A Nanoporous Alumina Membrane Based Electrochemical Biosensor for Histamine Determination with Biofunctionalized Magnetic Nanoparticles Concentration and Signal Amplification
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
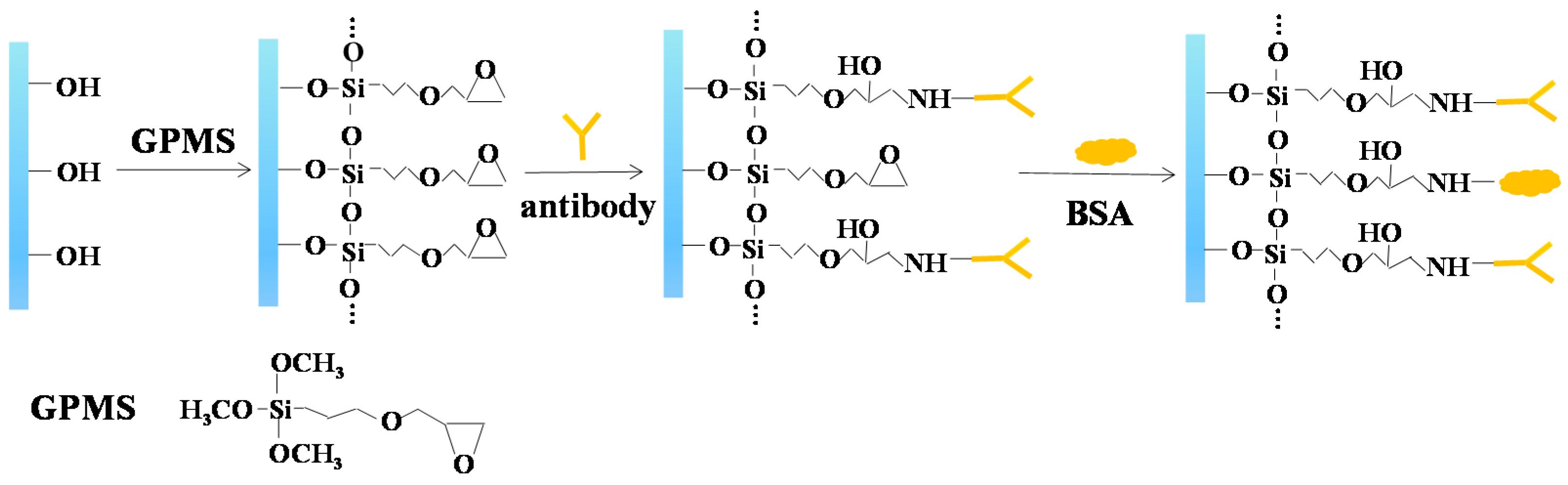
2.2. Biofunctionalization of Nanoporous Alumina Membrane
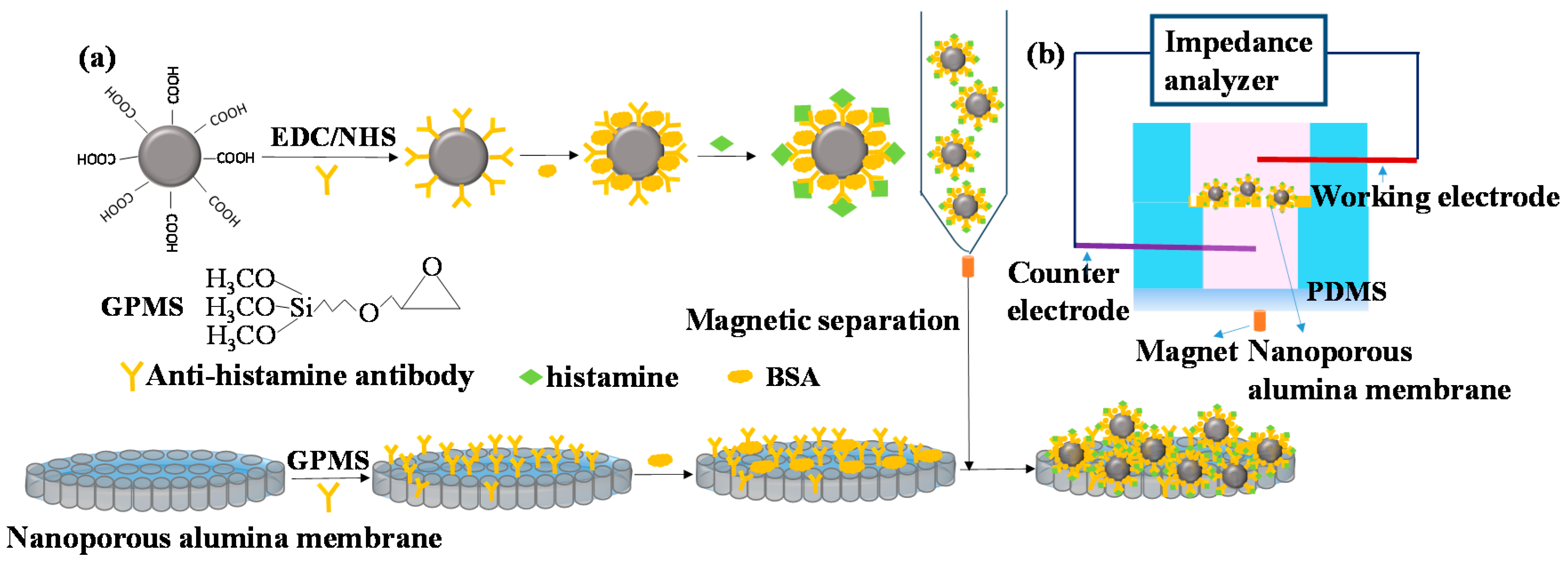
2.3. Magnetic Nanoparticles Functionalization
2.4. Histamine Extraction from Saury Fish
2.5. Histamine Concentration to Biofunctionalized Nanoporous Alumina Membrane
2.6. Characterization
2.7. Electrochemical Impedance Spectroscopy Measurement
3. Results and Discussion
3.1. Mechanism of Histamine Detection by Electrochemical Biosensor
3.2. Characterization of Magnetic Nanoparticles and Nanoporous Alumina Membranes
3.3. Antibody Immobilization on Nanoporous Membrane
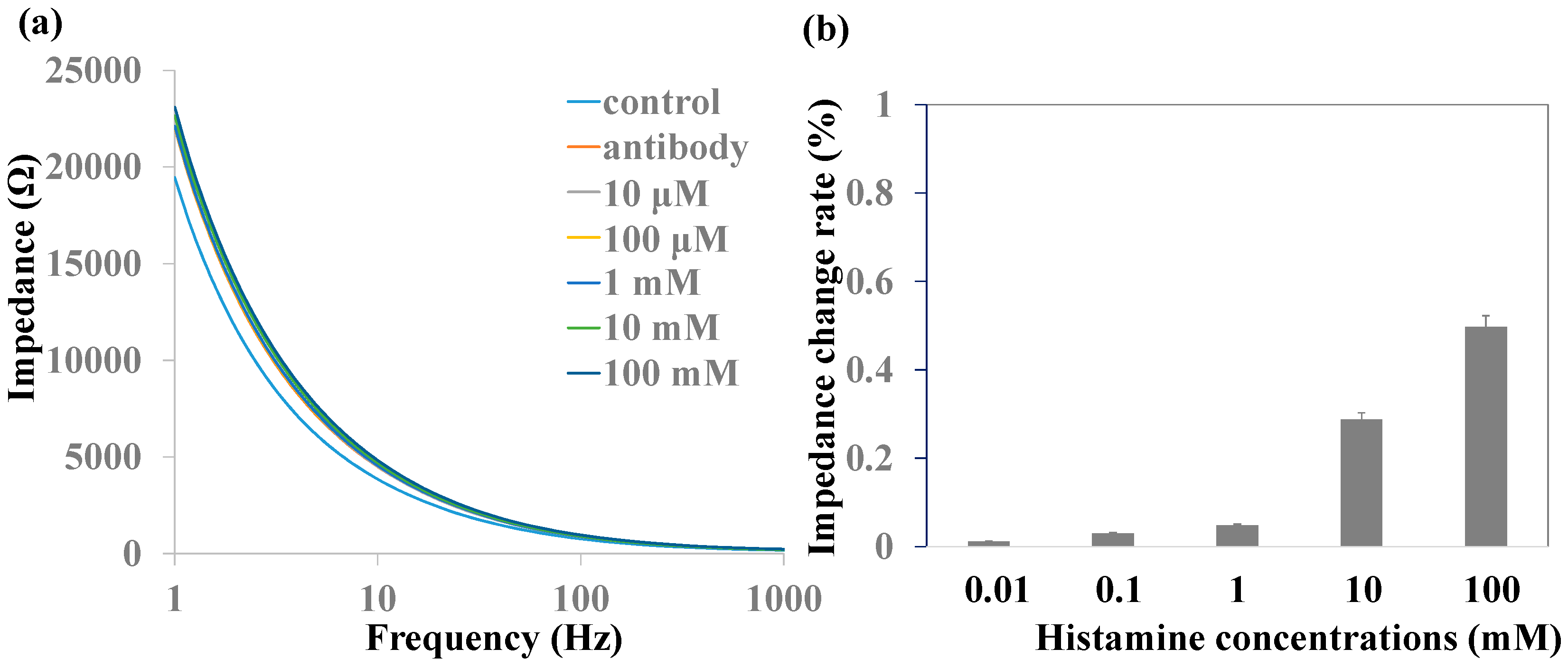
3.4. Histamine Detection without MNPs
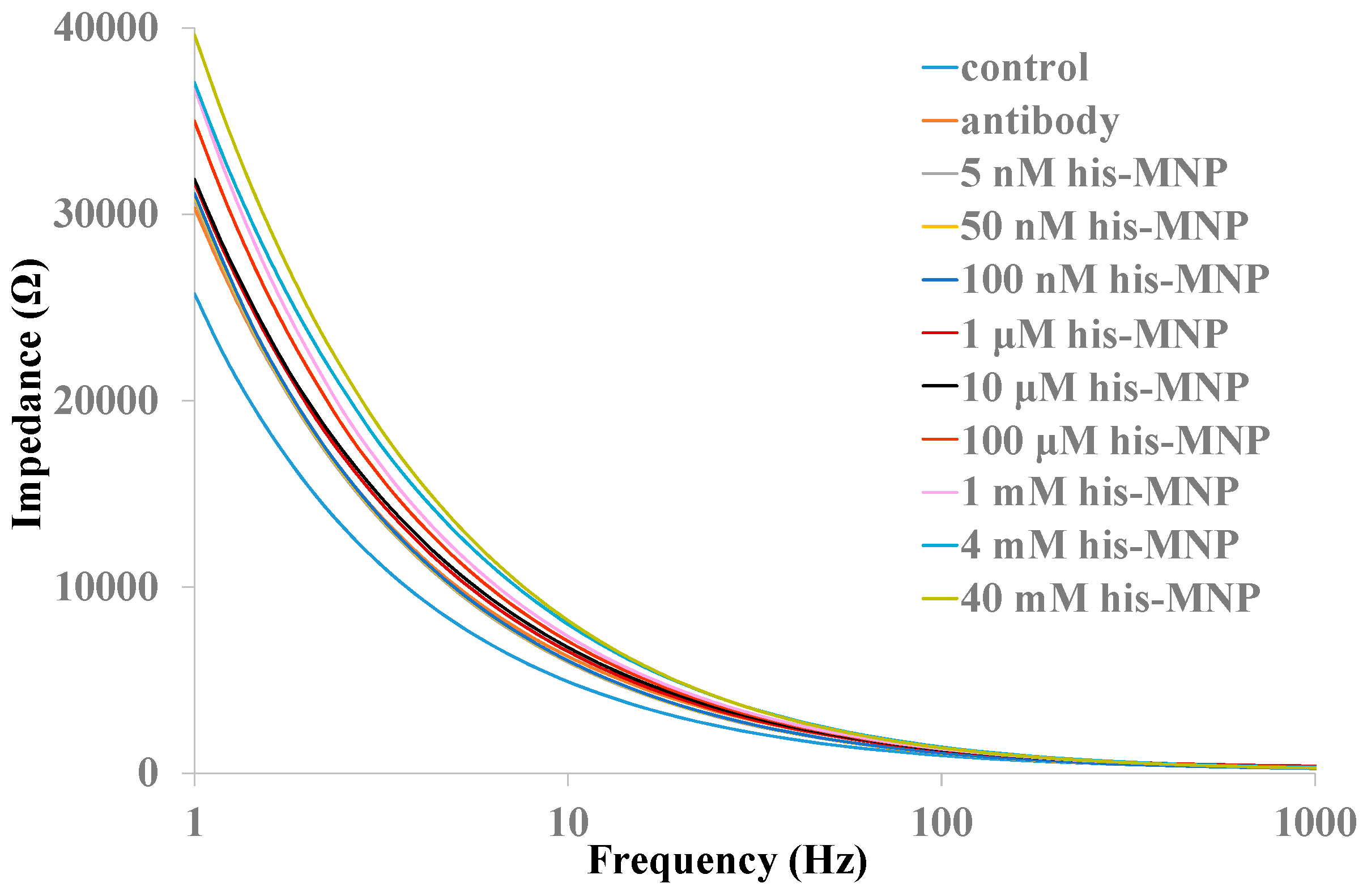
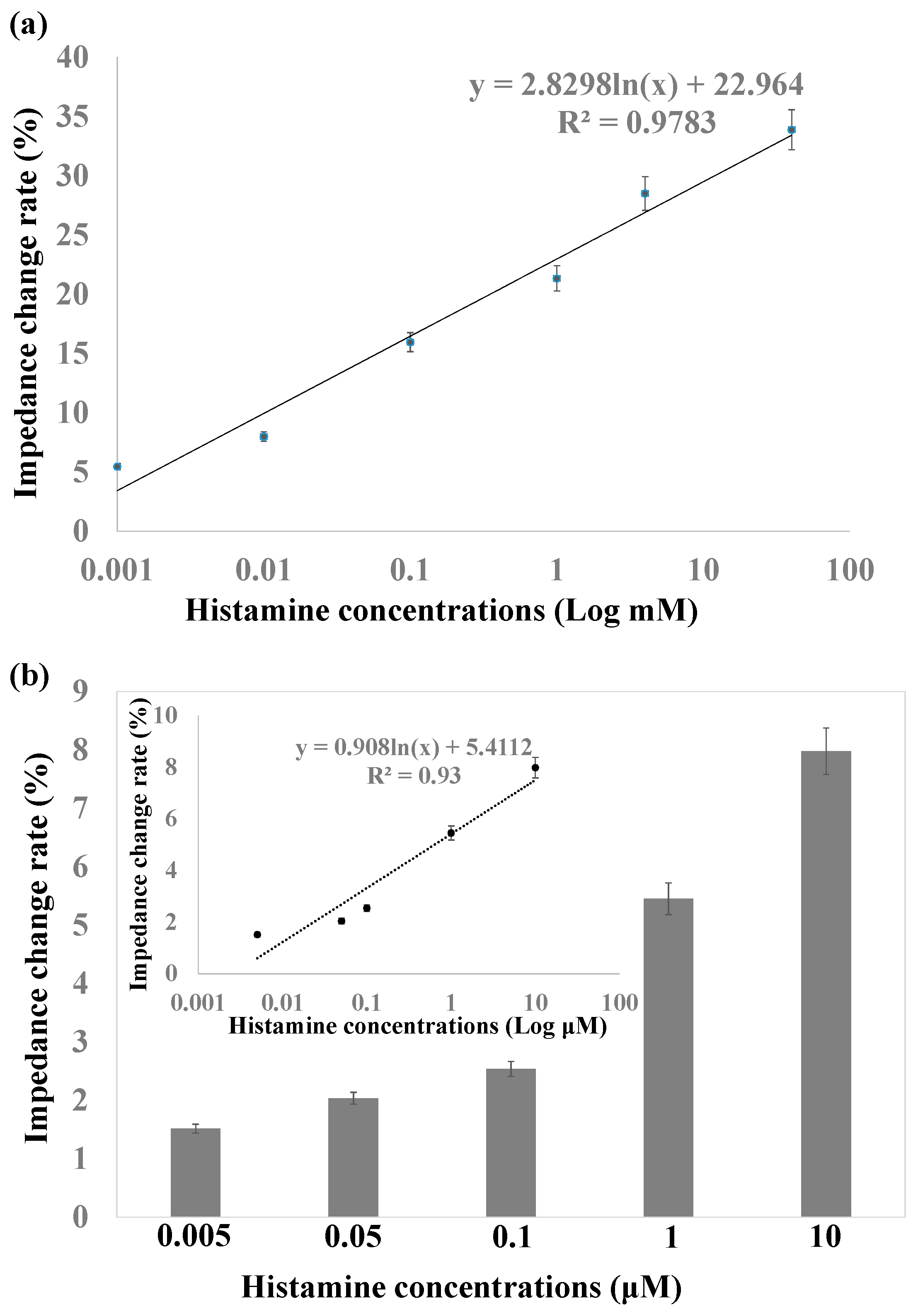
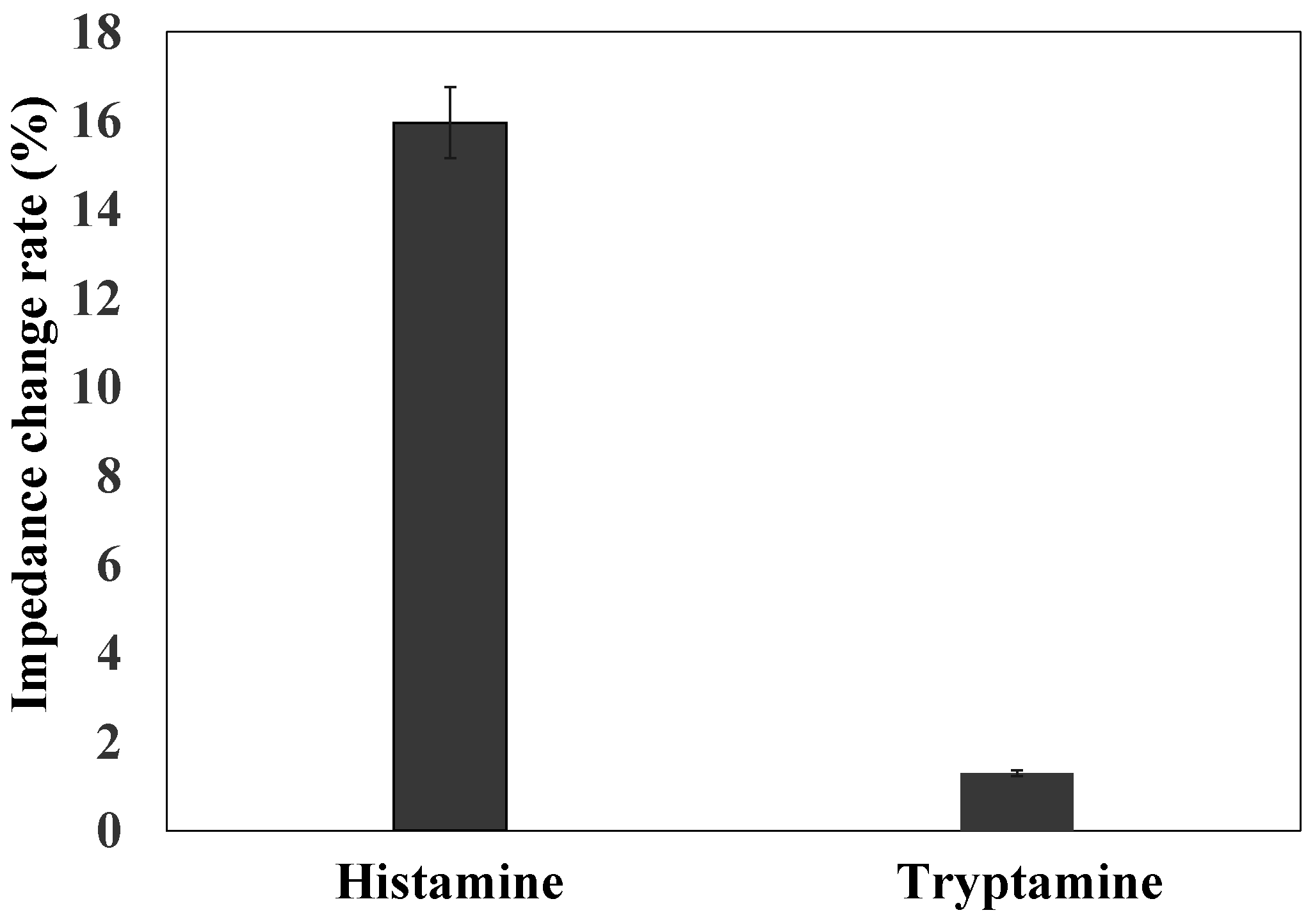
3.5. The Effect of MNPs on Histamine Detection
3.6. Histamine Determination in Saury
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Al Bulushi, I.; Poole, S.; Deeth, H.C.; Dykes, G.A. Biogenic amines in fish: Roles in intoxication, spoilage, and nitrosamine formation—A review. Crit. Rev. Food Sci. Nutr. 2009, 49, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, V. Biosensors in fish production and quality control. Biosens. Bioelectron. 2001, 17, 147–157. [Google Scholar] [CrossRef]
