Room-Temperature H2 Gas Sensing Characterization of Graphene-Doped Porous Silicon via a Facile Solution Dropping Method
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Synthesis of the P-Si Substrate
2.3. Synthesis of Graphene-Doped Porous Silicon Heterostructure
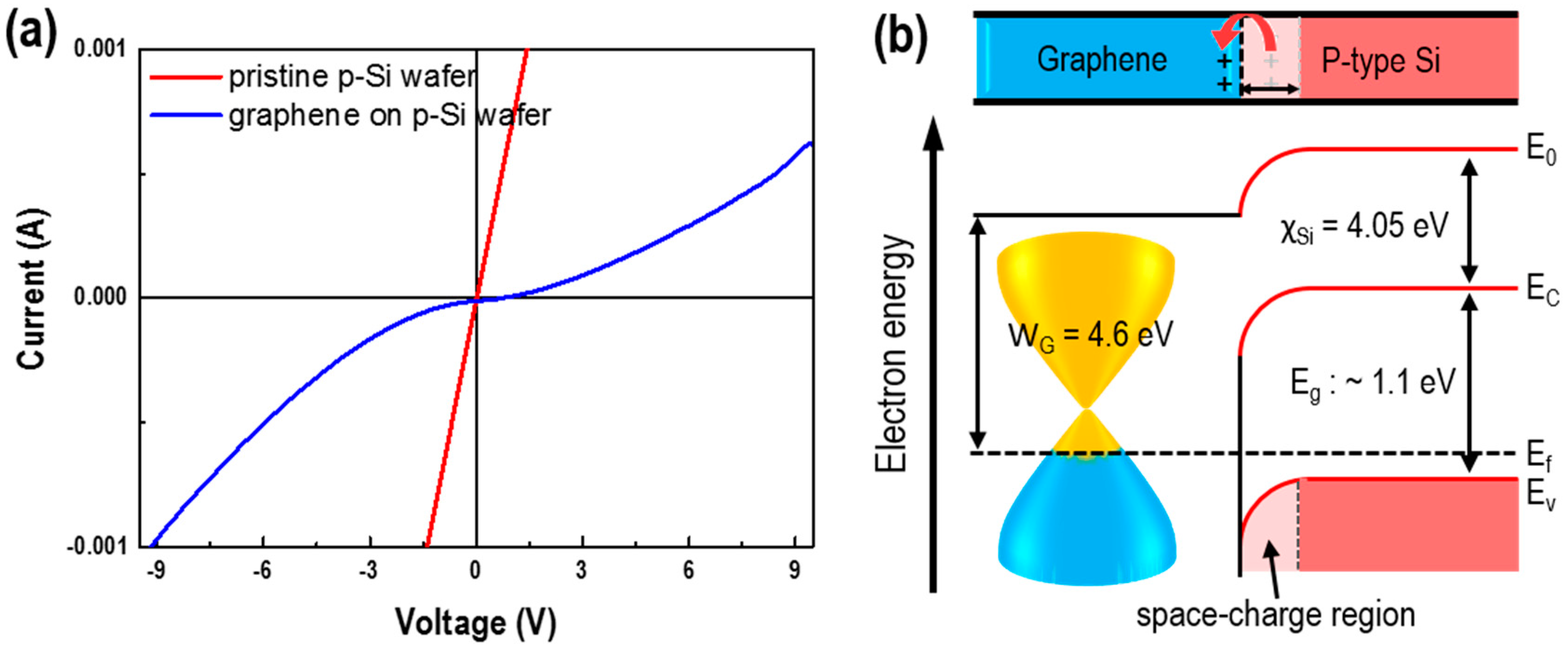
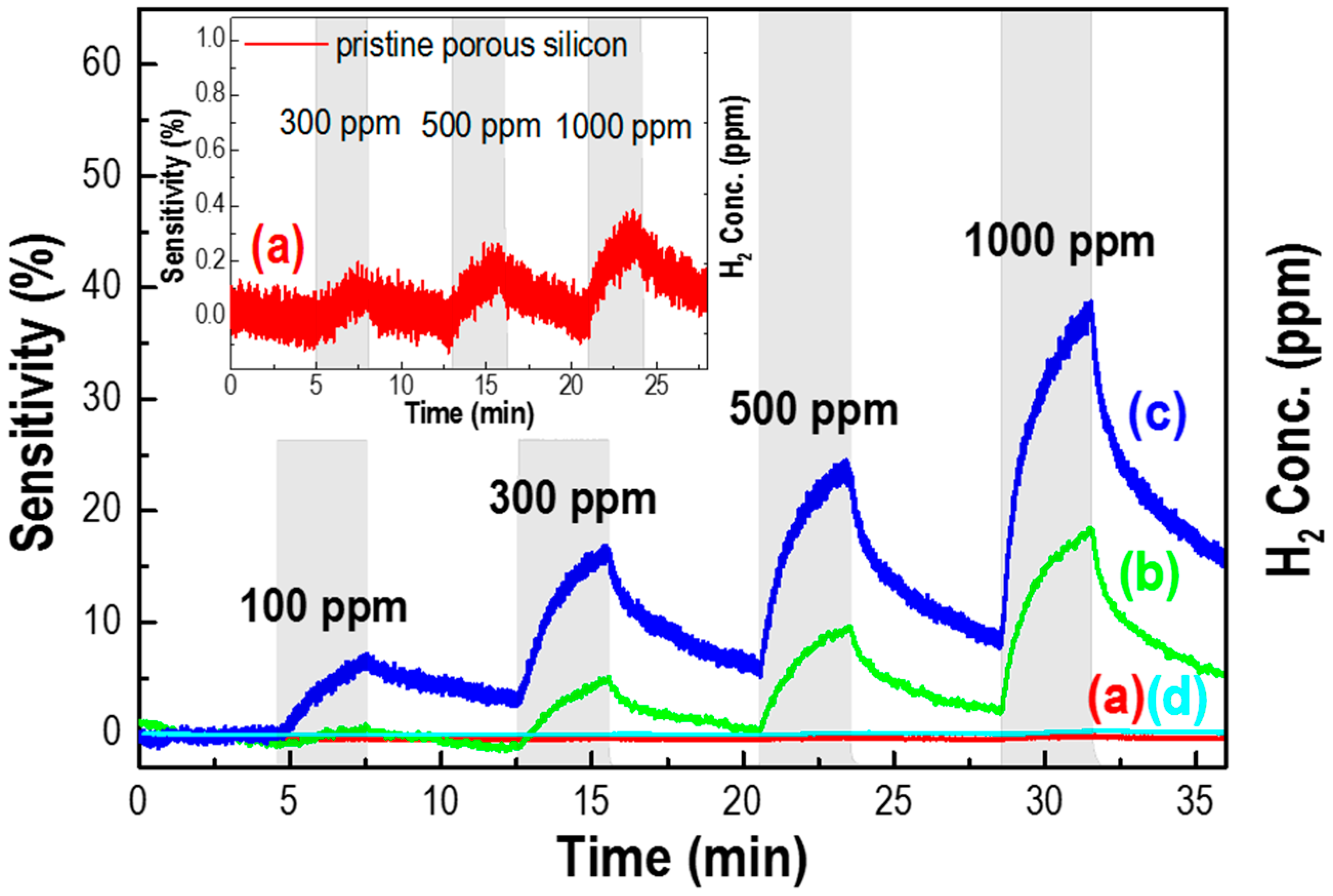
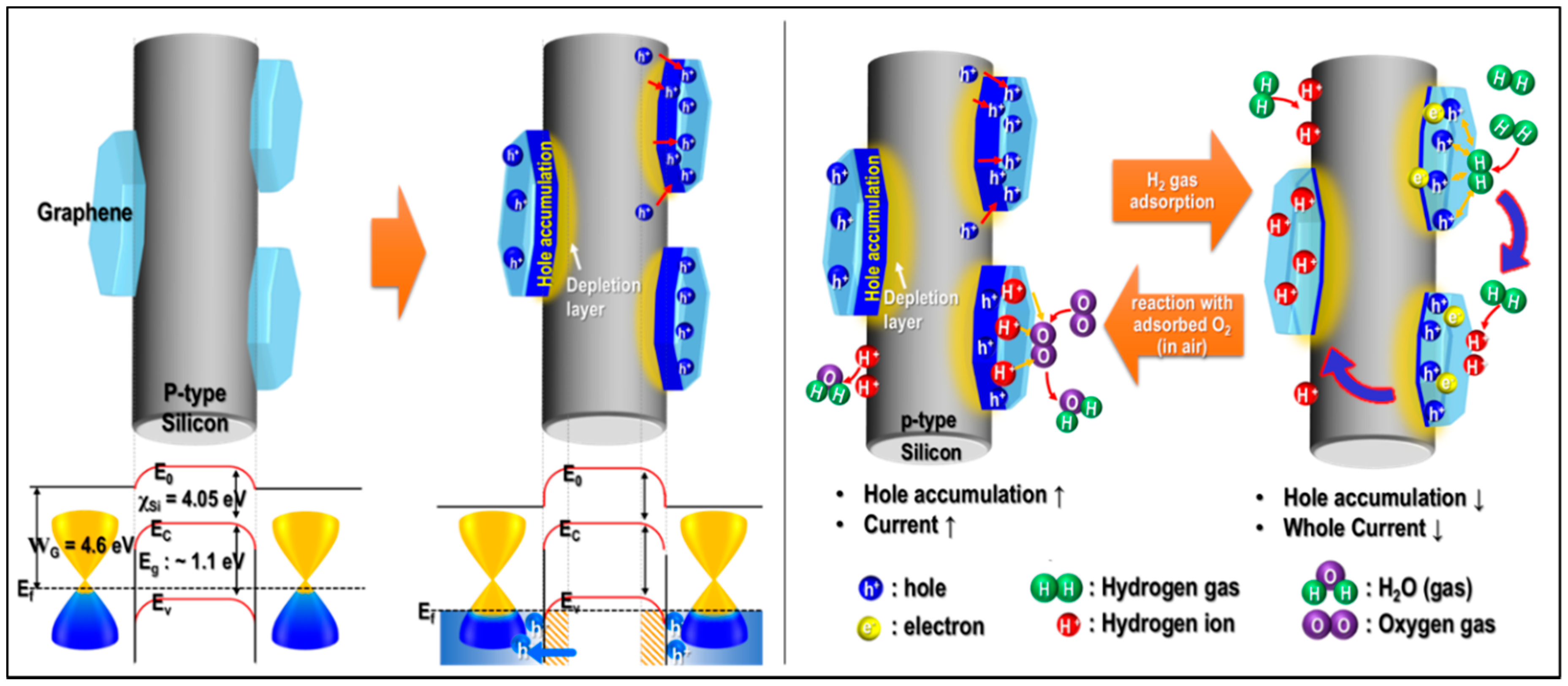
3. Results
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anand, K.; Singh, O.; Singh, M.P.; Kaur, J.; Singh, R.C. Hydrogen sensor based on graphene/ZnO nanocomposite. Sens. Actuators B Chem. 2014, 195, 409–415. [Google Scholar] [CrossRef]
- Sayago, I.; Terrado, E.; Lafuente, E.; Horrillo, M.; Maser, W.K.; Benito, A.; Navarro, R.; Urriolabeitia, E.; Martinez, M.; Gutierrez, J. Hydrogen sensors based on carbon nanotubes thin films. Synth. Metals 2005, 148, 15–19. [Google Scholar] [CrossRef]
- Mubeen, S.; Zhang, T.; Yoo, B.; Deshusses, M.A.; Myung, N.V. Palladium nanoparticles decorated single walled carbon nanotube hydrogen sensor. J. Phys. Chem. C 2007, 111, 6321–6327. [Google Scholar] [CrossRef]
- Plecenik, A.; Haidry, A.; Plecenik, T.; Durina, P.; Truchly, M.; Mosko, M.; Grancic, B.; Gregor, M.; Roch, T.; Satrapinskyy, L. Metal oxide gas sensors on the nanoscale. In Proceedings of the Micro- and Nanotechnology Sensors, Systems, and Applications VI, Volume 9083, Baltimore, MD, USA, 4 June 2014. [Google Scholar]
- Tommasi, A.; Cocuzza, M.; Perrone, D.; Pirri, C.F.; Mosca, R.; Villani, M.; Delmonte, N.; Zappettini, A.; Calestani, D.; Marasso, S.L. Modeling, Fabrication and Testing of a Customizable Micromachined Hotplate for Sensor Applications. Sensors 2017, 17, 62. [Google Scholar] [CrossRef] [PubMed]
