Development of Diamond and Silicon MEMS Sensor Arrays with Integrated Readout for Vapor Detection
Abstract
:1. Introduction
2. Material and Methods
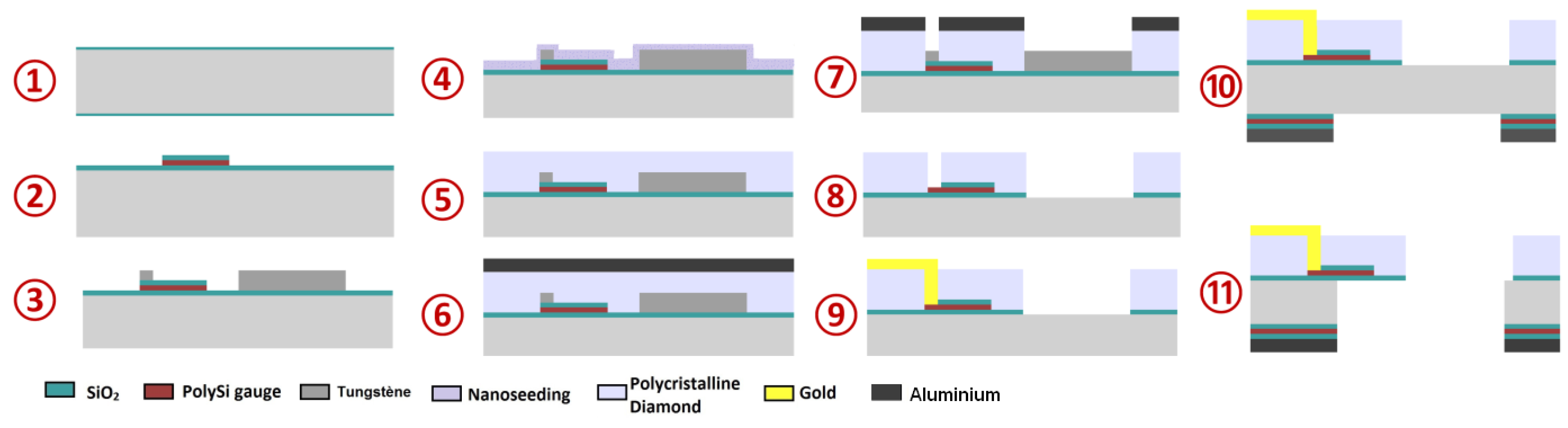
2.1. Microcantilevers
2.2. Microcantilever Functionalization
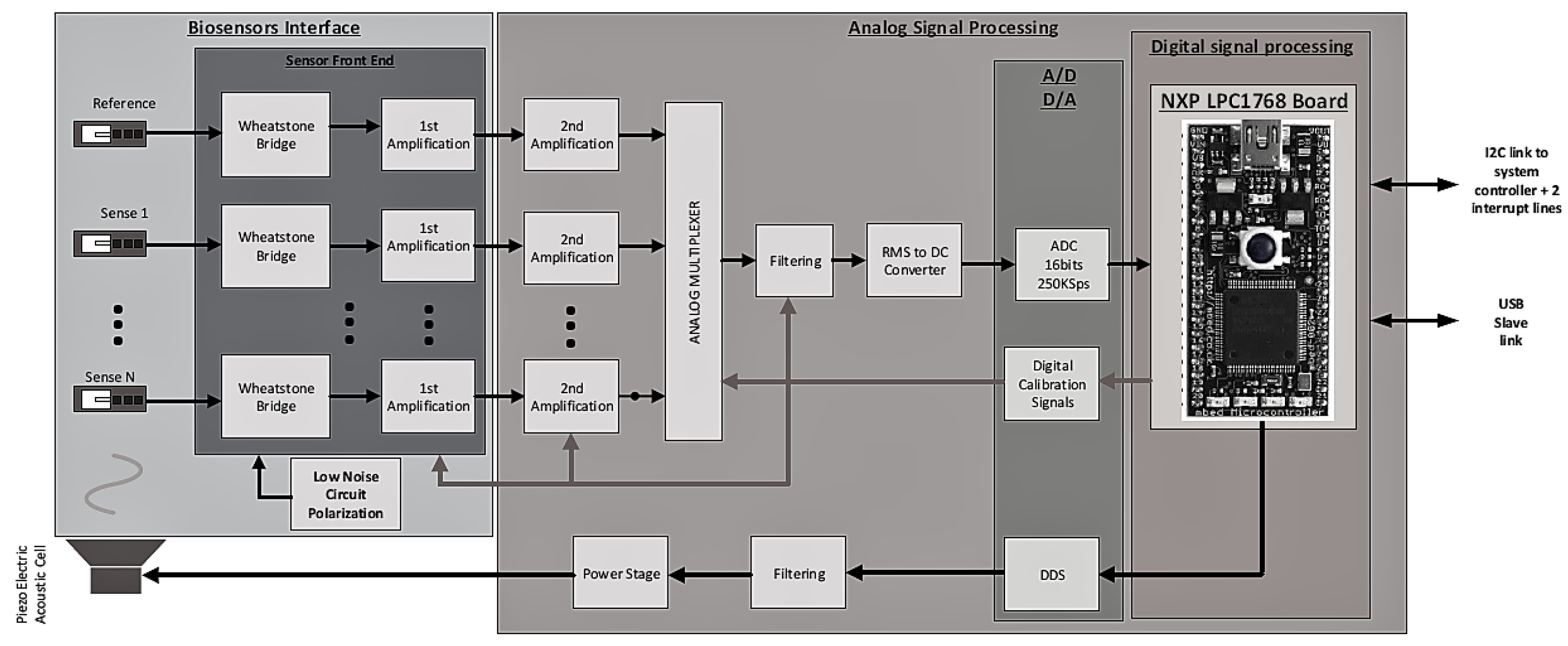
2.3. Gas Cell and Electronic System
- -
- Small dimensions: for electronic nose applications, it is not possible to use a measuring instrument such as gain-phase analyzers;
- -
- A human–machine interface: an interface is necessary for the visualization of the data in real time and for the intervention on the measurement configurations;
- -
- An analog processing interface to detect the responses of the sensors with sufficient resolution;
- -
- A memory to store a certain quantity of measurements and calibration data of the sensors;
- -
- An autonomous architecture: calibration and diagnostics executed autonomously, without user intervention;
- -
- Communication interfaces: to communicate with other modules (pump control system, human–machine interface, etc).
