Reagent-Less and Robust Biosensor for Direct Determination of Lactate in Food Samples
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Apparatus
2.2. Procedures
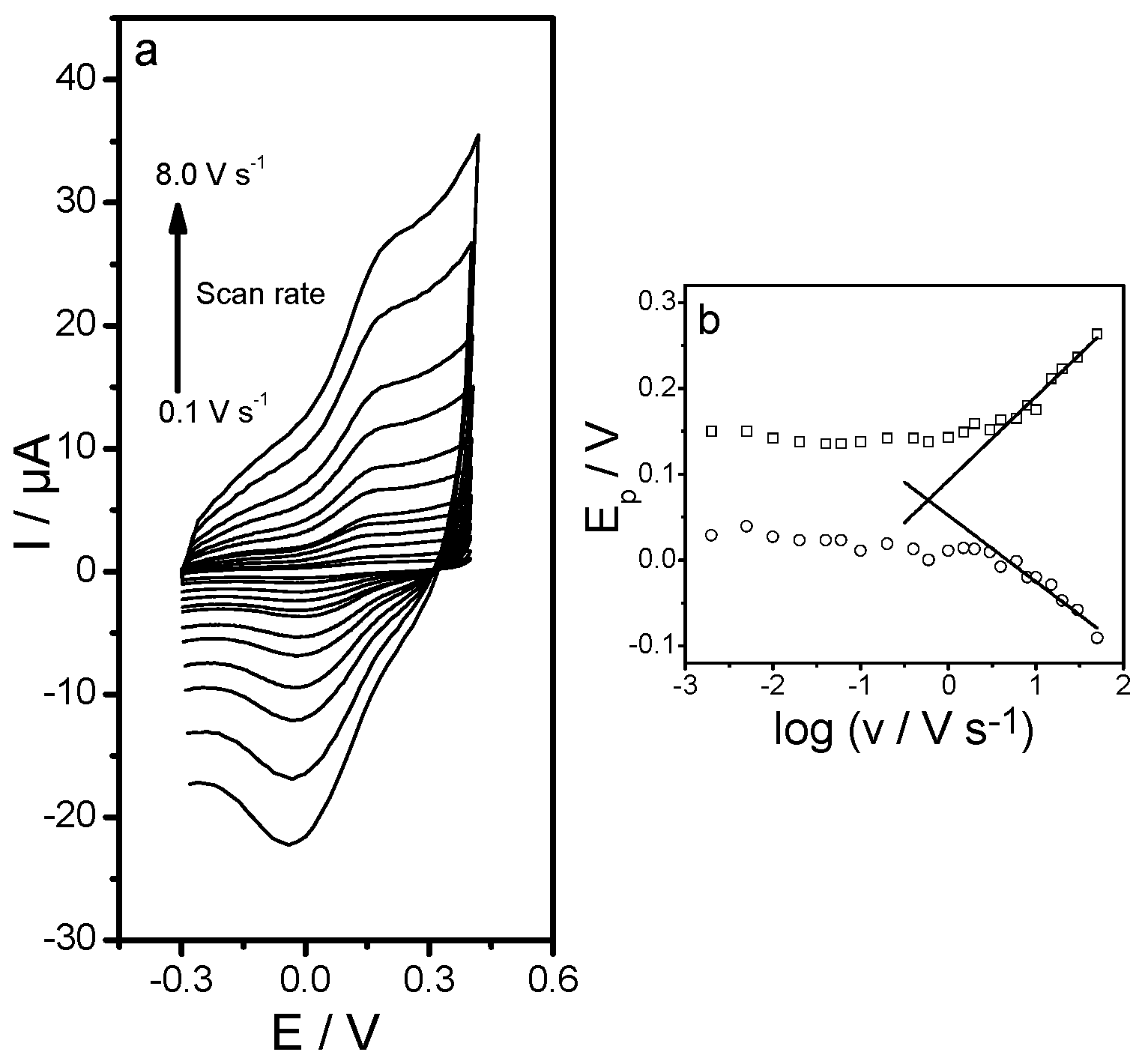
2.2.1. Synthesis of 3,4DHS Capped Gold Nanoparticles (3,4DHS–AuNPs)
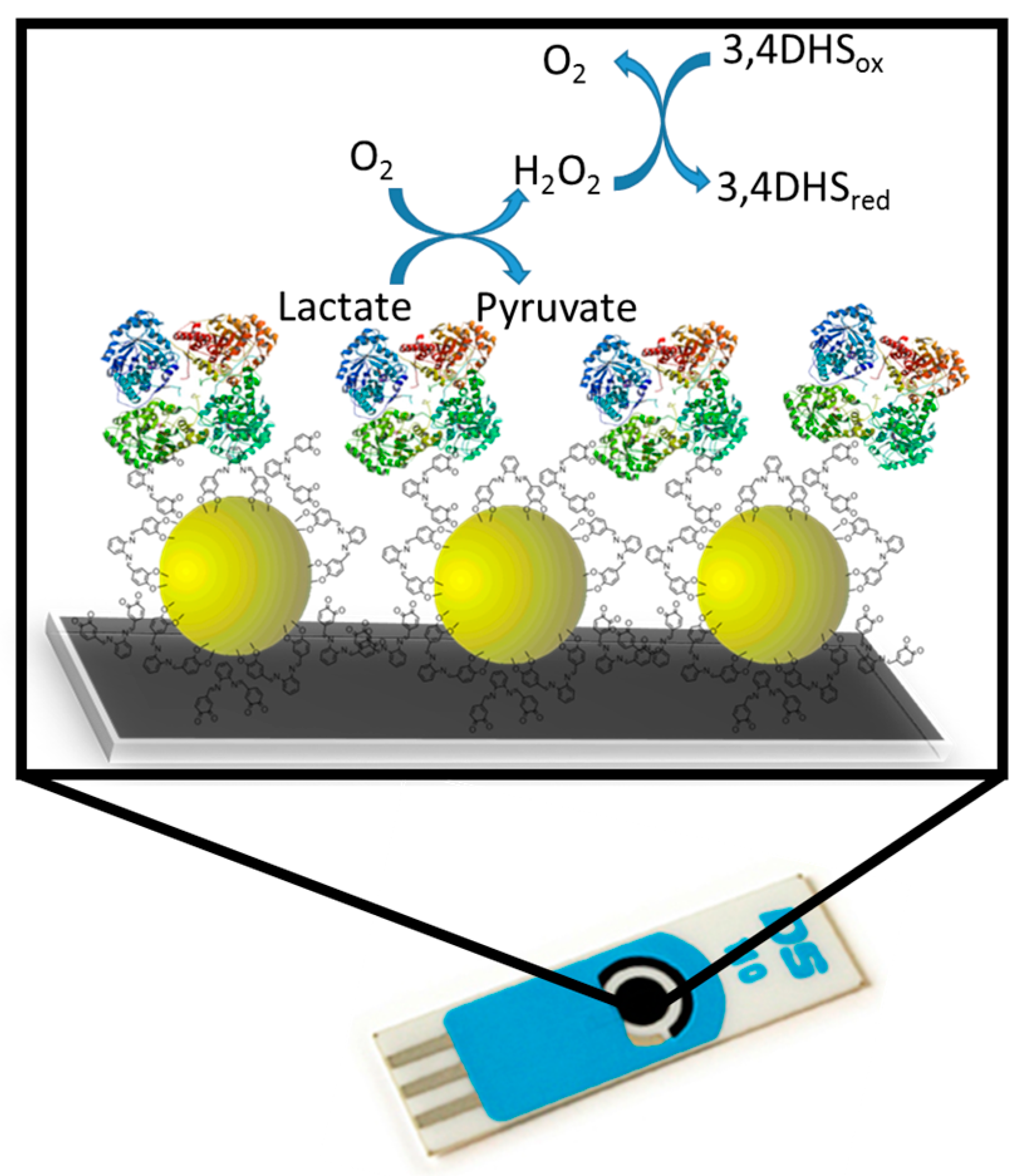
2.2.2. Biosensor Preparation
2.2.3. Determination of Lactate in Food Samples
3. Results and Discussion
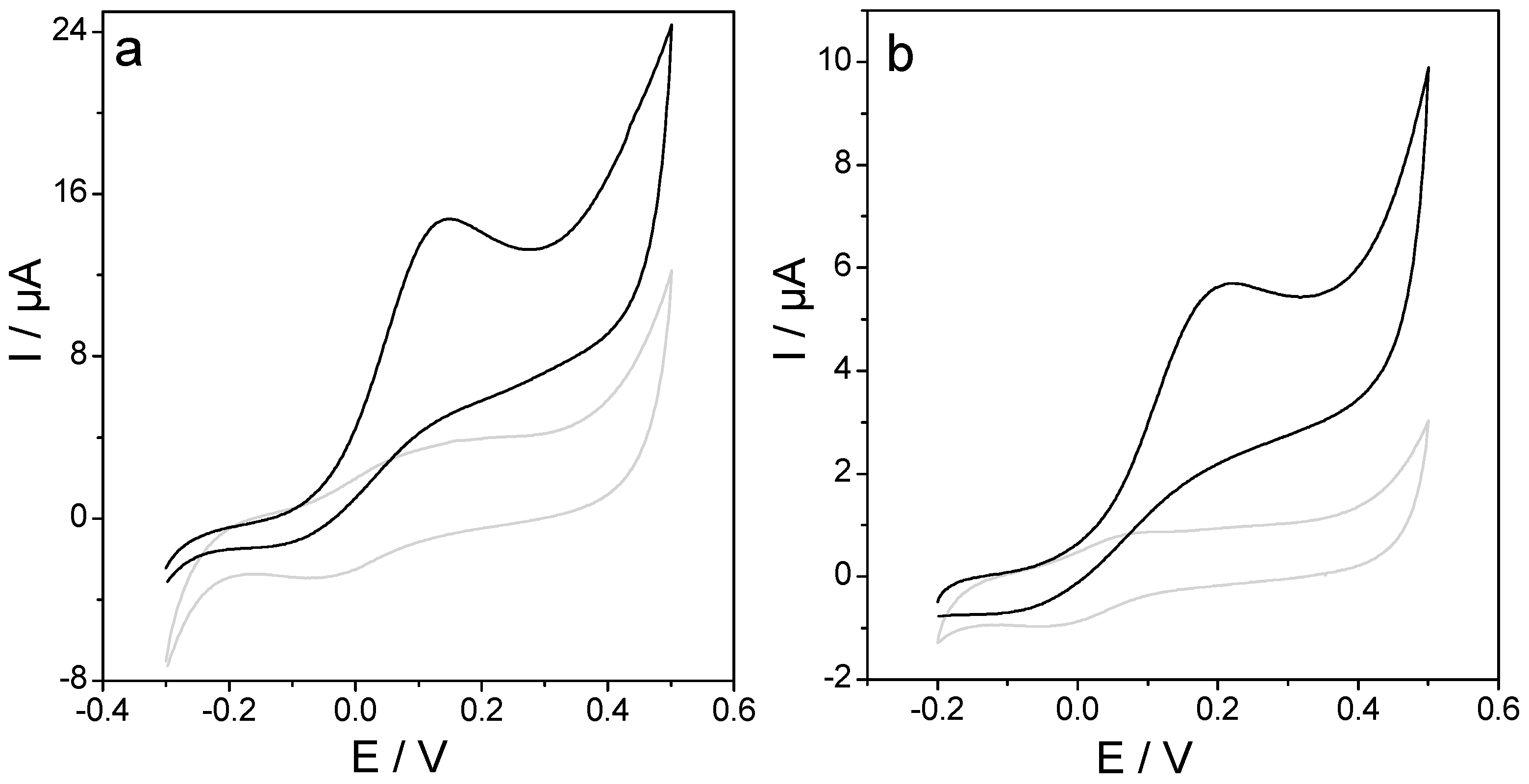
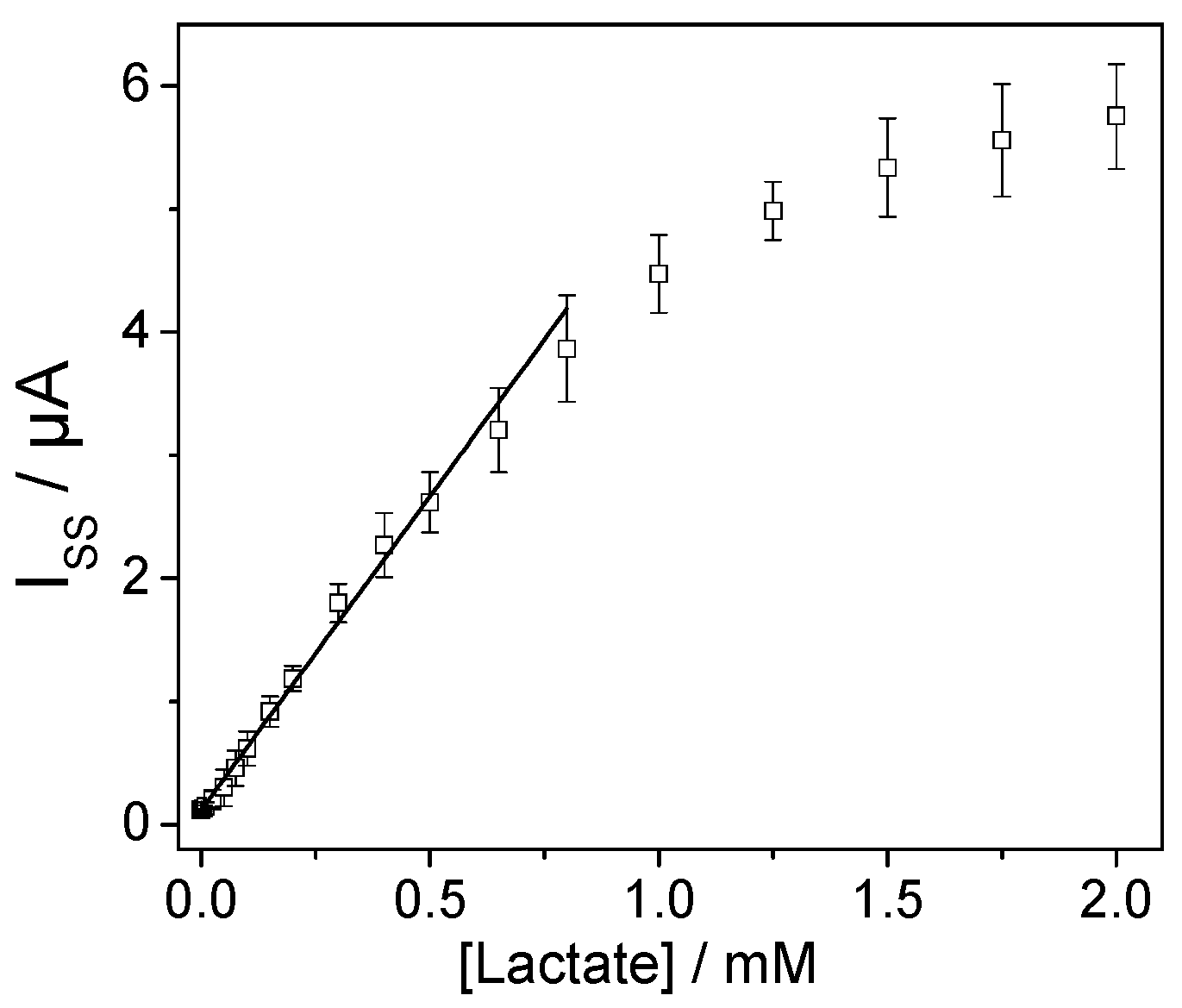
3.1. Lactate Oxidase Biosensor Development
3.2. Study of Common Interfering Substances on the Response of Lactate
3.3. Determination of Lactate in Real Samples
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stanley, W.C.; Gertz, E.W.; Wisneski, J.A.; Morris, D.L.; Neese, R.A.; Brooks, G.A. Systemic lactate kinetics during graded exercise in man. Am. J. Physiol. 1985, 249, E595–E602. [Google Scholar] [PubMed]
- Kemp, G. Lactate accumulation, proton buffering, and pH change in ischemically exercising muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 289, R895–R901. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.A. Anaerobic threshold: Review of the concept and directions for future research. Med. Sci. Sports Exerc. 1985, 17, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Valenza, F.; Aletti, G.; Fossali, T.; Chevallard, G.; Sacconi, F.; Irace, M.; Gattinoni, L. Lactate as a marker of energy failure in critically ill patients: Hypothesis. Crit. Care 2005, 9, 588–593. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Karlsson, J.; Willerson, J.T.; Leshin, S.J.; Mullins, C.B.; Mitchell, J.H. Skeletal-muscle metabolites in patients with cardiogenic-shock or severe congestive heart failure. Scand. J. Clin. Lab. Investig. 1975, 35, 73–79. [Google Scholar] [CrossRef]
- Sayeed, M.M.; Murthy, P.N.A. Adenine nucleotide and lactate metabolism in the lung in endotoxin shock. Circ. Shock 1981, 8, 657–666. [Google Scholar] [PubMed]
- DeBacker, D.; Creteur, J.; Zhang, H.B.; Norrenberg, M.; Vincent, J.L. Lactate production by the lungs in acute lung injury. Am. J. Respir. Crit. Care Med. 1997, 156, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Kruse, J.A.; Zaidi, S.A.; Carlson, R.W. Significance of blood lactate levels in critically ill patients with liver disease. Am. J. Med. 1987, 83, 77–82. [Google Scholar] [CrossRef]
- Hirschhaeuser, F.; Sattler, U.G.A.; Mueller-Klieser, W. Lactate: A metabolic key player in cancer. Cancer Res. 2011, 71, 6921–6925. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Tumor metabolism: Cancer cells give and take lactate. J. Clin. Investig. 2008, 118, 3835–3837. [Google Scholar] [CrossRef] [PubMed]
- Przybyt, M. Lactate biosensors for food industry. Biotechnol. Food Sci. 2014, 78, 71–88. [Google Scholar]
- Nikolaus, N.; Strehlitz, B. Amperometric lactate biosensors and their application in (sports) medicine, for life quality and wellbeing. Microchim. Acta 2008, 160, 15–55. [Google Scholar] [CrossRef]
- Shinbo, T.; Sugiura, M.; Kamo, N. Potentiometric enzyme electrode for lactate. Anal. Chem. 1979, 51, 100–104. [Google Scholar] [CrossRef]
