Implementing Morpholino-Based Nucleic Acid Sensing on a Portable Surface Plasmon Resonance Instrument for Future Application in Environmental Monitoring
Abstract
1. Introduction
2. Materials and Methods
2.1. General Assay Procedures and Materials
2.1.1. SPR Instrument
2.1.2. Probes
2.1.3. Analytes
2.1.4. Buffers
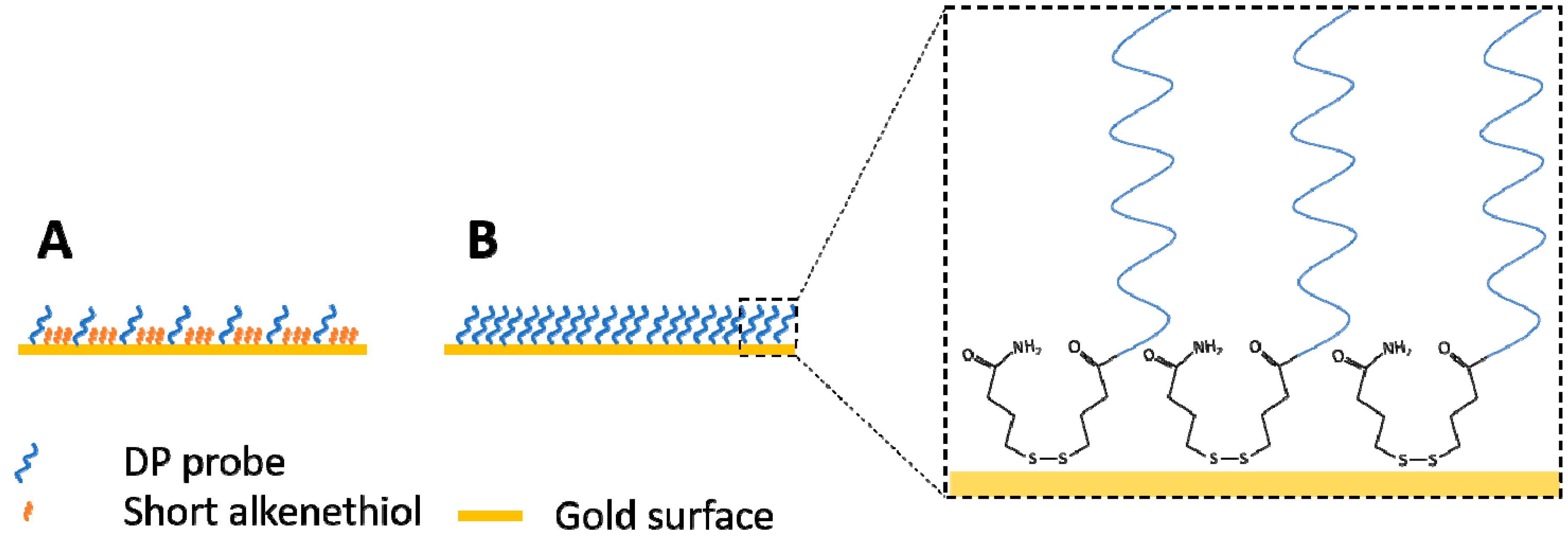
2.1.5. Preparation of Sensor Surfaces
2.1.6. Experimental Procedures
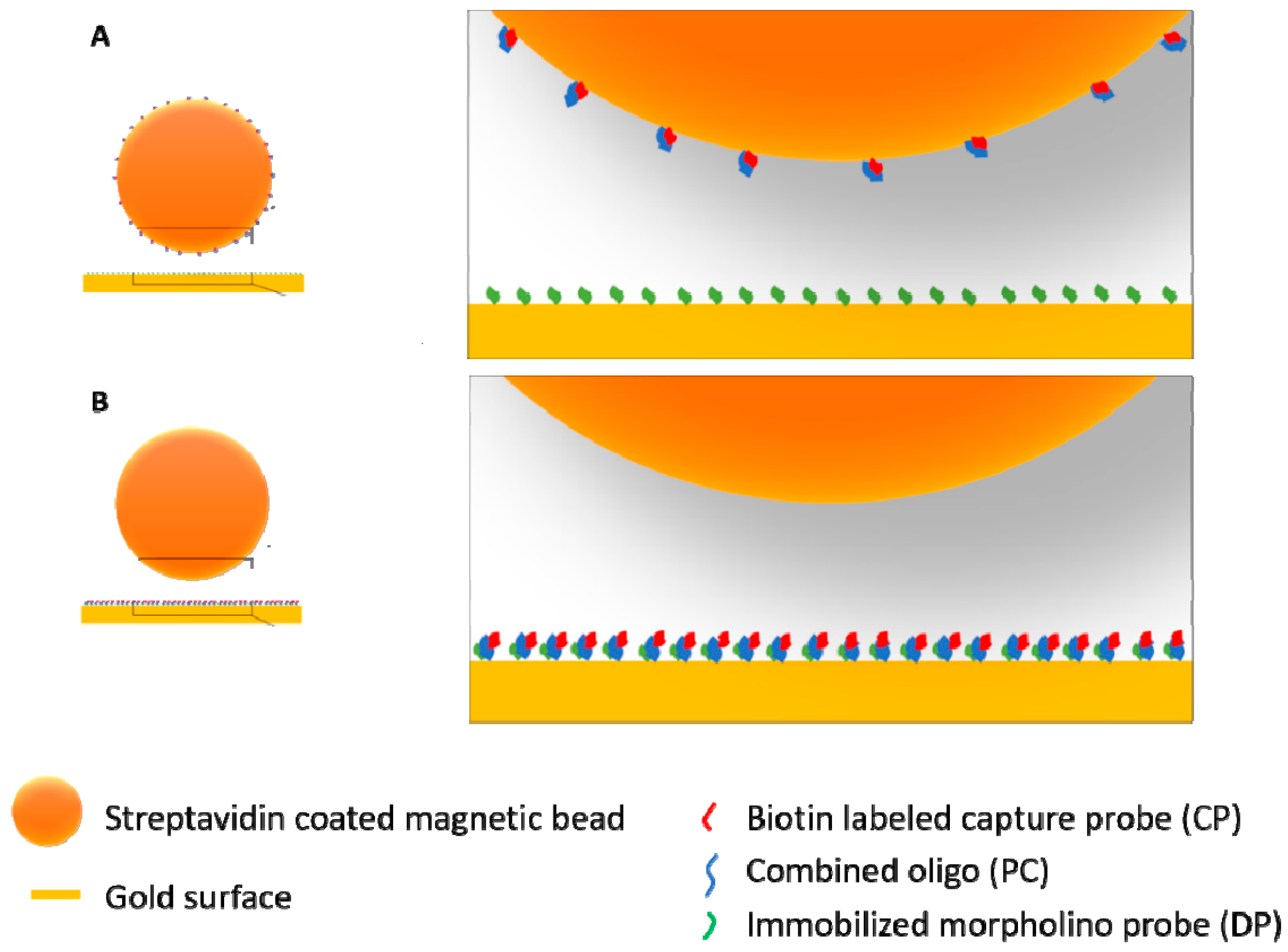
2.1.7. Preparation of Oligos and Magnetic Beads
2.2. Testing Specificity
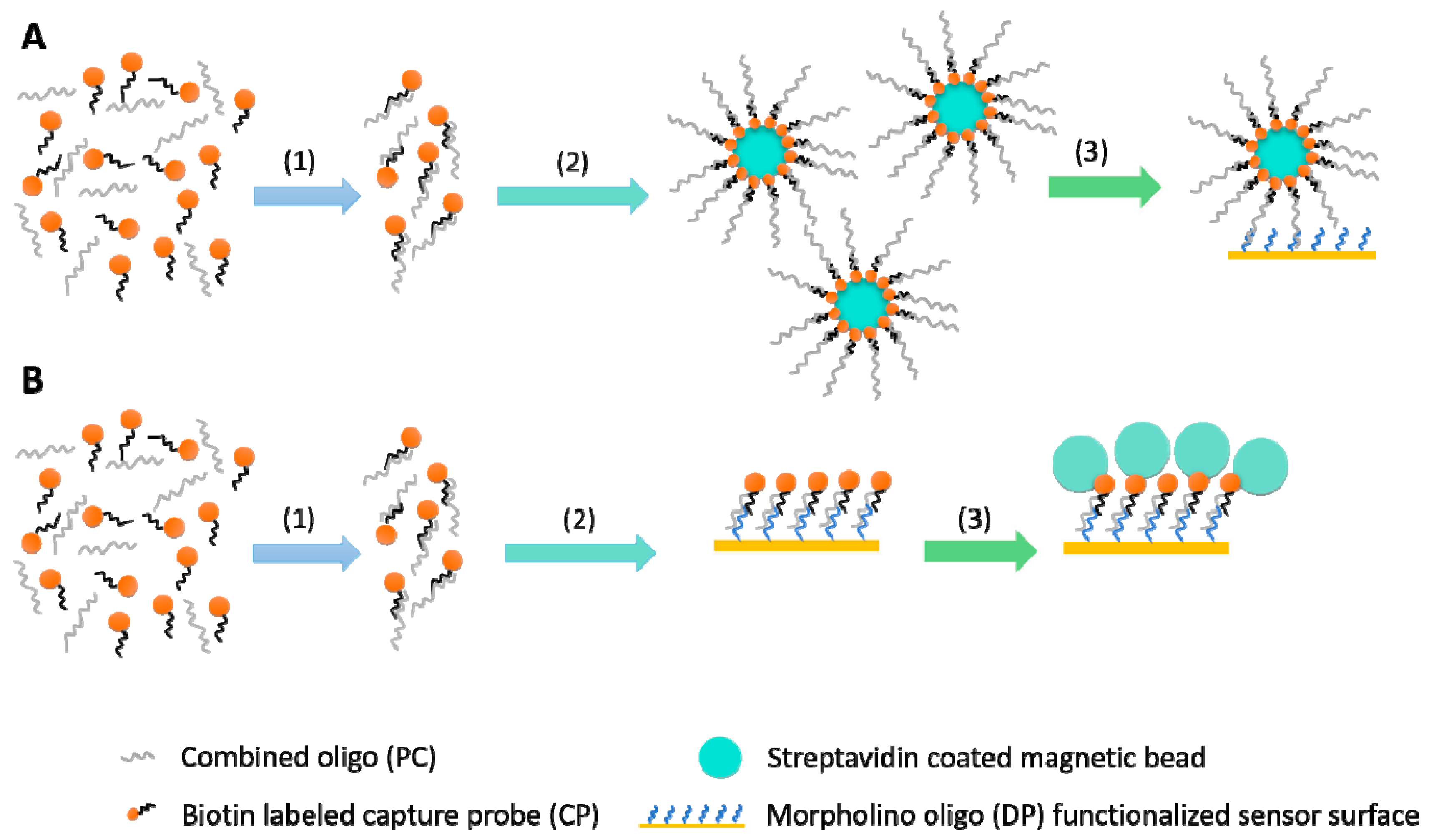
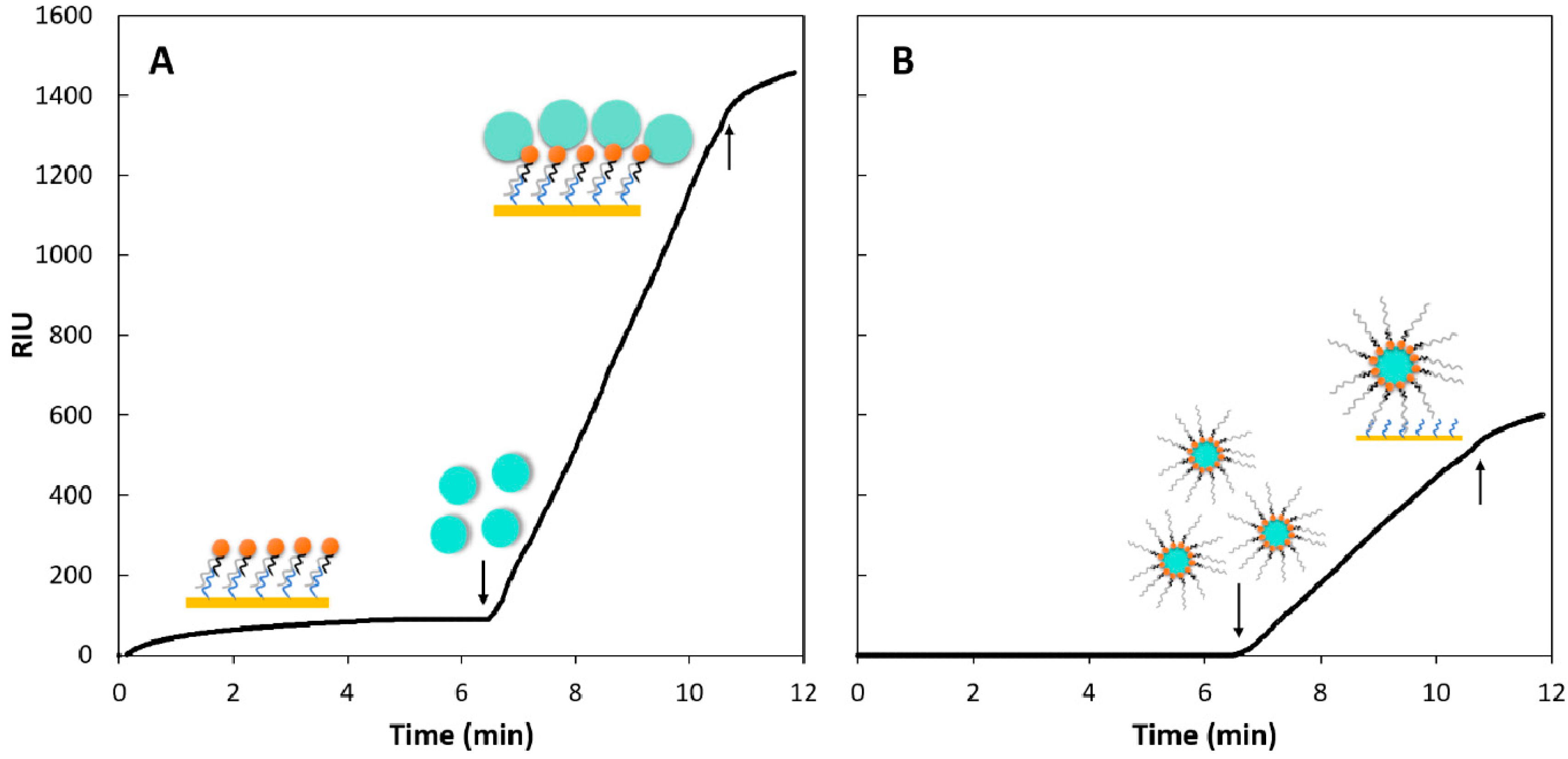
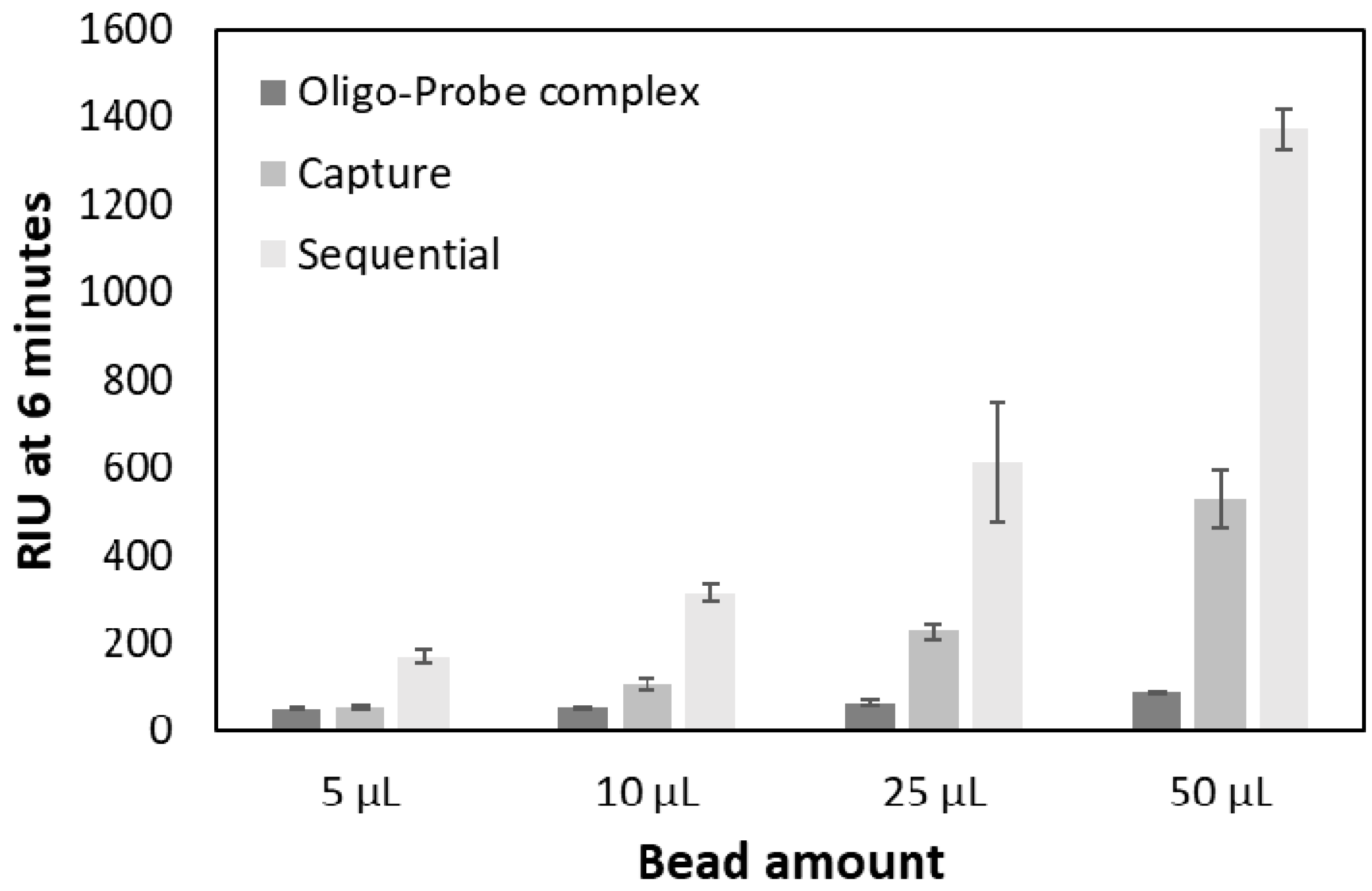
2.3. Testing Two Signal Amplification Strategies
2.3.1. Sequential Assay
2.3.2. Capture Assay
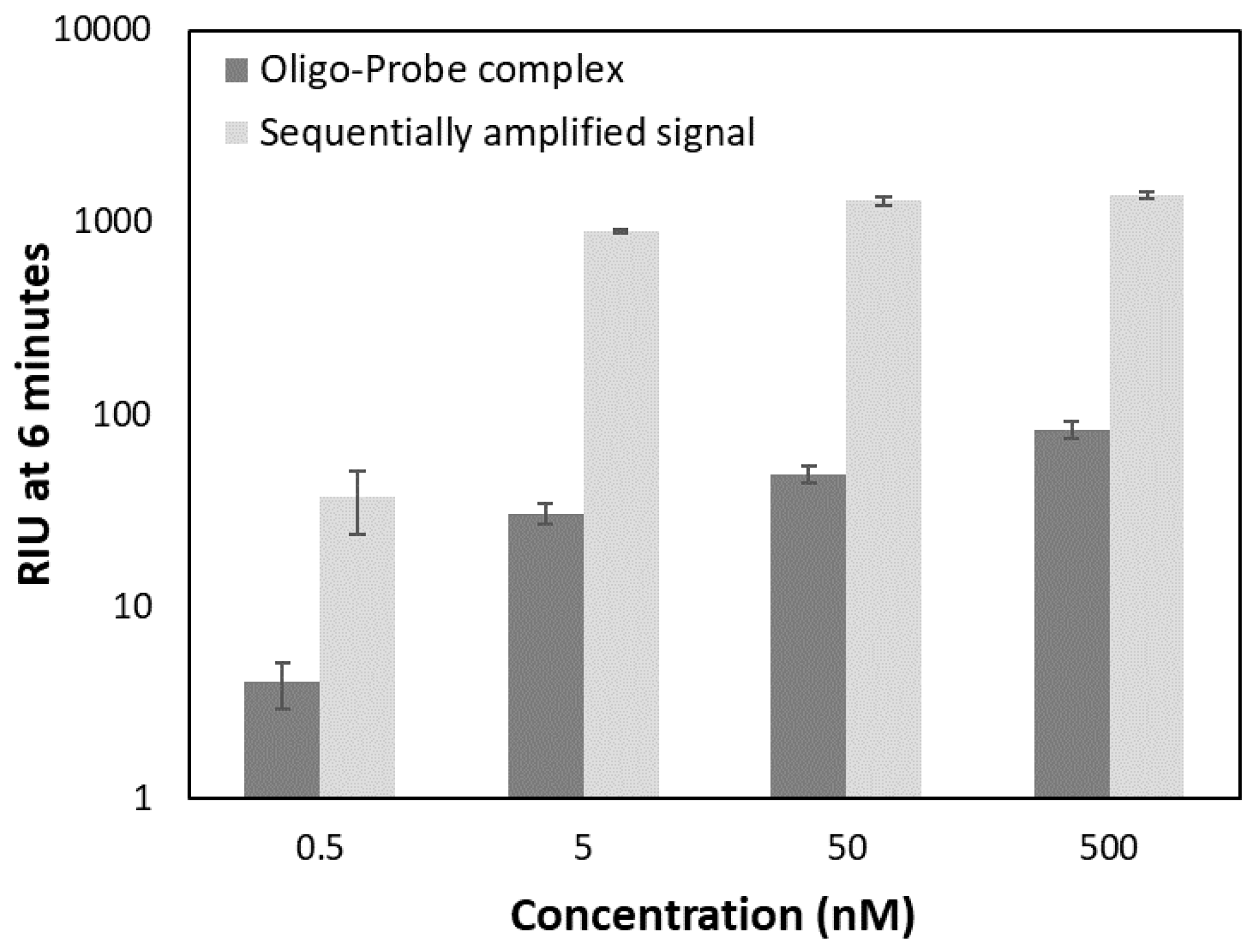
2.4. Testing the Detection Limits
3. Results and Discussion
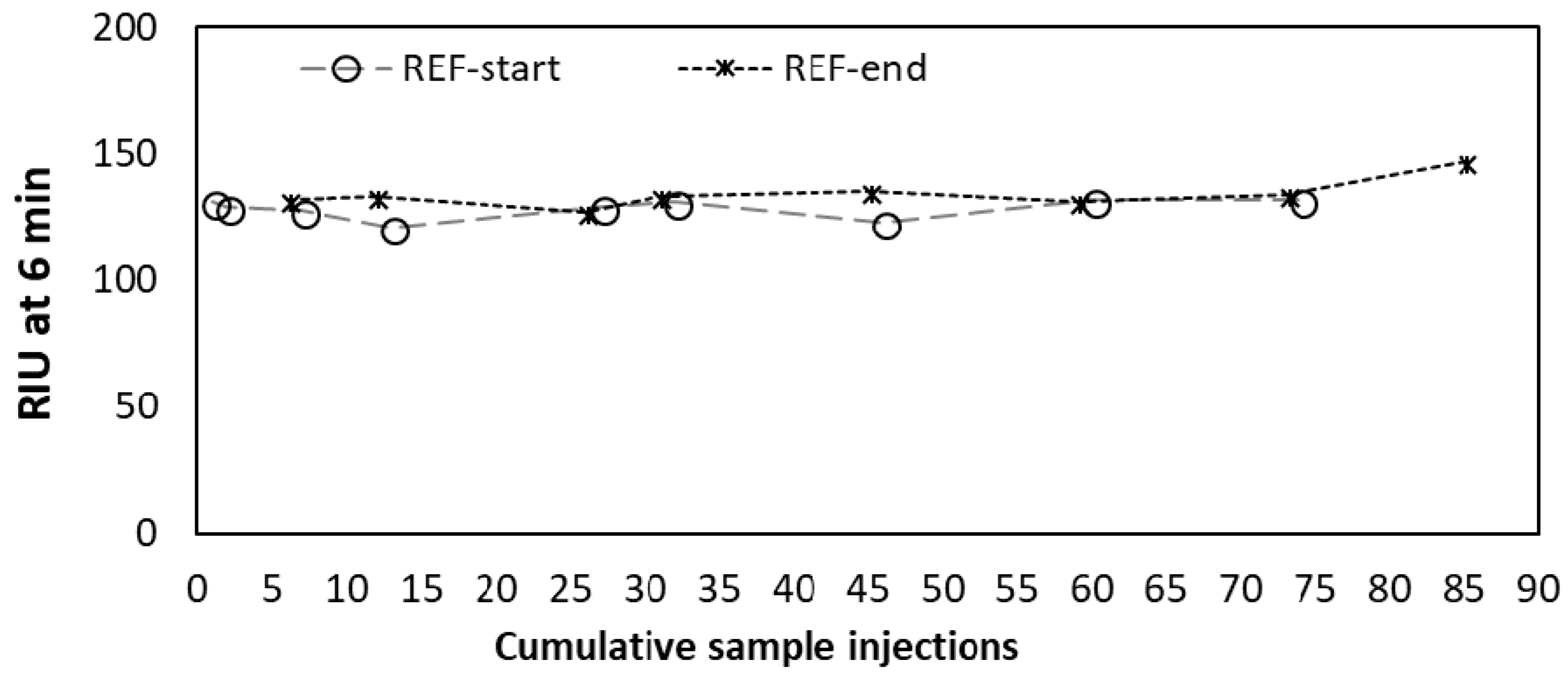