- Peng, J.; Fang, K.; Xie, D.; Ding, B.; Yin, J.; Cui, X.; Zhang, Y.; Liu, J. Development of an automated on-line-pre-column derivatization procedure for sensitive determination of histamine in food with high-performance liquid chromatography-fluorescence detection. J. Chromatogr. A 2008, 1209, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Ausländer, D.; Eggerschwiler, B.; Kemmer, C.; Geering, B.; Ausländer, S.; Fussenegger, M. A designer cell-based histamine-specific human allergy profiler. Nat. Commun. 2014, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lehane, L.; Olley, J. Histamine fish poisoning revisited. Int. J. Food Microbiol. 2000, 58, 1–37. [Google Scholar] [CrossRef]
- Ching, M.K.; Fatimah, A.B.; Abu, B.S.; Lee, Y.H.; Rahman, W.; Low, S.B. An amperometric biosensor for the rapid assessment of histamine level in tiger prawn (Penaeus monodon) spoilage. Food Chem. 2007, 105, 1636–1641. [Google Scholar]
- Degefua, H.; Amareb, M.; Tessemaa, M.; Admassiea, S. Lignin modified glassy carbon electrode for the electrochemical determination of histamine in human urine and wine samples. Electrochim. Acta 2014, 121, 307–314. [Google Scholar] [CrossRef]
- Jain, A.; Gupta, M.; Verma, K.K. Salting-out assisted liquid–liquid extraction for the determination of biogenic amines in fruit juices and alcoholic beverages after derivatization with 1-naphthylisothiocyanate and high performance liquid chromatography. J. Chromatogr. A 2015, 1422, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.; Sato, M.; Han, Y.; Tan, Z.; Yamaguchi, T.; Nakano, T. A simple and rapid method for histamine analysis in fish and fishery products by TLC determination. Food Control 2011, 22, 1154–1157. [Google Scholar] [CrossRef]