- Omole, O.O.; Brocks, D.R.; Nappert, G.; Naylor, J.M.; Zello, G.A. High-performance liquid chromatographic assay of (+/−)-lactic acid and its enantiomers in calf serum. J. Chromatogr. B Biomed. Sci. Appl. 1999, 727, 23–29. [Google Scholar] [CrossRef]
- Galbán, J.; Demarcos, S.; Castillo, J.R. Fluorometric–enzymatic lactate determination based on enzyme cytochrome b2 fluorescence. Anal. Chem. 1993, 65, 3076–3080. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.Q.; Huang, Y.M.; Huang, C.Z. Chemiluminescence biosensor system for lactic acid using natural animal tissue as recognition element. Biosens. Bioelectron. 2005, 21, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Goh, J.H.; Mason, A.; Al-Shamma’a, A.I.; Field, M.; Shackcloth, M.; Browning, P. Non invasive microwave sensor for the detection of lactic acid in cerebrospinal fluid (CSF). J. Phys. Conf. Ser. 2011, 307, 012017. [Google Scholar] [CrossRef]
- Sartain, F.K.; Yang, X.; Lowe, C.R. Holographic lactate sensor. Anal. Chem. 2006, 78, 5664–5670. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Sherry, A.D.; Malloy, C.R. Noninvasive monitoring of lactate dynamics in human forearm muscle after exhaustive exercise by 1H-magnetic resonance spectroscopy at 7 tesla. Magn. Reson. Med. 2013, 70, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Razumas, V.; Kanapieniene, J.; Nylander, T.; Engstrom, S.; Larsson, K. Electrochemical biosensors for glucose, lactate, urea, and creatinine based on enzymes entrapped in a cubic liquid crystalline phase. Anal. Chim. Acta 1994, 289, 155–162. [Google Scholar] [CrossRef]
