Simultaneous Detection of Ammonium and Nitrate in Environmental Samples Using on Ion-Selective Electrode and Comparison with Portable Colorimetric Assays
Abstract
1. Introduction
2. Experimental
2.1. Reagents
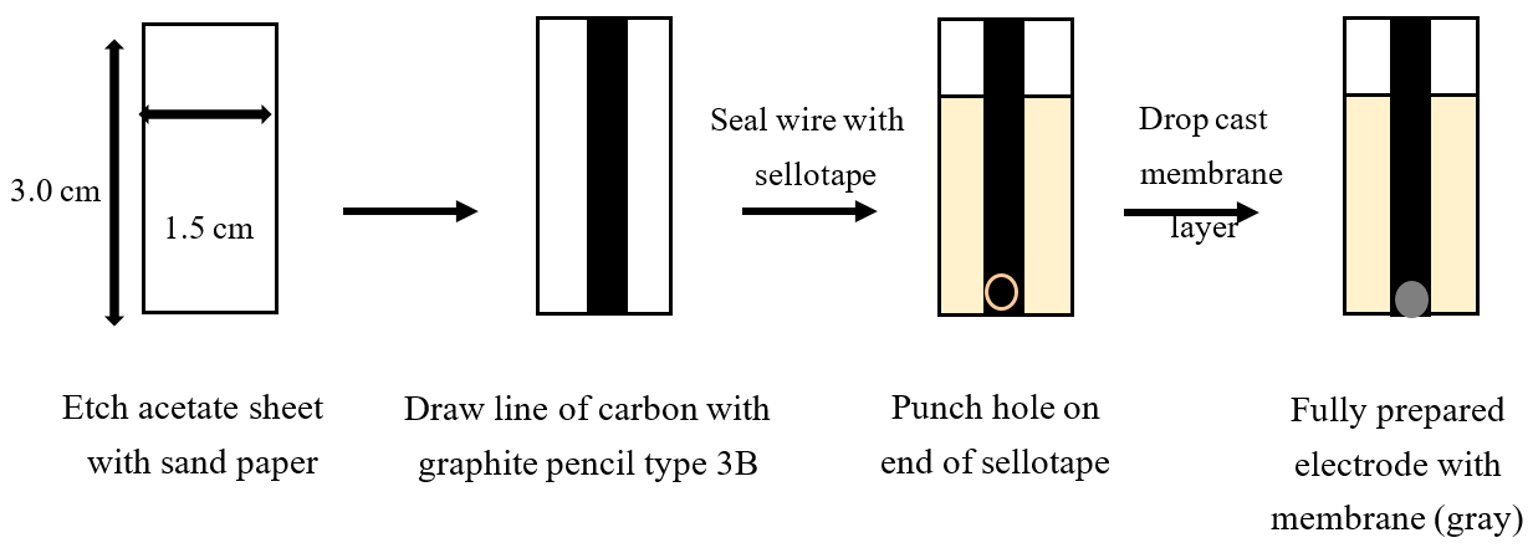
2.2. Preparation of Pencil-Drawn Electrodes
2.3. Preparation of Sensing Membrane for Nitrate and Ammonium Detection
2.4. EMF Measurements
2.5. Analysis of NO3− and NH4+ in Soil and Water Samples Using ISEs
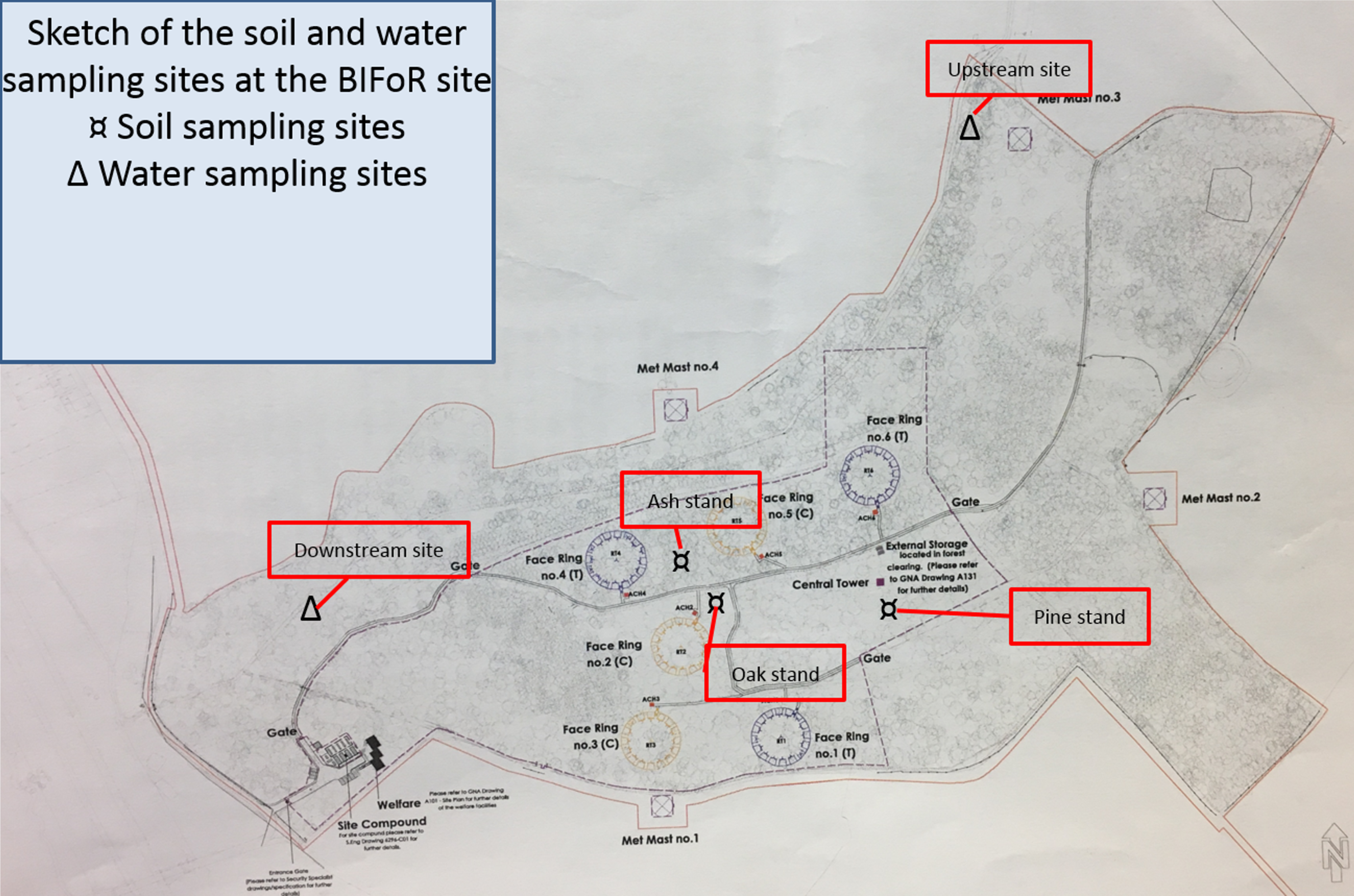
2.5.1. Study Sites and Sampling
2.5.2. Background Soil Analysis
2.5.3. Extraction Procedure of NH4+ and NO3− from Soil
2.5.4. Analysis of NH4+ and NO3− Using ISEs
2.6. Analysis of NO3− and NH4+ in Water and Soil Samples Using Portable Colorimetric Assays
2.7. Selectivity Measurements
3. Results and Discussion
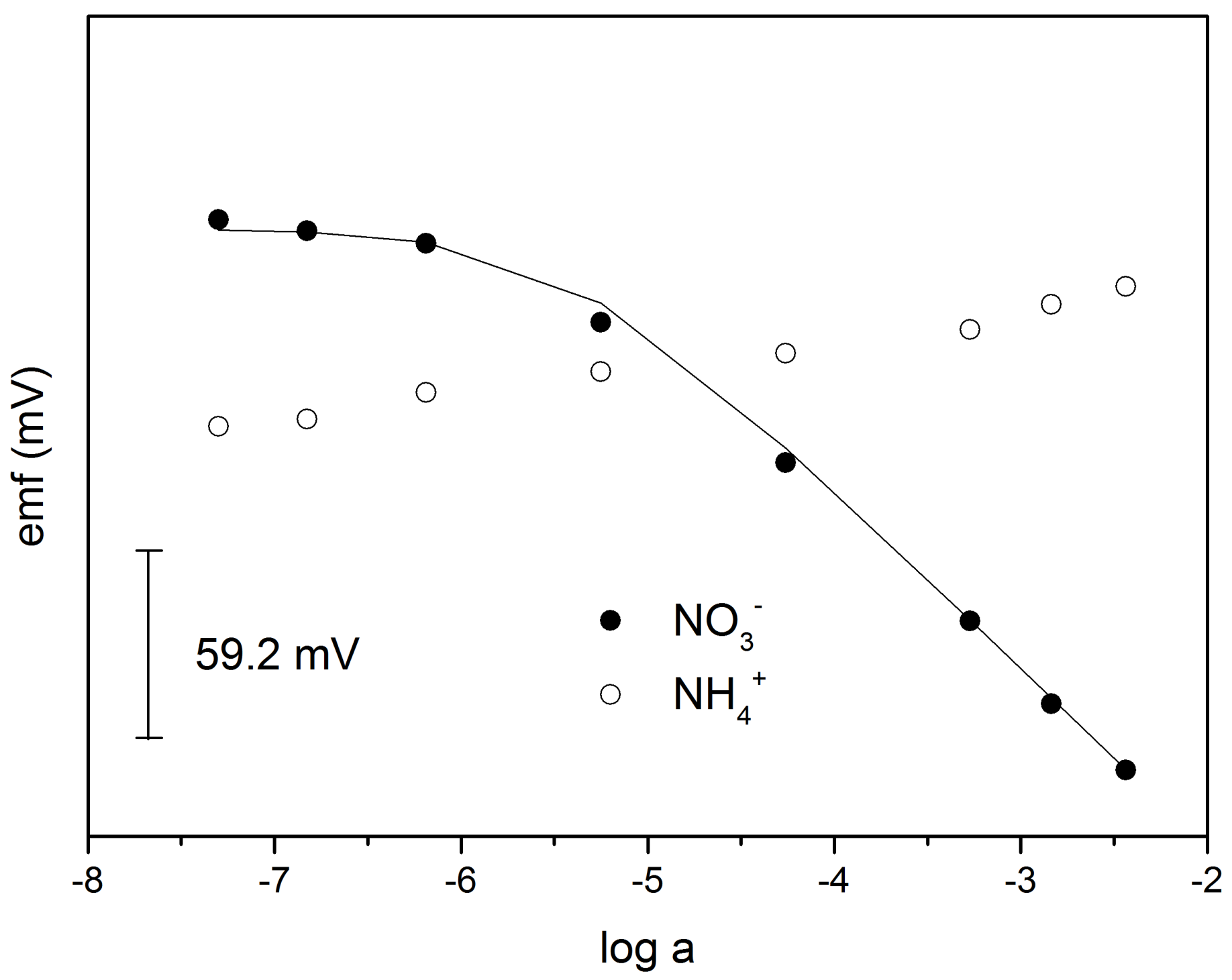
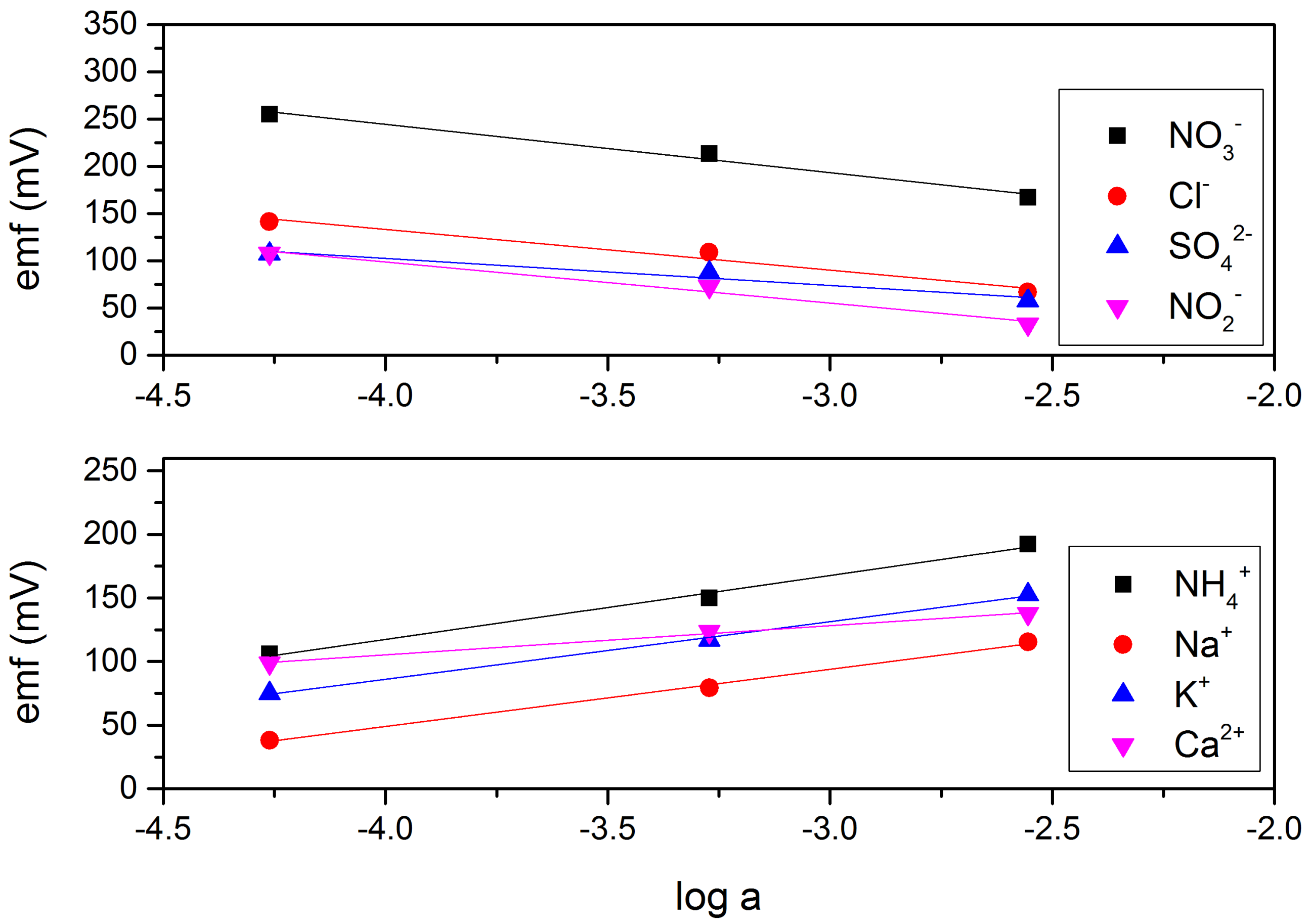
3.1. The Role of ILs in ISE
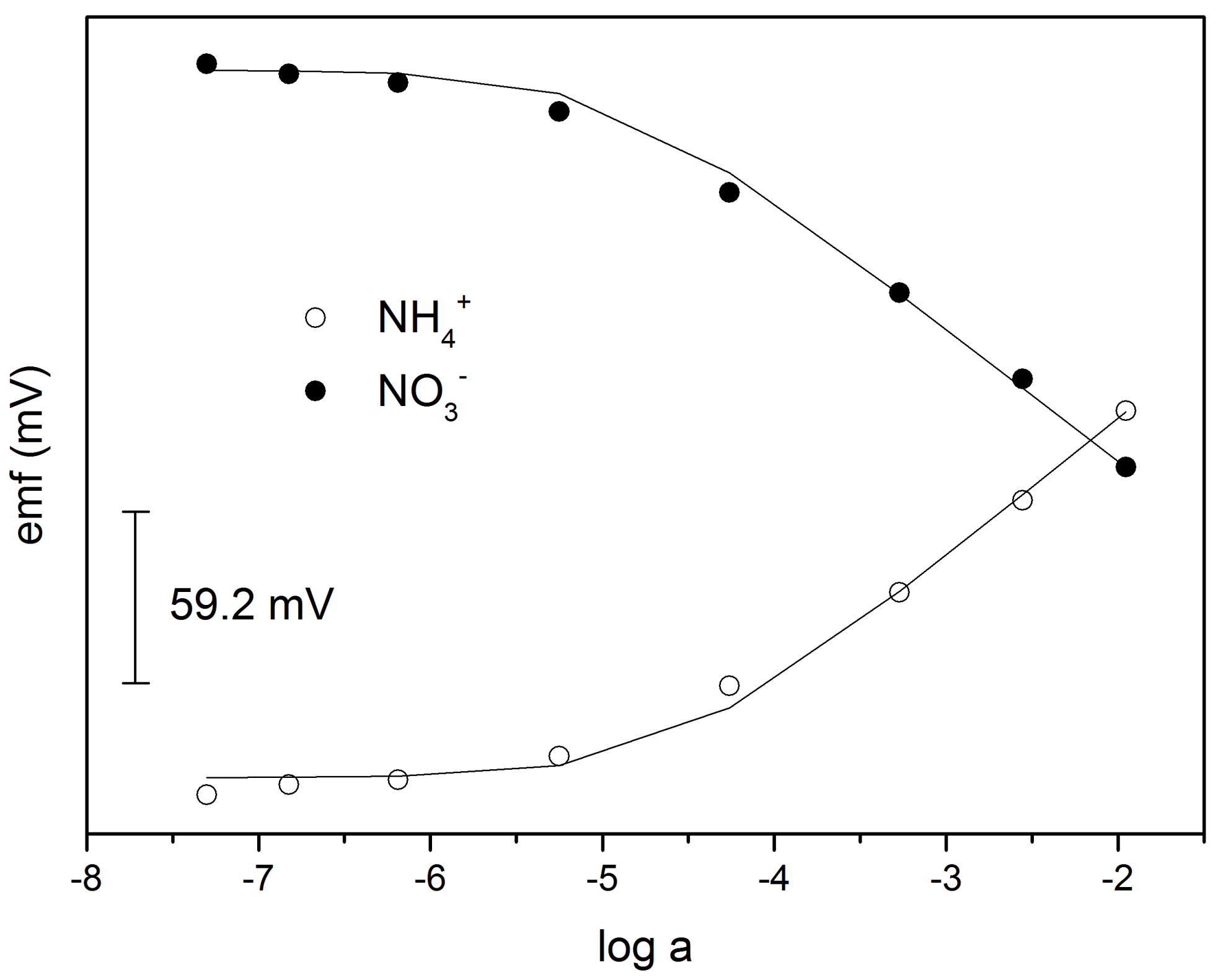
3.2. MMA-DMA—Based ISE and Determination of NO3− and NH4+ in Water and Soil Samples
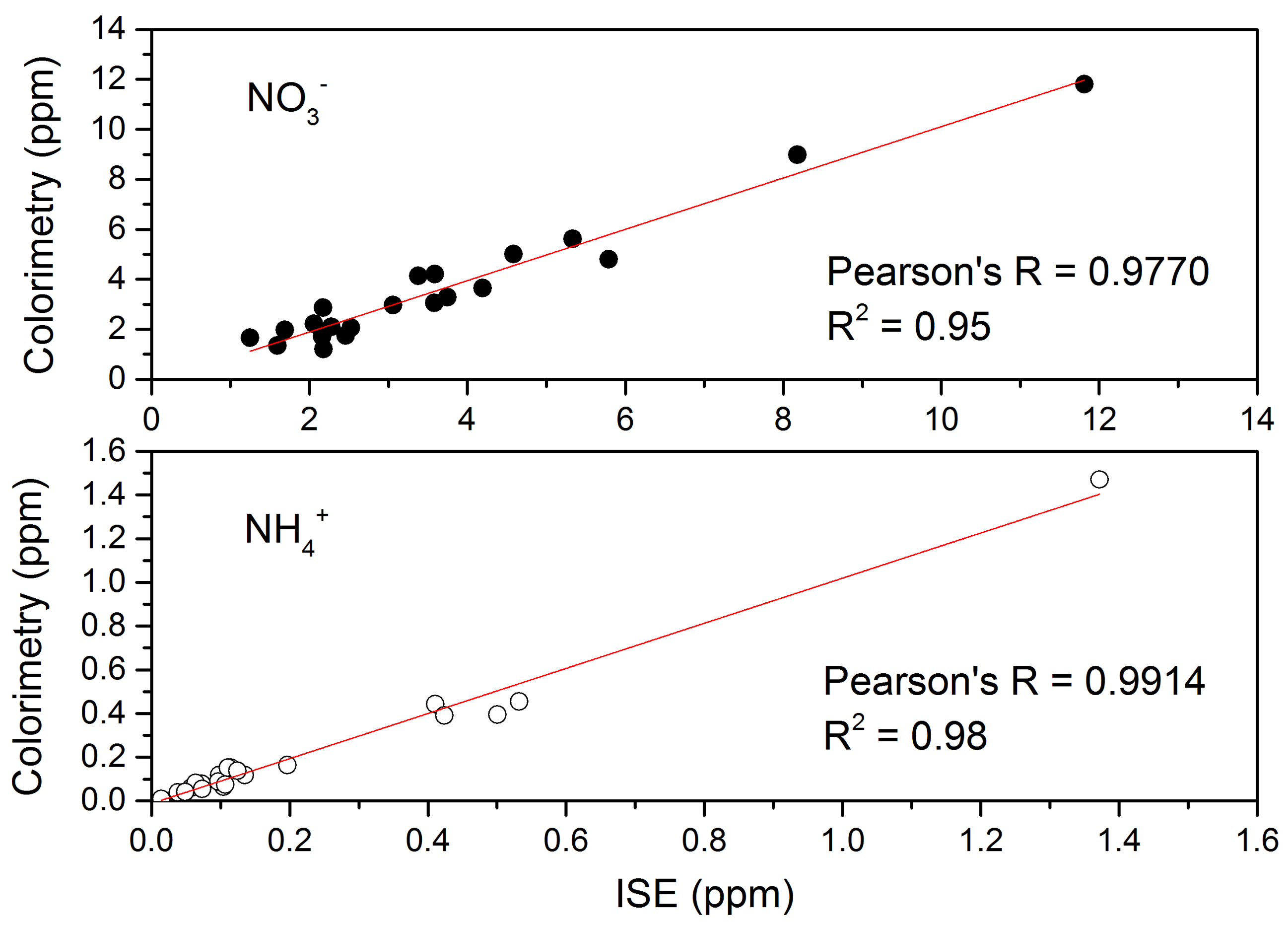
3.3. Application of MMA-DMA—Based ISEs in Environmental Analysis and Comparison to Colorimetric Assay
3.3.1. Sample Preparation
3.3.2. Sample Analysis
