Binding Affinity of a Highly Sensitive Au/Ag/Au/Chitosan-Graphene Oxide Sensor Based on Direct Detection of Pb2+ and Hg2+ Ions
Abstract
:1. Introduction
2. Materials and Methods
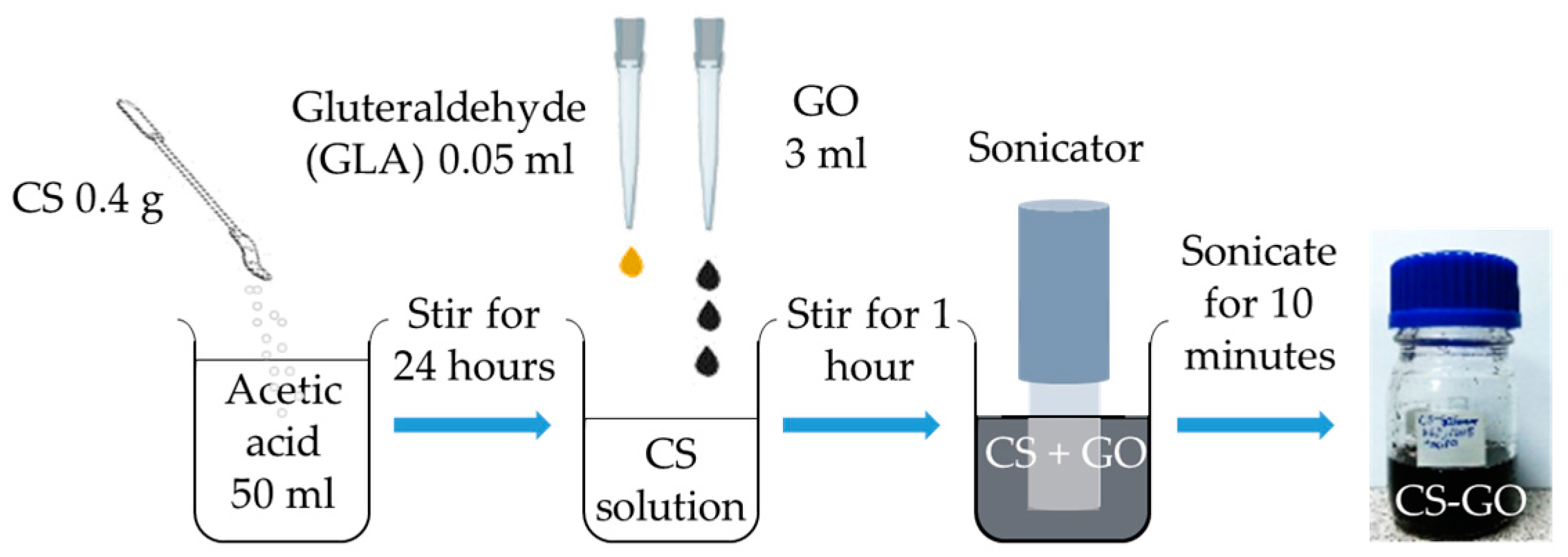
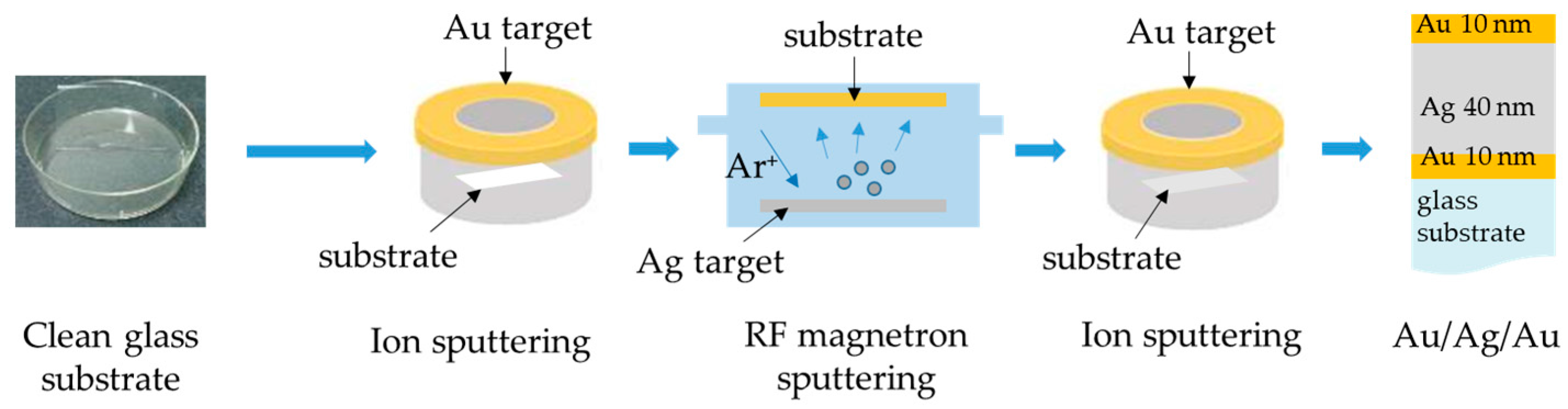
2.1. Fabrication of Au/Ag/Au/CS–GO SPR Sensor
2.2. Characterisation of Au/Ag/Au/CS–GO Nanostructure
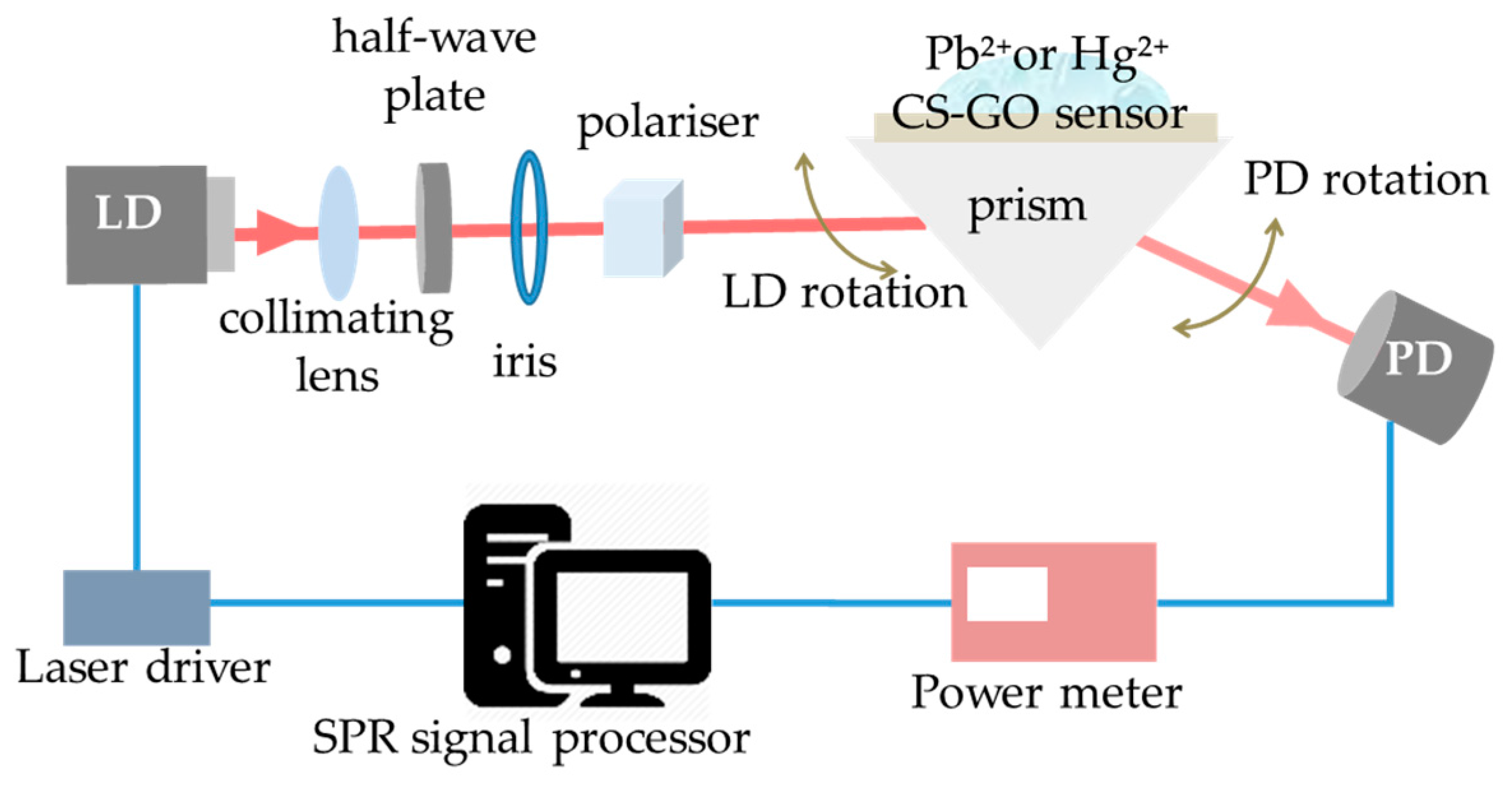
2.3. Execution of SPR Measurement
3. Results and Discussion
3.1. Elemental Composition of Au/Ag/Au/CS–GO SPR Sensor
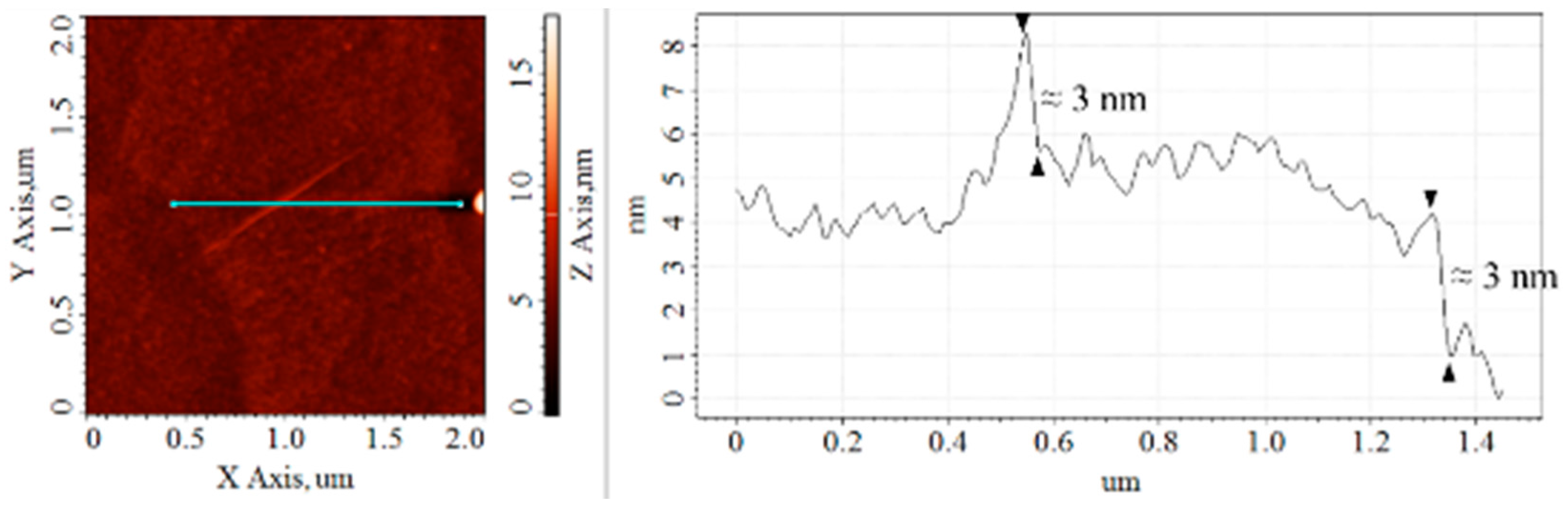
3.2. Cross-Sectional Analysis and Height Profile of the CS–GO Nanolayer
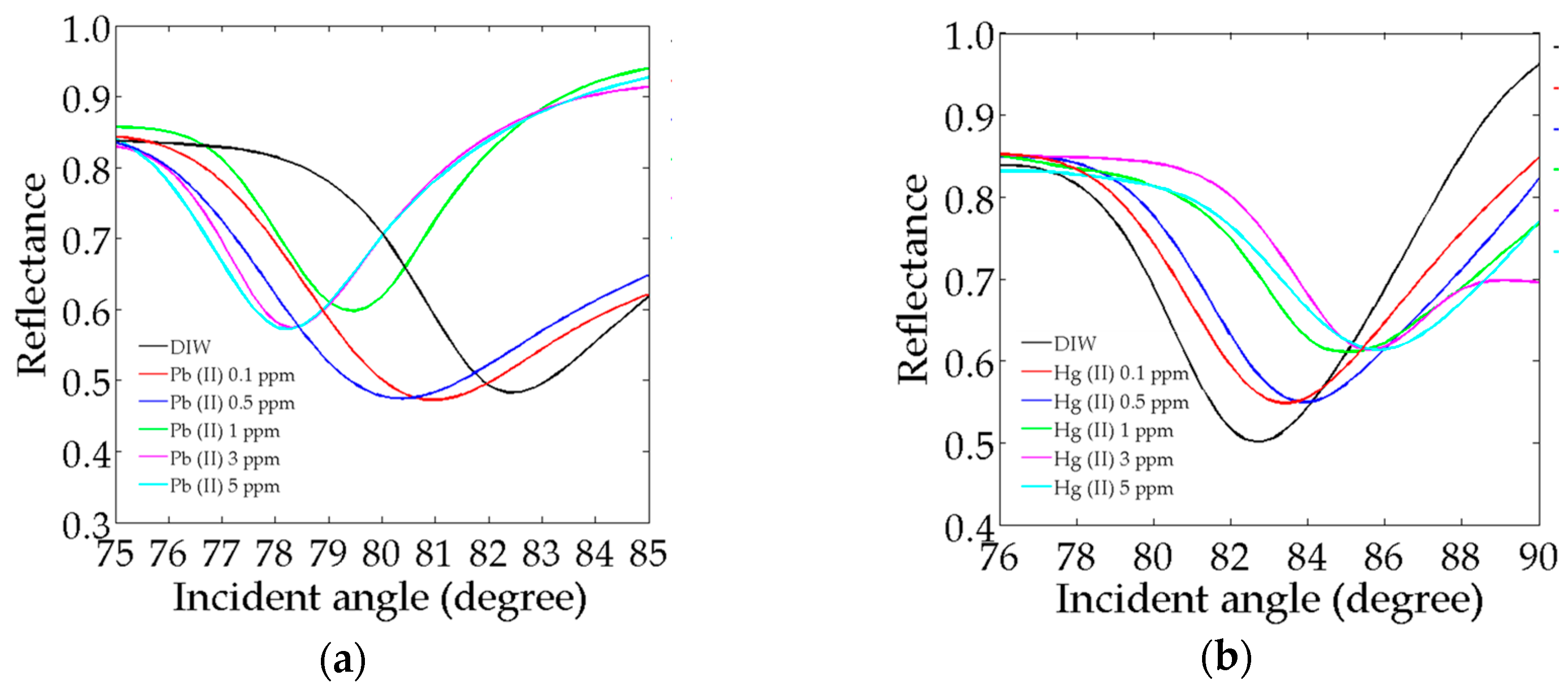
3.3. SPR Reflectivity in Pb2+ and Hg2+ Ions
Full-Width-Half-Maximum (FWHM)
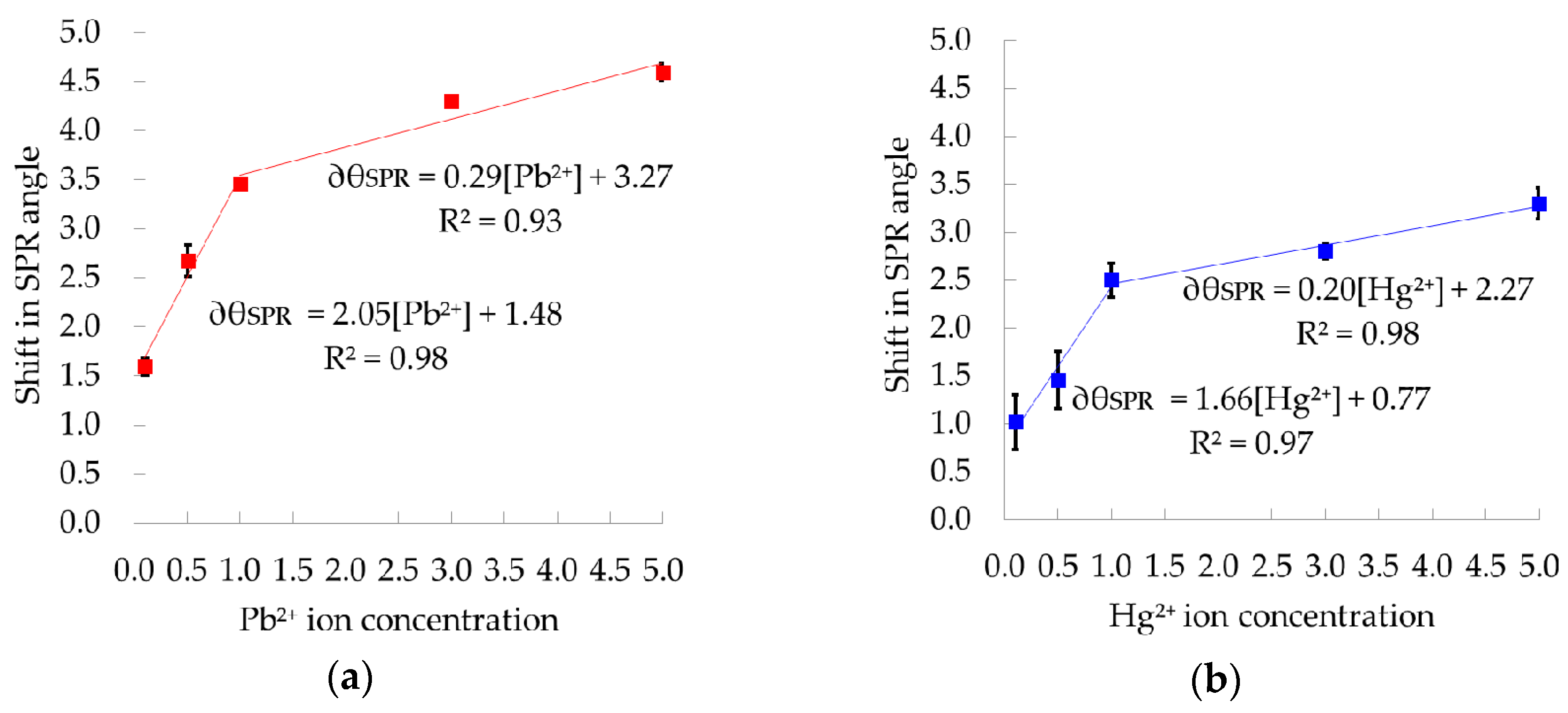
3.4. Calibration Curves for Pb2+ and Hg2+ Ions
Repeatability
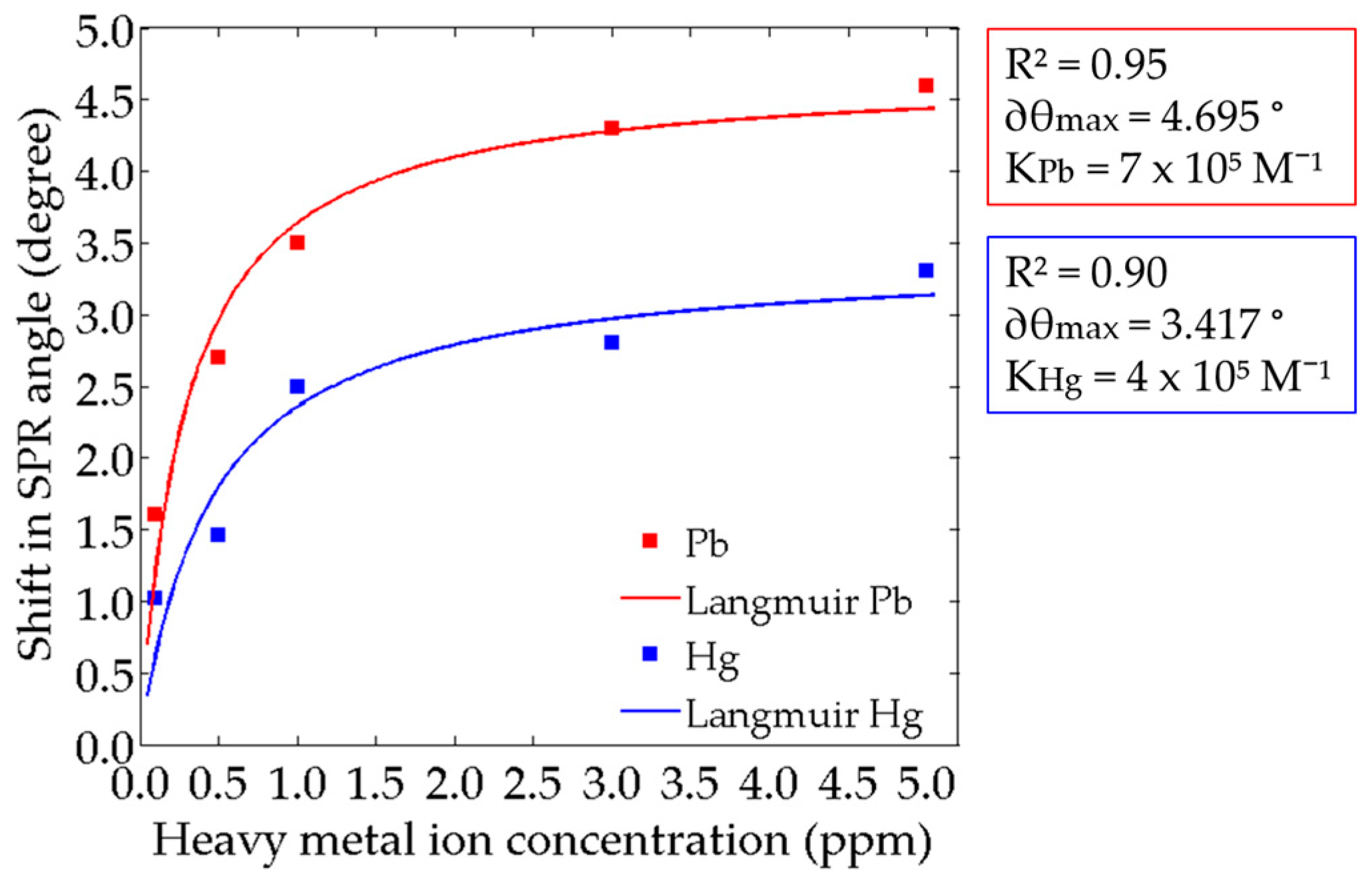
3.5. Binding Affinity Constant, K
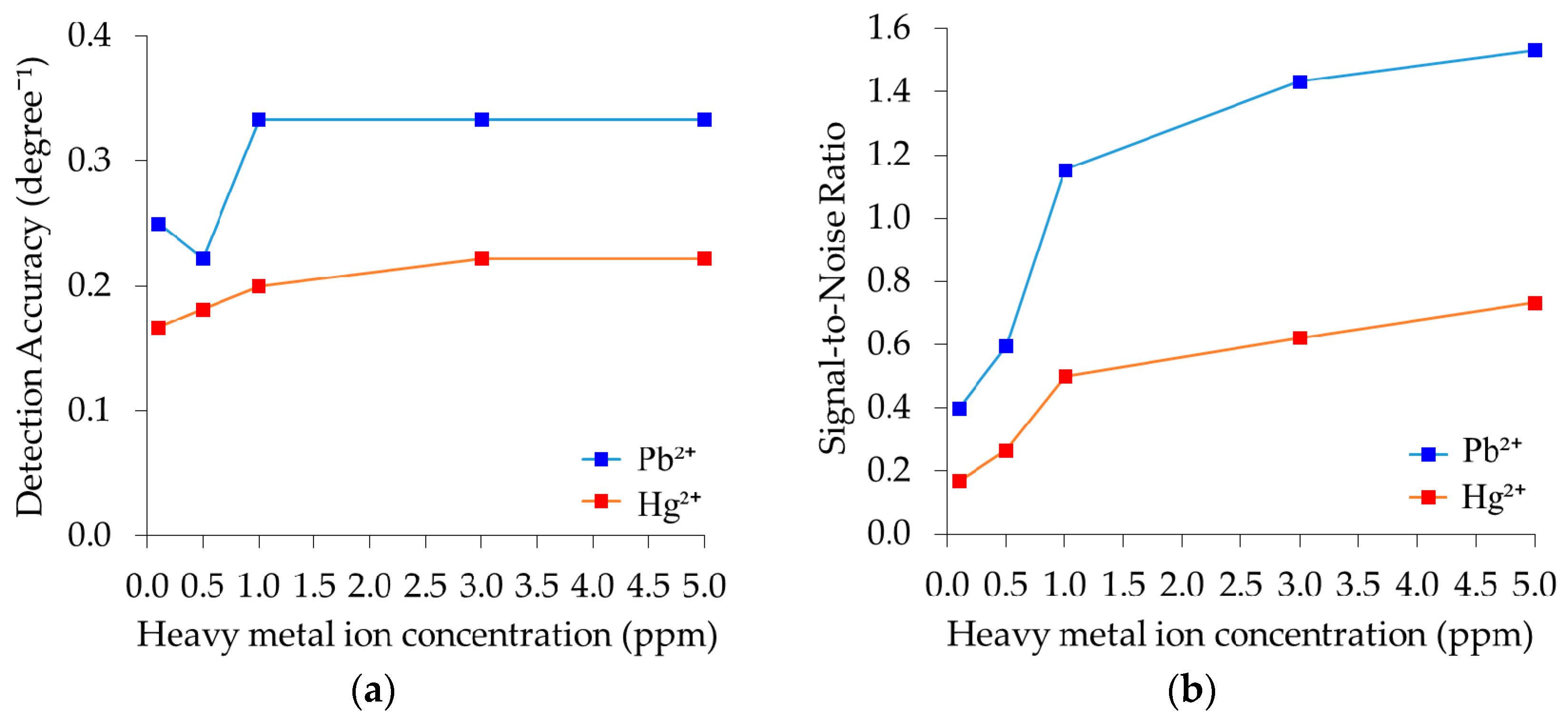
3.6. Detection Accuracy and Signal-to-Noise-Ratio
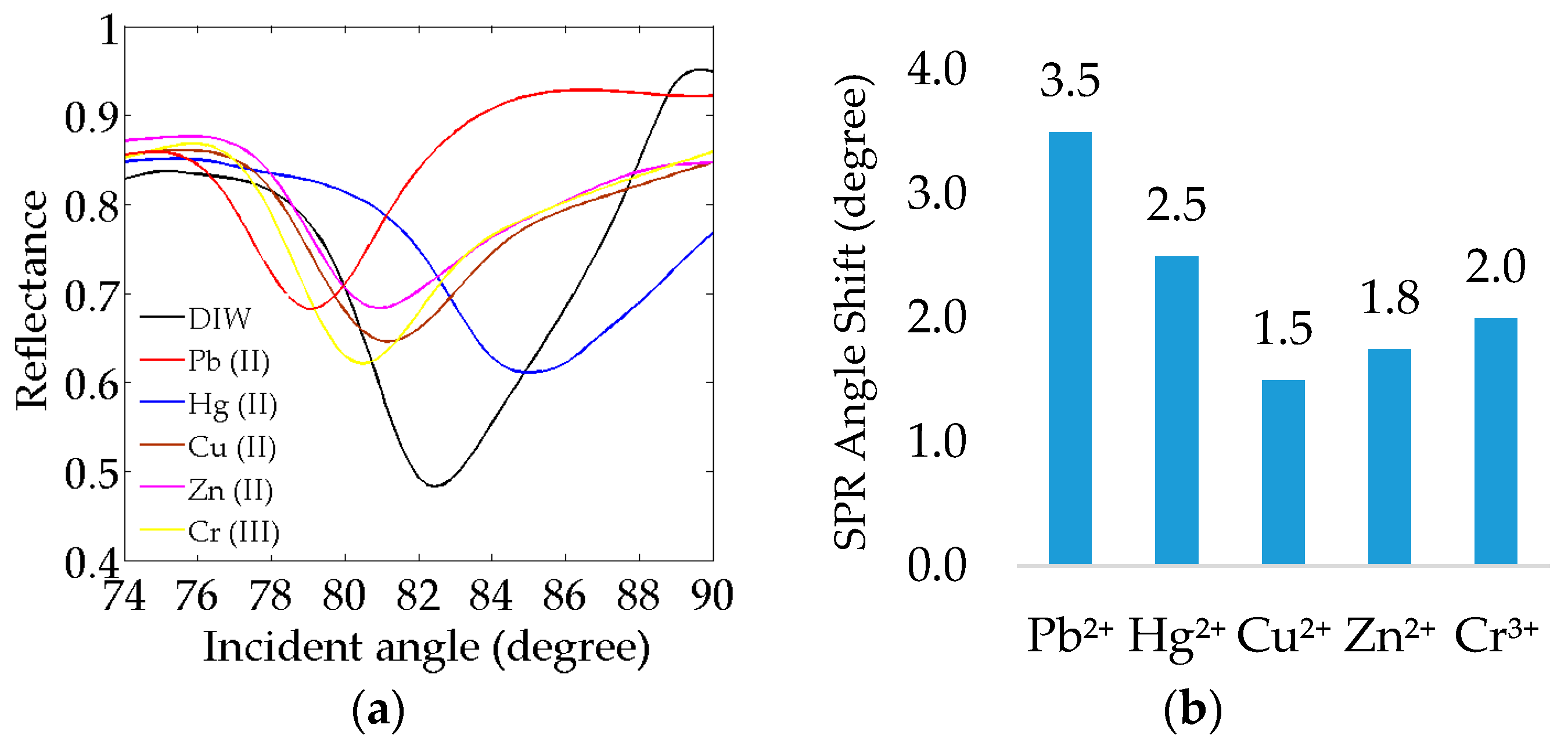
3.7. Shift in SPR Angle for Various Heavy Metal Ions
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McDonnell, J.M. Surface plasmon resonance: Towards an understanding of the mechanisms of biological molecular recognition. Curr. Opin. Chem. Biol. 2001, 5, 572–577. [Google Scholar] [CrossRef]
- Kastritis, P.L.; Bonvin, A.M. On the binding affinity of macromolecular interactions: Daring to ask why proteins interact. J. R. Soc. Interface 2013, 10, 20120835. [Google Scholar] [CrossRef] [PubMed]
- Cennamo, N.; Massarotti, D.; Galatus, R.; Conte, L.; Zeni, L. Performance comparison of two sensors based on surface plasmon resonance in a plastic optical fiber. Sensors 2013, 13, 721–735. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-M.; Lin, L.-Y. Immobilization of metallothionein as a sensitive biosensor chip for the detection of metal ions by surface plasmon resonance. Biosens. Bioelectron. 2004, 20, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Latour, R.A. Determination of the adsorption free energy for peptide-surface interactions by spr spectroscopy. Langmuir 2008, 24, 6721–6729. [Google Scholar] [CrossRef] [PubMed]
- O’Shannessy, D.J. Determination of kinetic rate and equilibrium binding constants for macromolecular interactions: A critique of the surface plasmon resonance literature. Curr. Opin. Biotechnol. 1994, 5, 65–71. [Google Scholar] [CrossRef]
