Scalable Microfabrication Procedures for Adhesive-Integrated Flexible and Stretchable Electronic Sensors
Abstract
:1. Introduction
2. Experimental Section
Process Description

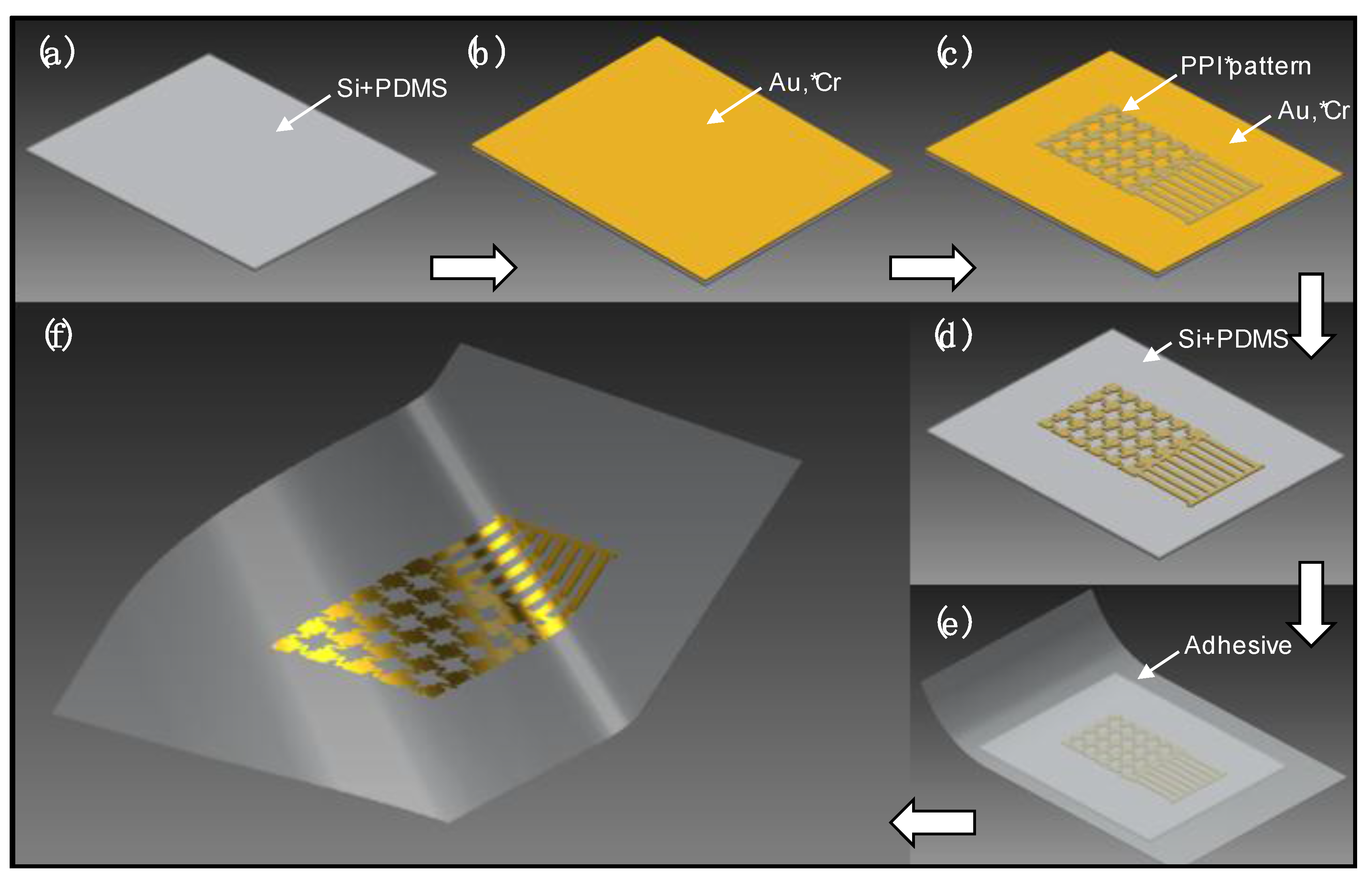
3. Results
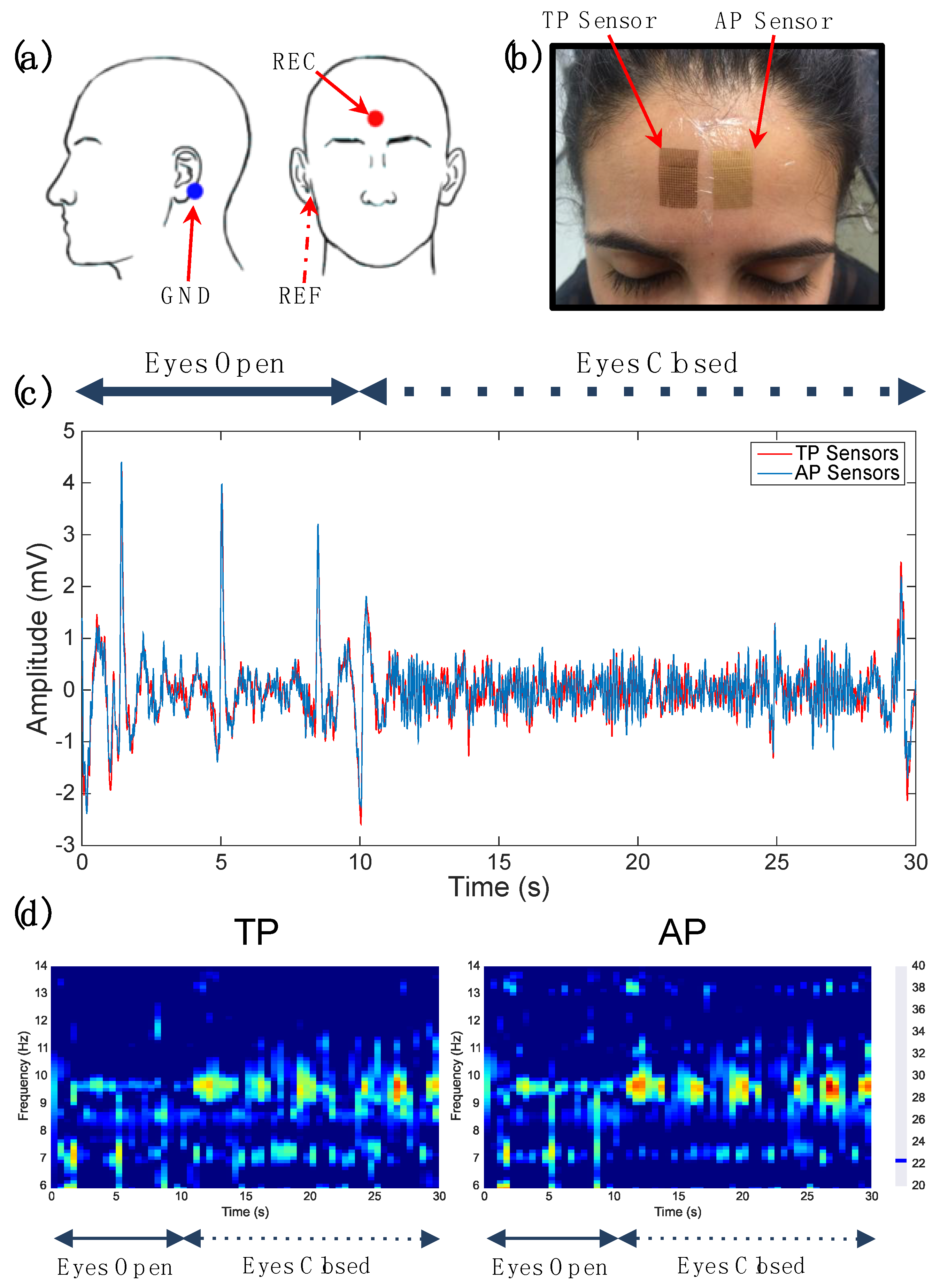
3.1. Qualitative Comparison of Fabrication Methods for TP and AP Sensors


3.2. Fabricating on PDMS Surfaces

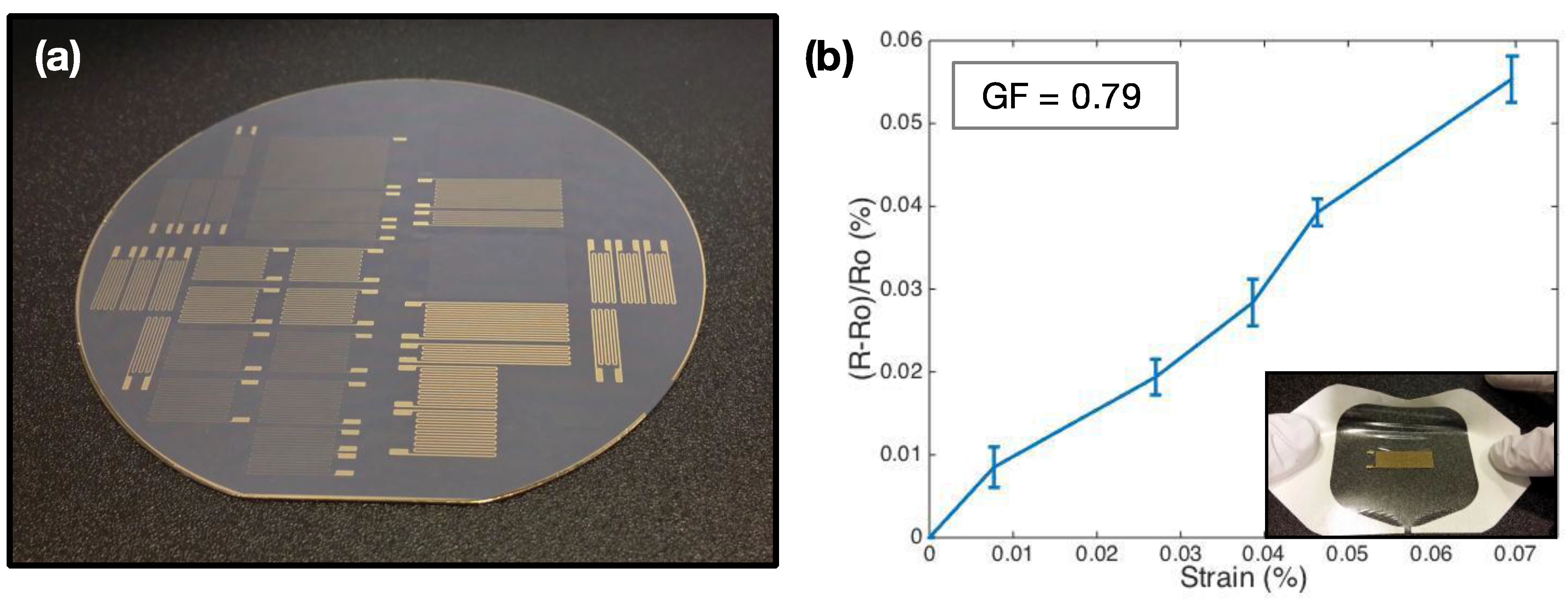
3.3. Quantitative Comparison of TP and AP Sensors
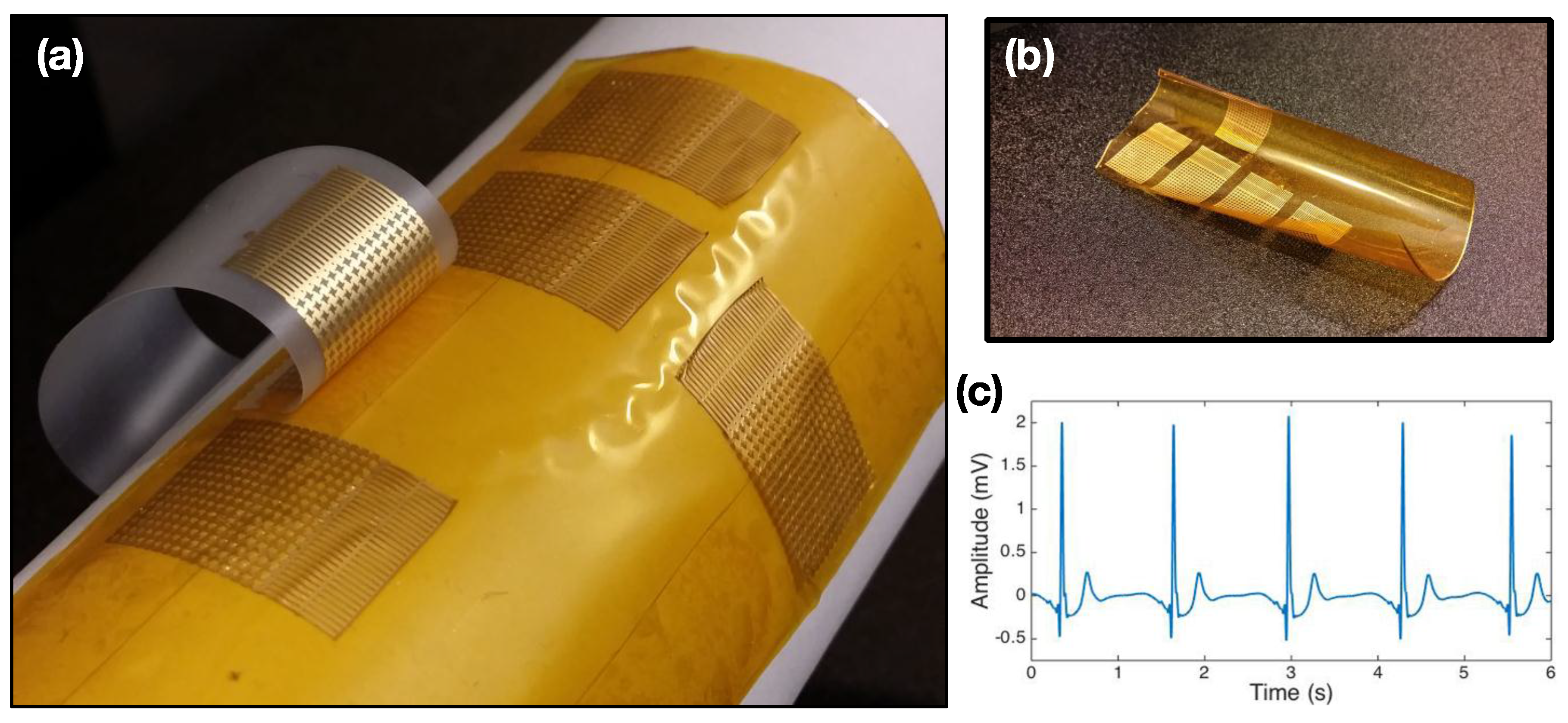
3.4. Non-Electrical AP Sensors
3.5. AP Sensors on Flexible Donor Substrate
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Søndergaard, R.R.; Hösel, M.; Krebs, F.C. Roll-to-roll fabrication of large area functional organic materials. J. Polym. Sci. Part B Polym. Phys. 2013, 51, 16–34. [Google Scholar] [CrossRef]
- Metters, J.P.; Kadara, R.O.; Banks, C.E. New directions in screen printed electroanalytical sensors: An overview of recent developments. Analyst 2011, 136, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- Harada, S.; Honda, W.; Arie, T.; Akita, S.; Takei, K. Fully printed, highly sensitive multifunctional artificial electronic whisker arrays integrated with strain and temperature sensors. ACS Nano 2014, 8, 3921–3927. [Google Scholar] [CrossRef] [PubMed]
- Windmiller, J.R.; Bandodkar, A.J.; Valdés-Ramírez, G.; Parkhomovsky, S.; Martinez, A.G.; Wang, J. Electrochemical sensing based on printable temporary transfer tattoos. Chem. Commun. 2012, 48, 6794–6796. [Google Scholar] [CrossRef] [PubMed]
- Bandodkar, A.J.; Molinnus, D.; Mirza, O.; Guinovart, T.; Windmiller, J.R.; Valdés-Ramírez, G.; Andradec, F.J.; Schöning, M.J.; Wang, J. Epidermal tattoo potentiometric sodium sensors with wireless signal transduction for continuous non-invasive sweat monitoring. Biosens. Bioelectron. 2014, 54, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Olmos, A.; Fernández-Salmerón, J.; Lopez-Ruiz, N.; Torres, A.R.; Capitan-Vallvey, L.F.; Palma, A.J. Screen printed flexible radiofrequency identification tag for oxygen monitoring. Anal. Chem. 2013, 85, 11098–11105. [Google Scholar] [CrossRef] [PubMed]