3.3.3. Precision and Efficiency
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sutton, M.A.; Bleeker, A.; Howard, C.M.; Erisman, J.W.; Abrol, Y.P.; Bekunda, M.; Datta, A.; Davidson, E.; de Vries, W.; Oenema, O.; et al. Our Nutrient World: The Challenge to Produce More Food and Energy With Less Pollution; Centre for Ecology & Hydrology: Lancaster, UK, 2013; ISBN 978-1-906698-40-9. [Google Scholar]
- Wang, Z.H.; Miao, Y.; Li, S.X. Wheat responses to ammonium and nitrate N applied at different sown and input times. Field Crops Res. 2016, 199, 10–20. [Google Scholar] [CrossRef]
- García-Robledo, E.; Corzo, A.; Papaspyrou, S. A fast and direct spectrophotometric method for the sequential determination of nitrate and nitrite at low concentrations in small volumes. Mar. Chem. 2014, 162, 30–36. [Google Scholar] [CrossRef]
- Tang, I.H.; Sundari, R.; Lintang, H.O.; Yuliati, L. Detection of nitrite and nitrate ions in water by graphene oxide as a potential fluorescence sensor. IOP Conf. Ser. Mater. Sci. Eng. 2016, 107, 012027. [Google Scholar] [CrossRef]
- Martínková, E.; Křžek, T.; Coufal, P. Determination of nitrites and nitrates in drinking water using capillary electrophoresis. Chem. Pap. 2014, 68. [Google Scholar] [CrossRef]
