A Force-Visualized Silicone Retractor Attachable to Surgical Suction Pipes
Abstract
:1. Introduction
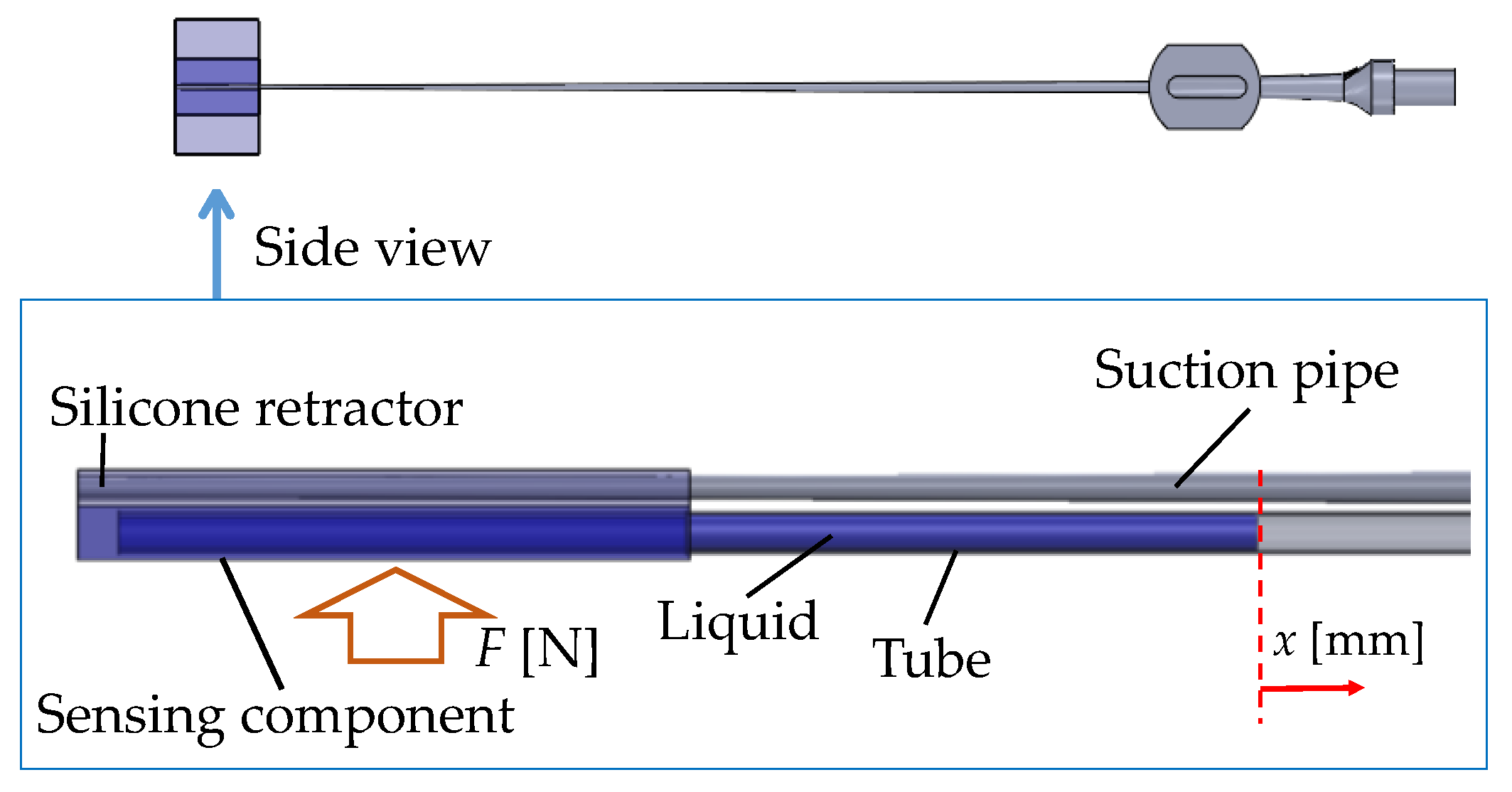
- Force visualization mechanism utilizing a liquid: A liquid was installed into the sensing component of the silicone retractor. A transparent tube was connected to the sensing component to observe the liquid level, which is displaced according to the load. The load is visually measurable by the liquid displacement.
- High usability: The silicone retractor is easily set up by installing a suction pipe into the hole in the retractor. The retractor was composed entirely of silicone and connected to PTFE tubes. Thus, the benefits of the system include the absence of electrical components, disposability, easy sterilization/disinfection, magnetic resonance imaging (MRI) compatibility, and low cost. The sensor was installed in only one area where the largest force is exerted to minimize the damage to the brain tissue. Moreover, the fact that there is only one sensing area minimizes the space occupied by the sensor and enhances the usability of the device.
- Multiple functions: The proposed instrument provides the functions of retraction, suction, and force sensing via attachment to the suction pipe.
- Hybrid soft and hard structure: The silicone retractor must be stiff for retracting tissues, while the sensing component inside the retractor must be soft to provide high sensitivity. To realize both these functions, a hybrid soft and hard structure was adapted. A thin retractor was developed using hard retracting and soft sensing parts.
2. Related Work
3. Force-Sensible Silicone Retractor Based on Force Visualization Utilizing a Liquid
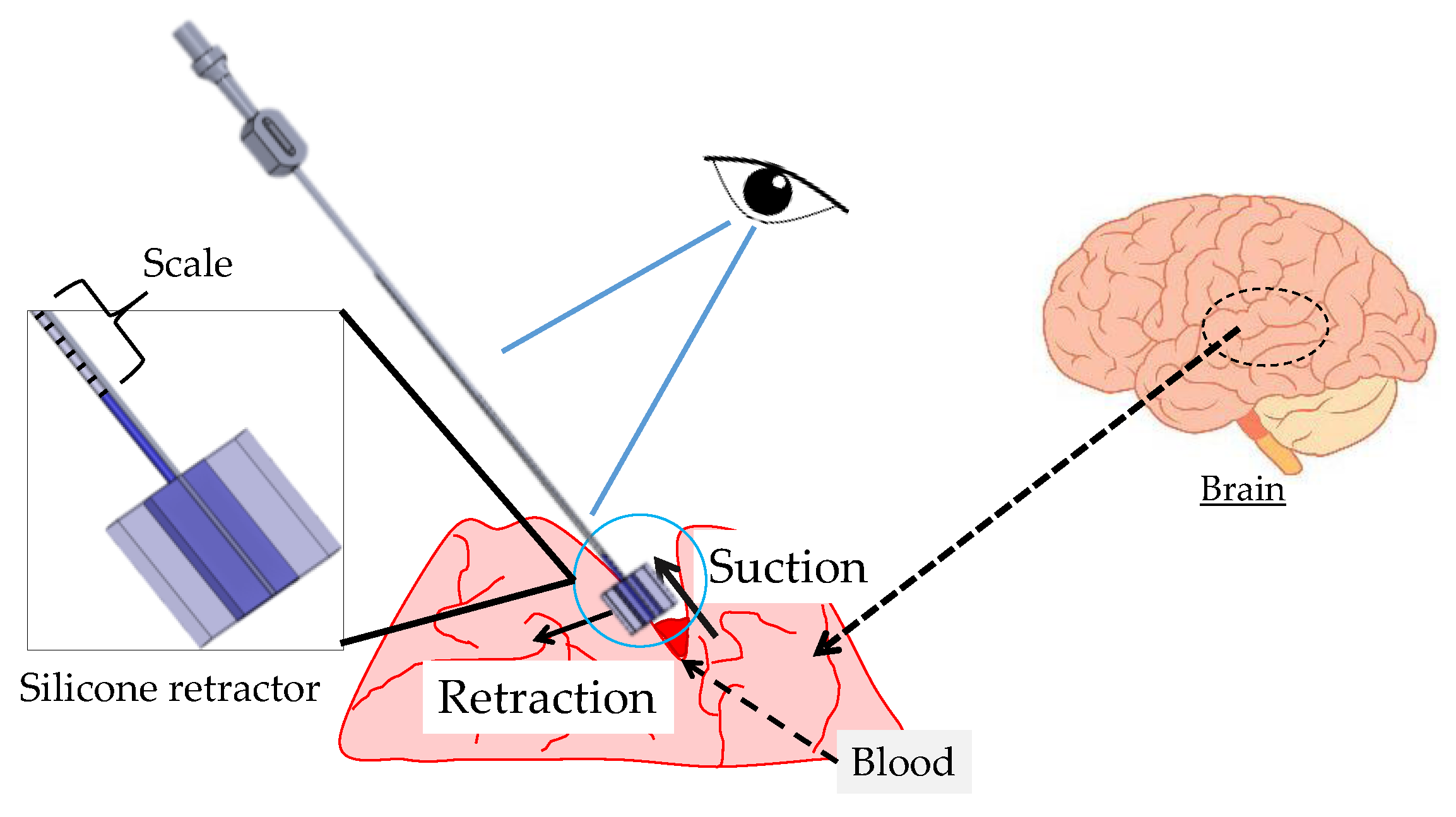
3.1. Target Situation
3.2. Design Requirements
- Suction, retraction, and retracting force measurement are available simultaneously.
