Sensing Responses Based on Transfer Characteristics of InAs Nanowire Field-Effect Transistors
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
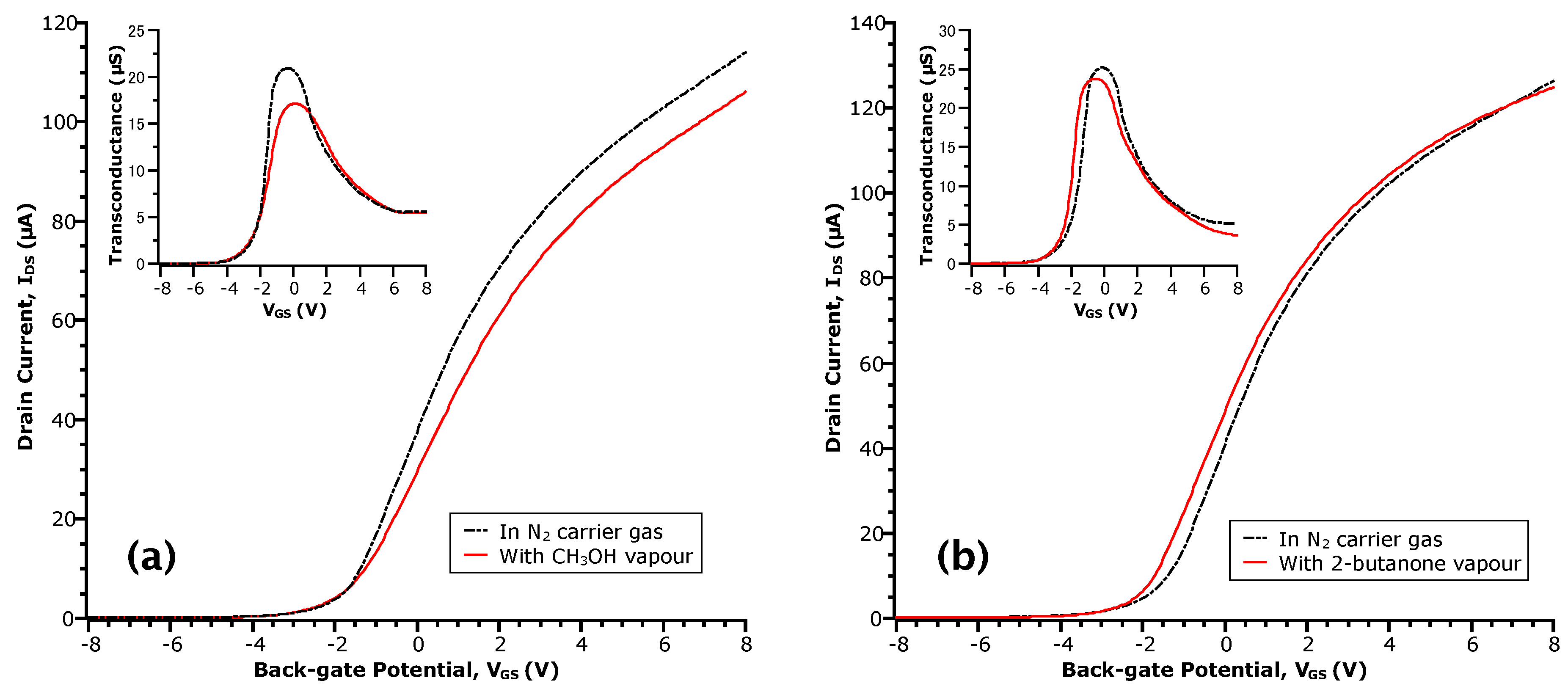
3.1. FET Transfer Curves in Acetic Acid
3.2. Physical versus Chemical Adsorption
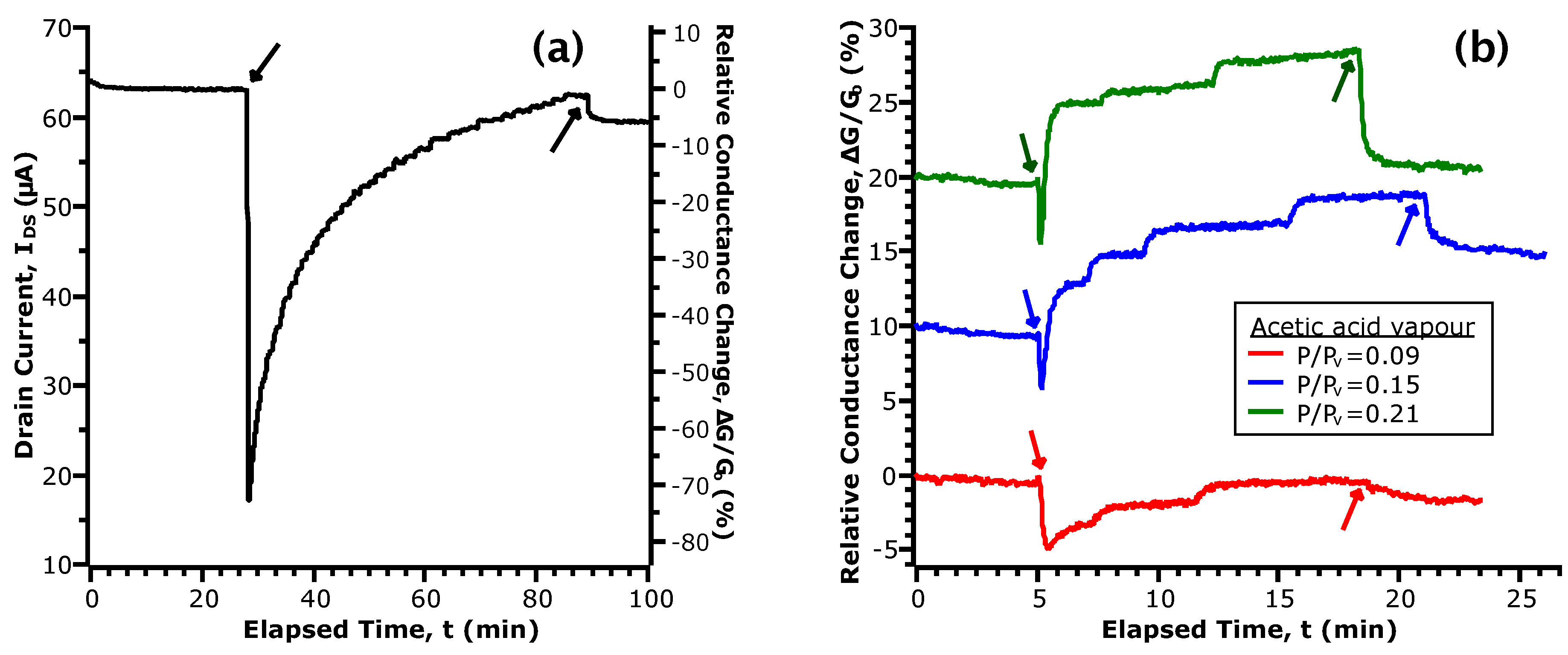
3.3. Current Transients in Acetic Acid
3.4. Correlation of Sensor Response
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ko, S.; Jang, J. Controlled Amine Functionalization on Conducting Polypyrrole Nanotubes as Effective Transducers for Volatile Acetic Acid. Biomacromolecules 2007, 8, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Ma, S.; Sun, A.; Qin, R.; Yang, F.; Li, X.; Li, F.; Yang, X. Characterization of electrospun Pr-doped ZnO nanostructure for acetic acid sensor. Sens. Actuators B Chem. 2014, 193, 326–333. [Google Scholar] [CrossRef]
- Mane, G.P.; Talapaneni, S.N.; Anand, C.; Varghese, S.; Iwai, H.; Ji, Q.; Ariga, K.; Mori, T.; Vinu, A. Preparation of Highly Ordered Nitrogen-Containing Mesoporous Carbon from a Gelatin Biomolecule and Its Excellent Sensing of Acetic Acid. Adv. Funct. Mater. 2012, 22, 3596–3604. [Google Scholar] [CrossRef]
- Shehada, N.; Cancilla, J.C.; Torrecilla, J.S.; Pariente, E.S.; Brönstrup, G.; Christiansen, S.; Johnson, D.W.; Leja, M.; Davies, M.P.A.; Liran, O.; et al. Silicon Nanowire Sensors Enable Diagnosis of Patients via Exhaled Breath. ACS Nano 2016, 10, 7047–7057. [Google Scholar] [CrossRef] [PubMed]
- Dryahina, K.; Pospíšilová, V.; Sovová, K.; Shestivska, V.; Kubišta, J.; Spesyvyi, A.; Pehal, F.; Turzíková, J.; Votruba, J.; Spaněl, P. Exhaled breath concentrations of acetic acid vapour in gastro-esophageal reflux disease. J. Breath Res. 2014, 8, 037109. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.; Sovová, K.; Dryahina, K.; Doušová, T.; Dřevínek, P.; Španěl, P. Breath concentration of acetic acid vapour is elevated in patients with cystic fibrosis. J. Breath Res. 2016, 10, 021002. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Zhong, Z.; Wang, D.; Wang, W.U.; Lieber, C.M. High Performance Silicon Nanowire Field Effect Transistors. Nano Lett. 2003, 3, 149–152. [Google Scholar] [CrossRef]
- Kolmakov, A.; Moskovits, M. Chemical Sensing and Catalysis by One-Dimensional Metal-Oxide Nanostructures. Ann. Rev. Mater. Res. 2004, 34, 151–180. [Google Scholar] [CrossRef]