- Vitali, L.; Valese, A.C.; Azevedo, M.S.; Gonzaga, L.V.; Costa, A.C.O.; Piovezan, M.; Vistuba, J.P.; Micke, G.A. Development of a fast and selective separation method to determine histamine in tuna fish samples using capillary zone electrophoresis. Talanta 2013, 106, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Köse, S.; Kaklikkaya, N.; Koral, S.; Tufan, B.; Buruk, K.C.; Aydin, F. Commercial test kit and the determination of histamine in traditional (ethnic) fish products-evaluation against an EU accepted HPLC method. Food Chem. 2011, 125, 1490–1497. [Google Scholar] [CrossRef]
- May, K.M.L.; Wang, Y.; Bachas, L.G.; Anderson, K.W. Development of a whole-cell-based biosensor for detecting histamine as a model toxin. Anal. Chem. 2004, 76, 4156–4161. [Google Scholar] [CrossRef] [PubMed]
- Cash, K.J.; Clark, H.A. Phosphorescent nanosensors for in vivo tracking of histamine levels. Anal. Chem. 2013, 85, 6312–6318. [Google Scholar] [CrossRef] [PubMed]
- An, D.; Chen, Z.; Zheng, J.; Chen, S.; Wang, L.; Su, W. Polyoxomatelate functionalized tris(2,2-bipyridyl)dichlororuthenium(II) as the probe for electrochemiluminescence sensing of histamine. Food Chem. 2016, 194, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Yang, J.Y.; Xiao, Z.L.; Zeng, D.P.; Li, Y.J.; Shen, Y.D.; Sun, Y.M.; Lei, H.T.; Wang, H.; Xu, Z.L. A sensitivity-enhanced heterologous immunochromatographic assay based on a monoclonal antibody for the rapid detection of histamine in saury samples. RSC Adv. 2015, 5, 78833–78840. [Google Scholar] [CrossRef]
