Electrochemical Aptasensor for Myoglobin-Specific Recognition Based on Porphyrin Functionalized Graphene-Conjugated Gold Nanocomposites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.2. Synthesis of the TCPP–Gr and TCPP–Gr/AuNPs
2.3. Fabrication of the Sensing Interface
2.4. Electrochemical Detection of Myoglobin
3. Results and Discussion
3.1. The Principle of the Aptasensor
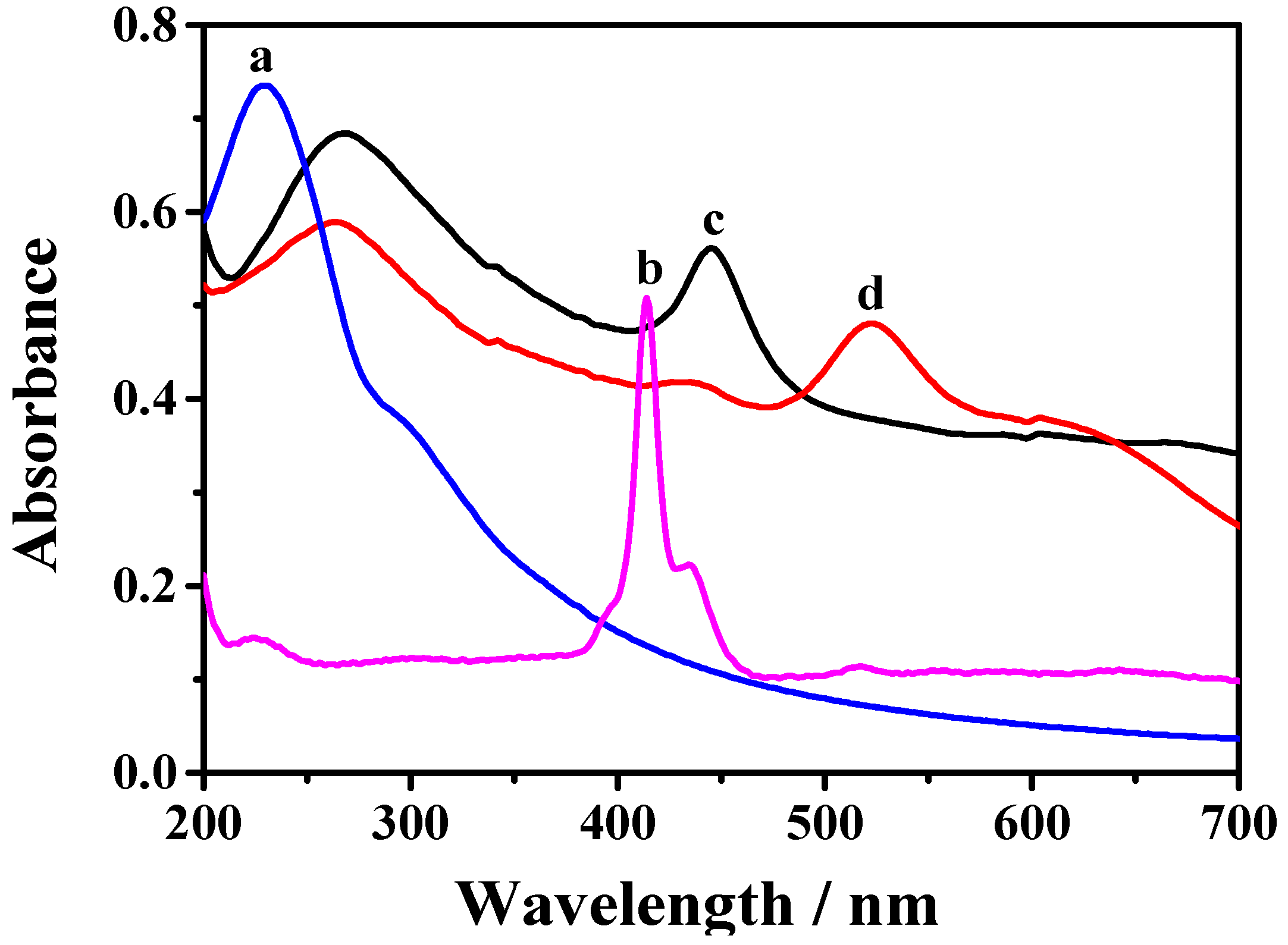
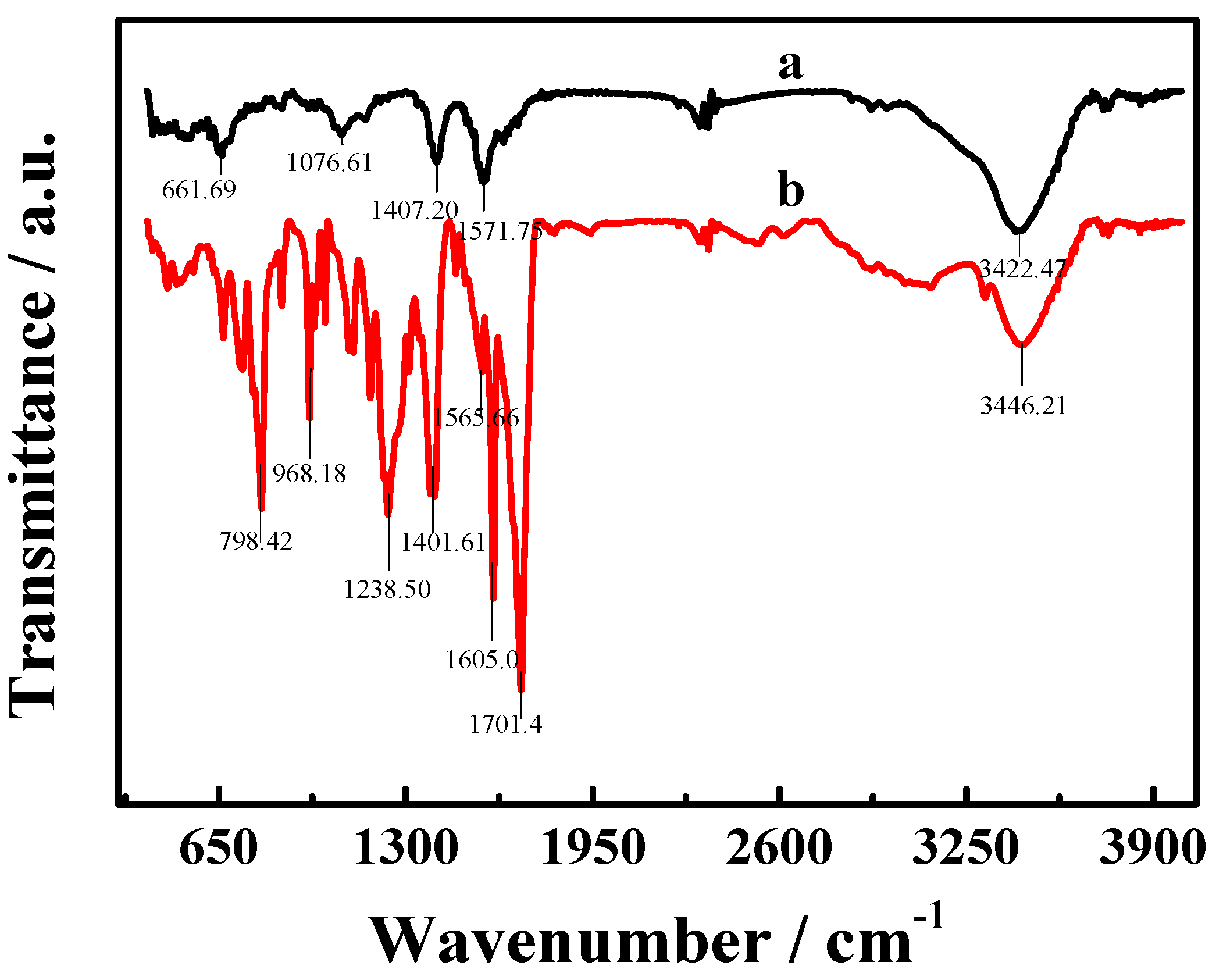
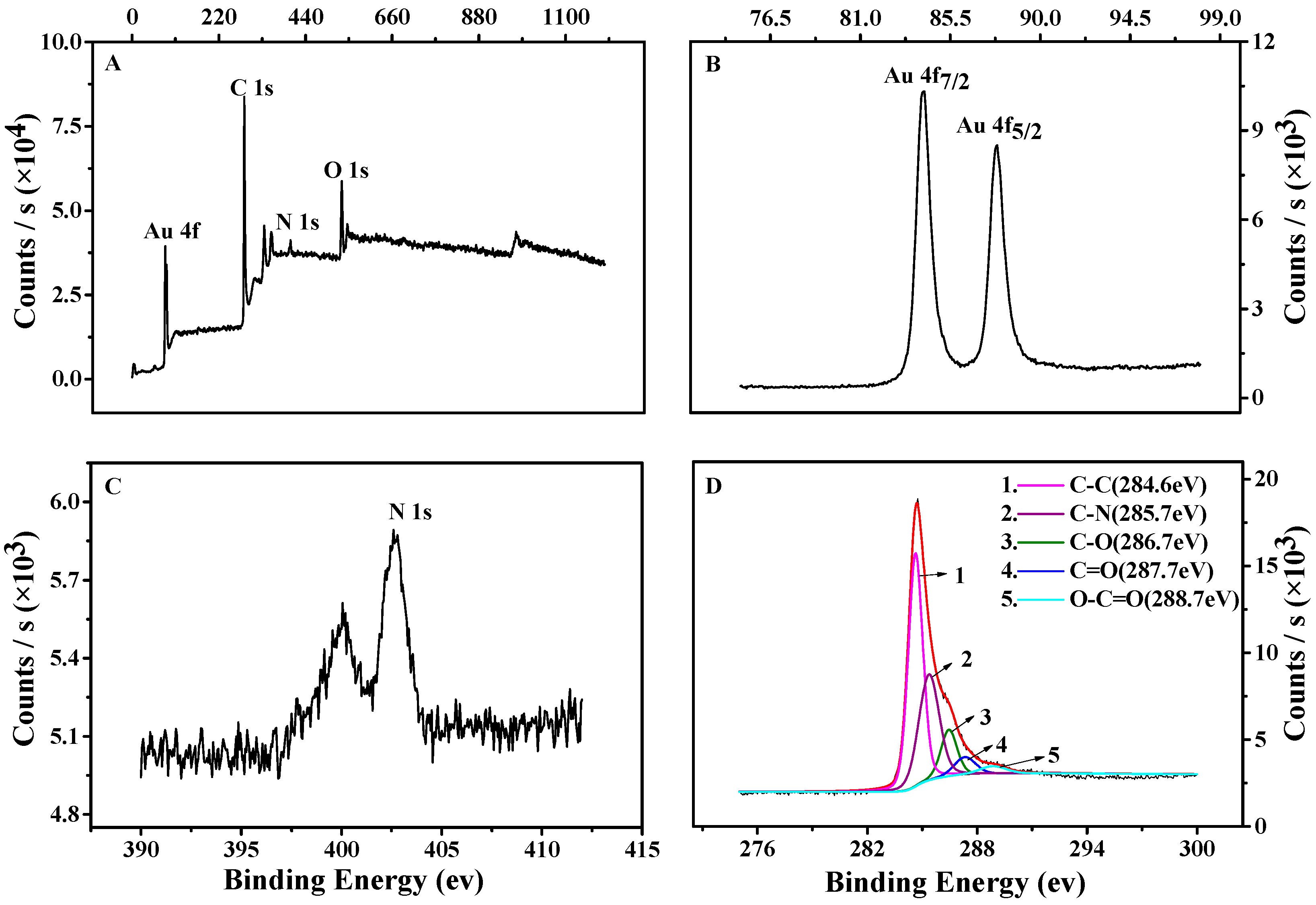
3.2. Characterization of the TCPP–Gr/AuNPs
3.3. Characterization of the Aptasensor
3.4. Optimization of Experimental Conditions
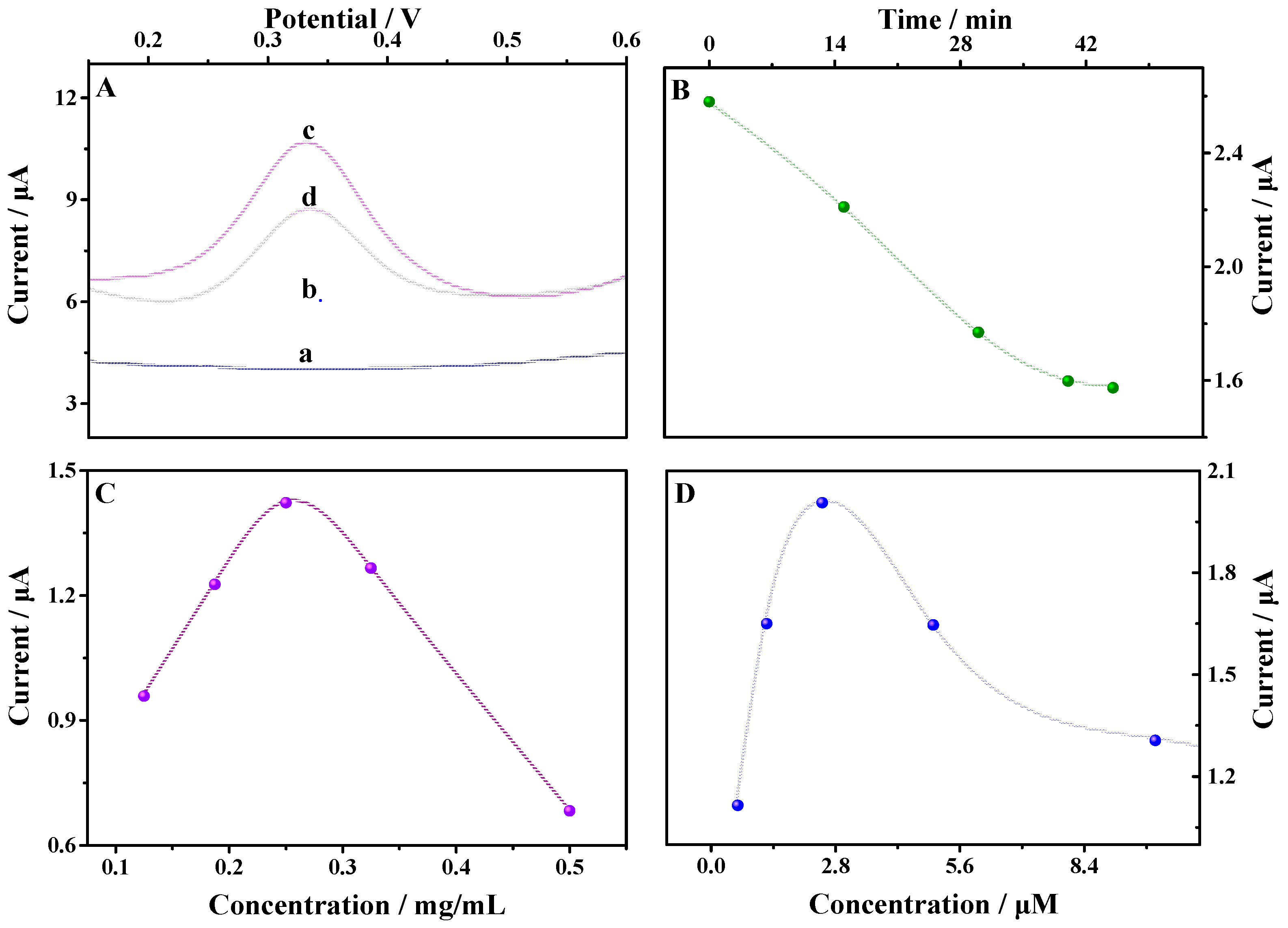
3.4.1. Influence of the Incubation Time
3.4.2. Influence of the TCPP–Gr/AuNPs Concentration
3.4.3. Influence of the MbBA Concentration
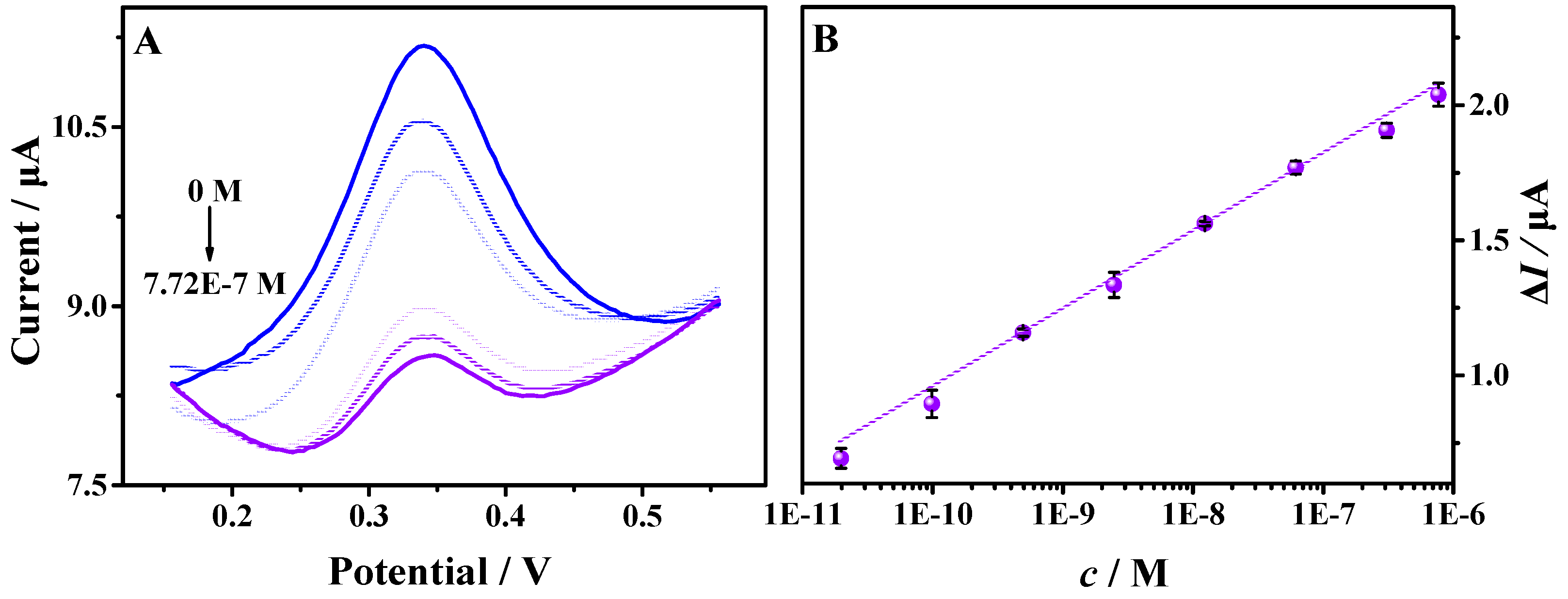
3.5. Analytical Application of the Aptasensor
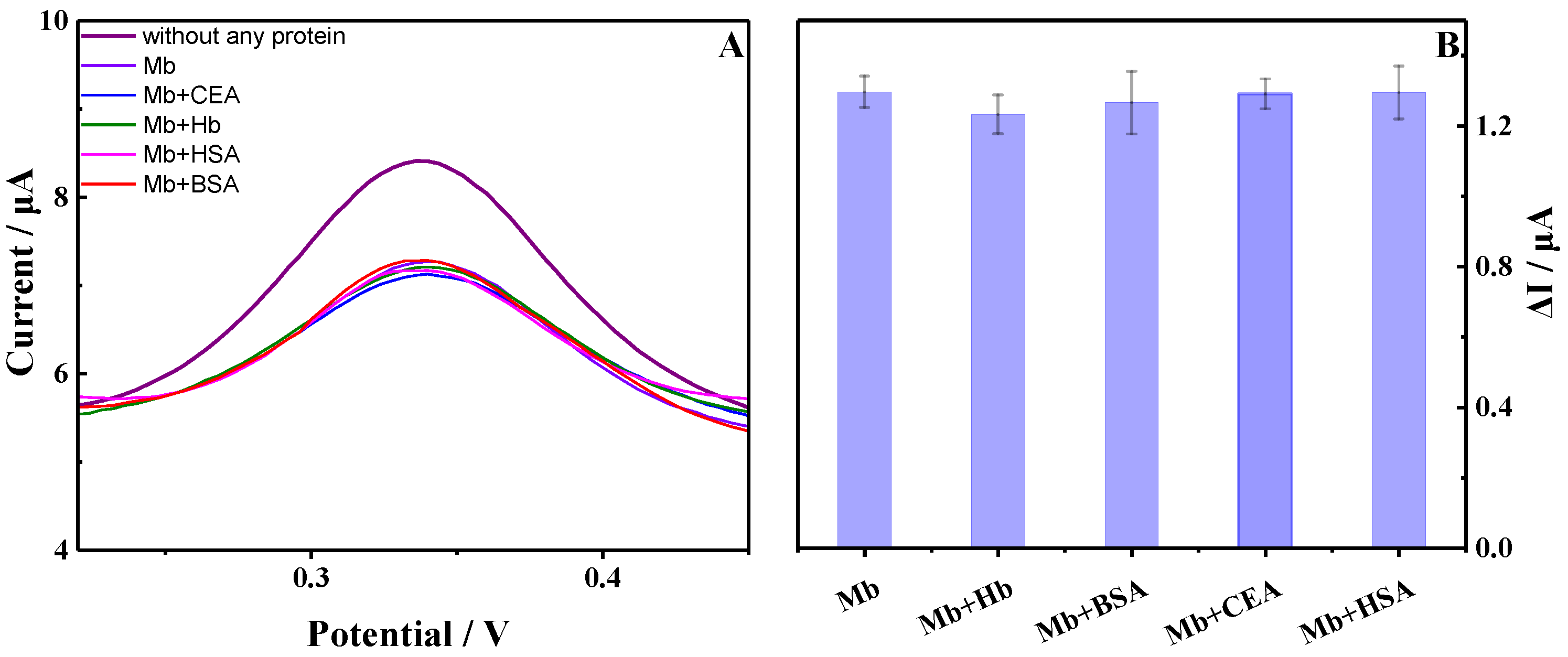
3.6. The Reproducibility, Stability and Specificity of the Aptasensor
3.7. Real Sample Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AMI | Acute Myocardial Infarction |
| AuNPs | Gold Nanoparticles |
| BSA | Bovine Serum Albumin |
| CEA | Carcinoembryonic Antigen |
| DPV | Differential Pulse Voltammetry |
| FT-IR | Fourier Infrared Spectrometer |
| GCE | Glassy Carbon Electrode |
| GO | Graphene Oxide |
| HAS | Human Serum Albumin |
| HAuCl4 | Chloroauric Acid |
| HB | Hemoglobin |
| LOD | Limit of Detection |
| MbBA | Myoglobin Binding Aptamer |
| MCH | 6-mercapto-l-hexanol |
| PBS | Phosphate Buffered Solution |
| PDDA | Poly Dimethyl Diallyl Ammonium Chloride |
| RSD | Relative Standard Deviation |
| SCE | Saturated Calomel Electrode |
| TCEP | Tris (2-carboxyethyl) Phosphine Hydrochloride |
