Smartphone-Based Food Diagnostic Technologies: A Review
Abstract
:1. Introduction
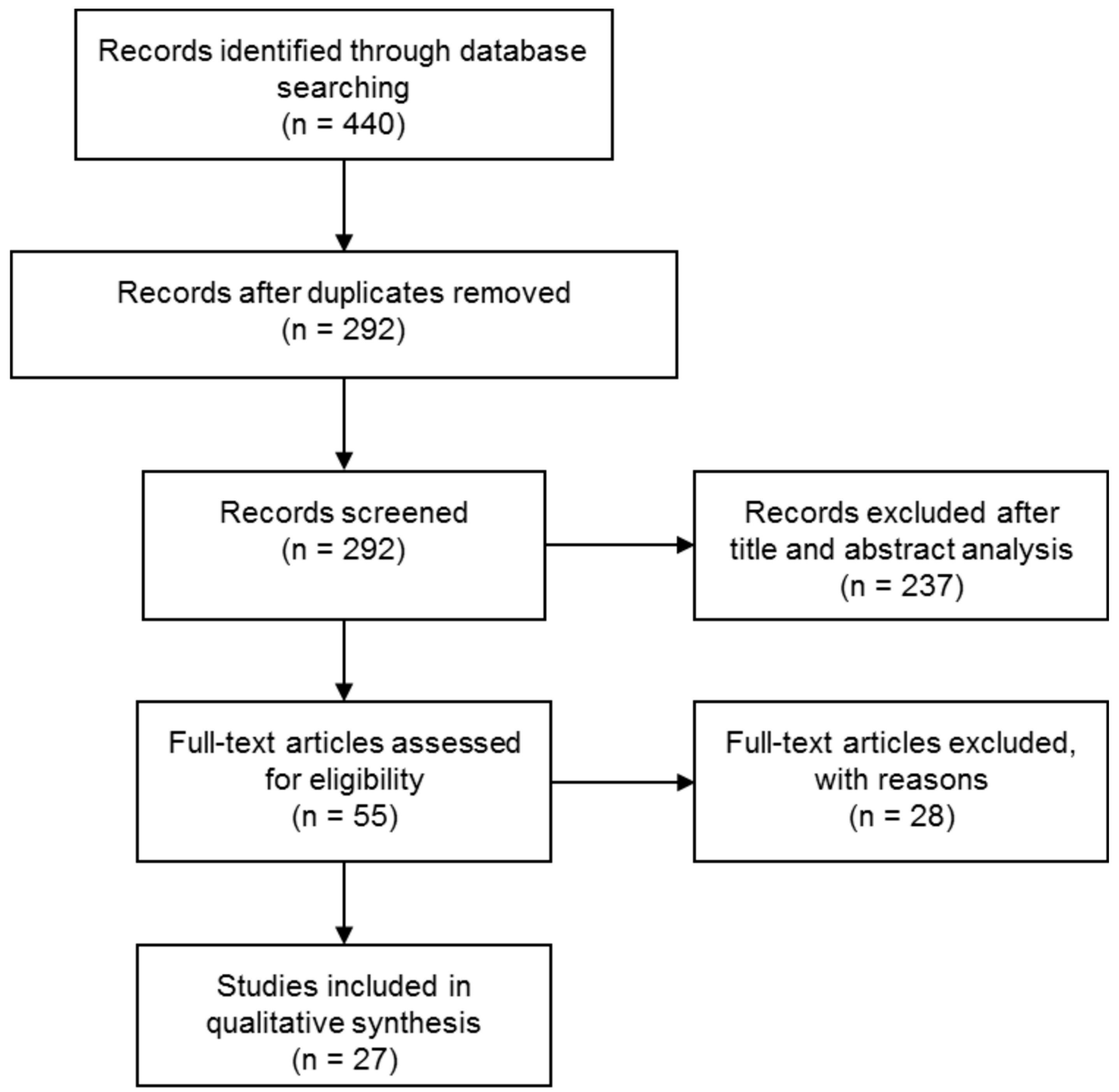
2. Materials and Methods
3. Results
3.1. Lab-On-Smartphone Biosensors
3.1.1. Fluorescence Imaging Using Smartphone
3.1.2. Smartphone-Based Colorimetric Readers
3.1.3. Smartphone-Based Electro-Analytical Platforms
3.2. Smartphone Spectroscopy
4. Emerging Market of Smartphone-Based Food Diagnostic Platforms
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Horizon Prize Food Scanner 1. Available online: http://ec.europa.eu/research/horizonprize/index.cfm?prize=food-scanner (accessed on 1 February 2017).