3. Experimental
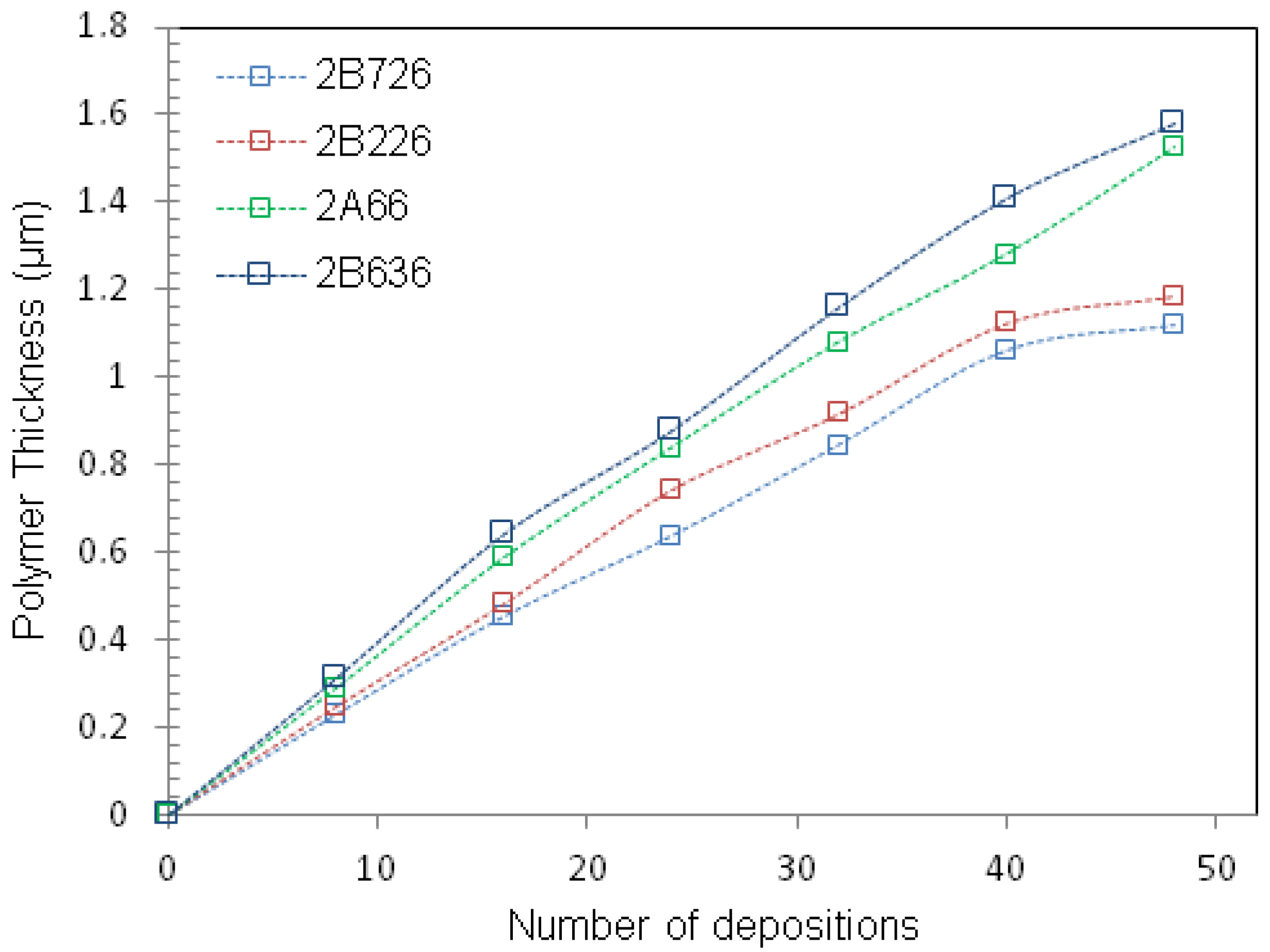
3.1. Sensor Preparations
3.2. Measurement Set-Up
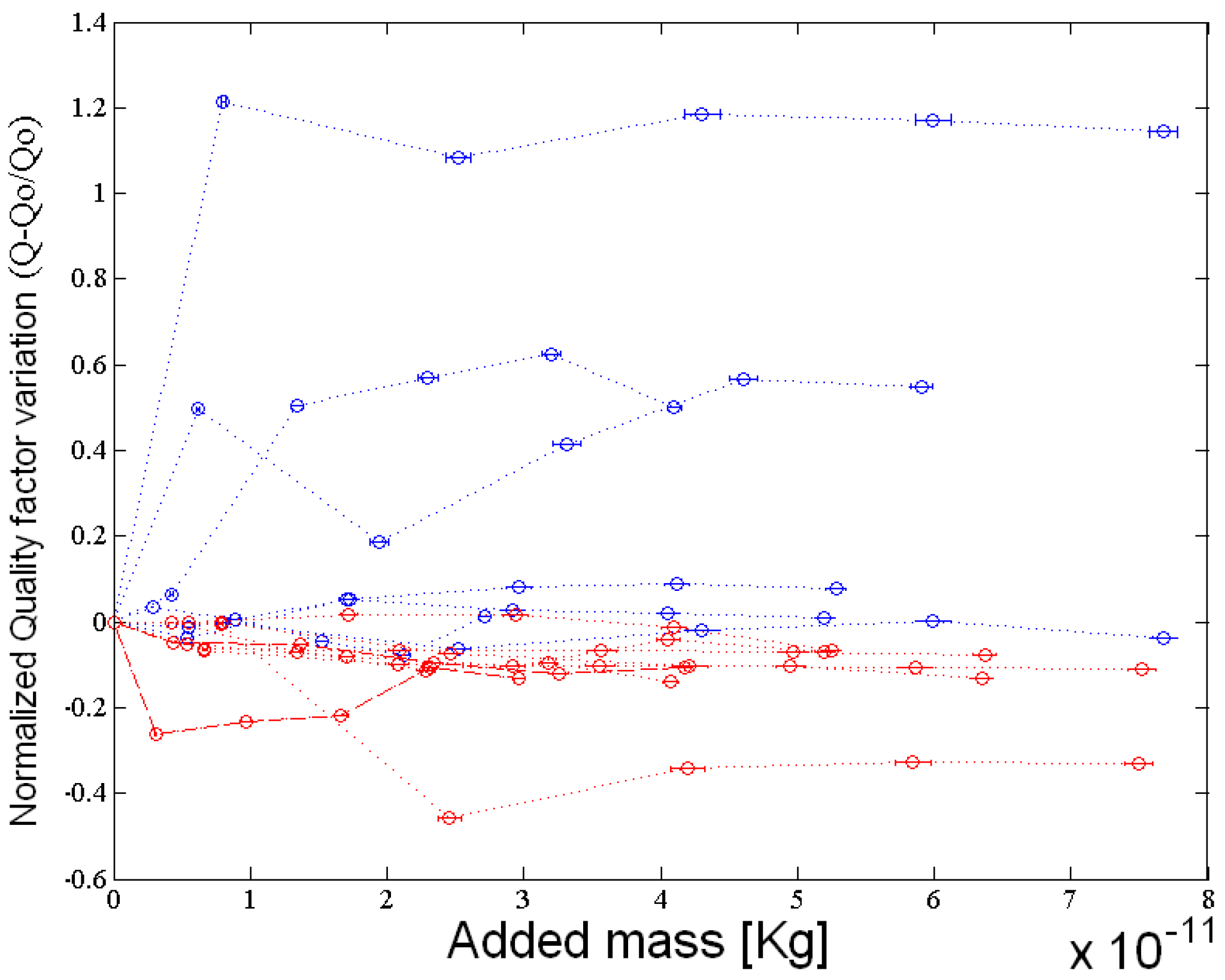
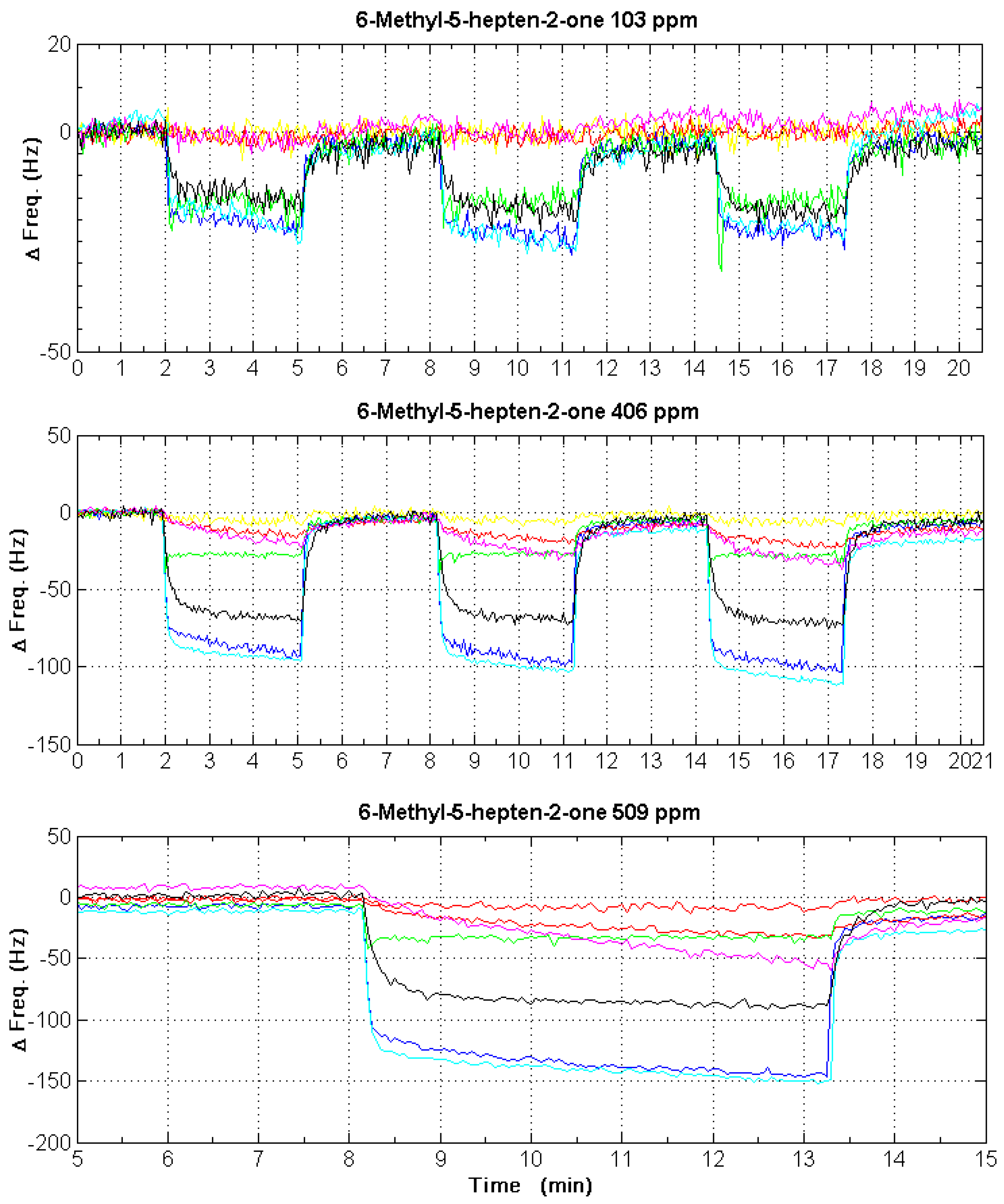
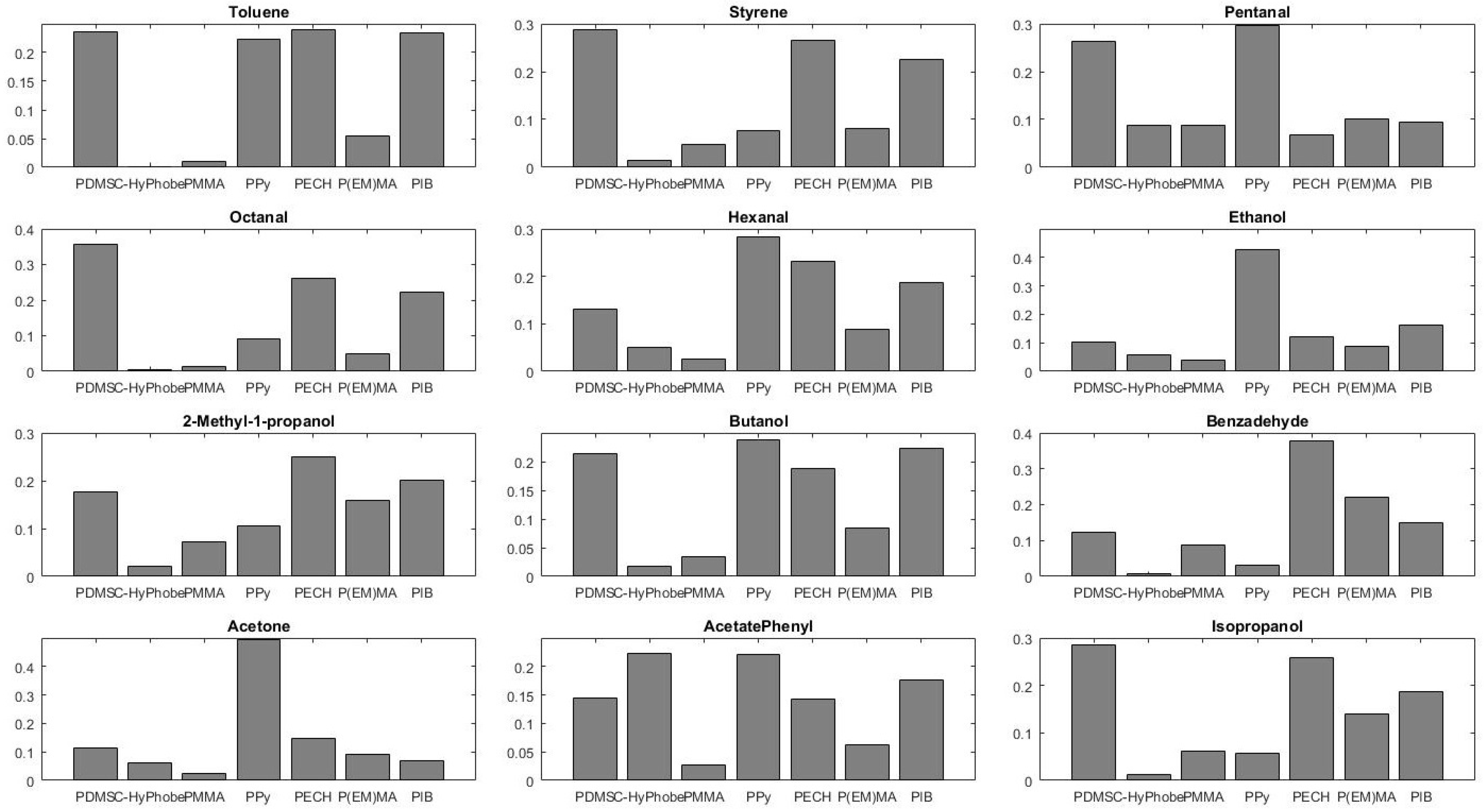
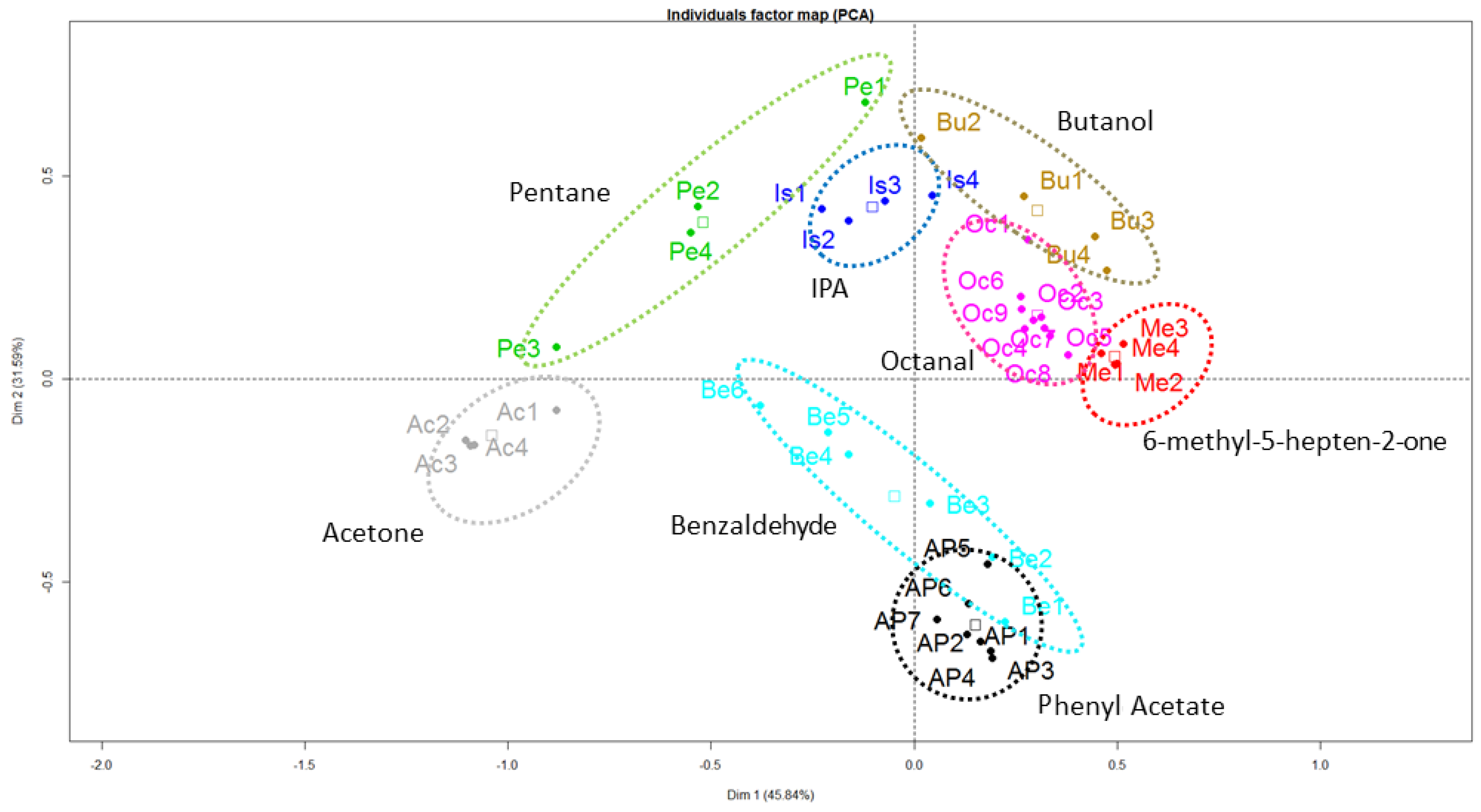
4. Results
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bushdid, C.; Magnasco, M.O.; Vosshall, L.B.; Keller, A. Humans can discriminate more than 1 trillion olfactory stimuli. Science 2014, 343, 1370–1372. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Matsunami, H. Mechanisms of olfaction. In Bioelectronic Nose; Springer: Berlin, Germany, 2014; pp. 23–45. [Google Scholar]
- Schaller, E.; Bosset, J.O.; Escher, F. ‘Electronic noses’ and their application to food. LWT Food Sci. Technol. 1998, 31, 305–316. [Google Scholar] [CrossRef]
- Delpha, C.; Siadat, M.; Lumbreras, M. An electronic nose for the discrimination of forane 134a and carbon dioxide in a humidity controlled atmosphere. Sens. Actuators B Chem. 2001, 78, 49–56. [Google Scholar] [CrossRef]
- Yinon, J. Peer reviewed: detection of explosives by electronic noses. Anal. Chem. 2003, 75, 98A. [Google Scholar] [CrossRef]
- Dragonieri, S.; Schot, R.; Mertens, B.J.; Le Cessie, S.; Gauw, S.A.; Spanevello, A.; Resta, O.; Willard, N.P.; Vink, T.J.; Rabe, K.F.; et al. An electronic nose in the discrimination of patients with asthma and controls. J. Allergy Clin. Immunol. 