- Caron, W.O.; Lamhamedi, M.S.; Viens, J.; Messaddeq, Y. Practical Application of Electrochemical Nitrate Sensor under Laboratory and Forest Nursery Conditions. Sensors 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- López Pasquali, C.E.; Fernández Hernando, P.; Durand Alegría, J.S. Spectrophotometric simultaneous determination of nitrite, nitrate and ammonium in soils by flow injection analysis. Anal. Chim. Acta 2007, 600, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Diamond, D.; Coyle, S.; Scarmagnani, S.; Hayes, J. Wireless Sensor Networks and Chemo-/Biosensing. Chem. Rev. 2008, 108, 652–679. [Google Scholar] [CrossRef] [PubMed]
- Fay, C.; Anastasova, S.; Slater, C.; Buda, S.T.; Shepherd, R.; Corcoran, B.; O’Connor, N.E.; Wallace, G.G.; Radu, A.; Diamond, D. Wireless ion-selective electrode autonomous sensing system. IEEE Sens. J. 2011, 11, 2374–2382. [Google Scholar] [CrossRef]
- Radu, A.; Radu, T.; McGraw, C.M.; Dillingham, P.W.; Anastasova-Ivanova, S.; Diamond, D. Ion-Selective Electrodes in Environmental Analysis. J. Serb. Chem. Soc. 2013, 78, 1729–1761. [Google Scholar] [CrossRef]
- Nitrate Sensing in the Soil. Available online: www.cambridgeconsultants.com/insights/nitrate-sensing-in-the-soil (accessed on 26 August 2018).
