A Hybrid FPGA-Based System for EEG- and EMG-Based Online Movement Prediction
Abstract
:1. Introduction
Contributions and Structure of the Paper
2. Related Work
2.1. EEG-Based Movement Prediction
2.2. EMG-Based Movement Prediction
2.3. Hybrid BCI Systems
2.4. Mobile and Embedded BCI Systems
2.5. Field Programmable Gate Arrays
2.6. Dataflow Architectures and Hardware Acceleration
3. Hard- and Software Architecture of the Hybid System
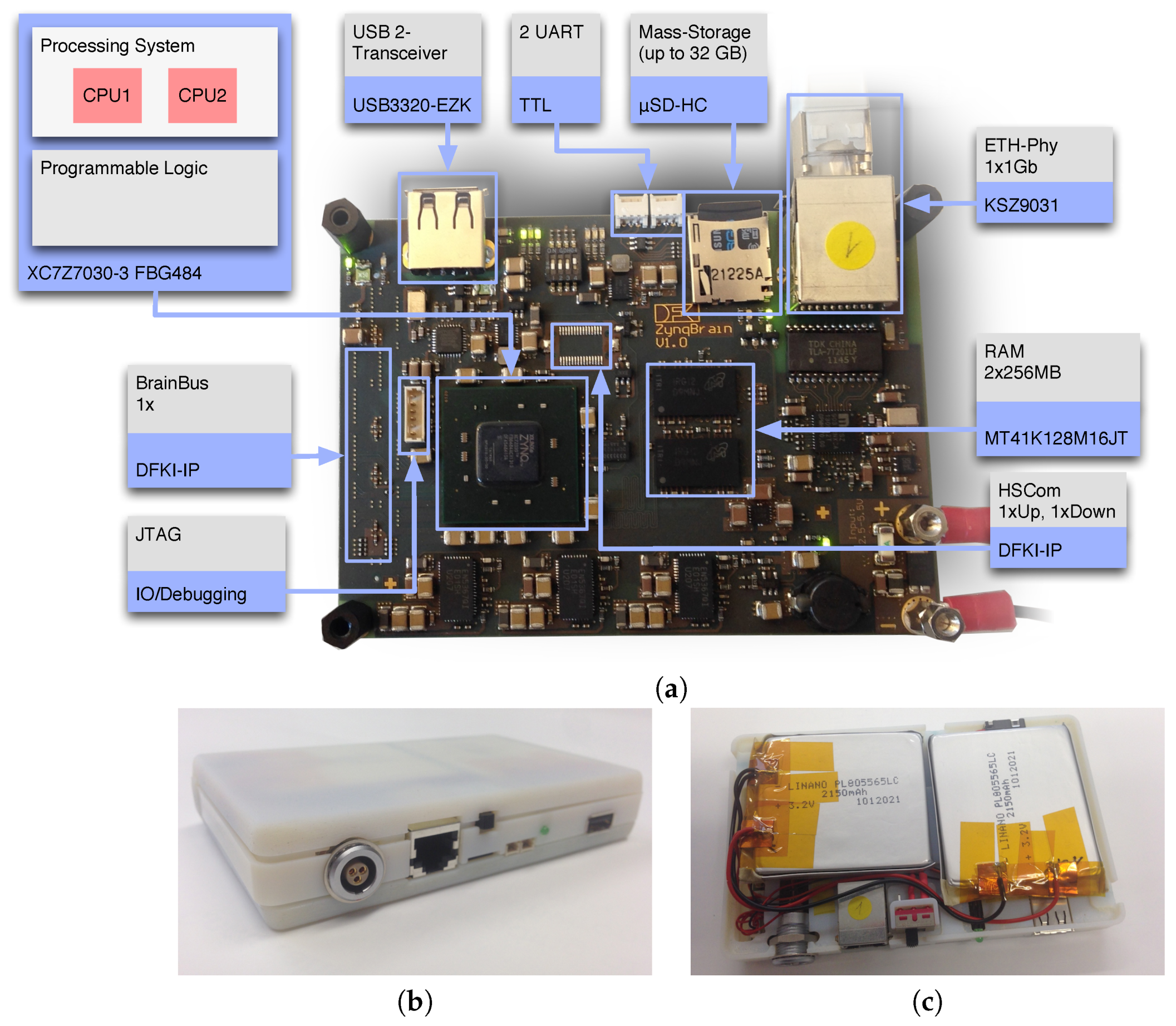
3.1. The ZynqBrain Electronics Platform
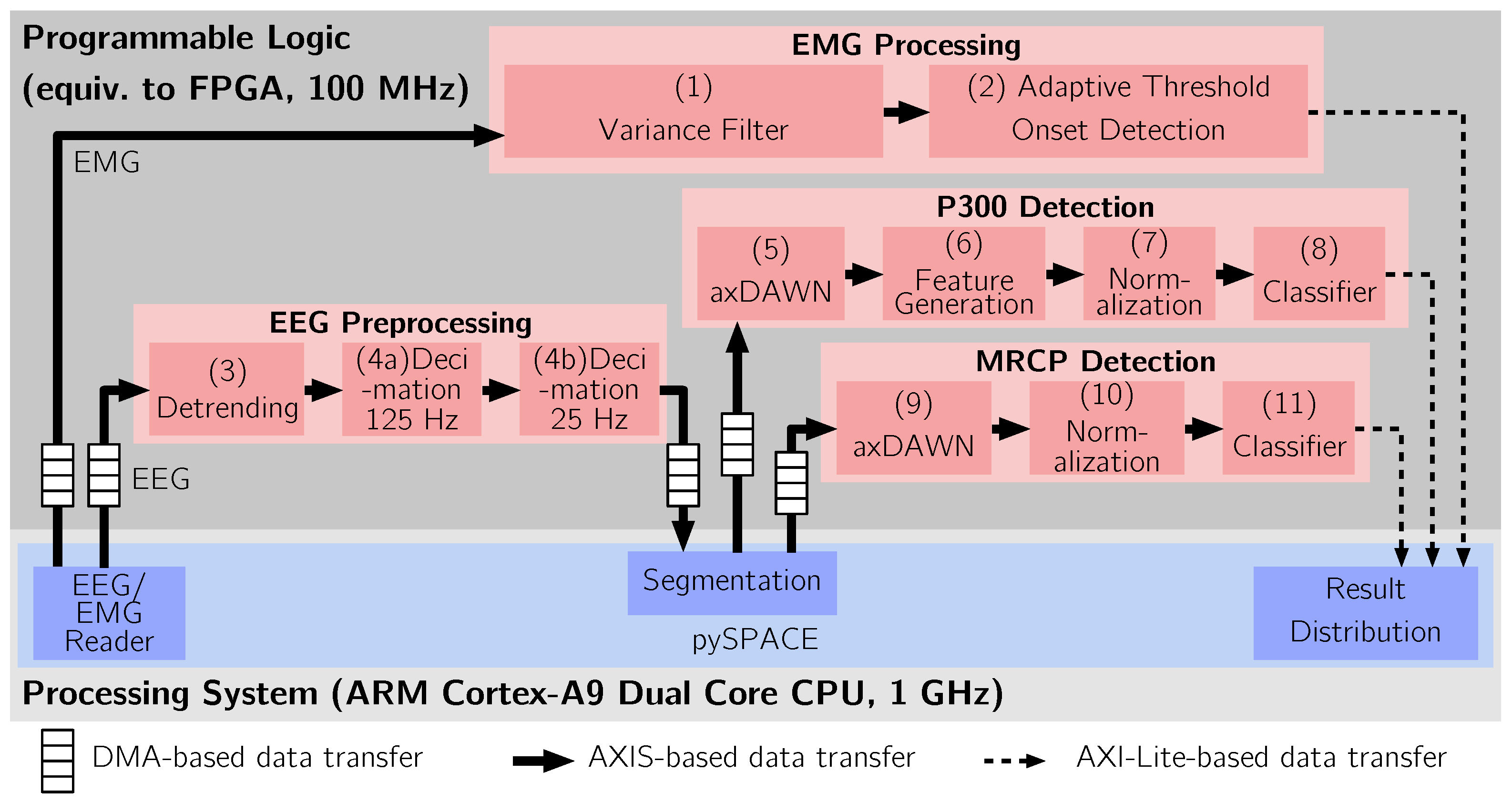
3.2. Dataflow Hardware Accelerator Architecture
3.3. Software Architecture
3.4. Investigated Physiological Signals
- EMG: Similarly, the EMG is used as a signal that indicates an upcoming movement.
- P300: The P300 is not directly related to an upcoming movement. However, it can be used to select one of several different modes of a BCI system. In this paper, we follow this approach. It is assumed that the subject is most of the time in an idle state and switches to an active state following the instruction of a human or virtual trainer or therapist. In such an application, the successful detection of the P300 in response to a command can be used as an indicator that the subject will perform a movement in the near future. Subsequent to the command, we expect a movement in a time window with a length of 5 s.
3.5. Combination of Signals for Hybrid Movement Prediction
- MRCP: The prediction of the movement onset is based only on the prediction of the MRCP.
- EMG: The prediction of the movement onset is based only on EMG analysis.
- MRCP or EMG (MoE): The prediction of the movement onset is based on the combination of the MRCP and EMG predictions. The combination is obtained using a logical or combination of the single predictions. Formally, let be the predictions of the EMG and EEG of an upcoming movement at time t, respectively, where 1 represents the upcoming movement. The combined prediction is then given by .
- MRCP and EMG (MaE): Similar to MoE, but the combination is obtained using a logical and, i.e., .
- P300 and MRCP (PaM): As discussed above, the P300 is used as a switch to select between an idle and an active state. Specifically, let denote the detection of the P300 at time . If , we set for and compute .
- P300 and EMG (PaE): Similar to PaM, but the EMG is used for the movement detection, i.e., we compute .
- P300 and MRCP or EMG (PaMoE): This combination is based on the combinations MoE and PaM, i.e., we compute .
- P300 and MRCP and EMG (PaMaE): Similar to PaMaE with .
4. Applied Signal Processing and Machine Learning Procedures
4.1. EMG Processing
- (1)
- Variance Filter: To obtain a signal with smoothed baseline noise and enlarged signal amplitudes during movement phases, a running variance method was used for preprocessing of the EMG signals. The calculation was based on [144], but was computed as described in [145] to calculate the running variance v at time t as:where is the window length. Variance filtering does not change the dimensionality of the data.
- (2)
- Adaptive Threshold Comparison and Classification: The actual onset detection was based on the comparison of the variance-filtered signal with an adaptive threshold. The threshold was computed aswith the mean value, the standard deviation, the length of the window for the mean and standard deviation and p the sensitivity factor of the threshold [146]. To compute the standard deviation, the methods described in [145] were used again. The adaptive threshold compensates slow drifts in the EMG signals or changing noise levels in the signal caused by, e.g., resistance changes at the electrode side. The classification was performed using a threshold comparison. That is, a segment was classified as belonging to the movement class if the single sample of the variance filtered signal of more than a previously chosen number of channels were higher than T. Due to the adaptive threshold based movement detection, the flow for EMG-based movement prediction required no explicit training phase. A single classification was computed for each data segment.
4.2. EEG Preprocessing
- (3)
- Detrending: First, detrending was used to remove slowly varying signal components using an IIR filter [147], which could otherwise produce a bias in the data.
- (4)
- (a + b) Decimation: Subsequently, the sampling rate was decimated in two steps [148,149], the first step reduced the sampling frequency from 5 kHz to 125 Hz. In the second step, a further reduction of the sampling frequency to 25 Hz was performed. The anti-alias FIR of the second step was parameterized so that all frequencies greater than 4 Hz were attenuated as proposed in [150,151]. In the decimation, each data segment is reduced to a c dimensional vector. The output of the second decimation step was sent back to the main memory for further segmentation in software.
4.3. Data Segmentation
4.4. P300 Processing
- (5)
- Spatial Filtering: The axDAWN spatial filter [140] was applied to decrease the number of channels to four, creating a dimensional matrix. This operation can be realized as a matrix multiplication (using DSP48 [121] slices if realized in hardware) and was implemented using a specialized accelerator for matrix operations [152].
- (6)
- Feature Generation: To generate features for classification the time samples were transformed into local straight line features, i.e., polynomial features of order one [47,57]. To this end, every segment was divided into subsegments of length of 400 ms that are shifted by 120 ms. A polynomial was fitted to each subsegment. The polynomials allow to describe the P300 by a series of slope values. The extracted slopes were combined in a single 24-dimensional feature vector.
- (7)
- Feature Standardization: A further operation was component-wise feature standardization, i.e., we computed , where are the element-wise mean and standard deviation of all vectors in the training data set, respectively. This operation removes bias from the data due to a constant offset. Feature standardization does not change the dimensionality of the feature vector.
- (8)
- Classification: A Passive-Aggressive Algorithm, type-I (PA-1) [153] was used for classification. The PA-1 classifier is a linear binary online classifier, that is based on a maximum-margin hyperplane and can be applied to nonseparable problems. Preliminary investigations showed no significant differences in classification performance to a linear soft-margin Support Vector Machine (SVM). Similarly to a soft-margin SVM, the PA-1 provides a hyperparameter C to control regularization. C was optimized using a grid-search in the range . A stratified three-fold nested cross validation was used for model selection on the training data [154]. Subsequent to the classification, a threshold correction [155] was used to compensate the bias in the data related to class imbalance.
4.5. MRCP Processing
- (9)
- Spatial Filtering and Feature Generation: Since the MRCP is reflected by a temporal pattern in the data, time-domain features can be used for the detection of the MRCP. Accordingly, the axDAWN spatial filter [140] can be applied for the MRCP detection to reduce the number of channels to a single channel, i.e., creating a 20 dimensional feature vector.
- (10)
- Feature Standardization: The feature vectors were standardized using the approach discussed in Section 4.4 (7), leaving the dimension of the feature vector unchanged.