- Gustiananda, M.; Andreoni, A.; Tabares, L.C.; Tepper, A.W.J.W.; Fortunato, L.; Aartsma, T.J.; Canters, G.W. Sensitive detection of histamine using fluorescently labeled oxido-reductases. Biosens. Bioelectron. 2012, 31, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Pietrzyk, A.; Suriyanarayanan, S.; Kutner, W.; Chitta, R.; D’Souza, F. Selective Histamine Piezoelectric Chemosensor Using a Recognition Film of the Molecularly Imprinted Polymer of Bis(bithiophene) Derivatives. Anal. Chem. 2009, 81, 2633–2643. [Google Scholar] [CrossRef] [PubMed]
- Basozabal, I.; Guerreiro, A.; Gomez-Caballero, A.; Goicolea, M.A.; Barrio, R.J. Direct potentiometric quantification of histamine using solid-phase imprinted nanoparticles as recognition elements. Biosens. Bioelectron. 2014, 58, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Henao-Escobar, W.; del Torno-de Román, L.; Domínguez-Renedo, O.; Alonso-Lomillo, M.A. Dual enzymatic biosensor for simultaneous amperometric determination of histamine and putrescine. Food Chem. 2016, 190, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Leonardo, S.; Campàs, M. Electrochemical enzyme sensor arrays for the detection of the biogenic amines histamine, putrescine and cadaverine using magnetic beads as immobilisation supports. Microchim. Acta 2016, 183, 1881–1890. [Google Scholar] [CrossRef]
- Delle, L.E.; Huck, C.; Bäcker, M.; Müller, F.; Grandthyll, S.; Jacobs, K.; Lilischkis, R.; Vu, X.T.; Schöning, M.J.; Wagner, P.; et al. Impedimetric immunosensor for the detection of histamine based on reduced graphene oxide. Phys. Status Solidi A 2015, 212, 1327–1334. [Google Scholar] [CrossRef]
