Glucose Sensing for Diabetes Monitoring: Recent Developments
Abstract
1. Introduction
2. Glucose Monitoring
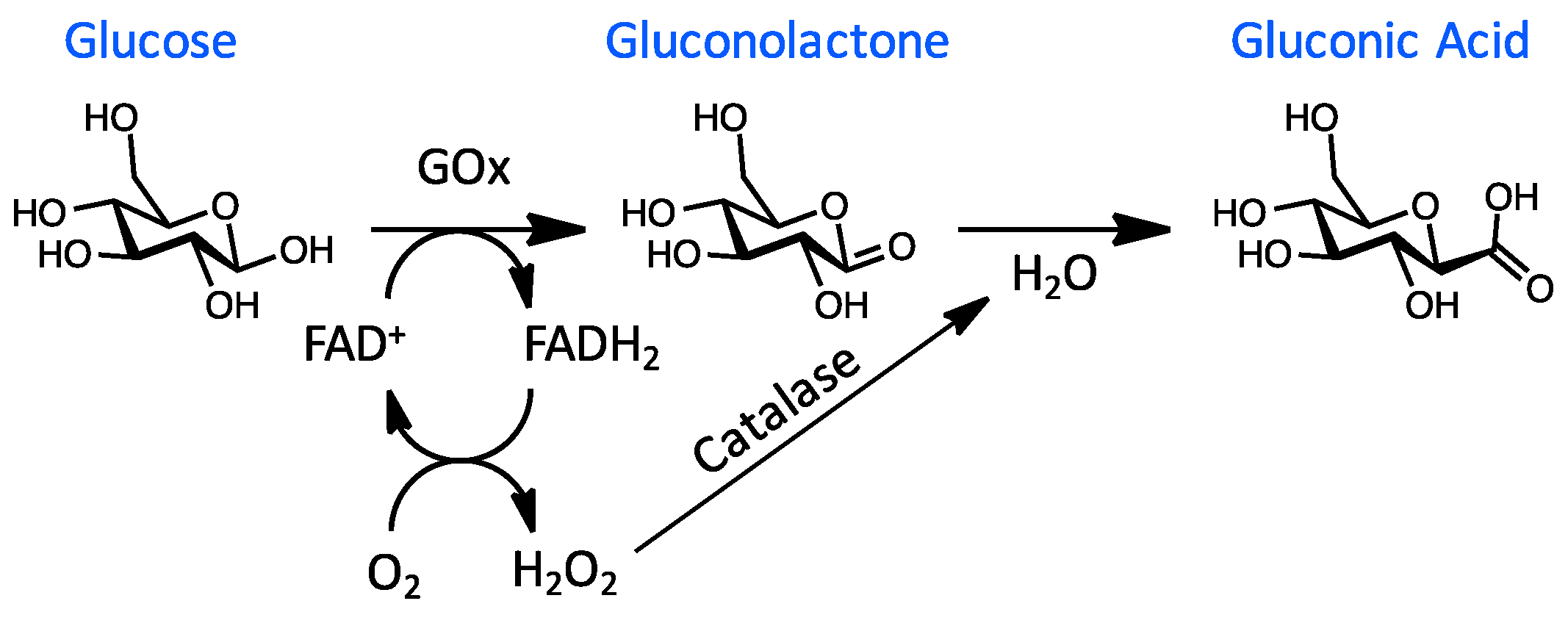
2.1. Glucose Monitoring Methods in Blood
2.2. Monitoring Glucose in Alternative Physiological Fluids
2.2.1. Interstitial Fluid
2.2.2. Urine
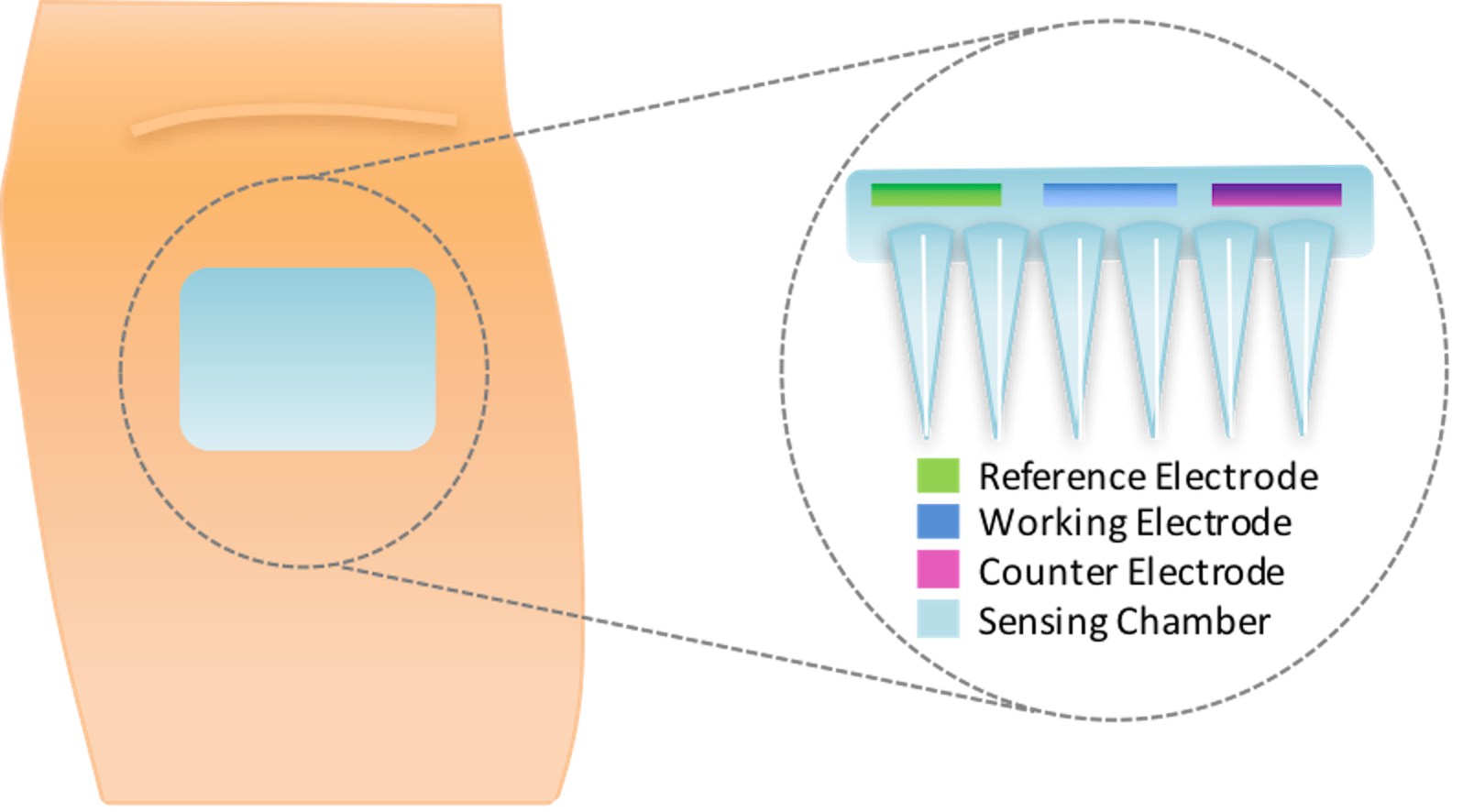
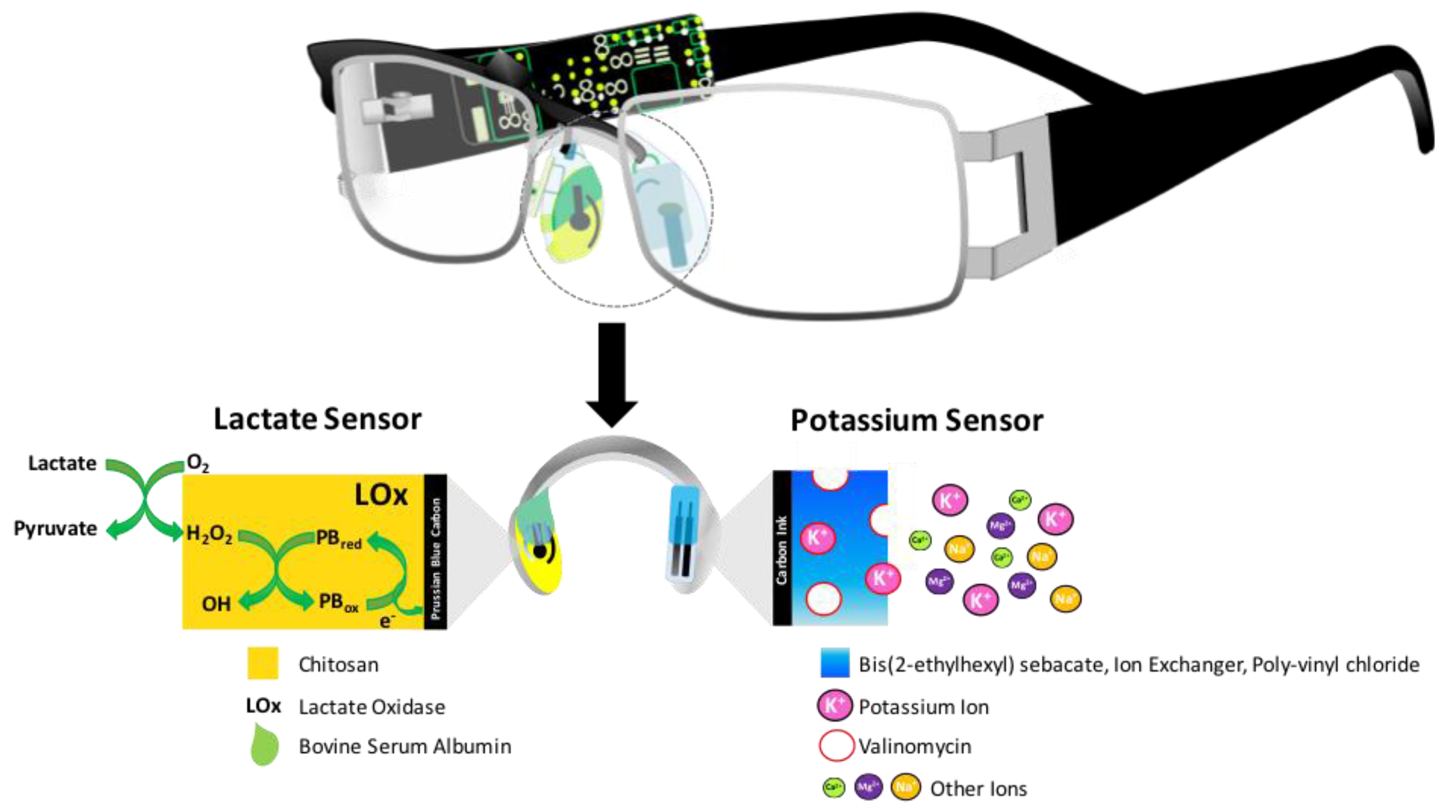
2.2.3. Sweat
2.2.4. Breath Analysis
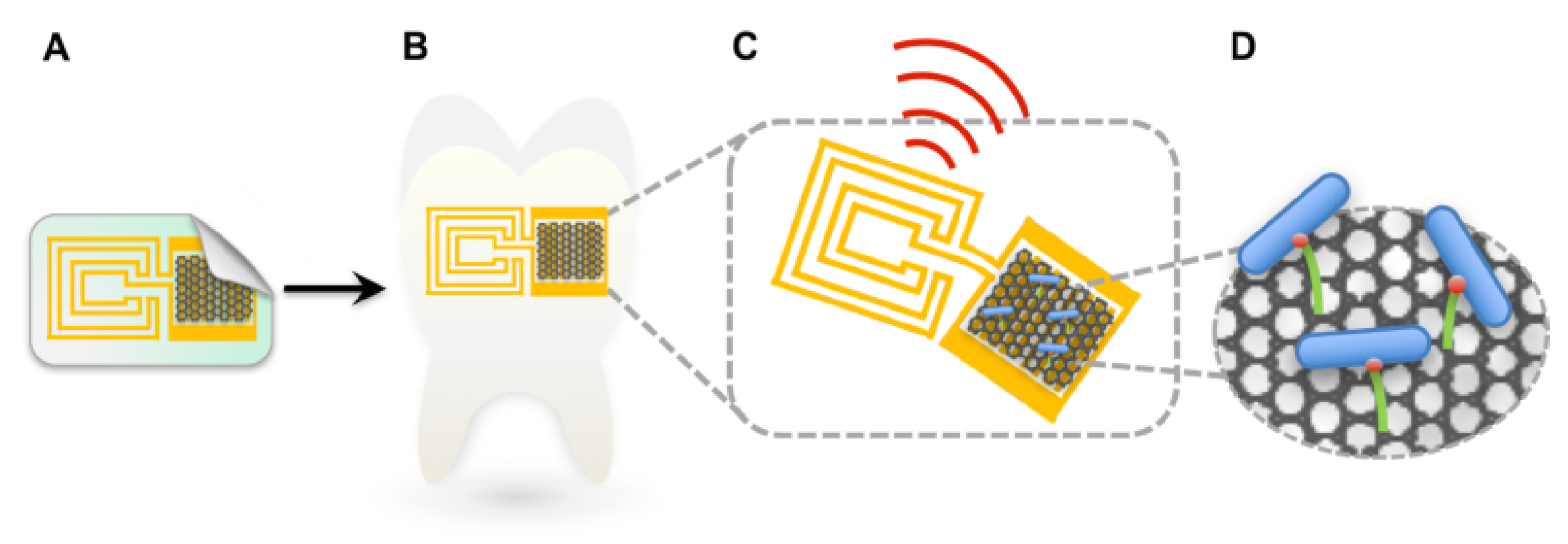
2.2.5. Saliva
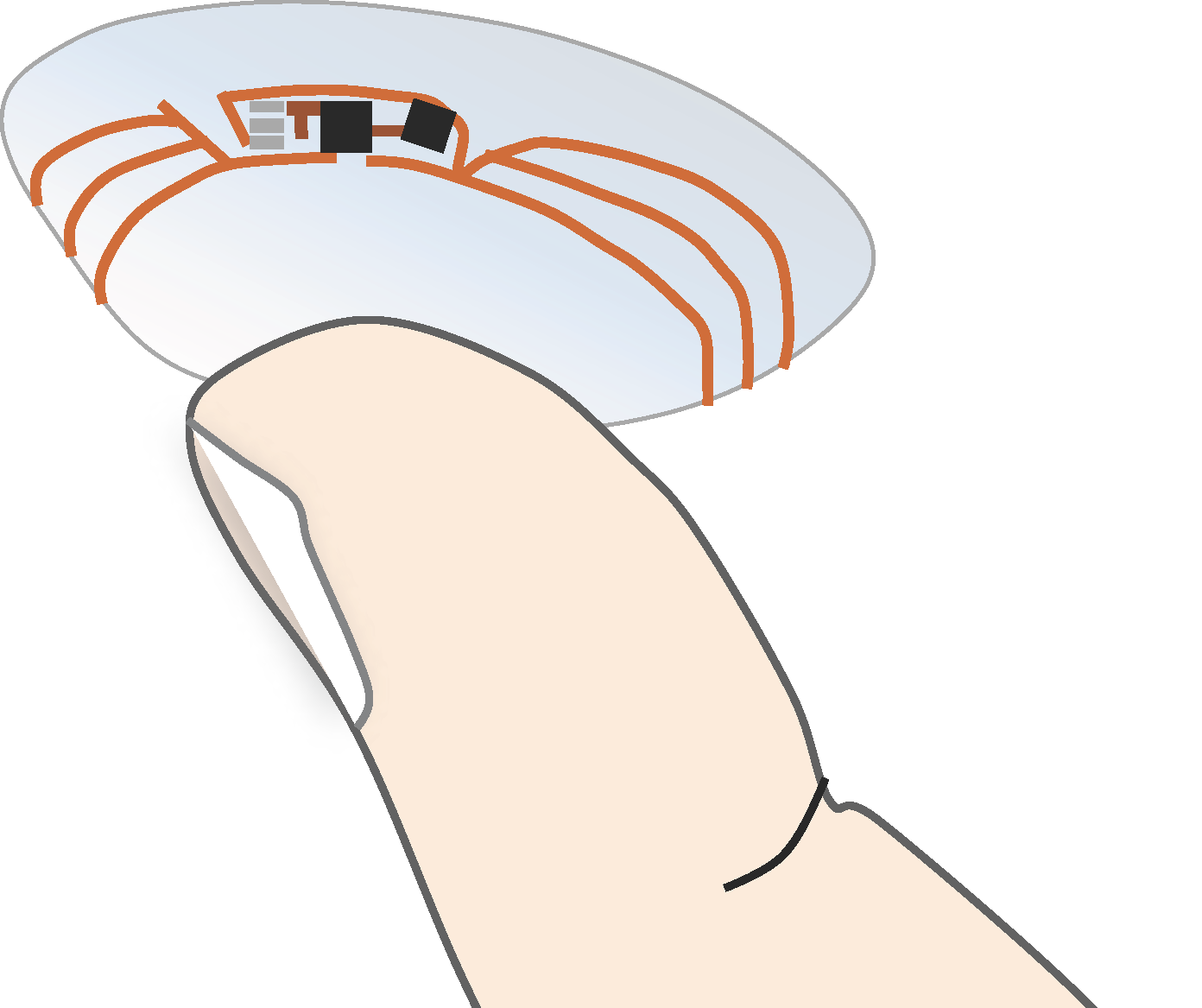
2.2.6. Ocular Fluid
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Food and Drug Administration. Glucowatch for diabetes. WHO Drug Inf. 2002, 16, 1. [Google Scholar]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2004, 27, 5–10. [Google Scholar]
- Sabokdast, M.; Habibi-Rezaei, M.; Moosavi-Movahedi, A.A.; Ferdousi, M.; Azimzadeh-Irani, E.; Poursasan, N. Protection by beta-hydroxybutyric acid against insulin glycation, lipid peroxidation and microglial cell apoptosis. DARU J. Pharm. Sci. 2015, 23. [Google Scholar] [CrossRef] [PubMed]
- Coster, S.; Gulliford, M.C.; Seed, P.T.; Powrie, J.K.; Swaminathan, R. Monitoring blood glucose control in diabetes mellitus: A systematic review. Health Technol. Assess 2000, 4, 1–93. [Google Scholar]
- Torpy, J.M.; Lynm, C.; Glass, R.M. Diabetes. J. Am. Med. Assoc. 2009, 301, 1620. Available online: http://jama.jamanetwork.com/ (accessed on 10 July 2014). [CrossRef] [PubMed]
- Center for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States. Available online: http://www.cdc.gov/diabetes/data/statistics/2014StatisticsReport.html (accessed on 18 August 2016).
