Carbon Nanomaterial Based Biosensors for Non-Invasive Detection of Cancer and Disease Biomarkers for Clinical Diagnosis
Abstract
1. Introduction
2. Construction of Biosensors for Biomarkers Detection
2.1. Electrochemical Biosensors
2.2. Optical-Based Biosensors
2.3. Piezoelectric-Based Biosensors
3. Detection of Cancer and Disease Biomarkers
3.1. CNT-Based Biomarker Biosensors
3.1.1. CNT-Based Biosensors for Cancer Biomarker Detection
3.1.2. CNT-Based Biosensors for Disease Biomarker Detection
3.2. Graphene-Based Biomarker Biosensors
3.2.1. Graphene-Based Biosensors for Cancer Biomarker Detection
3.2.2. Graphene-Based Biosensors for Disease Biomarker Detection
3.3. Carbon Quantum-Dot-Based Biomarker Biosensors
4. Conclusions and Outlook
Acknowledgments
Conflicts of Interest
References
- Zhou, Q.; Hu, H.-G.; Hou, L. Discover, Develop & Validate—Advance and Prospect of Tumor Biomarkers. Clin. Lab. 2015, 61, 1589–1599. [Google Scholar] [PubMed]
- Tripathi, K.M.; Kim, T.Y.; Losic, D.; Tung, T.T. Recent advances in engineered graphene and composites for detection of volatile organic compounds (VOCs) and non-invasive diseases diagnosis. Carbon 2016, 110, 97–129. [Google Scholar] [CrossRef]
- Susana Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Electrochemical Genosensing of Circulating Biomarkers. Sensors 2017, 17, 866. [Google Scholar] [CrossRef] [PubMed]
- Butler, J. Enzyme-Linked Immunosorbent Assay. J. Immunoass. Immunochem. 2000, 21, 165–209. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, S.J. Radioimmunoassay: Review of basic principles. Semin. Nucl. Med. 1975, 5, 125–152. [Google Scholar] [CrossRef]
- Schmalzing, D.; Nashabeh, W. Capillary electrophoresis based immunoassays: A critical review. Electrophoresis 1997, 18, 2184–2193. [Google Scholar] [CrossRef] [PubMed]
- Diamandis, E.P.; van der Merwe, D.E. Plasma Protein Profiling by Mass Spectrometry for Cancer Diagnosis: Opportunities and Limitations. Clin. Cancer Res. 2005, 11, 963–965. [Google Scholar] [PubMed]
- Widjojoatmodjo, M.N.; Fluit, A.C.; Torensma, R.; Verdonk, G.P.; Verhoef, J. The magnetic immuno polymerase chain reaction assay for direct detection of salmonellae in fecal samples. J. Clin. Microbiol. 1992, 30, 3195–3199. [Google Scholar] [PubMed]
- Wang, L.; Zhang, Y.; Wu, A.; Wei, G. Designed graphene-peptide nanocomposites for biosensor applications: A review. Anal. Chim. Acta 2017. [Google Scholar] [CrossRef]
- Grieshaber, D.; MacKenzie, R.; Vörös, J.; Reimhult, E. Electrochemical Biosensors—Sensor Principles and Architectures. Sensors 2008, 8, 1400–1458. [Google Scholar] [CrossRef] [PubMed]
- Pohanka, M. The Piezoelectric Biosensors: Principles and Applications, a Review. Int. J. Electrochem. Sci. 2017, 12, 496–506. [Google Scholar] [CrossRef]
- Touhami, A. Biosensors and Nanobiosensors: Design and Applications. In Nanomedicine; Seifalian, A., Kalaskar, M., Eds.; One Central Press (OCP): Altrincham, UK, 2014; Chapter 15; pp. 374–403. [Google Scholar]
- Krebsz, M.; Pasinszki, T.; Tung, T.T.; Losic, D. Development of Vapor/Gas Sensors from Biopolymer Composites. In Biopolymer Composites in Electronics; Sadasivuni, K.K., Ponnamma, D., Kim, J., Cabibihan, J.-J., AlMaadeed, M.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; Chapter 14; pp. 385–403. [Google Scholar]
- Tiwari, J.N.; Vij, V.; Kemp, K.C.; Kim, K.S. Engineered Carbon-Nanomaterial-Based Electrochemical Sensors for Biomolecules. ACS Nano 2016, 10, 46–80. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, A.; Chua, C.K.; Bonanni, A.; Pumera, M. Electrochemistry of Graphene and Related Materials. Chem. Rev. 2014, 114, 7150–7188. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xiong, Q.; Xiao, F.; Duan, H. 2D nanomaterials based electrochemical biosensors for cancer diagnosis. Biosens. Bioelectron. 2017, 89, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Hasanzadeh, M.; Shadjou, N. What are the reasons for low use of graphene quantum dots in immunosensing of cancer biomarkers? Mater. Sci. Eng. C 2017, 71, 1313–1326. [Google Scholar] [CrossRef] [PubMed]
- Tung, T.T.; Nine, M.J.; Krebsz, M.; Pasinszki, T.; Coghlan, C.J.; Tran, D.N.H.; Losic, D. Recent Advances in Sensing Applications of Graphene Assemblies and Their Composites. Adv. Funct. Mater. 2017. accepted. [Google Scholar]
- Tilmaciu, C.-M.; Morris, M.C. Carbon nanotube biosensors. Front. Chem. 2015, 3, 1–82. [Google Scholar] [CrossRef] [PubMed]
- Patil, A.V.; Fernandes, F.B.; Bueno, P.R.; Davis, J.J. Graphene-based protein biomarker Detection. Bioanalysis 2015, 7, 725–742. [Google Scholar] [CrossRef] [PubMed]
- Cruz, S.M.; Girão, A.F.; Gonçalves, G.; Marques, P.A. Graphene: The Missing Piece for Cancer Diagnosis? Sensors 2016, 16, 137. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Fan, H.; Li, Y.; Zhang, Y.; Liang, H.; Wei, Q. Ultrasensitive electrochemical immunoassay for squamous cell carcinoma antigen using dumbbell-like Pt-Fe3O4 nanoparticles as signal amplification. Biosens. Bioelectron. 2013, 46, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Yagati, A.K.; Choi, Y.; Park, J.; Choi, J.-W.; Jun, H.-S.; Cho, S. Silver nanoflower-reduced graphene oxide composite based micro-disk electrode for insulin detection in serum. Biosens. Bioelectron. 2016, 80, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Tabasi, A.; Noorbakhsh, A.; Sharifi, E. Reduced graphene oxide-chitosan-aptamer interface as new platform for ultrasensitive detection of human epidermal growth factor receptor 2. Biosens. Bioelectron. 2017, 95, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, J.; Wang, T.; Li, D.; Xi, F.; Wang, J.; Wang, E. Three-dimensional electrochemical immunosensor for sensitive detection of carcinoembryonic antigen based on monolithic and macroporous graphene foam. Biosens. Bioelectron. 2015, 65, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; Huang, J.-Y.; Bao, T.; Zhou, J.; Xia, H.-X.; Zhang, X.-H.; Wang, S.-F.; Zhao, Y.-D. Increased electrocatalyzed performance through hairpin oligonucleotide aptamer-functionalized gold nanorods labels and graphene-streptavidin nanomatrix: Highly selective and sensitive electrochemical biosensor of carcinoembryonic antigen. Biosens. Bioelectron. 2016, 83, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, H.; Aldalbahi, A.; Zuo, X.; Fan, C.; Mi, X. Fluorescent biosensors enabled by graphene and graphene oxide. Biosens. Bioelectron. 2017, 89, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Fan, J.; Ye, W.; Zhang, H.; Cong, Y.; Xiao, J. A Highly Specific Graphene Platform for Sensing Collagen Triple Helix. J. Mater. Chem. B 2016, 6, 1064–1069. [Google Scholar] [CrossRef]