- (11)
- Classification: The 20 dimensional feature vector was classified using a Passive-Aggressive Algorithm, type-I [153]. Similar to the P300 detection, the regularization hyper-parameter C was optimized in the range using a 3-fold nested cross validation. Again, we observed no significant differences in classification performance to a linear soft-margin SVM in preliminary investigations and applied a threshold correction to compensate class imbalance as proposed in [155].
5. Experimental Evaluation
- A preliminary offline evaluation to test and evaluate the system regarding different parameters and combination of modalities and select the best parameters for the online evaluation.
- A subsequent online evaluation to verify that the system works in a real application.
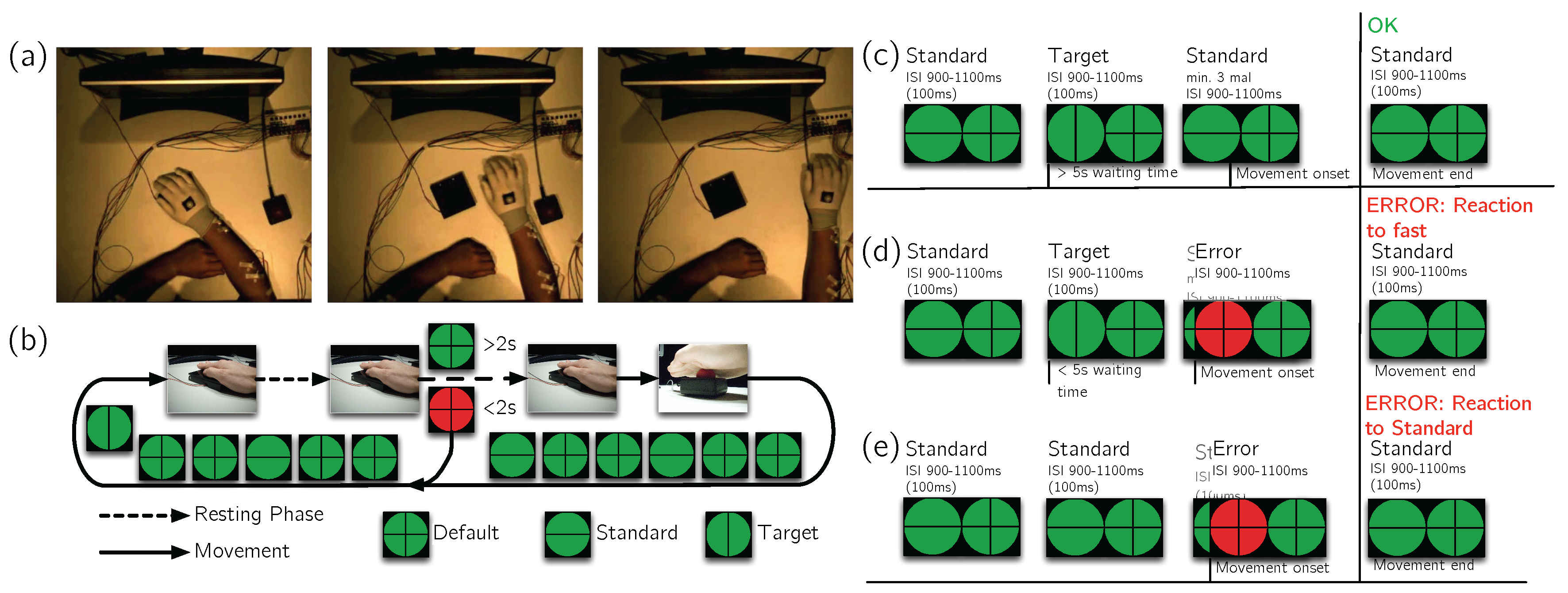
5.1. Experimental Setup
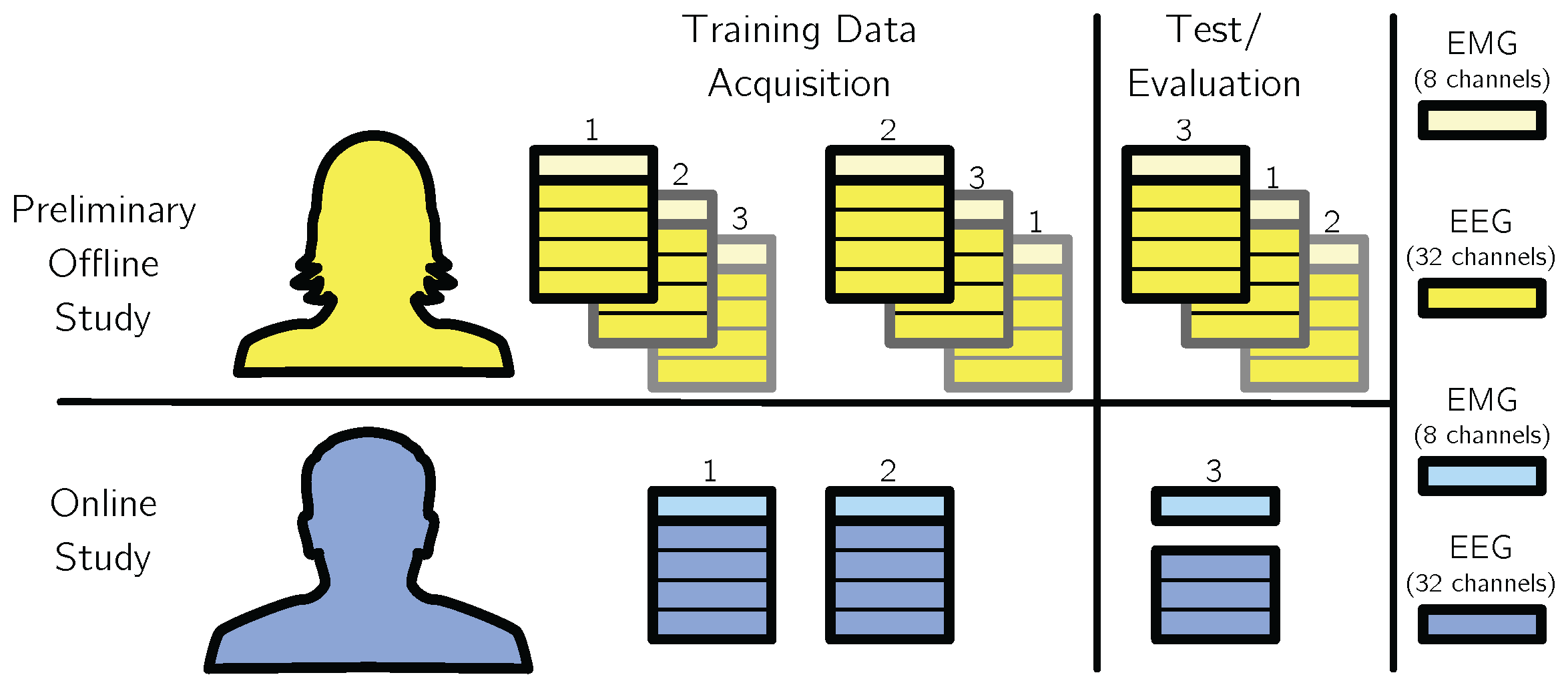
5.2. Data Acquisition
- Magma-Box Setup: In this setup, a Magma box with two Brain Products amplifier PCI cards were used, the first PCI card was used to acquire 128 channels of EEG, the second card to acquire 8 channels of EMG data. Electrodes I1, OI1h, OI2h, and I2 of the EEG were used for recording the electrooculogram, which is not considered in the following analysis. The Magma-Box setup was used for the offline evaluation and recording of the training data for the online evaluation.
- USB-Box Setup: In this setup, a Brain Products USB 2 Adapter was used to record the data. Since it is restricted to four BrainAmp DC amplifiers, three amplifiers were used for EEG data acquisition and one amplifier was used for EMG data acquisition.
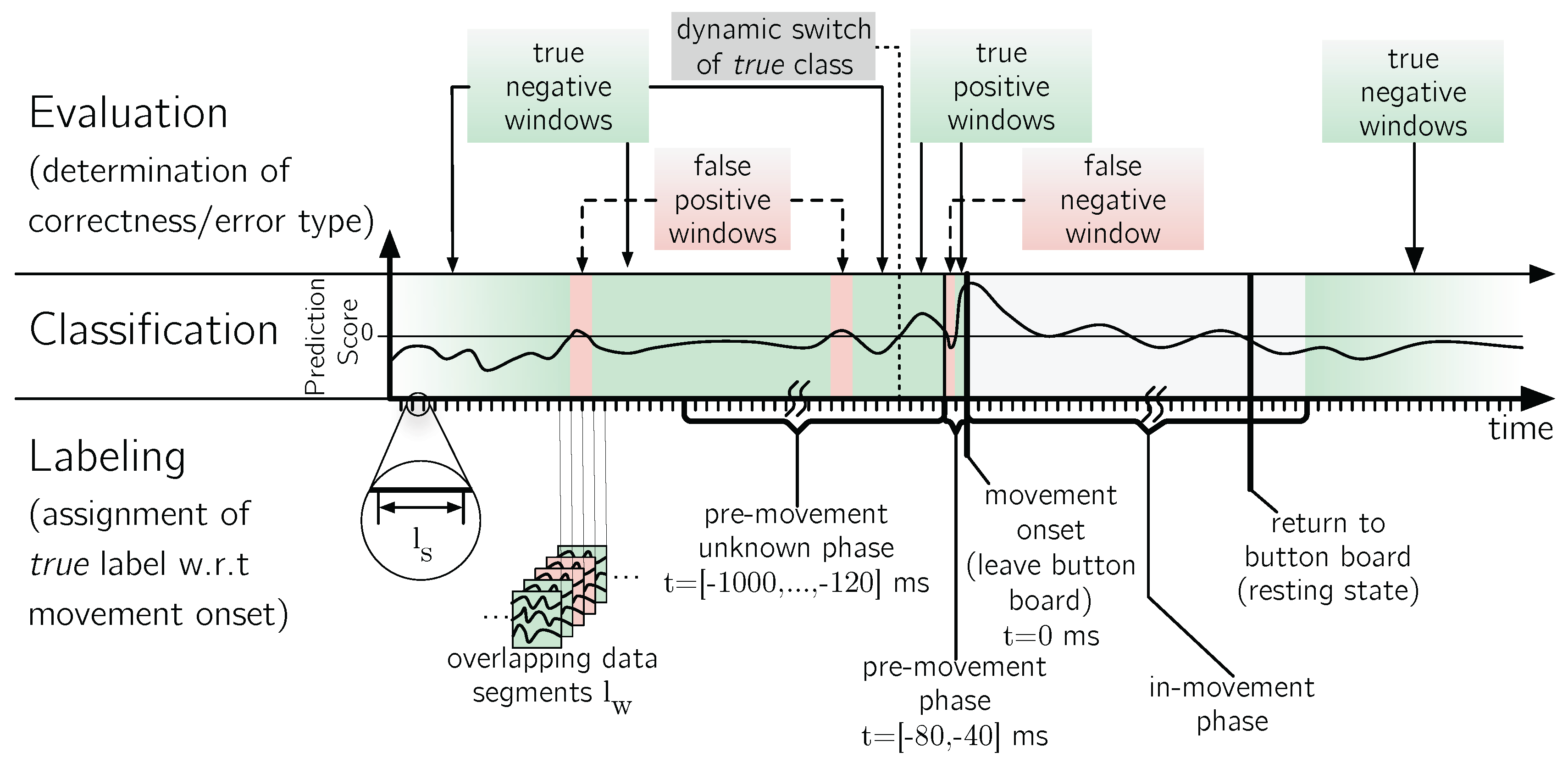
5.3. Evaluation Procedures
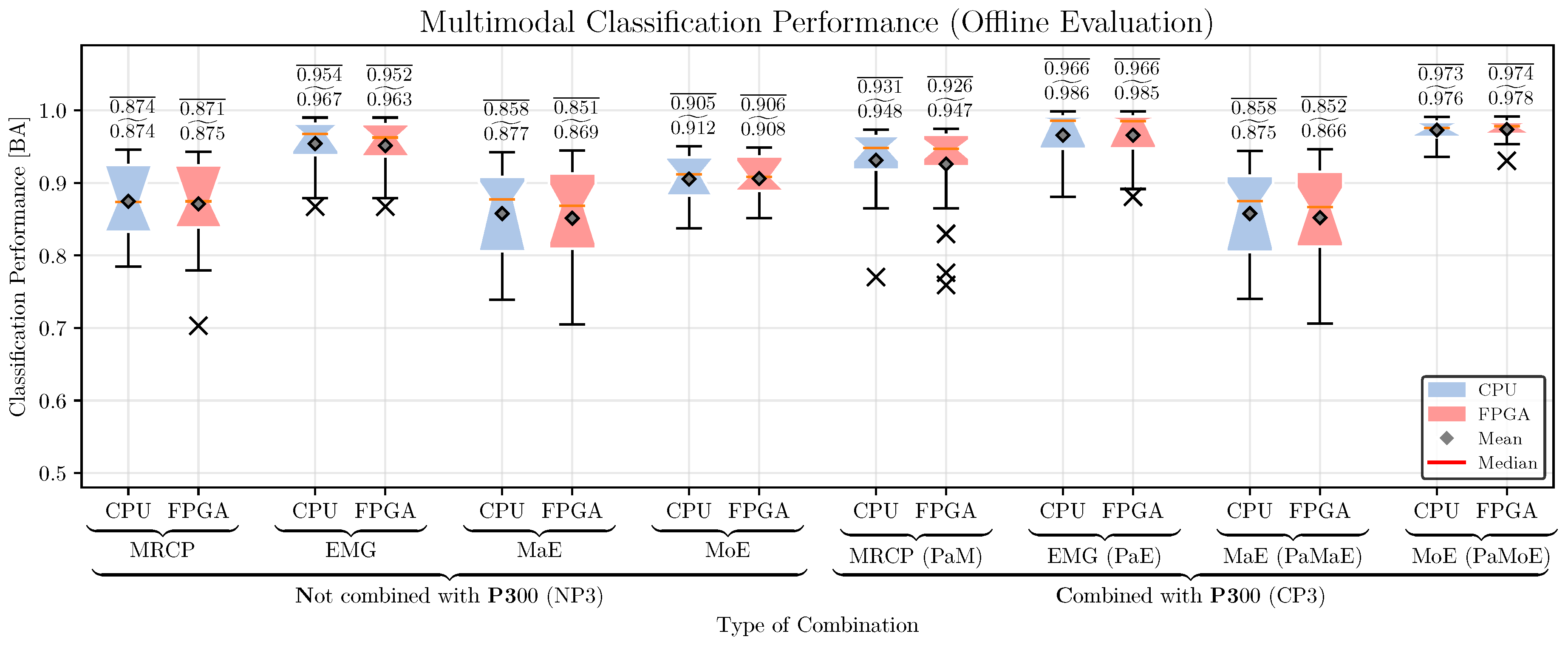
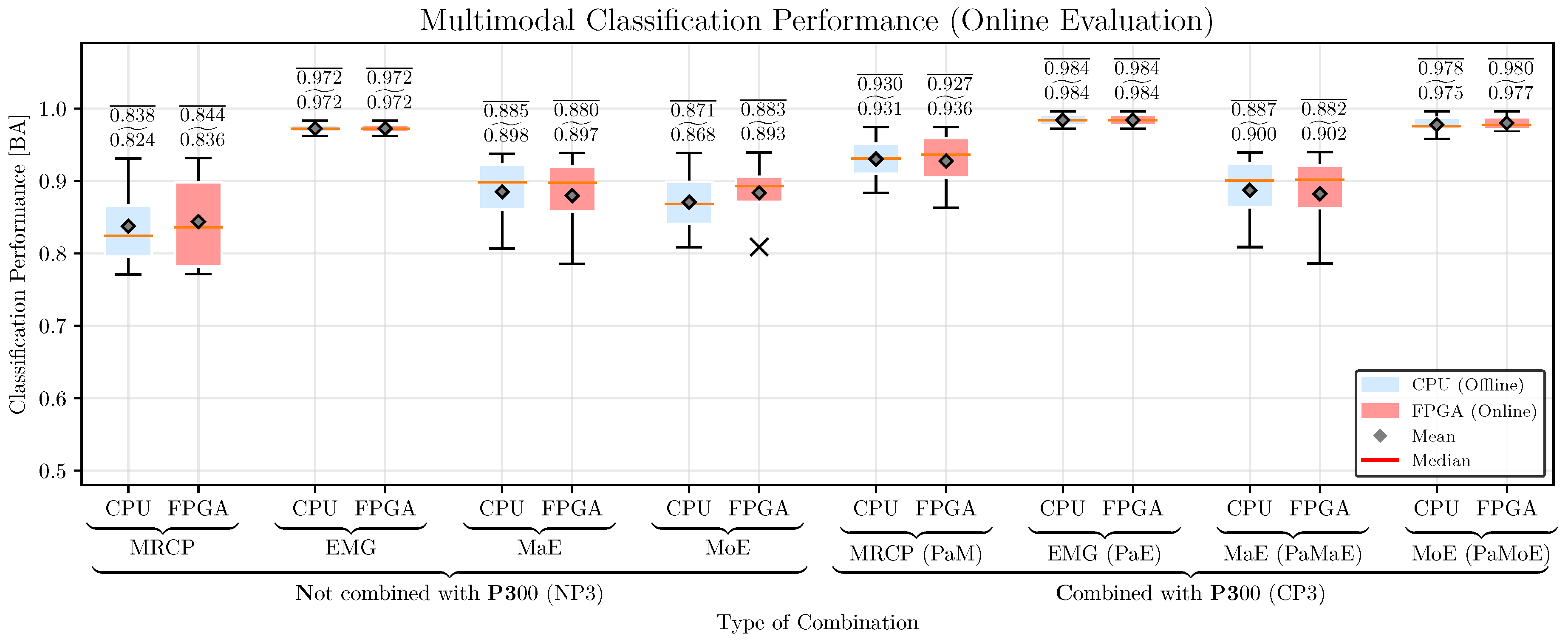
5.4. Classification Performance
5.5. Computing Performance: Reference Systems for Comparison
- A mobile processor-based system (the dual core ARM CPU of the ZC7030 PS, running at 1 GHz, 512 MB DDR SDRAM at 533 MHz), denoted as Mobile CPU-based Reference System (MRS) in the following.
- A standard desktop PC with an 8-core Intel(R) Core(TM) i7 CPU that was running at 3.07 GHz and a Linux Ubuntu operating system, version 14.4. denoted as Standard-PC Reference System (SRS) in the following.
5.6. Measurement of Power Consumption
6. Results and Discussion
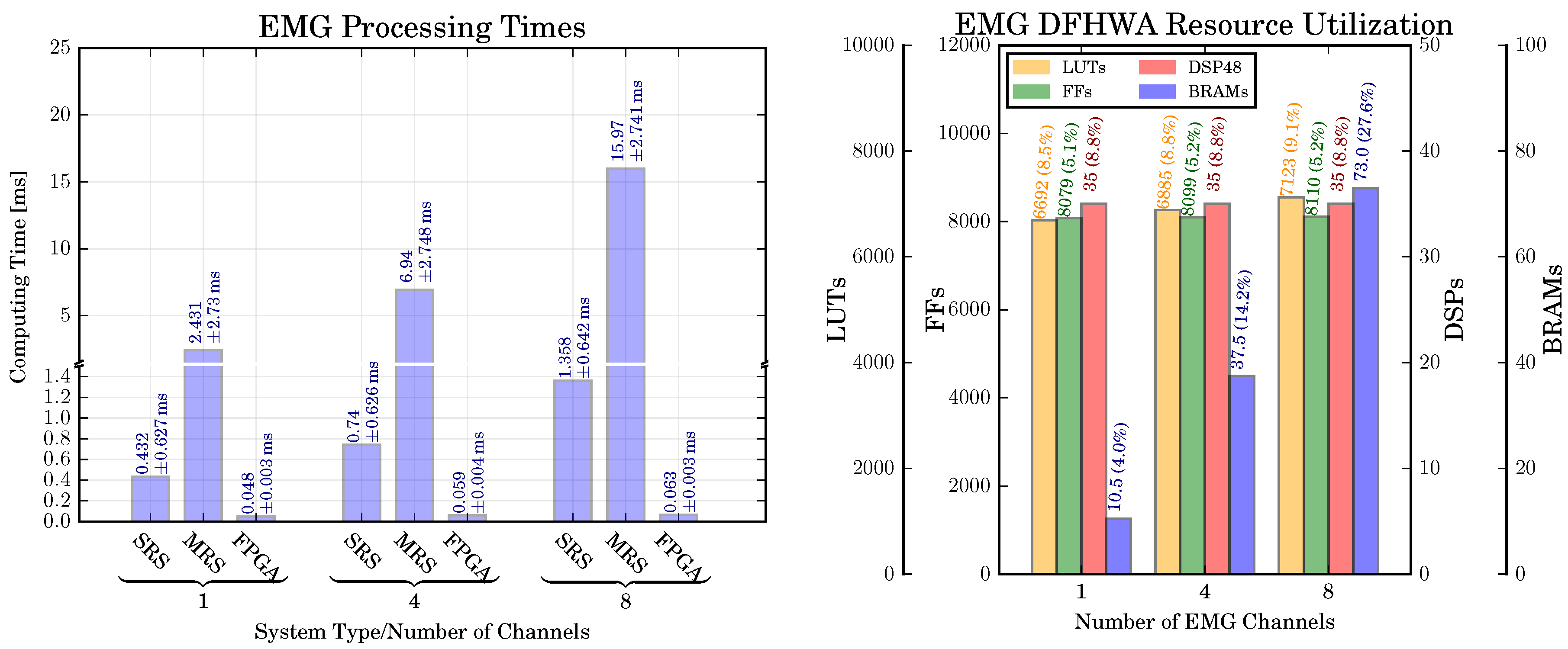
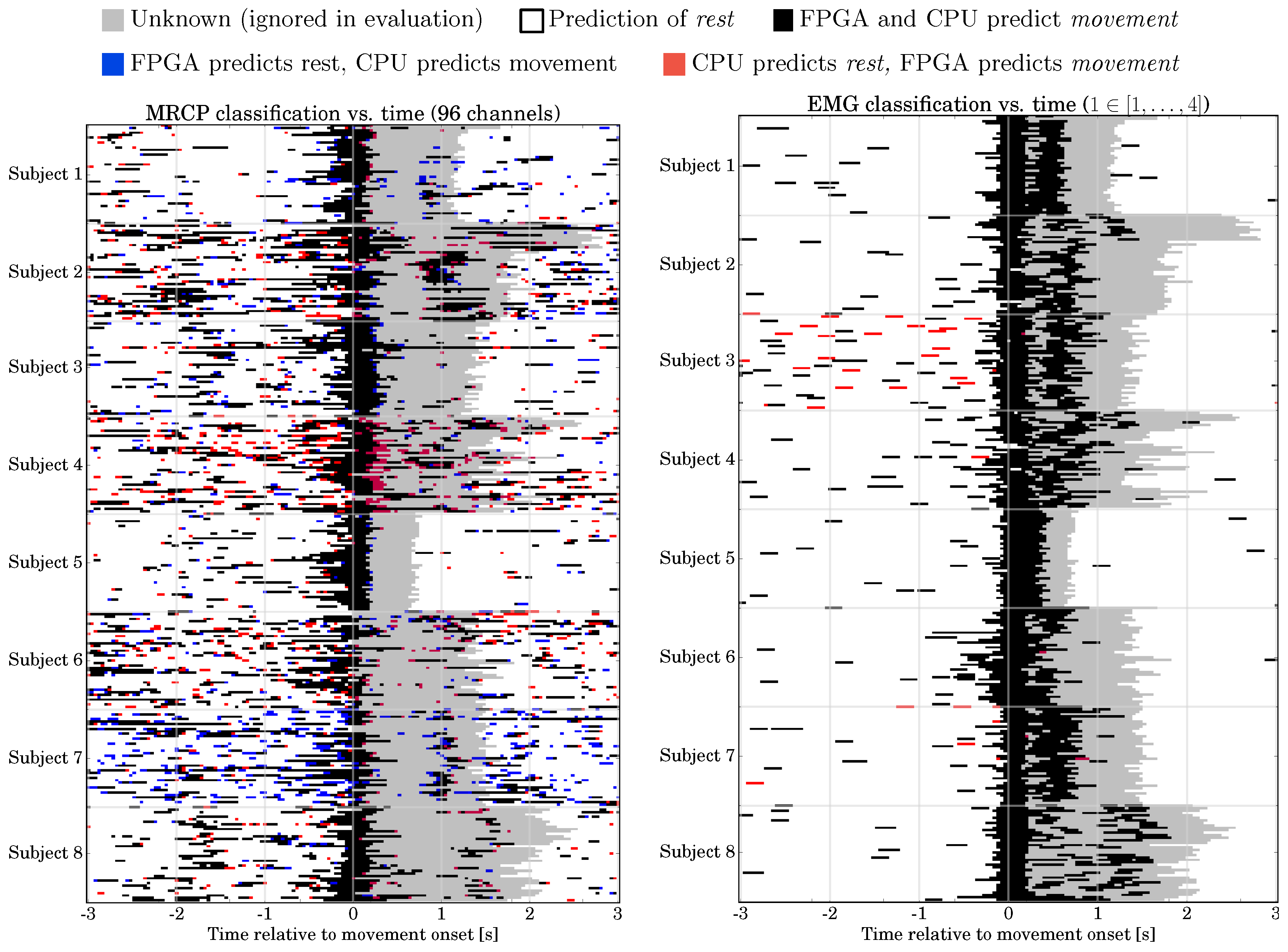
6.1. EMG-Based Movement Prediction: Classification Performance, Computing Time, Resource Utilization
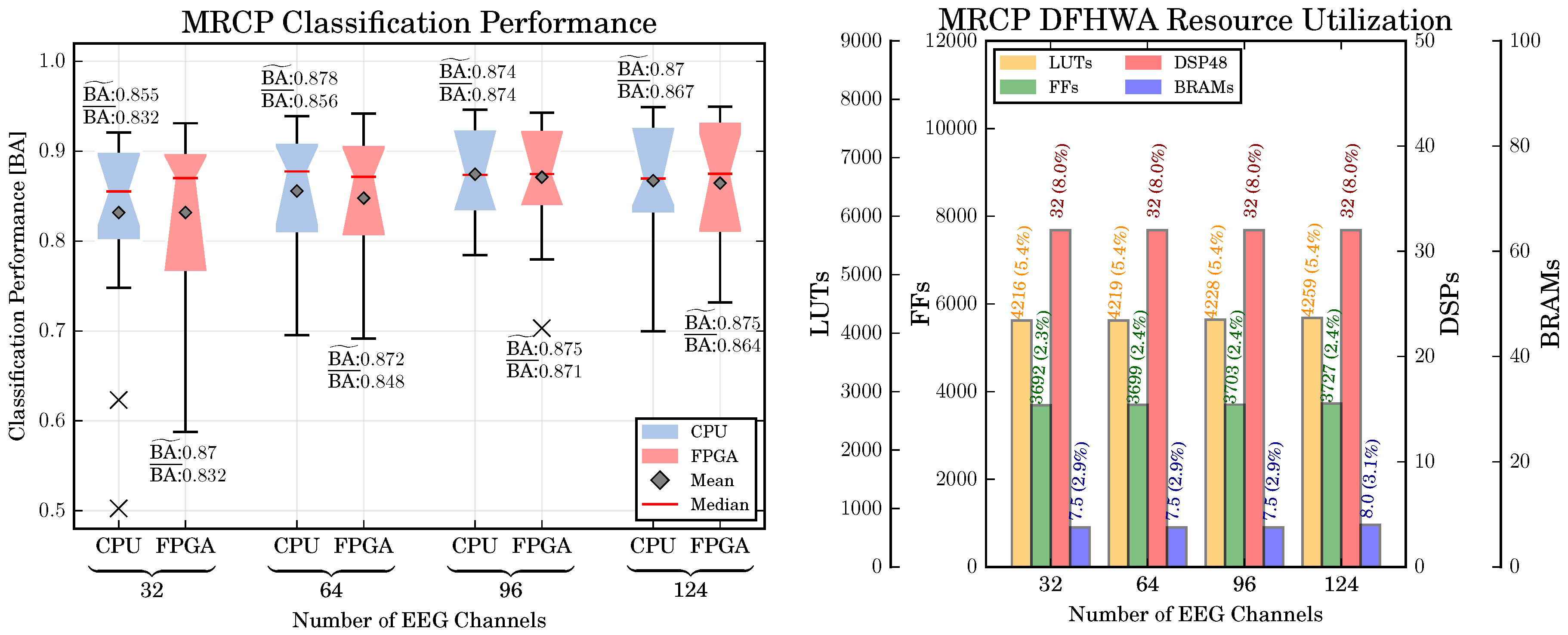
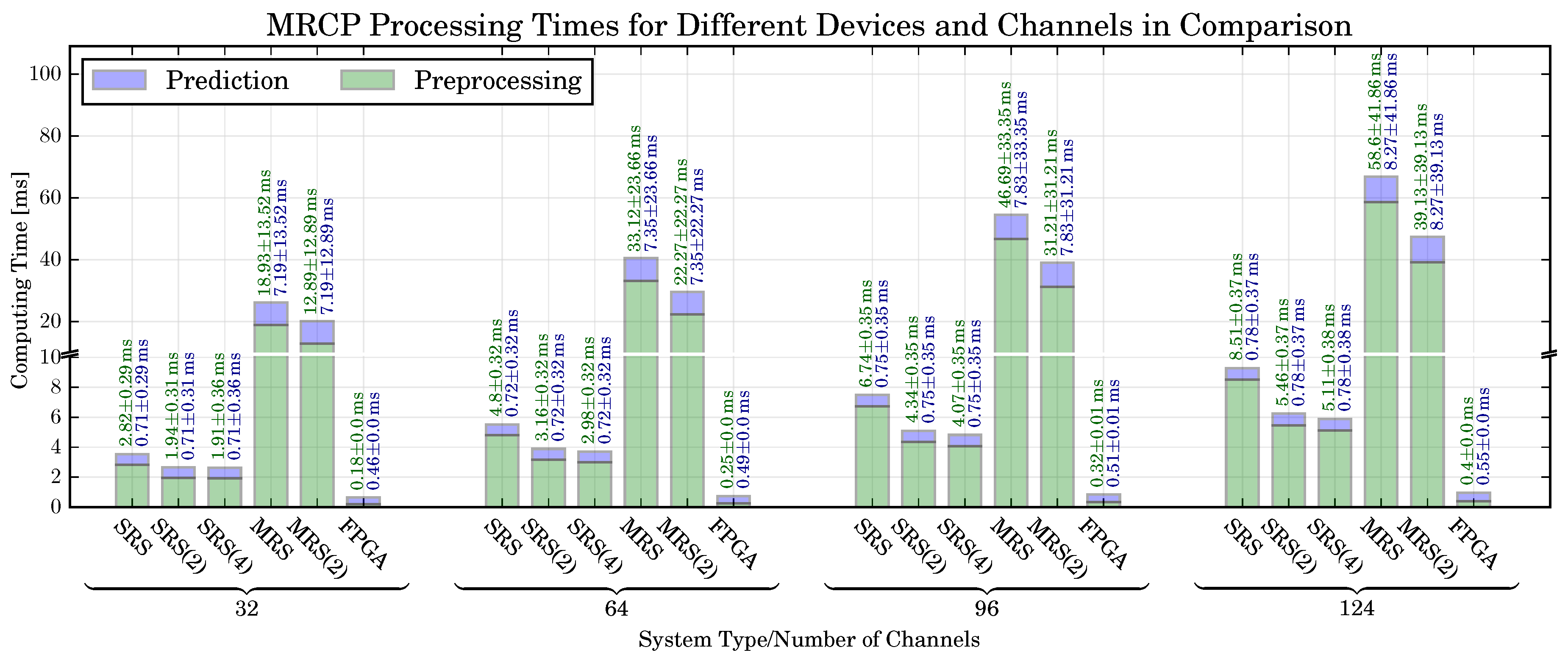
6.2. MRCP-Based Movement Prediction: Classification Performance, Computing Time, Resource Utilization
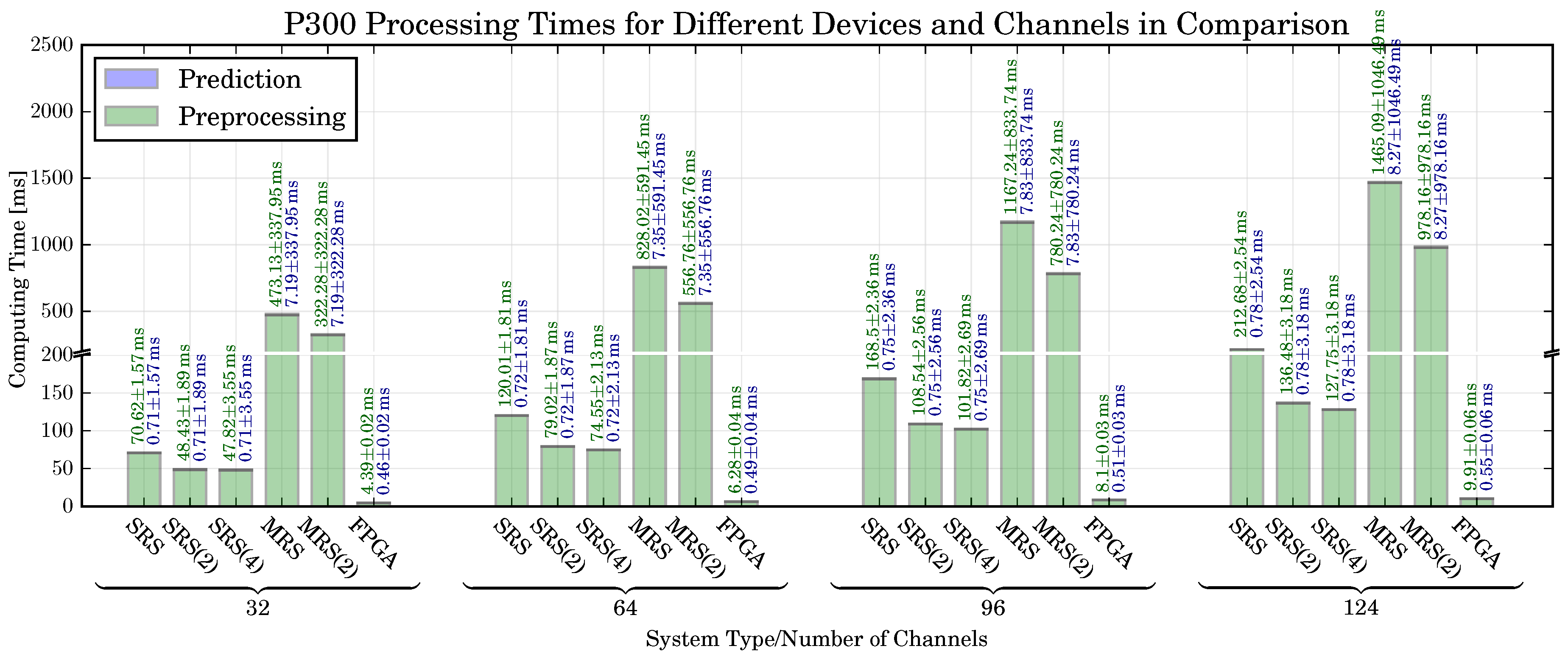
6.3. P300 Detection: Classification Performance, Computing Time, Resource Utilization
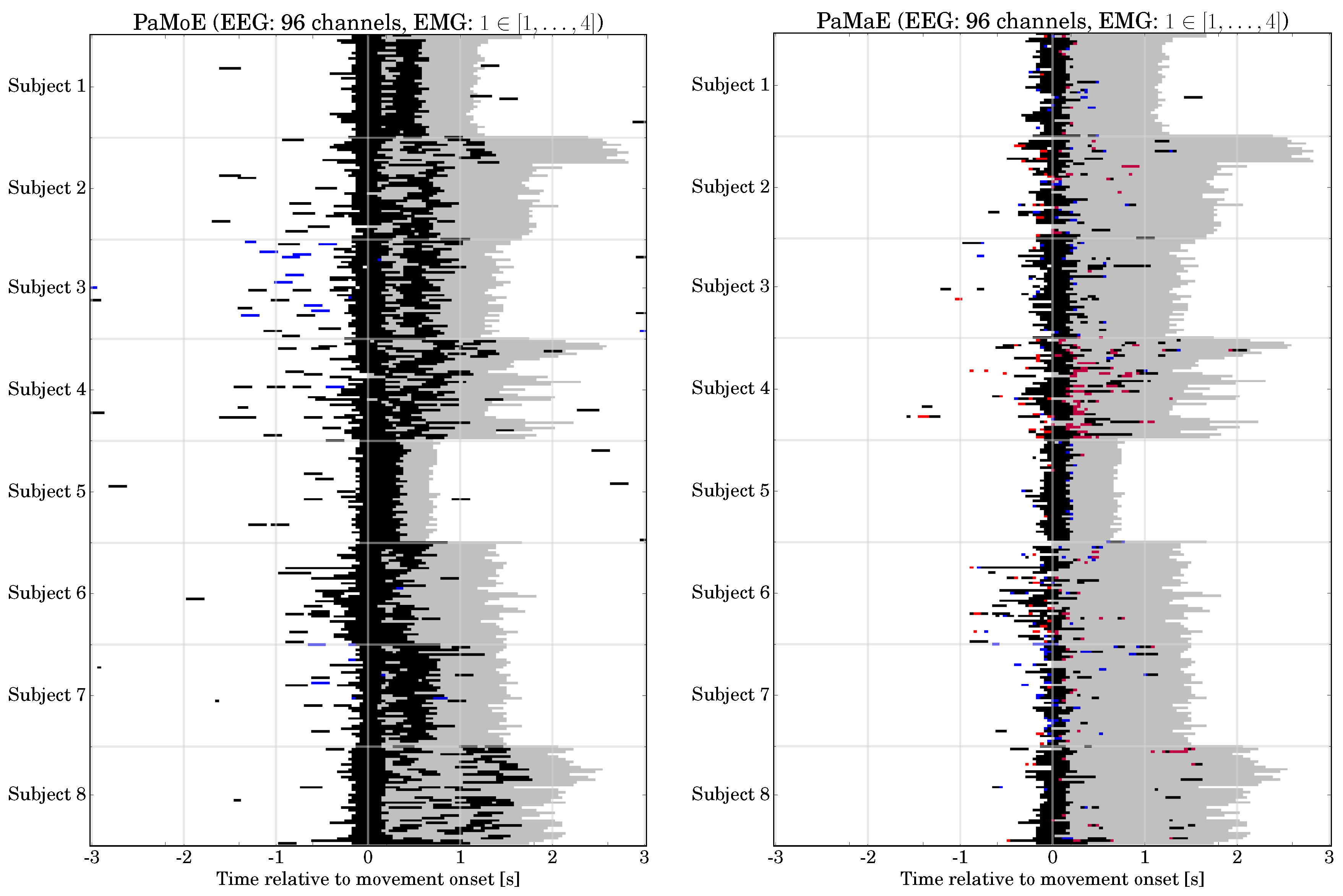
6.4. Hybrid Movement Prediction: Classification Performance, Prediction Time
6.5. Prediction Time
6.6. Power Consumption
6.7. Final Hybrid System: Verification in Application
6.8. Summary
6.9. Comparison to Previous Work
6.10. Limitations
7. Conclusions and Future Directions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ASIC | Application-Specific Integrated Circuit |
| BA | Balanced Accuracy |
| BCI | Brain-Computer Interface |
| BRAM | Block Random Access Memory |
| CPU | Central Processing Unit |
| DFHWA | Dataflow Hardware Accelerator |
| DMA | Direct Memory Access |
| DSP | Digital Signal Processing |
| EMG | Electromyography |
| EOG | Electrooculography |
| ERD/ERS | Event-Related Desynchronization/Synchronization |
| EEG | Electroencephalography |
| ERP | Event-Related Potential |
| FIFO | First-In, First-Out |
| FIR | Finite Impulse Response |
| FF | Flip Flop |
| FNR | False Negative Rate |
| FPGA | Field Programmable Gate Array |
| FPR | False Positive Rate |
| GPU | Graphics Processing Unit |
| IIR | Infinite Impulse Response |
| ISI | Inter-Stimulus Interval |
| IRQ | Interrupt ReQuest |
| LUT | Look-Up Table |
| MAC | Multiply ACcumulate |
| ML | Machine Learning |
| MRCP | Movement Related Cortical Potential |
| MRS | Mobile CPU-based Reference System |
| PE | Processing Element |
| PL | Programmable Logic |
| PS | Processing System |
| SDF | Synchronous Dataflow |
| SoC | System-on-Chip |
| SSVEP | Steady-State Visual Evoked Potential |
| SNR | Signal-to-Noise Ratio |
| SVM | Support Vector Machine |
| SRS | Standard-PC Reference System |
| TPR | True Positive Rate |
| TNR | True Negative Rate |
References
- Birbaumer, N.; Ghanayim, N.; Hinterberger, T.; Iversen, I.; Kotchoubey, B.; Kübler, A.; Perelmouter, J.; Taub, E.; Flor, H. A spelling device for the paralysed. Nature 1999, 398, 297–298. [Google Scholar] [CrossRef] [PubMed]
- Guger, C.; Harkam, W.; Hertnaes, C.; Pfurtscheller, G. Prosthetic control by an EEG-based brain-computer interface (BCI). In Proceedings of the 5th European Conference for the Advancement of Assistive Technology (AATE), Düsseldorf, Germany, 1–4 November 1999; pp. 590–595. [Google Scholar]
- Pfurtscheller, G.; Guger, C.; Müller, G.; Krausz, G.; Neuper, C. Brain oscillations control hand orthosis in a tetraplegic. Neurosci. Lett. 2000, 292, 211–214. [Google Scholar] [CrossRef]
- Wolpaw, J.; Birbaumer, N.; McFarland, D.; Pfurtscheller, G.; Vaughan, T. Brain-computer interfaces for communication and control. J. Clin. Neurophysiol. 2002, 113, 767–791. [Google Scholar] [CrossRef]
- Blankertz, B.; Dornhege, G.; Krauledat, M.; Müller, K.R.; Kunzmann, V.; Losch, F.; Curio, G. The Berlin brain-computer interface: EEG-based communication without subject training. IEEE Trans. Neural Syst. Rehabil. Eng. 2006, 14, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Birbaumer, N. Breaking the silence: Brain-computer interfaces (BCI) for communication and motor control. J. Psychophysiol. 2006, 43, 517–532. [Google Scholar] [CrossRef] [PubMed]
- Leeb, R.; Keinrath, C.; Friedman, D.; Guger, C.; Scherer, R.; Neuper, C.; Garau, M.; Antley, A.; Steed, A.; Slater, M.; et al. Walking by thinking: The brainwaves are crucial, not the muscles! Presence 2006, 15, 500–514. [Google Scholar] [CrossRef]
- Enzinger, C.; Ropele, S.; Fazekas, F.; Loitfelder, M.; Gorani, F.; Seifert, T.; Reiter, G.; Neuper, C.; Pfurtscheller, G.; Müller-Putz, G. Brain motor system function in a patient with complete spinal cord injury following extensive brain–computer interface training. Exp. Brain. Res. 2008, 190, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Pfurtscheller, G.; Müller-Putz, G.R.; Scherer, R.; Neuper, C. Rehabilitation with Brain-Computer Interface Systems. Computer 2008, 41, 58–65. [Google Scholar] [CrossRef]
- Kirchner, E.; Wöhrle, H.; Bergatt, C.; Kim, S.; Metzen, J.; Kirchner, F. Towards Operator Monitoring via Brain Reading—An EEG-based Approach for Space Applications. In Proceedings of the 10th International Symposium on Artificial Intelligence, Robotics and Automation in Space (iSAIRAS-10), Sapporo, Japan, 29 August–1 September 2010; ESA: Sapporo, Japan, 2010. [Google Scholar]