- Annanouch, F.E.; Gràcia, I.; Figueras, E.; Llobet, E.; Cané, C.; Vallejos, S. Localized aerosol-assisted CVD of nanomaterials for the fabrication of monolithic gas sensor microarrays. Sens. Actuators B Chem. 2015, 216, 374–383. [Google Scholar] [CrossRef]
- Solntsev, V.; Litovchenko, V.; Gorbanyuk, T.; Evtukh, A. Influence of H2S and H2 adsorption on characteristics of MIS structures with Si porous layers. Semiconduct. Phys. Quantum Electron. Optoelectron. 2008, 11, 381–384. [Google Scholar]
- Korotcenkov, G. Handbook of Gas Sensor Materials: Properties, Advantages and and Shortcomings for Applications; Springer: New York, NY, USA, 2013; ISBN 978-1-4614-7165-3. [Google Scholar]
- Lundström, I.; Spetz, A.; Winquist, F.; Ackelid, U.; Sundgren, H. Catalytic metals and field-effect devices—A useful combination. Sens. Actuators B Chem. 1990, 1, 15–20. [Google Scholar] [CrossRef]
- Gupta, D.; Dutta, D.; Kumar, M.; Barman, P.B.; Sarkar, C.K.; Basu, S.; Hazra, S.K. A low temperature hydrogen sensor based on palladium nanoparticles. Sens. Actuators B Chem. 2014, 196, 215–222. [Google Scholar] [CrossRef]
- Emtsev, K.V.; Bostwick, A.; Horn, K.; Jobst, J.; Kellogg, G.L.; Ley, L.; McChesney, J.L.; Ohta, T.; Reshanov, S.A.; Röhrl, J. Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide. Nat. Mater. 