- Bratlie, K.M.; York, R.L.; Invernale, M.A.; Langer, R.; Anderson, D.G. Materials for diabetes therapeutics. Adv. Healthc. Mater. 2012, 1, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Nwaneri, C. Diabetes mellitus: A complete ancient and modern historical perspective. WebmedCentral Diabetes 2015, 8, WMC004831. Available online: http://www.webmedcentral.com/article_view/4831 (accessed on 17 February 2015).
- Toghill, K.E.; Compton, R.G. Electrochemical non-enzymatic glucose sensors: A perspective and an evaluation. Int. J. Electrochem. Sci. 2010, 5, 1246–1301. [Google Scholar]
- Yadava, J.; Rani, A.; Singh, V.; Murari, B.M. Prospects and limitations of non-invasive blood glucose monitoring using near-infrared spectroscopy. Biomed. Signal Process. Control 2015, 18, 214–227. [Google Scholar] [CrossRef]
- Pandey, R.; Paidi, S.K.; Valdez, T.A.; Zhang, C.; Spegazzini, N.; Dasari, R.R.; Barman, I. Noninvasive monitoring of blood glucose with raman spectroscopy. Acc. Chem. Res. 2017, 50, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Spegazzini, N.; Barman, I.; Dingari, N.C.; Pandey, R.; Soares, J.S.; Ozaki, Y.; Dasari, R.R. Spectroscopic approach for dynamic bioanalyte tracking with minimal concentration information. Sci. Rep. 2014, 4, 7013. [Google Scholar] [CrossRef] [PubMed]
- Yoo, E.H.; Lee, S.Y. Glucose biosensors: An overview of use in clinical practice. Sensors 2010, 10, 4558–4576. [Google Scholar] [CrossRef] [PubMed]
- Corrie, S.J.; Coffey, J.W.; Islam, J.; Markey, K.A.; Kendall, M.A.F. Blood, sweat, and tears: Developing clinically relevant protein biosensors for integrated body fluid analysis. Analyst 2015, 140, 4350–4364. [Google Scholar] [CrossRef] [PubMed]
- Bandodkar, A.J.; Wang, J. Non-invasive wearable electrochemical sensors: A review. Trends Biotechnol. 2014, 32, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Coyle, S.; Curto, V.F.; Benito-Lopez, F.; Florea, L.; Diamond, D. Wearable bio and chemical sensors. In Wearable Sensors; Elsevier Inc.: Amsterdam, The Netherlands, 2014; pp. 65–83. [Google Scholar]
- Matzeu, G.; Florea, L.; Diamond, D. Advances in wearable chemical sensor design for monitoring biological fluids. Sens. Actuators B Chem. 2015, 21, 403–418. [Google Scholar] [CrossRef]
- Makaram, P.; Owens, D.; Aceros, J. Trends in Nanomaterial-Based Non-Invasive Diabetes Sensing Technologies. Diagnostics 2014, 4, 27–46. [Google Scholar] [CrossRef] [PubMed]
- Do Amaral, C.E.F.; Wolf, B. Current development in non-invasive glucose monitoring. Med. Eng. Phys. 2008, 30, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. Ophthalmic glucose monitoring using disposable contact lenses—A review. J. Fluoresc. 2004, 14, 617–633. [Google Scholar] [CrossRef] [PubMed]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. Fluorescence sensors for monosaccharides based on the 6-methylquinolinium nucleus and boronic acid moiety: Potential application to ophthalmic diagnostics. Talanta 2005, 65, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Clark, L.C., Jr.; Lyons, C. Electrode systems for continuous monitoring in cardiovascular surgery. Ann. N. Y. Acad. Sci. 1962, 102, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Price, C.P. Point-of-care testing in diabetes mellitus. Clin. Chem. Lab. Med. 2003, 41, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- D’Costa, E.J.; Higgins, I.J.; Turner, A.P. Quinoprotein glucose dehydrogenase and its application in an amperometric glucose sensor. Biosensors 1986, 2, 71–87. [Google Scholar] [CrossRef]
- Heller, A.; Feldman, B. Electrochemical glucose sensors and their applications in diabetes management. Chem. Rev. 2008, 108, 2482–2505. [Google Scholar] [CrossRef] [PubMed]
- Bankar, S.B.; Bule, M.V.; Singhal, R.S.; Ananthanarayan, L. Glucose oxidase - an overview. Biotechnol. Adv. 2009, 27, 489–501. [Google Scholar] [CrossRef] [PubMed]
- Guilbault, G.G.; Lubrano, G.J. An enzyme electrode for the amperometric determination of glucose. Anal. Chim. Acta 1973, 64, 439–455. [Google Scholar] [CrossRef]
- Magers, T.A.; Tabb, D.L. A Composition and Test Device Based on a Benzidine-Type Indicator for Detecting the Presence of a Constituent in a Test Sample. European Patent Office, Espacenet EP19800106766 19801104, 23 November 1983. [Google Scholar]
- Wang, J. Glucose biosensors: 40 Years of advances and challenges. Electroanalysis 2001, 13, 983–988. [Google Scholar] [CrossRef]
- Clark, L.C., Jr. Monitor and control of blood and tissue oxygen tensions. Trans. Am. Soc. Artif. Intern. Organs 1956, 2, 41–48. [Google Scholar]
- Hilditch, P.; Green, M. Disposable electrochemical biosensors. Analyst 1991, 116, 1217–1220. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.; Holman, R.; Brown, E.; Streemson, J.; Watson, A.; Hughes, S. Pen-sized digital 30-s blood glucose meter. Lancet 1987, 1, 778–779. [Google Scholar] [CrossRef]
- Fang, H.; Kaur, G.; Wang, B. Progress in boronic acid-based fluorescent glucose sensors. J. Fluoresc. 2004, 14, 481–489. [Google Scholar] [CrossRef]
- Pickup, J.C.; Hussain, F.; Evans, N.D.; Rolinski, O.J.; Birch, D.J.S. Fluorescence-based glucose sensors. Biosens. Bioelectron. 2005, 20, 2555–2565. [Google Scholar] [CrossRef] [PubMed]
- Wang, J. Electrochemical glucose biosensors. Chem. Rev. 2008, 108, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. Boronic acid fluorescent sensors for monosaccharide signalling based on the 6-methoxyquinolinium heterocyclic nucleus: Progress toward noninvasive and continuous glucose monitoring. Bioorgan. Med. Chem. 2005, 13, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. Ophthalmic glucose sensing: A novel monosaccharide sensing disposable and colourless contact lens. Analyst 2004, 129, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. Noninvasive continuous monitoring of physiological glucose using a monosaccharide-sensing contact lens. Anal. Chem. 2004, 76, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. A glucose sensing contact lens: A non-invasive technique for continuous physiological glucose monitoring. J. Fluoresc. 2003, 13, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Bondi, M.C.; Wolfbeis, O.S. Oxygen optrode for use in a fiber-optic glucose biosensor. Anal. Chem. 1990, 62, 2377–2380. [Google Scholar] [CrossRef] [PubMed]
- Larin, K.V.; Motamedi, M.; Ashitkov, T.V.; Esenaliev, R.O. Specificity of noninvasive blood glucose sensing using optical coherence tomography technique: A pilot study. Phys. Med. Biol. 2003, 48, 1371–1390. [Google Scholar] [CrossRef]
- Nathan, D.M. The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study at 30 years: Overview. Diabetes Care 2014, 37, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Clarke, A.; O’Kelly, S. Glucose Monitoring Systems. Available online: http://www.diabetes.ie/living-with-diabetes/educational-articles/diabetes-and-research-articles/continuous-glucose-monitoring-cgm-systems/ (accessed on 8 October 2014).