- Peeters, M.; Troost, F.J.; Mingels, R.H.G.; Welsch, T.; van Grinsven, B.; Vranken, T.; Ingebrandt, S.; Thoelen, R.; Cleij, T.J.; Wagner, P. Impedimetric Detection of Histamine in Bowel Fluids Using Synthetic Receptors with pH-Optimized Binding Characteristics. Anal. Chem. 2013, 85, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Ruan, C.; Yang, L.; Li, Y. Immunobiosensor chips for detection of Escherichia coli O157:H7 using electrochemical impedance spectroscopy. Anal. Chem. 2002, 74, 4814–4820. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, B.; Merkoç, A. Nanomaterials based biosensors for food analysis applications. Trends Food Sci. Technol. 2011, 22, 625–639. [Google Scholar] [CrossRef]
- Wu, S.M.; Ye, W.W.; Yang, M.; Taghipoor, M.; Meissner, R.; Brugger, J.; Renaud, P. Impedance sensing of DNA immobilization and hybridization by microfabricated alumina nanopore membranes. Sens. Actuators B Chem. 2015, 216, 105–112. [Google Scholar] [CrossRef]
- Wang, L.J.; Liu, Q.J.; Hu, Z.Y.; Zhang, Y.F.; Wu, C.S.; Yang, M.; Wang, P. A novel electrochemical biosensor based on dynamic polymerase-extending hybridization for E. coli O157:H7 DNA detection. Talanta 2009, 78, 647–652. [Google Scholar] [CrossRef] [PubMed]