| TCPP | Meso-tetra (4-carboxyphenyl) Porphyrin |
| TCPP-Gr | Meso-tetra (4-carboxyphenyl) Porphyrin Functionalized Graphene Composite |
| TEM | Transmission Electron Microscopy |
| UV-vis | Ultraviolet Visible Absorption Spectrum |
| XPS | X-ray Photoelectron Spectroscopy |
References
- Zhu, J.M.; Zou, N.L.; Mao, H.J.; Wang, P.; Zhu, D.N.; Ji, H.Y.; Cong, H.; Sun, C.J.; Wang, H.M.; Zhang, F.; et al. Evaluation of a modified lateral flow immunoassay for detection of high-sensitivity cardiac troponin I and myoglobin. Biosens. Bioelectron. 2013, 42, 522–525. [Google Scholar] [CrossRef] [PubMed]
- Stillman, A.E.; Oudkerk, M.; Bluemke, D.; Bremerich, J.; Esteves, F.P.; Garcia, E.V.; Gutberlet, M.; Hundley, W.G.; Jerosch-Herold, M.; Kuijpers, D.; et al. Assessment of acute myocardial infarction: Current status and recommendations from the north american society for cardiovascular imaging and the european society of cardiac radiology. Int. J. Cardiovas. Imaging 2011, 27, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.B.; Laios, I.; Green, S.; Gornet, T.G.; Wong, S.S.; Parmley, L.; Tonnesen, A.S.; Plaisier, B.; Orlando, R. Immunoassays for serum and urine myoglobin-myoglobin clearance assessed as a risk factor for acute-renal-failure. Clin. Chem. 1994, 40, 796–802. [Google Scholar] [PubMed]
- Naveena, B.M.; Faustman, C.; Tatiyaborworntham, N.; Yin, S.; Ramanathan, R.; Mancini, R.A. Detection of 4-hydroxy-2-nonenal adducts of turkey and chicken myoglobins using mass spectrometry. Food Chem. 2010, 122, 836–840. [Google Scholar] [CrossRef]