- Foster, C.W.; Metters, J.P.; Kampouris, D.K.; Banks, C.E. Ultraflexible screen-printed graphitic electroanalytical sensing platforms. Electroanalysis 2014, 26, 262–274. [Google Scholar] [CrossRef]
- Kim, D.-H.; Lu, N.; Ma, R.; Kim, Y.-S.; Kim, R.-H.; Wang, S.; Wu, J.; Won, S.M.; Tao, H.; Islam, A.; et al. Epidermal electronics. Science 2011, 333, 838–843. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-H.; Kim, Y.-S.; Wu, J.; Liu, Z.; Song, J.; Kim, H.-S.; Huang, Y.Y.; Hwang, K.-C.; Rogers, J.A. Ultrathin silicon circuits with strain-isolation layers and mesh layouts for high-performance electronics on fabric, vinyl, leather, and paper. Adv. Mater. 2009, 21, 3703–3707. [Google Scholar] [CrossRef]
- Ying, M.; Bonifas, A.P.; Lu, N.; Su, Y.; Li, R.; Cheng, H.; Ameen, A.; Huang, Y.; Rogers, J.A. Silicon nanomembranes for fingertip electronics. Nanotechnology 2012. [Google Scholar] [CrossRef] [PubMed]
- Son, D.; Lee, J.; Qiao, S.; Ghaffari, R.; Kim, J.; Lee, J.E.; Song, C.; Kim, S.J.; Lee, D.J.; Jun, S.W.; et al. Multifunctional wearable devices for diagnosis and therapy of movement disorders. Nat. Nanotechnol. 2014, 9, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Jang, K.-I.; Chung, H.U.; Xu, S.; Lee, C.H.; Luan, H.; Jeong, J.; Cheng, H.; Kim, G.-T.; Han, S.Y.; Lee, J.W.; et al. Soft network composite materials with deterministic and bio-inspired designs. Nat. Commun. 2015. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhang, Y.; Cho, J.; Lee, J.; Huang, X.; Jia, L.; Fan, J.A.; Su, Y.; Su, J.; Zhang, H.; et al. Stretchable batteries with self-similar serpentine interconnects and integrated wireless recharging systems. Nat. Commun. 2013. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.-W.; Kim, M.K.; Cheng, H.; Yeo, W.-H.; Huang, X.; Liu, Y.; Zhang, Y.; Huang, Y.; Rogers, J.A. Capacitive epidermal electronics for electrically safe, long-term electrophysiological measurements. Adv. Healthcare Mater. 2014, 3, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Banks, A.; Cheng, H.; Xie, Z.; Xu, S.; Jang, K.-I.; Lee, J.W.; Liu, Z.; Gutruf, P.; Huang, X.; et al. Epidermal electronics with advanced capabilities in near-field communication. Small 2015, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jang, K.-I.; Han, S.Y.; Xu, S.; Mathewson, K.E.; Zhang, Y.; Jeong, J.