- De la Escosura-Muñiz, A.; Chunglok, W.; Surareungchai, W.; Merkoçi, A. Nanochannels for diagnostic of thrombin-related diseases in human blood. Biosens. Bioelectron. 2013, 40, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.; Leung, P.H.M.; Zhang, Y.; Liu, Z.B.; Xiao, L.D.; Ye, W.W.; Zhang, X.; Li, Y.; Yang, M. A PDMS microfluidic impedance immunosensor for E. coli O157:H7 and Staphylococcus aureus detection via antibody-immobilized nanoporous membrane. Sens. Actuators B Chem. 2011, 159, 328–335. [Google Scholar] [CrossRef]
- Joung, C.K.; Kim, H.N.; Lim, M.C.; Jeon, T.J.; Kim, H.Y.; Kim, Y.R. A nanoporous membrane-based impedimetric immunosensor for label-free detection of pathogenic bacteria in whole milk. Biosens. Bioelectron. 2013, 44, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Tsang, M.K.; Ye, W.W.; Wang, G.J.; Li, J.M.; Yang, M.; Hao, J.H. Ultrasensitive detection of Ebola virus oligonucleotide based on upconversion nanoprobe/nanoporous membrane system. ACS Nano 2016, 10, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.J.; Liu, Z.B.; Mak, A.F.T.; Yang, M. Nanoporous membrane-based cell chip for the study of anti-cancer drug effect of retinoic acid with impedance spectroscopy. Talanta 2009, 80, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.W.; Shi, J.Y.; Chan, C.Y.; Zhang, Y.; Yang, M. A nanoporous membrane based impedance sensing platform for DNA sensing with gold nanoparticle amplification. Sens. Actuators B Chem. 2014, 193, 877–882. [Google Scholar] [CrossRef]
- Chan, K.Y.; Ye, W.W.; Zhang, Y.; Xiao, L.D.; Leung, P.H.M.; Li, Y.; Yang, M. Ultrasensitive detection of E. coli O157:H7 with biofunctional magnetic bead concentration via nanoporous membrane based electrochemical immunosensor. Biosens. Bioelectron. 