- Kirchner, E.; Kim, S.; Straube, S.; Seeland, A.; Wöhrle, H.; Krell, M.; Tabie, M.; Fahle, M. On the Applicability of Brain Reading for Predictive Human-Machine Interfaces in Robotics. PLoS ONE 2013, 8, e81732. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, E.; Albiez, J.; Seeland, A.; Jordan, M.; Kirchner, F. Towards Assistive Robotics for Home Rehabilitation. In Proceedings of the 6th International Conference on Biomedical Electronics and Devices (BIODEVICES-13), Barcelona, Spain, 11–14 February 2013; pp. 168–177. [Google Scholar]
- Iosa, M.; Morone, G.; Cherubini, A.; Paolucci, S. The Three Laws of Neurorobotics: A Review on What Neurorehabilitation Robots Should Do for Patients and Clinicians. J. Med. Biol. Eng. 2016, 36, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lum, P.S.; Burgar, C.G.; Shor, P.C.; Majmundar, M.; Van der Loos, M. Robot-assisted movement training compared with conventional therapy techniques for the rehabilitation of upper-limb motor function after stroke. Arch. Phys. Med. Rehabil. 2002, 83, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Krebs, H.I.; Volpe, B.T.; Aisen, M.L.; Hening, W.; Adamovich, S.; Poizner, H.; Subrahmanyan, K.; Hogan, N. Robotic Applications in Neuromotor Rehabilitation. Robotica 2003, 21, 3–11. [Google Scholar] [CrossRef]
- Kwakkel, G.; Kollen, B.J.; Krebs, H.I. Effects of robot-assisted therapy on upper limb recovery after stroke: A systematic review. Neurorehabil. Neural Repair 2008, 22, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Lo, A.C.; Guarino, P.D.; Richards, L.G.; Haselkorn, J.K.; Wittenberg, G.F.; Federman, D.G.; Ringer, R.J.; Wagner, T.H.; Krebs, H.I.; Volpe, B.T.; et al. Robot-Assisted Therapy for Long-Term Upper-Limb Impairment after Stroke. N. Engl. J. Med. 2010, 362, 1772–1783. [Google Scholar] [CrossRef] [PubMed]
- Díaz, I.; Gil, J.J.; Sánchez, E. Lower-limb robotic rehabilitation: Literature review and challenges. J. Robot. 2011, 2011, 759764. [Google Scholar] [CrossRef]
- Maciejasz, P.; Eschweiler, J.; Gerlach-Hahn, K.; Jansen-Troy, A.; Leonhardt, S. A survey on robotic devices for upper limb rehabilitation. J. NeuroEng. Rehabil. 2014, 11, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Novak, D.; Riener, R. A survey of sensor fusion methods in wearable robotics. Robot. Auton. Syst. 2015, 73, 155–170. [Google Scholar] [CrossRef]
- Lambercy, O.; Maggioni, S.; Lünenburger, L.; Gassert, R.; Bolliger, M. Robotic and wearable sensor technologies for measurements/clinical assessments. In Neurorehabilitation Technology; Springer International Publishing: Cham, Switzerland, 2016; pp. 183–207. [Google Scholar]
- Proietti, T.; Crocher, V.; Roby-Brami, A.; Jarrassé, N. Upper-Limb Robotic Exoskeletons for Neurorehabilitation: A Review on Control Strategies. IEEE Rev. Biomed. Eng. 2016, 9, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Hogan, N.; Krebs, H. Physically interactive robotic technology for neuromotor rehabilitation. Prog. Brain. Res. 2011, 192, 59–68. [Google Scholar] [PubMed]
- Benitez, L.; Tabie, M.; Will, N.; Schmidt, S.; Jordan, M.; Kirchner, E. Exoskeleton Technology in Rehabilitation: Towards an EMG-Based Orthosis System for Upper Limb Neuromotor Rehabilitation. J. Robot. 2013, 2013, 610589. [Google Scholar] [CrossRef]
- Kirchner, E.; Tabie, M.; Seeland, A. Multimodal Movement Prediction—Towards an Individual Assistance of Patients. PLoS ONE 2014, 9, e85060. [Google Scholar] [CrossRef] [PubMed]
- López-Larraz, E.; Montesano, L.; Gil-Agudo, Á.; Minguez, J. Continuous decoding of movement intention of upper limb self-initiated analytic movements from pre-movement EEG correlates. J. NeuroEng. Rehabil. 2014, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Riener, R.; Novak, D. Movement Onset Detection and Target Estimation for Robot-Aided Arm Training. Automatisierungstechnik 2015, 63, 286–298. [Google Scholar] [CrossRef]
- Lopez-Larraz, E.; Trincado-Alonso, F.; Rajasekaran, V.; Perez-Nombela, S.; del Ama, A.J.; Aranda, J.; Minguez, J.; Gil-Agudo, A.; Montesano, L. Control of an Ambulatory Exoskeleton with a Brain-Machine Interface for Spinal Cord Injury Gait Rehabilitation. Front. Neurosci. 2016, 10, 359. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, E.; Will, N.; Simnofske, M.; Vaca Benitez, L.V.; Bongardt, B.; Krell, M.; Kumar, S.; Mallwitz, M.; Seeland, A.; Tabie, M.; et al. Recupera-Reha: Exoskeleton Technology with Integrated Biosignal Analysis for Sensorimotor Rehabilitation. In Smart ASSIST; Elsevier: Hamburg, Germany, 2016; pp. 504–517. [Google Scholar]
- Soekadar, S.R.; Witkowski, M.; Gómez, C.; Opisso, E.; Medina, J.; Cortese, M.; Cempini, M.; Carrozza, M.C.; Cohen, L.G.; Birbaumer, N.; et al. Hybrid EEG/EOG-based brain/neural hand exoskeleton restores fully independent daily living activities after quadriplegia. Sci. Robot. 2016, 5. [Google Scholar] [CrossRef]
- Schaechter, J.D. Motor rehabilitation and brain plasticity after hemiparetic stroke. Prog. Neurobiol. 2004, 73, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Dobkin, B. Brain-computer interface technology as a tool to augment plasticity and outcomes for neurological rehabilitation. J. Physiol. 2007, 579, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Ang, K.; Guan, C.; Chua, K.; Ang, B.; Kuah, C.; Wang, C.; Phua, K.; Chin, Z.; Zhang, H. A clinical study of motor imagery-based brain-computer interface for upper limb robotic rehabilitation. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Minneapolis, MN, USA, 3–6 September 2009; pp. 5981–5984. [Google Scholar]
- Daly, J.; Wolpaw, J. Brain-computer interfaces in neurological rehabilitation. Lancet Neurol. 2008, 7, 1032–1043. [Google Scholar] [CrossRef]
- Ang, K.; Chua, K.; Phua, K.; Wang, C.; Chin, Z.; Kuah, C.; Low, W.; Guan, C. A randomized controlled trial of EEG-based motor imagery brain-computer interface robotic rehabilitation for stroke. Clin. EEG Neurosci. 2014, 46, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Van Dokkum, L.; Ward, T.; Laffont, I. Brain computer interfaces for neurorehabilitation—Its current status as a rehabilitation strategy post-stroke. Ann. Phys. Rehabil. Med. 2015, 58, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Makeig, S.; Gramann, K.; Jung, T.P.; Sejnowski, T.J.; Poizner, H. Linking brain, mind and behavior. Int. J. Psychophysiol. 2009, 73, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Kranczioch, C.; Zich, C.; Schierholz, I.; Sterr, A. Mobile EEG and its potential to promote the theory and application of imagery-based motor rehabilitation. Int. J. Psychophysiol. 2014, 91, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Edlinger, G.; Guger, C. Laboratory PC and mobile pocket PC brain-computer interface architectures. In Proceedings of the 27th International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Shanghai, China, 17–18 January 2006; Volume 5, pp. 5347–5350. [Google Scholar]