-W.; Kim, G.-T.; Webb, R.C.; Lee, J.W.; Dawidczyk, T.J.; et al. Rugged and breathable forms of stretchable electronics with adherent composite substrates for transcutaneous monitoring. Nat. Commun. 2014. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-K.; Kim, B.J.; Jang, H.; Yoon, S.C.; Lee, C.; Hong, B.H.; Rogers, J.A.; Cho, J.H.; Ahn, J.-H. Stretchable graphene transistors with printed dielectrics and gate electrodes. Nano Lett. 2011, 11, 4642–4646. [Google Scholar] [CrossRef] [PubMed]
- Viventi, J.; Kim, D.-H.; Moss, J.D.; Kim, Y.-S.; Blanco, J.A.; Annetta, N.; Hicks, A.; Xiao, J.; Huang, Y.; Callans, D.J.; et al. A conformal, bio-interfaced class of silicon electronics for mapping cardiac electrophysiology. Sci. Transl. Med. 2010, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Viventi, J.; Kim, D.-H.; Vigeland, L.; Frechette, E.S.; Blanco, J.A.; Kim, Y.-S.; Avrin, A.E.; Tiruvadi, V.R.; Hwang, S.-W.; Vanleer, A.C.; et al. Flexible, foldable, actively multiplexed, high-density electrode array for mapping brain activity in vivo. Nat. Neurosci. 2011, 14, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Suo, Z.; Lacour, S.P.; Wagner, S. Compliant thin film patterns of stiff materials as platforms for stretchable electronics. J. Mater. Res. 2005, 20, 3274–3277. [Google Scholar] [CrossRef]
- Ware, T.; Simon, D.; Hearon, K.; Liu, C.; Shah, S.; Reeder, J.; Khodaparast, N.; Kilgard, M.P.; Maitland, D.J.; Rennaker, R.L., II; et al. Three-dimensional flexible electronics enabled by shape memory polymer substrates for responsive neural interfaces. Macromol. Mater. Eng. 2012, 297, 1193–1202. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.; Ware, T.; Marcotte, R.; Lund, B.R.; Smith, D.W.; di Prima, M.; Rennaker, R.L.; Voit, W. A comparison of polymer substrates for photolithographic processing of flexible bioelectronics. Biomed. Microdevices 2013, 15, 925–939. [Google Scholar] [CrossRef] [PubMed]
- Lipomi, D.J.; Vosgueritchian, M.; Tee, B.C.-K.; Hellstrom, S.L.; Lee, J.A.; Fox, C.H.; Bao, Z. Skin-like pressure and strain sensors based on transparent elastic films of carbon nanotubes. Nat. Nanotechnol. 2011, 6, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, G.; Tee, B.C.-K.; Mei, J.; Appleton, A.L.; Kim, D.H.; Wang, H.; Bao, Z. Flexible polymer transistors with high pressure sensitivity for application in electronic skin and health monitoring. Nat. Commun. 2013. [Google Scholar] [CrossRef] [PubMed]