- Patolsky, F.; Zheng, G.; Hayden, O.; Lakadamyali, M.; Zhuang, X.; Lieber, C.M. Electrical detection of single viruses. Proc. Natl. Acad. Sci. USA. 2004, 101, 14017–14022. [Google Scholar] [CrossRef] [PubMed]
- Stern, E.; Klemic, J.F.; Routenberg, D.A.; Wyrembak, P.N.; Turner-Evans, D.B.; Hamilton, A.D.; LaVan, D.A.; Fahmy, T.M.; Reed, M.A. Label-free immunodetection with CMOS-compatible semiconducting nanowires. Nature 2007, 445, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Ramgir, N.S.; Yang, Y.; Zacharias, M. Nanowire-based sensors. Small 2010, 6, 1705–1722. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Cai, B.; Tang, Q.; Tong, Y.; Liu, Y. One-Dimensional Nanostructure Field-Effect Sensors for Gas Detection. Sensors 2014, 14, 13999–14020. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Chang, Y.E.; Sawtelle, S.D.; Wipf, M.; Duan, X.; Reed, M.A.; Member, S. Silicon Nanowire Field-Effect Transistors — A Versatile Class of Potentiometric Nanobiosensors. IEEE Access 2015, 3, 287–302. [Google Scholar] [CrossRef]
- Wolkenstein, T. The Electron Theory of Catalysis on Semiconductors. Adv. Catal. 1960, 12, 189–264. [Google Scholar]
- Kiselev, V.F.; Krylov, O.V. Electronic Phenomena in Adsorption and Catalysis on Semiconductors and Dielectrics; Number 7 in Springer Series in Materials Science; Springer: Berlin Heidelberg, Germany, 1987. [Google Scholar]
- Li, J.; Pud, S.; Petrychuk, M.; Offenhäusser, A.; Vitusevich, S. Sensitivity enhancement of Si nanowire field effect transistor biosensors using single trap phenomena. Nano Lett. 2014, 14, 3504–3509. [Google Scholar] [CrossRef] [PubMed]
- Salfi, J.; Savelyev, I.G.; Blumin, M.; Nair, S.V.; Ruda, H.E. Direct observation of single-charge-detection capability of nanowire field-effect transistors. Nat. Nanotechnol. 2010, 5, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Knopfmacher, O.; Tarasov, A.; Fu, W.; Wipf, M.; Niesen, B.; Calame, M.; Schönenberger, C. Nernst Limit in Dual-Gated Si-Nanowire FET Sensors. Nano Lett. 2010, 10, 2268–2274. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Zhou, W.; Jiang, X.; Hong, G.; Fu, T.M.; Lieber, C.M. General Strategy for Biodetection in High Ionic Strength Solutions Using Transistor-Based Nanoelectronic Sensors. Nano Lett. 2015, 15, 2143–2148. [Google Scholar] [CrossRef] [PubMed]
- Puppo, F.; Traversa, F.L.; Ventra, M.D.; Micheli, G.D.; Carrara, S. Surface trap mediated electronic transport in biofunctionalized silicon nanowires. Nanotechnology 2016, 27, 345503. [Google Scholar] [CrossRef] [PubMed]
