A Reliable and Simple Method for Fabricating a Poly(Dimethylsiloxane) Electrospray Ionization Chip with a Corner-Integrated Emitter
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.2. Microchip Design and Fabrication
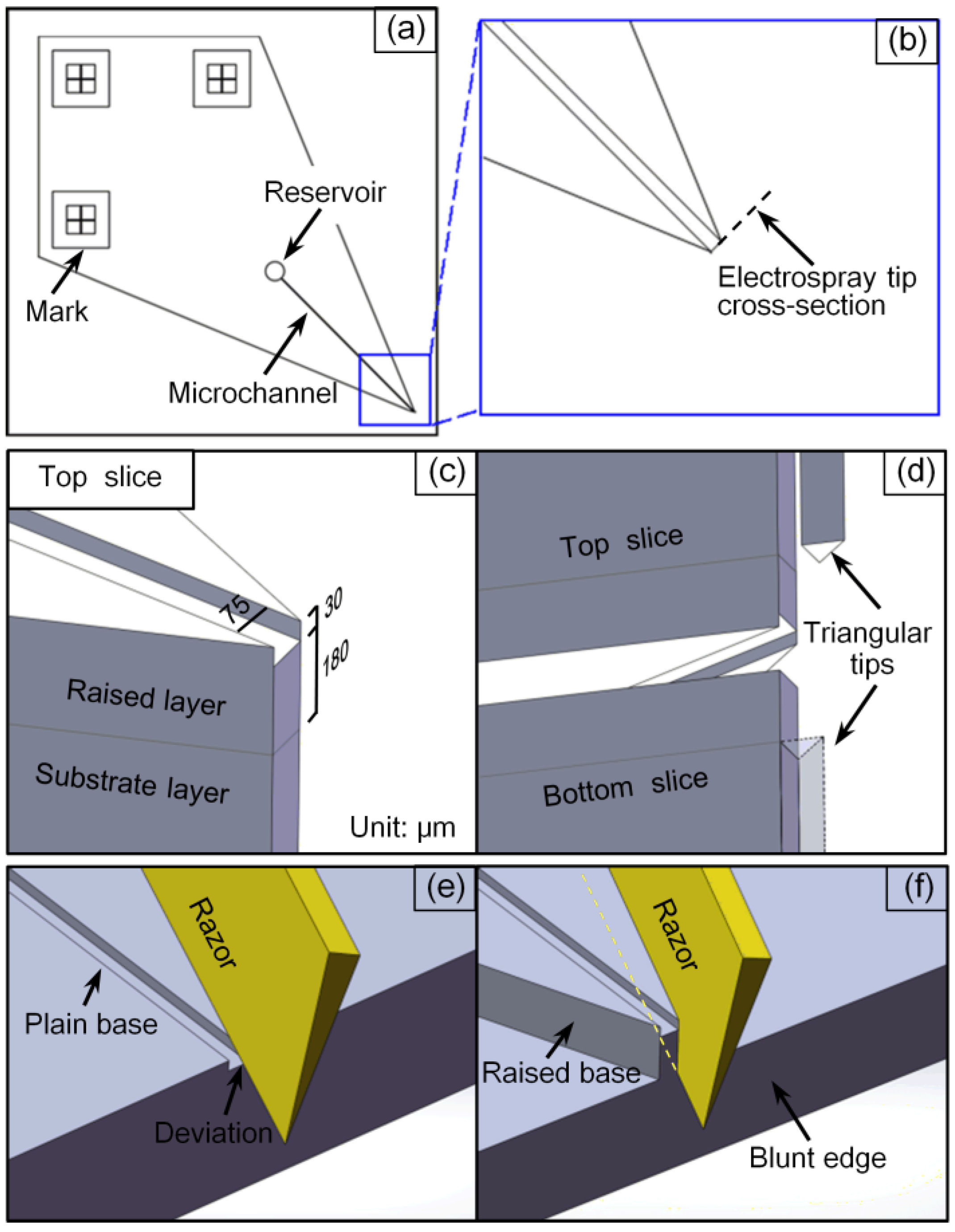
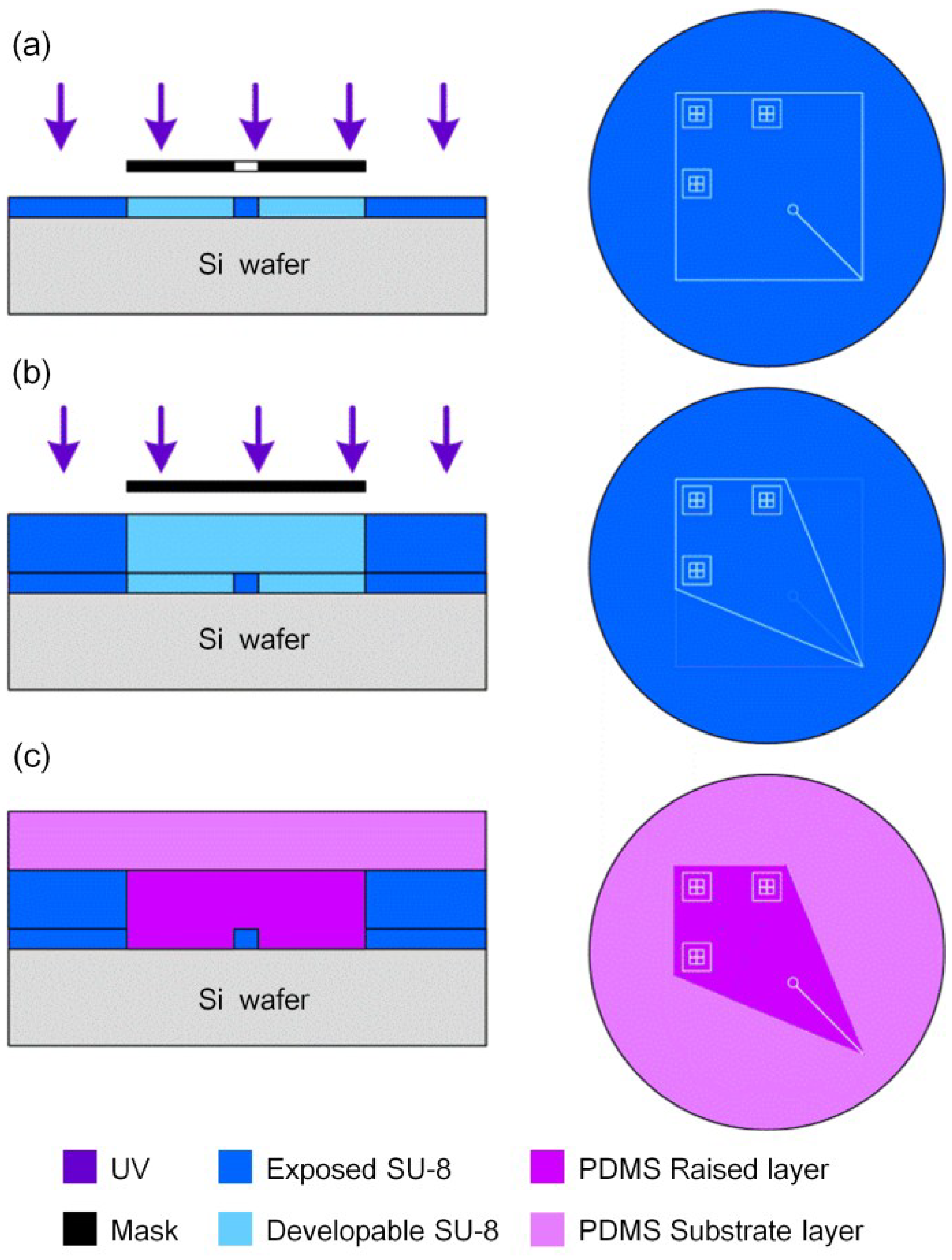
2.3. Mass Spectrometry

3. Results and Discussion
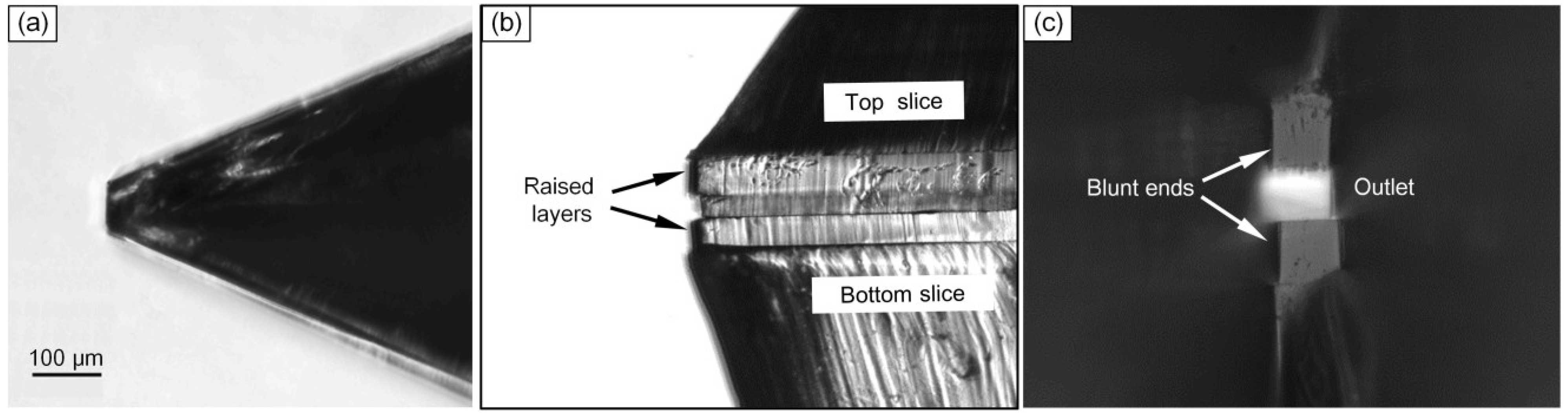
3.1. Characteristics of the Chip Fabrication

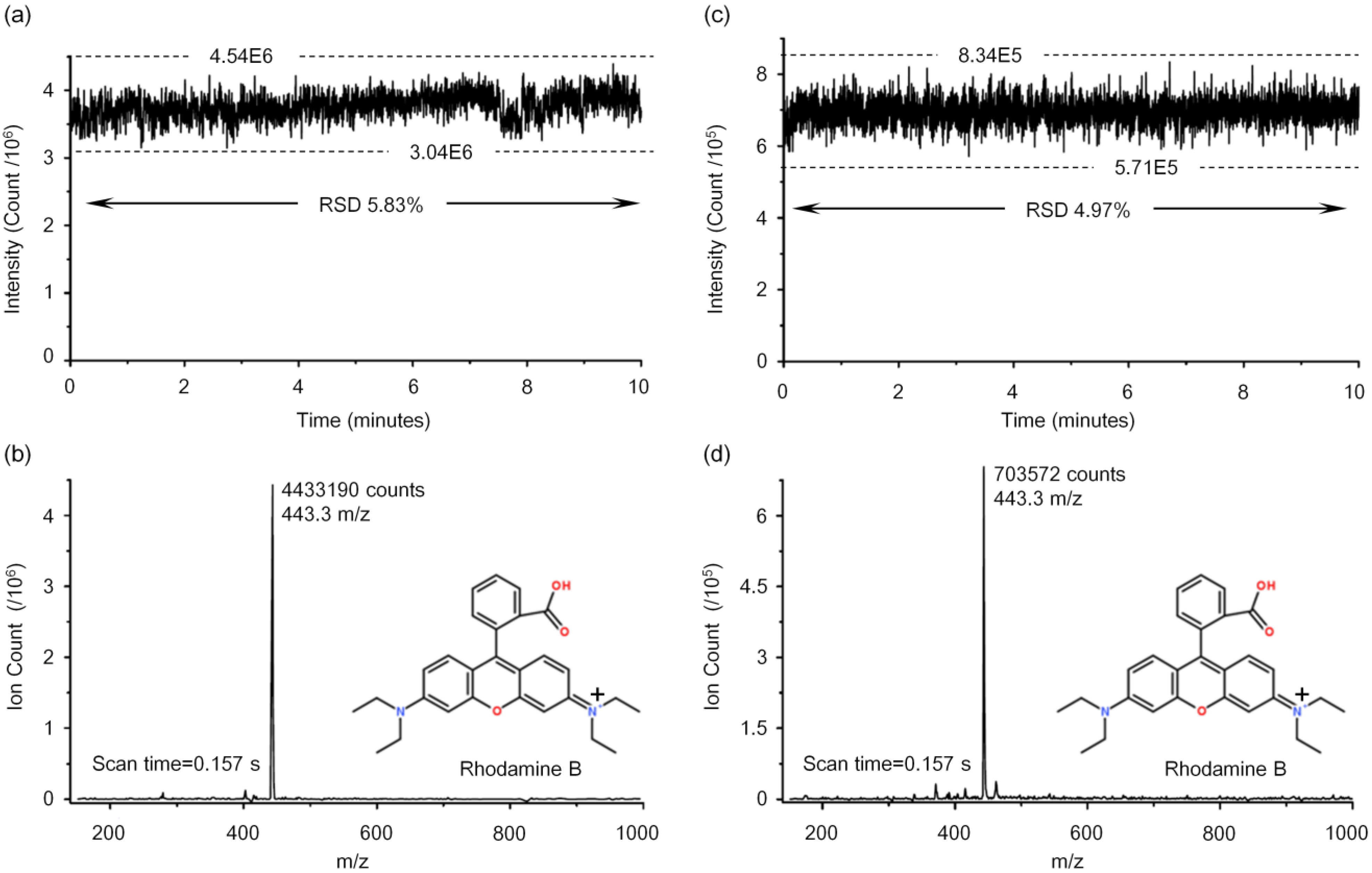
3.2. Performance Tests


4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jacobson, S.C.; Culbertson, C.T.; Daler, J.E.; Ramsey, J.M. Microchip Structures for Submillisecond Electrophoresis. Anal. Chem. 1998, 70, 3476–3480. [Google Scholar]
- Culbertson, C.T.; Jacobson, S.C.; Ramsey, J.M. Microchip Devices for High-Efficiency Separations. Anal. Chem. 2000, 72, 5814–5819. [Google Scholar] [CrossRef] [PubMed]
- Qu, H.Y.; Wang, H.T.; Huang, Y.; Zhong, W.; Lu, H.J.; Kong, J.L.; Yang, P.Y.; Liu, B.H. Stable Microstructured Network for Protein Patterning on a Plastic Microfluidic Channel: Strategy and Characterization of On-Chip Enzyme Microreactors. Anal. Chem. 2004, 76, 6426–6433. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.F.; Kelly, R.T.; Tang, K.Q.; Smith, R.D. Ultrasensitive Nanoelectrospray Ionization-Mass Spectrometry Using Poly(dimethylsiloxane) Microchips with Monolithically Integrated Emitters. Analyst 2010, 135, 2296–2302. [Google Scholar] [CrossRef] [PubMed]
- Koster, S.; Verpoorte, E. A Decade of Microfluidic Analysis Coupled with Electrospray Mass Spectrometry: An Overview. Lab Chip 2007, 7, 1394–1412. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, R.S.; Ramsey, J.M. Generating Electrospray from Microchip Devices Using Electroosmotic Pumping. Anal. Chem. 1997, 69, 1174–1178. [Google Scholar] [CrossRef]