- Kaltenbrunner, M.; Sekitani, T.; Reeder, J.; Yokota, T.; Kuribara, K.; Tokuhara, T.; Drack, M.; Schwödiauer, R.; Graz, I.; et al. An ultra-lightweight design for imperceptible plastic electronics. Nature 2013, 499, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Jeong, G.S.; Baek, D.-H.; Jung, H.C.; Song, J.H.; Moon, J.H.; Hong, S.W.; Kim, I.Y.; Lee, S.-H. Solderable and electroplatable flexible electronic circuit on a porous stretchable elastomer. Nat. Commun. 2012. [Google Scholar] [CrossRef] [PubMed]
- Forrest, S.R. The Path to ubiquitous and low-cost organic electronic appliances on plastic. Nature 2004, 428, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Yoon, J.; Yun, S.O.; Hwang, Y.; Jang, H.S.; Ko, H.C. Ultrathin sticker-type ZnO thin film transistors formed by transfer printing via topological confinement of water-soluble sacrificial polymer in dimple structure. Adv. Funct. Mater. 2013, 23, 1475–1482. [Google Scholar] [CrossRef]
- Linder, V.; Gates, B.D.; Ryan, D.; Parviz, B.A.; Whitesides, G.M. Water-soluble sacrificial layers for surface micromachining. Small. 2005, 1, 730–736. [Google Scholar] [CrossRef]
- Yim, K.H.; Zheng, Z.; Liang, Z.; Friend, R.H.; Huck, W.T.S.; Kim, J.-S. Efficient conjugated-polymer optoelectronic devices fabricated by thin-film transfer-printing technique. Adv. Funct. Mater. 2008, 18, 1012–1019. [Google Scholar] [CrossRef]
- Zhao, B.; Ji, G.; Gao, X. Transfer printing of magnetic structures with enhanced performance using a new type of water-soluble sacrificial layer. RSC Adv. 2015, 5, 56959–56966. [Google Scholar] [CrossRef]
- Carlson, A.; Bowen, A.M.; Huang, Y.; Nuzzo, R.G.; Rogers, J.A. Transfer printing techniques for materials assembly and micro/nanodevice fabrication. Adv. Mater. 2012, 24, 5284–5318. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.-H.; Kim, Y.-S.; Lee, J.; Ameen, A.; Shi, L.; Li, M.; Wang, S.; Ma, R.; Jin, S.H.; et al. multifunctional epidermal electronics printed directly onto the skin. Adv. Mater. 2013, 25, 2773–2778. [Google Scholar] [CrossRef] [PubMed]