- Gnedenko, O.V.; Mezentsev, Y.V.; Molnar, A.A.; Lisitsa, A.V.; Ivanov, A.S.; Archakov, A.I. Highly sensitive detection of human cardiac myoglobin using a reverse sandwich immunoassay with a gold nanoparticle-enhanced surface plasmon resonance biosensor. Anal. Chim. Acta 2013, 759, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Xi, J.W.; Hou, F.; Han, L.; Li, G.J.; Gong, S.X.; Chen, C.X.; Sun, W. Application of three-dimensional reduced graphene oxide-gold composite modified electrode for direct electrochemistry and electrocatalysis of myoglobin. Mater. Sci. Eng. C Mater. 2016, 58, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, X.H.; Yang, X.H.; Liu, F.; Wang, K.M. Visual detection of myoglobin via g-quadruplex dnazyme functionalized gold nanoparticles-based colorimetric biosensor. Sens. Actuator B Chem. 2015, 212, 440–445. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, F.; Yang, X.; Wang, K.; Wang, H.; Deng, X. Sensitive point-of-care monitoring of cardiac biomarker myoglobin using aptamer and ubiquitous personal glucose meter. Biosens. Bioelectron. 2015, 64, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, D.W.; LeBlanc, G.; Meschievitz, M.E.; Cliffel, D.E. Electrochemical sensors and biosensors. Anal. Chem. 2012, 84, 685–707. [Google Scholar] [CrossRef] [PubMed]
- Ronkainen, N.J.; Halsall, H.B.; Heineman, W.R. Electrochemical biosensors. Chem. Soc. Rev. 2010, 39, 1747–1763. [Google Scholar] [CrossRef] [PubMed]
- Moreira, F.T.C.; Dutra, R.A.F.; Noronha, J.P.C.; Sales, M.G.F. Electrochemical biosensor based on biomimetic material for myoglobin detection. Electrochim. Acta 2013, 107, 481–487. [Google Scholar] [CrossRef]