2009, 8, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.; Jiang, D.; Katsnelson, M.; Grigorieva, I.; Dubonos, S.; Firsov, A. Two-dimensional gas of massless Dirac fermions in graphene. Nature 2005, 438, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Xiang, C.; Jiang, D.; Zou, Y.; Chu, H.; Qiu, S.; Zhang, H.; Xu, F.; Sun, L.; Zheng, L. Ammonia sensor based on polypyrrole–graphene nanocomposite decorated with titania nanoparticles. Ceram. Int. 2015, 41, 6432–6438. [Google Scholar] [CrossRef]
- Peng, Y.; Ye, J.; Zheng, L.; Zou, K. The hydrogen sensing properties of Pt–Pd/reduced graphene oxide based sensor under different operating conditions. Rsc Adv. 2016, 6, 24880–24888. [Google Scholar] [CrossRef]
- Capone, S.; Forleo, A.; Francioso, L.; Rella, R.; Siciliano, P.; Spadavecchia, J.; Presicce, D.; Taurino, A. Solid state gas sensors: State of the art and future activities. J. Optoelectron. Adv. Mater. 2003, 5, 1335–1348. [Google Scholar] [CrossRef]
- Kong, J.; Franklin, N.R.; Zhou, C.; Chapline, M.G.; Peng, S.; Cho, K.; Dai, H. Nanotube molecular wires as chemical sensors. Science 2000, 287, 622–625. [Google Scholar] [CrossRef] [PubMed]
- Chourou, M.L.; Fukami, K.; Sakka, T.; Virtanen, S.; Ogata, Y.H. Metal-assisted etching of p-type silicon under anodic polarization in HF solution with and without H2O2. Electrochim. Acta 2010, 55, 903–912. [Google Scholar] [CrossRef]
- Biswas, S.; Drzal, L.T. Multilayered Nanoarchitecture of Graphene Nanosheets and Polypyrrole Nanowires for High Performance Supercapacitor Electrodes. Chem. Mater. 2010, 22, 5667–5671. [Google Scholar] [CrossRef]
- Kim, J.; Oh, S.D.; Kim, J.H.; Shin, D.H.; Kim, S.; Choi, S.-H. Graphene/Si-nanowire heterostructure molecular sensors. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Novikov, D.S. Numbers of donors and acceptors from transport measurements in graphene. Appl. Phys. Lett. 2007, 91, 102102. [Google Scholar] [CrossRef]