- Ding, Y.; Liu, J.; Jin, X.; Lu, H.; Shen, G.; Yu, R. Poly-l-lysine/hydroxyapatite/carbon nanotube hybrid nanocomposite applied for piezoelectric immunoassay of carbohydrate antigen 19–9. Analyst 2008, 133, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Feng, T.; Wang, Y.; Qiao, X. Recent Advances of Carbon Nanotubes-based Electrochemical Immunosensors for the Detection of Protein Cancer Biomarkers. Electroanalysis 2017, 29, 662–675. [Google Scholar] [CrossRef]
- Justino, I.L.; Rocha-Santos, T.A.P.; Duarte, A.C. Advances in point-of-care technologies with biosensors based on carbon nanotubes. TrAC Trends Anal. Chem. 2013, 45, 24–36. [Google Scholar] [CrossRef]
- Teker, K.; Sirdeshmukh, R.; Sivakumar, K.; Lu, S.; Wickstrom, E.; Wang, H.-N.; Vo-Dinh, T.; Panchapakesan, B. Applications of Carbon Nanotubes for Cancer Research. NanoBiotechnology 2005, 1, 171–182. [Google Scholar]
- Okuno, J.; Maehashi, K.; Kerman, K.; Takamura, Y.; Matsumoto, K.; Tamiya, E. Label-free immunosensor for prostate-specific antigen based on single-walled carbon nanotube array-modified microelectrodes. Biosens. Bioelectron. 2007, 22, 2377–2381. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Huang, J.; Zhao, Y.; Zhao, S. Electrochemical immunosensor for prostate-specific antigen using a glassy carbon electrode modified with a nanocomposite containing gold nanoparticles supported with starch-functionalized multi-walled carbon nanotubes. Microchim. Acta 2012, 178, 81–88. [Google Scholar] [CrossRef]
- Yu, X.; Munge, B.; Patel, V.; Jensen, G.; Bhirde, A.; Gong, J.D.; Kim, S.N.; Gillespie, J.; Gutkind, J.S.; Papadimitrakopoulos, F.; et al. Carbon Nanotube Amplification Strategies for Highly Sensitive Immunodetection of Cancer Biomarkers. J. Am. Chem. Soc. 2006, 128, 11199–11205. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.J.; Xiang, C.L.; Sun, L.X.; Xu, F.; Zhou, H.Y. Ultrasensitive Prostate Specific Antigen Immunosensor Based on Gold Nanoparticles Functionalized Polypyrrole@Carbon Nanotubes. Asian J. Chem. 2014, 26, 8002–8006. [Google Scholar] [CrossRef]
- Lerner, M.B.; D’Souza, J.; Pazina, T.; Dailey, J.; Goldsmith, B.R.; Robinson, M.K.; Johnson, A.T.C. Hybrids of a Genetically Engineered Antibody and a Carbon Nanotube Transistor for Detection of Prostate Cancer Biomarkers. ACS Nano 2012, 6, 5143–5149. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Hong, S.; Singh, R.; Jang, J. Single-walled carbon nanotube based transparent immunosensor for detection of a prostate cancer biomarker osteopontin. Anal. Chim. Acta 2015, 869, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Song, C.; Zhang, Z.; Yang, J.; Zhou, L.; Zhang, X.; Xie, G. Ultrasensitive electrochemical detection of microRNA-21 combining layered nanostructure of oxidized single-walled carbon nanotubes and nanodiamonds by hybridization chain reaction. Biosens. Bioelectron. 2015, 70, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.V.; Piro, B.; Reisberg, S.; Tran, L.D.; Duc, H.T.; Pham, M.C. Label-free and reagentless electrochemical detection of microRNAs using a conducting polymer nanostructured by carbon nanotubes: Application to prostate cancer biomarker miR-141. Biosens. Bioelectron. 2013, 49, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.V.; Piro, B.; Reisberg, S.; Nguyen, L.H.; Nguyen, T.D.; Duc, H.T.; Pham, M.C. An electrochemical ELISA-like immunosensor for miRNAs detection based on screen-printed gold electrodes modified with reduced graphene oxide and carbon nanotubes. Biosens. Bioelectron. 2014, 62, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Ramnani, P.; Gao, Y.; Ozsoz, M.; Mulchandani, A. Electronic Detection of MicroRNA at Attomolar Level with High Specificity. Anal. Chem. 2013, 85, 8061–8064. [Google Scholar] [CrossRef] [PubMed]
- Tian, Q.; Wang, Y.; Deng, R.; Lin, L.; Liu, Y.; Li, J. Carbon nanotube enhanced label-free detection of microRNAs based on hairpin probe triggered solid-phase rolling-circle amplification. Nanoscale 2015, 7, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Peng, J.; Wang, J.; Tang, H.; Tan, L.; Xie, Q.; Yao, S. Carbon nanotube-based label-free electrochemical biosensor for sensitive detection of miRNA-24. Biosens. Bioelectron. 2014, 54, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, Y.; Wang, L.; Song, Y.; Luo, J.; Cai, X. A Label-Free Microelectrode Array Based on One-Step Synthesis of Chitosan-Multi-Walled Carbon Nanotube-Thionine for Ultrasensitive Detection of Carcinoembryonic Antigen. Nanomaterials 2016, 6, 132. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Willander, M.; Sharma, J.G.; Malhotra, B.D. A solution processed carbon nanotube modified conducting paper sensor for cancer detection. J. Mater. Chem. B 2015, 3, 9305–9314. [Google Scholar] [CrossRef]
- Feng, D.; Lu, X.; Dong, X.; Ling, Y.; Zhang, Y. Label-free electrochemical immunosensor for the carcinoembryonic antigen using a glassy carbon electrode modified with electrodeposited Prussian Blue, a graphene and carbon nanotube assembly and an antibody immobilized on gold nanoparticles. Microchim. Acta 2013, 180, 767–774. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, H.; Gao, X.; Chen, Z.; Lin, X. A novel immunosensor based on an alternate strategy of electrodeposition and self-assembly. Biosens. Bioelectron. 2012, 35, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Zhang, Y.; Chen, H.; Chen, Z.; Lin, X. Amperometric immunosensor for carcinoembryonic antigen detection with carbon nanotube-based film decorated with gold nanoclusters. Anal. Biochem. 2011, 414, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Li, X.; Wang, L.; Wu, Q.; Chen, Z.; Lin, X. Sandwich-type amperometric immunosensor for cancer biomarker based on signal amplification strategy of multiple enzyme-linked antibodies as probes modified with carbon nanotubes and concanavalin A. J. Electroanal. Chem. 2014, 732, 38–45. [Google Scholar] [CrossRef]
- Cheng, H.; Lai, G.; Fu, L.; Zhang, H.; Yu, A. Enzymatically catalytic deposition of gold nanoparticles by glucose oxidase-functionalized gold nanoprobe for ultrasensitive electro- chemical immunoassay. Biosens. Bioelectron. 2015, 71, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Liu, F.; Ge, S.; Yu, J.; Yan, M.; Song, X. A dual amplification strategy for ultrasensitive electrochemiluminescence immunoassay based on a Pt nanoparticles dotted grapheme-carbon nanotubes composite and carbon dots functionalized mesoporous Pt/Fe. Analyst 2014, 139, 1713–1720. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Zheng, J.; Su, X.; Wang, J.; Wu, W.; Hu, S. Ultrasensitive All-Carbon Photoelectrochemical Bioprobes for Zeptomole Immunosensing of Tumor Markers by an Inexpensive Visible Laser Light. Anal. Chem. 2013, 85, 10612–10619. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Wang, Y.; Cao, W.; Zhang, Y.; Yan, T.; Du, B.; Wei, Q. An ultrasensitive electrochemical immunosensor for CEA using MWCNT-NH2 supported PdPt nanocages as labels for signal amplification. J. Mater. Chem. B 2015, 3, 2006–2011. [Google Scholar] [CrossRef]