- Idustrial Internet of Things: Unleashing the Potential of Connected Products and Services. Available online: http://www3.weforum.org/docs/WEFUSA_IndustrialInternet_Report2015.pdf (accessed on 22 January 2015).
- Atzori, L.; Iera, A.; Morabito, G. The Internet of Things: A Survey. Comput. Networks 2010, 54, 2787–2805. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Yasuda, T.; Tsutsui, T. Flexible organic field-effect transistors fabricated by the electrode-peeling transfer with an assist of self-assembled monolayer. Appl. Phys. Lett. 2003, 82, 4373–4375. [Google Scholar] [CrossRef]
- Ilic, B.; Craighead, H.G. Topographical patterning of chemically sensitive biological materials using a polymer-based dry lift off. Biomed. Microdevices 2000, 2, 317–322. [Google Scholar] [CrossRef]
- Defranco, J.A.; Schmidt, B.S.; Lipson, M.; Malliaras, G.G. Photolithographic patterning of organic electronic materials. Org. Electron. 2006, 7, 22–28. [Google Scholar] [CrossRef]
- Yoon, J.; Baca, A.J.; Park, S.-I.; Elvikis, P.; Geddes, J.B., III; Li, L.; Kim, R.H.; Xiao, J.; Wang, S.; Kim, T.-H.; et al. Ultrathin silicon solar microcells for semitransparent, mechanically flexible and microconcentrator module designs. Nat. Mater. 2008, 7, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-I.; Xiong, Y.; Kim, R.-H.; Elvikis, P.; Meitl, M.; Kim, D.-H.; Wu, J.; Yoon, J.; Yu, C.-J.; Liu, Z.; et al. Printed assemblies of inorganic light-emitting diodes for deformable and semitransparent displays. Science 2009, 325, 977–981. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.-J.; Kim, T.-I.; Kim, H.-S.; Wells, S.A.; Jo, S.; Ahmed, N.; Jung, Y.H.; Won, S.M.; Bower, C.A.; Rogers, J.A. Fabrication of releasable single-crystal silicon–metal oxide field-effect devices and their deterministic assembly on foreign substrates. Adv. Funct. Mater. 2011, 21, 3029–3036. [Google Scholar] [CrossRef]
- Van Krevelen, D.W.; te Nijenhuis, K. Properties of Polymers; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Bowden, N.; Brittain, S.; Evans, A.G.; Hutchinson, J.W.; Whitesides, G.M. Spontaneous formation of ordered structures in thin films of metals supported on an elastomeric polymer. Nature 1998, 393, 146–149. [Google Scholar]