- De Mol, N.J.; Fischer, M.J. Kinetic and thermodynamic analysis of ligand-receptor interactions: Spr applications in drug development. In Handbook of Surface Plasmon Resonance; Royal Society of Chemistry: Cambridge, UK, 2008; pp. 123–172. [Google Scholar]
- Forzani, E.S.; Foley, K.; Westerhoff, P.; Tao, N. Detection of arsenic in groundwater using a surface plasmon resonance sensor. Sens. Actuators B Chem. 2007, 123, 82–88. [Google Scholar] [CrossRef]
- Forzani, E.S.; Zhang, H.; Chen, W.; Tao, N. Detection of heavy metal ions in drinking water using a high-resolution differential surface plasmon resonance sensor. Environ. Sci. Technol. 2004, 39, 1257–1262. [Google Scholar] [CrossRef]
- Wang, R.; Wang, W.; Ren, H.; Chae, J. Detection of copper ions in drinking water using the competitive adsorption of proteins. Biosens. Bioelectron. 2014, 57, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, M.; Wang, Y.; Toledo, F.; Zhou, F. Studies of metal ion binding by apo-metallothioneins attached onto preformed self-assembled monolayers using a highly sensitive surface plasmon resonance spectrometer. Sens. Actuators B Chem. 2007, 123, 784–792. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, S.J. What is a “heavy metal”? J. Chem. Educ. 1997, 74, 1374. [Google Scholar] [CrossRef]
- Nagajyoti, P.C.; Lee, K.D.; Sreekanth, T.V.M. Heavy metals, occurrence and toxicity for plants: A review. Environ. Chem. Lett. 2010, 8, 199–216. [Google Scholar] [CrossRef]
- Habila, M.A.; Yilmaz, E.; Alothman, Z.A.; Soylak, M. Combination of dispersive liquid–liquid microextraction and multivariate optimization for separation-enrichment of traces lead by flame atomic absorption spectrometry. J. Ind. Eng. Chem. 2016, 37, 306–311. [Google Scholar] [CrossRef]
- Taylor, H.E. Inductively Coupled Plasma-Mass Spectrometry: Practices and Techniques; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
- Shih, T.-T.; Hsieh, C.-C.; Luo, Y.-T.; Su, Y.-A.; Chen, P.-H.; Chuang, Y.-C.; Sun, Y.-C. A high-throughput solid-phase extraction microchip combined with inductively coupled plasma-mass spectrometry for rapid determination of trace heavy metals in natural water. Anal. Chim. Acta 2016, 916, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Jankowski, K.; Yao, J.; Kasiura, K.; Jackowska, A.; Sieradzka, A. Multielement determination of heavy metals in water samples by continuous powder introduction microwave-induced plasma atomic emission spectrometry after preconcentration on activated carbon. Spectrochim. Acta Part B At. Spectrosc. 2005, 60, 369–375. [Google Scholar] [CrossRef]