2007, 120, 856–862. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Feng, L.; Kemling, J.W.; Musto, C.J.; Suslick, K.S. An optoelectronic nose for the detection of toxic gases. Nat. Chem. 2009, 1, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Montuschi, P.; Santonico, M.; Mondino, C.; Pennazza, G.; Mantini, G.; Martinelli, E.; Capuano, R.; Ciabattoni, G.; Paolesse, R.; Di Natale, C.; et al. Diagnostic performance of an electronic nose, fractional exhaled nitric oxide, and lung function testing in asthma. CHEST J. 2010, 137, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.D.; Baietto, M. Advances in electronic-nose technologies developed for biomedical applications. Sensors 2011, 11, 1105–1176. [Google Scholar] [CrossRef] [PubMed]
- Persaud, K.; Dodd, G. Analysis of discrimination mechanisms in the mammalian olfactory system using a model nose. Nature 1982, 299, 352–355. [Google Scholar] [CrossRef] [PubMed]
- Gardner, J.W.; Bartlett, P.N. Sensors and Sensory Systems for an Electronic Nose; Springer: Berlin, Germany, 1992. [Google Scholar]
- Hatfield, J.; Neaves, P.; Hicks, P.; Persaud, K.; Travers, P. Towards an integrated electronic nose using conducting polymer sensors. Sens. Actuators B Chem. 1994, 18, 221–228. [Google Scholar] [CrossRef]
- Yang, Y.M.; Yang, P.Y.; Wang, X.R. Electronic nose based on SAWS array and its odor identification capability. Sens. Actuators B Chem. 2000, 66, 167–170. [Google Scholar] [CrossRef]