- Ozcan, A. Mobile phones democratize and cultivate next-generation imaging, diagnostics and measurement tools. Lab Chip 2014, 14, 3187–3194. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.Y.-C.; Hsu, M.-Y.; Chen, S.-J.; Hwang, D.-K.; Yen, T.-H.; Cheng, C.-M. Point-of-Care Detection Devices for Food Safety Monitoring: Proactive Disease Prevention. Trends Biotechnol. 2017, 35, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Liu, Q. Biosensors and bioelectronics on smartphone for portable biochemical detection. Biosens. Bioelectron. 2016, 75, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Bao, Y.; Wang, D.; Wang, W.; Niu, L. Smartphones for sensing. Sci. Bull. 2016, 61, 190–201. [Google Scholar] [CrossRef]
- Daponte, P.; Vito, L.; de Picariello, F.; Riccio, M. State of the art and future developments of measurement applications on smartphones. Measurement 2013, 46, 3291–3307. [Google Scholar] [CrossRef]
- Xu, X.; Akay, A.; Wei, H.; Wang, S.; Pingguan-Murphy, B.; Erlandsson, B.E.; Li, X.; Lee, W.; Hu, J.; Wang, L.; et al. Advances in Smartphone-Based Point-of-Care Diagnostics. Proc. IEEE 2015, 103, 236–247. [Google Scholar] [CrossRef]
- Roda, A.; Michelini, E.; Zangheri, M.; Di Fusco, M.; Calabria, D.; Simoni, P. Smartphone-based biosensors: A critical review and perspectives. TrAC Trends 2016, 79, 317–325. [Google Scholar] [CrossRef]
- Liu, X.; Lin, T.-Y.; Lillehoj, P.B. Smartphones for Cell and Biomolecular Detection. Ann. Biomed. Eng. 2014, 42, 2205–2217. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Peretz-Soroka, H.; Liu, Y.; Lin, F. Novel developments in mobile sensing based on the integration of microfluidic devices and smartphones. Lab Chip 2016, 16, 943–958. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Sikora, U.; Ozcan, A. Quantum dot enabled detection of Escherichia coli using a cell-phone. Analyst 2012, 137, 2541–2544. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, S.K.J.; Zhu, H.; Phillips, S.; Shiledar, A.; Feng, S.; Tseng, D.; van Ginkel, L.A.; Nielen, M.W.F.; Ozcan, A. Cellphone-based detection platform for rbST biomarker analysis in milk extracts using a microsphere fluorescence immunoassay. Anal. Bioanal. Chem. 2014, 406, 6857–6866. [Google Scholar] [CrossRef] [PubMed]
- Mora, C.A.; Herzog, A.F.; Raso, R.A.; Stark, W.J. Programmable living material containing reporter micro-organisms permits quantitative detection of oligosaccharides. Biomaterials 2015, 61, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Coskun, A.F.; Wong, J.; Khodadadi, D.; Nagi, R.; Tey, A.; Ozcan, A. A personalized food allergen testing platform on a cellphone. Lab Chip 2013, 13, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, G.; Moon, J. Performance improvement of the one-dot lateral flow immunoassay for aflatoxin B1 by using a smartphone-based reading system. Sensors 2013, 13, 5109–5116. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Wang, R.; Bever, C.R.S.; Xing, S.; Hammock, B.D.; Pan, T. Smartphone-interfaced lab-on-a-chip devices for field-deployable enzyme-linked immunosorbent assay. Biomicrofluidics 2014, 8, 064101. [Google Scholar] [CrossRef] [PubMed]
- Park, T.S.; Baynes, C.; Cho, S.-I.; Yoon, J.-Y. Paper microfluidics for red wine tasting. RSC Adv. 2014, 4, 24356–24362. [Google Scholar] [CrossRef]
- Bueno, L.; Meloni, G.N.; Reddy, S.M.; Paixao, T.R.L.C. Use of plastic-based analytical device, smartphone and chemometric tools to discriminate amines. RSC Adv. 2015, 5, 20148–20154. [Google Scholar] [CrossRef]
- Masawat, P.; Harfield, A.; Namwong, A. An iPhone-based digital image colorimeter for detecting tetracycline in milk. Food Chem. 2015, 184, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Monosik, R.; dos Santos, V.B.; Angnes, L. A simple paper-strip colorimetric method utilizing dehydrogenase enzymes for analysis of food components. Anal. Methods 2015, 7, 8177–8184. [Google Scholar] [CrossRef]
- Yu, L.; Shi, Z.; Fang, C.; Zhang, Y.; Liu, Y.; Li, C. Disposable lateral flow-through strip for smartphone-camera to quantitatively detect alkaline phosphatase activity in milk. Biosens. Bioelectron. 2015, 69, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Qiu, X.; Wan, Z.; Zou, Q.; Su, K.; Hu, N.; Wang, P. A sensing smartphone and its portable accessory for on-site rapid biochemical detection of marine toxins. Anal. Methods 2016, 8, 6895–6902. [Google Scholar] [CrossRef]
