Electrochemical Impedance Sensors for Monitoring Trace Amounts of NO3 in Selected Growing Media
Abstract
:1. Introduction
2. Chemistry of the EIS Sensor
2.1. EIS Nitrate Sensor Structure
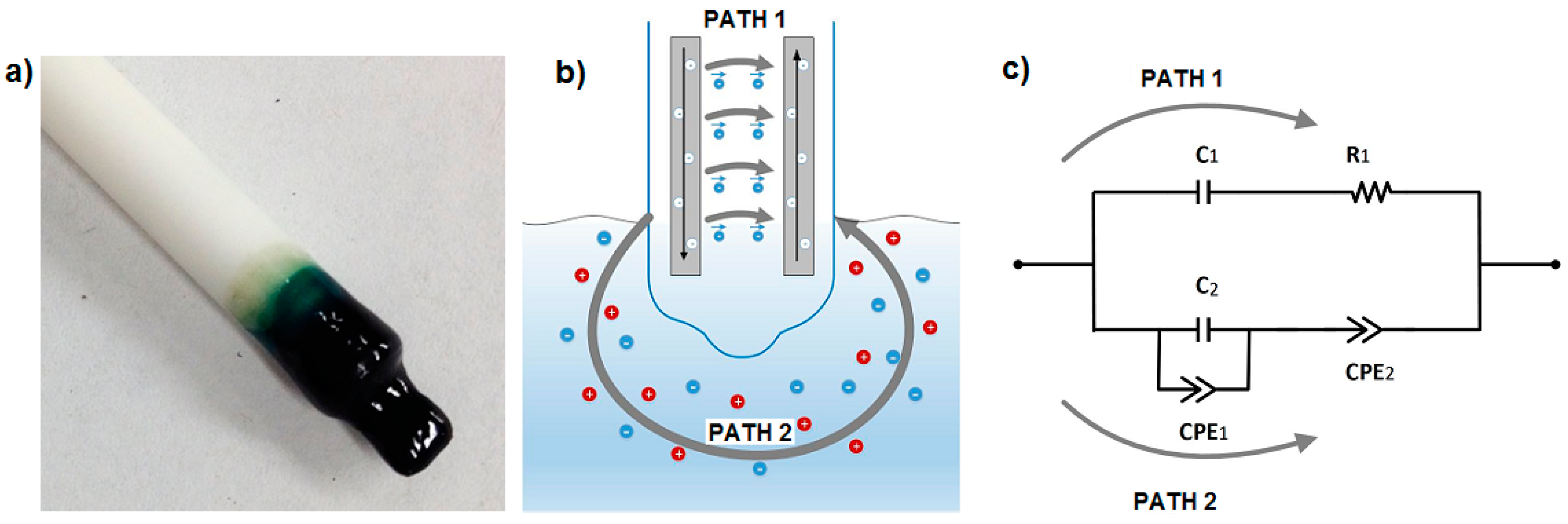
2.2. Medium under Test
3. Experimental Setup and Results
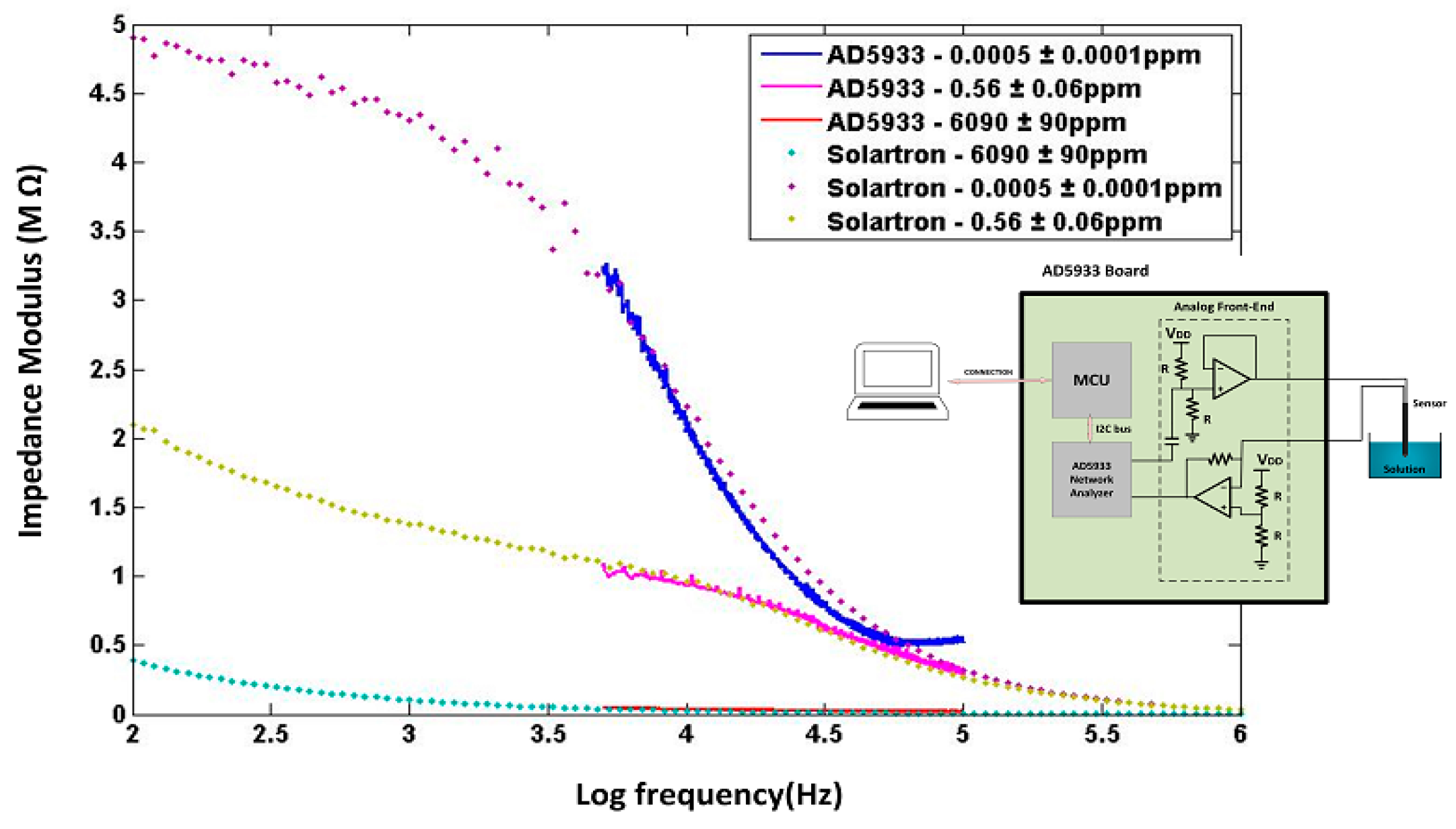
3.1. Measurement Setup