- Xilinx Inc. UG585 Zynq-7000 All Programmable SoC Technical Reference Manual, 1.10 ed.; Xilinx Inc.: San Jose, CA, USA, 2015. [Google Scholar]
- Pfurtscheller, G.; Allison, B.; Bauernfeind, G.; Brunner, C.; Solis Escalante, T.; Scherer, R.; Zander, T.; Mueller-Putz, G.; Neuper, C.; Birbaumer, N. The hybrid BCI. Front. Neurosci. 2010, 4, 3. [Google Scholar] [CrossRef] [PubMed]
- Lalitharatne, T.; Teramoto, K.; Hayashi, Y.; Kiguchi, K. Towards hybrid EEG-EMG-based control approaches to be used in bio-robotics applications: Current status, challenges and future directions. Paladyn 2013, 4, 147–154. [Google Scholar] [CrossRef]
- Villa-Parra, A.; Broche, L.; Delisle-Rodríguez, D.; Sagaró, R.; Bastos, T.; Frizera-Neto, A. Design of active orthoses for a robotic gait rehabilitation system. Front. Mech. Eng. 2015, 10, 242–254. [Google Scholar] [CrossRef]
- Grimm, F.; Walter, A.; Spüler, M.; Naros, G.; Rosenstiel, W.; Gharabaghi, A. Hybrid Neuroprosthesis for the Upper Limb: Combining Brain-Controlled Neuromuscular Stimulation with a Multi-Joint Arm Exoskeleton. Front. Neurosci. 2016, 10, 367. [Google Scholar] [CrossRef] [PubMed]
- Tabie, M.; Wöhrle, H.; Kirchner, E.A. Runtime Calibration of Online EEG-based Movement Prediction using EMG Signals. In Proceedings of the International Conference on Bio-inspired Systems and Signal Processing (BIOSIGNALS), Angers, France, 3–6 March 2014; pp. 284–288. [Google Scholar]
- Seeland, A.; Tabie, M.; Kim, S.; Kirchner, F.; Kirchner, E.A. Adaptive multimodal biosignal control for exoskeleton supported stroke rehabilitation. In Proceedings of the IEEE International Conference on Systems, Man, and Cybernetics (SMC-2017), Banff, AB, Canada, 1–4 October 2017. [Google Scholar]
- Kirchner, E.A.; Kim, S.K.; Tabie, M.; Wöhrle, H.; Maurus, M.; Kirchner, F. An Intelligent Man-Machine Interface-Multi-Robot Control Adapted for Task Engagement Based on Single-Trial Detectability of P300. Front. Hum. Neurosci. 2016, 10, 291. [Google Scholar] [CrossRef] [PubMed]
- Bai, O.; Rathi, V.; Lin, P.; Huang, D.; Battapady, H.; Fei, D.; Schneider, L.; Houdayer, E.; Chen, X.; Hallett, M. Prediction of human voluntary movement before it occurs. J. Clin. Neurophysiol. 2011, 122, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, J.; Serrano, J.; del Castillo, M.; Barrios, L.; Gallego, J.; Rocon, E. An EEG-Based Design for the Online Detection of Movement Intention. In Advances in Computational Intelligence; Cabestany, J., Rojas, I., Joya, G., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 370–377. [Google Scholar]
- Lew, E.; Chavarriaga, R.; Silvoni, S.; Millán, J. Detection of self-paced reaching movement intention from EEG signals. Front. Neuroeng. 2012, 5. [Google Scholar] [CrossRef] [PubMed]
- Pfurtscheller, G.; Neuper, C. Motor imagery activates primary sensorimotor area in humans. Neurosci. Lett. 1997, 239, 65–68. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Da Silva, F. Event-related EEG/MEG synchronization and desynchronization: Basic principles. J. Clin. Neurophysiol. 1999, 110, 1842–1857. [Google Scholar] [CrossRef]
- Stančák, A.; Feige, B.; Lücking, C.; Kristeva-Feige, R. Oscillatory cortical activity and movement-related potentials in proximal and distal movements. J. Clin. Neurophysiol. 2000, 111, 636–650. [Google Scholar] [CrossRef]
- Paradiso, G.; Cunic, D.; Saint-Cyr, J.; Hoque, T.; Lozano, A.; Lang, A.; Chen, R. Involvement of human thalamus in the preparation of self-paced movement. Brain 2004, 127, 2717–2731. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, H.; Hallett, M. What is the Bereitschaftspotential? J. Clin. Neurophysiol. 2006, 117, 2341–2356. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, E.; Metzen, J.H.; Duchrow, T.; Kim, S.; Kirchner, F. Assisting Telemanipulation Operators via Real-Time Brain Reading. In Proceedings of the Machine Learning in Real-time Applications Workshop, Paderborn, Germany, 15–18 September 2009. [Google Scholar]
- Woehrle, H.; Kirchner, E. Online Detection of P300 related Target Recognition Processes During a Demanding Teleoperation Task. In Proceedings of the International Conference on Physiological Computing Systems, (PhyCS 2014), Lissabon, Portugal, 7–9 January 2014; ScitePress: Setúbal, Portugal, 2014; pp. 13–19. [Google Scholar]
- Seeland, A.; Woehrle, H.; Straube, S.; Kirchner, E. Online Movement Prediction in a Robotic Application Scenario. In Proceedings of the 6th International IEEE EMBS Conference on Neural Engineering (NER), San Diego, CA, USA, 6–8 November 2013; pp. 41–44. [Google Scholar]
- Müller, K.R.; Krauledat, M.; Dornhege, G.; Curio, G.; Blankertz, B. Machine learning techniques for brain-computer interfaces. Biomed. Tech. 2004, 49, 11–22. [Google Scholar]
- Lotte, F.; Congedo, M.; Lécuyer, A.; Lamarche, F.; Arnaldi, B. A review of classification algorithms for EEG-based brain-computer interfaces. J. Neural Eng. 2007, 4, R1. [Google Scholar] [CrossRef] [PubMed]
- Krusienski, D.; Grosse-Wentrup, M.; Galain, F.; Coyle, D.; Miller, K.; Forney, E.; Anderson, C. Critical issues in state-of-the-art brain computer interface signal processing. J. Neural Eng. 2011, 8, 025002. [Google Scholar] [CrossRef] [PubMed]
- Kleissen, R.; Buurke, J.; Harlaar, J.; Zilvold, G. Electromyography in the biomechanical analysis of human movement and its clinical application. Gait Posture 1998, 8, 143–158. [Google Scholar] [CrossRef]
- Reaz, M.B.I.; Hussain, M.S.; Mohd-Yasin, F. Techniques of EMG signal analysis: Detection, processing, classification and applications. Biol. Proced. Online 2006, 8, 11–35. [Google Scholar] [CrossRef] [PubMed]
- Tabie, M.; Kirchner, E. EMG Onset Detection-Comparison of different methods for a movement prediction task based on EMG. In Proceedings of the 6th International Conference on Bio-inspired Systems and Signal Processing (BIOSIGNALS-13), Barcelona, Spain, 11–14 February 2013; pp. 242–247. [Google Scholar]
- Fleischer, C.; Reinicke, C.; Hommel, G. Predicting the intended motion with EMG signals for an exoskeleton orthosis controller. In Proceedings of the IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Edmonton, AB, Canada, 2–6 August 2005; pp. 2029–2034. [Google Scholar]
- Suberbiola, A.; Zulueta, E.; Lopez-Guede, J.M.; Etxeberria-Agiriano, I.; Van Caesbroeck, B. Arm Orthosis/Prosthesis Control Based on Surface EMG Signal Extraction. In Hybrid Artificial Intelligent Systems; Springer: Berlin/Heidelberg, Germany, 2013; pp. 510–519. [Google Scholar]
- Loconsole, C.; Dettori, S.; Frisoli, A.; Avizzano, C.A.; Bergamasco, M. An EMG-based approach for on-line predicted torque control in robotic-assisted rehabilitation. In Proceedings of the 2014 IEEE Haptics Symposium (HAPTICS), Houston, TX, USA, 23–26 February 2014; pp. 181–186. [Google Scholar]