- Levin, S.; Krishnan, S.; Rajkumar, S.; Halery, N.; Balkunde, P. Monitoring of fluoride in water samples using a smartphone. Sci. Total Environ. 2016, 551–552, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, Y.; Bao, X.; Han, J.; Xia, J.; Tian, X.; Ni, L. A smartphone-based colorimetric reader coupled with a remote server for rapid on-site catechols analysis. Talanta 2016, 160, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.M.; Kim, S.W.; Jeon, J.W.; Kim, J.H.; Kim, H.S.; Cho, J.H.; Lee, W.H.; Paek, S.H. Food contamination monitoring via internet of things, exemplified by using pocket-sized immunosensor as terminal unit. Sens. Actuators B Chem. 2016, 233, 148–156. [Google Scholar] [CrossRef]
- DuVall, J.A.; Borba, J.C.; Shafagati, N.; Luzader, D.; Shukla, N.; Li, J.; Kehn-Hall, K.; Kendall, M.M.; Feldman, S.H.; Landers, J.P. Optical Imaging of Paramagnetic Bead-DNA Aggregation Inhibition Allows for Low Copy Number Detection of Infectious Pathogens. PLoS ONE 2015, 10, e0129830. [Google Scholar] [CrossRef] [PubMed]
- Dou, Y.; Jiang, Z.; Deng, W.; Su, J.; Chen, S.; Song, H.; Aldalbahi, A.; Zuo, X.; Song, S.; Shi, J.; Fan, C. Portable detection of clenbuterol using a smartphone-based electrochemical biosensor with electric field-driven acceleration. J. Electroanal. Chem. 2016, 781, 339–344. [Google Scholar] [CrossRef]
- Giordano, G.F.; Vicentini, M.B.R.; Murer, R.C.; Augusto, F.; Ferrão, M.F.; Helfer, G.A.; da Costa, A.B.; Gobbi, A.L.; Hantao, L.W.; Lima, R.S. Point-of-use electroanalytical platform based on homemade potentiostat and smartphone for multivariate data processing. Electrochim. Acta 2016, 219, 170–177. [Google Scholar] [CrossRef]
- Liang, P.-S.; Park, T.S.; Yoon, J.-Y. Rapid and reagentless detection of microbial contamination within meat utilizing a smartphone-based biosensor. Sci. Rep. 2014, 4, 5953. [Google Scholar] [CrossRef] [PubMed]
- Bouyé, C.; Kolb, H.; D’Humières, B. Mini and micro spectrometers pave the way to on-field advanced analytics. In Proceedings of the SPIE-Photonics Instrumentation Engineering III, San Francisco, CA, USA, 13 February 2016; SPIE: Bellingham, WA, USA, 2016; Volume 9754. [Google Scholar]
- Mignani, A.G.; Mencaglia, A.A.; Baldi, M.; Ciaccheri, L. SpiderSpec: A low-cost compact colorimeter with IoT functionality. In Proceedings of the SPIE Fifth Asia-Pacific Optical Sensors Conference, Jeju, Korea, 20 May 2015; Lee, B., Lee, S.B., Rao, Y., Eds.; SPIE: Bellingham, WA, USA, 2015; Volume 9655. [Google Scholar]
- Das, A.J.; Wahi, A.; Kothari, I.; Raskar, R. Ultra-portable, wireless smartphone spectrometer for rapid, non-destructive testing of fruit ripeness. Sci. Rep. 2016, 6, 32504. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.; Lee, H.; Cho, H.; Moon, S.-H.; Kim, E.-K.; Kim, M.S. Detection of fecal contamination on beef meat surfaces using handheld fluorescence imaging device (HFID). In Proceedings of the SPIE Sensing for Agriculture and Food Quality and Safety VIII, Baltimore, MD, USA, 17 April 2016; Kim, M.S., Chao, K., Chin, B.A., Eds.; SPIE: Bellingham, WA, USA, 2016; Volume 9864. [Google Scholar]
- Rissanen, A.; Saari, H.; Rainio, K.; Stuns, I.; Viherkanto, K.; Holmlund, C.; Nakki, I.; Ojanen, H. MEMS FPI-based smartphone hyperspectral imager. In Proceedings of the SPIE Next-Generation Spectroscopic Technologies IX, Baltimore, MD, USA, 17 April 2016; Druy, M.A., Crocombe, R.A., Eds.; SPIE: Bellingham, WA, USA, 2016; Volume 9855. [Google Scholar]
- Sasikumar, H.; Prasad, V.; Pal, P.; Varma, M.M. Diffractive Interference Optical Analyzer (DiOPTER). In Proceedings of the SPIE Optical Diagnostics and Sensing XVI: Toward Point-of-Care Diagnostics, San Francisco, CA, USA, 13 February 2016; Cote, G.L., Ed.; SPIE: Bellingham, WA, USA, 2016; Volume 9715. [Google Scholar]
- Yu, X.; Lu, Q.; Gao, H.; Ding, H. Development of a handheld spectrometer based on a linear variable filter and a complementary metal-oxide-semiconductor detector for measuring the internal quality of fruit. J. Near Infrared Spectrosc. 2016, 24, 69–76. [Google Scholar] [CrossRef]
- CELLMIC | We Innovate Mobile Diagnostics |. Available online: http://www.cellmic.com/ (accessed on 20 March 2017).