3.2. Measurement Results
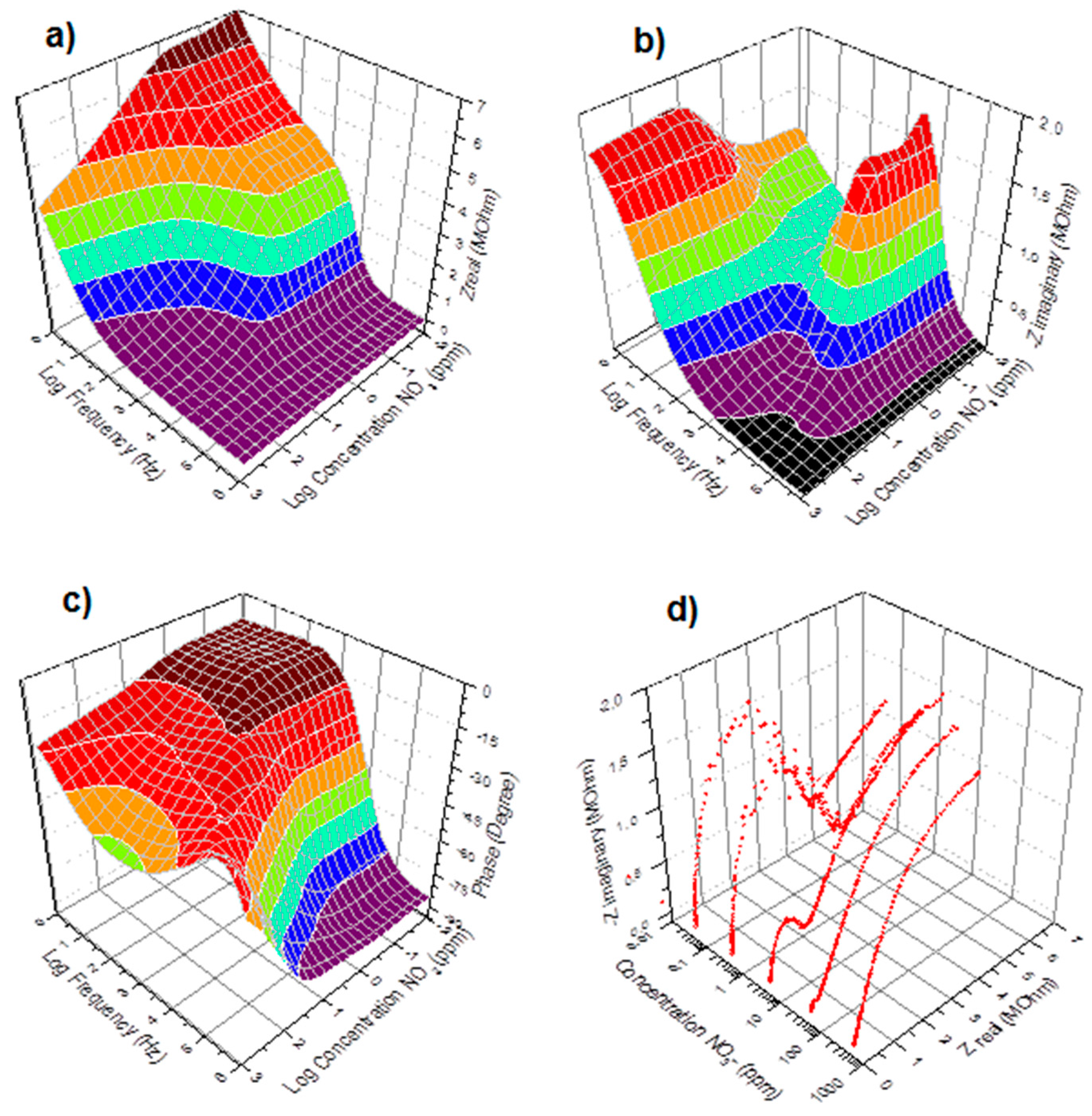
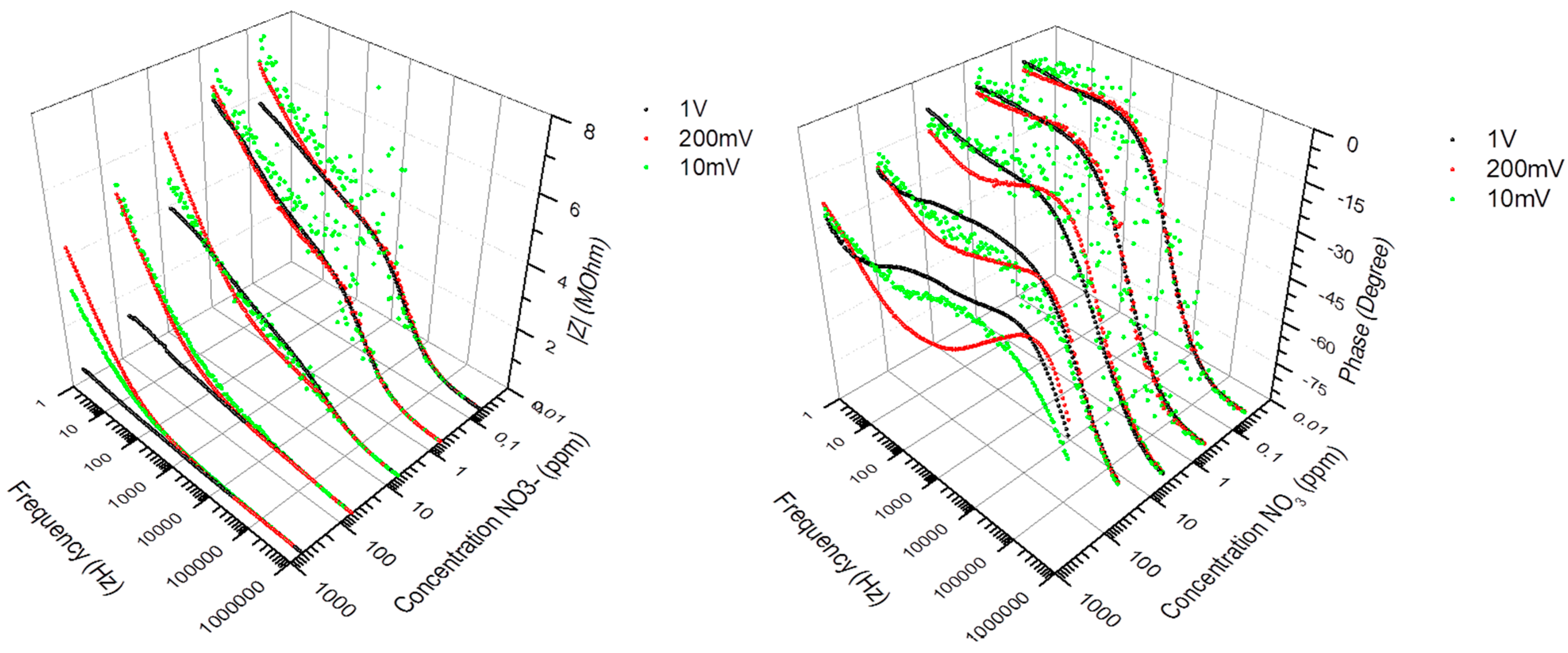
3.3. Measurement Non-Linearity
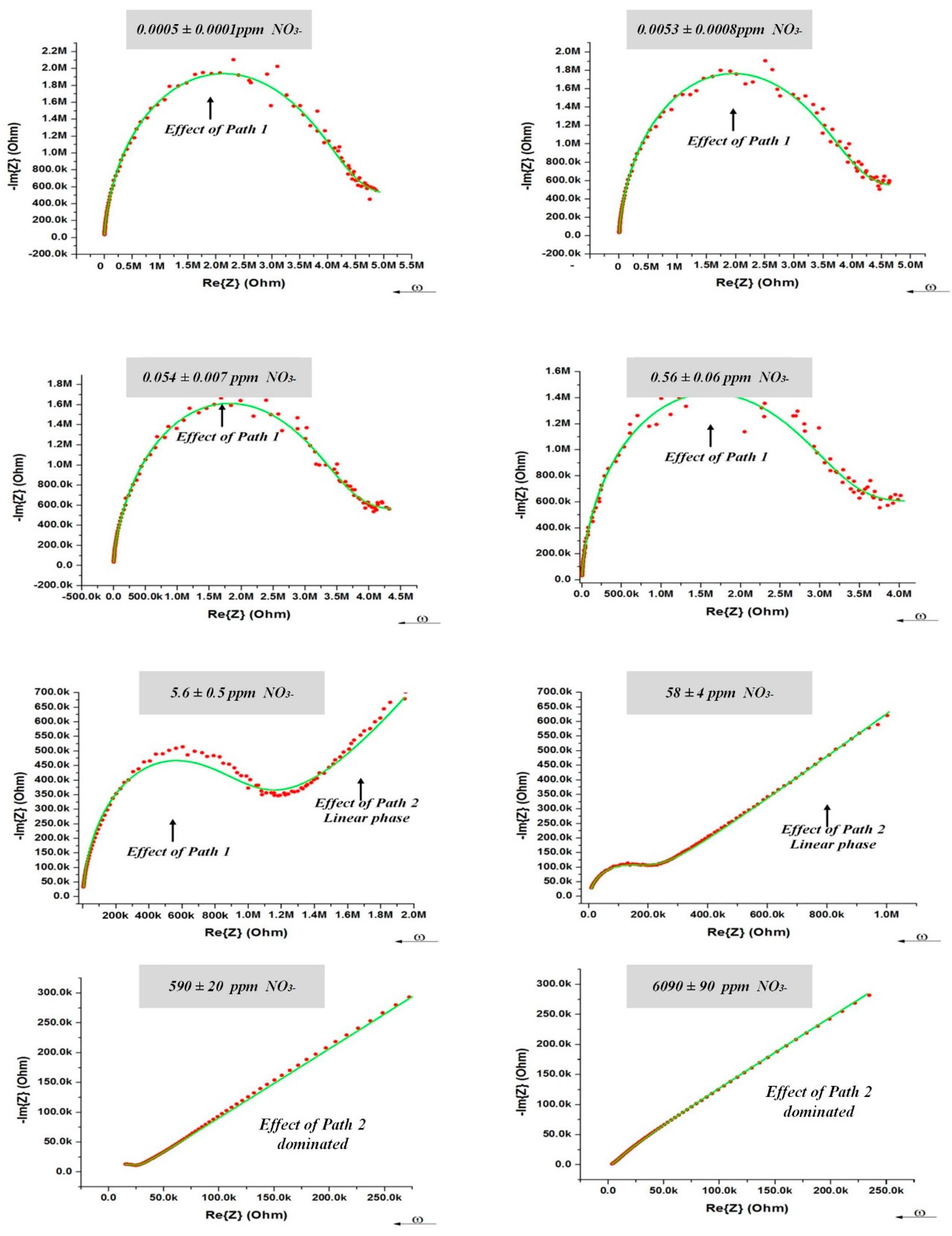