- Lee, H.Y.; Choi, J.S.; Guruprasath, P.; Lee, B.H.; Cho, Y.W. An electrochemical biosensor based on a myoglobin-specific binding peptide for early diagnosis of acute myocardial infarction. Anal. Sci. 2015, 31, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Leca-Bouvier, B.; Blum, L.J. Biosensors for protein detection: A review. Anal. Lett. 2005, 38, 1491–1517. [Google Scholar] [CrossRef]
- Vestergaard, M.; Kerman, K.; Tamiya, E. An overview of label-free electrochemical protein sensors. Sensors 2007, 7, 3442–3458. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, Y.; Yang, P.; Liu, B. Label-free aptasensor based on electrodeposition of gold nanoparticles on graphene and its application in the quantification of adenosine triphosphate. Electrochim. Acta 2015, 172, 88–93. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Service, R.F. Materials science carbon sheets an atom thick give rise to graphene dreams. Science 2009, 324, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zeng, G.M.; Tang, L.; Chen, J.; Zhu, Y.; He, X.X.; He, Y. Electrochemical sensor based on electrodeposited graphene-Au modified electrode and nanoAu carrier amplified signal strategy for attomolar mercury detection. Anal. Chem. 2015, 87, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Patil, A.J.; Vickery, J.L.; Scott, T.B.; Mann, S. Aqueous stabilization and self-assembly of graphene sheets into layered bio-nanocomposites using DNA. Adv. Mater. 2009, 21, 3159. [Google Scholar] [CrossRef]
- Akca, S.; Foroughi, A.; Frochtzwajg, D.; Postma, H.W.C. Competing interactions in DNA assembly on graphene. PLoS ONE 2011, 6, e18442. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bai, X.; Wang, X.; Shiu, K.-K.; Zhu, Y.; Jiang, H. Highly sensitive graphene–Pt nanocomposites amperometric biosensor and its application in living cell H2O2 detection. Anal. Chem. 2014, 86, 9459–9465. [Google Scholar] [CrossRef] [PubMed]