- El Barbri, N.; Mirhisse, J.; Ionescu, R.; El Bari, N.; Correig, X.; Bouchikhi, B.; Llobet, E. An electronic nose system based on a micro-machined gas sensor array to assess the freshness of sardines. Sens. Actuators B Chem. 2009, 141, 538–543. [Google Scholar] [CrossRef]
- McDonagh, C.; Burke, C.S.; MacCraith, B.D. Optical chemical sensors. Chem. Rev. 2008, 108, 400–422. [Google Scholar] [CrossRef] [PubMed]
- Lang, H.P.; Hegner, M.; Gerber, C. Cantilever array sensors. Mater. Today 2005, 8, 30–36. [Google Scholar] [CrossRef]
- Lavrik, N.V.; Sepaniak, M.J.; Datskos, P.G. Cantilever transducers as a platform for chemical and biological sensors. Rev. Sci. Instrum. 2004, 75, 2229–2253. [Google Scholar] [CrossRef]
- Possas, M.; Rousseau, L.; Ghassemi, F.; Lissorgues, G.; Scorsone, E.; Bergonzo, P. Fabrication and micromechanical characterization of polycrystalline diamond microcantilevers. In Proceedings of the 2014 Symposium on Design, Test, Integration and Packaging of MEMS/MOEMS (DTIP), Cannes, France, 1–4 April 2014; pp. 1–5. [Google Scholar]
- Manai, R.; Scorsone, E.; Rousseau, L.; Ghassemi, F.; Abreu, M.P.; Lissorgues, G.; Tremillon, N.; Ginisty, H.; Arnault, J.; Tuccori, E.; et al. Grafting odorant binding proteins on diamond bio-MEMS. Biosens. Bioelectron. 2014, 60, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Qin, G.; Darne, C.; Cai, C.; Wosik, W.; Pei, S.S. Fabrication of short and thin silicon cantilevers for AFM with SOI wafers. Sens. Actuators A Phys. 2006, 126, 369–374. [Google Scholar] [CrossRef]