3.4. Equivalent Electrical Circuit
3.5. Sensor Circuit Miniaturization
4. Field Testing in Tree Nursery Under ISO/IEC 17025 Certifications
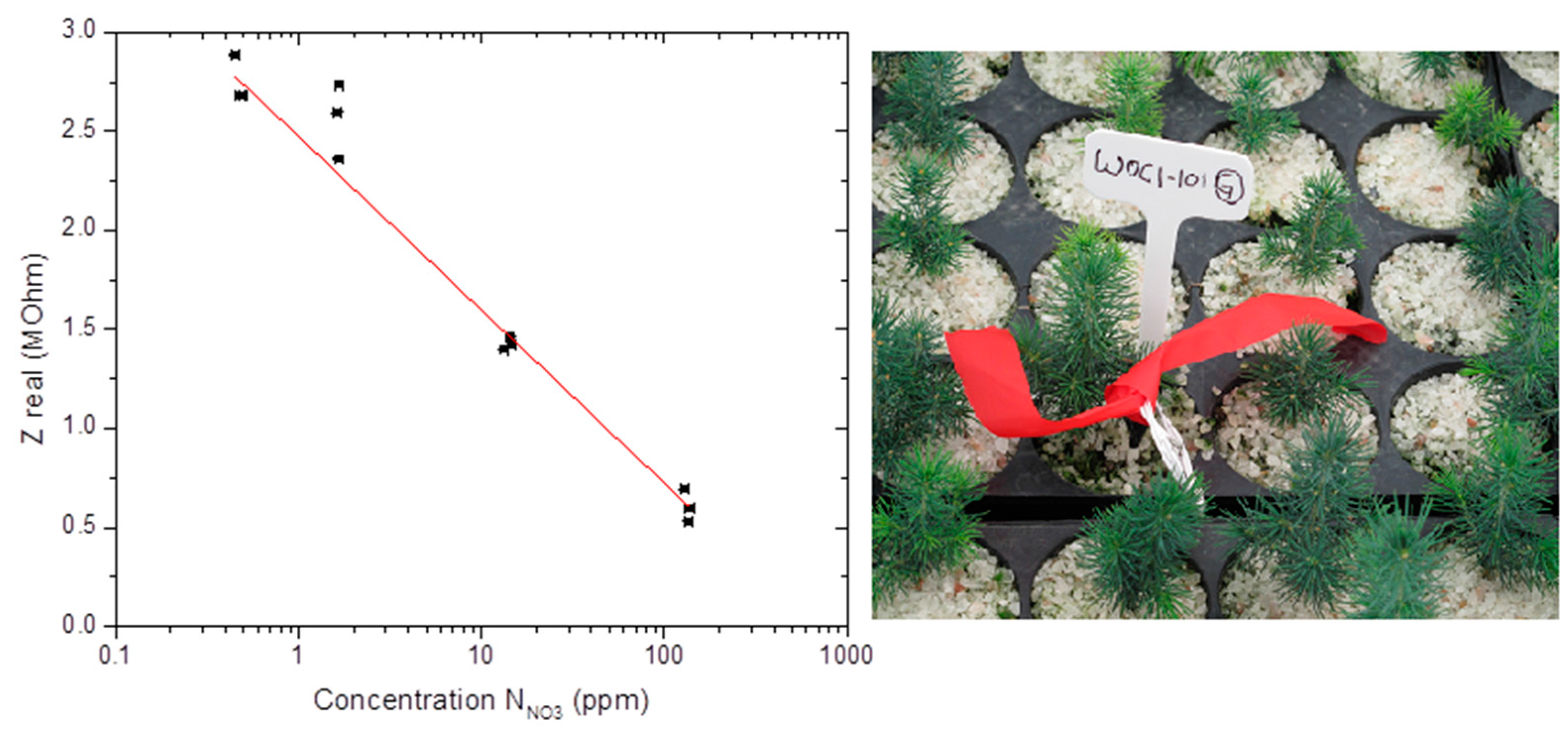
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schaffers, H.; Komninos, N.; Pallot, M.; Trousse, B.; Nilsson, M. The Future Internet; Springer: Berlin, Germany, 2011; Volume 6656, pp. 431–446. [Google Scholar]
- Landis, T.D. The Container Tree Nursery Manual; US Department of Agriculture: Washington, DC, USA, 1990; Volume 2.
