquez, L.; Del Carlo, M.; Sergi, M.; Compagnone, D.; Escarpa, A. Press-printed conductive carbon clack nanoparticle films for molecular detection at the microscale. Chem. A Eur. J. 2016, 22, 12761–12766. [Google Scholar] [CrossRef] [PubMed]
- Della Pelle, F.; Del Carlo, M.; Sergi, M.; Compagnone, D.; Escarpa, A. Press-transferred carbon black nanoparticles on board of microfluidic chips for rapid and sensitive amperometric determination of phenyl carbamate pesticides in environmental samples. Microchim. Acta 2016, 183, 3143–3149. [Google Scholar] [CrossRef]
- Gan, T.; Hu, S. Electrochemical sensors based on graphene materials. Microchim. Acta 2011, 175, 1–19. [Google Scholar] [CrossRef]
- Martín, A.; Escarpa, A. Graphene: The cutting-edge interaction between chemistry and electrochemistry. TrAC Trends Anal. Chem. 2014, 56, 13–26. [Google Scholar] [CrossRef]
- Del Carlo, M.; Capoferri, D.; Gladich, I.; Guida, F.; Forazato, C.; Navarini, L.; Compagnone, D.; Laio, A.; Berti, F. In silico design of short peptides as sensing elements for phenolic compounds. ACS Sens. 2016, 1, 279–286. [Google Scholar] [CrossRef]
- Ziyatdinova, G.; Galandova, J.; Labuda, J. Impedimetric nanostructured disposable DNA-based biosensors for the detection of deep DNA damage and effect of antioxidants. Int. J. Electrochem. Sci. 2008, 3, 223–235. [Google Scholar]
- Del Carlo, M.; Compagnone, D. Recent strategies for the biological sensing of pesticides: From the design to the application in real samples. Bioanal. Rev. 2010, 1, 159–176. [Google Scholar] [CrossRef]
- Gul, I.; Ahmad, M.S.; Naqvi, S.M.S.; Hussain, A.; Wali, R.; Farooqi, A.A.; Ahmed, I. Polyphenol oxidase (PPO) based biosensors for detection of phenolic compounds: A Review. J. Appl. Biol. Biotechnol. 2017, 5, 72–85. [Google Scholar] [CrossRef]
- Diaconu, M.; Litescu, S.C.; Radu, G.L. Laccase-MWCNT-chitosan biosensor—A new tool for total polyphenolic content evaluation from in vitro cultivated plants. Sens. Actuators B Chem. 2010, 145, 800–806. [Google Scholar] [CrossRef]
- Di Fusco, M.; Tortolini, C.; Deriu, D.; Mazzei, F. Laccase-based biosensor for the determination of polyphenol index in wine. Talanta 2010, 81, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Rawal, R.; Chawla, S.; Pundir, C.S. Polyphenol biosensor based on laccase immobilized onto silver nanoparticles/multiwalled carbon nanotube/polyaniline gold electrode. Anal. Biochem. 2011, 419, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Chawla, S.; Rawal, R.; Sharma, S.; Pundir, C.S. An amperometric biosensor based on laccase immobilized onto nickel nanoparticles/carboxylated multiwalled carbon nanotubes/polyaniline modified gold electrode for determination of phenolic content in fruit juices. Biochem. Eng. J. 2012, 68, 76–84. [Google Scholar] [CrossRef]
