Detection of Craving for Gaming in Adolescents with Internet Gaming Disorder Using Multimodal Biosignals
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
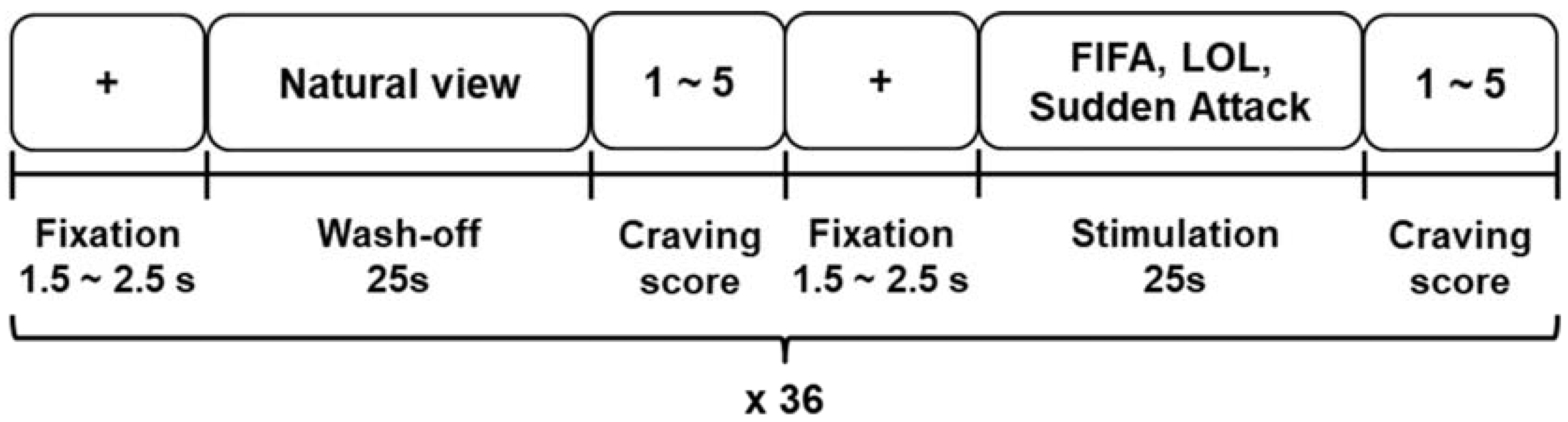
2.2. Experimental Paradigm and Bio-Signal Acquisition
2.3. Processing Multiple Biosignals
2.3.1. Processing of PPG and GSR Signals
2.3.2. Processing of EOG Signals
2.4. Statistical Tests
2.5. Classification of Craving States
3. Results
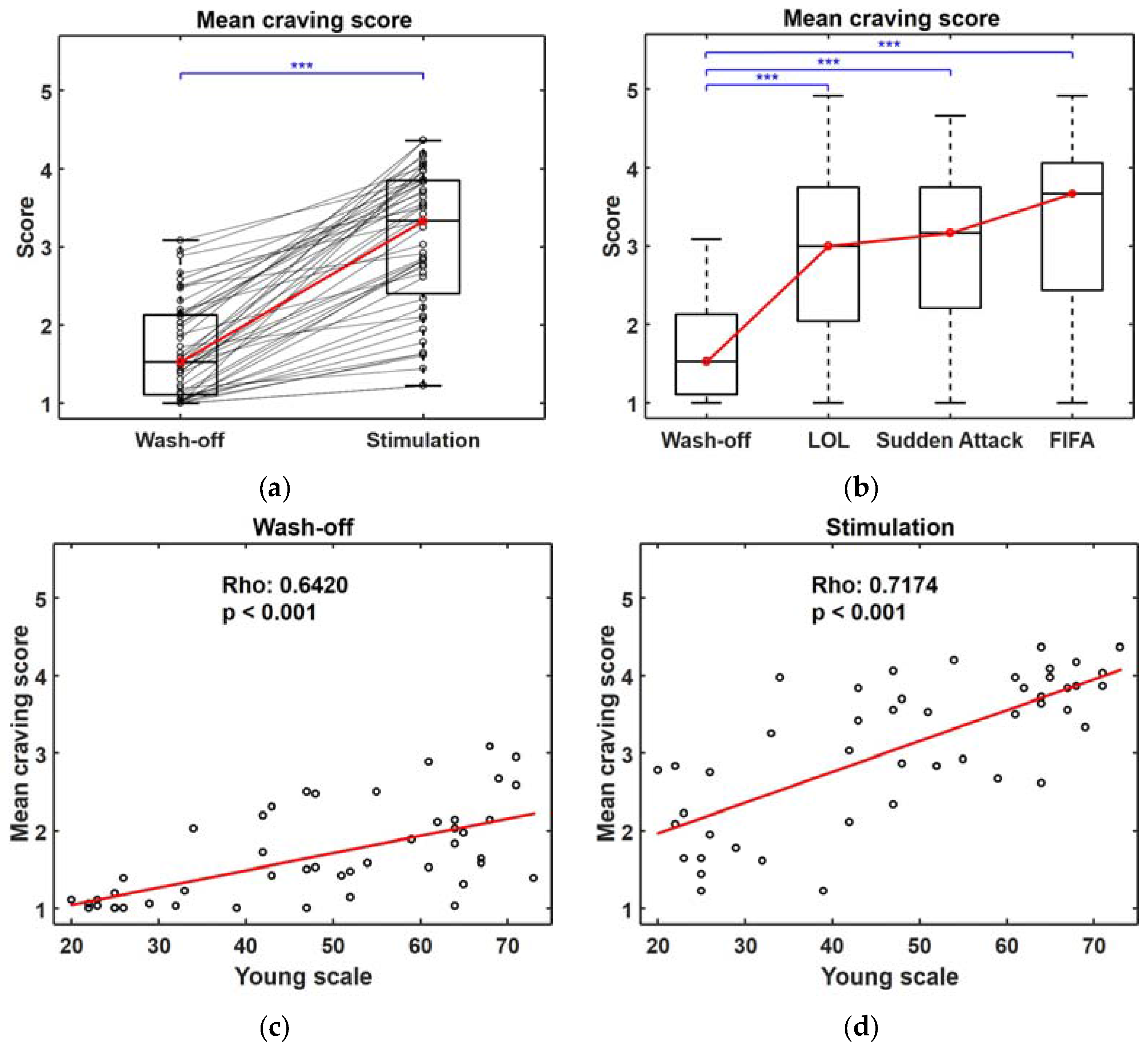
3.1. Self-Reported Craving Score
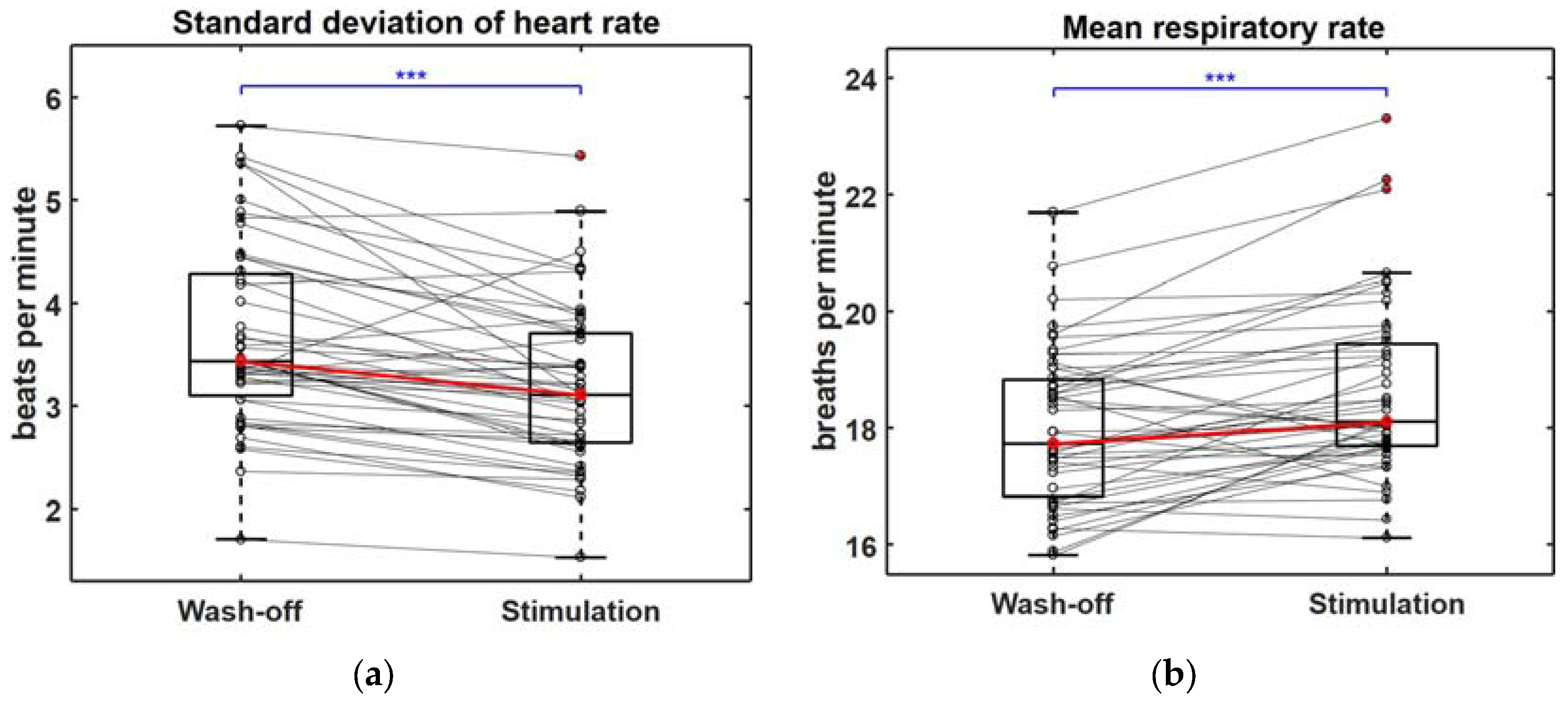
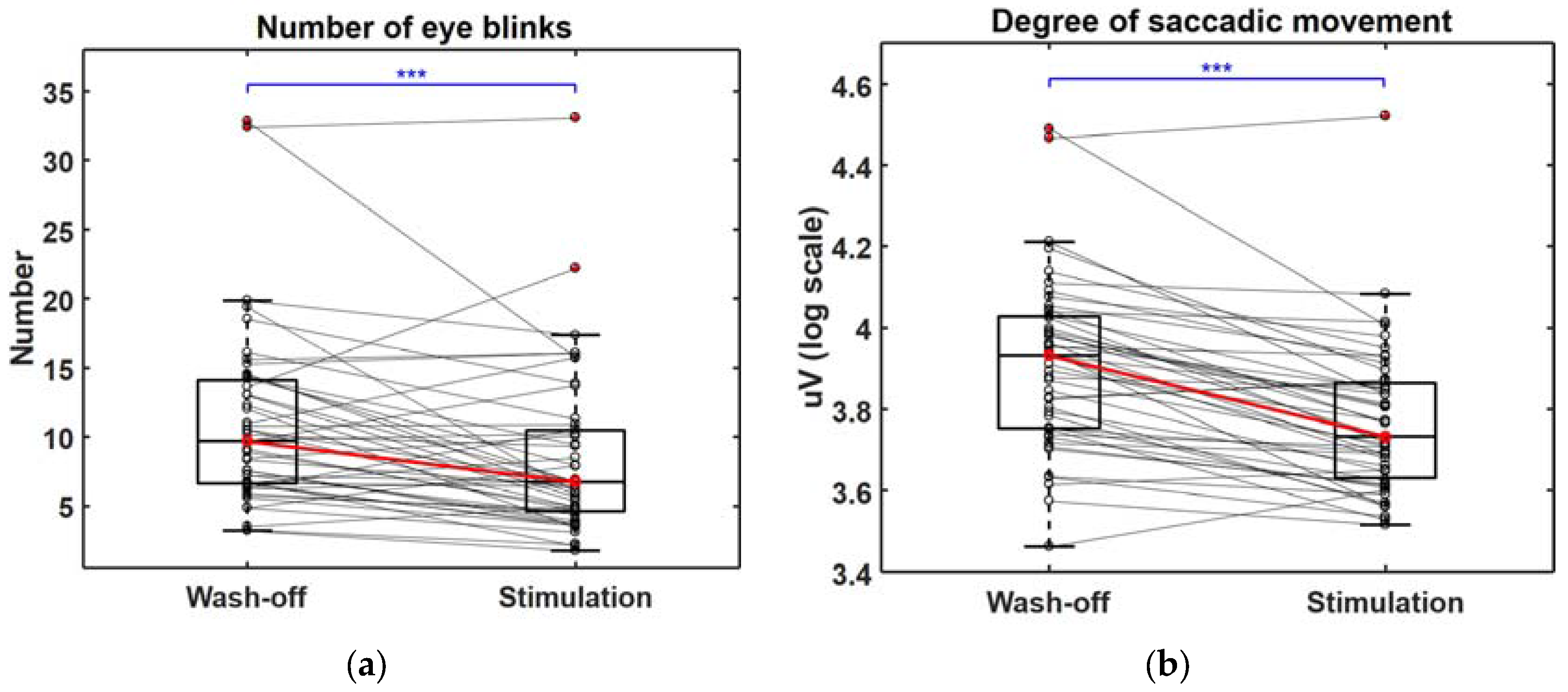
3.2. Changes in Multiple Biosignals When Craving for Gaming Was Aroused
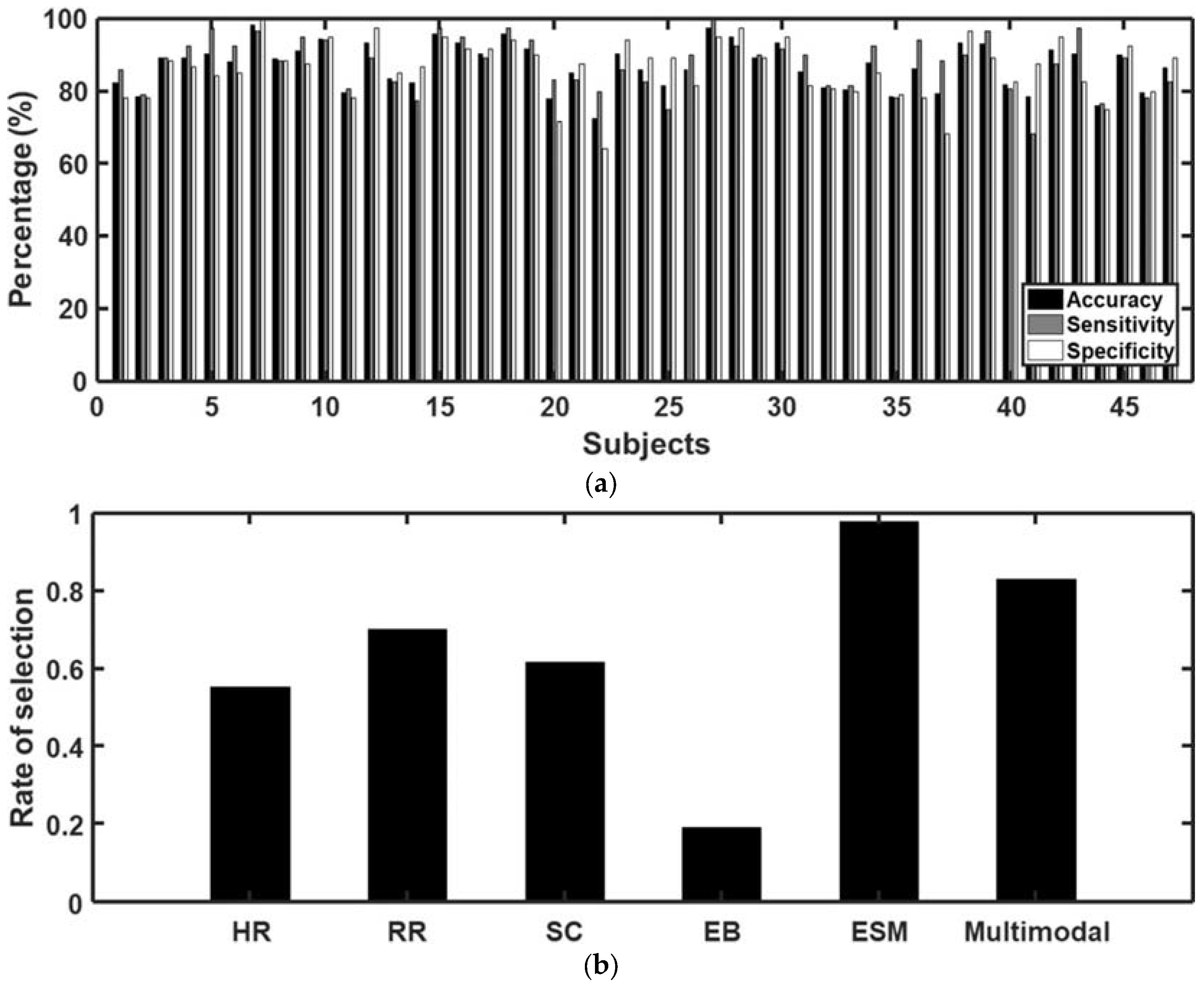
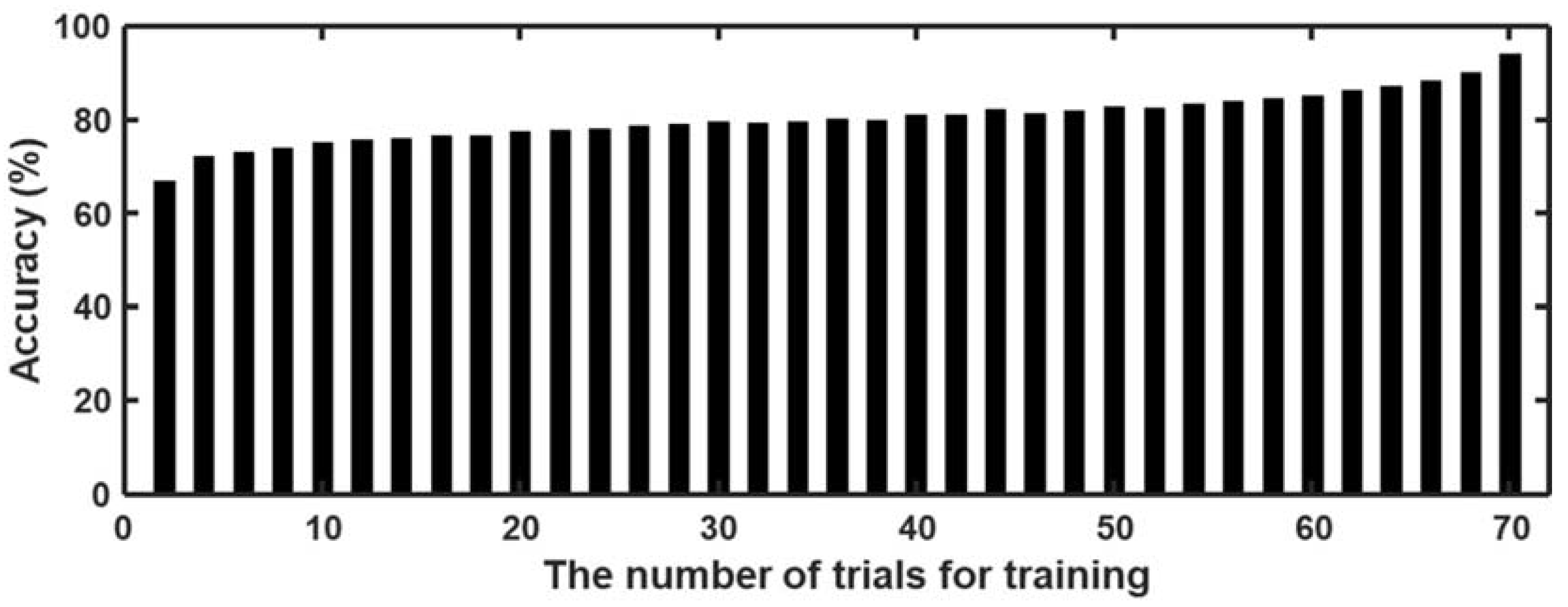
3.3. Classification of Craving State Using Machine Learning
4. Discussion and Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A. Calculation of Features Listed in Table 1
References
- Petry, N.M.; O’brien, C.P. Internet gaming disorder and the dsm-5. Addiction 2013, 108, 1186–1187. [Google Scholar] [CrossRef] [PubMed]
- Potenza, M.N. Should addictive disorders include non-substance-related conditions? Addiction 2006, 101, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Franken, I.H. Drug craving and addiction: Integrating psychological and neuropsychopharmacological approaches. Prog. Neuropsychopharmacol. Biol. Psychiatry 2003, 27, 563–579. [Google Scholar] [CrossRef]