- Johnson, S.; Sprehn, G.; Saykin, A. Intact motor imagery in chronic upper limb hemiplegics: Evidence for activity-independent action representations. J. Cogn. Neurosci. 2002, 14, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Ang, K.; Guan, C.; Chua, K.; Ang, B.; Kuah, C.; Wang, C.; Puah, K.; Chin, Z.; Zhang, H. A Large Clinical Study on the Ability of Stroke Patients to Use an EEG-Based Motor Imagery Brain-Computer Interface. Clin. EEG Neurosci. 2011, 42, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.; Lin, C.; McDowell, K.; Wickenden, A.E.; Gramann, K.; Jung, T.; Ko, L.; Chang, J. Biosensor technologies for augmented brain-computer interfaces in the next decades. Proc. IEEE 2012, 100, 1553–1566. [Google Scholar] [CrossRef]
- McFarland, D.J.; Wolpaw, J.R. Brain-Computer Interface Operation of Robotic and Prosthetic Devices. Computer 2008, 41, 52–56. [Google Scholar] [CrossRef]
- Rebsamen, B.; Burdet, E.; Zeng, Q.; Zhang, H.; Ang, M.; Teo, C.L.; Guan, C.; Laugier, C. Hybrid P300 and Mu-beta Brain Computer Interface to Operate a Brain Controlled Wheelchair. In Proceedings of the 2nd International Convention on Rehabilitation Engineering & Assistive Technology, Bangkok, Thailand, 13–15 May 2008; pp. 51–55. [Google Scholar]
- Gancet, J.; Ilzkovitz, M.; Motard, E.; Nevatia, Y.; Letier, P.; de Weerdt, D.; Cheron, G.; Hoellinger, T.; Seetharaman, K.; Petieau, M.; et al. MINDWALKER: Going one step further with assistive lower limbs exoskeleton for SCI condition subjects. In Proceedings of the 4th IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics (BioRob), Rome, Italy, 24–27 June 2012; pp. 1794–1800. [Google Scholar]
- Do, A.; Wang, P.; King, C.; Chun, S.; Nenadic, Z. Brain-computer interface controlled robotic gait orthosis. J. NeuroEng. Rehabil. 2013, 10, 111. [Google Scholar] [CrossRef] [PubMed]
- Looned, R.; Webb, J.; Xiao, Z.; Menon, C. Assisting drinking with an affordable BCI-controlled wearable robot and electrical stimulation: A preliminary investigation. J. NeuroEng. Rehabil. 2014, 11, 51. [Google Scholar] [CrossRef] [PubMed]
- Allison, B.; Leeb, R.; Brunner, C.; Müller-Putz, G.; Bauernfeind, G.; Kelly, J.; Neuper, C. Toward smarter BCIs: Extending BCIs through hybridization and intelligent control. J. Neural. Eng. 2012, 9, 013001. [Google Scholar] [CrossRef] [PubMed]
- Amiri, S.; Fazel-Rezai, R.; Asadpour, V. A review of hybrid brain-computer interface systems. Adv. Hum. Comput. Interact. 2013, 2013, 1. [Google Scholar] [CrossRef]
- Lotte, F.; Guan, C. An efficient P300-based brain-computer interface with minimal calibration time. In Proceedings of the Assistive Machine Learning for People with Disabilities symposium (NIPS’09 Symposium), Vancouver, BC, Canada, 12 December 2009. [Google Scholar]
- Wöhrle, H.; Kirchner, E. Online Classifier Adaptation for the Detection of P300 Target Recognition Processes in a Complex Teleoperation Scenario. In Physiological Computing Systems; da Silva, H., Holzinger, A., Fairclough, S., Majoe, D., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 105–118. [Google Scholar]
- Su, Y.; Qi, Y.; Luo, J.; Wu, B.; Yang, F.; Li, Y.; Zhuang, Y.; Zheng, X.; Chen, W. A hybrid brain-computer interface control strategy in a virtual environment. J. Zhejiang Univ. 2011, 12, 351–361. [Google Scholar] [CrossRef]
- Riechmann, H.; Hachmeister, N.; Ritter, H.; Finke, A. Asynchronous, parallel on-line classification of P300 and ERD for an efficient hybrid BCI. In Proceedings of the 5th International IEEE/EMBS Conference on Neural Engineering (NER), Cancun, Mexico, 27 April–1 May 2011; pp. 412–415. [Google Scholar]
- Leeb, R.; Sagha, H.; Chavarriaga, R.; del R Millán, J. A hybrid brain–computer interface based on the fusion of electroencephalographic and electromyographic activities. J. Neural Eng. 2011, 8, 025011. [Google Scholar] [CrossRef] [PubMed]
- Kiguchi, K.; Hayashi, Y. Motion Estimation Based on EMG and EEG Signals to Control Wearable Robots. In Proceedings of the IEEE International Conference on Systems, Man, and Cybernetics, Manchester, UK, 13–16 October 2013; pp. 4213–4218. [Google Scholar]
- Novak, D.; Omlin, X.; Leins-Hess, R.; Riener, R. Predicting targets of human reaching motions using different sensing technologies. IEEE Trans. Biomed. Eng. 2013, 60, 2645–2654. [Google Scholar] [CrossRef] [PubMed]
- De Venuto, D.; Annese, V.F.; de Tommaso, M.; Vecchio, E.; Sangiovanni Vincentelli, A.L. Combining EEG and EMG Signals in a Wireless System for Preventing Fall in Neurodegenerative Diseases. In Ambient Assisted Living: Italian Forum 2014; Springer International Publishing: Cham, Switzerland, 2015; pp. 317–327. [Google Scholar]
- Kawase, T.; Sakurada, T.; Koike, Y.; Kansaku, K. A hybrid BMI-based exoskeleton for paresis: EMG control for assisting arm movements. J. Neural Eng. 2017, 14, 016015. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Samuel, O.; Zhang, X.; Wang, H.; Fang, P.; Li, G. A motion-classification strategy based on sEMG-EEG signal combination for upper-limb amputees. J. NeuroEng. Rehabil. 2017, 14, 2. [Google Scholar] [CrossRef] [PubMed]
- Corbett, E.A.; Sachs, N.A.; Körding, K.P.; Perreault, E.J. Multimodal decoding and congruent sensory information enhance reaching performance in subjects with cervical spinal cord injury. Front. Neurosci. 2014, 8, 123. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, G.; Bifulco, P.; Calvo, R.; Cesarelli, M.; Jin, C.; Schaik, A. A mobile EEG system with dry electrodes. In Proceedings of the IEEE Biomedical Circuits and Systems Conference, Baltimore, MD, USA, 20–22 November 2008; pp. 273–276. [Google Scholar]
- Liao, L.D.; Wang, I.J.; Chen, S.F.; Chang, J.Y.; Lin, C.T. Design, fabrication and experimental validation of a novel dry-contact sensor for measuring electroencephalography signals without skin preparation. Sensors 2011, 11, 5819–5834. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.T.; Liao, L.D.; Liu, Y.H.; Wang, I.J.; Lin, B.S.; Chang, J.Y. Novel dry polymer foam electrodes for long-term EEG measurement. IEEE Trans. Biomed. Eng. 2011, 58, 1200–1207. [Google Scholar] [PubMed]
- Vos, M.D.; Kroesen, M.; Emkes, R.; Debener, S. P300 speller BCI with a mobile EEG system: Comparison to a traditional amplifier. J. Neural Eng. 2014, 11, 036008. [Google Scholar] [CrossRef] [PubMed]
- Ehinger, B.; Fischer, P.; Gert, A.; Kaufhold, L.; Weber, F.; Pipa, G.; König, P. Kinesthetic and vestibular information modulate alpha activity during spatial navigation: A mobile EEG study. Front. Hum. Neurosci. 2014, 8, 71. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.C.; Chien, T.Y.; Chen, Y.C.; Tsai, S.H.; Fang, W.C.; Lin, B.S. A Wearable Channel Selection-Based Brain-Computer Interface for Motor Imagery Detection. Sensors 2016, 16, 213. [Google Scholar] [CrossRef] [PubMed]
- Matthews, R.; Turner, P.; McDonald, N.J.; Ermolaev, K.; Mc Manus, T.; Shelby, R.A.; Steindorf, M. Real time workload classification from an ambulatory wireless EEG system using hybrid EEG electrodes. In Proceedings of the 2008 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 20–25 August 2008; pp. 5871–5875. [Google Scholar]