- Jans, H.; Huo, Q. Gold nanoparticle-enabled biological and chemical detection and analysis. Chem. Soc. Rev. 2012, 41, 2849–2866. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.-J.; Liu, Y.-J.; Zhang, J.-Z.; Cao, J.-T.; Liu, Y.-M. Aptamer/Au nanoparticles/cobalt sulfide nanosheets biosensor for 17β-estradiol detection using a guanine-rich complementary DNA sequence for signal amplification. Biosens. Bioelectron. 2015, 67, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wang, Y.; Wei, H.; Dong, S. Amplified electrochemical aptasensor taking aunps based sandwich sensing platform as a model. Biosens. Bioelectron. 2008, 23, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Wang, F.; Yang, D.; Gao, Y.; Li, H. Electrochemical sensing platform for l-cysh based on nearly uniform Au nanoparticles decorated graphene nanosheets. Mater. Sci. Eng. C 2014, 38, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Vidotti, M.; Carvalhal, R.F.; Mendes, R.K.; Ferreira, D.C.M.; Kubota, L.T. Biosensors based on gold nanostructures. J. Braz. Chem. Soc. 2011, 22, 3–20. [Google Scholar] [CrossRef]
- Yang, F.; Wang, P.L.; Wang, R.G.; Zhou, Y.; Su, X.O.; He, Y.J.; Shi, L.; Yao, D.S. Label free electrochemical aptasensor for ultrasensitive detection of ractopamine. Biosens. Bioelectron. 2016, 77, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.-P.; Zhou, Y.; Wang, J.; Zhu, J.-J. Electrogenerated chemiluminescence resonance energy transfer between Ru(bpy)32+ electrogenerated chemiluminescence and gold nanoparticles/graphene oxide nanocomposites with graphene oxide as coreactant and its sensing application. Anal. Chem. 2016, 88, 5469–5475. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Yao, Y.; Li, Z.Z.; Zhang, X.H.; Chen, J.H. A new amplified impedimetric aptasensor based on the electron transfer ability of Au nanoparticles and their affinity with aptamer. J. Electroanal. Chem. 2015, 757, 243–249. [Google Scholar] [CrossRef]