- Liu, H.; Kong, J.; Deng, J. An Amperometric Lactate Sensor Using Tetrathiafulvalene in Polyester Ionomer Film as Electron Transfer. Anal. Lett. 1995, 28, 563–579. [Google Scholar] [CrossRef]
- Pfeiffer, D.; Mollor, B.; Klimon, N.; Szoponik, J. Amperometric lactate oxidase catheter for real-time lactate monitoring based on thin film technology. Biosens. Bioelectron. 1997, 12, 539–550. [Google Scholar] [CrossRef]
- Suman, S.; Singhal, R.; Sharma, A.L.; Malthotra, B.D.; Pundir, C.S. Development of a lactate biosensor based on conducting copolymer bound lactate oxidase. Sens. Actuators B Chem. 2005, 107, 768–772. [Google Scholar] [CrossRef]
- Chaubey, A.; Pande, K.K.; Pandey, M.K.; Singh, V.S. Signal amplification by substrate recycling on polyaniline/lactate oxidase/lactate dehydrogenase bienzyme electrodes. Appl. Biochem. Biotechnol. 2001, 96, 239–248. [Google Scholar] [CrossRef]
- Qhobosheane, M.; Santra, S.; Zhang, P.; Tan, W. Biochemically functionalized silica nanoparticles. Analyst 2001, 126, 1274–1278. [Google Scholar] [CrossRef] [PubMed]
- Briones, M.; Casero, E.; Petit Dominquez, M.D.; Ruiz, M.A.; Parra-Alfambra, A.M.; Pariente, F.; Lorenzo, E.; Vazquez, L. Diamond nanoparticles based biosensors for efficient glucose and lactate determination. Biosens. Bioelectron. 2015, 68, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Boero, C.; Carrara, S.; Vecchio, G.D.; Calza, L.; Micheli, G.D. Highly Sensitive Carbon Nanotube-Based Sensing for Lactate and Glucose Monitoring in Cell Culture. IEEE Trans. Nanobiosci. 