- Lin, C.T.; Chen, Y.C.; Huang, T.Y.; Chiu, T.T.; Ko, L.W.; Liang, S.F.; Hsieh, H.Y.; Hsu, S.H.; Duann, J.R. Development of Wireless Brain Computer Interface With Embedded Multitask Scheduling and its Application on Real-Time Driver’s Drowsiness Detection and Warning. IEEE Trans. Biomed. Eng. 2008, 55, 1582–1591. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.T.; Ko, L.W.; Chiou, J.C.; Duann, J.R.; Huang, R.S.; Liang, S.F.; Chiu, T.W.; Jung, T.P. Noninvasive Neural Prostheses Using Mobile and Wireless EEG. Proc. IEEE 2008, 96, 1167–1183. [Google Scholar]
- Riera, A.; Dunne, S.; Cester, I.; Ruffini, G. STARFAST: A wireless wearable EEG/ECG biometric system based on the ENOBIO sensor. In Proceedings of the 6th International Workshop on Wearable Micro- and Nanosystems for Personalised Health, Oslo, Norway, 24–26 June 2008. [Google Scholar]
- Lin, C.T.; Ko, L.W.; Chang, M.H.; Duann, J.R.; Chen, J.Y.; Su, T.P.; Jung, T.P. Review of wireless and wearable electroencephalogram systems and brain-computer interfaces—A mini-review. Gerontology 2009, 56, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.T.; Ko, L.W.; Chang, C.J.; Wang, Y.T.; Chung, C.H.; Yang, F.S.; Duann, J.R.; Jung, T.P.; Chiou, J.C. Wearable and wireless brain-computer interface and its applications. In Proceedings of the International Conference on Foundations of Augmented Cognition, San Diego, CA, USA, 9–24 July 2009; pp. 741–748. [Google Scholar]
- Brown, L.; van de Molengraft, J.; Yazicioglu, R.F.; Torfs, T.; Penders, J.; Van Hoof, C. A low-power, wireless, 8-channel EEG monitoring headset. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology (EMBC), Buenos Aires, Argentina, 31 August–4 September 2010; pp. 4197–4200. [Google Scholar]
- Tolbert, J.R.; Kabali, P.; Brar, S.; Mukhopadhyay, S. An accuracy aware low power wireless EEG unit with information content based adaptive data compression. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 5417–5420. [Google Scholar]
- Higgins, G.; Faul, S.; McEvoy, R.; McGinley, B.; Glavin, M.; Marnane, W.; Jones, E. EEG compression using JPEG2000: How much loss is too much? In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology (EMBC), Buenos Aires, Argentina, 31 August–4 September 2010; pp. 614–617. [Google Scholar]
- Casson, A.J.; Yates, D.C.; Smith, S.J.M.; Duncan, J.S.; Rodriguez-Villegas, E. Wearable Electroencephalography. What is it, why is it needed, and what does it entail? IEEE Eng. Med. Biol. Mag. 2010, 29, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Cheng, C.; Jung, T. Developing stimulus presentation on mobile devices for a truly portable SSVEP-based BCI. In Proceedings of the 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 5271–5274. [Google Scholar]
- Webb, J.; Xiao, Z.G.; Aschenbrenner, K.; Herrnstadt, G.; Menon, C. Towards a portable assistive arm exoskeleton for stroke patient rehabilitation controlled through a brain computer interface. In Proceedings of the 4th IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics (BioRob), Rome, Italy, 24–27 June 2012; pp. 1299–1304. [Google Scholar]
- Shyu, K.; Lee, P.; Lee, M.; Lin, M.; Lai, R.; Chiu, Y. Development of a low-cost FPGA-based SSVEP BCI multimedia control system. IEEE Trans. Biomed. Circuits Syst. 2010, 4, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Khurana, K.; Gupta, P.; Panicker, R.; Kumar, A. Development of an FPGA-based real-time P300 speller. In Proceedings of the 22nd International Conference on Field Programmable Logic and Applications (FPL), Oslo, Norway, 29–31 August 2012; pp. 551–554. [Google Scholar]
- Shyu, K.; Chiu, Y.; Lee, P.; Lee, M.; Sie, J.; Wu, C.; Wu, Y.; Tung, P. Total Design of an FPGA-Based Brain-Computer Interface Control Hospital Bed Nursing System. IEEE Trans. Ind. Electron. 2013, 60, 2731–2739. [Google Scholar] [CrossRef]
- Annese, V.F.; Crepaldi, M.; Demarchi, D.; de Venuto, D. A digital processor architecture for combined EEG/EMG falling risk prediction. In Proceedings of the Design, Automation Test in Europe Conference & Exhibition (DATE), Dresden, Germany, 14–18 March 2016; pp. 714–719. [Google Scholar]
- Belwafi, K.; Ghaffari, F.; Djemal, R.; Romain, O. A Hardware/Software Prototype of EEG-based BCI System for Home Device Control. J. Signal. Process. Syst. 2016, 1–17. [Google Scholar] [CrossRef]
- Chi, Y.; Wang, Y.; Wang, Y.; Maier, C.; Jung, T.; Cauwenberghs, G. Dry and Noncontact EEG Sensors for Mobile Brain-Computer Interfaces. IEEE Trans. Neural Syst. Rehabil. Eng. 2012, 20, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Page, A.; Sagedy, C.; Smith, E.; Attaran, N.; Oates, T.; Mohsenin, T. A Flexible Multichannel EEG Feature Extractor and Classifier for Seizure Detection. IEEE Trans. Circuits Syst. II Express Briefs 2015, 62, 109–113. [Google Scholar] [CrossRef]
- Bailey, D. Design for Embedded Image Processing on FPGAs, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- McAllister, J. FPGA-based DSP. In Handbook of Signal Processing Systems; Springer: New York, NY, USA, 2013; pp. 363–392. [Google Scholar]
- Vanderbauwhede, W.; Benkrid, K. High-Performance Computing Using FPGAs; Springer: New York, NY, USA, 2013. [Google Scholar]
- García, G.; Jara, C.; Pomares, J.; Alabdo, A.; Poggi, L.; Torres, F. A survey on FPGA-Based sensor systems: Towards intelligent and reconfigurable low-power sensors for computer vision, control and signal processing. Sensors 2014, 14, 6247–6278. [Google Scholar] [CrossRef] [PubMed]
- Trimberger, S. Three Ages of FPGAs: A Retrospective on the First Thirty Years of FPGA Technology. Proc. IEEE 2015, 103, 318–331. [Google Scholar] [CrossRef]
- Tessier, R.; Pocek, K.; DeHon, A. Reconfigurable Computing Architectures. Proc. IEEE 2015, 103, 332–354. [Google Scholar] [CrossRef]
- Xilinx Inc. UG473 7 Series FPGAs Memory Resources User Guide, 1.11 ed.; Xilinx Inc.: San Jose, CA, USA, 2014. [Google Scholar]
- Xilinx Inc. UG479 7 Series DSP48E1 Slice User Guide, 1.8th ed.; Xilinx Inc.: San Jose, CA, USA, 2014. [Google Scholar]
- Altera Corp. Altera’s User-Customizable ARM-Based SoC; Altera Corp.: San Jose, CA, USA, 2014. [Google Scholar]
- Krell, M.; Straube, S.; Seeland, A.; Wöhrle, H.; Teiwes, J.; Metzen, J.H.; Kirchner, E.; Kirchner, F. pySPACE—A signal processing and classification environment in Python. Front. Neuroinf. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Renard, Y.; Lotte, F.; Gibert, G.; Congedo, M.; Maby, E.; Delannoy, V.; Bertrand, O.; Lécuyer, A. Openvibe: An open-source software platform to design, test, and use brain–computer interfaces in real and virtual environments. Presence 2010, 19, 35–53. [Google Scholar] [CrossRef]
- Martin, G.; Smith, G. High-level synthesis: Past, present, and future. IEEE Des. Test 2009, 26, 18–25. [Google Scholar] [CrossRef]
- Windh, S.; Ma, X.; Halstead, R.J.; Budhkar, P.; Luna, Z.; Hussaini, O.; Najjar, W.A. High-Level Language Tools for Reconfigurable Computing. Proc. IEEE 2015, 103, 390–408. [Google Scholar] [CrossRef]