- Schultz, G.A.; Corso, T.N.; Prosser, S.J.; Zhang, S. A Fully Integrated Monolithic Microchip Electrospray Device for Mass Spectrometry. Anal. Chem. 2000, 72, 4058–4063. [Google Scholar] [CrossRef] [PubMed]
- Mery, E.; Ricoul, F.; Sarrut, N.; Constantin, O.; Delapierre, G.; Garin, J.; Vinet, F. A Silicon Microfluidic Chip Integrating an Ordered Micropillar Array Separation Column and a Nano-Electrospray Emitter for LC/MS Analysis of Peptides. Sens. Actuators B Chem. 2008, 134, 438–446. [Google Scholar] [CrossRef]
- Su, S.Q.; Gibson, G.T.T.; Mugo, S.M.; Marecak, D.M.; Oleschuk, R.D. Microstructured Photonic Fibers as Multichannel Electrospray Emitters. Anal. Chem. 2009, 81, 7281–7287. [Google Scholar] [CrossRef] [PubMed]
- Chambers, A.G.; Ramsey, J.M. Microfluidic Dual Emitter Electrospray Ionization Source for Accurate Mass Measurements. Anal. Chem. 2012, 84, 1446–1451. [Google Scholar] [CrossRef] [PubMed]
- Mellors, J.S.; Black, W.A.; Chambers, A.G.; Starkey, J.A.; Lacher, N.A.; Ramsey, J.M. Hybrid Capillary/Microfluidic System for Comprehensive Online Liquid Chromatography-Capillary Electrophoresis-Electrospray Ionization-Mass Spectrometry. Anal. Chem. 2013, 85, 4100–4106. [Google Scholar] [CrossRef] [PubMed]
- Arscott, S. SU-8 as a Material for Lab-on-a-Chip-Based Mass Spectrometry. Lab Chip 2014, 14, 3668–3689. [Google Scholar] [CrossRef] [PubMed]