- Paul, K.B.; Singh, V.; Vanjari, S.R.K.; Singh, S.G. One step biofunctionalized electrospun multiwalled carbon nanotubes embedded zinc oxide nanowire interface for highly sensitive detection of carcinoma antigen-125. Biosens. Bioelectron. 2017, 88, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yuan, R.; Chai, Y.; Min, L.; Li, W.; Xu, Y. Electrochemical sensing platform based on tris(2,2′-bipyridyl)cobalt(III) and multiwall carbon nanotubes-Nafion composite for immunoassay of carcinoma antigen-125. Electrochim. Acta 2009, 54, 7242–7247. [Google Scholar] [CrossRef]
- Fayazfar, H.; Afshar, A.; Dolati, M.; Dolati, A. DNA impedance biosensor for detection of cancer, TP53 gene mutation, based on gold nanoparticles/aligned carbon nanotubes modified electrode. Anal. Chim. Acta 2014, 836, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gao, C.; Shu, G.; Wang, Y.; Liu, X. The enzyme electrocatalytic immunosensor based on functional composite nanofibers for sensitive detection of tumor suppressor protein p53. J. Electroanal. Chem. 2015, 756, 101–107. [Google Scholar] [CrossRef]
- Wang, X.; Shu, G.; Gao, C.; Yang, Y.; Xu, Q.; Tang, M. Electrochemical biosensor based on functional composite nanofibers for detection of K-ras gene via multiple signal amplification strategy. Anal. Biochem. 2014, 466, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.K.; Bold, B.; Lee, W.K.; Jeon, M.H.; An, K.H.; Jeong, S.Y.; Shim, Y.K. D-(+)-Galactose-Conjugated Single-Walled Carbon Nanotubes as New Chemical Probes for Electrochemical Biosensors for the Cancer Marker Galectin-3. Int. J. Mol. Sci. 2011, 12, 2946–2957. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, Q.; Qian, C.; Hao, N.; Xu, L.; Yao, C. Electrochemical aptasensor for mucin 1 based on dual signal amplification of poly(o-phenylenediamine) carrier and functionalized carbon nanotubes tracing tag. Biosens. Bioelectron. 2015, 64, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Arkan, E.; Saber, R.; Karimi, Z.; Shamsipur, M. A novel antibody-antigen based impedimetric immunosensor for low level detection of HER2 in serum samples of breast cancer patients via modification of a gold nanoparticles decorated multiwall carbon nanotube-ionic liquid electrode. Anal. Chim. Acta 2015, 874, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Asav, E.; Sezgintürk, M.K. A novel impedimetric disposable immunosensor for rapid detection of a potential cancer biomarker. Int. J. Biol. Macromol. 2014, 66, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Munge, B.S.; Fisher, J.; Millord, L.N.; Krause, C.E.; Dowda, R.S.; Rusling, J.F. Sensitive electrochemical immunosensor for matrix metalloproteinase-3 based on single-wall carbon nanotubes. Analyst 2010, 135, 1345–1350. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.-C.; Chen, H.-Y.; Wang, Y.; Moochhala, S.M.; Alagappan, P.; Liedberg, B. Immunosensor based on carbon nanotube/manganese dioxide electrochemical tags. Anal. Chim. Acta 2015, 853, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Li, Z.; Wei, X.; Huang, R.; Qi, H.; Gao, Q.; Li, C.; Zhang, C. Detection and discrimination of alpha-fetoprotein with a label-free electrochemical impedance spectroscopy biosensor array based on lectin functionalized carbon nanotubes. Talanta 2013, 111, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Han, J.; Jiang, L.; Wang, Y.; Li, Y.; Dong, Y.; Wei, Q. An ultrasensitive sandwich-type electrochemical immunosensor based on signal amplification strategy of gold nanoparticles functionalized magnetic multi-walled carbon nanotubes loaded with lead ions. Biosens. Bioelectron. 2015, 68, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, M.; Singh, A.; Kaur, S.; Arora, K. Enhancing Lung Cancer Diagnosis: Electrochemical Simultaneous Bianalyte Immunosensing Using Carbon Nanotubes-Chitosan Nanocomposite. Appl. Biochem. Biotechnol. 2014, 174, 1188–1200. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Tirado, E.; Salvo, C.; González-Cortés, A.; Yánnez-Sedenno, P.; Langa, F.; Pingarrón, J.M. Electrochemical immunosensor for simultaneous determination of interleukin-1 beta and tumor necrosis factor alpha in serum and saliva using dual screen printed electrodes modified with functionalized doubleewalled carbon nanotubes. Anal. Chim. Acta 2017, 959, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Pal, M.; Kuzikov, A.V.; Bulko, T.; Suprun, E.V.; Shumyantseva, V.V. Impedimetric immunosensor for detection of cardiovascular disorder risk biomarker. Mater. Sci. Eng. C 2016, 68, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; He, J.; Gao, L.; Zhang, J.; Yu, C. Immunoassay for netrin 1 via a glassy carbon electrode modified with multi-walled carbon nanotubes, thionine and gold nanoparticles. Microchim. Acta 2015, 182, 2115–2122. [Google Scholar] [CrossRef]
- Herrasti, Z.; Martínez, F.; Baldrich, E. Carbon nanotube wiring for signal amplification of electrochemical magneto immunosensors: Application to myeloperoxidase detection. Anal. Bioanal. Chem. 2014, 406, 5487–5493. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Liu, B.; Liu, C.; Xie, G. Amperometric Immunosensor for Myeloperoxidase in Human Serum Based on a Multi-wall Carbon Nanotubes-Ionic Liquid-Cerium Dioxide Film-modified Electrode. Bull. Korean Chem. Soc. 2010, 31, 3259–3264. [Google Scholar] [CrossRef]
- Liu, B.; Lu, L.; Li, Q.; Xie, G. Disposable electrochemical immunosensor for myeloperoxidase based on the indium tin oxide electrode modified with an ionic liquid composite film containing gold nanoparticles, poly(o-phenylenediamine) and carbon nanotubes. Microchim. Acta 2011, 173, 513–520. [Google Scholar] [CrossRef]
- Navamani, J.; Palanisamy, R.; Gurusamy, R.; Ramasamy, M.; Arumugam, S. Development of Nanoprobe for the Determination of Blood Cholesterol. J. Biosens. Bioelectron. 2012, 3. [Google Scholar] [CrossRef]