- Presnova, G.; Presnov, D.; Krupenin, V.; Grigorenko, V.; Trifonov, A.; Andreeva, I.; Ignatenko, O.; Egorov, A.; Rubtsova, M. Biosensor based on a silicon nanowire field-effect transistor functionalized by gold nanoparticles for the highly sensitive determination of prostate specific antigen. Biosens. Bioelectron. 2017, 88, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Sakata, T.; Matsuse, Y. In situ electrical monitoring of cancer cells invading vascular endothelial cells with semiconductor-based biosensor. Genes Cells 2017, 22, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.R.; Akbar, S.A.; Morris, P.A. Nanoscale metal oxide-based heterojunctions for gas sensing: A review. Sens. Actuators B Chem. 2014, 204, 250–272. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, J.H. Highly sensitive and selective gas sensors using p-type oxide semiconductors: Overview. Sens. Actuators B Chem. 2014, 192, 607–627. [Google Scholar] [CrossRef]
- Dayeh, S.A. Electron transport in indium arsenide nanowires. Semiconduct. Sci. Technol. 2010, 25, 1–20. [Google Scholar] [CrossRef]
- Noguchi, M.; Hirakawa, K.; Ikoma, T. Intrinsic electron accumulation layers on reconstructed clean InAs(100) surfaces. Phys. Rev. Lett. 1991, 66, 2243–2246. [Google Scholar] [CrossRef] [PubMed]
- Halpern, E.; Cohen, G.; Gross, S.; Henning, A.; Matok, M.; Kretinin, A.V.; Shtrikman, H.; Rosenwaks, Y. Measuring surface state density and energy distribution in InAs nanowires. Phys. Status Solidi A 2014, 211, 473–482. [Google Scholar] [CrossRef]
- Petrovykh, D.Y.; Yang, M.J.; Whitman, L.J. Chemical and electronic properties of sulfur-passivated InAs surfaces. Surf. Sci. 2003, 523, 231–240. [Google Scholar] [CrossRef]
- Du, J.; Liang, D.; Tang, H.; Gao, X.P. InAs Nanowire Transistors as Gas Sensor and the Response Mechanism. Nano Lett. 2009, 9, 4348–4351. [Google Scholar] [CrossRef] [PubMed]
- Dedigama, A.; Angelo, M.; Torrione, P.; Kim, T.H.; Wolter, S.; Lampert, W.; Atewologun, A.; Edirisoorya, M.; Collins, L.; Kuech, T.F.; et al. Hemin-Functionalized InAs-Based High Sensitivity Room Temperature NO Gas Sensors. J. Phys. Chem. C 2012, 116, 826–833. [Google Scholar] [CrossRef]
- Offermans, P.; Crego-Calama, M.; Brongersma, S.H. Gas Detection with Vertical InAs Nanowire Arrays. Nano Lett. 2010, 10, 2412–2415. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Fu, M.; Li, X.; Shi, T.; Ning, Z.; Wang, X.; Yang, T.; Chen, Q. Study on the response of InAs nanowire transistors to H2O and NO2. Sens. Actuators B Chem. 2015, 209, 456–461. [Google Scholar] [CrossRef]