- Huck, W.T.S.; Bowden, N.; Onck, P.; Pardoen, T.; Hutchinson, J.W.; Whitesides, G.M. Ordering of spontaneously formed buckles on planar surfaces. Langmuir 2000, 16, 3497–3501. [Google Scholar] [CrossRef]
- Lacour, S.P.; Jones, J.; Wagner, S.; Li, T.; Suo, Z. Strechable interconnects for elastics electronic surfaces. IEEE Proc. 2005, 93, 1459–1466. [Google Scholar] [CrossRef]
- Adrega, T.; Lacour, S.P. Stretchable gold conductors embedded in PDMS and patterned by photolithography: fabrication and electromechanical characterization. J. Micromech. Microeng. 2010, 20, 055025–055033. [Google Scholar] [CrossRef]
- Niedermeyer, E.; Lopes da Silva, F.H. Electroencephalography: Basic Principles, Clinical Applications, and Related Fields; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2012. [Google Scholar]
- McAdams, E.T.; Jossinet, J.; Subramanian, R.; McCauley, R.G.E. Characterization of gold electrodes in phosphate buffered saline solution by impedance and noise measurements for biological applications. In Proceedings of the 28th IEEE EMBS Annual International Conference, New York, NY, USA, 30 August–3 September 2006.
- Tallgrena, P.; Vanhataloa, S.; Kailaa, K.; Voipioa, J. Evaluation of commercially available electrodes and gels for recording of slow EEG potentials. Clin. Neurophysiol. 2005, 116, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Ba, D.; Babadi, B.; Purdon, P.L.; Brown, E.N. Robust spectrotemporal decomposition by Iteratively reweighted least squares. Proc. Natl. Acad. Sci. USA. 2014, 111, E5336–E5345. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.-D.; Wang, I.-J.; Chen, S.-F.; Chang, J.-Y.; Lin, C.-T. Design, fabrication and experimental validation of a novel dry-contact sensor for measuring electroencephalography signals without skin preparation. Sensors 2011, 11, 5819–5834. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Gu, Y.; Fei, P.; Mai, W.; Gao, Y.; Yang, R.; Bao, G.; Wang, Z.L. Flexible piezotronic strain sensor. Nano Lett. 2008, 8, 3035–3040. [Google Scholar] [CrossRef] [PubMed]
- Chang, N.-K.; Su, C.-C.; Chang, S.-H. Fabrication of single-walled carbon nanotube flexible strain sensors with high sensitivity. Appl. Phys. Lett. 2008, 92. [Google Scholar] [CrossRef]