- Bakker, E.; Buhlmann, P.; Pretsch, E. Polymer Membrane Ion-Selective Electrodes—What are the Limits? Electroanalysis 1999, 11, 915–933. [Google Scholar] [CrossRef]
- Bergveld, P. Thirty Years of ISFETOLOGY. Sens. Actuators B Chem. 2003, 88, 1–20. [Google Scholar] [CrossRef]
- Ballesteros-Gomez, A.; Rubio, S. Recent Advances in Environmental Analysis. Anal. Chem. 2011, 83, 4579–4613. [Google Scholar] [CrossRef] [PubMed]
- McBratney, A.; Whelan, B.; Ancev, T. Future Directions of Precision Agriculture. Precis. Agric. 2005, 6, 7–23. [Google Scholar] [CrossRef]
- Corwin, D.L.; Lesch, S.M. Application of Soil Electrical Conductivity to Precision Agriculture: Theory, Principles, and Guidelines. Agron. J. 2003, 95, 455–471. [Google Scholar] [CrossRef]
- Chighladze, G. Estimation of Change in Soil Water Nitrate-Nitrogen Concentration Using Impedance Spectra. Ph.D. Dissertation, Iowa State University, IA, USA, 2011. [Google Scholar]
- Macdonald, J.R.; Barsoukov, E. Impedance Spectroscopy Theory, Experiment, and Applications; Wiley-Interscience: Hoboken, NJ, 2005. [Google Scholar]
- Mu, X.; Wei, J.; Dong, J.; Ke, W. In Situ Corrosion Monitoring of Mild Steel in a Simulated Tidal Zone without Marine Fouling Attachment by Electrochemical Impedance Spectroscopy. J. Mater. Sci. Technol. 2014, 30, 1043–1050. [Google Scholar] [CrossRef]
- De Beer, C.; Barendse, P.S.; Pillay, P. Fuel Cell Condition Monitoring Using Optimized Broadband Impedance Spectroscopy. Ind. Electron. 2015, 62, 5306–5316. [Google Scholar] [CrossRef]
- Stejskal, K.; Krizkova, S.; Adam, V.; Sures, B. Bio-Assessing of Environmental Pollution via Monitoring of Metallothionein Level Using Electrochemical Detection. Sens. J. 2008, 8, 1578–1585. [Google Scholar] [CrossRef]
- Tomkiewicz, D.; Piskier, T. A plant based sensing method for nutrition stress monitoring. Precis. Agric. 2012, 13, 370–383. [Google Scholar] [CrossRef]
- Muñoz-Huerta, R.F.; Ortiz-Melendez, A.J.; Guevara-Gonzalez, R.G.; Torres-Pacheco, I.; Herrera-Ruiz, G.; Contreras-Medina, L.M.; Prado-Olivarez, J.; Ocampo-Velazquez, R.V. An Analysis of Electrical Impedance Measurements Applied for Plant N Status Estimation in Lettuce (Lactuca sativa). Sensors 2014, 14, 11492–11503. [Google Scholar] [CrossRef] [PubMed]
- Selwyna, P.G.C.; Loganathan, P.R.; Begam, K.H. Development of electrochemical biosensor for breast cancer detection using gold nanoparticle doped CA 15-3 antibody and antigen interaction. In Proceedings of 2013 International Conference on Signal Processing Image Processing & Pattern Recognition (ICSIPR), Coimbatore, India, 7–8 February 2013; pp. 75–81.
- Chowdhurya, A.D.; Gangopadhyayb, R.; De, A. Highly sensitive electrochemical biosensor for glucose, DNA and protein using gold-polyaniline nanocomposites as a common matrix. Sens. Actuators B Chem. 2014, 190, 348–356. [Google Scholar] [CrossRef]
- Ardakani, M.M.; Dastanpour, A.; Niasari, M.S. A highly selective nitrate electrode based on a tetramethyl cyclotetra-decanato-nickel(II) complex. J. Electroanal. Chem. 2004, 568, 1–6. [Google Scholar] [CrossRef]
- Solartron. 1260 Impedance/Gain-Phase Analyzer Operating Manual, Issue CB; January 1996. Available online: https://mm.ece.ubc.ca/mediawiki/images/8/8b/1260_manual.pdf (accessed on 4 April 2015).
- Lasia, A. Electrochemical Impedance Spectroscopy and Its Applications, 1st ed.; Springer: New York, NY, USA, 2014; pp. 7–66. [Google Scholar]
- Wang, L.; Zhao, J.S.; He, X.M.; Gao, J.; Li, J.J.; Wan, C.R.; Jiang, C.Y. Electrochemical Impedance Spectroscopy (EIS) Study of LiNi1/3Co1/3Mn1/3O2 for Li-ion Batteries. J. Electrochem. Sci. 2012, 7, 345–353. [Google Scholar]
- Rioux, M.; Ledemi, Y.; Viens, J.; Morency, S.; Ghaffari, S.A.; Messaddeq, Y. Optically-transparent and electrically-conductive AgI-AgPO3-WO3 glass fibers. RSC Adv. 2015, 5, 40236–40248. [Google Scholar]
- Bondarenko, A.S.; Ragoisha, G.A. Progress in Chemometrics Research; Pomerantsev, A.L., Ed.; Nova Science Publishers: New York, NY, USA, 2005; pp. 89–102. Available online: http://www.abc.chemistry.bsu.by/vi/analyser/ (accessed on 4 April 2015).