- Batz, N.G.; Mellors, J.S.; Alarie, J.P.; Ramsey, J.M. Chemical Vapor Deposition of Aminopropyl Silanes in Microfluidic Channels for Highly Efficient Microchip Capillary Electrophoresis-Electrospray Ionization-Mass Spectrometry. Anal. Chem. 2014, 86, 3493–3500. [Google Scholar] [CrossRef] [PubMed]
- Dahlin, A.P.; Wetterhall, M.; Liljegren, G.; Bergstrom, S.K.; Andren, P.; Nyholm, L.; Markides, K.E.; Bergquist, J. Capillary Electrophoresis Coupled to Mass Spectrometry from a Polymer Modified Poly(dimethylsiloxane) Microchip with an Integrated Graphite Electrospray Tip. Analyst 2005, 130, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Sung, W.C.; Makamba, H.; Chen, S.H. Chip-Based Microfluidic Devices Coupled with Electrospray Ionization-Mass Spectrometry. Electrophoresis 2005, 26, 1783–1791. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.L.; Bai, H.Y.; Lin, T.Y.; Fuh, M.R. Microfluidic Chip-Based Liquid Chromatography Coupled to Mass Spectrometry for Determination of Small Molecules in Bioanalytical Applications. Electrophoresis 2012, 33, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Svedberg, M.; Veszelei, M.; Axelsson, J.; Vangbo, M.; Nikolajeff, F. Poly(dimethylsiloxane) Microchip: Microchannel with Integrated Open Electrospray Tip. Lab Chip 2004, 4, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.T.; Tang, K.; Irimia, D.; Toner, M.; Smith, R.D. Elastomeric Microchip Electrospray Emitter for Stable Cone-Jet Mode Operation in the Nanoflow Regime. Anal. Chem. 2008, 80, 3824–3831. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Kelly, R.T.; Tang, K.; Smith, R.D. Membrane-Based Emitter for Coupling Microfluidics with Ultrasensitive Nanoelectrospray Ionization-Mass Spectrometry. Anal. Chem. 2011, 83, 5797–5803. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Knapp, D.R. Microfabrication of Polydimethylsiloxane Electrospray Ionization Emitters. J. Chromatogr. A 2001, 924, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Truong, T.Q.; Nguyen, N.T. A Polymeric Piezoelectric Micropump Based on Lamination Technology. J. Micromech. Microeng. 2004, 14, 632–638. [Google Scholar] [CrossRef]
- Gao, L.; Song, Q.Y.; Patterson, G.E.; Cooks, R.G.; Ouyang, Z. Handheld Rectilinear Ion Trap Mass Spectrometer. Anal. Chem. 2006, 78, 5994–6002. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Sugiarto, A.; Harper, J.D.; Cooks, R.G.; Ouyang, Z. Design and Characterization of a Multisource Hand-Held Tandem Mass Spectrometer. Anal. Chem. 2008, 80, 7198–7205. [Google Scholar] [CrossRef] [PubMed]
- Li, L.F.; Chen, T.C.; Ren, Y.; Hendricks, P.I.; Cooks, R.G.; Ouyang, Z. Mini 12, Miniature Mass Spectrometer for Clinical and Other Applications-Introduction and Characterization. Anal. Chem. 2014, 86, 2909–2916. [Google Scholar] [CrossRef] [PubMed]
- Mellors, J.S.; Jorabchi, K.; Smith, L.M.; Ramsey, J.M. Integrated Microfluidic Device for Automated Single Cell Analysis Using Electrophoretic Separation and Electrospray Ionization Mass Spectrometry. Anal. Chem. 2010, 82, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.N.; Whitesides, G.M. Soft lithography. Annu. Rev. Mater. Sci. 1998, 28, 153–184. [Google Scholar] [CrossRef]
- Unger, M.A.; Chou, H.P.; Thorsen, T.; Scherer, A.; Quake, S.R. Monolithic Microfabricated Valves and Pumps by Multilayer Soft Lithography. Science 2000, 288, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Mellors, J.S.; Gorbounov, V.; Ramsey, R.S.; Ramsey, J.M. Fully Integrated Glass Microfluidic Device for Performing High-Efficiency Capillary Electrophoresis and Electrospray Ionization Mass Spectrometry. Anal. Chem. 2008, 80, 6881–6887. [Google Scholar] [CrossRef] [PubMed]