- Patel, J.N.; Kaminska, B.; Gray, B.L.; Gates, B.D. A sacrificial SU-8 mask for direct metallization on PDMS. J. Micromech. Microeng. 2009, 19, 115014–115024. [Google Scholar] [CrossRef]
- Patel, J.N.; Kaminska, B.; Gray, B.L.; Gates, B.D. PDMS as a sacrificial substrate for SU-8-based biomedical and microfluidic applications. J. Micromech. Microeng. 2008, 18, 095028–095039. [Google Scholar] [CrossRef]
- Melzer, M.; Karnaushenko, D.; Lin, G.; Baunack, S.; Makarov, D.; Schmidt, O. Direct transfer of magnetic sensor devices to elastomeric supports for stretchable electronics. Adv. Mater. 2015, 27, 1333–1338. [Google Scholar] [CrossRef] [PubMed]
- McDonald, J.C.; Duffy, D.C.; Anderson, J.R.; Chiu, D.T.; Wu, H.; Schueller, O.J.A.; Whitesides, G.M. Fabrication of microfluidic systems in poly (dimethylsiloxane). Electrophoresis 2000, 21, 27–40. [Google Scholar] [CrossRef]
- Schmid, H.; Wolf, H.; Allenspach, R.; Riel, H.; Karg, S.; Michel, B.; Delamarche, E. Preparation of metallic films on elastomeric stamps and their application for contact processing and contact printing. Adv. Funct. Mater. 2003, 13, 145–153. [Google Scholar] [CrossRef]
- Sun, Y.; Lacour, S.P.; Brooks, R.A.; Rushton, N.; Fawcett, J.; Cameron, R.E. Assessment of the biocompatibility of photosensitive polyimide for implantable medical device use. J. Biomed. Mater. Res. Part A 2009, 90, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Harbert, M.J.; Rosenberg, S.S.; Mesa, D.; Sinha, M.; Karanjia, N.P.; Nespeca, M.; Coleman, T.P. Demonstration of the use of epidermal electronics in neurological monitoring. Ann. Neurol. 2013, 74, S76–S77. [Google Scholar]
- Merrill, D.R.; Bikson, M.; Jeffrys, J.G.R. Design, electrical stimulation of excitable tissue: Design of efficacious and safe protocols. J. Neurosci. Method. 2005, 141, 171–198. [Google Scholar] [CrossRef] [PubMed]
- Ghomashchi, A.; Zheng, Z.; Majaj, N.; Trumpis, M.; Kiorpes, L.; Viventi, J. A low-cost, open-source, wireless electrophysiology system. In Proceedings of 36th Annual International Conference of the IEEE: Engineering in Medicine and Biology Society (EMBC), Chicago, IL, USA, 26–30 August 2014.