- The silicone retractor is attachable to the suction pipe.
- There is no electric component in the silicone retractor.
- The force is visualized.
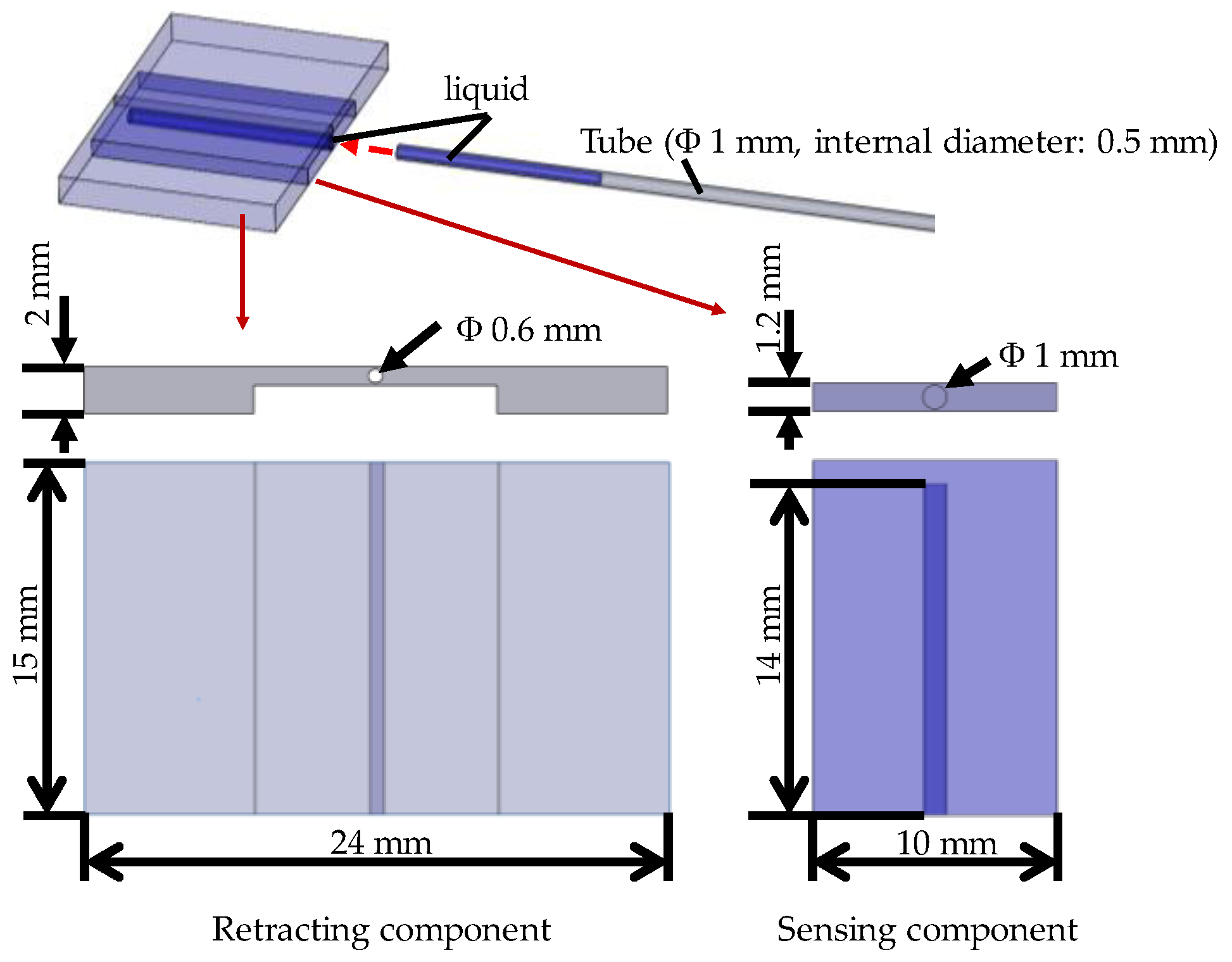
- The dimensions should include a width of less than 25 mm, length of less than 15 mm, and thickness of less than 2 mm.
- The force range should be 0.00–1.00 N, the resolution should be less than 0.05 N, and the minimum sensed force should be 0.10 N.