- Badugu, R.; Lakowicz J, R.; Geddes C, D. Wavelength-ratiometric and colorimetric robes for glucose determination. Dyes Pigments 2006, 68, 159–163. [Google Scholar] [CrossRef]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. A glucose-sensing contact lens: From bench top to patient. Curr. Opin. Biotechnol. 2005, 16, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.; Turner, A.P.F. Home blood glucose biosensors: A commerical perspective. Biosens. Bioelectron. 2005, 20, 2435–2453. [Google Scholar] [CrossRef] [PubMed]
- Boiroux, D.; Batora, V.; Hagdrup, M.; Tarnik, M.; Murgas, J.; Schmidt, S.; Norgaard, K.; Poulsen, N.K.; Madsen, H.; Jorgensen, J.B. Comparison of prediction models for a dual-hormone artifical pancreas. IFAC-PapersOnLine 2015, 48, 7–12. [Google Scholar] [CrossRef]
- Nishida, K.; Shimoda, S.; Ichinose, K.; Araki, E.; Shichiri, M. What is the artificial endocrine pancreas? Mechanism and history. World J. Gastroenterol. 2009, 15, 4105–4110. [Google Scholar] [CrossRef] [PubMed]
- Albisser, A.M.; Leibel, B.S.; Ewart, T.G.; Davidovac, Z.; Botz, C.K.; Zingg, W.; Schipper, H.; Gander, R. Clinical control of diabetes by the artificial pancreas. Diabetes 1974, 23, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Bindra, D.S.; Zhang, Y.; Wilson, G.S.; Sternberg, R.; Thévenot, D.R.; Moatti, D.; Reach, G. Design and in vitro studies of a needle-type glucose sensor for subcutaneous monitoring. Anal. Chem. 1991, 63, 1692–1696. [Google Scholar] [CrossRef] [PubMed]
- McGreevy, R. Flash glucose monitoring latest concept in testing. The Irish Times. 22 October 2013. Available online: www.irishtimes.com (accessed on 22 October 2013).
- Worsley, G.J.; Tourniaire, G.A.; Medlock, K.E.S.; Sartain, F.K.; Harmer, H.E.; Thatcher, M.; Horgan, A.M.; Pritchard, J. Continuous blood glucose monitoring with a thin-film optical sensor. Clin. Chem. 2007, 53, 1820–1826. [Google Scholar] [CrossRef] [PubMed]
- Fox, L.A.; Beck, R.W.; Xing, D.; Chase, H.P.; Gilliam, L.K.; Hirsch, I.; Kollman, C.; Laffel, L.; Lee, J.; Ruedy, K.J.; et al. Variation of interstitial glucose measurements assessed by continuous glucose monitors in healthy nondiabetic individuals. Diabetes Care 2010, 33, 1297–1299. [Google Scholar] [CrossRef] [PubMed]
- Koschinsky, T.; Heinemann, L. Sensors for glucose monitoring: Technical and clinical aspects. Diabetes Metab. Res. Rev. 2001, 17, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Song, C.; Hong, Y.S.; Kim, M.S.; Cho, H.R.; Kang, T.; Shin, K.; Choi, S.H.; Hyeon, T.; Kim, D. Wearable/disposable sweat-based glucose monitoring device with multistage transdermal drug delivery module. Sci. Adv. 2017, 3, e1601314. [Google Scholar] [CrossRef] [PubMed]
- Heikenfeld, J. Non-invasive analyte access and sensing through eccrine sweat: Challenges and outlook circa 2016. Electroanalysis 2016, 28, 1242–1249. [Google Scholar] [CrossRef]
- Gupta, S.; Sandhu, S.V.; Bansal, H.; Sharma, D. Comparison of salivary and serum glucose levels in diabetic patients. J. Diabetes Sci. Technol. 2014, 9, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Baliga, S.; Muglikar, S.; Kale, R. Salivary pH: A diagnostic biomarker. J. Indian Soc. Periodontol. 2013, 17, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Sun, M.; Wang, Z.; Chen, Z.; Zhao, X.; Yuan, Y.; Li, Y.; Wang, C. A portable real-time ringdown breath acetone analyser: Toward potential diabetic screening and management. Sensors 2016, 16, 1199. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J. Exhaled breath condensate pH assays. Immunol. Allergy Clin. N. Am. 2007, 27, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.K. Non-invasive glucose monitoring technology in diabetes management: A review. Anal. Chim. Acta 2012, 750, 16–77. [Google Scholar] [CrossRef] [PubMed]
- Jina, A.; Tierney, M.J.; Tamada, J.A.; McGill, S.; Desai, S.; Chua, B.; Chang, A.; Christiansen, M. Design, development, and evaluation of a novel microneedle array-based continuous glucose monitor. J. Diabetes Sci. Technol. 2014, 8, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Zhi, Z.; Khan, F.; Pickup, J.C. Multilayer nanoencapsulation: A nanomedicine technology for diabetes research and management. Diabetes Res. Clin. Pract. 2013, 100, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Li, S.; Schultz, J.S.; Wang, Q.; Lin, Q. A MEMS affinity glucose sensor using a biocompatible glucose-responsive polymer. Sens. Actuators B Chem. 2009, 140, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Rao, G.; Guy, H.R.; Glikfeld, P.; LaCourse, W.R.; Leung, L.; Tamada, J.; Potts, R.O.; Azimi, N. Reverse iontophoresis: Noninvasive glucose monitoring in vivo in humans. Pharm. Res. 1995, 12, 1869–1873. [Google Scholar] [CrossRef] [PubMed]