- McRae-Clark, A.L.; Carter, R.E.; Price, K.L.; Baker, N.L.; Thomas, S.; Saladin, M.E.; Giarla, K.; Nicholas, K.; Brady, K.T. Stress-and cue-elicited craving and reactivity in marijuana-dependent individuals. Psychopharmacology (Berl.) 2011, 218, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Franken, I.H.; Kroon, L.Y.; Wiers, R.W.; Jansen, A. Selective cognitive processing of drug cues in heroin dependence. J. Psychopharmacol. 2000, 14, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.J.; Sayette, M.A.; Fiez, J.A. Prefrontal responses to drug cues: A neurocognitive analysis. Nat. Neurosci. 2004, 7, 211–214. [Google Scholar] [CrossRef] [PubMed]
- Ko, C.-H.; Liu, G.-C.; Hsiao, S.; Yen, J.-Y.; Yang, M.-J.; Lin, W.-C.; Yen, C.-F.; Chen, C.-S. Brain activities associated with gaming urge of online gaming addiction. J. Psychiatr. Res. 2009, 43, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Burton, S.M.; Tiffany, S.T. The effect of alcohol consumption on craving to smoke. Addiction 1997, 92, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.; Bovbjerg, D.H.; Erblich, J. Smoking-related videos for use in cue-induced craving paradigms. Addict. Behav. 2007, 32, 3034–3044. [Google Scholar] [CrossRef] [PubMed]