3.1. General Assay Procedures
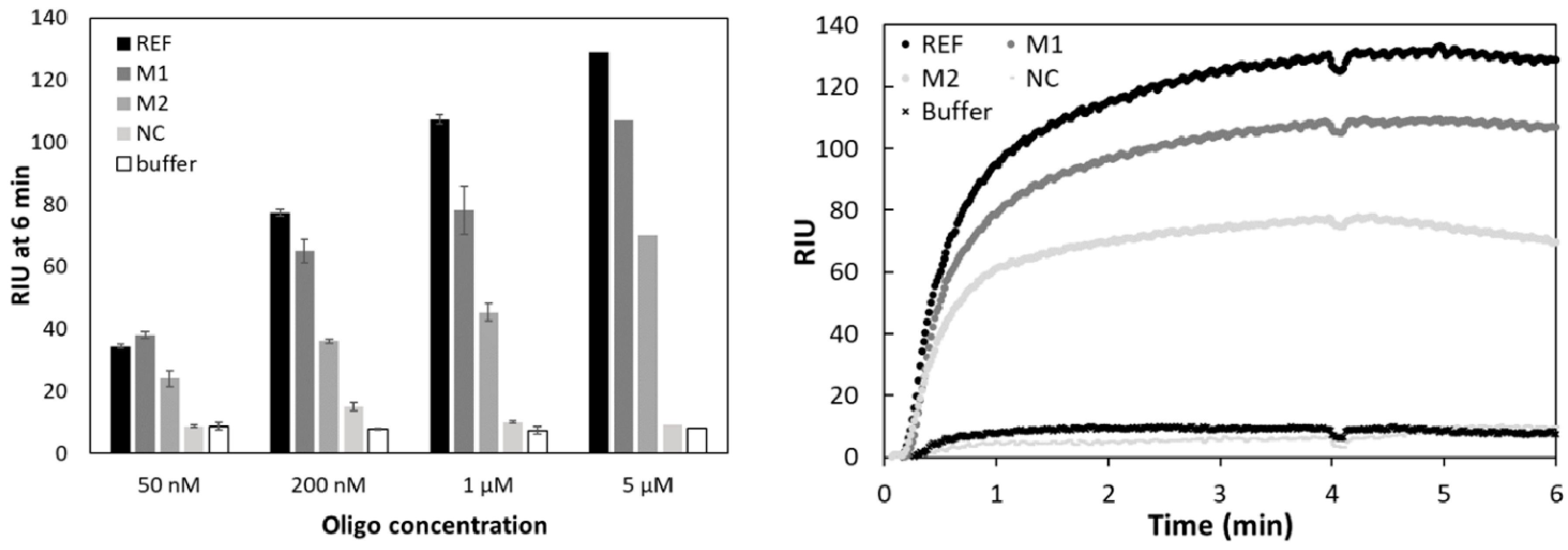
3.2. Testing Specificity
3.3. Testing Two Signal Amplification Strategies
3.4. Testing the Detection Limits
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Medlin, L.K.; Orozco, J. Molecular techniques for the detection of organisms in aquatic environments, with emphasis on harmful algal bloom species. Sensors 2017, 17, 1184. [Google Scholar] [CrossRef] [PubMed]
- McQuillan, J.S.; Robidart, J.C. Molecular-biological sensing in aquatic environments: Recent developments and emerging capabilities. Curr. Opin. Biotechnol. 2017, 45, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Ottesen, E.A. Probing the living ocean with ecogenomic sensors. Curr. Opin. Microbiol. 2016, 31, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Yamahara, K.M.; Demir-Hilton, E.; Preston, C.M.; Marin, R., 