- Son, M.; Kim, D.; Park, K.S.; Hong, S.; Park, T.H. Detection of aquaporin-4 antibody using aquaporin-4 extracellular loop-based carbon nanotube biosensor for the diagnosis of neuromyelitis optica. Biosens. Bioelectron. 2016, 78, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Lerner, M.B.; Dailey, J.; Goldsmith, B.R.; Brisson, D.; Johnson, A.T.C. Detecting Lyme disease using antibody-functionalized single-walled carbon nanotube transistors. Biosens. Bioelectron. 2013, 45, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Lee, H.J. Electrochemical sandwich-type biosensors for α−1 antitrypsin with carbon nanotubes and alkaline phosphatase labeled antibody-silver nanoparticles. Biosens. Bioelectron. 2017, 89, 959–963. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Yoo, G.; Chang, Y.W.; Kim, H.J.; Jose, J.; Kim, E.; Pyun, J.-C.; Yoo, K.-H. A carbon nanotube metal semiconductor field effect transistor-based biosensor for detection of amyloid-beta in human serum. Biosens. Bioelectron. 2013, 50, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Pundir, S.; Chauhan, N.; Narang, J.; Pundir, C.S. Amperometric choline biosensor based on multiwalled carbon nanotubes/zirconium oxide nanoparticles electrodeposited on glassy carbon electrode. Anal. Biochem. 2012, 427, 26–32. [Google Scholar] [CrossRef] [PubMed]
- An, Y.; Zhu, G.; Bi, W.; Lu, L.; Feng, C.; Xu, Z.; Zhang, W. Highly sensitive electrochemical immunoassay integrated with polymeric nanocomposites and enhanced SiO2@Au core-shell nanobioprobes for SirT1 determination. Anal. Chim. Acta 2017, 966, 54–61. [Google Scholar] [CrossRef] [PubMed]
- De Gracia Villa, M.; Jiménez-Jorquera, C.; Haro, I.; José Gomara, M.; Sanmartí, R.; Fernández-Sánchez, C.; Mendoza, E. Carbon nanotube composite peptide-based biosensors as putative diagnostic tools for rheumatoid arthritis. Biosens. Bioelectron. 2011, 27, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Drouvalakis, K.A.; Bangsaruntip, S.; Hueber, W.; Kozar, L.G.; Utz, P.J.; Dai, H. Peptide-coated nanotube-based biosensor for the detection of disease-specific autoantibodies in human serum. Biosens. Bioelectron. 2008, 23, 1413–1421. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.-S.; Chen, S.-Y.; Huang, X.-J.; Wang, L.-S.; Wang, H.-Y.; Wang, J.-F. Simple approach for ultrasensitive electrochemical immunoassay of Clostridium difficile toxin B detection. Biosens. Bioelectron. 2014, 53, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Anirudhan, T.S.; Alexander, S.; Lilly, A. Surface modified multiwalled carbon nanotube based molecularly imprinted polymer for the sensing of dopamine in real samples using potentiometric method. Polymer 2014, 55, 4820–4831. [Google Scholar] [CrossRef]
- Prasad, B.B.; Jauhari, D.; Tiwari, M.P. A dual-template imprinted polymer-modified carbon ceramic electrode for ultra trace simultaneous analysis of ascorbic acid and dopamine. Biosens. Bioelectron. 2013, 50, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.R.; Ma, Y.; Parajuli, R.R.; Balogun, Y.; Lai, W.Y.-C.; He, H. A Nonoxidative Sensor Based on a Self-Doped Polyaniline/Carbon Nanotube Composite for Sensitive and Selective Detection of the Neurotransmitter Dopamine. Anal. Chem. 2007, 79, 2583–2587. [Google Scholar] [CrossRef] [PubMed]
- Canevari, T.C.; Raymundo-Pereira, P.A.; Landers, R.; Benvenutti, E.V.; Machado, S.A.S. Sol-gel thin-film based mesoporous silica and carbon nanotubes for the determination of dopamine, uric acid and paracetamol in urine. Talanta 2013, 116, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wang, Q.; Jin, J.; Wu, P.; Wang, H.; Yu, S.; Zhang, H.; Cai, C. Low-Potential Detection of Endogenous and Physiological Uric Acid at Uricase-Thionine-Single-Walled Carbon Nanotube Modified Electrodes. Anal. Chem. 2010, 82, 2448–2455. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Musameh, M. Carbon-nanotubes doped polypyrrole glucose biosensor. Anal. Chim. Acta 2005, 539, 209–213. [Google Scholar] [CrossRef]
- Valentini, F.; Galache Fernandez, L.; Tamburri, E.; Palleschi, G. Single Walled Carbon Nanotubes/polypyrrole-GOx composite films to modify gold microelectrodes for glucose biosensors: Study of the extended linearity. Biosens. Bioelectron. 2013, 43, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Diagnostics Strategies with Electrochemical Affinity Biosensors Using Carbon Nanomaterials as Electrode Modifiers. Diagnostics 2017, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Bollella, P.; Fusco, G.; Tortolini, C.; Sanzò, G.; Favero, G.; Gorton, L.; Antiochia, R. Beyond graphene: Electrochemical sensors and biosensors for biomarkers detection. Biosens. Bioelectron. 2017, 89, 152–166. [Google Scholar] [CrossRef] [PubMed]
- Janegitz, B.C.; Silva, T.A.; Wong, A.; Ribovski, L.; Vicentini, F.C.; Sotomayor, M.P.T.; Fatibello-Filho, O. The application of graphene for in vitro and in vivo electrochemical biosensing. Biosens. Bioelectron. 2017, 89, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Justino, C.I.L.; Gomes, A.R.; Freitas, A.C.; Duarte, A.C.; Rocha-Santos, T.A.P. Graphene based sensors and biosensors. TrAC Trends Anal. Chem. 2017, 91, 53–66. [Google Scholar] [CrossRef]
- Vilela, P.; El-Sagheer, A.; Millar, T.M.; Brown, T.; Muskens, O.L.; Kanaras, A.G. Graphene Oxide-Upconversion Nanoparticle Based Optical Sensors for Targeted Detection of mRNA Biomarkers Present in Alzheimer’s Disease and Prostate Cancer. ACS Sens. 2017, 2, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Cui, T. An ultrasensitive and low-cost graphene sensor based on layer-by-layer nano self-assembly. Appl. Phys. Lett. 2011, 98, 073116. [Google Scholar] [CrossRef]
- Zhang, B.; Li, Q.; Cui, T. Ultra-sensitive suspended graphene nanocomposite cancer sensors with strong suppression of electrical noise. Biosens. Bioelectron. 2012, 31, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, B.; Cui, T. Towards intrinsic graphene biosensor: A label-free, suspended single crystalline graphene sensor for multiplex lung cancer tumor markers detection. Biosens. Bioelectron. 2015, 72, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, B.; Cui, T. TiO2 and shrink induced tunable nano self-assembled graphene composites for label free biosensors. Sens. Actuators B Chem. 2015, 216, 337–342. [Google Scholar] [CrossRef]
- Li, L.; Li, W.; Yang, H.; Ma, C.; Yu, J.; Yan, M.; Song, X. Sensitive origami dual-analyte electrochemical immunodevice based on polyaniline/Au-paper electrode and multi-labeled 3D graphene sheets. Electrochim. Acta 2014, 120, 102–109. [Google Scholar] [CrossRef]
- Chen, X.; Jia, X.; Han, J.; Ma, J.; Ma, Z. Electrochemical immunosensor for simultaneous detection of multiplex cancer biomarkers based on graphene nanocomposites. Biosens. Bioelectron. 2013, 50, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, N.; Ma, Z. Platinum porous nanoparticles hybrid with metal ions as probes for simultaneous detection of multiplex cancer biomarkers. Biosens. Bioelectron. 2014, 53, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Liu, N.; Yuan, J.; Ma, Z. Triple tumor markers assay based on carbon-gold nanocomposite. Biosens. Bioelectron. 2015, 70, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Chai, Y.; Zhuo, Y.; Yuan, R. Ultrasensitive simultaneous detection of four biomarkers based on hybridization chain reaction and biotin-streptavidin signal amplification strategy. Biosens. Bioelectron. 2015, 68, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Md Ali, A.; Mondal, K.; Jiao, Y.; Oren, S.; Xu, Z.; Sharma, A.; Dong, L. Microfluidic Immuno-Biochip for Detection of Breast Cancer Biomarkers Using Hierarchical Composite of Porous Graphene and Titanium Dioxide Nanofibers. ACS Appl. Mater. Interfaces 2016, 8, 20570–20582. [Google Scholar] [CrossRef] [PubMed]