- LaRowe, S.D.; Saladin, M.E.; Carpenter, M.J.; Upadhyaya, H.P. Reactivity to nicotine cues over repeated cue reactivity sessions. Addict. Behav. 2007, 32, 2888–2899. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.; Fox, H.C.; Hong, K.A.; Bergquist, K.; Bhagwagar, Z.; Siedlarz, K.M. Enhanced negative emotion and alcohol craving, and altered physiological responses following stress and cue exposure in alcohol dependent individuals. Neuropsychopharmacology 2009, 34, 1198–1208. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.W.; Wang, J.W.; Huang, A.C.W. Differentiation of internet addiction risk level based on autonomic nervous responses: The internet-addiction hypothesis of autonomic activity. Cyberpsychol. Behav. Soc. Netw. 2010, 13, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, D.-L.; Hsiao, T.-C. Respiratory sinus arrhythmia reactivity of internet addiction abusers in negative and positive emotional states using film clips stimulation. Biomed. Eng. Online 2016, 15, 69. [Google Scholar] [CrossRef] [PubMed]
- Carroll, K.M.; Ball, S.A.; Martino, S.; Nich, C.; Babuscio, T.A.; Nuro, K.F.; Gordon, M.A.; Portnoy, G.A.; Rounsaville, B.J. Computer-assisted delivery of cognitive-behavioral therapy for addiction: A randomized trial of cbt4cbt. Am. J. Psychiatry 2008, 165, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Horrell, T.; El-Baz, A.; Baruth, J.; Tasman, A.; Sokhadze, G.; Stewart, C.; Sokhadze, E. Neurofeedback effects on evoked and induced eeg gamma band reactivity to drug-related cues in cocaine addiction. J. Neurother. 2010, 14, 195–216. [Google Scholar] [CrossRef] [PubMed]