- Eremia, S.A.V.; Vasilescu, I.; Radoi, A.; Litescu, S.C.; Radu, G.L. Disposable biosensor based on platinum nanoparticles-reduced graphene oxide-laccase biocomposite for the determination of total polyphenolic content. Talanta 2013, 110, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Lanzellotto, C.; Favero, G.; Antonelli, M.L.; Tortolini, C.; Cannistraro, S.; Coppari, E.; Mazzei, F. Nanostructured enzymatic biosensor based on fullerene and gold nanoparticles: Preparation, characterization and analytical applications. Biosens. Bioelectron. 2014, 55, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Vasilescu, I.; Eremia, S.A.V.; Kusko, M.; Radoi, A.; Vasile, E.; Radu, G.L. Molybdenum disulphide and graphene quantum dots as electrode modifiers for laccase biosensor. Biosens. Bioelectron. 2016, 75, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Vlamidis, Y.; Gualandi, I.; Tonelli, D. Amperometric biosensors based on reduced GO and MWCNTs composite for polyphenols detection in fruit juices. J. Electroanal. Chem. 2017, 799, 285–292. [Google Scholar] [CrossRef]
- Nadifiyine, S.; Haddam, M.; Mandli, J.; Chadel, S.; Blanchard, C.C.; Marty, J.L.; Amine, A. Amperometric biosensor based on tyrosinase immobilized onto a carbon black paste electrode for phenol determination in olive oil. Anal. Lett. 2013, 46, 2705–2726. [Google Scholar] [CrossRef]
- Rodríguez-Delgado, M.M.; Alemán-Nava, G.S.; Rodríguez-Delgado, J.M.; Dieck-Assad, G.; Martínez-Chapa, S.O.; Barceló, D.; Parra, R. Laccase-based biosensors for detection of phenolic compounds. TrAC Trends Anal. Chem. 2015, 74, 21–45. [Google Scholar] [CrossRef]
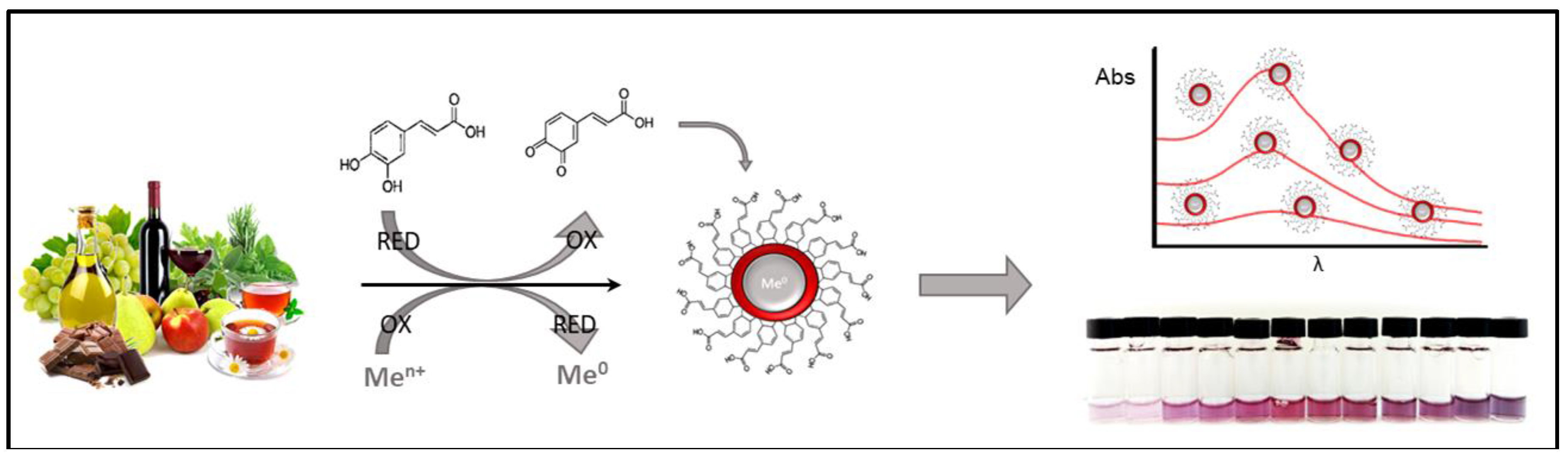
| Work Aim | Nanomaterial | Food Matrix | Sample Pretreatment | Method Principles | Detection Mechanism | Ref. |
|---|---|---|---|---|---|---|
| AOC samples comparison | AuNPs | Fruit extracts | LLE | Au reduction, mild condition | Optical: LSPR | [47] |
| AOC new index proposal/sample evaluation | AuNPs | Olive oil | SPE | Au reduction, mild condition | Optical: LSPR | [46] |
| TP new index proposal/sample evaluation | AuNPs | Olive oil, Chocolate | No required (sample dilution in DMSO) | Au reduction in fat matrix, DMSO strategic solvent | Optical: LSPR | [48] |
| AOC samples evaluation | AuNPs | Rapeseed | LLE | Au reduction at pH 4.6 | Optical: LSPR | [49] |
| AOC paper based sensor realization | AuNPs | Tea, Wine | Infusion/No required | Au reduction on paper | Visual, Optical (LSPR) | [44] |
| AOC sample evaluation | AgNPs | Fruit juices, Olive Oil | Infusion/LLE | AgNPs seed-growth | Optical (LSPR) | [50] |
| AOC sample evaluation | AgNPs | Ginger | LLE | AgNPs seed-growth | Optical (LSPR) | [51] |
| AOC samples evaluation | AgNPs | Rapeseed | LLE | Ag reduction, 1 h | Optical (LSPR) | [52] |
| AOC evaluation | Ceria NPs | Teas, medicinal mushrooms | Infusion | Ce(IV) reduction and ceria NPs colour inhibition, both on paper | Visual, Optical (LSPR) | [53] |
| AOC sample evaluation | Cerium oxide NPs | Rapeseed and its By-Products | LLE | Ce(IV) reduction, mild condition 60 min | Optical (LSPR) | [54] |
| AOC sample evaluation | Iron oxide NPs | Rapessed oil | LLE | Fe (III) reduction, mild condition 50 min | Optical (LSPR) | [55] |
| AOC and total catechins evaluation | Rhodium NPs | Teas | Infusion | RhNPS LSPR shifting | Optical (LSPR) | [56] |