- Lima, A.F.; Da Costa, M.C.; Ferreira, D.C.; Richter, E.M.; Munoz, R.A. Fast ultrasound-assisted treatment of inorganic fertilizers for mercury determination by atomic absorption spectrometry and microwave-induced plasma spectrometry with the aid of the cold-vapor technique. Microchem. J. 2015, 118, 40–44. [Google Scholar] [CrossRef]
- Neupane, L.N.; Oh, E.-T.; Park, H.J.; Lee, K.-H. Selective and sensitive detection of heavy metal ions in 100% aqueous solution and cells with a fluorescence chemosensor based on peptide using aggregation-induced emission. Anal. Chem. 2016, 88, 3333–3340. [Google Scholar] [CrossRef] [PubMed]
- Ermakova, E.; Michalak, J.; Meyer, M.; Arslanov, V.; Tsivadze, A.; Guilard, R.; Bessmertnykh-Lemeune, A. Colorimetric Hg2+ sensing in water: From molecules toward low-cost solid devices. Org. Lett. 2013, 15, 662–665. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Zhou, T.; Song, D.; Zhang, L. Monodispersed au nanoparticles decorated graphene as an enhanced sensing platform for ultrasensitive stripping voltammetric detection of mercury(ii). Sens. Actuators B Chem. 2010, 150, 491–497. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, X.; Li, C.; He, X.; Liu, G. On-site detection of heavy metals in agriculture land by a disposable sensor based virtual instrument. Comput. Electron. Agric. 2016, 123, 176–183. [Google Scholar] [CrossRef]
- Martín-Yerga, D.; Costa-García, A. Recent advances in the electrochemical detection of mercury. Curr. Opin. Electrochem. 2017. [Google Scholar] [CrossRef]
- Lu, Z.; Yang, S.; Yang, Q.; Luo, S.; Liu, C.; Tang, Y. A glassy carbon electrode modified with graphene, gold nanoparticles and chitosan for ultrasensitive determination of lead (ii). Microchim. Acta 2013, 180, 555–562. [Google Scholar] [CrossRef]
- Zhou, W.; Li, C.; Sun, C.; Yang, X. Simultaneously determination of trace Cd2+ and Pb2+ based on l-cysteine/graphene modified glassy carbon electrode. Food Chem. 2016, 192, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.A.; Hwang, T.; Dugasani, S.R.; Amin, R.; Kulkarni, A.; Park, S.H.; Kim, T. Graphene based fiber optic surface plasmon resonance for bio-chemical sensor applications. Sens. Actuators B Chem. 2013, 187, 426–433. [Google Scholar] [CrossRef]