- Ozcan, A.; Coskun, A.; Wong, J. Allergen Testing Platform for Use with Mobile Electronic Devices. U.S. Pat. App. 14/053,475, 14 October 2013. [Google Scholar]
- OrganaDx Sensor | Portable Food Tester Analyzer. Available online: http://www.cdxlife.com/organa-sensor/ (accessed on 20 March 2017).
- Hosono, S.; Qi, W.; Sato, S.; Suzuki, Y.; Fujiwara, M.; Hiramatsu, H.; Suzuki, S.; Abeygunawardhana, P.K.W.; Wada, K.; Nishiyama, A.; et al. Proposal of AAA-battery-size one-shot ATR Fourier spectroscopic imager for on-site analysis-Simultaneous measurement of multi-components with high accuracy. In Proceedings of the SPIE 9314, Optics and Biophotonics in Low-Resource Settings, San Francisco, CA, USA, 7 February 2015; SPIE: Bellingham, WA, USA, 2015; Volume 9314. [Google Scholar]
- Rouse, R. Apparatus for Detection and Delivery of Volatilized Compounds and RELATED Methods. U.S. Patent Application 14/905,780, 14 July 2014. [Google Scholar]
- Gailius, D. Electronic Nose for Determination of Meat Freshness. U.S. Patent Application 14/376,939, 8 June 2014. [Google Scholar]
- FOODsniffer. Available online: http://www.myfoodsniffer.com/ (accessed on 20 March 2017).
- Sundvor, S.; Portela, S.; Ward, J.; Walton, J. System and Method for Detection of Target Substances. U.S. Patent Application 15/265,171, 26 September 2016. [Google Scholar]
- Nima-A Portable Gluten Tester | Nima. Available online: https://nimasensor.com/ (accessed on 20 March 2017).
- Antila, J.; Kantojärvi, U.; Mäkynen, J. Optical Measurement System. WO2016071572A1 2016. [Google Scholar]
- Food Scanner. Available online: https://www.spectralengines.com/products/food-scanner (accessed on 23 March 2017).
- Watson, W.; Correa, I. Analyzing and Correlating Spectra, Identifying Samples and Their Ingredients, and Displaying Related Personalized Information. US Patent 9,212,996, 5 August 2015. [Google Scholar]
- Tellspec–Beam Your Health Up–TellSpec. Available online: http://tellspec.com/en/ (accessed on 20 March 2017).