- Escabí, M.A.; Read, H.L.; Viventi, J.; Kim, D.-H.; Higgins, N.C.; Storace, D.A.; Liu, A.S.K.; Gifford, A.M.; Burke, J.F.; Campisi, M.; et al. A high-density, high-channel count, multiplexed µ ECoG array for auditory-cortex recordings. J. Neurophysiol. 2014, 112, 1566–1583. [Google Scholar] [CrossRef] [PubMed]
- Mercier, P.P.; Lysaght, A.C.; Bandyopadhyay, S.; Chandrakasan, A.P.; Stankovic, K.M. Energy extraction from the biologic battery in the inner ear. Nat. Biotechnol. 2012, 30, 1240–1244. [Google Scholar] [CrossRef] [PubMed]
- Report to the President: Accelerating U.S. Advanced Manufacturing. Available online: http://www.whitehouse.gov/sites/default/files/microsites/ostp/PCAST/amp20_report_final.pdf (accessed on 30 October 2014).
- Flexible Hybrid Electronics Manufacturing Innovation (FHEMI) Institute. Available online: http://manufacturing.gov/docs/fhemi-faq.pdf (accessed on 1 April 2015).
- Patrito, N.; McLachlan, J.M.; Faria, S.N.; Chan, J.; Norton, P.R. A novel metal-protected plasma treatment for the robust bonding of polydimethylsiloxane. Lab Chip 2007, 7, 1813–1818. [Google Scholar] [CrossRef] [PubMed]
- Efimenko, K.; Wallace, W.E.; Genzer, J. Surface modification of sylgard-184 poly(dimethyl siloxane) networks by ultraviolet and ultraviolet/ozone treatment. J. Coll. Interface Sci. 2002, 254, 306–315. [Google Scholar] [CrossRef]
- Ginn, B.T.; Steinbock, O. Polymer surface modification using microwave-oven-generated plasma brent. Langmuir 2003, 19, 8117–8118. [Google Scholar] [CrossRef]
- Armani, D.; Liu, C.; Aluru, N. Re-configurable fluid circuits by PDMS elastomer micromachining. In Proceedings of the 12th IEEE International Conference: Micro Electro Mechanical Systems, Orlando, FL, USA, 17–21 January 1999.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, D.Y.; Kim, Y.-S.; Ornelas, G.; Sinha, M.; Naidu, K.; Coleman, T.P. Scalable Microfabrication Procedures for Adhesive-Integrated Flexible and Stretchable Electronic Sensors. Sensors 2015, 15, 23459-23476. https://doi.org/10.3390/s150923459
Kang DY, Kim Y-S, Ornelas G, Sinha M, Naidu K, Coleman TP. Scalable Microfabrication Procedures for Adhesive-Integrated Flexible and Stretchable Electronic Sensors. Sensors. 2015; 15(9):23459-23476. https://doi.org/10.3390/s150923459
Chicago/Turabian StyleKang, Dae Y., Yun-Soung Kim, Gladys Ornelas, Mridu Sinha, Keerthiga Naidu, and Todd P. Coleman. 2015. "Scalable Microfabrication Procedures for Adhesive-Integrated Flexible and Stretchable Electronic Sensors" Sensors 15, no. 9: 23459-23476. https://doi.org/10.3390/s150923459
APA StyleKang, D. Y., Kim, Y.-S., Ornelas, G., Sinha, M., Naidu, K., & Coleman, T. P. (2015). Scalable Microfabrication Procedures for Adhesive-Integrated Flexible and Stretchable Electronic Sensors. Sensors, 15(9), 23459-23476. https://doi.org/10.3390/s150923459