- Scorsone, E.; Saada, S.; Arnault, J.; Bergonzo, P. Enhanced control of diamond nanoparticle seeding using a polymer matrix. J. Appl. Phys. 2009, 106, 014908. [Google Scholar] [CrossRef]
- Fritz, J.; Baller, M.; Lang, H.; Rothuizen, H.; Vettiger, P.; Meyer, E.; Güntherodt, H.J.; Gerber, C.; Gimzewski, J. Translating biomolecular recognition into nanomechanics. Science 2000, 288, 316–318. [Google Scholar] [CrossRef] [PubMed]
- Arntz, Y.; Seelig, J.D.; Lang, H.; Zhang, J.; Hunziker, P.; Ramseyer, J.; Meyer, E.; Hegner, M.; Gerber, C. Label-free protein assay based on a nanomechanical cantilever array. Nanotechnology 2002, 14, 86. [Google Scholar] [CrossRef]
- Thundat, T.; Wachter, E.; Sharp, S.; Warmack, R. Detection of mercury vapor using resonating microcantilevers. Appl. Phys. Lett. 1995, 66, 1695–1697. [Google Scholar] [CrossRef]
- Baselt, D.; Fruhberger, B.; Klaassen, E.; Cemalovic, S.; Britton, C.; Patel, S.; Mlsna, T.; McCorkle, D.; Warmack, B. Design and performance of a microcantilever-based hydrogen sensor. Sens. Actuators B Chem. 2003, 88, 120–131. [Google Scholar] [CrossRef]
- Maute, M.; Raible, S.; Prins, F.; Kern, D.; Ulmer, H.; Weimar, U.; Göpel, W. Detection of volatile organic compounds (VOCs) with polymer-coated cantilevers. Sens. Actuators B Chem. 1999, 58, 505–511. [Google Scholar] [CrossRef]