- Skalicky, S.; Wood, C.; ukowiak, M.; Ryan, M. High level synthesis: Where are we? A case study on matrix multiplication. In Proceedings of the International Conference on Reconfigurable Computing and FPGAs (ReConFig), Cancun, Mexico, 9–11 December 2013; pp. 1–7. [Google Scholar]
- Dennis, J. First version of a data flow procedure language. In Programming Symposium; Lecture Notes in Computer Science; Robinet, B., Ed.; Springer: Berlin/Heidelberg, Germany, 1974; Volume 19, pp. 362–376. [Google Scholar]
- Lee, E.; Messerschmitt, D. Synchronous data flow. Proc. IEEE 1987, 75, 1235–1245. [Google Scholar] [CrossRef]
- Lee, E.A.; Parks, T.M. Dataflow process networks. Proc. IEEE 1995, 83, 773–801. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Deprettere, E.; Leupers, R.; Takala, J. Handbook of Signal Processing Systems; Springer: New York, NY, USA, 2013. [Google Scholar]
- Wöhrle, H.; Kirchner, F. Reconfigurable Hardware-Based Acceleration for Machine Learning and Signal Processing. In Formal Modeling and Verification of Cyber-Physical Systems; Lecture Notes in Computer Science; Drechsler, R., Kühne, U., Eds.; Springer: Wiesbaden, Germany, 2015. [Google Scholar]
- Williamson, M.C.; Lee, E.A. Synthesis of parallel hardware implementations from synchronous dataflow graph specifications. In Proceedings of the Conference Record of the 30th Asilomar Conference on Signals, Systems and Computers, Pacific Grove, CA, USA, 3–6 November 1996; pp. 1340–1343. [Google Scholar]
- Roquier, G.; Bezati, E.; Mattavelli, M. Hardware and software synthesis of heterogeneous systems from dataflow programs. J. Electr. Comp. Eng. 2012, 2012, 484962. [Google Scholar] [CrossRef]
- Wöhrle, H.; Teiwes, J.; Krell, M.; Seeland, A.; Kirchner, E.; Kirchner, F. Reconfigurable Dataflow Hardware Accelerators for Machine Learning and Robotics. Proceedings of European Conference on Machine Learning and Principles and Practice of Knowledge Discovery in Databases, Nancy, France, 15–19 September 2014. [Google Scholar]
- Yazdanpanah, F.; Alvarez-Martinez, C.; Jimenez-Gonzalez, D.; Etsion, Y. Hybrid dataflow/von-Neumann architectures. IEEE Trans. Parallel Distrib. Syst. 2014, 25, 1489–1509. [Google Scholar] [CrossRef]
- Zenzes, M.; Kampmann, P.; Stark, T.; Schilling, M. NDLCom: Simple Protocol for Heterogeneous Embedded Communication Networks. In Proceedings of the Embedded World Exhibition & Conference, Nuremberg, Germany, 23–25 February 2016. [Google Scholar]
- Bartsch, S.; Manz, M.; Kampmann, P.; Dettmann, A.; Hanff, H.; Langosz, M.; von Szadkowski, K.; Hilljegerdes, J.; Simnofske, M.; Kloss, P.; et al. Development and Control of the Multi-Legged Robot Mantis. In Proceedings of the 47th International Symposium on Robotics (ISR-2016), Munich, Germany, 21–22 June 2016. [Google Scholar]
- Rivet, B.; Souloumiac, A.; Attina, V.; Gibert, G. xDAWN Algorithm to Enhance Evoked Potentials: Application to Brain Computer Interface. IEEE Trans. Biomed. Eng. 2009, 56, 2035–2043. [Google Scholar] [CrossRef] [PubMed]
- Wöhrle, H.; Krell, M.; Straube, S.; Kim, S.; Kirchner, E.; Kirchner, F. An Adaptive Spatial Filter for User-Independent Single Trial Detection of Event-Related Potentials. IEEE Trans. Biomed. Eng. 2015, 62, 1696–1705. [Google Scholar] [CrossRef] [PubMed]
- van der Walt, S.; Colbert, S.; Varoquaux, G. The NumPy Array: A Structure for Efficient Numerical Computation. IEEE Comput. Sci. Eng. 2011, 13, 22–30. [Google Scholar] [CrossRef]
- Jones, E.; Oliphant, T.; Peterson, P. SciPy: Open Source Scientific Tools For Python. 2001. Available online: http://www.scipy.org (accessed on 21 January 2017).
- Seeland, A.; Manca, L.; Kirchner, F.; Kirchner, E. Spatio-temporal Comparison Between ERD/ERS and MRCP-based Movement Prediction. In Proceedings of the 8th International Conference on Bio-inspired Systems and Signal Processing (BIOSIGNALS-15), Lisbon, Portugal, 12–15 January 2015. [Google Scholar]
- Nikolic, M.; Krarup, C. EMGTools, an Adaptive and Versatile Tool for Detailed EMG Analysis. IEEE Trans. Biomed. Eng. 2011, 58, 2707–2718. [Google Scholar] [CrossRef] [PubMed]
- Krell, M.; Tabie, M.; Wöhrle, H.; Kirchner, E. Memory and Processing Efficient Formula for Moving Variance Calculation in EEG and EMG Signal Processing. In Proceedings of the International Congress on Neurotechnology, Electronics and Informatics (NEUROTECHNIX 2013), Vilamoura, Portugal, 16–17 November 2015; pp. 41–45. [Google Scholar]
- Semmaoui, H.; Drolet, J.; Lakhssassi, A.; Sawan, M. Setting Adaptive Spike Detection Threshold for Smoothed TEO Based on Robust Statistics Theory. IEEE Trans. Biomed. Eng. 2012, 59, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Dick, C.; Harris, F. FPGA signal processing using sigma-delta modulation. IEEE Signal Process. Mag. 2000, 17, 20–35. [Google Scholar] [CrossRef]
- Crochiere, R.; Rabiner, L. Optimum FIR digital filter implementations for decimation, interpolation, and narrow-band filtering. IEEE Trans. Acoust. Speech Signal Process. 1975, 23, 444–456. [Google Scholar] [CrossRef]
- Bellanger, M. Computation rate and storage estimation in multirate digital filtering with half-band filters. IEEE Trans. Acoust. Speech Signal. Process. 1977, 25, 344–346. [Google Scholar] [CrossRef]
- Jansen, B.; Allam, A.; Kota, P.; Lachance, K.; Osho, A.; Sundaresan, K. An exploratory study of factors affecting single trial P300 detection. IEEE Trans. Biomed. Eng. 2004, 51, 975–978. [Google Scholar] [CrossRef] [PubMed]
- Ghaderi, F.; Kim, S.; Kirchner, E. Effects of eye artifact removal methods on single trial P300 detection, a comparative study. J. Neurosci. Methods 2014, 221, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Wöhrle, H.; Kirchner, F. CAEMO—A high performance matrix coprocessor for SoCs. Microprocess. Microsyst. 2017. submitted. [Google Scholar]
- Crammer, K.; Dekel, O.; Keshet, J.; Shalev-Shwartz, S.; Singer, Y. Online Passive-Aggressive Algorithms. J. Mach. Learn. Res. 2006, 7, 551–585. [Google Scholar]
- Cawley, G.C.; Talbot, N. On over-fitting in model selection and subsequent selection bias in performance evaluation. J. Mach. Learn. Res. 2010, 11, 2079–2107. [Google Scholar]
- Metzen, J.; Kirchner, E. Rapid Adaptation of Brain Reading Interfaces based on Threshold Adjustment. In Proceedings of the 35th Annual Conference of the German Classification Society, (GfKl-2011), Farankfurt, Germany, 30 August–2 September 2011; p. 138. [Google Scholar]
- Straube, S.; Krell, M. How to evaluate an agent’s behaviour to infrequent events? Reliable performance estimation insensitive to class distribution. Front. Comput. Neurosci. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- OpenMP Architecture Review Board. OpenMP Application Program Interface, Version 3.0. May 2008. Available online: http://www.openmp.org/wp-content/uploads/spec30.pdf (accessed on 14 December 2016).