- Agilent Impedance Measurement Handbook: A Guide to Measurement Technology and Techniques, 4th Edition. Available online: http://cp.literature.agilent.com/litweb/pdf/5950-3000.pdf (accessed on 4 April 2015).
- Analog Devices. 1 MSPS, 12 Bit Impedance Converter Network Analyzer, Datasheet AD5933. Available online: http://www.analog.com/media/en/technical-documentation/data-sheets/AD5933.pdf (accessed on 4 April 2015).
- Matsiev, L. Improving Performance and Versatility of Systems Based on Single-Frequency DFT Detectors such as AD5933. Electronics 2015, 4, 1–34. [Google Scholar] [CrossRef]
- Analog Devices. EVAL-AD5933EB Document. Available online: http://www.analog.com/media/en/technicaldocumentation/evaluationdocumentation/537700023EVAL_AD5933EB.pdf (accessed on 15 June 2015).
- Lamhamedi, M.S.; Chamberland, H.; Bernier, P.Y.; Tremblay, F.M. Clonal variation in morphology, growth, physiology, anatomy and ultrastructure of container-grown white spruce somatic seedlings. Tree Physiol. 2000, 20, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Lamhamedi, M.S.; Lambany, G.; Margolis, H.A.; Renaud, M.; Veilleux, L.; Bernier, P.Y. Growth, physiology and leachate losses in Picea glauca seedlings (1 + 0) grown in air-slit containers under different irrigation regimes. Can. J. For. Res. 2001, 31, 1968–1980. [Google Scholar] [CrossRef]
- ISO/IEC 17025 relates to the International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC), and specifies the general requirements for the competence to carry out tests and/or calibrations, including sampling. It covers testing and calibration performed using standard methods, non-standard methods, and laboratory-developed methods. http://www.iso.org/iso/catalogue_detail.htm?csnumber=39883 (accessed on 14 July 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghaffari, S.A.; Caron, W.-O.; Loubier, M.; Normandeau, C.-O.; Viens, J.; Lamhamedi, M.S.; Gosselin, B.; Messaddeq, Y. Electrochemical Impedance Sensors for Monitoring Trace Amounts of NO3 in Selected Growing Media. Sensors 2015, 15, 17715-17727. https://doi.org/10.3390/s150717715
Ghaffari SA, Caron W-O, Loubier M, Normandeau C-O, Viens J, Lamhamedi MS, Gosselin B, Messaddeq Y. Electrochemical Impedance Sensors for Monitoring Trace Amounts of NO3 in Selected Growing Media. Sensors. 2015; 15(7):17715-17727. https://doi.org/10.3390/s150717715
Chicago/Turabian StyleGhaffari, Seyed Alireza, William-O. Caron, Mathilde Loubier, Charles-O. Normandeau, Jeff Viens, Mohammed S. Lamhamedi, Benoit Gosselin, and Younes Messaddeq. 2015. "Electrochemical Impedance Sensors for Monitoring Trace Amounts of NO3 in Selected Growing Media" Sensors 15, no. 7: 17715-17727. https://doi.org/10.3390/s150717715
APA StyleGhaffari, S. A., Caron, W.-O., Loubier, M., Normandeau, C.-O., Viens, J., Lamhamedi, M. S., Gosselin, B., & Messaddeq, Y. (2015). Electrochemical Impedance Sensors for Monitoring Trace Amounts of NO3 in Selected Growing Media. Sensors, 15(7), 17715-17727. https://doi.org/10.3390/s150717715