- Upadhyay, S.; Frederiksen, R.; Lloret, N.; Vico, L.D.; Krogstrup, P.; Jensen, J.H.; Martinez, K.L.; Nygård, J. Indium arsenide nanowire field-effect transistors for pH and biological sensing. Appl. Phys. Lett. 2014, 104, 203504. [Google Scholar] [CrossRef]
- Tarasov, A.; Wipf, M.; Stoop, R.L.; Bedner, K.; Fu, W.; Guzenko, V.A.; Knopfmacher, O.; Calame, M.; Schönenberger, C. Understanding the Electrolyte Background for Biochemical Sensing with Ion-Sensitive Field-Effect Transistors. ACS Nano 2012, 6, 9291–9298. [Google Scholar] [CrossRef] [PubMed]
- Paska, Y.; Stelzner, T.; Christiansen, S.; Haick, H. Enhanced Sensing of Nonpolar Volatile Organic Compounds by Silicon Nanowire Field Effect Transistors. ACS Nano 2011, 5, 5620–5626. [Google Scholar] [CrossRef] [PubMed]
- Paska, Y.; Stelzner, T.; Assad, O.; Tisch, U.; Christiansen, S.; Haick, H. Molecular Gating of Silicon Nanowire Field-Effect Transistors with Nonpolar Analytes. ACS Nano 2012, 6, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Haick, H. Effect of Functional Groups on the Sensing Properties of Silicon Nanowires toward Volatile Compounds. ACS Appl. Mater. Interfaces 2013, 5, 2289–2299. [Google Scholar] [CrossRef] [PubMed]
- Jewett, S.A.; Ivanisevic, A. Wet-Chemical Passivation of InAs: Toward Surfaces with High Stability and Low Toxicity. Acc. Chem. Res. 2012, 45, 1451–1459. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.H.; Mei, X.Y.; Kim, D.; Blumin, M.; Ruda, H.E. Growth of Au-catalyzed ordered GaAs nanowire arrays by molecular-beam epitaxy. Appl. Phys. Lett. 2002, 81, 5177–5179. [Google Scholar] [CrossRef]
- Fan, Z.; Ho, J.C.; Jacobson, Z.A.; Yerushalmi, R.; Alley, R.L.; Razavi, H.; Javey, A. Wafer-Scale Assembly of Highly Ordered Semiconductor Nanowire Arrays by Contact Printing. Nano Lett. 2008, 8, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Lynall, D.; Byrne, K.; Shik, A.; Nair, S.V.; Ruda, H.E. Surface Properties from Transconductance in Nanoscale Systems. Nano Lett. 2016, 16, 6028–6035. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.P.A.; Zheng, G.; Lieber, C.M. Subthreshold Regime has the Optimal Sensitivity for Nanowire FET Biosensors. Nano Lett. 2010, 10, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Lu, J.G. Gate-refreshable nanowire chemical sensors. Appl. Phys. Lett. 2005, 86, 123510. [Google Scholar] [CrossRef]