- Dehghani-Arani, F.; Rostami, R.; Nadali, H. Neurofeedback training for opiate addiction: Improvement of mental health and craving. Appl. Psychophysiol. Biofeedback 2013, 38, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Young, K.S. Internet addiction: The emergence of a new clinical disorder. Cyberpsychol. Behav. 1998, 1, 237–244. [Google Scholar] [CrossRef]
- Beard, K.W.; Wolf, E.M. Modification in the proposed diagnostic criteria for internet addiction. CyberPsychol. Behav. 2001, 4, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, J.-Y.; Im, C.-H. Fast and robust real-time estimation of respiratory rate from photoplethysmography. Sensors 2016, 16, 1494. [Google Scholar] [CrossRef] [PubMed]
- Green, S.R.; Kragel, P.A.; Fecteau, M.E.; LaBar, K.S. Development and validation of an unsupervised scoring system (autonomate) for skin conductance response analysis. Int. J. Psychophysiol. 2014, 91, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-R.; Chang, W.-D.; Kim, S.; Im, C.-H. Real-time “eye-writing” recognition using electrooculogram. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.-D.; Cha, H.-S.; Kim, K.; Im, C.-H. Detection of eye blink artifacts from single prefrontal channel electroencephalogram. Comput. Methods Progr. Biomed. 