- Cuartero, M.; Bakker, E. Environmental water analysis with membrane electrodes. Curr. Opin. Electrochem. 2017, 3, 97–105. [Google Scholar] [CrossRef]
- Cuartero, M.; Pankratova, N.; Cherubini, T.; Crespo, G.A.; Massa, F.; Confalonieri, F.; Bakker, E. In Situ Detection of Species Relevant to the Carbon Cycle in Seawater with Submersible Potentiometric Probes. Environ. Sci. Technol. Lett. 2017, 4, 410–415. [Google Scholar] [CrossRef]
- Mendecki, L.; Fayose, T.; Stockmal, K.A.; Wei, J.; Granados-Focil, S.; McGraw, C.M.; Radu, A. Robust and ultrasensitive polymer membrane-based carbonate-selective electrodes. Anal. Chem. 2015, 87, 7515–7518. [Google Scholar] [CrossRef] [PubMed]
- Schazmann, B.; Demey, S.; Ali, Z.W.; Plissart, M.S.; Brennan, E.; Radu, A. Robust, Bridge-less Ion-selective Electrodes with Significantly Reduced Need for Pre-and Post-application Handling. Electroanalysis 2018, 30, 740–747. [Google Scholar] [CrossRef]
- Wang, C.; Yuan, H.; Duan, Z.; Xiao, D. Integrated multi-ISE arrays with improved sensitivity, accuracy and precision. Sci. Rep. 2017, 7, 44771. [Google Scholar] [CrossRef] [PubMed]
- Fayose, T.; Mendecki, L.; Ullah, S.; Radu, A. Single strip solid contact ion selective electrodes on a pencil-drawn electrode substrate. Anal. Methods 2017, 9, 1213–1220. [Google Scholar] [CrossRef]