- Wu, L.; Feng, L.Y.; Ren, J.S.; Qu, X.G. Electrochemical detection of dopamine using porphyrin-functionalized graphene. Biosens. Bioelectron. 2012, 34, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.X.; Lei, Y.; Li, C.M. Porphyrin functionalized graphene for sensitive electrochemical detection of ultratrace explosives. Electroanalysis 2011, 23, 885–893. [Google Scholar] [CrossRef]
- Gerasimov, J.Y.; Schaefer, C.S.; Yang, W.; Grout, R.L.; Lai, R.Y. Development of an electrochemical insulin sensor based on the insulin-linked polymorphic region. Biosens. Bioelectron. 2013, 42, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Radi, A.-E.; Acero Sánchez, J.L.; Baldrich, E.; O’Sullivan, C.K. Reagentless, reusable, ultrasensitive electrochemical molecular beacon aptasensor. J. Am. Chem. Soc. 2006, 128, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Lubin, A.A.; Heeger, A.J.; Plaxco, K.W. Label-free electronic detection of thrombin in blood serum by using an aptamer-based sensor. Angew. Chem. Int. Ed. 2005, 44, 5456–5459. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, L.; Yang, X.; Wang, K.; Chen, N.; Zhou, C.; Luo, B.; Du, S. Evaluation of medicine effects on the interaction of myoglobin and its aptamer or antibody using atomic force microscopy. Anal. Chem. 2015, 87, 2242–2248. [Google Scholar] [CrossRef] [PubMed]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339–1339. [Google Scholar] [CrossRef]