- Goldring, D.; Sharon, D. Low-Cost Spectrometry System for End-User Food Analysis. US Patent 9,377,396, 31 October 2016. [Google Scholar]
- Consumer Physics. Available online: https://www.consumerphysics.com/myscio/ (accessed on 20 March 2017).
- Contreras-Naranjo, J.; Wei, Q. Mobile phone-based microscopy, sensing, and diagnostics. IEEE J. Sel. 2016, 22, 392–405. [Google Scholar] [CrossRef]






| Detection Target | Methodology | Materials | LoD/Test Time/Performance | Smartphone Use | Reference |
|---|---|---|---|---|---|
| Escherichia coli in water | Fluorescent imaging | Antibody, quantum dots, UV LED | 5–10 CFU mL−1 | Cellphone imaging with camera attachment | [11] |
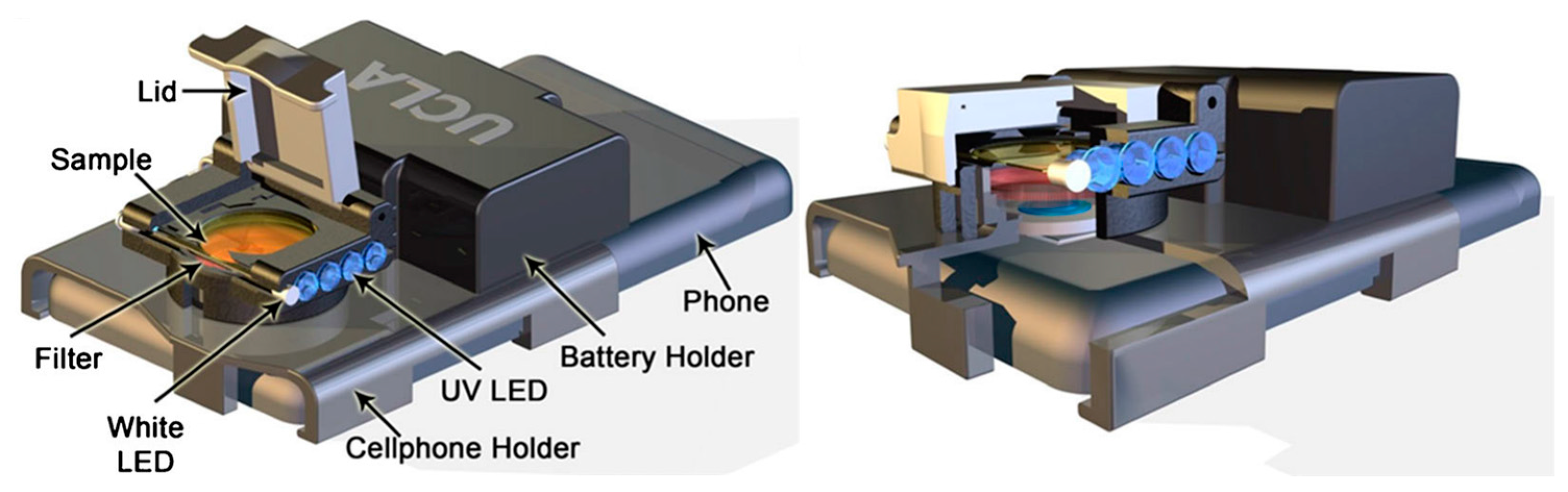
| rbST antibodies in milk | Microsphere fluorescent immunoassay | Antibody, quantum dots, UV LED and white LED | 80% true-positive rate and 95% true-negative rate | Cellphone imaging with camera attachment | [12] |
| Lactose and galactose in undiluted food samples | Engineered bacteria fluorescence | Blue light and optical filter | 1–1000 mM | Cellphone imaging | [13] |
| Peanut allergen in food samples | Colorimetric assays | ELISA allergen test kit, cellphone attachment with 2 test tubes and 2 LEDs | ~1 parts per million (ppm), 20-min preparation phase | Cellphone assay with camera attachment | [14] |
| Aflatoxin B1 in maize | Lateral flow immunoassay | Paper strip, close-up lens and a white LED | 5 μg/kg | Smartphone imaging via LFIA reader adapter | [15] |
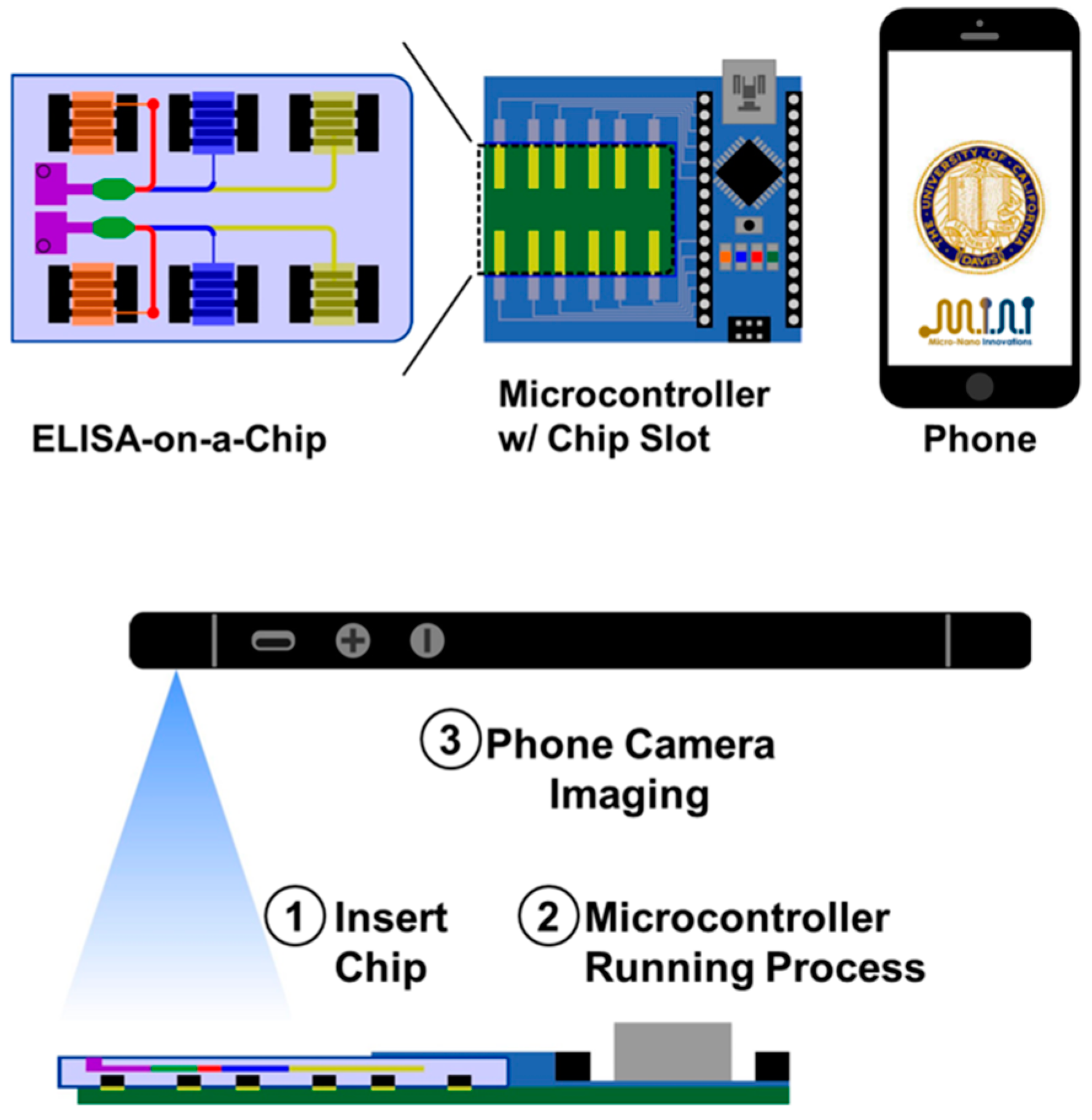
| BDE-47 in food sample | Microfluidics and competitive ELISA | Arduino Nano, PCB, microfluidic chip | Readout time of 15 min and input sample volume considerably reduced | Smartphone as power source, imaging reader and cloud sender | [16] |
| Red wine properties | Paper microfluidics, colorimetric assay, and PCA | Chemical dyes | Successful distinction of red wines by their grape varieties and oxidation. | Smartphone imaging | [17] |
| Amines as indication of foodborne pathogens in meat | Membrane technology colorimetry and unsupervised chemometric tools | Dyes | Down to 1 ppm concentration of amine | Camera imaging | [18] |