- Rajesh; Gao, Z.; Vishnubhotla, R.; Ducos, P.; Serrano, M.D.; Ping, J.; Robinson, M.K.; Johnson, A.T.C. Genetically Engineered Antibody Functionalized Platinum Nanoparticles Modified CVD-Graphene Nanohybrid Transistor for the Detection of Breast Cancer Biomarker, HER3. Adv. Mater. Interfaces 2016, 3, 1600124. [Google Scholar]
- Akter, R.; Jeong, B.; Choi, J.-S.; Md Rahman, A. Ultrasensitive Nanoimmunosensor by coupling non-covalent functionalized graphene oxide platform and numerous ferritin labels on carbon nanotubes. Biosens. Bioelectron. 2016, 80, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Shiddiky, M.J.A.; Kithva, P.H.; Kozak, D.; Trau, M. An electrochemical immunosensor to minimize the nonspecific adsorption and to improve sensitivity of protein assays in human serum. Biosens. Bioelectron. 2012, 38, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Guo, Z.; Liu, Y.; Guo, A.; Lou, W.; Fan, D.; Wei, Q. Sandwich-type electrochemical immunosensor using dumbbell-like nanoparticles for the determination of gastric cancer biomarker CA72-4. Talanta 2015, 134, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Mazloum-Ardakani, M.; Hosseinzadeh, L. A Sensitive Electrochemical Aptasensor for TNF-α Based on Bimetallic Ag@Pt Core-Shell Nanoparticle Functionalized Graphene Nanostructures as Labels for Signal Amplification. J. Electrochem. Soc. 2016, 163, B119–B124. [Google Scholar] [CrossRef]
- Cheng, F.-F.; He, T.-T.; Miao, H.-T.; Shi, J.-J.; Jiang, L.-P.; Zhu, J.-J. Electron Transfer Mediated Electrochemical Biosensor for MicroRNAs Detection Based on Metal Ion Functionalized Titanium Phosphate Nanospheres at Attomole Level. ACS Appl. Mater. Interfaces 2015, 7, 2979–2985. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Li, W.; Wu, P.; Zhang, H.; Cai, C. Fluorescence Quenching of Graphene Oxide Integrating with the Site-Specific Cleavage of the Endonuclease for Sensitive and Selective MicroRNA Detection. Anal. Chem. 2013, 85, 2536–2542. [Google Scholar] [CrossRef] [PubMed]
- Cai, B.; Huang, L.; Zhang, H.; Sun, Z.; Zhang, Z.; Zhang, G.-J. Gold Nanoparticles-Decorated Graphene Field-Effect Transistor Biosensor for Femtomolar MicroRNA Detection. Biosens. Bioelectron. 2015, 74, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Hizir, M.S.; Balcioglu, M.; Rana, M.; Robertson, N.M.; Yigit, M.V. Simultaneous Detection of Circulating OncomiRs from Body Fluids for Prostate Cancer Staging Using Nanographene Oxide. ACS Appl. Mater. Interfaces 2014, 6, 14772–14778. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hu, X.; Shi, S.; Zhang, Y.; Yao, T. Three label-free thrombin aptasensors based on aptamers and [Ru(bpy)2(o-mopip)]2+. J. Mater. Chem. B 2016, 4, 1361–1367. [Google Scholar] [CrossRef]
- Wang, X.; Wang, C.; Qu, K.; Song, Y.; Ren, J.; Miyoshi, D.; Sugimoto, N.; Qu, X. Ultrasensitive and Selective Detection of a Prognostic Indicator in Early-Stage Cancer Using Graphene Oxide and Carbon Nanotubes. Adv. Funct. Mater. 2010, 20, 3967–3971. [Google Scholar] [CrossRef]
- Feng, L.; Wu, L.; Wang, J.; Ren, J.; Miyoshi, D.; Sugimoto, N.; Qu, X. Detection of a Prognostic Indicator in Early-Stage Cancer Using Functionalized Graphene-Based Peptide Sensors. Adv. Mater. 2012, 24, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, F.; Zaidi, S.A.; Koo, C.M. Highly sensitive electrochemical sensor based on environmentally friendly biomass-derived sulfur-doped graphene for cancer biomarker detection. Sens. Actuators B Chem. 2017, 241, 716–724. [Google Scholar] [CrossRef]
- Jia, L.-P.; Liu, J.-F.; Wang, H.-S. Electrochemical performance and detection of 8-Hydroxy-2′-deoxyguanosine at single-stranded DNA functionalized graphene modified glassy carbon electrode. Biosens. Bioelectron. 2015, 67, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Azzouzi, S.; Rotariu, L.; Benito, A.M.; Maser, W.K.; Ali, M.B.; Bala, C. A novel amperometric biosensor based on gold nanoparticles anchored on reduced graphene oxide for sensitive detection of l-lactate tumor biomarker. Biosens. Bioelectron. 2015, 69, 280–286. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Wang, Q.; Mandler, D.; Li, M.; Boukherroub, R.; Szunerits, S. Detection of folic acid protein in human serum using reduced graphene oxide electrodes modified by folic-acid. Biosens. Bioelectron. 2016, 75, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Song, E.; Cheng, D.; Song, Y.; Jiang, M.; Yu, J.; Wang, Y. A graphene oxide-based FRET sensor for rapid and sensitive detection of matrix metalloproteinase 2 in human serum sample. Biosens. Bioelectron. 2013, 47, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Sharma, J.G.; Maji, S.; Malhotra, B.D. Nanostructured zirconia decorated reduced graphene oxide based efficient biosensing platform for non-invasive oral cancer detection. Biosens. Bioelectron. 2016, 78, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Du, B.; Zhang, X.; Guo, A.; Zhang, Y.; Wu, D.; Ma, H.; Wei, Q. Ultrasensitive enzyme-free immunoassay for squamous cell carcinoma antigen using carbon supported Pd-Au as electrocatalytic labels. Anal. Chim. Acta 2014, 833, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wei, Q.; Zhang, Y.; Wu, D.; Ma, H.; Guo, A.; Du, B. A sandwich type immunosensor using Pd-Pt nanocrystals as labels for sensitive detection of human tissue polypeptide antigen. Nanotechnology 2014, 25, 055102. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zou, G.; Wei, Q. Ultrasensitive electrochemical immunosensor for quantitative detection of tumor specific growth factor by using Ag@CeO2 nanocomposite as labels. Talanta 2016, 156–157, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Zhang, X.; Li, X.; Li, R.; Du, B.; Wei, Q. Electrochemical immunosensor for detecting typical bladder cancer biomarker based on reduced graphene oxide-tetraethylene pentamine and trimetallic AuPdPt nanoparticles. Talanta 2015, 143, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-C.; Chen, Y.-T.; Tsai, R.-Y.; Chen, M.-C.; Chen, S.-L.; Xiao, M.-C.; Chen, C.-L.; Hua, M.-Y. A sensitive and selective magnetic graphene composite-modified polycrystalline-silicon nanowire field-effect transistor for bladder cancer diagnosis. Biosens. Bioelectron. 