3rd; Pargett, D.; Roman, B.; Jensen, S.; Birch, J.M.; Boehm, A.B.; Scholin, C.A. Simultaneous monitoring of faecal indicators and harmful algae using an in-situ autonomous sensor. Lett. Appl. Microbiol. 2015, 61, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Palchetti, I.; Mascini, M. Nucleic acid biosensors for environmental pollution monitoring. Analyst 2008, 133, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Justino, C.I.L.; Duarte, A.C.; Rocha-Santos, T.A.P. Recent progress in biosensors for environmental monitoring: A review. Sensors 2017, 17, 2918. [Google Scholar] [CrossRef] [PubMed]
- Diercks-Horn, S.; Metfies, K.; Jäckel, S.; Medlin, L.K. The algadec device: A semi-automated rrna biosensor for the detection of toxic algae. Harmful Algae 2011, 10, 395–401. [Google Scholar] [CrossRef]
- Soelberg, S.D.; Furlong, C.E. Biosensors and bioassays for ecological risk monitoring and assessment. In Environmental Toxicology; Laws, E., Ed.; Springer: New York, NY, USA, 2013; pp. 121–142. [Google Scholar]
- Du, Y.; Dong, S. Nucleic acid biosensors: Recent advances and perspectives. Anal. Chem. 2017, 89, 189–215. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.H.; Park, J.; Kang, S.; Kim, M. Surface plasmon resonance: A versatile technique for biosensor applications. Sensors 2015, 15, 10481–10510. [Google Scholar] [CrossRef] [PubMed]
- Homola, J.; Yee, S.S.; Gauglitz, G. Surface plasmon resonance sensors: Review. Sens. Actuators B Chem. 1999, 54, 3–15. [Google Scholar] [CrossRef]
- Foudeh, A.M.; Daoud, J.T.; Faucher, S.P.; Veres, T.; Tabrizian, M. Sub-femtomole detection of 16s rrna from legionella pneumophila using surface plasmon resonance imaging. Biosens. Bioelectron. 