3.3. Principle of Force Sensing
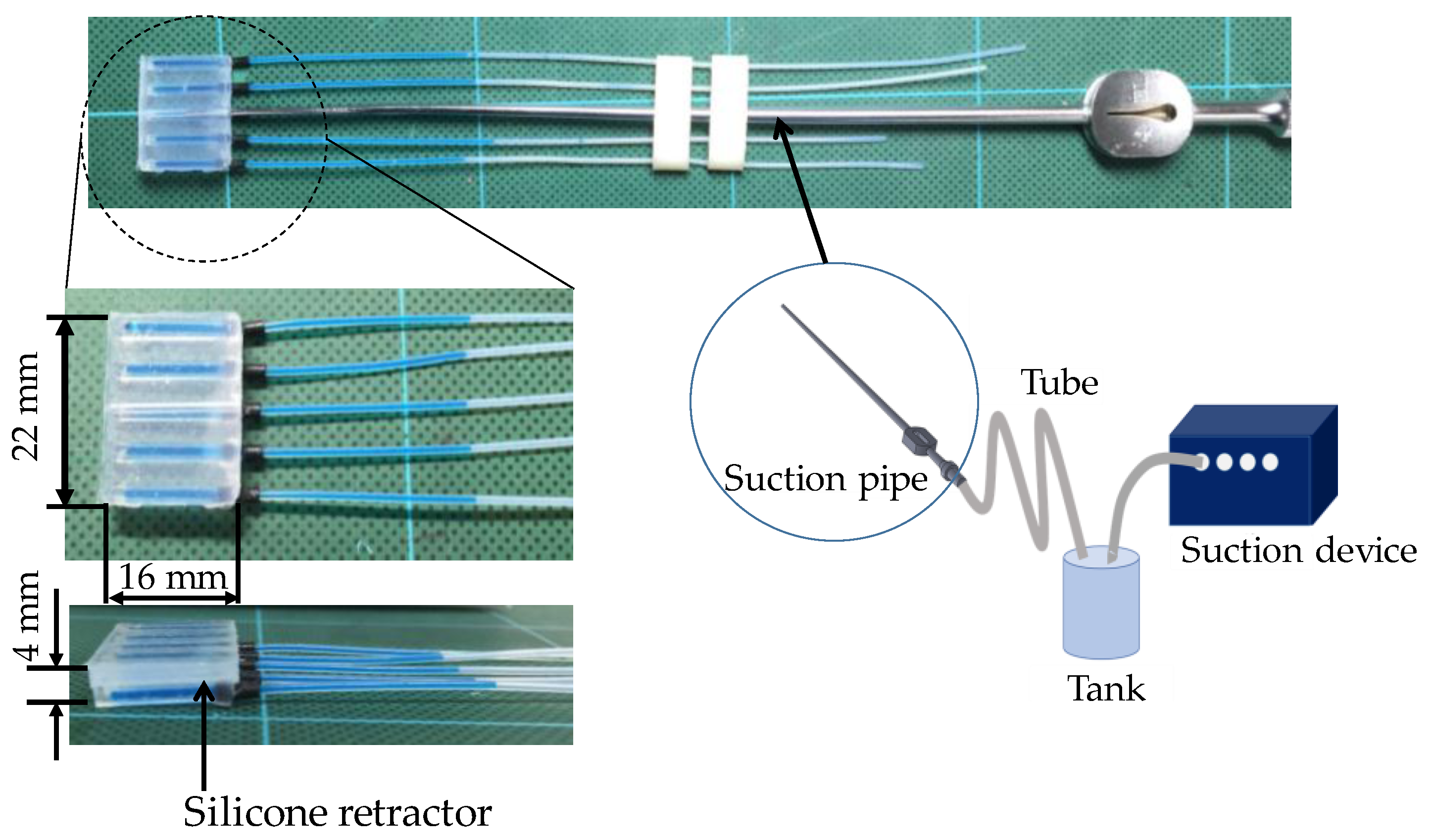
3.4. Structure of the Silicone Retractor with an Embedded Force-Sensing System
4. Design of the Sensor
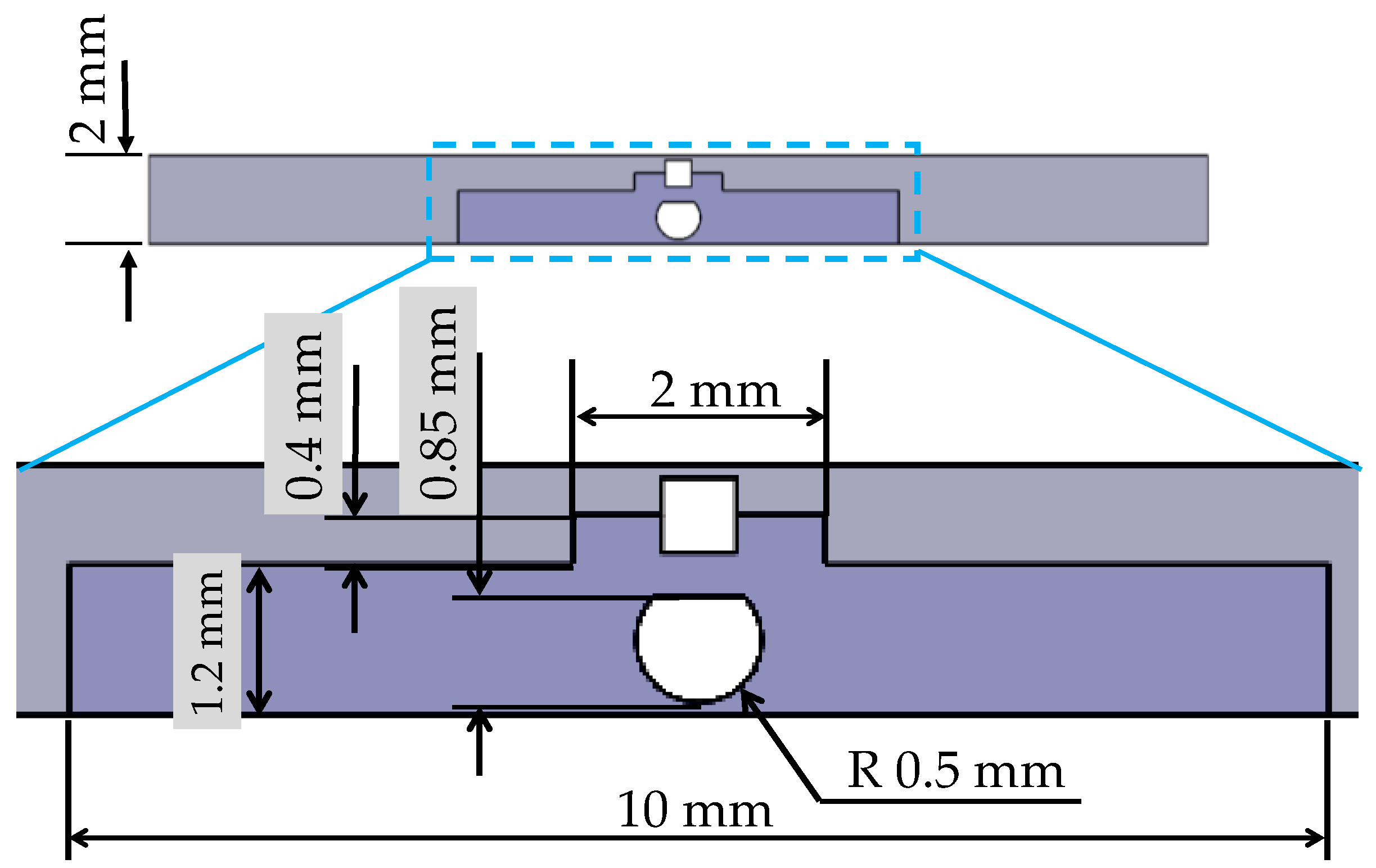
4.1. Design of the Hole for the Sensor
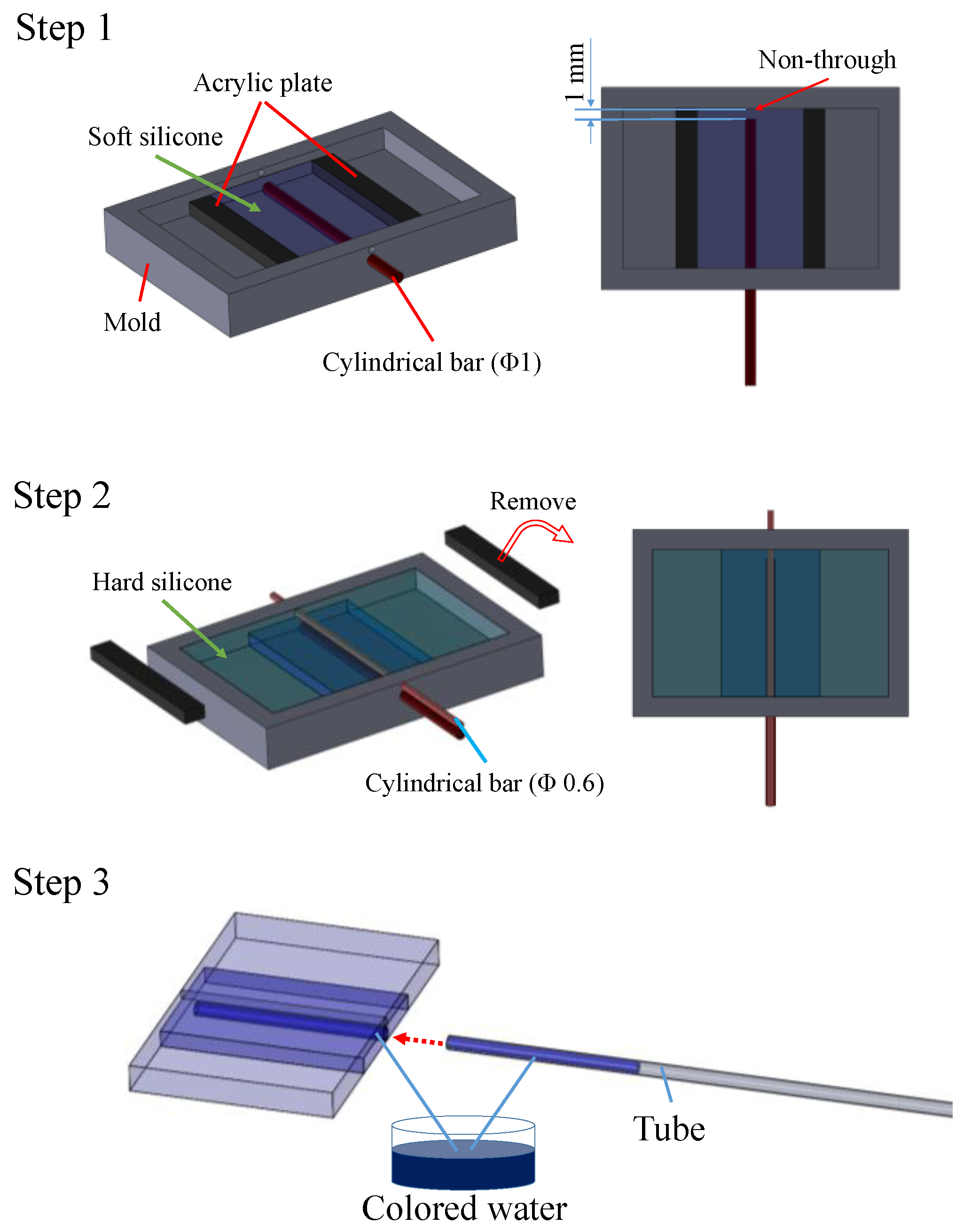
4.1.1. Procedure
4.1.2. Derivation of the Liquid Displacement
4.1.3. Relationship between the Retracting Force and the Liquid Displacement
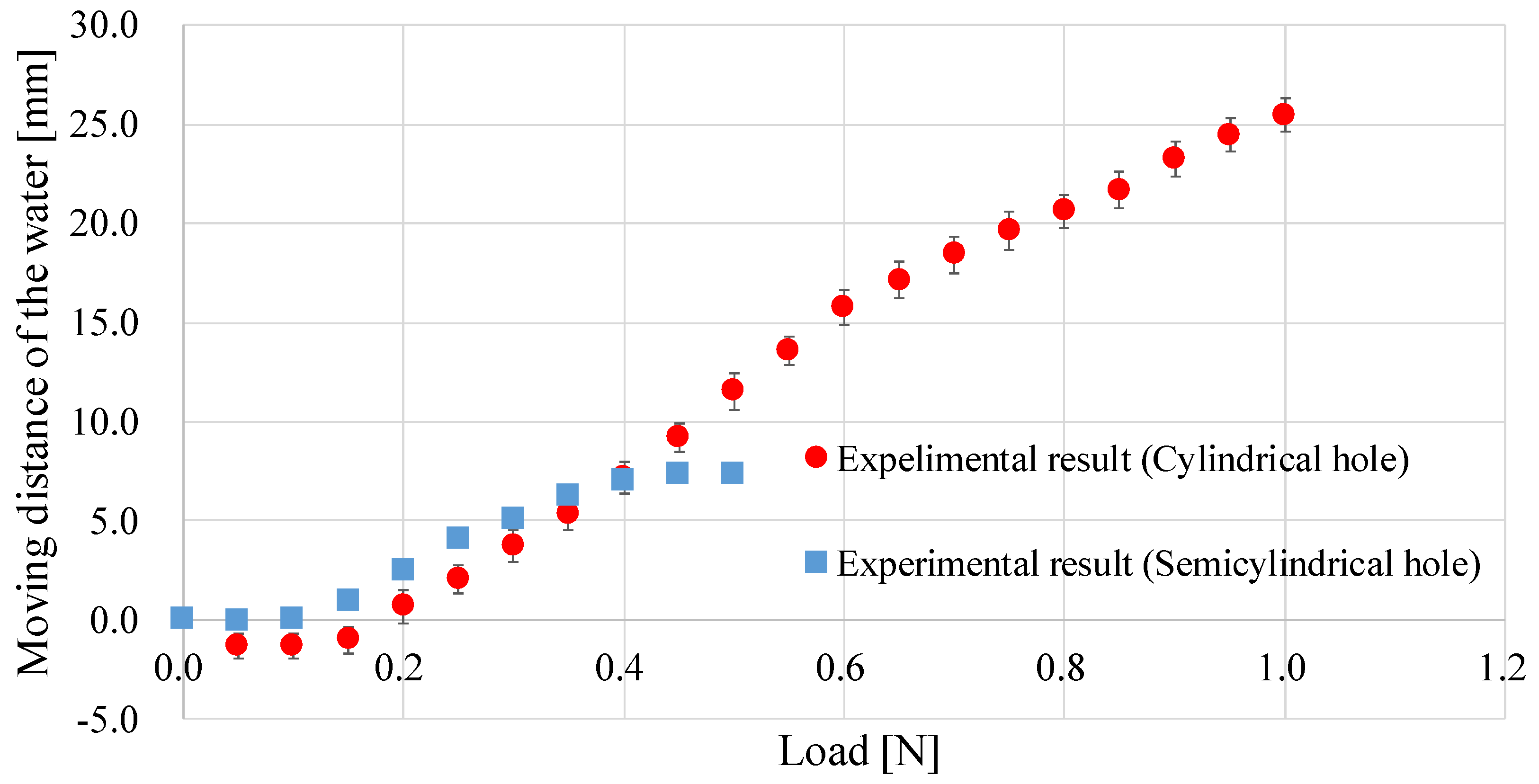
4.2. Improvement of the Sensor Structure
5. Experimental Evaluation on Soft Tissues
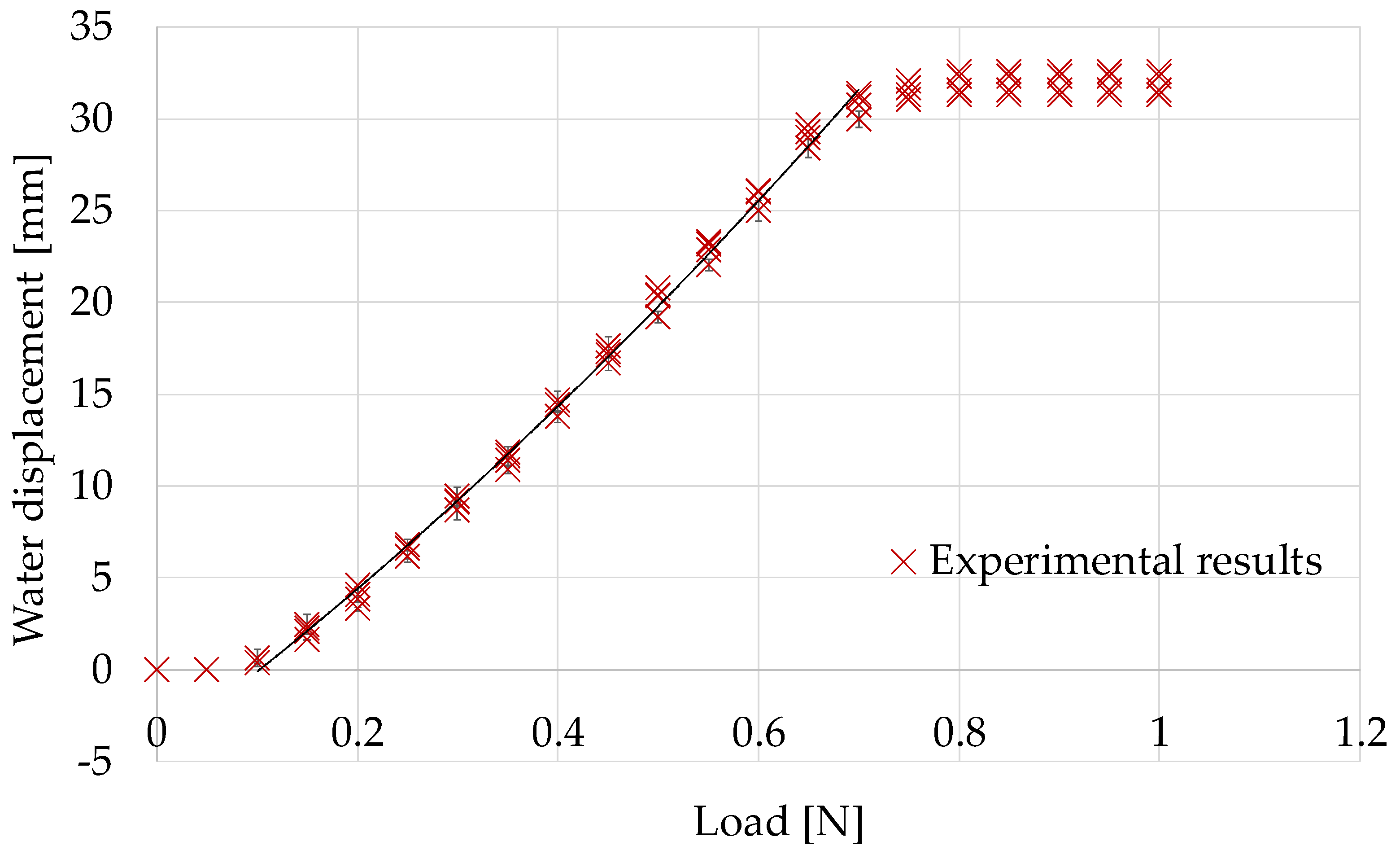
5.1. Calibration While Retracting Flat and Rigid Surfaces
5.2. Calibration While Retracting Gelatin (Young’s Modulus, 10 kPa)
5.3. Evaluation on Curved and Soft Surfaces
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Koyama, T.; Iwai, T.; Yoneyama, T.; Kagawa, H.; Hayashi, Y.; Nakada, M.; Watanabe, T. Incompressible liquid based Force sensible silicone retractor attachable to surgical suction instrument. In Proceedings of the 2016 IEEE International Conference on Advanced Intelligent Mechatronics (AIM), Banff, AB, Canada, 12–15 July 2016; pp. 1398–1404. [Google Scholar]
- Puangmali, P.; Althoefer, K.; Seneviratne, L.D.; Murphy, D.; Dasgupta, P. State-of-the-Art in Force and Tactile Sensing for Minimally Invasive Surgery. IEEE Sens. J. 2008, 8, 371–381. [Google Scholar] [CrossRef]
- Westebring-van der Putten, E.P.; Goossens, R.H.M.; Jakimowicz, J.J.; Dankelman, J. Haptics in Minimally Invasive Surgery—A Review. Minim. Invasive Ther. Allied Technol. 2008, 17, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Okamura, A.M. Haptic Feedback in Robot-Assisted Minimally Invasive Surgery. Curr. Opin. Urol. 2009, 19, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Vander Poorten, E.V.B.; Demeester, E.; Lammertse, P. Haptic Feedback for Medical Applications, A Survey. In Proceedings of the 13th International Conference on New Actuators, Bremen, Germany, 18–21 June 2012; pp. 18–20. [Google Scholar]
- Tiwana, M.I.; Redmond, S.J.; Lovell, N.H. A Review of Tactile Sensing Technologies with Applications in Biomedical Engineering. Sens. Actuators A Phys. 2012, 179, 17–31. [Google Scholar] [CrossRef]