- Bagavathiappan, S.; Saravanan, T.; Philip, J.; Jayakumar, T.; Raj, B.; Karunanithi, R.; Panicker, T.M.R.; Korath, M.P.; Jagadeesan, K. Infrared thermal imaging for detection of peripheral vascular disorders. J. Med. Phys. 2009, 34, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Oberli, M.A.; Schoellhammer, C.M.; Langer, R.; Blankschtein, D. Ultrasound-enhanced transdermal delivery: Recent advances and future challenges. Ther. Deliv. 2014, 5, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Hanashi, T.; Yamazaki, T.; Tsugawa, W.; Ikebukuro, K.; Sode, K. BioRadioTransmitter: A self-powered wireless glucose-sensing system. J. Diabetes Sci. Technol. 2011, 5, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.J.; Pishko, M.V.; Gefrides, C.C.; McShane, M.J.; Coté, G.L. A fluorescence-based glucose biosensor using concanavalin a and dextran encapsulated in a poly(ethylene glycol) hydrogel. Anal. Chem. 1999, 71, 3126–3132. [Google Scholar] [CrossRef] [PubMed]
- Henderson, T.A.; Morries, L.D. Near-infrared photonic energy penetration: Can infrared phototherapy effectively reach the human brain? Neuropsychaiatr. Dis. Treat. 2015, 11, 2191–2208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Du, Y.; Wang, M.L. On-chip highly sensitive saliva glucose sensing using multilayer films composed of single-walled carbon nanotubes, gold nanoparticles, and glucose oxidase. Sens. Bio-Sens. Res. 2015, 4, 96–102. [Google Scholar] [CrossRef]
- Yao, H.; Shum, A.J.; Cowan, M.; Lahdesmaki, I.; Parvis, B.A. A contact lens with embedded sensor for monitoring tear glucose level. Biosens. Bioelectron. 2011, 26, 3290–3296. [Google Scholar] [CrossRef] [PubMed]
- Glennon, T.; O’Quigley, C.; McCaul, M.; Matzeu, G.; Beirne, S.; Wallace, G.G.; Stroiescu, F.; O’Mahoney, N.; White, P.; Diamond, D. SWEATCH: A wearable platform for harvesting and analysing sweat sodium content. Electroanalysis 2016, 28, 1–8. [Google Scholar] [CrossRef]
- Heikenfeld, J. Technological leap for sweat sensing. Nature 2016, 529, 475–476. [Google Scholar] [CrossRef] [PubMed]
- Heikenfeld, J. Let them see you sweat: Your sweat may bring medical diagnostics to fitbits and fuelbands. IEEE Spectr. 2014, 51, 46–63. [Google Scholar] [CrossRef]
- Morris, D.; Coyle, S.; Wu, Y.; Lau, K.T.; Wallace, G.; Diamond, D. Bio-sensing textile based patch with integrated optical detection system for sweat monitoring. Sens. Actuators B Chem. 2009, 139, 231–236. [Google Scholar] [CrossRef]
- Mitsubayashi, K.; Suzuki, M.; Tamiya, E.; Karube, I. Analysis of metabolites in sweat as a measure of physical condition. Anal. Chim. Acta 1994, 289, 27–34. [Google Scholar] [CrossRef]
- Bandodkar, A.J.; You, J.-M.; Kim, N.-H.; Gu, Y.; Kumar, R.; Mohan, A.M.V.; Kurniawan, J.; Imani, S.; Nakagawa, T.; Parish, B.; et al. Soft, stretchable, high power density electronic skin-based biofuel cells for scavenging energy from human sweat. Energy Environ. Sci. 2017, 10, 1581–1589. [Google Scholar] [CrossRef]
- Abellán-Llobregat, A.; Jeerapan, I.; Bandodkar, A.; Vidal, L.; Canals, A.; Wang, J.; Morallón, E. A stretchable and screen-printed electrochemical sensor for glucose determination in human perspiration. Biosens. Bioelectron. 2017, 91, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Sempionatto, J.R.; Nakagawa, T.; Pavinatto, A.; Mensah, S.T.; Imani, S.; Mercier, P.; Wang, J. Eyeglasses based wireless electrolyte and metabolite sensor platform. Lab Chip 2017, 17, 1834–1842. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Emaminejad, S.; Nyein, H.Y.Y.; Challa, S.; Chen, K.; Peck, A.; Fahad, H.M.; Ota, H.; Shiraki, H.; Kiriya, D.; et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016, 529, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Liu, Y.; Chen, K.; Shin, W.-J.; Lu, C.-J.; Kong, G.-W.; Patnaik, D.; Lee, S.-H.; Cortes, J.F.; Rogers, J.A. Stretchable, wireless sensors and functional substrates for epidermal characterization of sweat. Small 2014, 10, 3083–3090. [Google Scholar] [CrossRef] [PubMed]
- Jang, K.-I.; Jung, H.N.; Lee, J.W.; Xu, S.; Liu, Y.H.; Ma, Y.; Jeong, J.-W.; Song, Y.M.; Kim, J.; Kim, B.H.; et al. Ferromagnetic, folded electrode composite as a soft interface to the skin for long-term electrophysiological recording. Adv. Funct. Mater. 2016, 26, 7281–7290. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, J.V.; Harvey, A.S.; Wrennall, J.; Zacharin, M.; Berkovic, S.F. Transcallosal resection of hypothalamic hamartomas, with control of seizures, in children with gelastic epilepsy. Neurosurgery 2001, 48, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Cram, J.R.; Steger, J.C. EMG scanning in the diagnosis of chronic pain. Biofeedback Self-Regul. 1983, 8, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Parkey, R.W.; Bonte, F.J.; Meyer, S.L.; Atkins, J.M.; Curry, G.L.; Stokely, E.M.; Willerson, J.T. A new method for radionuclide imaging of acute myocardial infarction in humans. Circulation 1974, 50, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Staman, J.A.; Fitzgerald, C.R.; Dawson, W.W.; Barris, M.C.; Hood, C.I. The eog and choroidal malignant melanomas. Doc. Ophthalmol. 1980, 49, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Fishberg, E.H.; Beirman, W. Acid-base balance in sweat. J. Biol. Chem. 1932, 97, 433–441. [Google Scholar]