- Bobacka, J. Conducting Polymer-Based Solid-State Ion-Selective Electrodes. Electroanalysis 2006, 18, 7–18. [Google Scholar] [CrossRef]
- Crespo, G.A.; Macho, S.; Rius, F.X. Ion-Selective Electrodes Using Carbon Nanotubes as Ion-to-Electron Transducers. Anal. Chem. 2008, 80, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Chumbimuni-Torres, K.Y.; Rubinova, N.; Radu, A.; Kubota, L.T.; Bakker, E. Solid contact potentiometric sensors for trace level measurements. Anal. Chem. 2006, 78, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Ivaska, A. Applications of ionic liquids in electrochemical sensors. Anal. Chim. Acta 2008, 607, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Kakiuchi, T.; Tsujioka, N.; Kurita, S.; Iwami, Y. Phase-boundary potential across the nonpolarized interface between the room-temperature molten salt and water. Electrochem. Commun. 2003, 5, 159–164. [Google Scholar] [CrossRef]
- Shvedene, N.V.; Chernyshov, D.V.; Khrenova, M.G.; Formanovsky, A.A.; Baulin, V.E.; Pletnev, I.V. Ionic Liquids Plasticize and Bring Ion-Sensing Ability to Polymer Membranes of Selective Electrodes. Electroanalysis 2006, 18, 1416–1421. [Google Scholar] [CrossRef]