2016, 124, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.-D.; Cha, H.-S.; Kim, S.H.; Im, C.-H. Development of an electrooculogram-based eye-computer interface for communication of individuals with amyotrophic lateral sclerosis. J. Neuroeng. Rehabil. 2017, 14, 89. [Google Scholar] [CrossRef] [PubMed]
- Lilliefors, H.W. On the kolmogorov-smirnov test for normality with mean and variance unknown. J. Am. Stat. Assoc. 1967, 62, 399–402. [Google Scholar] [CrossRef]
- Chang, C.-C.; Lin, C.-J. Libsvm: A library for support vector machines. ACM Trans. Intell. Syst. Technol. 2011, 2. [Google Scholar] [CrossRef]
- Sun, Y.; Ying, H.; Seetohul, R.M.; Xuemei, W.; Ya, Z.; Qian, L.; Guoqing, X.; Ye, S. Brain fmri study of crave induced by cue pictures in online game addicts (male adolescents). Behav. Brain Res. 2012, 233, 563–576. [Google Scholar] [CrossRef] [PubMed]
- Styn, M.A.; Bovbjerg, D.H.; Lipsky, S.; Erblich, J. Cue-induced cigarette and food craving: A common effect? Addict. Behav. 2013, 38, 1840–1843. [Google Scholar] [CrossRef] [PubMed]
- Miedl, S.F.; Büchel, C.; Peters, J. Cue-induced craving increases impulsivity via changes in striatal value signals in problem gamblers. J. Neurosci. 2014, 34, 4750–4755. [Google Scholar] [CrossRef] [PubMed]
- Silverthorn, D.U.; Ober, W.C.; Garrison, C.W.; Silverthorn, A.C.; Johnson, B.R. Human Physiology: An Integrated Approach; Pearson/Benjamin Cummings: San Francisco, CA, USA, 2009. [Google Scholar]