| Polyphenols evaluation | Ti(IV)oxo, ZnO, SiO2, ZrO2, TiO2, Fe2O3, CeO2 nanoparticles | Green Tea | Infusion | MNPs-oxides surface complexation with polyphenols | Visual color changes | [57] |
| TP estimation | CdTe QDs | Teas | Infusion | CdTe QDs fluorescence quenching inhibition | Fluorescence | [58] |
| Fluorescence | CdTe QDs | Plant extracts | LLE | CdTe QDs laccase mediated/fluorescence quenching | Fluorescence | [59] |
| TP estimation | CdTe–sodium periodate | Tea, Herbal infusions | Infusion | CdTe QDs fluorescence quenching inhibition | Fluorescence | [60] |
| TP estimation | Graphene QDs | Olive oil extracts | LLE | Graphene QDs quenching | Fluorescence | [61] |
| AOC estimation | Graphene QD | Wine samples | Dilution | Graphene QDs quenching on paper | Fluorescence | [62] |
| Work Aim | Nanomaterial | Food Matrix | Sample Pretreatment | Method Principles | Detection Mechanism | Linear Range/LOD | Ref. |
|---|---|---|---|---|---|---|---|
| AOC sample evaluation | SWCNTs | Red/white wine | No required (sample dilution) | Polyphenols ox. at GCE-SWCNTs | DPV | Gallic acid: 5.0 × 10−7 to 1.5 × 10−5; 3.0 × 10−7 mol L−1 | [84] |
| TP estimation | MWCNTs | Red/white wine | No required (sample dilution) | Polyphenols ox. at GCE-MWCNTs | FIA-amperometric | Phenolic acids: 1.0 × 10−7 to 1.0 × 10−4 mol L−1 | [85] |
| o-Diphenol evaluation | MWCNTs-NH2-Mo | Olive oil | SPE | o-diphenols selective oxidation at SPE-Mo/MWCNTs-NH2 | FIA-amperometric | Catechol: 0.2–5.0 × 101 mg L−1 | [64] |
| AOC evaluation | MWCNTs | Cognac and brandie | No required (sample dilution) | Polyphenols ox. at GCE-MWCNTs | Amperometry | Ellagic acid: 0.7 × 10−6–5.3 × 10−5 mol L−1; 0.2 × 10−6 mol L−1 | [86] |
| AOC evaluation | MWCNTs | Coffee beans and infusions | Infusion | Polyphenols ox. at GCE-MWCNTs | DPV | Chlorogenic acid: 1.0 × 10−6 – 1.1 × 10−3 mol L−1; 0.21 × 10−7 mol L−1 | [87] |
| AOC evaluation | MWCNTs | Red/white wine | No required (sample dilution) | Polyphenols ox. at GCE-MWCNTs | Chronocoulometry | Gallic acid: 5.2 × 10−7–2.9 × 10−4 mol L−1; 1.4 × 10−7 mol L−1 | [88] |
| TP estimation | MWCNTs | Tea | Infusion | Polyphenols ox. at GCE-SWCNT/Polytyr | Amperometry | Gallic acid: 5.0 × 10−7 and 1.7 × 10−4 mol L−1; 8.8 × 10−9 mol L−1 | [89] |
| Total isoflavones and evaluation of polyphenols profiles. | SPE-MWCNTs | dietary supplement/apples and pears extracts | No required (sample dilution)/LLE | Chip flow injection/separation ox at and channel SPE-MWCNTs | Chip flow injection and separation/amperometric detection | Genistein: 0.3–1.5 × 101 mg L−1; 0.4 × 10−1 mg L−1. Phloridzin: 1.0 × 101–1.5 mg 102 L−1; 1.2 mg L−1 | [90] |
| Total glycosides (TG) and total aglycones (TA) Isoflavones | SWCNTs | soy extracts | LLE | Chip separation ox at and channel Press-produced MWCNTs transducer | Chip separation/amperometric detection | Genistein and Daidzein: 2.0 × 10−5–5.0 × 10−4 mol L−1; 2.0 × 10−5 mol L−1 | [91] |
| Polyphenols studies | CB | - | - | Polyphenols ox. at SPE-CB | SWV | Gallic acid: 1.0 × 10−5–1.0 × 10−4 mol L−1; 1.0 × 10−6 mol L−1 | [92] |