- Peng, B.; Zhu, J.; Liu, X.; Qin, Y. Potentiometric response of ion-selective membranes with ionic liquids as ion-exchanger and plasticizer. Sens. Actuators B Chem. 2008, 133, 308–314. [Google Scholar] [CrossRef]
- Shiddiky, M.J.; Torriero, A.A. Application of ionic liquids in electrochemical sensing systems. Biosens. Bioelectron. 2011, 26, 1775–1787. [Google Scholar] [CrossRef] [PubMed]
- Heng, L.Y.; Hall, E.A. Producing “Self-Plasticizing” Ion-Selective Membranes. Anal. Chem. 2000, 72, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Heng, L.Y.; Hall, E.A. Taking the Plasticizer out of Methacrylic-Acrylic Membranes for K+-Selective Electrodes. Electroanalysis 2000, 12, 187–193. [Google Scholar] [CrossRef]
- Qin, Y.; Peper, S.; Radu, A.; Ceresa, A.; Bakker, E. Plasticizer-Free Polymer Containing a Covalently Immobilized Ca -Selective Ionophore for Potentiometric and Optical Sensors. Anal. Chem. 2003, 75, 3038–3045. [Google Scholar] [CrossRef] [PubMed]
- Peper, S.; Ceresa, A.; Qin, Y.; Bakker, E. Plasticizer-free microspheres for ionophore-based sensing and extraction based on a methyl methacrylate-decyl methacrylate copolymer matrix. Anal. Chim. Acta 2003, 500, 127–136. [Google Scholar] [CrossRef]