2015, 66, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Chae, M.-S.; Lee, S.M.; Jeong, D.; Lee, B.C.; Lee, J.H.; Kim, Y.S.; Chang, S.T.; Hwang, K.S. Wafer-scale high-resolution patterning of reduced graphene oxide films for detection of low concentration biomarkers in plasma. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Qi, M.; Zhang, Y.; Cao, C.; Goldys, E.M. Nanocomposites of gold nanoparticles and graphene oxide towards an stable label-free electrochemical immunosensor for detection of cardiac marker troponin-I. Anal. Chim. Acta 2016, 909, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Liao, N.; Zhuo, Y.; Chai, Y.-Q.; Xiang, Y.; Han, J.; Yuan, R. Reagentless electrochemiluminescent detection of protein biomarker using graphene-based magnetic nanoprobes and poly-l-lysine as co-reactant. Biosens. Bioelectron. 2013, 45, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Xiang, G.; Yuan, R.; Chen, X.; Luo, F.; Jiang, D.; Huang, S.; Li, Y.; Pu, X. Procalcitonin sensitive detection based on grapheme-gold nanocomposite film sensor platform and single-walled carbon nanohorns/hollow Pt chains complex as signal tags. Biosens. Bioelectron. 2014, 60, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, H.S.; Sao, R.; Vaish, R. Label free selective detection of estriol using graphene oxide-based fluorescence sensor. J. Appl. Phys. 2014, 116, 034701. [Google Scholar] [CrossRef]
- Martín, A.; Batalla, P.; Hernández-Ferrer, J.; Martínez, M.T.; Escarpa, A. Graphene oxide nanoribbon-based sensors for the simultaneous bioelectrochemical enantiomeric resolution and analysis of amino acid biomarkers. Biosens. Bioelectron. 2015, 68, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.-L.; Su, C.-H.; Wu, J.-J. Synthesis of short graphene oxide nanoribbons for improved biomarker detection of Parkinson’s disease. Biosens. Bioelectron. 2015, 67, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.Y.; Huang, S.; Chang, J.; Heo, C.; Yao, F.; Adhikari, S.; Gunes, F.; Liu, L.C.; Lee, T.H.; Oh, E.S.; et al. ZnO Nanowire Arrays on 3D Hierarchical Graphene Foam: Biomarker Detection of Parkinson’s Disease. ACS Nano 2014, 8, 1639–1646. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Kuang, D.; Liu, S.; Liu, C.; Zhang, T. Template-assisted self-assembly method to prepare three-dimensional reduced graphene oxide for dopamine sensing. Sens. Actuators B Chem. 2014, 205, 120–126. [Google Scholar] [CrossRef]
- Liu, S.; Yan, J.; He, G.; Zhong, D.; Chen, J.; Shi, L.; Zhou, X.; Jiang, H. Layer-by-layer assembled multilayer films of reduced graphene oxide/gold nanoparticles for the electrochemical detection of dopamine. J. Electroanal. Chem. 2012, 672, 40–44. [Google Scholar] [CrossRef]
- Bai, Y.-F.; Zhang, Y.-F.; Zhou, A.-W.; Li, H.-W.; Zhang, Y.; Luong, J.H.T.; Cui, H.-F. Self-assembly of a thin highly reduced graphene oxide film and its high electrocatalytic activity. Nanotechnology 2014, 25, 405601. [Google Scholar] [CrossRef] [PubMed]
- Manna, B.; Raj, C.R. Covalent Functionalization and Electrochemical Tuning of Reduced Graphene Oxide for the Bioelectrocatalytic Sensing of Serum Lactate. J. Mater. Chem. B 2016, 4, 4585–4593. [Google Scholar] [CrossRef]
- Liu, S.; Tian, J.; Wang, L.; Luo, Y.; Lu, W.; Sun, X. Self-assembled graphene platelet-glucose oxidase nanostructures for glucose biosensing. Biosens. Bioelectron. 2011, 26, 4491–4496. [Google Scholar] [CrossRef] [PubMed]
- Barsan, M.M.; David, M.; Florescu, M.; Ţugulea, L.; Brett, C.M.A. A new self-assembled layer-by-layer glucose biosensor based on chitosan biopolymer entrapped enzyme with nitrogen doped graphene. Bioelectrochem. 2014, 99, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zhong, T.; Qi, W.; Wang, H. The Application of Assembled Inorganic and Organic Hybrid Nanoarchitecture of Prussian Blue/Polymers/Graphene in Glucose Biosensing. J. Inorg. Organomet. Polym. 2015, 25, 275–281. [Google Scholar] [CrossRef]
- Zeng, G.; Xing, Y.; Gao, J.; Wang, Z.; Zhang, X. Unconventional Layer-by-Layer Assembly of Graphene Multilayer Films for Enzyme-Based Glucose and Maltose Biosensing. Langmuir 2010, 26, 15022–15026. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Yu, Y.; Liu, X.; Ni, B.; Zhou, T.; Shi, G. Layer-by-layer self-assembly of functionalized graphene nanoplates for glucose sensing in vivo integrated with on-line microdialysis system. Biosens. Bioelectron. 2012, 32, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Yan, S.; Shi, Y. Direct electrochemical analysis of glucose oxidase on a graphene aerogel/gold nanoparticle hybrid for glucose biosensing. J. Solid State Electrochem. 2015, 19, 307–314. [Google Scholar] [CrossRef]
- Su, X.; Ren, J.; Meng, X.; Rena, X.; Tang, F. A novel platform for enhanced biosensing based on the synergy effects of electrospun polymer nanofibers and graphene oxides. Analyst 2013, 138, 1459–1466. [Google Scholar]
- Liang, B.; Fang, L.; Hu, Y.; Yang, G.; Zhu, Q.; Ye, X. Fabrication and application of flexible graphene silk composite film electrodes decorated with spiky Pt nanospheres. Nanoscale 2014, 6, 4264–4274. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, S.; Li, Y.; Deng, D.; Si, X.; Ding, Y.; He, H.; Luo, L.; Wang, Z. Electrospun graphene decorated MnCo2O4 composite nanofibers for glucose biosensing. Biosens. Bioelectron. 2015, 66, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Liu, Y.; Wang, Y.; Hu, L.; Ma, H.; Wang, G.; Wei, Q. Label-free Electrochemiluminescent Immunosensor for Detection of Prostate Specific Antigen based on Aminated Graphene Quantum Dots and Carboxyl Graphene Quantum Dots. Sci. Rep. 2016, 6, 20511. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Wu, H.; Shang, P.; Zeng, X.; Chi, Y. Immobilizing water-soluble graphene quantum dots with gold nanoparticles for a low potential electrochemiluminescence immunosensor. Nanoscale 2015, 7, 16366–16371. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, W.; Ma, C.; Yang, H.; Ge, S.; Yu, J. Paper-based electrochemiluminescence immunodevice for carcinoembryonic antigen using nanoporous gold-chitosan hybrids and graphene quantum dots functionalized Au@Pt. Sens. Actuators B Chem. 2014, 202, 314–322. [Google Scholar] [CrossRef]
- Yang, H.; Liu, W.; Ma, C.; Zhang, Y.; Wang, X.; Yu, J.; Song, X. Gold-silver nanocomposite-functionalized graphene based electrochemiluminescence immunosensor using graphene quantum dots coated porous PtPd nanochains as labels. Electrochim. Acta 2014, 123, 470–476. [Google Scholar] [CrossRef]