- Vollmer, M.; Hörth, P.; Rozing, G.; Couté, Y.; Grimm, R.; Hochstrasser, D.; Sanchez, J.C. Multi-Dimensional HPLC/MS of the Nucleolar Proteome Using HPLC-chip/MS. J. Sep. Sci. 2006, 29, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Huikko, K.; Ostman, P.; Grigoras, K.; Tuomikoski, S.; Tiainen, V.M.; Soininen, A.; Puolanne, K.; Manz, A.; Franssila, S.; Kostiainen, R.; et al. Poly(dimethylsiloxane) Electrospray Devices Fabricated with Diamond-Like Carbon-Poly(dimethylsiloxane) Coated SU-8 Masters. Lab Chip 2003, 3, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Liu, H.G.; Jiang, Y.Y.; Lin, J.M. Recent Advances in Microfluidics Combined with Mass Spectrometry: Technologies and Applications. Lab Chip 2013, 13, 3309–3322. [Google Scholar] [CrossRef] [PubMed]
- Oh, K.W.; Lee, K.; Ahn, B.; Furlani, E.P. Design of Pressure-Driven Microfluidic Networks Using Electric Circuit Analogy. Lab Chip 2012, 12, 515–545. [Google Scholar] [CrossRef] [PubMed]
- Brantom, P.G. Review of the Toxicology of a Number of Dyes Illegally Present in Food in the EU. EFSA J. 2005, 263, 1–71. [Google Scholar]
- Pei, J.; Li, Q.; Lee, M.S.; Valaskovic, G.A.; Kennedy, R.T. Analysis of Samples Stored as Individual Plugs in a Capillary by Electrospray Ionization Mass Spectrometry. Anal. Chem. 2009, 81, 6558–6561. [Google Scholar] [CrossRef] [PubMed]