| AOC evaluation of o-diphenols and m-phenols | CB | Olive oil | SPE | Polyphenols ox. at a Press-produced CB transducer | DPV | Tyrosol and hydroxytrosol: 1.0 × 10−5–7.5 × 10−5 mol L−1; 6.0 × 10−6 and 2.0 × 10−5 mol L−1, respectively | [93] |
| AOC evaluation | CB (Printex L6 carbon)-AgNPs | wine | No required (sample dilution) | Polyphenols ox. at GCE-CB/AgNPs | DPV | Gallic acid: 5.0 × 10−7–8.5 × 10−6 mol L−1; 6.6 × 10−8 mol L−1 | [94] |
| AOC evaluation | GR-boron-doped | Tea | Infusion | Polyphenols ox. at GCE-GR/boron doped | DPV | Gallic acid: 1.2 × 10−6–1.2 × 10−5 mol L−1 | [95] |
| AOC evaluation | GR-thermally reduced | Beer | No required (sample dilution) | Polyphenols ox. at GCE-GR/thermally reduced | DPV | Catechin: 1.2 × 10−6–1.2 × 10−5 mol L−1 | [96] |
| AOC evaluation | GR reduced-Fe2O3/Chitosan | Red/white wine | No required (sample dilution) | Polyphenols ox. at GCE-GR reduced-Fe2O3/Chit | DPV | Gallic acid: 1.0 × 10−6 mol L−1–1.0 × 10−4 mol L−1 ; 1.5 × 10−7 mol L−1 | [97] |
| AOC evaluation | GR-PEDOT-poly (styrenesulfonate) | herbs and herbal beverages | LLE/Dilution | Indirect evaluation via DPPH• ox. at SPE-GR/PEDOT/PSS | Chronoamperometry | Trolox: 5.0 × 10−6–3.0 × 10−5 mol L−1; 0.6 × 10−6 mol L−1 | [98] |
| Polyphenols evaluation (proof) | Maghemite NPs-tannic acid | blueberry | LLE | Polyphenols ox. at CPE-SAMN/TA | SWV | Hydroquinone: 2.5 × 10−5–5.0 × 10−4 mol L−1; 8.6 × 10−6 mol L−1 | [99] |
| AOC evaluation (o-diphenols) | Cerium (IV)oxide NPs | Red/white wine | No required (sample dilution) | Polyphenols ox at quinines, quinines reduction at SPE-CeO (IV)NPs | Amperometry | Gallic acid: 2.0 × 10−6–2.0 × 10−5 mol L−1; 1.5 × 10−6 mol L−1 | [100] |
| Polyphenols content double index | graphitized mesoporous carbon/Chitosan | Tea | Infusion | Polyphenols ox Polyphenols ox at GCE-Chit/GMC at two potentials | FIA-amperometric(double potential) | 1,2,3-THB, and (1,2,3-THB, 1,2-DHB,1,3-DHB): 0.1 × 10−6–1.0 × 10−4 mol L−1; 1.1 × 10−6 mol L−1 and 0.9 × 10−6 mol L−1, respectively | [101] |
| Polyphenols evaluation/resolve phenolic mixture | copper nanoparticles | Wine | No required (sample dilution) | epoxy–graphite-(bio-)electronic array | CV data input for artificial neural network | - | [102] |
| Work Aim | Nanomaterial | Enzyme/Biological Element | Immobilization | Food Matrix | Sample Pretreatment | Principle | Linear Range/LOD | Ref. |
|---|---|---|---|---|---|---|---|---|
| TP estimation | MWCNT-Chitosan | Laccase from Trametes versicolor | MWCNT/Chitosan electrodeposition entrapping | Sage and mint | LLE | Laccase phenols derived quinones Amperometric reduction at Gold sheet-MWCNT/CS | Rosmarinic acid: 9.1 × 10−7; 1.2 ×10−5; 2.3 × 10−7 mol·L−1 | [114] |
| TP index | SWCNT/MWNCT | Laccases from Trametes versicolor (TvL) and Trametes hirsuta (ThL) | PAP cross-linking | Red and white wine | No required (sample dilution) | Laccase phenols derived quinones FIA-Amperometric reduction at SPE-SWCNT | Gallic Acid: 0.1–1.7 × 101 mg·L−1; 0.1 mg·L−1 | [115] |