2014, 52, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wark, A.W.; Lee, H.J.; Corn, R.M. Single-nucleotide polymorphism genotyping by nanoparticle-enhanced surface plasmon resonance imaging measurements of surface ligation reactions. Anal. Chem. 2006, 78, 3158–3164. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Yan, Y.; Li, S.; Zhang, Y.; Cheng, W.; Cheng, Q.; Ding, S. Surface plasmon resonance biosensor for highly sensitive detection of microrna based on DNA super-sandwich assemblies and streptavidin signal amplification. Anal. Chim. Acta 2015, 874, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Scholin, C.; Doucette, G.; Jensen, S.; Roman, B.; Pargett, D.; Marin, R.; Preston, C.; Jones, W.; Feldman, J.; Everlove, C.; et al. Remote detection of marine microbes, small invertebrates, harmful algae, and biotoxins using the environmental sample processor (esp). Oceanography 2009, 22, 158–167. [Google Scholar] [CrossRef]
- Chinowsky, T.M.; Soelberg, S.D.; Baker, P.; Swanson, N.R.; Kauffman, P.; Mactutis, A.; Grow, M.S.; Atmar, R.; Yee, S.S.; Furlong, C.E. Portable 24-analyte surface plasmon resonance instruments for rapid, versatile biodetection. Biosens. Bioelectron. 2007, 22, 2268–2275. [Google Scholar] [CrossRef] [PubMed]
- Kroger, S.; Law, R.J. Biosensors for marine applications. We all need the sea, but does the sea need biosensors? Biosens. Bioelectron. 2005, 20, 1903–1913. [Google Scholar] [PubMed]
- Kröger, S.; Parker, E.R.; Metcalfe, J.D.; Greenwood, N.; Forster, R.M.; Sivyer, D.B.; Pearce, D.J. Sensors for observing ecosystem status. Ocean Sci. 2009, 5, 523–535. [Google Scholar] [CrossRef]
- Bagi, A.; Knapik, K.; Nilsen, M.M.; Krolicka, A.; Boccadoro, C.; Baussant, T. Environmental surveillance of marine systems using genosensors: Application to offshore activities. In Oilfield Microbiology; Whitby, C., Skovhus, T.L., Eds.; CRC Press: Boca Raton, FL, USA, In Prep.