- Dahiya, R.S.; Metta, G.; Valle, M.; Sandini, G. Tactile Sensing-From Humans to Humanoids. IEEE Trans. Robot. 2010, 26, 1–20. [Google Scholar] [CrossRef]
- Yoneyama, T.; Watanabe, T.; Kagawa, H.; Hamada, J.; Hayashi, Y.; Nakada, M. Force-Detecting Gripper and Force Feedback System for Neurosurgery Applications. Int. J. Comput. Assist. Radiol. Surg. 2013, 8, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Kanada, Y.; Yoneyama, T.; Watanabe, T.; Kagawa, H.; Sugiyama, N.; Tanaka, K.; Hanyu, T. Force Feedback Manipulating System for Neurosurgery. Procedia CIRP 2013, 5, 133–136. [Google Scholar] [CrossRef]
- Fujihira, Y.; Hanyu, T.; Kanada, Y.; Yoneyama, T.; Watanabe, T.; Kagawa, H. Gripping Force Feedback System for Neurosurgery. Int. J. Autom. Technol. 2014, 8, 83–94. [Google Scholar]
- Payne, C.J.; Marcus, H.J.; Yang, G.-Z. A Smart Haptic Hand-Held Device for Neurosurgical Microdissection. Ann. Biomed. Eng. 2015, 43, 2185–2195. [Google Scholar] [CrossRef] [PubMed]
- Marcus, H.J.; Payne, C.J.; Kailaya-Vasa, A.; Griffiths, S.; Clark, J.; Yang, G.-Z.; Darzi, A.; Nandi, D. A “Smart” Force-Limiting Instrument for Microsurgery: Laboratory and In Vivo Validation. PLoS ONE 2016, 11, e0162232. [Google Scholar] [CrossRef] [PubMed]
- Maddahi, Y.; Huang, J.; Huang, J.; Gan, L.S.; Hoshyarmanesh, H.; Zareinia, K.; Sutherland, G.R. Real-time measurement of tool-tissue interaction forces in neurosurgery: Quantification and analysis. In Proceedings of the 2016 IEEE International Conference on Advanced Intelligent Mechatronics (AIM), Banff, AB, Canada, 12–15 July 2016; pp. 1405–1410. [Google Scholar]
- Zareinia, K.; Maddahi, Y.; Gan, L.S.; Ghasemloonia, A.; Lama, S.; Sugiyama, T.; Yang, F.W.; Sutherland, G.R. A Force-Sensing Bipolar Forceps to Quantify Tool–Tissue Interaction Forces in Microsurgery. IEEE/ASME Trans. Mechatron. 2016, 21, 2365–2377. [Google Scholar] [CrossRef]
- Tadano, K.; Kawashima, K. Development of 4-DOFs Forceps with Force Sensing using Pneumatic Servo System. In Proceedings of the 2006 IEEE International Conference on Robotics and Automation, Orlando, FL, USA, 15–19 May 2006; pp. 2250–2255. [Google Scholar]
- Haraguchi, D.; Tadano, K.; Kawashima, K. A prototype of pneumatically-driven forceps manipulator with force sensing capability using a simple flexible joint. In Proceedings of the IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Francisco, CA, USA, 20–30 September 2011; pp. 931–936. [Google Scholar]