- Hill, D.; Binions, R. Breath analysis for medical diagnosis. Int. J. Smart Sens. Intell. Syst. 2012, 5, 401–440. [Google Scholar]
- Wilson, A.D. Advances in electronic-nose technologies for the detection of volatile biomarker metabolites in the human breath. Metabolites 2015, 5, 140–163. [Google Scholar] [CrossRef] [PubMed]
- Mathew, T.L.; Pownraj, P.; Abdulla, S.; Pullithadathil, B. Technologies for clinical diagnosis using expired human breath analysis. Diagnostics 2015, 5, 27–60. [Google Scholar] [CrossRef] [PubMed]
- Xing, R.; Xu, L.; Song, J.; Zhou, C.; Li, Q.; Liu, D.; Wei Song, H. Preparation and gas sensing properties of In2O3/Au nanorods for detection of volatile organic compounds in exhaled breath. Sci. Rep. 2015, 5, 10717. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Ohishi, K.; Gilbert, A.; Akasaka, M.; Yoshida, N.; Yoshimura, R. Measurement of natural carbon isotopic composition of acetone in human urine. Anal. Bioanal. Chem. 2016, 408, 1597–1607. [Google Scholar] [CrossRef] [PubMed]
- Shafiek, H.; Fiorentino, F.; Merino, J.L.; Lopez, C.; Oliver, A.; Segura, J.; de Paul, I.; Sibila, O.; Agusti, A.; Cosio, B.G. Using the electronic nose to identify airway infection during copd exacerbations. PLoS ONE 2015, 10, e0135199. [Google Scholar] [CrossRef] [PubMed]
- Pavlou, A.; Turner, A.P.; Magan, N. Recognition of anaerobic baterical isolates in vitro using electronic nose technology. Lett. Appl. Microbiol. 2002, 35, 366–369. [Google Scholar] [CrossRef] [PubMed]
- Pavlou, A.K.; Magan, N.; Jones, J.M.; Brown, J.; Klatser, P.; Turner, A.P. Detection of mycobaterium tuberculosis (tb) in vitro and in situ using an electronic nose in combination with a neural network system. Biosens. Bioelectron. 2004, 20, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Di Natale, C.; Macagnano, A.; Davide, F.; D’Amico, A.; Paolesse, R.; Boschi, T.; Faccio, M.; Ferri, G. An electronic nose for food analysis. Sens. Actuators B Chem. 1997, 44, 521–526. [Google Scholar] [CrossRef]
- Xu, S.; Zhou, Z.; Li, K.; Jamir, S.M.; Luo, X. Recognition of the duration and prediction of insect prevalence of stored rough rice infested by the red fluor beetle (tribolium castaneum herbst) using an electronic nose. Sensors (Basel) 2017, 17, 688. [Google Scholar] [CrossRef] [PubMed]
- Bikov, A.; Hernadi, M.; Korosi, B.Z.; Kunos, L.; Zsamboki, G.; Sutto, Z.; Tarnoki, A.D.; Tarnoki, D.L.; Losonczy, G.; Horvath, I. Expiratory flow rate, breath hold and anatomic dead space influence electronic nose ability to detect lung cancer. BMC Pulm. Med. 2014, 14, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Plaza, V.; Crespo, A.; Giner, J.; Merino, J.L.; Ramos-Barbon, D.; Mateus, E.F.; Torrego, A.; Cosio, B.G.; Agusti, A.; Sibila, O. Inflammatory asthma phenotype discrimination using an electronic nose breath analyzer. J. Investig. Allergol. Clin. Immunol. 2015, 25, 431–437. [Google Scholar] [PubMed]
- Chen-Kaminsky, S.; Nakhleh, M.; Perros, F.; Montani, D.; Girerd, B.; Garcia, G.; Simonneau, G.; Haick, H.; Humbert, M. A proof of concept for the detection and classification of pulmonary arterial hypertension through breath analysis with a sensor array. Am. J. Respir. Crit. Care Med. 2013, 188, 756–759. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Sheng, Y.; Sun, Y.; Feng, J.; Wang, S.; Zhang, J.; Xu, J.; Jiang, D. A glucose oxidase-coupled DNAzyme sensor for glucose detection in tears and saliva. Biosens. Bioelectron. 2015, 70, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Valdes-Ramirez, G.; Bandodkar, A.J.; Jia, W.; Martinez, A.G.; Ramirez, J.; Mercier, P.; Wang, J. Non-invasive mouthguard biosensor for continuous salivary monitoring of metabolites. Analyst 2013, 139, 1632–1636. [Google Scholar] [CrossRef] [PubMed]
- Mannoor, M.S.; Tao, H.; Clayton, J.D.; Sengupta, A.; Kaplan, D.L.; Naik, R.R.; Verma, N.; Omenetto, F.G.; McAlpine, M.C. Graphene-based wireless bacteria detection on tooth enamel. Nat. Commun. 2012, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.; Lahdesmaki, I.; Parvis, B.A. A contact lens with an integrated lactate sensor. Sens. Actuators B Chem. 2012, 162, 128–134. [Google Scholar] [CrossRef]
- King, L. Google Smart Contact Lens Focuses on Healthcare Billions. Available online: www.forbes.com/sites/leoking/2014/07/15/google-smart-contact-lens-focuses-on-healthcare-billions/ (accessed on 15 July 2014).
- Senior, M. Novartis signs up for Google smart lens. Nat. Biotechnol. 2014, 32, 856. [Google Scholar] [CrossRef] [PubMed]
- Scott, M. Novartis Joins with Google to Develop Contact Lens That Monitors Blood Sugar. Available online: https://www.nytimes.com/2014/07/16/business/international/novartis-joins-with-google-to-develop-contact-lens-to-monitor-blood-sugar.html?_r=1 (accessed on 15 July 2014).