2013, 41, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Cernei, N.; Heger, Z.; Kopel, P.; Skladanka, J.; Zitka, O.; Adam, V.; Kizek, R. Isolation of biogenic amines using paramagnetic microparticles off-line coupled with ion exchange liquid chromatography. Chromatographia 2014, 77, 1451–1459. [Google Scholar] [CrossRef]
- Cernei, N.; Lackova, Z.; Guran, R.; Hynek, D.; Skladanka, J.; Horky, P.; Zitka, O.; Adam, V. Determination of histamine in silages using nanomaghemite core (γ-Fe2O3)—Titanium dioxide shell nanoparticles off-line coupled with ion exchange chromatography. Int. J. Environ. Res. Public Health 2016, 13, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Zhang, Y.; White, H.S.; Harrell, C.C.; Martin, C.R. Electrophoretic capture and detection of nanoparticles at the opening of a membrane pore using scanning electrochemical microscopy. Anal. Chem. 2004, 76, 6108–6115. [Google Scholar] [CrossRef] [PubMed]
- Schmid, G. Materials in nanoporous alumina. J. Mater. Chem. 2002, 12, 1231–1238. [Google Scholar] [CrossRef]
- Ye, W.W.; Guo, J.B.; Chen, S.; Yang, M. Nanoporous membrane based impedance sensors to detect the enzymatic activity of botulinum neurotoxin A. J. Mater. Chem. B 2013, 1, 6544–6550. [Google Scholar] [CrossRef]
- Xu, H.; Aguilar, Z.P.; Yang, L.; Kuang, M.; Duan, H.; Xiong, Y.; Wei, H.; Wang, A. Antibody conjugated magnetic iron oxide nanoparticles for cancer cell separation in fresh whole blood. Biomaterials 2011, 32, 9758–9765. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhong, Y.; Wei, Y.; Ortiz, N.; Chen, F.; Wang, G. Microscopic movement of slow-diffusing nanoparticles in cylindrical nanopores studied with three-dimensional tracking. Anal. Chem. 2016, 88, 5122–5130. [Google Scholar] [CrossRef] [PubMed]