| TP evaluation | C-MWCNT-PANI/AgNPs | laccase from Ganoderma sp. | Covalent onto AgNPs/C-MWCNT/PANI | Tea leaves, alcoholic beverages, pharmaceutical formulations | Infusion/no required (sample dilution) | Laccase phenols derived quinones Amperometric reduction at gold electro-C-MWCNT-PANI/AgNPs | Guaiacol: 1.0 × 10−5–5.0 × 10−4 mol·L−1; 5.0 × 10−8 mol·L−1 | [116] |
| TP evaluation | C-MWCNT-PANI/NiNPs | laccase from Ganoderma sp. | Covalent onto NiNPs/C-MWCNT/PANI | Fruit juices | No required (sample dilution) | Laccase phenols derived quinones CV reduction at gold electrdo-C-MWCNT-PANI/NiNPs | Guaiacol: 1.0 × 10−5–5.0 × 10−4 mol·L−1; 5.0 × 10−8 mol·L−1 | [117] |
| TP evaluation | GRO-PtNPs | Laccase from: Trametes versicolor | Nafion entrapping | tea infusions | Infusion/ | Laccase phenols derived quinones Amperometric reduction at SPE-GRO/PtNPs/Nafion | Caffeic acid: 2.0 × 10−7–2.0 × 10−6 mol·L−1; 9.0 × 10−8 mol·L−1 | [118] |
| TP evaluation | Fullerene-AuNPs | Laccase from: Trametes versicolor | Covalent onto Au-SAM/AuNPs-Linker/Fullerenols/TvL | Red and white wine | No required (sample dilution) | Laccase phenols derived quinones FIA-amperometric reduction at gold electrode-SAM/AuNPs Linker/Fullerenols | Gallic acid: 3.0 × 10−5–3.0 × 10−4 mol·L−1; 6.0 × 10−6 mol·L−1 | [119] |
| TP evaluation | GRQDs-MoS2/nanoflakes | Laccase from: Trametes versicolor | Electrostatic interaction laccase/GRQDs | Red wine | No required (sample dilution) | Laccase phenols derived quinones Amperometric reduction at SPE-GRQDs-MoS2/nanoflakes | caffeic acid: 3.8 × 10−7–1.0 × 10−4 mol·L−1; 3.2 × 10−7 mol·L−1; | [120] |
| TP evaluation | GRO-MWCNTs | Laccase from: Trametes versicolor/tyrosinase from mushroom | BSA reticulated with GA/chitosan entrapping | fruit juices | Centrifugation (sample dilution) | Laccase/tyrosinase phenols derived quinones Amperometric reduction at GCE-GRO/MWCNTs | Catechol: 1.0 × 10−6–3.0 × 10−4 mol·L−1 ; 3.0 × 10−7 mol·L-1 | [121] |
| TP evaluation | CB | Tyrosinase from mushroom | Entrapping | Olive oil | LLE | Laccase phenols derived quinones Amperometric reduction at Carbon paste electrode-CB | Catechol: 1.25 × 10−8 to 1.5 × 10−4 mol·L−1; of 6.0 × 10−9 | [122] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Della Pelle, F.; Compagnone, D. Nanomaterial-Based Sensing and Biosensing of Phenolic Compounds and Related Antioxidant Capacity in Food. Sensors 2018, 18, 462. https://doi.org/10.3390/s18020462
Della Pelle F, Compagnone D. Nanomaterial-Based Sensing and Biosensing of Phenolic Compounds and Related Antioxidant Capacity in Food. Sensors. 2018; 18(2):462. https://doi.org/10.3390/s18020462
Chicago/Turabian StyleDella Pelle, Flavio, and Dario Compagnone. 2018. "Nanomaterial-Based Sensing and Biosensing of Phenolic Compounds and Related Antioxidant Capacity in Food" Sensors 18, no. 2: 462. https://doi.org/10.3390/s18020462
APA StyleDella Pelle, F., & Compagnone, D. (2018). Nanomaterial-Based Sensing and Biosensing of Phenolic Compounds and Related Antioxidant Capacity in Food. Sensors, 18(2), 462. https://doi.org/10.3390/s18020462