- Kornhuber, H.; Deecke, L. Hirnpotentialaänderungen bei Willkürbewegungen und passiven Bewegungen des Menschen: Bereitschaftspotential und reafferente Potentiale. Pflüg. Arch. Eur. J. Physiol. 1965, 284, 1–17. (In Germany) [Google Scholar] [CrossRef]
- Deecke, L.; Scheid, P.; Kornhuber, H. Distribution of readiness potential, pre-motion positivity, and motor potential of the human cerebral cortex preceding voluntary finger movements. Exp. Brain Res. 1969, 7, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Makeig, S. Predicting intended movement direction using EEG from human posterior parietal cortex. In International Conference on Foundations of Augmented Cognition; San Diego, CA, USA, 19–24 July 2009, pp. 437–446.
- Lew, E.Y.L.; Chavarriaga, R.; Silvoni, S.; Millán, J.d.R. Single trial prediction of self-paced reaching directions from EEG signals. Front. Neurosci. 2014, 8, 222. [Google Scholar] [CrossRef] [PubMed]
- Wöhrle, H.; Teiwes, J.; Kirchner, E.A.; Kirchner, F. A Framework for High Performance Embedded Signal Processing and Classification of Psychophysiological Data. In Proceedings of the International Conference on Biomedical Engineering and Technology (ICBET), Copenhagen, Denmark, 19–20 May 2013; pp. 60–66. [Google Scholar]
- Wöhrle, H.; Teiwes, J.; Tabie, M.; Seeland, A.; Kirchner, E.A.; Kirchner, F. Prediction of Movements by Online Analysis of Electroencephalogram with Dataflow Accelerators. In Proceedings of the International Congress on Neurotechnology, Electronics and Informatics (NEUROTECHNIXX), Rome, Italy, 25–26 November 2014; pp. 31–37. [Google Scholar]
- Johnson, S.H. Imagining the impossible: Intact motor representations in hemiplegics. Neuroreport 2000, 11, 729–732. [Google Scholar] [CrossRef] [PubMed]
- Platz, T.; Kim, I.H.; Pintschovius, H.; Winter, T.; Kieselbach, A.; Villringer, K.; Kurth, R.; Mauritz, K.H. Multimodal EEG analysis in man suggests impairment-specific changes in movement-related electric brain activity after stroke. Brain 2000, 123, 2475–2490. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, V.; Daly, I.; Pichiorri, F.; Mattia, D.; Müller-Putz, G.; Neuper, C. Relationship between electrical brain responses to motor imagery and motor impairment in stroke. Stroke 2012, 43, 2735–2740. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Zhang, T.; Shan, L. Cortical lateralization in stroke patients measured by event-related potentials during motor imagery. Mol. Med. Rep. 2013, 8, 1701–1707. [Google Scholar] [PubMed]
- Castro, A.; Diaz, F.; Sumich, A. Long-term neuroplasticity in spinal cord injury patients: A study on movement-related brain potentials. Int. J. Psychophysiol 2013, 87, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Jiang, N.; Vuckovic, A.; Hasan, M.; Mrachacz-Kersting, N.; Allan, D.; Fraser, M.; Nasseroleslami, B.; Conway, B.; Dremstrup, K.; Farina, D. Movement-related cortical potentials in paraplegic patients: Abnormal patterns and considerations for BCI-rehabilitation. Front. Neuroeng. 2014, 7, 35. [Google Scholar] [CrossRef] [PubMed]
- Park, W.; Kwon, G.H.; Kim, Y.H.; Lee, J.H.; Kim, L. EEG response varies with lesion location in patients with chronic stroke. J. NeuroEng. Rehabil. 2016, 13, 21. [Google Scholar] [CrossRef] [PubMed]
- Dewald, J.; Sheshadri, V.; Dawson, M.; Beer, R. Upper-limb discoordination in hemiparetic stroke: Implications for neurorehabilitation. Top. Stroke Rehabil. 2001, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Cesqui, B.; Tropea, P.; Micera, S.; Krebs, H. EMG-based pattern recognition approach in post stroke robot-aided rehabilitation: A feasibility study. J. Neuroeng. Rehabil. 2013, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Jochumsen, M.; Niazi, I.; Mrachacz-Kersting, N.; Jiang, N.; Farina, D.; Dremstrup, K. Comparison of spatial filters and features for the detection and classification of movement-related cortical potentials in healthy individuals and stroke patients. J. Neural Eng. 2015, 12, 056003. [Google Scholar] [CrossRef] [PubMed]
- Jochumsen, M.; Niazi, I.; Taylor, D.; Farina, D.; Dremstrup, K. Detecting and classifying movement-related cortical potentials associated with hand movements in healthy subjects and stroke patients from single-electrode, single-trial EEG. J. Neural Eng. 2015, 12, 056013. [Google Scholar]
- Kamavuako, E.; Jochumsen, M.; Niazi, I.; Dremstrup, K. Comparison of features for movement prediction from single-trial movement-related cortical potentials in healthy subjects and stroke patients. Comput. Intell. Neurosci. 2015, 2015, 71. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Farina, D.; Murguialday, A.R.; Dremstrup, K.; Birbaumer, N. Comparison of movement related cortical potential in healthy people and amyotrophic lateral sclerosis patients. Front. Neurosci. 2013, 7, 65. [Google Scholar] [CrossRef] [PubMed]
- Ando, T.; Moshnyaga, V.; Hashimoto, K. A low-power FPGA implementation of eye tracking. In Proceedings of the IEEE International Conference on Acoustics, Speech and Signal Processing (ICASSP), Kyoto, Japan, 25–30 March 2012; pp. 1573–1576. [Google Scholar]


| Programmable | Look-Up Tables | Flip-Flops | BRAM | DSP Slices |
|---|---|---|---|---|
| Logic Cells | (LUT) | (FF) | (36 kb Each) | (DSP48) |
| 125,000 | 78,600 | 157,200 | 265 | 400 |
| Not Combined with P300 (NP3) | Combined with P300 (CP3) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MRCP | EMG | MaE | MoE | MRCP | EMG | MaE | MoE | |||||||||
| CPU | FPGA | CPU | FPGA | CPU | FPGA | CPU | FPGA | CPU | FPGA | CPU | FPGA | CPU | FPGA | CPU | FPGA | |
| Mean FNR | 0.104 | 0.116 | 0.063 | 0.063 | 0.278 | 0.290 | 0.021 | 0.019 | 0.116 | 0.127 | 0.063 | 0.062 | 0.282 | 0.293 | 0.021 | 0.020 |
| Mean FPR | 0.147 | 0.142 | 0.029 | 0.034 | 0.006 | 0.007 | 0.169 | 0.168 | 0.022 | 0.021 | 0.005 | 0.006 | 0.002 | 0.002 | 0.033 | 0.033 |
| Mean Precision | 0.108 | 0.109 | 0.332 | 0.303 | 0.577 | 0.541 | 0.123 | 0.121 | 0.422 | 0.421 | 0.711 | 0.695 | 0.826 | 0.810 | 0.403 | 0.408 |
| Movement Prediction Times | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MRCP | EMG | MaE | MoE | |||||||||||||
| System | ||||||||||||||||
| SRS | 515 | 234 | 514 | 834 | 140 | 39 | 79 | 199 | 124 | 34 | 74 | 154 | 585 | 274 | 594 | 914 |
| SRS (2) | 516 | 235 | 515 | 835 | 140 | 39 | 79 | 199 | 125 | 35 | 75 | 155 | 586 | 275 | 595 | 915 |
| SRS (4) | 516 | 235 | 515 | 835 | 140 | 39 | 79 | 199 | 125 | 35 | 75 | 155 | 586 | 275 | 595 | 915 |
| MRS | N/A | N/A | 134 | 33 | 73 | 193 | N/A | N/A | N/A | N/A | ||||||
| MRS (2) | N/A | N/A | 134 | 33 | 73 | 193 | N/A | N/A | N/A | N/A | ||||||
| FPGA | 542 | 239 | 559 | 879 | 136 | 40 | 80 | 200 | 129 | 39 | 79 | 159 | 596 | 269 | 599 | 919 |
| PaM | PaE | PaMaE | PaMoE | |||||||||||||
| System | ||||||||||||||||
| SRS | 314 | 194 | 234 | 354 | 125 | 34 | 74 | 194 | 190 | 34 | 74 | 154 | 340 | 194 | 234 | 394 |
| SRS (2) | 315 | 195 | 235 | 355 | 126 | 35 | 75 | 195 | 191 | 35 | 75 | 155 | 340 | 195 | 235 | 395 |
| SRS (4) | 315 | 195 | 235 | 355 | 126 | 35 | 75 | 195 | 191 | 35 | 75 | 155 | 341 | 195 | 235 | 395 |
| MRS | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | ||||||||
| MRS (2) | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | ||||||||
| FPGA | 319 | 199 | 239 | 359 | 129 | 39 | 79 | 199 | 199 | 39 | 79 | 159 | 355 | 199 | 239 | 399 |
| SRS | Idle | 1 Core | 2 Cores | 4 Cores |
| EMG | 119.8 W | 130.2 W | 130.2 W | 130.2 W |
| MRCP | 119.8 W | 126.0 W | 126.4 W | 127.6 W |
| MaE, MoE | 119.8 W | 133.0 W | 133.3 W | 135.2 W |
| PaM | 119.8 W | 129.6 W | 130.2 W | 132.1 W |
| PaE | 119.8 W | 132.6 W | 132.6 W | 132.6 W |
| PaMaE, PaMoE | 119.8 W | 136.8 W | 138.1 W | 138.3 W |
| MRS | Idle | 1 Core | 2 Cores | |
| EMG | 2.98 W | 3.28 W | 3.23 W | |
| MRCP | 2.98 W | 3.23 W | 3.23 W | |
| MaE, MoE | 2.98 W | 3.30 W | 3.61 W | |
| PaM | 2.98 W | 3.29 W | 3.60 W | |
| PaE | 2.98 W | 3.31 W | 3.64 W | |
| PaMaE, PaMoE | 2.98 W | 3.32 W | 3.66 W | |
| FPGA | Idle | Computing | ||
| EMG | 3.45 W | 4.12 W | ||
| MRCP | 3.45 W | 4.12 W | ||
| MaE, MoE | 3.49 W | 4.41 W | ||
| PaM | 3.58 W | 4.48 W | ||
| PaE | 3.53 W | 4.49 W | ||
| PaMaE, PaMoE | 3.59 W | 4.51 W |
| Type of Resource | LUT | FF | 36 kb Each | DSP48 |
|---|---|---|---|---|
| DFHWA | 15 755 (20.0%) | 16 786 (10.7%) | 53.5 (20.1%) | 136 (34%) |
| Auxiliary Components | 6 084 (7.7%) | 7 529 (4.8%) | 7.5 (2.8%) | 0 (0%) |
| Total | 21 839 (27.8%) | 24 315 (15.4%) | 61 (23.0%) | 136 (34%) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wöhrle, H.; Tabie, M.; Kim, S.K.; Kirchner, F.; Kirchner, E.A. A Hybrid FPGA-Based System for EEG- and EMG-Based Online Movement Prediction. Sensors 2017, 17, 1552. https://doi.org/10.3390/s17071552
Wöhrle H, Tabie M, Kim SK, Kirchner F, Kirchner EA. A Hybrid FPGA-Based System for EEG- and EMG-Based Online Movement Prediction. Sensors. 2017; 17(7):1552. https://doi.org/10.3390/s17071552
Chicago/Turabian StyleWöhrle, Hendrik, Marc Tabie, Su Kyoung Kim, Frank Kirchner, and Elsa Andrea Kirchner. 2017. "A Hybrid FPGA-Based System for EEG- and EMG-Based Online Movement Prediction" Sensors 17, no. 7: 1552. https://doi.org/10.3390/s17071552
APA StyleWöhrle, H., Tabie, M., Kim, S. K., Kirchner, F., & Kirchner, E. A. (2017). A Hybrid FPGA-Based System for EEG- and EMG-Based Online Movement Prediction. Sensors, 17(7), 1552. https://doi.org/10.3390/s17071552