2011, 10, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Lamas-Ardisana, P.J.; Loaiza, O.A.; Añorga, L.; Jubete, E.; Borghei, M.; Ruiz, V.; Ochoteco, E.; Cabañero, G.; Grande, H.J. Disposable amperometric biosensor based on lactate oxidase immobilised on platinum nanoparticle-decorated carbon nanofiber and poly(diallyldimethylammonium chloride) films. Biosens. Bioelectron. 2014, 56, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Loaiza, O.A.; Lamas-Ardisana, P.J.; Añorga, L.; Jubete, E.; Ruiz, V.; Borghei, M.; Cabañero, G.; Grande, H.J. Graphitized carbon nanofiber-Pt nanoparticle hybrids as sensitive tool for preparation of screen printing biosensors. Detection of lactate in wines and ciders. Bioelectrochemistry 2015, 101, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Parra, A.; Casero, E.; Lorenzo, E.; Pariente, F.; Vazquez, L. Nanomechanical properties of globular proteins: Lactate oxidase. Langmuir 2007, 23, 2747–2754. [Google Scholar] [CrossRef] [PubMed]
- Makovos, E.B.; Liu, C.C. Measurements of lactate concentration using lactate oxidase and an electrochemical oxygen sensor. Biotechnol. Bioeng. 1985, 27, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Jin, Y.L. Sensitive lactate determination based on acclimated mixed bacteria and palygorskite co-modified oxygen electrode. Bioelectrochemistry 2011, 80, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Jena, B.K.; Raj, C.R. Amperometric l-lactate biosensor based on gold nanoparticles. Electroanalysis 2007, 19, 816–822. [Google Scholar] [CrossRef]