| Antibiotic residues in milk | SPE and fluorescence spectroscopy | Photography lightbox with fluorescent light | LoC 0.50 mL−1 and LoQ 1.50 µg mL−1 | Smartphone camera used as spectrometer | [19] |
| Glutamate in food compound, instant soup and wines | Paper-based colorimetric assay | Glutamate-specific enzyme | 0.028 mmol L−1 | Camera acquisition and analysis | [20] |
| ALP as indicator of incorrect milk pasteurization | Disposable lateral flow-through strip | Sample pad | 0.1 U L−1, within 10 min with a detection range of 0.1–150 U L−1 | Image acquisition and Matlab analysis | [21] |
| OA and STX in shellfish | Competitive immunoassay strip | 3D-printed smartphone strip adapter | 2.800 ng mL−1 for OA and 9.808 ng mL−1 for STX in 30 min | Camera acquisition via strip adapter and data processing | [22] |
| Fluoride in water | Colorimetric imaging | Compact sample chamber adapter for smartphone | Linear range 0–2 mg L−1 | Smartphone colorimeter | [23] |
| Catechols in water | Colorimetric imaging | 96-well sensor array, light-tight box, white LED | PCA, HCA and LDA for quality discrimination and PLS for quantitative determination | Smartphone colorimeter coupled to remote server | [24] |
| V. parahaemolyticus in fish samples | Colorimetric immunoassay | Biosensor cartridge, lens-free CMOS image sensor, Wi-Fi module | 1.4 × 104 CFU mL−1 | Dedicated app to operate the system and upload on internet server | [25] |
| Escherichia coli O157:H7 and Salmonella enterica | DNA transduction on microfluidic device | Magnetic beads | Down to 20 genomic copies of E. coli | Custom written app for cell phone image analysis | [26] |
| Clenbuterol | Electric field-driven immunoreaction | Functionalized electrodes | 0.076 ng mL−1 CLB in 6 min | USB Smartphone tool biochip | [27] |
| Pattern recognition of Brazilian honey samples | Cyclic voltammetry assay | Electrode of gold, homemade potentiostat with USB connection and Bluetooth module | Successfully generation of voltammetric fingerprints of numerous honey samples | Chemometric data processing on smartphone | [28] |