- Riazimehr, S.; Bablich, A.; Schneider, D.; Kataria, S.; Passi, V.; Yim, C.; Duesberg, G.S.; Lemme, M.C. Spectral sensitivity of graphene/silicon heterojunction photodetectors. Solid State Electron. 2016, 115, 207–212. [Google Scholar] [CrossRef]
- Xie, C.; Lv, P.; Nie, B.; Jie, J.; Zhang, X.; Wang, Z.; Jiang, P.; Hu, Z.; Luo, L.; Zhu, Z. Monolayer graphene film/silicon nanowire array Schottky junction solar cells. Appl. Phys. Lett. 2011, 99, 133113. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eom, N.S.A.; Cho, H.-B.; Song, Y.; Lee, W.; Sekino, T.; Choa, Y.-H. Room-Temperature H2 Gas Sensing Characterization of Graphene-Doped Porous Silicon via a Facile Solution Dropping Method. Sensors 2017, 17, 2750. https://doi.org/10.3390/s17122750
Eom NSA, Cho H-B, Song Y, Lee W, Sekino T, Choa Y-H. Room-Temperature H2 Gas Sensing Characterization of Graphene-Doped Porous Silicon via a Facile Solution Dropping Method. Sensors. 2017; 17(12):2750. https://doi.org/10.3390/s17122750
Chicago/Turabian StyleEom, Nu Si A., Hong-Baek Cho, Yoseb Song, Woojin Lee, Tohru Sekino, and Yong-Ho Choa. 2017. "Room-Temperature H2 Gas Sensing Characterization of Graphene-Doped Porous Silicon via a Facile Solution Dropping Method" Sensors 17, no. 12: 2750. https://doi.org/10.3390/s17122750
APA StyleEom, N. S. A., Cho, H.-B., Song, Y., Lee, W., Sekino, T., & Choa, Y.-H. (2017). Room-Temperature H2 Gas Sensing Characterization of Graphene-Doped Porous Silicon via a Facile Solution Dropping Method. Sensors, 17(12), 2750. https://doi.org/10.3390/s17122750