- Zimmermann, C.; Rebière, D.; Dejous, C.; Pistré, J.; Chastaing, E.; Planade, R. A love-wave gas sensor coated with functionalized polysiloxane for sensing organophosphorus compounds. Sens. Actuators B Chem. 2001, 76, 86–94. [Google Scholar] [CrossRef]
- Bietsch, A.; Zhang, J.; Hegner, M.; Lang, H.P.; Gerber, C. Rapid functionalization of cantilever array sensors by inkjet printing. Nanotechnology 2004, 15, 873. [Google Scholar] [CrossRef]
- Urbiztondo, M.; Peralta, A.; Pellejero, I.; Sesé, J.; Pina, M.; Dufour, I.; Santamaria, J. Detection of organic vapours with Si cantilevers coated with inorganic (zeolites) or organic (polymer) layers. Sens. Actuators B Chem. 2012, 171, 822–831. [Google Scholar] [CrossRef]
- Härtl, A.; Schmich, E.; Garrido, J.A.; Hernando, J.; Catharino, S.C.; Walter, S.; Feulner, P.; Kromka, A.; Steinmüller, D.; Stutzmann, M. Protein-modified nanocrystalline diamond thin films for biosensor applications. Nat. Mater. 2004, 3, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Carlisle, J.A. Covalent immobilization of glucose oxidase on conducting ultrananocrystalline diamond thin films. Diamond Relat. Mater. 2006, 15, 279–284. [Google Scholar] [CrossRef]