- Abad, J.M.; Bravo, I.; Pariente, F.; Lorenzo, E. Multi-tasking Schiff base ligand: A new concept of AuNPs synthesis. Anal. Bioanal. Chem. 2016, 408, 2329–2338. [Google Scholar] [CrossRef] [PubMed]
- Revenga-Parra, M.; Lorenzo, E.; Pariente, F. Synthesis and electrocatalytic activity towards oxidation of hydrazine of a new family of hydroquinone salophen derivatives: Application to the construction of hydrazine sensors. Sens. Actuators B Chem. 2005, 107, 678–687. [Google Scholar] [CrossRef]
- Haiss, W.; Thanh, N.T.K.; Aveyard, J.; Fernig, D.G. Determination of size and concentration of gold nanoparticles from UV-Vis spectra. Anal. Chem. 2007, 79, 4215–4221. [Google Scholar] [CrossRef] [PubMed]
- Abad, J.M.; Revenga-Parra, M.; Garcia, T.; Gamero, M.; Lorenzo, E.; Pariente, F. Interactions of Schiff-base ligands with gold nanoparticles: Structural, optical and electrocatalytic studies. Phys. Chem. Chem. Phys. 2011, 13, 5668–5678. [Google Scholar] [CrossRef] [PubMed]
- Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem. 1979, 101, 19–28. [Google Scholar] [CrossRef]
- Azzouzi, S.; Rotariu, L.; Benito, A.M.; Maser, W.K.; Ben Ali, M.; Bala, C. A novel amperometric biosensor based on gold nanoparticles anchored on reduced graphene oxide for sensitive detection of l-lactate tumor biomarker. Biosens. Bioelectron 2015, 69, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Nesakumar, N.; Sethuraman, S.; Krishnan, U.M.; Rayappan, J.B.B. Fabrication of lactate biosensor based on lactate dehydrogenase immobilized on cerium oxide nanoparticles. J. Colloid Interface Sci. 2013, 410, 158–164. [Google Scholar] [CrossRef] [PubMed]