- Leng, P.Q.; Zhao, F.L.; Yin, B.C.; Ye, B.C. A novel, colorimetric method for biogenic amine detection based on arylalkylamine N-acetyltransferase. Chem. Commun. 2015, 51, 8712–8714. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, W.; Xu, Y.; Zheng, L.; Zhang, Y.; Yang, M.; Sun, P. A Nanoporous Alumina Membrane Based Electrochemical Biosensor for Histamine Determination with Biofunctionalized Magnetic Nanoparticles Concentration and Signal Amplification. Sensors 2016, 16, 1767. https://doi.org/10.3390/s16101767
Ye W, Xu Y, Zheng L, Zhang Y, Yang M, Sun P. A Nanoporous Alumina Membrane Based Electrochemical Biosensor for Histamine Determination with Biofunctionalized Magnetic Nanoparticles Concentration and Signal Amplification. Sensors. 2016; 16(10):1767. https://doi.org/10.3390/s16101767
Chicago/Turabian StyleYe, Weiwei, Yifan Xu, Lihao Zheng, Yu Zhang, Mo Yang, and Peilong Sun. 2016. "A Nanoporous Alumina Membrane Based Electrochemical Biosensor for Histamine Determination with Biofunctionalized Magnetic Nanoparticles Concentration and Signal Amplification" Sensors 16, no. 10: 1767. https://doi.org/10.3390/s16101767
APA StyleYe, W., Xu, Y., Zheng, L., Zhang, Y., Yang, M., & Sun, P. (2016). A Nanoporous Alumina Membrane Based Electrochemical Biosensor for Histamine Determination with Biofunctionalized Magnetic Nanoparticles Concentration and Signal Amplification. Sensors, 16(10), 1767. https://doi.org/10.3390/s16101767