- Salfi, J.; Nair, S.V.; Savelyev, I.G.; Blumin, M.; Ruda, H.E. Evidence for nonlinear screening and enhancement of scattering by a single Coulomb impurity for dielectrically confined electrons in InAs nanowires. Phys. Rev. B 2012, 85, 235316. [Google Scholar] [CrossRef]
- Akbar, M.A.; Ali, A.A.S.; Amira, A.; Bensaali, F.; Benammar, M.; Hassan, M.; Bermak, A. An Empirical Study for PCA- and LDA-Based Feature Reduction for Gas Identification. IEEE Sens. J. 2016, 16, 5734–5746. [Google Scholar] [CrossRef]
- Wang, B.; Huynh, T.P.; Wu, W.; Hayek, N.; Do, T.T.; Cancilla, J.C.; Torrecilla, J.S.; Nahid, M.M.; Colwell, J.M.; Gazit, O.M.; et al. A Highly Sensitive Diketopyrrolopyrrole-Based Ambipolar Transistor for Selective Detection and Discrimination of Xylene Isomers. Adv. Mater. 2016, 28, 4012–4018. [Google Scholar] [CrossRef] [PubMed]


| Analyte | Formula | Pv a (kPa) | VTh (V) | Ioff (µA) | FE (cm2/V s) | SS (V/dec) | QS (e cm−2) |
|---|---|---|---|---|---|---|---|
| nitrogen | N2 | – | −1.6 to −1.8 | 0.12 to 0.42 | 540 to 770 | 1.6 to 2.2 | 1.3 to 1.6 × 1013 |
| methanol | CH3OH | 14 | −1.8 | 0.12 | 440 | 1.9 | 1.6 × 1013 |
| 2-butanone | CH3COC2H5 | 25 | −2.1 | 0.15 | 610 | 1.7 | 1.6 × 1013 |
| acetic acid | CH3COOH | 1.8 | – | – | – | – | – |
| = 0.09 | – | – | – | 1.6 | 50 | 13.6 | 1.0 × 1014 |
| = 0.15 | – | – | – | 16 | 110 | 23.6 | 1.8 × 1014 |
| = 0.21 | – | – | – | 25 | 110 | 29.1 | 2.2 × 1014 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tseng, A.C.; Lynall, D.; Savelyev, I.; Blumin, M.; Wang, S.; Ruda, H.E. Sensing Responses Based on Transfer Characteristics of InAs Nanowire Field-Effect Transistors. Sensors 2017, 17, 1640. https://doi.org/10.3390/s17071640
Tseng AC, Lynall D, Savelyev I, Blumin M, Wang S, Ruda HE. Sensing Responses Based on Transfer Characteristics of InAs Nanowire Field-Effect Transistors. Sensors. 2017; 17(7):1640. https://doi.org/10.3390/s17071640
Chicago/Turabian StyleTseng, Alex C., David Lynall, Igor Savelyev, Marina Blumin, Shiliang Wang, and Harry E. Ruda. 2017. "Sensing Responses Based on Transfer Characteristics of InAs Nanowire Field-Effect Transistors" Sensors 17, no. 7: 1640. https://doi.org/10.3390/s17071640
APA StyleTseng, A. C., Lynall, D., Savelyev, I., Blumin, M., Wang, S., & Ruda, H. E. (2017). Sensing Responses Based on Transfer Characteristics of InAs Nanowire Field-Effect Transistors. Sensors, 17(7), 1640. https://doi.org/10.3390/s17071640