- Salmi, Z.; Lamouri, A.; Decorse, P.; Jouini, M.; Boussadi, A.; Achard, J.; Gicquel, A.; Mahouche-Chergui, S.; Carbonnier, B.; Chehimi, M.M. Grafting polymer–protein bioconjugate to boron-doped diamond using aryl diazonium coupling agents. Diamond Relat. Mater. 2013, 40, 60–68. [Google Scholar] [CrossRef]
- Manai, R.; Habchi, M.; Kamouni-Belghiti, D.; Persuy, M.; Rousseau, L.; Abreu, M.P.; Grebert, D.; Badonnel, K.; Bergonzo, P.; Pajot-Augy, E.; et al. Diamond micro-cantilevers as transducers for olfactory receptors-based biosensors: Application to the receptors M71 and OR7D4. Sens. Actuators B Chem. 2017, 238, 1199–1206. [Google Scholar] [CrossRef]
- Dyer, D.C.; Gardner, J.W. High-precision intelligent interface for a hybrid electronic nose. Sens. Actuators A Phys. 1997, 62, 724–728. [Google Scholar] [CrossRef]
- Depari, A.; Ferrari, P.; Flammini, A.; Marioli, D.; Rosa, S.; Taroni, A. A new modular approach for low-cost electronic noses. In Proceedings of the Instrumentation and Measurement Technology Conference, Sorrento, Italy, 24–27 April 2006; pp. 578–583. [Google Scholar]
Sample Availability: Samples of the fabricated silicon and polycristalline diamond sensors are available from the authors. |





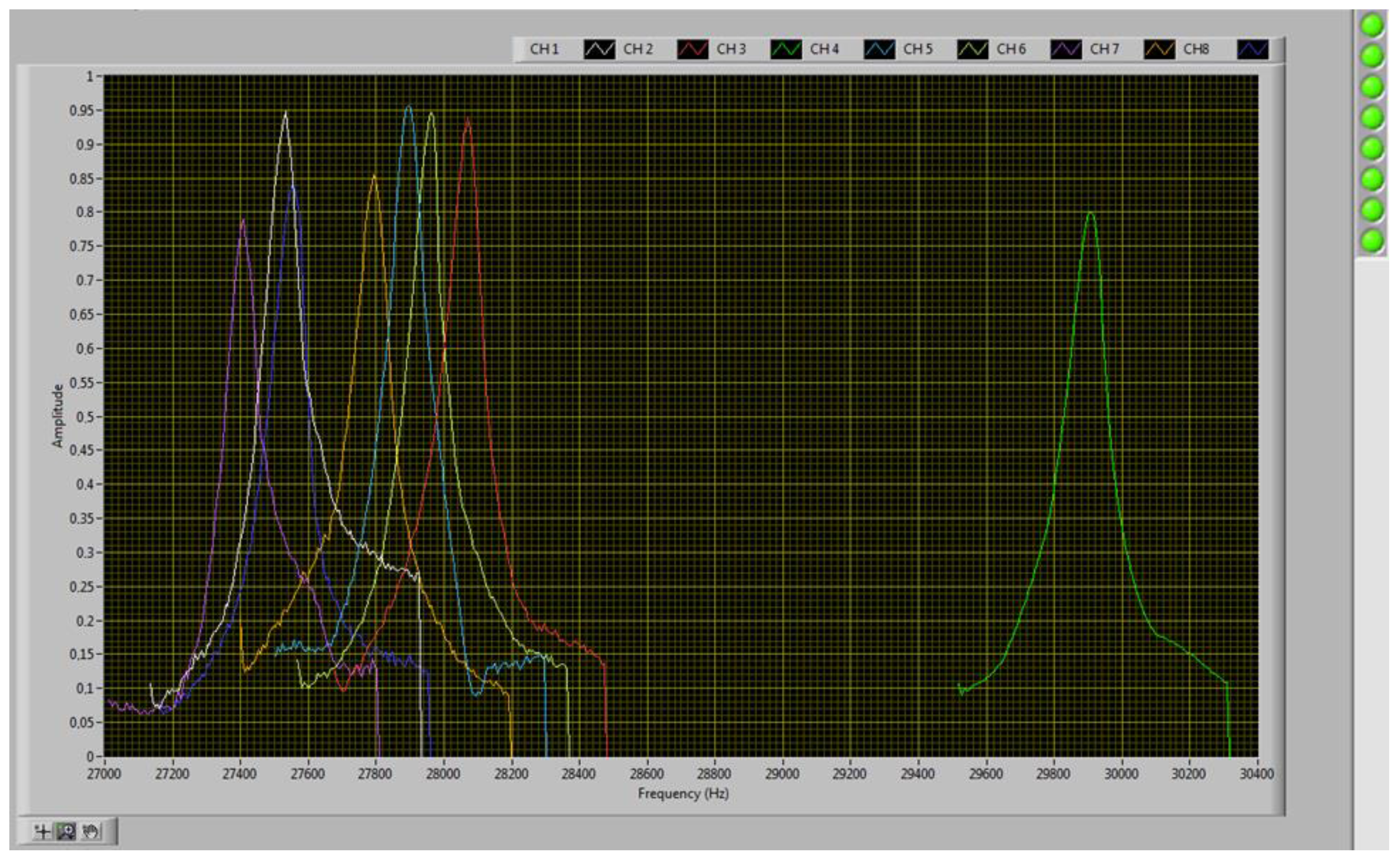

| Cantilever | Material | Sensitive Layer (Solvent > 90%) | Resonant Frequency [Hz] | |
|---|---|---|---|---|
| 1 | Silicon | PDMS | Polydimethylsiloxane | 113,954 |
| 2 | Diamond | Hydrophile treatment | - | 28,026 |
| 3 | Diamond | Hydrophobic treatment | - | 31,549 |