- Knott, V.J.; Naccache, L.; Cyr, E.; Fisher, D.J.; McIntosh, J.F.; Millar, A.M.; Villeneuve, C.M. Craving-induced eeg reactivity in smokers: Effects of mood induction, nicotine dependence and gender. Neuropsychobiology 2008, 58, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Yamamoto, Y.; Kitajo, K.; Takahashi, T.; Kitazawa, S. Synchronization of spontaneous eyeblinks while viewing video stories. Proc. R. Soc. B Biol. Sci. 2009, 276, 3635–3644. [Google Scholar] [CrossRef] [PubMed]
- Ledger, H. The effect cognitive load has on eye blinking. Plymouth Stud. Sci. 2013, 6, 206–223. [Google Scholar]
- Shin, Y.S.; Park, J.; Im, C.-H.; Lee, S.I.; Kim, I.Y.; Jang, D.P. Correlation between inter-blink interval and episodic encoding during movie watching. PLoS ONE 2015, 10, e0141242. [Google Scholar] [CrossRef] [PubMed]
- Mogg, K.; Bradley, B.P.; Field, M.; De Houwer, J. Eye movements to smoking-related pictures in smokers: Relationship between attentional biases and implicit and explicit measures of stimulus valence. Addiction 2003, 98, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Fishbein, D.; Hyde, C.; Eldreth, D.; London, E.D.; Matochik, J.; Ernst, M.; Isenberg, N.; Steckley, S.; Schech, B.; Kimes, A. Cognitive performance and autonomic reactivity in abstinent drug abusers and nonusers. Exp. Clin. Psychopharmacol. 