- Huang, K.-J.; Liu, Y.-J.; Shi, G.-W.; Yang, X.-R.; Liu, Y.-M. Label-free aptamer sensor for 17β-estradiol based on vanadium disulfide nanoflowers and Au nanoparticles. Sens. Actuators B Chem. 2014, 201, 579–585. [Google Scholar] [CrossRef]
- Ang, P.K.; Wang, S.; Bao, Q.; Thong, J.T.L.; Loh, K.P. High-throughput synthesis of graphene by intercalation–exfoliation of graphite oxide and study of ionic screening in graphene transistor. ACS Nano 2009, 3, 3587–3594. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.J.; Deng, L.; Li, J.; Guo, S.J.; Wang, E.K.; Dong, S.J. Hemin-graphene hybrid nanosheets with intrinsic peroxidase-like activity for label-free colorimetric detection of single-nucleotide polymorphism. ACS Nano 2011, 5, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Guo, Y.; Dong, C. Ultrasensitive and label-free electrochemical DNA biosensor based on water-soluble electroactive dye azophloxine-functionalized graphene nanosheets. Electrochim. Acta 2013, 113, 69–76. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Zhou, Z.X.; Zhai, Y.L.; Zhang, L.L.; Hong, W.; Zhang, Z.Q.; Dong, S.J. Label-free aptamer biosensor for thrombin detection based on functionalized graphene nanocomposites. Talanta 2015, 141, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Shorie, M.; Ganguli, A.K.; Sabherwal, P. Graphene-CNT nanohybrid aptasensor for label free detection of cardiac biomarker myoglobin. Biosens. Bioelectron. 2015, 72, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Shorie, M.; Kumar, V.; Sabherwal, P.; Ganguli, A.K. Carbon quantum dots-mediated direct fluorescence assay for the detection of cardiac marker myoglobin. Curr. Sci. 2015, 108, 1595–1596. [Google Scholar]


| Platform | Detection Method | Linear Range (nm) | Detection Limit (nm) | Specificity | Reference |