- Patnaik, A.; Senthilnathan, K.; Jha, R. Graphene-based conducting metal oxide coated d-shaped optical fiber spr sensor. IEEE Photonics Technol. Lett. 2015, 27, 2437–2440. [Google Scholar] [CrossRef]
- Preechaburana, P.; Gonzalez, M.C.; Suska, A.; Filippini, D. Surface plasmon resonance chemical sensing on cell phones. Angew. Chem. 2012, 124, 11753–11756. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Q.; Chen, S.; Cheng, F.; Wang, H.; Peng, W. Surface plasmon resonance biosensor based on smart phone platforms. Sci. Rep. 2015, 5, 12864. [Google Scholar] [CrossRef] [PubMed]
- McIlwee, H.A.; Schauer, C.L.; Praig, V.G.; Boukherroub, R.; Szunerits, S. Thin chitosan films as a platform for spr sensing of ferric ions. Analyst 2008, 133, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Sadrolhosseini, A.R.; Moksin, M.M.; Yunus, W.M.M.; Talib, Z.A.; Abdi, M.M. Surface plasmon resonance detection of copper corrosion in biodiesel using polypyrrole-chitosan layer sensor. Opt. Rev. 2011, 18, 331–337. [Google Scholar] [CrossRef]
- Sadrolhosseini, A.R.; Noor, A.; Moksin, M.M.; Abdi, M.; Mohammadi, A. Application of polypyrrole-chitosan layer for detection of Zn (II) and Ni (II) in aqueous solutions using surface plasmon resonance. Int. J. Polym. Mater. Polym. Biomater. 2013, 62, 284–287. [Google Scholar] [CrossRef]
- Varma, A.; Deshpande, S.; Kennedy, J. Metal complexation by chitosan and its derivatives: A review. Carbohydr. Polym. 2004, 55, 77–93. [Google Scholar] [CrossRef]
- Abdi, M.M.; Abdullah, L.C.; Sadrolhosseini, A.R.; Mat Yunus, W.M.; Moksin, M.M.; Tahir, P.M. Surface plasmon resonance sensing detection of mercury and lead ions based on conducting polymer composite. PLoS ONE 2011, 6, e24578. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-W.; Lin, S.; Chang, C.-C. A reversible optical sensor based on chitosan film for the selective detection of copper ions. Biomed. Eng. Appl. Basis Commun. 2012, 24, 453–459. [Google Scholar] [CrossRef]
- Chiu, N.-F.; Huang, T.-Y.; Lai, H.-C. Graphene oxide based surface plasmon resonance. Biosensors 2013. [Google Scholar] [CrossRef]
- Fan, L.; Luo, C.; Sun, M.; Li, X.; Lu, F.; Qiu, H. Preparation of novel magnetic chitosan/graphene oxide composite as effective adsorbents toward methylene blue. Bioresour. Technol. 2012, 114, 703–706. [Google Scholar] [CrossRef] [PubMed]