- Kube, M.; Chernikova, T.N.; Al-Ramahi, Y.; Beloqui, A.; Lopez-Cortez, N.; Guazzaroni, M.E.; Heipieper, H.J.; Klages, S.; Kotsyurbenko, O.R.; Langer, I.; et al. Genome sequence and functional genomic analysis of the oil-degrading bacterium Oleispira antarctica. Nat. Commun. 2013, 4, 2156. [Google Scholar] [CrossRef] [PubMed]
- Gentile, G.; Bonsignore, M.; Santisi, S.; Catalfamo, M.; Giuliano, L.; Genovese, L.; Yakimov, M.M.; Denaro, R.; Genovese, M.; Cappello, S. Biodegradation potentiality of psychrophilic bacterial strain Oleispira antarctica rb-8(t). Mar. Pollut. Bull. 2016, 105, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Królicka, A.; Boccadoro, C.; Mæland, M.; Preston, C.; Birch, J.; Scholin, C.; Baussant, T. Detection of oil leaks by quantifying hydrocarbonoclastic bacteria in cold marine environments using the environmental sample processor. In Proceedings of the 37th AMOP Technical Seminar on Environmental Contamination and Response, Canmore, AB, Canada, 3–5 June 2014. [Google Scholar]
- Sipova, H.; Homola, J. Surface plasmon resonance sensing of nucleic acids: A review. Anal. Chim. Acta 2013, 773, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Fu, G.; Kong, J.; Zhou, S.; Scafa, N.; Zhang, X. Advancement of nucleic acid biosensors based on morpholino. Am. J. Biomed. Sci. 2015, 7, 40–51. [Google Scholar] [CrossRef]
- Moulton, H.M.; Moulton, J.D. Morpholino Oligomers: Methods and Protocols, 1st ed.; Humana Press: New York, NY, USA, 2017; p. 284. [Google Scholar]
- Levicky, R.; Koniges, U.; Tercero, N. Diagnostic applications of morpholinos and label-free electrochemical detection of nucleic acids. Methods Mol. Biol. 2017, 1565, 181–190. [Google Scholar] [PubMed]
- Soelberg, S.D.; Chinowsky, T.; Geiss, G.; Spinelli, C.B.; Stevens, R.; Near, S.; Kauffman, P.; Yee, S.; Furlong, C.E. A portable surface plasmon resonance sensor system for real-time monitoring of small to large analytes. J. Ind. Microbiol. Biotechnol. 2005, 32, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The silva ribosomal rna gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.P.; Liao, J.C.; Zhang, Y.H.; Gau, V.; Mastali, M.; Babbitt, J.T.; Grundfest, W.S.; Churchill, B.M.; McCabe, E.R.; Haake, D.A. Rapid, species-specific detection of uropathogen 16s rdna and rrna at ambient temperature by dot-blot hybridization and an electrochemical sensor array. Mol. Genet. Metab. 2005, 84, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Liao, T.; Li, X.; Tong, Q.; Zou, K.; Zhang, H.; Tang, L.; Sun, Z.; Zhang, G.J. Ultrasensitive detection of micrornas with morpholino-functionalized nanochannel biosensor. Anal. Chem. 2017, 89, 5511–5518. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Lu, M.; Dinel, M.P.; Blain, P.; Peng, W.; Gu, H.; Masson, J.F. Hybridization conditions of oligonucleotide-capped gold nanoparticles for spr sensing of microrna. Biosens. Bioelectron. 2018, 109, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Tercero, N.; Wang, K.; Gong, P.; Levicky, R. Morpholino monolayers: Preparation and label-free DNA analysis by surface hybridization. J. Am. Chem. Soc. 2009, 131, 4953–4961. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zeng, G.M.; Tang, L.; Zhang, Y.; Li, Y.P.; Liu, Y.Y.; Li, Z.; Wu, M.S.; Luo, J. Electrochemical detection of pseudomonas aeruginosa 16s rrna using a biosensor based on immobilized stem-loop structured probe. Enzym. Microb. Technol. 2011, 49, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Heng, J.B.; Ho, C.; Kim, T.; Timp, R.; Aksimentiev, A.; Grinkova, Y.V.; Sligar, S.; Schulten, K.; Timp, G. Sizing DNA using a nanometer-diameter pore. Biophys. J. 2004, 87, 2905–2911. [Google Scholar] [CrossRef] [PubMed]
- Tercero, N.; Wang, K.; Levicky, R. Capacitive monitoring of morpholino-DNA surface hybridization: Experimental and theoretical analysis. Langmuir 2010, 26, 14351–14358. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Yao, D.; Knoll, W. Oligonucleotide hybridization studied by a surface plasmon diffraction sensor (spds). Nucleic Acids Res. 2004, 32, e75. [Google Scholar] [CrossRef] [PubMed]
- Parab, H.J.; Jung, C.; Lee, J.H.; Park, H.G. A gold nanorod-based optical DNA biosensor for the diagnosis of pathogens. Biosens. Bioelectron. 2010, 26, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Hao, K.; He, Y.; Lu, H.; Pu, S.; Zhang, Y.; Dong, H.; Zhang, X. High-sensitive surface plasmon resonance microrna biosensor based on streptavidin functionalized gold nanorods-assisted signal amplification. Anal. Chim. Acta 2017, 954, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Da-Silva, E.; Barthelmebs, L.; Baudart, J. Development of a pcr-free DNA-based assay for the specific detection of vibrio species in environmental samples by targeting the 16s rrna. Environ. Sci. Pollut. Res. Int. 2017, 24, 5690–5700. [Google Scholar] [CrossRef] [PubMed]