2005, 13, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.S.; Prichep, L.S.; Ciplet, D.; O’Leary, S.; Tom, M.; Howard, B.; Rotrosen, J.; John, E.R. Quantitative electroencephalographic studies of cue-induced cocaine craving. Clin. EEG 2003, 34, 110–123. [Google Scholar] [CrossRef]
- Stork, M. Some methods systems and sensors which are possible for driver’s drowsiness estimation. In Latest Trends in Circuits, Automatic Control and Signal Processing; WSEAS Press: Barcelona, Spain, 2012; pp. 87–94. [Google Scholar]
- Min, K.B.; Min, J.-Y.; Paek, D.; Cho, S.-I.; Son, M. Is 5-minute heart rate variability a useful measure for monitoring the autonomic nervous system of workers? Int. Heart J. 2008, 49, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Wikipedia. Heart Rate Variability. Available online: https://en.wikipedia.org/wiki/Heart_rate_variability (accessed on 21 October 2017).
| Feature (Unit) | Description | Feature (Unit) | Description |
|---|---|---|---|
| stdHR (beats per minute) | Standard deviation of heart rate | DHSM (µV) | Degree of horizontal saccadic movement |
| mHR (beats per minute) | Mean heart rate | DVSM (μV) | Degree of vertical saccadic movement |
| stdRR (breaths per minute) | Standard deviation of respiratory rate | mDHV (μV) | Mean of DHSM and DVSM |
| mRR (breaths per minute) | Mean respiratory rate | DSM (μV) | Degree of saccadic movement |