- Szunerits, S.; Maalouli, N.; Wijaya, E.; Vilcot, J.-P.; Boukherroub, R. Recent advances in the development of graphene-based surface plasmon resonance (spr) interfaces. Anal. Bioanal. Chem. 2013, 405, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Kyzas, G.Z.; Deliyanni, E.A.; Matis, K.A. Graphene oxide and its application as an adsorbent for wastewater treatment. J. Chem. Technol. Biotechnol. 2014, 89, 196–205. [Google Scholar] [CrossRef]
- Lokman, N.F.; Bakar, A.A.A.; Suja, F.; Abdullah, H.; Rahman, W.B.W.A.; Huang, N.-M.; Yaacob, M.H. Highly sensitive spr response of au/chitosan/graphene oxide nanostructured thin films toward Pb (II) ions. Sens. Actuators B Chem. 2014, 195, 459–466. [Google Scholar] [CrossRef]
- Kamaruddin, N.H.; Bakar, A.A.A.; Yaacob, M.H.; Mahdi, M.A.; Zan, M.S.D.; Shaari, S. Enhancement of chitosan-graphene oxide spr sensor with a multi-metallic layers of au–ag–au nanostructure for lead (ii) ion detection. Appl. Surf. Sci. 2016, 361, 177–184. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Introduction to United States Environmental Protection Agency Hazardous Waste Identification (40 cfr parts 261); United States Environmental Protection Agency: Washington, DC, USA, 2005.
- Bustos-Ramírez, K.; Martínez-Hernández, A.L.; Martínez-Barrera, G.; Icaza, M.; Castaño, V.M.; Velasco-Santos, C. Covalently bonded chitosan on graphene oxide via redox reaction. Materials 2013, 6, 911–926. [Google Scholar] [CrossRef] [PubMed]
- Salam, A.; Pawlak, J.J.; Venditti, R.A.; El-tahlawy, K. Synthesis and characterization of starch citrate–chitosan foam with superior water and saline absorbance properties. Biomacromolecules 2010, 11, 1453–1459. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.S.K.; Jiang, S.-J. Chitosan-functionalized graphene oxide: A novel adsorbent an efficient adsorption of arsenic from aqueous solution. J. Environ. Chem. Eng. 2016, 4, 1698–1713. [Google Scholar] [CrossRef]
- Kim, D.S.; Dhand, V.; Rhee, K.Y.; Park, S.-J. Study on the effect of silanization and improvement in the tensile behavior of graphene-chitosan-composite. Polymers 2015, 7, 527–551. [Google Scholar] [CrossRef]