- White, T.P.; Wood, T.D. Reproducibility in Fabrication and Analytical Performance of Polyaniline-Coated Nanoelectrospray Emitters. Anal. Chem. 2003, 75, 3660–3665. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Karas, M.; Dulcks, T. Effect of Different Solution Flow Rates on Analyte Ion Signals in Nano-ESI MS, or: When does ESI Turn into Nano-ESI? J. Am. Soc. Mass Spectrom. 2003, 14, 492–500. [Google Scholar] [PubMed]
- Wang, J.D.; Douville, N.J.; Takayama, S.; ElSayed, M. Quantitative Analysis of Molecular Absorption into PDMS Microfluidic Channels. Ann. Biomed. Eng. 2012, 40, 1862–1873. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qian, X.; Xu, J.; Yu, C.; Chen, Y.; Yu, Q.; Ni, K.; Wang, X. A Reliable and Simple Method for Fabricating a Poly(Dimethylsiloxane) Electrospray Ionization Chip with a Corner-Integrated Emitter. Sensors 2015, 15, 8931-8944. https://doi.org/10.3390/s150408931
Qian X, Xu J, Yu C, Chen Y, Yu Q, Ni K, Wang X. A Reliable and Simple Method for Fabricating a Poly(Dimethylsiloxane) Electrospray Ionization Chip with a Corner-Integrated Emitter. Sensors. 2015; 15(4):8931-8944. https://doi.org/10.3390/s150408931
Chicago/Turabian StyleQian, Xiang, Jie Xu, Cilong Yu, Yan Chen, Quan Yu, Kai Ni, and Xiaohao Wang. 2015. "A Reliable and Simple Method for Fabricating a Poly(Dimethylsiloxane) Electrospray Ionization Chip with a Corner-Integrated Emitter" Sensors 15, no. 4: 8931-8944. https://doi.org/10.3390/s150408931
APA StyleQian, X., Xu, J., Yu, C., Chen, Y., Yu, Q., Ni, K., & Wang, X. (2015). A Reliable and Simple Method for Fabricating a Poly(Dimethylsiloxane) Electrospray Ionization Chip with a Corner-Integrated Emitter. Sensors, 15(4), 8931-8944. https://doi.org/10.3390/s150408931