| mNSC (no unit) | Mean amplitude of normalized skin conductance response | CHV (μV2) | Covariance of horizontal EOG and vertical EOG |
| minNSC (no unit) | Minimum amplitude of normalized skin conductance response | CHP (μV) | Covariance of horizontal EOG and PPG |
| NE (no unit) | The number of eye blinks | CVP (μV) | Covariance of vertical EOG and PPG |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Ha, J.; Chang, W.-D.; Park, W.; Kim, L.; Im, C.-H. Detection of Craving for Gaming in Adolescents with Internet Gaming Disorder Using Multimodal Biosignals. Sensors 2018, 18, 102. https://doi.org/10.3390/s18010102
Kim H, Ha J, Chang W-D, Park W, Kim L, Im C-H. Detection of Craving for Gaming in Adolescents with Internet Gaming Disorder Using Multimodal Biosignals. Sensors. 2018; 18(1):102. https://doi.org/10.3390/s18010102
Chicago/Turabian StyleKim, Hodam, Jihyeon Ha, Won-Du Chang, Wanjoo Park, Laehyun Kim, and Chang-Hwan Im. 2018. "Detection of Craving for Gaming in Adolescents with Internet Gaming Disorder Using Multimodal Biosignals" Sensors 18, no. 1: 102. https://doi.org/10.3390/s18010102
APA StyleKim, H., Ha, J., Chang, W.-D., Park, W., Kim, L., & Im, C.-H. (2018). Detection of Craving for Gaming in Adolescents with Internet Gaming Disorder Using Multimodal Biosignals. Sensors, 18(1), 102. https://doi.org/10.3390/s18010102