- Kawahara, T.; Tanaka, S.; Kaneko, M. Non-Contact Stiffness Imager. Int. J. Robot. Res. 2006, 25, 537–549. [Google Scholar] [CrossRef]
- Fukuda, T.; Tanaka, Y.; Fujiwara, M.; Sano, A. Softness measurement by forceps-type tactile sensor using acoustic reflection. In Proceedings of the 2015 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Hamburg, Germany, 28 September–3 October 2015; pp. 3791–3796. [Google Scholar]
- Peirs, J.; Clijnen, J.; Reynaerts, D.; Brussel, H.; Van Herijgers, P.; Corteville, B.; Boone, S. A Micro Optical Force Sensor for Force Feedback during Minimally Invasive Robotic Surgery. Sens. Actuators A Phys. 2004, 115, 447–455. [Google Scholar] [CrossRef]
- Tada, M.; Sasaki, S.; Ogasawara, T. Development of an Optical 2-axis Force Sensor Usable in MRI Environments. IEEE Sens. Proc. 2002, 2, 984–989. [Google Scholar]
- Puangmali, P.; Liu, H.; Seneviratne, L.D.; Dasgupta, P.; Althoefer, K. Miniature 3-Axis Distal Force Sensor for Minimally Invasive Surgical Palpation. IEEE/ASME Trans. Mechatron. 2012, 17, 646–656. [Google Scholar] [CrossRef]
- Polygerinos, P.; Seneviratne, L.D.; Razavi, R.; Schaeffter, T.; Althoefer, K. Triaxial Catheter-Tip Force Sensor for MRI-Guided Cardiac Procedures. IEEE/ASME Trans. Mechatron. 2013, 18, 386–396. [Google Scholar] [CrossRef]
- Polygerinos, P.; Ataollahi, A.; Schaeffter, T.; Razavi, R.; Seneviratne, L.D.; Althoefer, K. MRI-Compatible Intensity-Modulated Force Sensor for Cardiac Catheterization Procedures. IEEE Trans. Biomed. Eng. 2011, 58, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Polygerinos, P.; Zbyszewski, D.; Schaeffter, T.; Razavi, R.; Seneviratne, L.D.; Althoefer, K. MRI-Compatible Fiber-Optic Force Sensors for Catheterization Procedures. IEEE Sens. J. 2010, 10, 1598–1608. [Google Scholar] [CrossRef]
- Polygerinos, P.; Seneviratne, L.D.; Althoefer, K. Modeling of Light Intensity-Modulated Fiber-Optic Displacement Sensors. IEEE Trans. Instrum. Meas. 2011, 60, 1408–1415. [Google Scholar] [CrossRef]