- Rich, M.; Mendecki, L.; Mensah, S.T.; Blanco-Martinez, E.; Armas, S.; Calvo-Marzal, P.; Radu, A.; Chumbimuni-Torres, K.Y. Circumventing traditional conditioning protocols in polymer membrane-based ion-selective electrodes. Anal. Chem. 2016, 88, 8404–8408. [Google Scholar] [CrossRef] [PubMed]
- BIFoR FACE—Birmingham Institute of Forest Research—University of Birmingham. Available online: https://www.birmingham.ac.uk/research/activity/bifor/face/index.aspx (accessed on 28 August 2018).
- Korfmacher, J.L.; Musselman, R.C. Evaluation of Storage and Filtration Protocols for Alpine/Subalpine Lake Water Quality Samples. Environ. Monit. Assess. 2007, 131, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.; Gandhi, J.; Zhang, C.C. Analysis of inorganic nitrogen and related anions in high salinity water using ion chromatography with tandem UV and conductivity detectors. J. Chromatogr. Sci. 2011, 49, 596–602. [Google Scholar] [CrossRef] [PubMed]
- Grizzetti, B.; Bouraoui, F.; Billen, G.; van Grinsven, H.; Cardoso, A.C.; Thieu, V.; Garnier, J.; Curtis, C.; Howarth, R.W.; Johnes, P. Nitrogen as a threat to European water quality. Available online: http://centaur.reading.ac.uk/20869/1/28387ENA_c17.pdf (accessed on 1 June 2018).