|---|---|---|---|---|---|
| 3D RGO–Au/CILE | CV | 200–36,000 | 60 | No | [6] |
| Hemin/G-quadruplet/AuNPs | Colorimetric | 0–1000 | 2.5 | Antibody | [7] |
| Au/DSP/Peptide | CV | 1.1–105 | 58 | Myoglobin specific binding peptide | [12] |
| rGO/CNT | CV | 0.058–235 | 0.020 | Aptamer | [42] |
| POC | PGM | 0–200 | 0.050 | aptamer | [8] |
| CQDs | Fluorescence | 0.059–5.9 | 0.059 | Anti-Mb-aptamer | [43] |
| TCPP–Gr/AuNPs | DPV | 0.020–770 | 0.0067 | Aptamer | This work |
| Samples | Added (nM) | Found (nM) | Recovery/% |
|---|---|---|---|
| 1 | 0.152 | 0.159 | 104.6 |
| 2 | 1.56 | 1.51 | 96.8 |
| 3 | 9.88 | 10.5 | 106.3 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, G.; Liu, Z.; Wang, L.; Guo, Y. Electrochemical Aptasensor for Myoglobin-Specific Recognition Based on Porphyrin Functionalized Graphene-Conjugated Gold Nanocomposites. Sensors 2016, 16, 1803. https://doi.org/10.3390/s16111803
Zhang G, Liu Z, Wang L, Guo Y. Electrochemical Aptasensor for Myoglobin-Specific Recognition Based on Porphyrin Functionalized Graphene-Conjugated Gold Nanocomposites. Sensors. 2016; 16(11):1803. https://doi.org/10.3390/s16111803
Chicago/Turabian StyleZhang, Guojuan, Zhiguang Liu, Li Wang, and Yujing Guo. 2016. "Electrochemical Aptasensor for Myoglobin-Specific Recognition Based on Porphyrin Functionalized Graphene-Conjugated Gold Nanocomposites" Sensors 16, no. 11: 1803. https://doi.org/10.3390/s16111803
APA StyleZhang, G., Liu, Z., Wang, L., & Guo, Y. (2016). Electrochemical Aptasensor for Myoglobin-Specific Recognition Based on Porphyrin Functionalized Graphene-Conjugated Gold Nanocomposites. Sensors, 16(11), 1803. https://doi.org/10.3390/s16111803