- Liu, H.; Puangmali, P.; Zbyszewski, D.; Elhage, O.; Dasgupta, P.; Dai, J.S.; Seneviratne, L.; Althoefer, K. An Indentation Depth-Force Sensing Wheeled Probe for Abnormality Identification during Minimally Invasive Surgery. Proc. Inst. Mech. Eng. H J. Eng. Med. 2010, 224, 751–763. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, R.; Packirisamy, M.; Dargahi, J.; Cecere, R. Discretely Loaded Beam-Type Optical Fiber Tactile Sensor for Tissue Manipulation and Palpation in Minimally Invasive Robotic Surgery. IEEE Sens. J. 2012, 12, 22–32. [Google Scholar] [CrossRef]
- Xie, H.; Liu, H.; Seneviratne, L.D.; Althoefer, K. An Optical Tactile Array Probe Head for Tissue Palpation during Minimally Invasive Surgery. IEEE Sens. J. 2014, 14, 3283–3291. [Google Scholar] [CrossRef]
- Tan, U.X.; Yang, B.; Gullapalli, R.; Desai, J.P. Tri-Axial MRI Compatible Fiber-Optic Force Sensor. IEEE Trans. Robot. 2011, 27, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Fischer, G.S. A 3-Axis Optical Force/Torque Sensor for Prostate Needle Placement in Magnetic Resonance Imaging Environments. In Proceedings of the 2009 IEEE International Conference on Technologies for Practical Robot Applications, Woburn, MA, USA, 9–10 November 2009; pp. 5–9. [Google Scholar]
- Turkseven, M.; Ueda, J. Design of an MRI Compatible Haptic Interface. In Proceedings of the 2011 IEEE/RSJ International Conference on Intelligent Robots and Systems, San Francisco, CA, USA, 25–30 September 2011; pp. 2139–2144. [Google Scholar]
- Su, H.; Zervas, M.; Furlong, C.; Fischer, G.S. A Miniature Mri-Compatible Fiber-Optic Force Sensor Utilizing Fabry-Perot Interferometer. In Mems and Nanotechnology; Springer: New York, NY, USA, 2011; pp. 131–136. [Google Scholar]
- Su, H.; Zervas, M.; Cole, G.A.; Furlong, C.; Fischer, G.S. Real-Time MRI-Guided Needle Placement Robot with Integrated Fiber Optic Force Sensing. In Proceedings of the 2011 IEEE International Conference on Robotics and Automation, Shanghai, China, 9–13 May 2011; pp. 1583–1588. [Google Scholar]
- Liu, X.; Iordachita, I.I.; He, X.; Taylor, R.H.; Kang, J.U. Miniature Fiber-Optic Force Sensor based on Low-Coherence Fabry-Pérot Interferometry for Vitreoretinal Microsurgery. Biomed. Opt. Express 2012, 3, 1062–1076. [Google Scholar] [CrossRef] [PubMed]
- Tomo, T.P.; Somlor, S.; Schmitz, A.; Jamone, L.; Huang, W.; Kristanto, H.; Sugano, S. Design and Characterization of A Three-Axis Hall Effect-Based Soft Skin Sensor. Sensors 2016, 16, 491. [Google Scholar] [CrossRef] [PubMed]