| Detection Target | Methodology | Materials | LoD/Performance | Smartphone Use | Reference |
|---|---|---|---|---|---|
| Microbial spoilage on beef | Mie scattering | Positioning stages, 880 nm NIR LED | 101 CFU mL−1 to 108 CFU mL−1 | Built-in gyro sensor and camera spectroscopy | [32] |
| Generic application | Spectroscopic colorimetry | 3D printed housing, LED array, Phidgets board, and VIS-spectrometer | Good agreement to certified spectra with dE/E ranging from 0.5% to 1.5% | IoT device to be used with smartphone | [33] |
| Glucose and ethanol in alcoholic beverages | FTIR spectroscopy and independent component analysis | Graphite light source, ATR prisms, 2-dimensional light receiving device for smartphone | Wavelength resolution 0.057 μm | Proposed as a bean-size spectroscopic module to be mounted on smartphones | [34] |
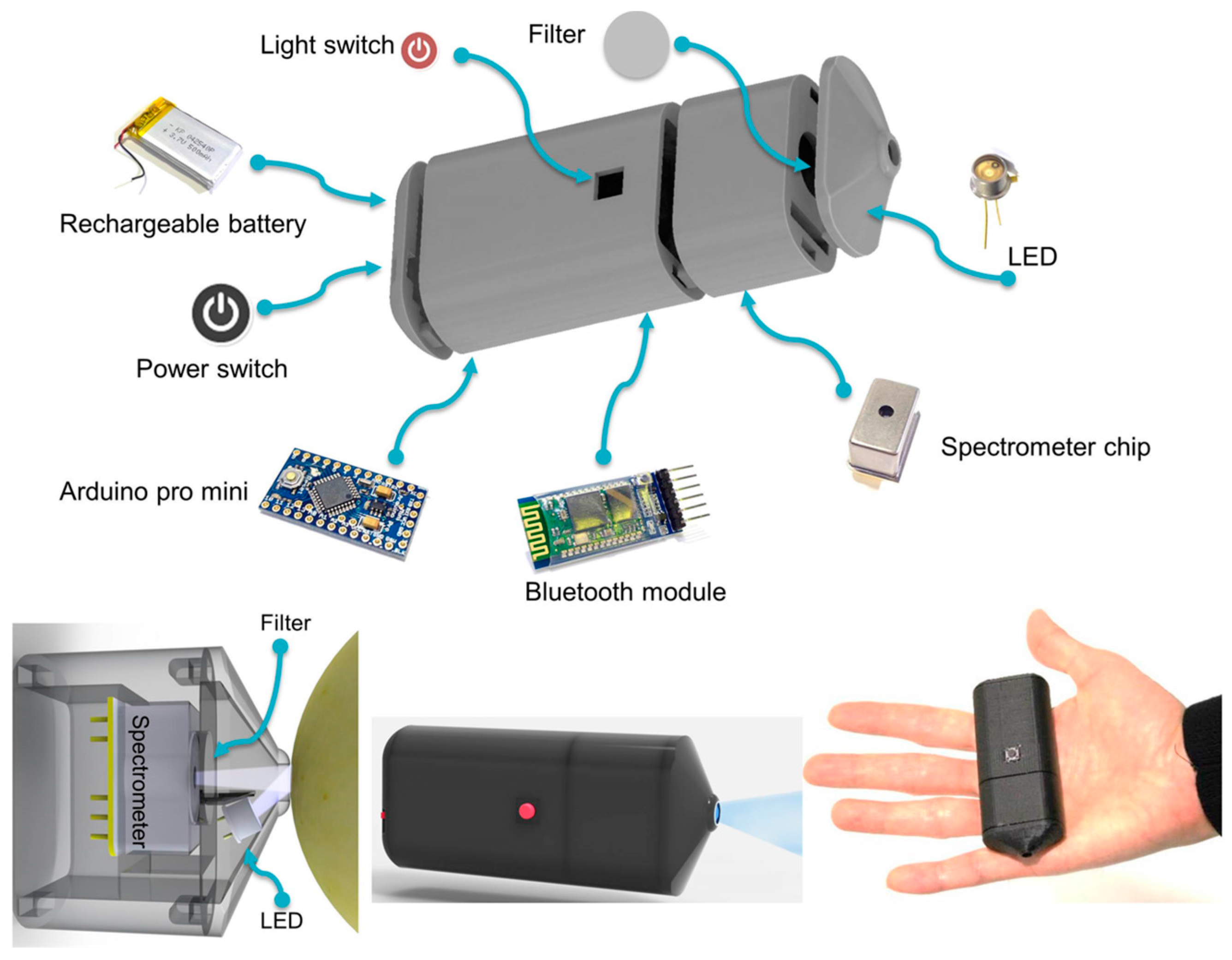
| ChlF detection in a variety of apple samples | UV fluorescence spectroscopy | UV LED, nozzle-like enclosure VIS-spectrometer, Arduino pro mini µ, Bluetooth | Satisfactory agreement observed between ripeness and fluorescence signals | Dedicated app interface on smartphone to communicate, receive, plot, and analyse spectral data | [35] |
| E. coli contamination on meat | Fluorescence-based imaging | 4405-nm 10 W LEDs, CCD camera, optical filter at 670 nm, and Wi-Fi transmitter | Localization of most fecal contamination spots successfully identified | Outlined real-time broadcasting to monitoring device such as smartphone | [36] |
| Generic food sensing application | Hyper-spectral imaging | Tunable MEMS FPI, Bluetooth | Operation range 450–550 nm with spectral resolution 8–15 nm @FWHM | Mobile phone-compatible hyper-spectral imager | [37] |
| Food quality testing | Diffractive interference refractometry | 5 mW semiconductor red laser, circular spatial filter, Si detectors, and a PDMS device | LoD of 4 × 10−4 RIU | Outlined smartphone interface based on transmission mode configuration | [38] |