- Yan, T.; Zhang, H.; Huang, D.; Feng, S.; Fujita, M.; Gao, X.-D. Chitosan-functionalized graphene oxide as a potential immunoadjuvant. Nanomaterials 2017, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Fen, Y.W.; Yunus, W.M.M.; Yusof, N.A. Surface plasmon resonance optical sensor for detection of Pb2+ based on immobilized p-tert-butylcalix[4]arene-tetrakis in chitosan thin film as an active layer. Sens. Actuators B Chem. 2012, 171–172, 287–293. [Google Scholar] [CrossRef]
- Fen, Y.W.; Yunus, W.M.M.; Talib, Z.A. Analysis of Pb (ii) ion sensing by crosslinked chitosan thin film using surface plasmon resonance spectroscopy. Opt.-Int. J. Light Electron. Opt. 2013, 124, 126–133. [Google Scholar] [CrossRef]
- Saha, S.; Sarkar, P. Differential pulse anodic stripping voltammetry for detection of as (iii) by chitosan-fe (oh) 3 modified glassy carbon electrode: A new approach towards speciation of arsenic. Talanta 2016, 158, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Gupta, B.D. Fabrication and characterization of a surface plasmon resonance based fiber optic sensor using gel entrapment technique for the detection of low glucose concentration. Sens. Actuators B Chem. 2013, 177, 589–595. [Google Scholar] [CrossRef]
- Tamir, T. Leaky waves in planar optical waveguides. Nouvelle Revue d'Optique 1975, 6, 273–284. [Google Scholar] [CrossRef]
- Tamir, T.; Oliner, A.A. The spectrum of electromagnetic waves guided by a plasma layer. Proc. IEEE 1963, 51, 317–332. [Google Scholar] [CrossRef]
- Yin, X.; Hesselink, L.; Liu, Z.; Fang, N.; Zhang, X. Large positive and negative lateral optical beam displacements due to surface plasmon resonance. Appl. Phys. Lett. 2004, 85, 372–374. [Google Scholar] [CrossRef]
- Kim, N.-H.; Kim, T.W.; Byun, K.M. How to avoid a negative shift in reflection-type surface plasmon resonance biosensors with metallic nanostructures. Opt. Expr. 2014, 22, 4723–4730. [Google Scholar] [CrossRef] [PubMed]
- Zynio, S.; Samoylov, A.; Surovtseva, E.; Mirsky, V.; Shirshov, Y. Bimetallic layers increase sensitivity of affinity sensors based on surface plasmon resonance. Sensors 2002, 2, 62–70. [Google Scholar] [CrossRef]
- Lee, K.-S.; Son, J.M.; Jeong, D.-Y.; Lee, T.S.; Kim, W.M. Resolution enhancement in surface plasmon resonance sensor based on waveguide coupled mode by combining a bimetallic approach. Sensors 2010, 10, 11390–11399. [Google Scholar] [CrossRef] [PubMed]