| 4 | Silicon | PMMA | Polymethylmethacrylate | 127,136 |
| 5 | Silicon | PAC | Poly(acetylene) | 59,769 |
| 6 | Silicon | PECH | Polyepichlorohydrin | 126,627 |
| 7 | Silicon | P(EM)MA | Poly(ethylene-co-methyl methacrylate) | 124,236 |
| 8 | Silicon | PIB | Polyisobutylene | 125,388 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Possas-Abreu, M.; Ghassemi, F.; Rousseau, L.; Scorsone, E.; Descours, E.; Lissorgues, G. Development of Diamond and Silicon MEMS Sensor Arrays with Integrated Readout for Vapor Detection. Sensors 2017, 17, 1163. https://doi.org/10.3390/s17061163
Possas-Abreu M, Ghassemi F, Rousseau L, Scorsone E, Descours E, Lissorgues G. Development of Diamond and Silicon MEMS Sensor Arrays with Integrated Readout for Vapor Detection. Sensors. 2017; 17(6):1163. https://doi.org/10.3390/s17061163
Chicago/Turabian StylePossas-Abreu, Maira, Farbod Ghassemi, Lionel Rousseau, Emmanuel Scorsone, Emilie Descours, and Gaelle Lissorgues. 2017. "Development of Diamond and Silicon MEMS Sensor Arrays with Integrated Readout for Vapor Detection" Sensors 17, no. 6: 1163. https://doi.org/10.3390/s17061163
APA StylePossas-Abreu, M., Ghassemi, F., Rousseau, L., Scorsone, E., Descours, E., & Lissorgues, G. (2017). Development of Diamond and Silicon MEMS Sensor Arrays with Integrated Readout for Vapor Detection. Sensors, 17(6), 1163. https://doi.org/10.3390/s17061163