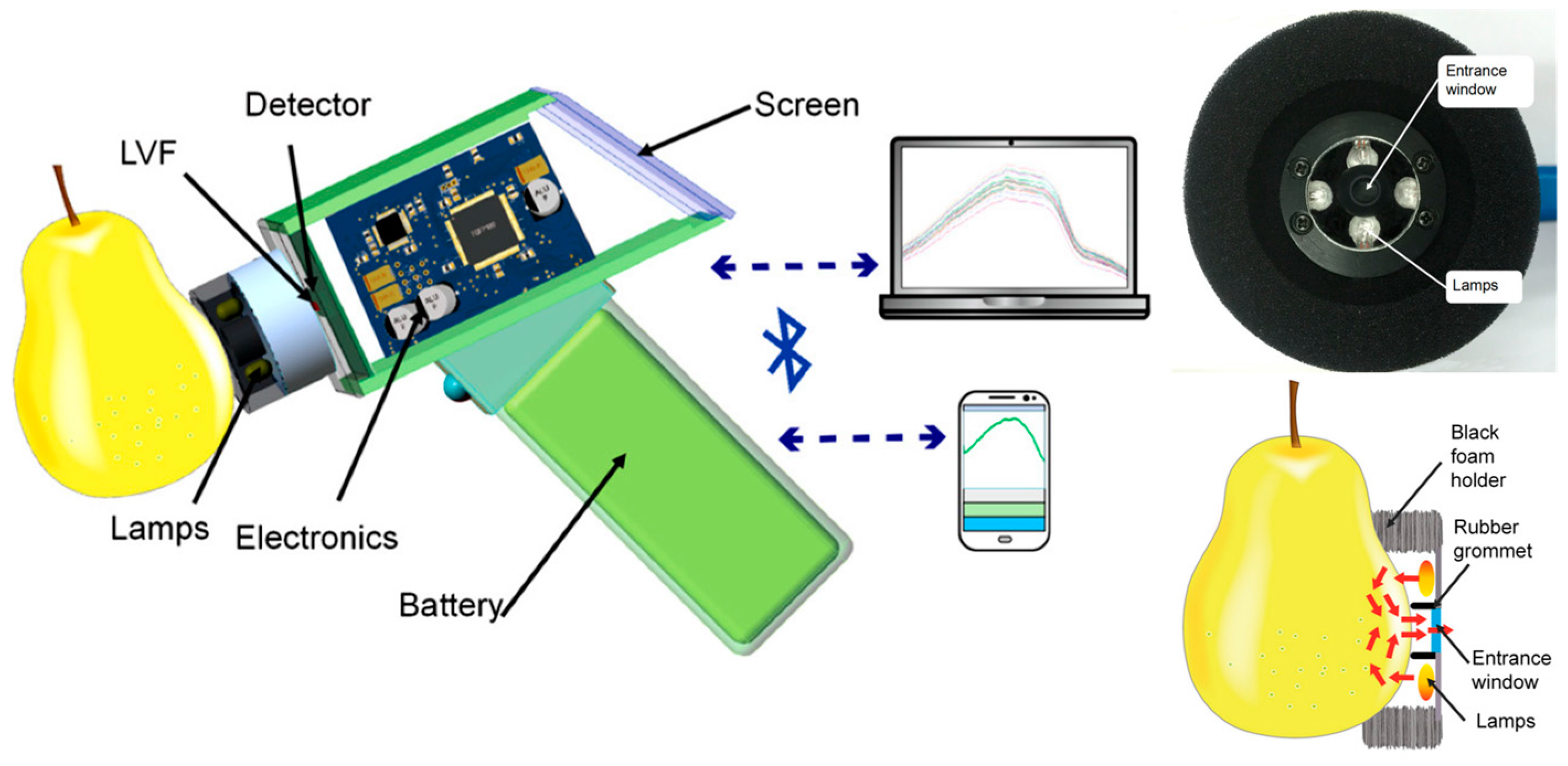
| Sugar content prediction in pears | NIR spectrometry and PLS | 4 tungsten lamps, LVF 620–1080 nm and CMOS linear detector array | Low power, SNR ratio up to 5000, R2 0.96, SEC 0.29° Bx and SEP 0.46° Bx | Instrument wirelessly operated with smartphone | [39] |
| Platform | Approximate Cost | Reference |
|---|---|---|
| Smartphone with fluorescence microscope attachment | Attachment of around $140, significantly reduced compared to the equipment costs for the reference method | [12] |
| Akvo Caddisfly | Expected to retail at $75, without the phone and mapping system, plus $0.3 for each test | [23] |
| PiBA assay coupled to LAMP | Reagent cost for PiBA is a fraction of a cent. Overall cost reduction is ~10-fold respect to the reference (fluorescence reagents for qPCR) | [26] |
| Smartphone-based analytical platform with homemade potentiostat | Based on CheapStat potentiostat which requires less than eighty dollars for its manufacturing, while the most commercial potentiostats cost a few thousands of dollars | [28] |
| Smartphone spectrometer | Entire assembly along with the smartphone can be realized under $250, while reference spectrometer platforms costs are $4000 and $1200 | [32] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rateni, G.; Dario, P.; Cavallo, F. Smartphone-Based Food Diagnostic Technologies: A Review. Sensors 2017, 17, 1453. https://doi.org/10.3390/s17061453
Rateni G, Dario P, Cavallo F. Smartphone-Based Food Diagnostic Technologies: A Review. Sensors. 2017; 17(6):1453. https://doi.org/10.3390/s17061453
Chicago/Turabian StyleRateni, Giovanni, Paolo Dario, and Filippo Cavallo. 2017. "Smartphone-Based Food Diagnostic Technologies: A Review" Sensors 17, no. 6: 1453. https://doi.org/10.3390/s17061453
APA StyleRateni, G., Dario, P., & Cavallo, F. (2017). Smartphone-Based Food Diagnostic Technologies: A Review. Sensors, 17(6), 1453. https://doi.org/10.3390/s17061453