- Bakker, E. Determination of Unbiased Selectivity Coefficients of Neutral Carrier-Based Cation-Selective Electrodes. Anal. Chem. 1997, 69, 1061–1069. [Google Scholar] [CrossRef]
- Mendecki, L.; Callan, N.; Ahern, M.; Schazmann, B.; Radu, A. Influence of Ionic Liquids on the Selectivity of Ion Exchange-Based Polymer Membrane Sensing Layers. Sensors 2016, 16, 1106. [Google Scholar] [CrossRef] [PubMed]
- Kakiuchi, T.; Yoshimatsu, T.; Nishi, N. New Class of Ag/AgCl Electrodes Based on Hydrophobic Ionic Liquid Saturated with AgCl. Anal. Chem. 2007, 79, 7187–7191. [Google Scholar] [CrossRef] [PubMed]
- Dillingham, P.W.; Radu, T.; Diamond, D.; Radu, A.; McGraw, C.M. Bayesian Methods for Ion Selective Electrodes. Electroanalysis 2012, 24, 316–324. [Google Scholar] [CrossRef]
| Membrane | Ammonium I Ionophore | Additive (ETH 500) | Ion Exchanger | Plasticizer | Polymer | ||
|---|---|---|---|---|---|---|---|
| Concentration (mmol/kg) | Concentration (mmol/kg) | Type | Concentration (mmol/kg) | Type | Concentration (wt%) | ||
| 1 | TDACl | 5 | DOS | 66 | PVC | ||
| 2,3 | TDACl | 5 | IL | 66 | PVC | ||
| 4,5 | TDACl | 5 | DOS+IL | 33 + 33 | PVC | ||
| 5,6 | IL | 5 | DOS | 66 | PVC | ||
| 7 | 10 | NaTFPB | 5 | DOS | 66 | PVC | |
| 8,9 | 10 | NaTFPB | 5 | IL | 66 | PVC | |
| 10,11 | 10 | NaTFPB | 5 | DOS+IL | 33 + 33 | PVC | |
| 12 | 10 | TDACl | 5 | N/A | MMA-DMA | ||
| 13 | 10 | 10 | NaTFPB | 5 | N/A | MMA-DMA | |
| Soil Sample | pH | Moisture Content |
|---|---|---|
| (g/g) | ||
| Ash | 6.14 ± 0.04 | 0.35 ± 0.04 |
| Oak | 5.86 ± 0.02 | 0.29 ± 0.05 |
| Scottish pine | 6.03 ± 0.02 | 0.14 ± 0.03 |
| NH4+-Selective Electrode | NO3−-Selective Electrode | ||||
|---|---|---|---|---|---|
| Ion | Slope ± S.D. | ± S.D. | Ion | Slope ± S.D. | ± S.D. |
| Na+ | 45.67 ± 1.0 | −1.56 ± 0.01 | Cl− | −42.89 ± 0.9 | −2.24 ± 0.05 |
| K+ | 46.88 ± 1.5 | −0.65 ± 0.20 | SO42− | −24.82 ± 1.9 | −2.59 ± 0.80 |
| Ca2+ | 25.45 ± 2.6 | −2.77 ± 0.20 | NO2− | −42.80 ± 0.8 | −2.83 ± 0.08 |
| NH4+ | 52.96 ± 0.5 | 0 | NO3− | −49.45 ± 1.3 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choosang, J.; Numnuam, A.; Thavarungkul, P.; Kanatharana, P.; Radu, T.; Ullah, S.; Radu, A. Simultaneous Detection of Ammonium and Nitrate in Environmental Samples Using on Ion-Selective Electrode and Comparison with Portable Colorimetric Assays. Sensors 2018, 18, 3555. https://doi.org/10.3390/s18103555
Choosang J, Numnuam A, Thavarungkul P, Kanatharana P, Radu T, Ullah S, Radu A. Simultaneous Detection of Ammonium and Nitrate in Environmental Samples Using on Ion-Selective Electrode and Comparison with Portable Colorimetric Assays. Sensors. 2018; 18(10):3555. https://doi.org/10.3390/s18103555
Chicago/Turabian StyleChoosang, Jittima, Apon Numnuam, Panote Thavarungkul, Proespichaya Kanatharana, Tanja Radu, Sami Ullah, and Aleksandar Radu. 2018. "Simultaneous Detection of Ammonium and Nitrate in Environmental Samples Using on Ion-Selective Electrode and Comparison with Portable Colorimetric Assays" Sensors 18, no. 10: 3555. https://doi.org/10.3390/s18103555
APA StyleChoosang, J., Numnuam, A., Thavarungkul, P., Kanatharana, P., Radu, T., Ullah, S., & Radu, A. (2018). Simultaneous Detection of Ammonium and Nitrate in Environmental Samples Using on Ion-Selective Electrode and Comparison with Portable Colorimetric Assays. Sensors, 18(10), 3555. https://doi.org/10.3390/s18103555