- Tsukayama, H. Google’s Smart Contact Lens: What It Does and How It Works. Available online: http://www.washingtonpost.com/business/technology/googles-smart-contact-lens-what-it-does-and-how-it-works/2014/01/17/96b938ec-7f80-11e3-93c1-0e888170b723_story.html (accessed on 17 January 2014).
- Kleinman, J. Google’s Smart Contact Lenses Move One Step Closer to Launch. Available online: https://www.technobuffalo.com/2014/07/15/google-smart-contact-lens/ (accessed on 15 July 2014).
- Watt, B.E.; Proudfoot, A.T.; Vale, J.A. Hydrogen peroxide poisoning. Toxicol. Rev. 2004, 23, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Reid, R.C.; Minteer, S.D.; Gale, B.K. Contact lens biofuel cells tested in a synthetic tear solution. Biosens. Bioelectron. 2015, 68, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J. Google Contact Lenses: Tech Giant Licenses Smart Contact Lens Technology to Help Diabetics and Glasses Wearers. Available online: http://www.independant.co.uk/life-style/gadgets-and-tech/google-licenses-smart-contact-lens-technology-to-help-diabetics-and-glasses-wearers-9607368.html (accessed on 15 July 2014).
- Iguchi, S.; Kudo, H.; Saito, T.; Ogawa, M.; Satio, H.; Otsuka, K.; Funakubo, A.; Mitsubayashi, K. A flexible and wearable biosensor for tear glucose measurement. Biomed. Microdevices 2007, 9, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Kagie, A.; Bishop, D.K.; Burdick, J.; La Belle, J.T.; Dymond, R.; Felder, R.; Wang, J. Flexible rolled thick-film miniaturized flow-cell for minimally invasive amperometric sensing. Electroanalysis 2008, 20, 1610–1614. [Google Scholar] [CrossRef]
- Badugu, R.; Lakowicz, J.R.; Geddes, C.R. Wavelength-ratiometric and colorimetric robes for glucose determination. Dyes Pigments 2006, 68, 159–163. [Google Scholar] [CrossRef]
- Geddes, C.D.; Badugu, R.; Lakowicz, R.J. Quaternary Nitrogen Heterocyclic Compounds for Detecting Aqueous Monosaccharides in Physiological Fluids. U.S. Patent US20070020182A1, 25 January 2007. [Google Scholar]
- Jeong, J.W.; Arnob, M.P.; Baek, K.-M.; Lee, S.Y.; Shih, W.-C.; Jung, Y.S. 3D cross-point plasmonic nanoarchitectures containing dense and regular hot spots for surface-enhanced ramen spectroscopy analysis. Adv. Mater. 2016, 28, 8695–8704. [Google Scholar] [CrossRef] [PubMed]
- Moser, T.; Celma, C.; Lebert, A.; Charrault, E.; Brooke, R.; Murphy, P.J.; Browne, G.; Young, R.; Higgs, T.; Evans, D. Hydrophilic organic electrode on flexible hydrogels. ACS Appl. Mater. Interfaces 2016, 8, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Kell, M. Making Magic: The Smart Contact Lens. Available online: https://www.mivision.com.au/makingmagic-the-smart-contact-lens/ (accessed on 21 June 2017).




| Physiological Fluid | Biomarker | Concentration for Healthy Patients’ | Concentration for Diabetic Patients’ | pH |
|---|---|---|---|---|
| Blood | Glucose | 4.9–6.9 mM [19] | 2–40 mM [20,40] | 7.35–7.45 [14] |
| Interstitial Fluid | Glucose | 3.9–6.6 [53] | 1.99–22.2 [54] | 7.2–7.4 [14] |
| Urine | Glucose | 2.78–5.55 mM [18] | >5.55 mM [18] | 4.5–8 [14] |
| Sweat | Glucose | 0.06–0.11 mM [55] | 0.01–1 mM [55] | 4.5–7 [56] |
| Saliva | Glucose | 0.23–0.38 mM [57] | 0.55–1.77 mM [57] | 6.2–7.6 [58] |
| Ocular Fluid | Glucose | 0.05–0.5 mM [21] | 0.5–5 mM [20,21] | 6.5–7.6 [18] |
| Breath | Acetone | 0.1–2 ppm [59] | 0.1–103.7 ppm [59] | 7.4–8.1 [60] |
| Sensing Technique | Explanation | Reference |
|---|---|---|
| Reverse Iontophoresis | Reverse iontophoresis is the passing of a current over the skin to drive ions from the interstitial fluid and onto the surface of the skin, where they can be analysed. This results due to the increased negative charge across the skin which drives cations from the body on to the skin surface. | [65] |
| Thermal Emission | Thermal emission, infrared thermography, infrared imaging or thermal imaging is a non-contact tool that uses the surface body temperature to gain insight in to the detection of diseases, flow of blood or muscular performance of an individual. | [66] |
| Photoacoustic Detection | Photoacoustic spectroscopy uses a form of light absorption to detect an analyte concentration in a biofluid, such as blood. Light from a laser source is absorbed by blood which generates heat in a localised region. The heat induces ultrasonic pressure waves in blood, which can be detected by an ultrasonic transducer and directly related to an analyte concentration that absorbs at the laser wavelength. | [10] |
| Sonophoresis | Transdermal delivery of drugs by ultrasound. | [67] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruen, D.; Delaney, C.; Florea, L.; Diamond, D. Glucose Sensing for Diabetes Monitoring: Recent Developments. Sensors 2017, 17, 1866. https://doi.org/10.3390/s17081866
Bruen D, Delaney C, Florea L, Diamond D. Glucose Sensing for Diabetes Monitoring: Recent Developments. Sensors. 2017; 17(8):1866. https://doi.org/10.3390/s17081866
Chicago/Turabian StyleBruen, Danielle, Colm Delaney, Larisa Florea, and Dermot Diamond. 2017. "Glucose Sensing for Diabetes Monitoring: Recent Developments" Sensors 17, no. 8: 1866. https://doi.org/10.3390/s17081866
APA StyleBruen, D., Delaney, C., Florea, L., & Diamond, D. (2017). Glucose Sensing for Diabetes Monitoring: Recent Developments. Sensors, 17(8), 1866. https://doi.org/10.3390/s17081866