| Compound | Current Ratio 1 | |
|---|---|---|
| 1:1 | 1:0.1 | |
| Tartaric acid | 1.05 | 0.97 |
| Citric acid | 1.08 | 1.00 |
| Acetic acid | 1.16 | 0.88 |
| Ascorbic acid | 1.76 | 1.08 |
| Glucose | 1.07 | 0.96 |
| Fructose | 1.01 | 0.99 |
| Methanol | 1.12 | 0.97 |
| Ethanol | 1.10 | 0.94 |
| Lactate Content (g/L) ± SD 1 | Lactate Content (g/L) ± SD 1 | |
|---|---|---|
| Sample | (LOx Biosensor) | (Commercial Assay Kit) |
| White wine | 1.7 ± 0.1 | 1.73 ± 0.02 |
| Beer | 0.10 ± 0.02 | 0.100 ± 0.001 |
| Yogurt | 5.4 ± 0.6 | 5.7 ± 0.4 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bravo, I.; Revenga-Parra, M.; Pariente, F.; Lorenzo, E. Reagent-Less and Robust Biosensor for Direct Determination of Lactate in Food Samples. Sensors 2017, 17, 144. https://doi.org/10.3390/s17010144
Bravo I, Revenga-Parra M, Pariente F, Lorenzo E. Reagent-Less and Robust Biosensor for Direct Determination of Lactate in Food Samples. Sensors. 2017; 17(1):144. https://doi.org/10.3390/s17010144
Chicago/Turabian StyleBravo, Iria, Mónica Revenga-Parra, Félix Pariente, and Encarnación Lorenzo. 2017. "Reagent-Less and Robust Biosensor for Direct Determination of Lactate in Food Samples" Sensors 17, no. 1: 144. https://doi.org/10.3390/s17010144
APA StyleBravo, I., Revenga-Parra, M., Pariente, F., & Lorenzo, E. (2017). Reagent-Less and Robust Biosensor for Direct Determination of Lactate in Food Samples. Sensors, 17(1), 144. https://doi.org/10.3390/s17010144