- Zijlstra, P.; Paulo, P.M.R.; Yu, K.; Xu, Q.-H.; Orrit, M. Chemical interface damping in single gold nanorods and its near elimination by tip-specific functionalization. Angew. Chem. Int. Ed. 2012, 51, 8352–8355. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cheng, Z.; Singh, V.; Zheng, Z.; Wang, Y.; Li, S.; Song, L.; Zhu, J. Stable and sensitive silver surface plasmon resonance imaging sensor using trilayered metallic structures. Anal. Chem. 2013, 86, 1430–1436. [Google Scholar] [CrossRef] [PubMed]
- Raether, H. Surface Plasmons on Smooth Surfaces; Springer: Berlin, Germany, 1988. [Google Scholar]
- Jose, J.; Segerink, F.; Korterik, J.; Gomez-Casado, A.; Huskens, J.; Herek, J.; Offerhaus, H. Enhanced surface plasmon polariton propagation length using a buried metal grating. J. Appl. Phys. 2011, 109, 064906. [Google Scholar] [CrossRef]
- Bayramoglu, G.; Yakup Arica, M.; Bektas, S. Removal of Cd(ii), Hg(ii), and Pb(ii) ions from aqueous solution using p(hema/chitosan) membranes. J. Appl. Polym. Sci. 2007, 106, 169–177. [Google Scholar] [CrossRef]
- Pearson, R.G. Hard and soft acids and bases. J. Am. Chem. Soc. 1963, 85, 3533–3539. [Google Scholar] [CrossRef]
- Parr, R.G.; Pearson, R.G. Absolute hardness: Companion parameter to absolute electronegativity. J. Am. Chem. Soc. 1983, 105, 7512–7516. [Google Scholar] [CrossRef]
- Pearson, R.G. Chemical hardness and density functional theory. J. Chem. Sci. 2005, 117, 369–377. [Google Scholar] [CrossRef]
- Fan, L.; Luo, C.; Sun, M.; Li, X.; Qiu, H. Highly selective adsorption of lead ions by water-dispersible magnetic chitosan/graphene oxide composites. Colloids Surf. B Biointerfaces 2013, 103, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Gedam, A.H.; Dongre, R.S.; Bansiwal, A.K. Synthesis and characterization of graphite doped chitosan composite for batch adsorption of lead (ii) ions from aqueous solution. Adv. Mater. Lett. 2015, 6, 59–67. [Google Scholar] [CrossRef]
- Weiner, E.R. Applications of Environmental Aquatic Chemistry: A Practical Guide; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Sharma, A.K.; Jha, R.; Gupta, B.D. Fiber-optic sensors based on surface plasmon resonance: A comprehensive review. Sens. J. IEEE 2007, 7, 1118–1129. [Google Scholar] [CrossRef]
- Sharma, A.K.; Gupta, B.D. On the performance of different bimetallic combinations in surface plasmon resonance based fiber optic sensors. J. Appl. Phys. 2007, 101, 093111. [Google Scholar] [CrossRef]