- Jamone, L.; Natale, L.; Metta, G.; Sandini, G. Highly Sensitive Soft Tactile Sensors for an Anthropomorphic Robotic Hand. IEEE Sens. J. 2015, 15, 4226–4233. [Google Scholar] [CrossRef]
- Ribeiro, P.; Khan, M.A.; Alfadhel, A.; Kosel, J.; Franco, F.; Cardoso, S.; Bernardino, A.; Schmitz, A.; Santos-Victor, J.; Jamone, L. Bioinspired Ciliary Force Sensor for Robotic Platforms. IEEE Robot. Autom. Lett. 2017, 2, 971–976. [Google Scholar] [CrossRef]
- Takaki, T.; Omasa, Y.; Ishii, I.; Kawahara, T.; Okajima, M. Force Visualization Mechanism Using a Moiré fringe Applied to Endoscopic Surgical Instruments. In Proceedings of the 2010 IEEE International Conference on Robotics and Automation, Anchorage, AK, USA, 3–7 May 2010; pp. 3648–3653. [Google Scholar]
- Watanabe, T.; Iwai, T.; Fujihira, Y.; Wakako, L.; Kagawa, H.; Yoneyama, T. Force Sensor Attachable to Thin Fiberscopes/Endoscopes Utilizing High Elastic Fabric. Sensors 2014, 14, 5207–5220. [Google Scholar] [CrossRef] [PubMed]
- Iwai, T.; Fujihira, Y.; Wakako, L.; Kagawa, H.; Yoneyama, T.; Watanabe, T. Three-Axis Force Visualizing System for Fiberscopes Utilizing Highly Elastic Fabric. In Proceedings of the IEEE/ASME International Conference on Advanced Intelligent Mechatronics (AIM), Besacon, France, 8–11 July 2014; pp. 1110–1115. [Google Scholar]
- Watanabe, T.; Iwai, T.; Koyama, T.; Yoneyama, T. Stiffness Measurement System Using Endoscopes with a Visualization Method. IEEE Sens. J. 2016, 16, 5889–5897. [Google Scholar] [CrossRef]
- Ohka, M.; Mitsuya, Y.; Matsunaga, Y.; Takeuchi, S. Sensing Characteristics of an Optical Three-Axis Tactile Sensor under Combined Loading. Robotica 2004, 22, 213–221. [Google Scholar] [CrossRef]
- Kamiyama, K.; Vlack, K.; Mizota, T.; Kajimoto, H.; Kawakami, K.; Tachi, S. Vision-based Sensor for Real-Time Measuring of Surface Traction Fields. IEEE Comput. Graph. Appl. 2005, 25, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Koyama, T.; Yoneyama, T.; Nakada, M. Force-Sensing Silicone Retractor for Attachment to Surgical Suction Pipes. Sensors 2016, 16, 1133. [Google Scholar] [CrossRef] [PubMed]
- Zinnia, S.X.; Rona, J.L.; Stephanie, S.C.; Anning, Y.; Marla, K.P.; Sean, P.M.; Pierre, D.M. Evidence of Changes in Brain Tissue Stiffness after Ischemic Stroke Derived From ltrasound-Based Elastography. Ultrasound Med. 2013, 32, 485–494. [Google Scholar]