- Zhao, H.; Chang, Y.; Liu, M.; Gao, S.; Yu, H.; Quan, X. A universal immunosensing strategy based on regulation of the interaction between graphene and graphene quantum dots. Chem. Commun. 2013, 49, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Al-Ogaidi, I.; Gou, H.; Aguilar, Z.P.; Guo, S.; Melconian, A.K.; Al-kazaz, A.K.A.; Meng, F.; Wu, N. Detection of the ovarian cancer biomarker CA-125 using chemiluminescence resonance energy transfer to graphene quantum dots. Chem. Commun. 2014, 50, 1344–1346. [Google Scholar] [CrossRef] [PubMed]
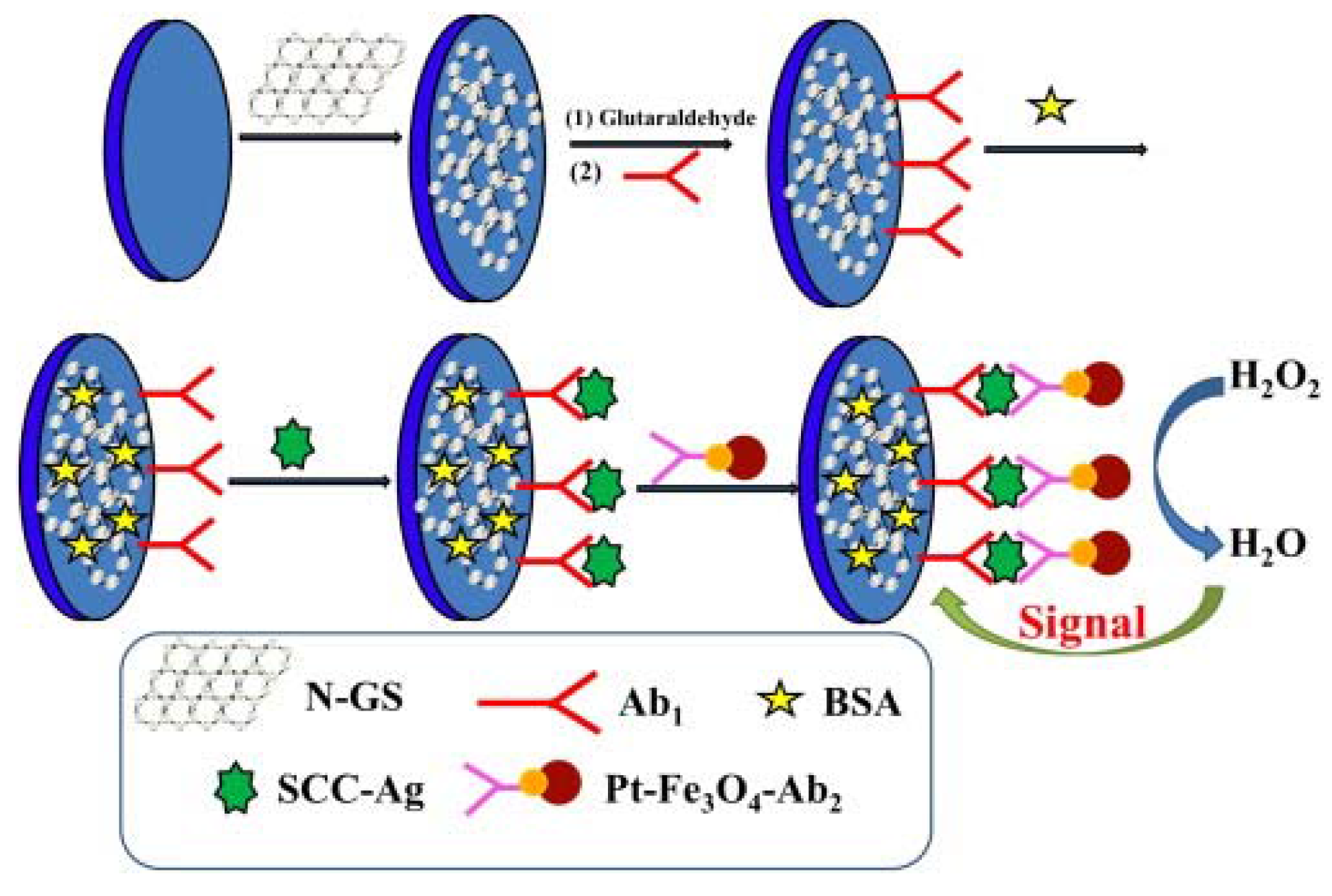

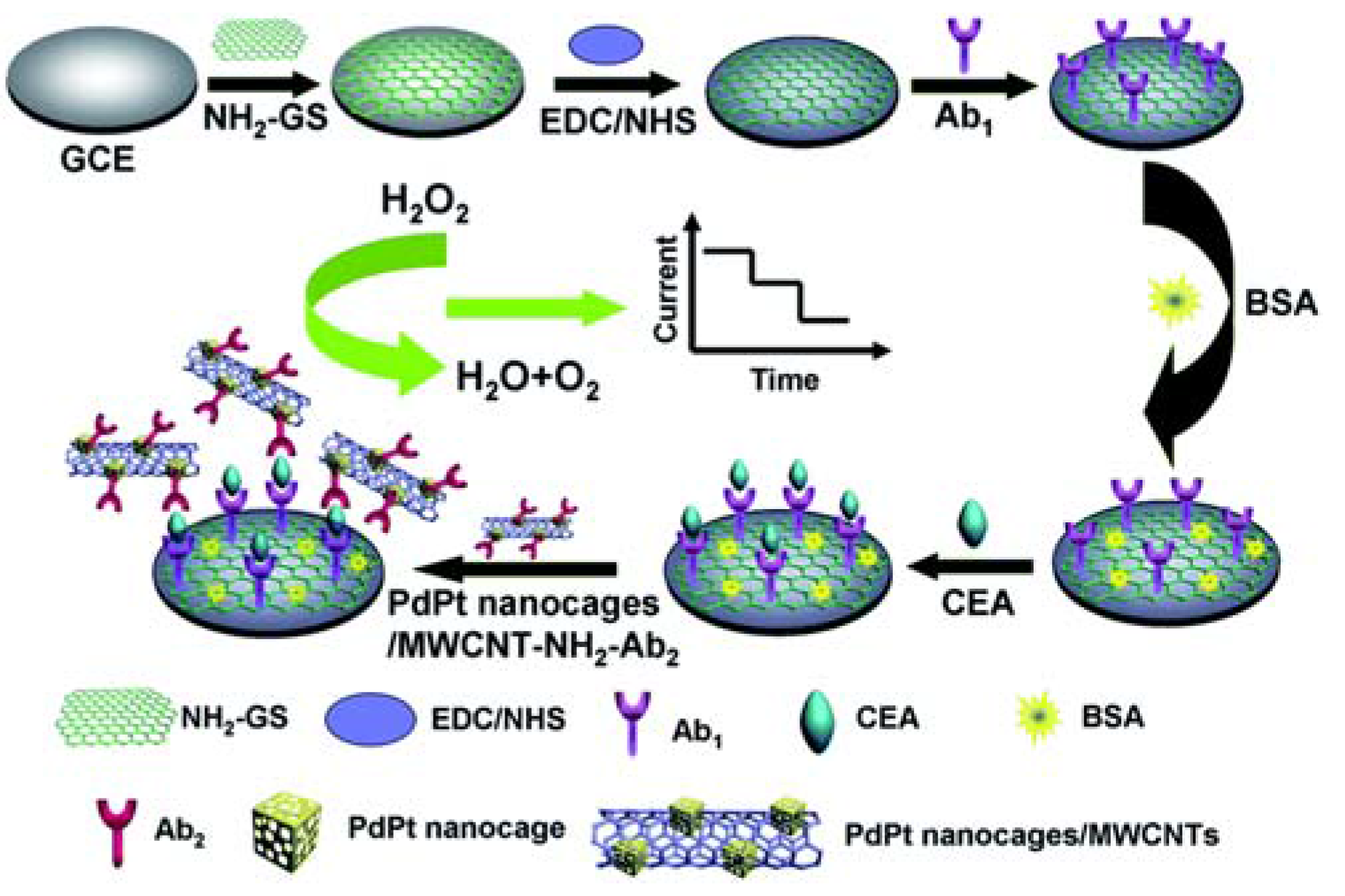
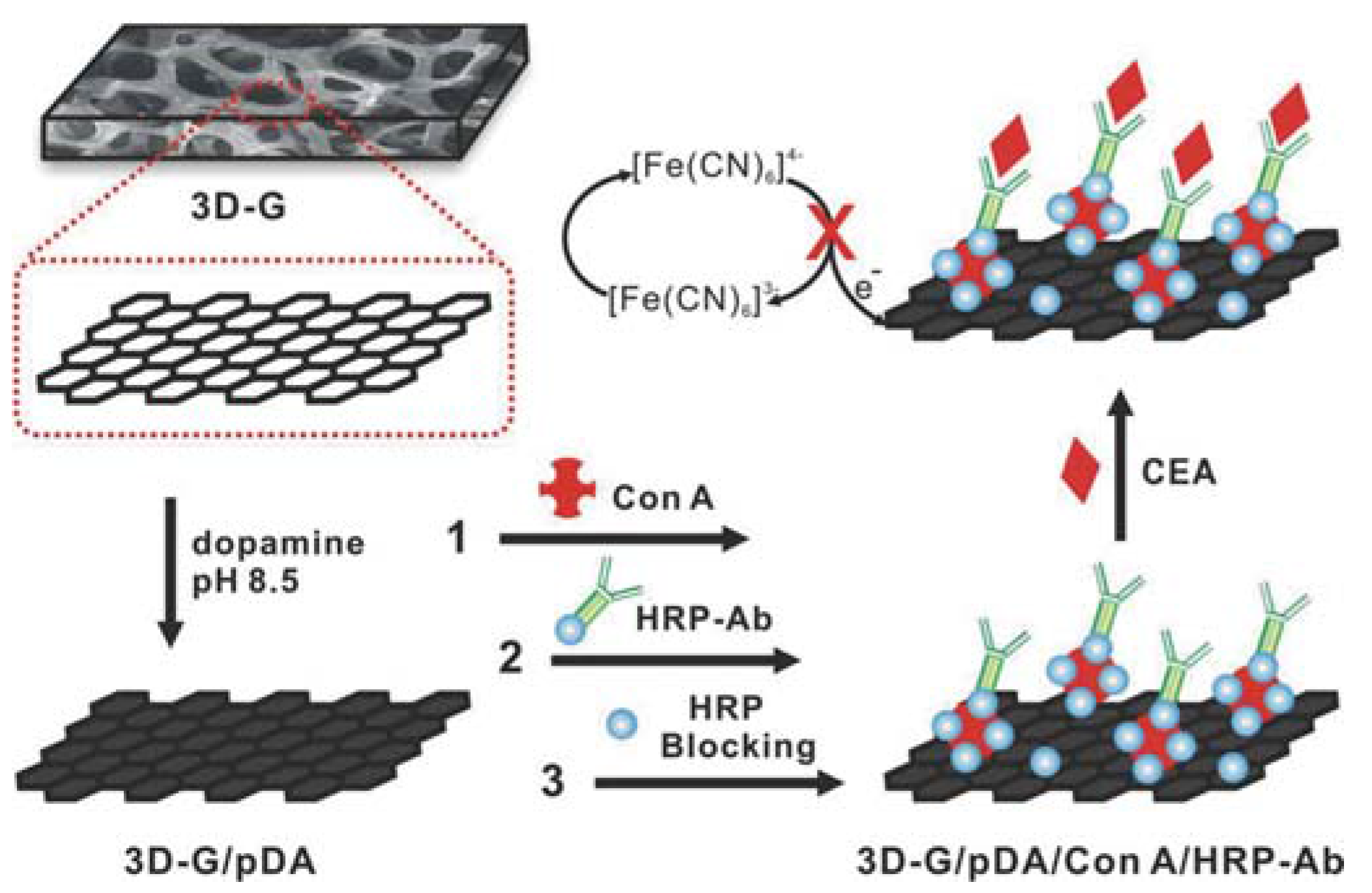
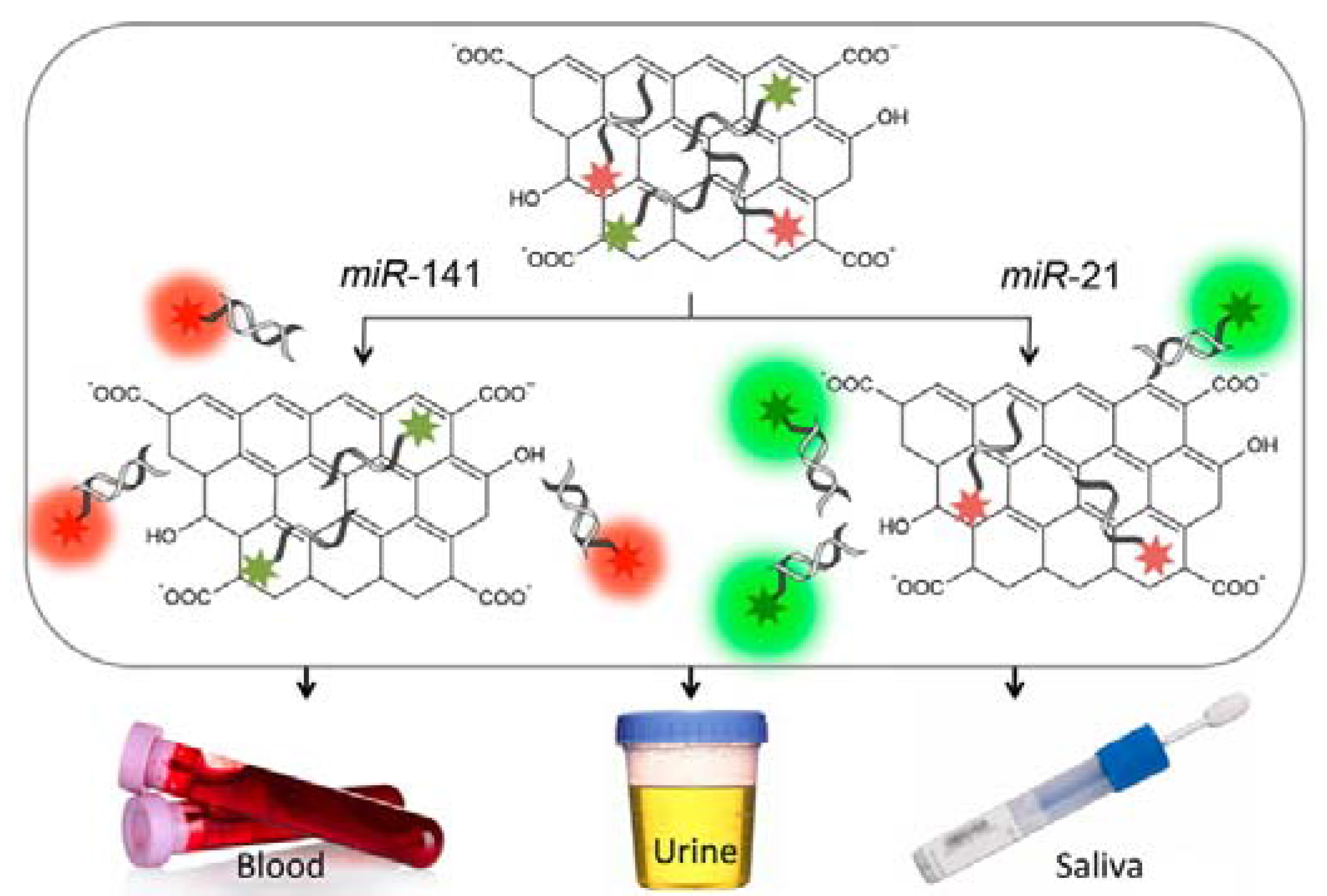

| Sensor Platform//Label | Analyte | Instr. Techn.a | Linearity Range | LOD | References |
|---|---|---|---|---|---|
| Pt/SWCNT/anti-PSA | PSA | CHI | n.a. | 0.25 ng/mL | [33] |
| GCE/starch-MWCNT-Nafion/AuNP/anti-PSA | PSA | CV | 0.01–0.5, 0.5–3.0 ng/mL | 7 pg/mL | [34] |
| PG/SWCNT/anti-PSA//anti-PSA/MWCNT/HRP | PSA | AMP | 0.4–40 ng/mL | 4 pg/mL | [35] |
| GCE/PPy@MWCNT/AuNP/anti-PSA//HRP-anti-PSA | PSA | DPV | 0.002–20 ng/mL | 1 pg/mL | [36] |
| FET/CNT/anti-OPN | OPN | CHI | 1 pg/mL–1 µg/mL | 1 pg/mL | [37] |
| Au/ITO/SWCNT/anti-OPN | OPN | CHI | 1 pg/mL–1 µg/mL | 0.3 pg/mL | [38] |
| Au/SWCNT/NDI/SWCNT/AuNP/TSP-cDNA//AuNP-hemin-G-quadruplex DNAzyme | miRNA-21 | DPV | 10 fM–1 nM | 1.95 fM | [39] |
| GCE/ox-MWCNT/poly(JUG-JUGA)/cDNA | miRNA-141 | SWV | 10 fM–100 pM | 8 fM | [40] |
| GSPE/rGO/ox-MWCNT/cDNA//anti-miRNA/HRP-anti-miRNA | miRNA-141 | SWV | 10 fM–1 nM | 10 fM | [41] |
| Au/SWCNT/protein p19 | miRNA-122a | CHI | 1 aM–10 fM | 1 aM | [42] |
| GCE/CNT/NH2-HP DNA/CP/RCA | miRNA let-7 | DPV | 10–1000 fM | 1.2 fM | [43] |
| GCE/MWCNT/cDNA | miRNA-24 | DPV | 1 pM–1 nM, 1–10 nM | 1 pM | [44] |
| Au/CS-MWCNT-Thi/anti-CEA | CEA | DPV | 1 pg/mL–100 ng/mL | 0.5 pg/mL | [45] |
| CP/PEDOT:PSS-CNT/anti-CEA | CEA | AMP | 2–15 ng/mL | n.a. | [46] |
| GCE/(PB-NP/RGO-MWCNT)5/AuNP/anti-CEA | CEA | DPV | 0.2–1, 1–40 ng/mL | 60 pg/mL | [47] |
| GCE/(PB-NP/MWCNT-PEI-AuNP)5/CS-AuNP/anti-CEA | CEA | AMP | 0.5–2, 2–160 ng/mL | 80 pg/mL | [48] |
| GCE/(PDDA-MWCNT/PSS)2/PDDA-MWCNT/AuNP/anti-CEA | CEA | AMP | 0.1–2, 2–160 ng/mL | 60 pg/mL | [49] |
| Au/l-Cys/anti-CEA//MWCNT/PDDA/HRP/ConA/HRP-anti-CEA | CEA | DPV | 0.05–200 ng/mL | 18 pg/mL | [50] |
| SPCE/MWCNT/anti-CEA//AuNR/GlOx/anti-CEA | CEA | DPV | 0.01–100 ng/mL | 4.2 pg/mL | [51] |
| GCE/RGO-MWCNT/Pt/anti-CEA//Pt/Fe@CD/anti-CEA | CEA | ECL | 0.003–600 ng/mL | 0.8 pg/mL | [52] |
| ITO/MWCNT-PABA/anti-CEA//MWCNT-CR/C60/anti-CEA | CEA | PEC | 0.001–100 ng/mL | 0.1 pg/mL | [53] |
| GCE/GS-NH2/anti-CEA//PdPt/MWCNT-NH2/anti-CEA | CEA | AMP | 0.001–20 ng/mL | 0.2 pg/mL | [54] |
| GCE/MWCNT-ZnO/anti-CA125 | CA125 | DPV | 0.001–1000 U/mL | 0.00113 U/mL | [55] |
| GCE/MWCNT-Nafion/Co(bpy)33+/AuNP/anti-CA125 | CA125 | CV | 1–30, 30–150 U/mL | 0.36 U/mL | [56] |
| QCM/PLL/HA/MWCNT/anti-CA 19-9 | CA 19-9 | QCM | 12.5–270 U/mL | 8.3 U/mL | [29] |
| Ta/MWCNT/AuNP/cDNA | TP53 | EIS | 1 fM–100 nM | 10 aM | [57] |
| GCE/MWCNT-PA6-PTH/anti-AGp53//HRP@anti-AGp53 | AGp53 | DPV | 2–2000 pg/mL | 1 pg/mL | [58] |
| GCE/MWCNT-PA6/PTH/ssDNA1//AuNP-ssDNA2 | K-ras | DPV | 0.1–100 pM | 30 fM | [59] |
| FET/SWCNT-galactose | Galectin-3 | CHI | 156–312.5 ng/mL | n.a. | [60] |
| Au/PoPD/AuNP/aptamer//aptamer/Thi/AuNP/SiO2@MWCNT | MUC 1 | DPV | 1–100 nM | 1 pM | [61] |
| MWCNT-CILE/AuNP/HDT/AuNP/anti-HER2 | HER2 | EIS | 10–110 ng/mL | 7.4 ng/mL | [62] |
| SPCE/SWCNT/anti-HER-3 | HER-3 | EIS | 2–14 fg/mL | 2 fg/mL | [63] |
| PG/SWCNT/anti-MMP-3//anti-MMP-3/polystyrene/HRP | MMP-3 | AMP | 4–300 pg/mL | 4 pg/mL | [64] |
| GCE/CS-AuNP/anti-AFP//HOOC-SWCNT-MnO2 | AFP | LSV | 0.2–100 ng/mL | 40 pg/mL | [65] |
| SPCE/SWCNT/WGA | AFP | EIS | 1–100 pg/mL | 0.1 pg/mL | [66] |
| GCE/AuNP/anti-AFP//Pb2+@Au@MWCNT-Fe3O4/anti-AFP | AFP | AMP | 10 fg/mL–100 ng/mL | 3.33 fg/mL | [67] |
| GRT/SWCNT-CS/anti-anti-MAGE A2 or A11 | anti-MAGE A2 | DPV | 5 fg/mL–50 ng/mL | n.a. | [68] |
| anti-MAGE A11 | |||||
| SPCE/HOOC-Phe-DWCNT/Mix&Go/anti-TNF,anti-IL/ /biotin-anti-TNF, biotin-anti-IL/poly-HRP-strept | TNF-α | AMP | 1–200 pg/mL | 0.85 pg/mL | [69] |
| IL-1β | 0.5–100 pg/mL | 0.38 pg/mL |
| Sensor Platform//Label | Analyte | Instr. Techn.a | Linearity Range | LOD | References |
|---|---|---|---|---|---|
| SPE-MWCNT//anti-Mgb | myoglobin | EIS | 0.1–90 ng/mL | 0.08 ng/mL | [70] |
| GCE/c-MWCNT-CS/Thi/AuNP/anti-netrin 1 | netrin 1 | DPV | 0.09–1800 pg/mL | 0.03 pg/mL | [71] |
| SPCE/MP-anti-MPO/SWCNT | MPO | CHA | n.a. | 55 ng/mL | [72] |
| GCE/DMF-MWCNT-EMIMBF4/CS-CeO2NP/anti-MPO | MPO | CV | 5–300 ng/mL | 0.2 ng/mL | [73] |
| ITO/PoPD-MWCNT-EMIMBr/AuNP/anti-MPO | MPO | CV | 0.2–23 and 23–300 ng/mL | 0.05 ng/mL | [74] |
| GCE/Nafion-ZnS-c-MWCNT-ChOx | cholesterol | CV | 1.3–11.6 mM | 0.26 mM | [75] |
| FET/CNT/loop peptide | ab-AQP4 | CHI | n.a. | 1 pg/mL | [76] |
| FET/SWCNT/anti-ag-Lyme | ag-Lyme | CHI | 1–3000 ng/mL | 1 ng/mL | [77] |
| SPCE/PTCA-MWCNT/AAT aptamer//ALP-anti-AAT/AgNP | AAT | DPV | 0.05–20 pM | 0.01 pM | [78] |
| FET/SWCNT/Au/anti-amyloid-β | amyloid-β | CHI | 1 pg/mL–1 ng/mL | 1 pg/mL | [79] |
| GCE/c-MWCNT/ZnO2-NP/AChE-ChlO | choline | CV | 0.05–200 µM | 0.01 µM | [80] |
| GCE/PAMAM-AuNP-MWCNT/anti-SirT1//SiO2@Au NP/HRP-anti-SirT1 | SirT1 | DPV | 20 pg/mL–500 ng/mL | 12.5 pg/mL | [81] |
| Au/c-MWCNT-PS/CFFCP1 peptide//anti-ACPA-HRP | ACPA | AMP | n.a. | n.a. | [82] |
| Au/SWCNT/CSPR peptide//anti-ACPA | ACPA | QCM | n.a. | n.a. | [83] |
| GCE/MWCNT/PB/CS/ anti-Tcd B//GO/HRP-anti-Tcd B/HRP | Tcd B | DPV | 0.003–320 ng/mL | 0.7 pg/mL | [84] |
| Cu/MWCNTs-MIP | dopamine | PT | 1–10,000 nM | 1.0 nM | [85] |
| CCE/MWCNTs-MIP | dopamine | DPASV | 0.75–34 ng/mL | 0.21 ng/mL | [86] |
| Au/ssDNA-SWCNT/PABA/Nafion | dopamine | DPV | n.a. | 40 pM | [87] |
| GCE/c-MWCNT/SiO2 | dopamine | DPV | 0.5–6 µM | 14 nM | [88] |
| GCE/ox-MWCNT/SiO2 | uric acid | DPV | 0.5–10 µM | 0.068 µM | [88] |
| GCE/SWCNT/Thi/UOx | uric acid | AMP | 2 µM–2 mM | 0.5 µM | [89] |
| GCE/PPy/c-MWCNT/GlOx | glucose | AMP | 4–50 mM | 0.2 mM | [90] |
| Au/SWCNT/PPy/GlOx | glucose | AMP | 0.56–100 mM | 0.05 mM | [91] |
| Sensor Platform//Label | Analyte | Instr. Techn.a | Linearity Range | LOD | References |
|---|---|---|---|---|---|
| GO/UCNP/ssDNA | PCA3 | FS | n.a. | 0.5 pM | [96] |
| FET/(PDDA+PSS)2(PDDA+GR)5/anti-PSA | PSA | CHI | 4 fg/mL–4 µg/mL | 0.11 fM | [97] |
| FET/(PDDA+PSS)2(PDDA+GR)5/anti-PSA | PSA | CHI | 0.4 fg/mL–4 µg/mL | 11 aM | [98] |
| FET/GR/anti-ANXA2, or anti-ENO1, or anti-VEGF | ANXA2, ENO1, VEGF | CHI | 1 pg/mL–1 µg/mL | 0.1 pg/mL | [99] |
| FET/(PDDA+GR)2(PDDA+TiO2) (PDDA+GR)2/anti-ANXA2, or anti-ENO1, or anti-VEGF | ANXA2, ENO1, VEGF | CHI | 100 fg/mL–1 µg/mL | 100 fg/mL | [100] |
| GF/pDA/ConA/HRP-anti-CEA | CEA | DPV | 0.1–750 ng/mL | 90 pg/mL | [25] |