- Yu, X.; Xu, D.; Cheng, Q. Label-free detection methods for protein microarrays. Proteomics 2006, 6, 5493–5503. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Forzani, E.S.; Tao, N. Detection of heavy metal ions in water by high-resolution surface plasmon resonance spectroscopy combined with anodic stripping voltammetry. Anal. Chem. 2007, 79, 4427–4432. [Google Scholar] [CrossRef] [PubMed]
- Homola, J.R. SURFACE Plasmon Resonance Based Sensors; Springer Science and Business Media: Heidelberg, Germany, 2006; Volume 4. [Google Scholar]
| Heavy Metal Ions | SPR Sensor | K (M−1) | References |
|---|---|---|---|
| Ni2+ | MUA-(His)6- | 4.0 × 108 | [9] |
| Cu2+ | MUA/Gly-Gly-His | 6.0 × 108 | |
| Cu2+ | Cysteamine/His-Gly-Gly | 4.0 × 106 | |
| Fe3+ | CS | 9.5 × 105 | [30] |
| Cd2+ | Apo-metallothionein | 4.2 × 105 | [11] |
| Hg2+ | Apo-metallothionein | 2.7 × 103 | |
| Cu2+ | Polypyrrole–CS | 1.3 × 104 | [31] |
| Zn2+ | Polypyrrole–CS | 2.3 × 104 | [32] |
| Ni2+ | Polypyrrole–CS | 1.7 × 104 | |
| Cu2+ | Albumin | 2.3 × 102 | [10] |
| Pb2+ | Albumin | 2.4 × 102 | |
| Hg2+ | Albumin | 4.3 × 102 |
| Element | Weight Percentage (%) |
|---|---|
| Carbon (C) | 19.25 |
| Oxygen (O) | 32.62 |
| Silver (Ag) | 27.58 |
| Gold (Au) | 20.54 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamaruddin, N.H.; Bakar, A.A.A.; Mobarak, N.N.; Zan, M.S.D.; Arsad, N. Binding Affinity of a Highly Sensitive Au/Ag/Au/Chitosan-Graphene Oxide Sensor Based on Direct Detection of Pb2+ and Hg2+ Ions. Sensors 2017, 17, 2277. https://doi.org/10.3390/s17102277
Kamaruddin NH, Bakar AAA, Mobarak NN, Zan MSD, Arsad N. Binding Affinity of a Highly Sensitive Au/Ag/Au/Chitosan-Graphene Oxide Sensor Based on Direct Detection of Pb2+ and Hg2+ Ions. Sensors. 2017; 17(10):2277. https://doi.org/10.3390/s17102277
Chicago/Turabian StyleKamaruddin, Nur Hasiba, Ahmad Ashrif A. Bakar, Nadhratun Naiim Mobarak, Mohd Saiful Dzulkefly Zan, and Norhana Arsad. 2017. "Binding Affinity of a Highly Sensitive Au/Ag/Au/Chitosan-Graphene Oxide Sensor Based on Direct Detection of Pb2+ and Hg2+ Ions" Sensors 17, no. 10: 2277. https://doi.org/10.3390/s17102277
APA StyleKamaruddin, N. H., Bakar, A. A. A., Mobarak, N. N., Zan, M. S. D., & Arsad, N. (2017). Binding Affinity of a Highly Sensitive Au/Ag/Au/Chitosan-Graphene Oxide Sensor Based on Direct Detection of Pb2+ and Hg2+ Ions. Sensors, 17(10), 2277. https://doi.org/10.3390/s17102277