- Nishimura, T.; Fujihira, Y.; Adachi, R.; Watanabe, T. New Condition for Tofu Stable Grasping with Fluid Fingertips. In Proceedings of the 2016 IEEE International Conference on Advanced Intelligent Mechatronics (CASE), Fort Worth, TX, USA, 21–24 August 2016; pp. 335–341. [Google Scholar]







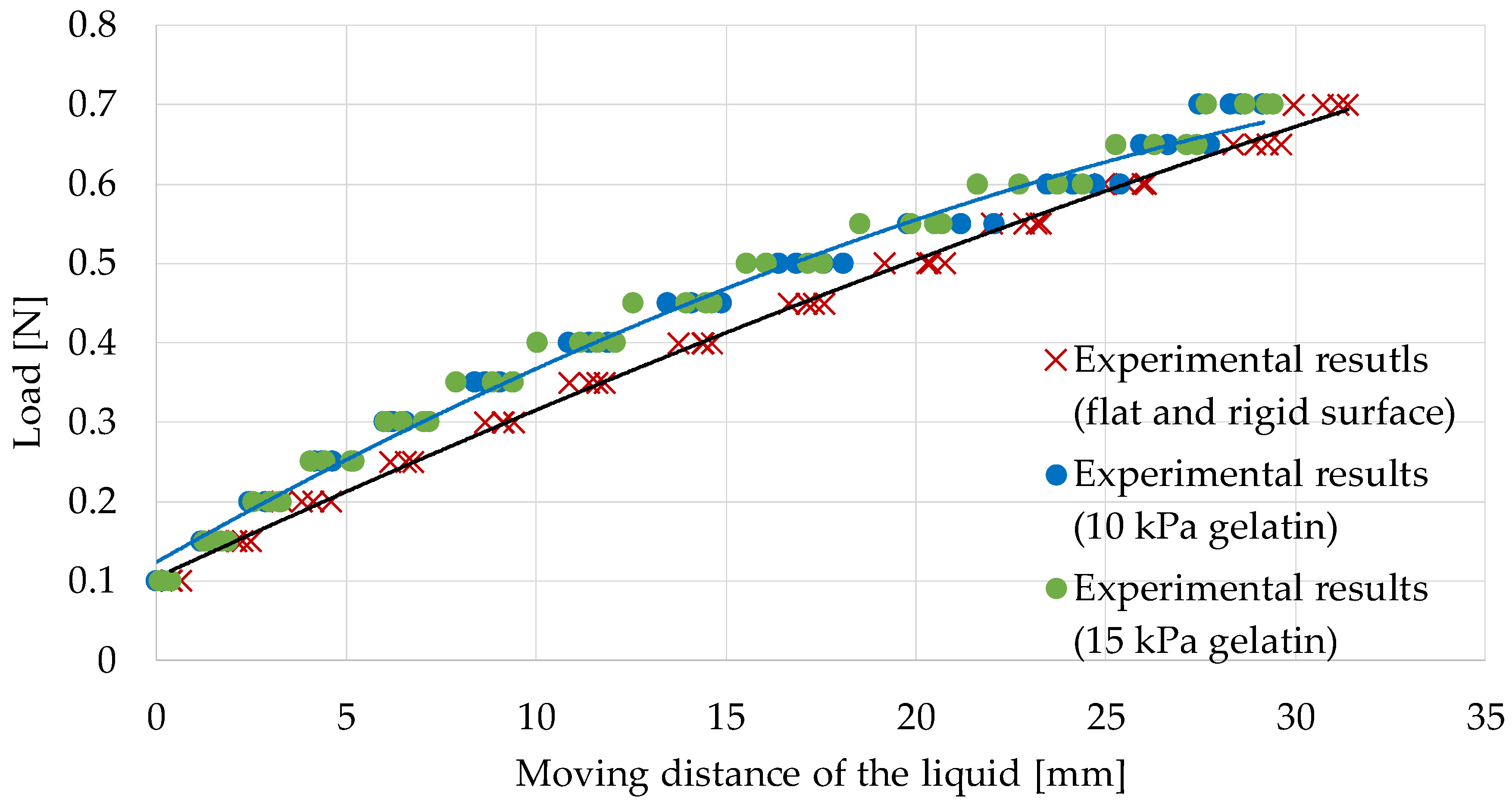

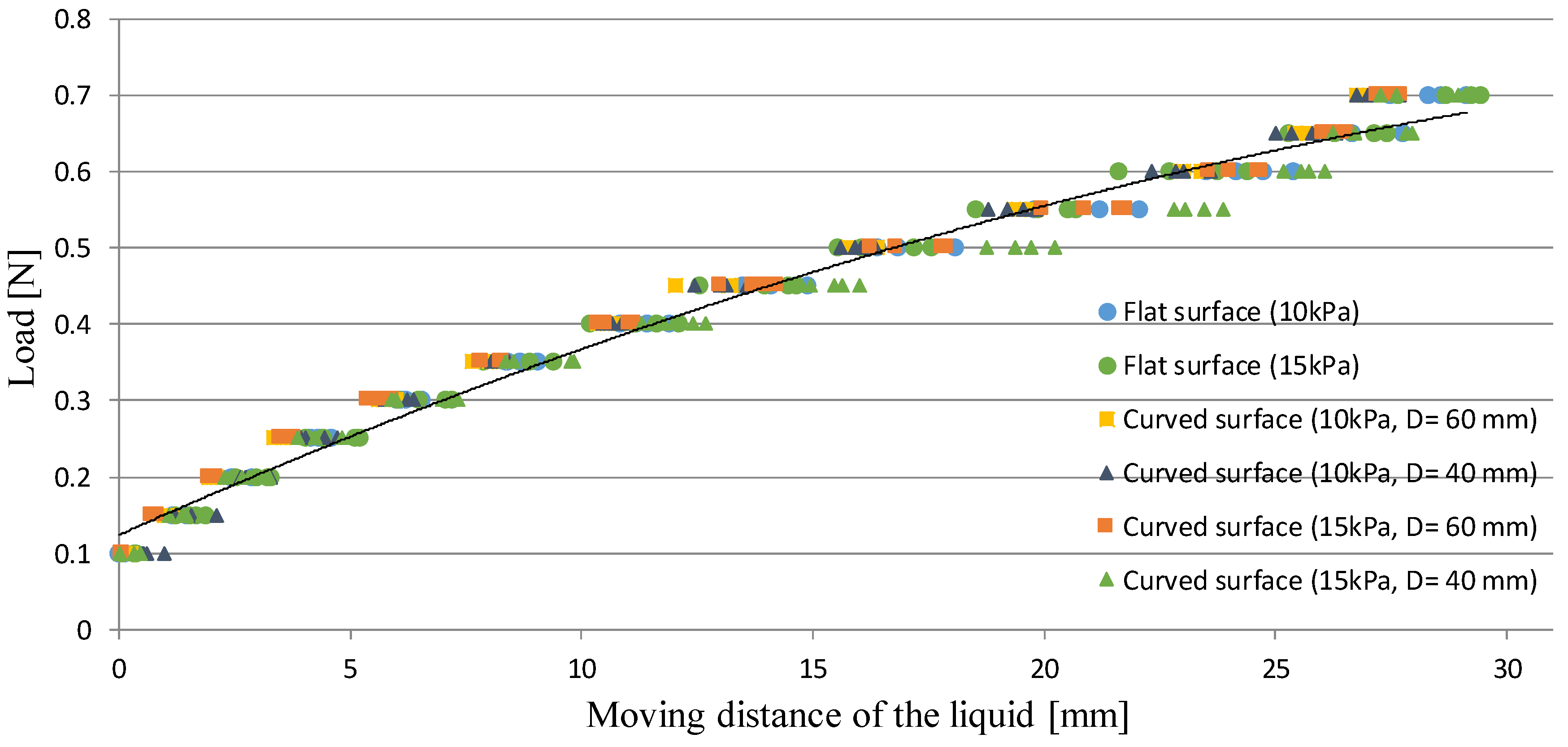
| Dimension of the Polynomial Function | Coefficient of Determination: R2 | RMSE (Root Mean Squared Error) [N] |
|---|---|---|
| 2 | 0.99 | 0.011 |
| Dimension [mm] | Young’s Modulus [kPa] | Composition Ratio |
|---|---|---|
| 30 × 32 × 16 | 15 | Water:gelatin powder = 100 mL:8 g |
| 30 × 32 × 16 | 10 | Water:gelatin powder = 100 mL:7 g |
| Young’s Modulus [kPa] | RMSE (Root Mean Squared Error) [N] |
|---|---|
| 10 | 0.051 |
| 15 | 0.054 |
| Young’s Modulus [kPa] | RMSE (Root Mean Squared Error) [N] |
|---|---|
| 15 | 0.017 |
| Diameter [mm] | Young’s Modulus [kPa] | Composition Ratio |
|---|---|---|
| 60 | 15 | Water:gelatin powder = 100 mL:8 g |
| 40 | 15 | Water:gelatin powder = 100 mL:8 g |
| 60 | 10 | Water:gelatin powder = 100 mL:7 g |
| 40 | 10 | Water:gelatin powder = 100 mL:7 g |
| Diameter [mm] | Young’s Modulus [kPa] | RMSE (Root Mean Squared Error) [N] |
|---|---|---|
| 60 | 15 | 0.026 |
| 40 | 15 | 0.027 |
| 60 | 10 | 0.028 |
| 40 | 10 | 0.026 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, T.; Koyama, T.; Yoneyama, T.; Nakada, M. A Force-Visualized Silicone Retractor Attachable to Surgical Suction Pipes. Sensors 2017, 17, 773. https://doi.org/10.3390/s17040773
Watanabe T, Koyama T, Yoneyama T, Nakada M. A Force-Visualized Silicone Retractor Attachable to Surgical Suction Pipes. Sensors. 2017; 17(4):773. https://doi.org/10.3390/s17040773
Chicago/Turabian StyleWatanabe, Tetsuyou, Toshio Koyama, Takeshi Yoneyama, and Mitsutoshi Nakada. 2017. "A Force-Visualized Silicone Retractor Attachable to Surgical Suction Pipes" Sensors 17, no. 4: 773. https://doi.org/10.3390/s17040773
APA StyleWatanabe, T., Koyama, T., Yoneyama, T., & Nakada, M. (2017). A Force-Visualized Silicone Retractor Attachable to Surgical Suction Pipes. Sensors, 17(4), 773. https://doi.org/10.3390/s17040773