| GCE/GR/SA-CS//HP-DNA-AuNR-HRP | CEA | DPV | 5 pg/mL–50 ng/mL | 1.5 pg/mL | [26] |
| PWE/AuNPs/PANI/anti-CEA,anti-AFP//rGO/MB,Fc-COOH/anti-CEA,anti-AFP | CEA, AFP | DPV | 1 pg/mL–100 ng/mL | 0.5 pg/mL, 0.8 pg/mL | [101] |
| GCE/CS-AuNP/anti-CEA,anti-AFP//anti-CEA-TB-CGS/anti-AFP-PB-CGS | CEA, AFP | DPV | 0.5–60 ng/mL | 0.1 ng/Ml, 0.05 ng/mL | [102] |
| GCE/IL-rGO/anti-CEA,anti-AFP//anti-CEA-PtNP-Cd2+,anti-AFP-PtNP-Cu2+ | CEA, AFP | DPV | 0.05–200 ng/mL | 0.002 ng/mL, 0.05 ng/mL | [103] |
| GCE/IL-rGO/PSS/anti-CEA,anti-AFP,anti-PSA//anti-CEA-Thi-CAuNP,anti-AFP-Cd2+-CAuNP,anti-PSA-DAP-CAuNP | CEA, AFP, PSA | SWV | 0.01–100 ng/mL | 2.7 pg/mL, 3.1 pg/mL, 4.8 pg/mL | [104] |
| GCE/GR-AuNP/anti-CEA,AFP,CA125,PSA//SA/biotin-dsDNA/SA/biotin-anti-CEA,AFP,CA125,PSA/Au/SiO2-Fe3O4 | CEA, AFP, CA125 PSA | DPV | 0.2–600, 0.2–800, 0.2–1000, 0.2–800 pg/mL | 48 fg/mL, 62 fg/mL, 77 fg/mL, 60 fg/mL | [105] |
| GF-nTiO2/anti-ErbB2 | ErbB2 | DPV | 1.0 fM–0.1 µM | n.a. | [106] |
| GCE/rGO-CS/aptamer/MB | HER2 | DPV | 0.5–2, 2–75 ng/mL | 0.21 ng/mL | [24] |
| FET/GR/PtNP/scFv-anti-HER3 | HER3 | CHI | 300 fg/mL–300 ng/mL | 300 fg/mL | [107] |
| Au/Cys/GO/Py-COOH/anti-CA 15-3//anti-CA 15-3/MWCNT/Ferritin | CA 15-3 | DPV | 0.05–100 U/mL | 0.009 U/mL | [108] |
| ITO/APES/pNiPAM/anti-MSLN//scFv-MSLN/GO/CdSe QDs | MSLN | SWASV | n.a. | 0.5 pg/mL | [109] |
| GCE/rGO-TEPA/anti-CA72-4//anti-CA72-4/ PtPd-Fe3O4 | CA72-4 | EIS | 0.001–10 U/mL | 0.0003 U/mL | [110] |
| SPCE/AuNP-GR/CS/aptamer//aptamer-Ag@Pt-Gr | TNF-α | DPV | 5–70 pg/mL | 1.64 pg/mL | [111] |
| GCE/PEI-RGO/AuNP/cDNA1//cDNA2/TiP-Cd2+/Ru(NH3)63+ | miRNA-21 | SWV | 1 aM–10 pM | 0.76 aM | [112] |
| GO/FAM-ssDNA | miRNA-126 | FS | 0.02–100 pM | 3.0 fM | [113] |
| FET/rGO/AuNP/PNA | miRNA let7b | CHI | 10 fM–100 pM | 10 fM | [114] |
| GO/FAM-anti-miR-21/Cy5-anti-miR-141 ssDNA | miRNA-21 miRNA-141 | FS | n.a. | 2.0 nM, 1.2 nM | [115] |
| GO-RuOMO-aptamers | thrombin | FS | 3.7–613 nM | 0.76 nM | [116] |
| GO/ FITC-HAKRRLIF | cyclin A2 | FS | n.a. | 0.5 nM | [117] |
| GCE/CCG/TCPP/hexapeptide | cyclin A2 | EIS | 0.5–10 pM | 0.32 pM | [118] |
| GCE/SRGO | 8OHdG | DPV | 2 nM–20 µM | 1 nM | [119] |
| GCE/GR/ss-DNA | 8OHdG | CV | 0.0056–1.155, 1.155–11.655, 11.655–36.155 µM | 0.875 nM | [120] |
| SPCE/RGO-AuNP/LDH | l-lactate | AMP | 0.01–5 mM | 0.13 µM | [121] |
| Au/rGO/FA | FAP | DPV | 1–200 pM | 1 pM | [122] |
| GO/ Pep-FITC | MMP-2 | FS | 10–150 ng/mL | 2.5 ng/mL | [123] |
| ITO/rGO-ZrO2/APTES/anti-CYFRA-21-1 | CYFRA-21-1 | DPV | 2–22 ng/mL | 0.122 ng/mL | [124] |
| GCE/N-GS/CS/anti-SCCA//anti-SCCA/Pt-Fe3O4 | SCCA | EIS | 0.05–18 ng/mL | 15.3 pg/mL | [22] |
| GCE/N-GS/anti-SCCA//anti-SCCA/Pd-Au/C | SCCA | EIS | 0.005–2 ng/mL | 1.7 pg/mL | [125] |
| GCE/GO/anti-hTPA//anti-hTPA/Pd-Pt NP | hTPA | EIS | 0.005–15 ng/mL | 1.2 pg/mL | [126] |
| GCE/rGO-TEPA/anti-TSGF//anti-TSGF/Ag@CeO2 | TSGF | CV | 0.5–100 pg/mL | 0.2 pg/mL | [127] |
| GCE/rGO-TEPA/AuPdPt NP/anti-NMP22 | NMP22 | DPV | 0.04–20 U/mL | 0.01 U/mL | [128] |
| FET-poly-SiNW/MGLA-anti-APOA2 | APOA2 | CHI | 19.5 pg/mL–1.95 µg/mL | 6.7 pg/mL | [129] |
| Sensor Platform//Label | Analyte | Instr. Techn.a | Linearity Range | LOD | References |
|---|---|---|---|---|---|
| MEMS/rGO/anti-Aβ | Aβ40 | CHI | 100 fg/mL–100 pg/mL | 100 fg/mL | [130] |
| GO/UCNP/ssDNA | BACE-1 | FS | n.a. | 0.5 pM | [96] |
| GCE/GO-Ph-AuNP/anti-cTnI//FcGO/anti-cTnI | cTnI | SWV | 0.05–3 ng/mL | 0.05 ng/mL | [131] |
| GCE/PLL/AuNP/anti-T3//Fe3O4@GO/Ru(bpy)32+/anti-T3 | T3 | ECL | 0.1 pg/mL–10 ng/mL | 0.03 pg/mL | [132] |
| GCE/rGO-Au/anti-PCT//SWCNH/HPtC/Thi/anti-PCT/HRP | PCT | CV | 1.0 pg/mL–2.0 ng/mL | 0.43 pg/mL | [133] |
| GO | estriol | FS | 1.3–10 nM | 1.3 nM | [134] |
| SPCE/GONR/DAAO | d-Tyr | DPV | 0.25–1.0 mM | 60 µM | [135] |
| ITO/rGO-AgNF/MPA/anti-insulin | insulin | EIS | 1–1000 ng/mL | 70 pg/mL | [23] |
| GCE/GONR-Nafion | dopamine | DPV | 0.1–8.5 µM | 24 nM | [136] |
| GCE/GONR-Nafion | uric acid | DPV | 0.1–8.5 µM | 98 nM | [136] |
| ITO/GF/ZnO NWA | dopamine | DPV | 0–40 µM | 1 nM | [137] |
| ITO/GF/ZnO NWA | uric acid | DPV | 0–40 µM | 1 nM | [137] |
| GCE/3D-RGO | dopamine | DPV | 5 µM–1 mM | 0.17 µM | [138] |
| GCE/(PDDA)1(PSS-RGO/PAMAM-AuNPs)20 | dopamine | DPV | 1–60 µM | 0.02 µM | [139] |
| GCE/(rGO)3(Nafion) | dopamine | LSV | 0.5–30 µM | 0.2 µM | [140] |
| GCE/rGO-PhNHOH/LDH | l-lactate | AMP | 0–90 µM | 2.5 µM | [141] |
| GCE/rGO-CS/GlOx | Glucose | CV | 2–22 mM | 20 µM | [142] |
| Au/[CS(NGR+GlOx)/ PSS/CS(NGR+GlOx] | Glucose | CHA | 0.2–1.8 mM | 64 µM | [143] |
| GCE/(rGO/PDDA-PB/GlOx/PDDA-PB)3 | Glucose | AMP | 0.1–6.5 mM | 6 µM | [144] |
| GCE/(PEI/PAA-rGO)3(PEI/GlOx)5 | Glucose | AMP | 0 -10 mM | 0.168 mM | [145] |
| GCE/(IL-RGO/SA-RGO)5/IL-RGO/GlOx | Glucose | AMP | 10–500 µM | 3.33 µM | [146] |
| GCE/GA-AuNP/GlOx | Glucose | AMP | 50–450 µM | 0.597 µM | [147] |
| Pt/GO/GlOx/CS/PVA fiber | Glucose | CV | 5 µM–3.5 mM | 5 µM | [148] |
| RGO/silk fiber/PtNP/GlOx | Glucose | AMP | 10 µM–10 mM | 1 µM | [149] |
| GCE/rGO/MnCo2O4 fiber/GlOx | Glucose | AMP | 0.005–800 µM | 1 nM | [150] |
| Sensor Platform//Label | Analyte | Instr. Techn.a | Linearity Range | LOD | References |
|---|---|---|---|---|---|
| GCE/Au/Ag-rGO/GR-QD/GO-QD/anti-PSA | PSA | ECL | 0.001–10 ng/mL | 0.29 pg/mL | [151] |
| GCE/rGO-QD/AuNP/anti-CEA | CEA | ECL | 0.02–80 ng/mL | 10 pg/mL | [152] |
| PWE/Au/CS/anti-CEA/ /anti-CEA/GO-QD/Au@PtNP | CEA | ECL | 0.001–10 ng/mL | 0.6 pg/mL | [153] |
| GCE/PVP-rGO/AgNP/AuNP/anti-CA199//anti-CA199/GO-QD/PtPdNPs | CA199 | ECL | 0.002–70 U/mL | 0.96 mU/mL | [154] |
| rGO/GO-QD/anti-IgG | IgG | FS | n.a. | 10 ng/mL | [155] |
| GO-QD/anti-CA125/ /anti-CA125-HRP | CA125 | CL | 0.1–600 U/mL | 0.05 U/mL | [156] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasinszki, T.; Krebsz, M.; Tung, T.T.; Losic, D. Carbon Nanomaterial Based Biosensors for Non-Invasive Detection of Cancer and Disease Biomarkers for Clinical Diagnosis. Sensors 2017, 17, 1919. https://doi.org/10.3390/s17081919
Pasinszki T, Krebsz M, Tung TT, Losic D. Carbon Nanomaterial Based Biosensors for Non-Invasive Detection of Cancer and Disease Biomarkers for Clinical Diagnosis. Sensors. 2017; 17(8):1919. https://doi.org/10.3390/s17081919
Chicago/Turabian StylePasinszki, Tibor, Melinda Krebsz, Thanh Tran Tung, and Dusan Losic. 2017. "Carbon Nanomaterial Based Biosensors for Non-Invasive Detection of Cancer and Disease Biomarkers for Clinical Diagnosis" Sensors 17, no. 8: 1919. https://doi.org/10.3390/s17081919
APA StylePasinszki, T., Krebsz, M., Tung, T. T., & Losic, D. (2017). Carbon Nanomaterial Based Biosensors for Non-Invasive Detection of Cancer and Disease Biomarkers for Clinical Diagnosis. Sensors, 17(8), 1919. https://doi.org/10.3390/s17081919