- Wages, J.M., Jr.; Wages, G.M.; Matthews, P.; Weller, D.; Summerton, J. Affinity purification of rna: Sequence-specific capture by nonionic morpholino probes. Biotechniques 1997, 23, 1116–1121. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; McQuistan, T.J.; Stanek, J.W.; Summerton, J.E.; Mata, J.E.; Squier, T.C. Detection of unique ebola virus oligonucleotides using fluorescently-labeled phosphorodiamidate morpholino oligonucleotide probe pairs. Anal. Biochem. 2018, 557, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.C.; Soelberg, S.D.; Near, S.; Furlong, C.E. Detection of cortisol in saliva with a flow-filtered, portable surface plasmon resonance biosensor system. Anal. Chem. 2008, 80, 6747–6751. [Google Scholar] [CrossRef] [PubMed]
- Soelberg, S.D.; Stevens, R.C.; Limaye, A.P.; Furlong, C.E. Surface plasmon resonance detection using antibody-linked magnetic nanoparticles for analyte capture, purification, concentration, and signal amplification. Anal. Chem. 2009, 81, 2357–2363. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.C.; Soelberg, S.D.; Eberhart, B.-T.L.; Spencer, S.; Wekell, J.C.; Chinowsky, T.M.; Trainer, V.L.; Furlong, C.E. Detection of the toxin domoic acid from clam extracts using a portable surface plasmon resonance biosensor. Harmful Algae 2007, 6, 166–174. [Google Scholar] [CrossRef]
- Wang, Y.; Dostalek, J.; Knoll, W. Magnetic nanoparticle-enhanced biosensor based on grating-coupled surface plasmon resonance. Anal. Chem. 2011, 83, 6202–6207. [Google Scholar] [CrossRef] [PubMed]
- Melaine, F.; Saad, M.; Faucher, S.; Tabrizian, M. Selective and high dynamic range assay format for multiplex detection of pathogenic pseudomonas aeruginosa, salmonella typhimurium, and legionella pneumophila rnas using surface plasmon resonance imaging. Anal. Chem. 2017, 89, 7802–7807. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.S.; Bukar, N.; Toulouse, J.L.; Pelechacz, D.; Robitaille, R.; Pelletier, J.N.; Masson, J.F. Miniature multi-channel spr instrument for methotrexate monitoring in clinical samples. Biosens. Bioelectron. 2015, 64, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.T.; Pourhassan-Moghaddam, M.; Wu, L.; Bai, P.; Thierry, B. Ultrasensitive detection of cancer prognostic mirna biomarkers based on surface plasmon enhanced light scattering. ACS Sens. 2017, 2, 635–640. [Google Scholar] [CrossRef] [PubMed]


| Name | Sequence (5′-3′) | MW (g/mol) |
|---|---|---|
| DP | CTAGCTAATCTCACTCAGGCTCAT-S-S-amide | 8243 |
| CP | Bio-TCTACGCATTTCACCGCTACA | 6706 |
| REF | ATGAGCCTGAGTGAGATTAGCTAG | 7457 |
| PC | ATGAGCCTGAGTGAGATTAGCTAG/iSp18/TGTAGCGGTGAAATGCGTAGA | 14,368 |
| M1 | ATGAACCTGAGTGAGATTAGCTAG | 7441 |
| M2 | ATGAACCTGAGTTAGATTAGCTAG | 7416 |
| NC | TGTAGCGGTGAAATGCGTAGA | 6550 |
| Step | Duration (s) | Pump Speed (µL/min) |
|---|---|---|
| Initialization | 120 | 20 |
| Baseline | 0 | 0 |
| Injection | 10 | 0 |
| Flow | 240 | 20 |
| Flush | 7 | n.a. |
| Flow | 120 | 20 |
| Regeneration | 120 | 100 |
| Flow | 120 | 100 |
| Amplification Factors (±SD) | ||||
|---|---|---|---|---|
| 5 µL | 10 µL | 25 µL | 50 µL | |
| Capture | 1.1 ± 0.03 | 2.0 ± 0.22 | 3.5 ± 0.31 | 6.0 ± 0.6 |
| Sequential | 3.2 ± 0.32 | 5.8 ± 0.38 | 9.7 ± 3.08 | 15.5 ± 0.70 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bagi, A.; Soelberg, S.D.; Furlong, C.E.; Baussant, T. Implementing Morpholino-Based Nucleic Acid Sensing on a Portable Surface Plasmon Resonance Instrument for Future Application in Environmental Monitoring. Sensors 2018, 18, 3259. https://doi.org/10.3390/s18103259
Bagi A, Soelberg SD, Furlong CE, Baussant T. Implementing Morpholino-Based Nucleic Acid Sensing on a Portable Surface Plasmon Resonance Instrument for Future Application in Environmental Monitoring. Sensors. 2018; 18(10):3259. https://doi.org/10.3390/s18103259
Chicago/Turabian StyleBagi, Andrea, Scott D. Soelberg, Clement E. Furlong, and Thierry Baussant. 2018. "Implementing Morpholino-Based Nucleic Acid Sensing on a Portable Surface Plasmon Resonance Instrument for Future Application in Environmental Monitoring" Sensors 18, no. 10: 3259. https://doi.org/10.3390/s18103259
APA StyleBagi, A., Soelberg, S. D., Furlong, C. E., & Baussant, T. (2018). Implementing Morpholino-Based Nucleic Acid Sensing on a Portable Surface Plasmon Resonance Instrument for Future Application in Environmental Monitoring. Sensors, 18(10), 3259. https://doi.org/10.3390/s18103259





