Development of a Microfluidic-Based Optical Sensing Device for Label-Free Detection of Circulating Tumor Cells (CTCs) Through Their Lactic Acid Metabolism
Abstract
:1. Introduction
2. Experimental Section
2.1. Design of Microfluidic Device
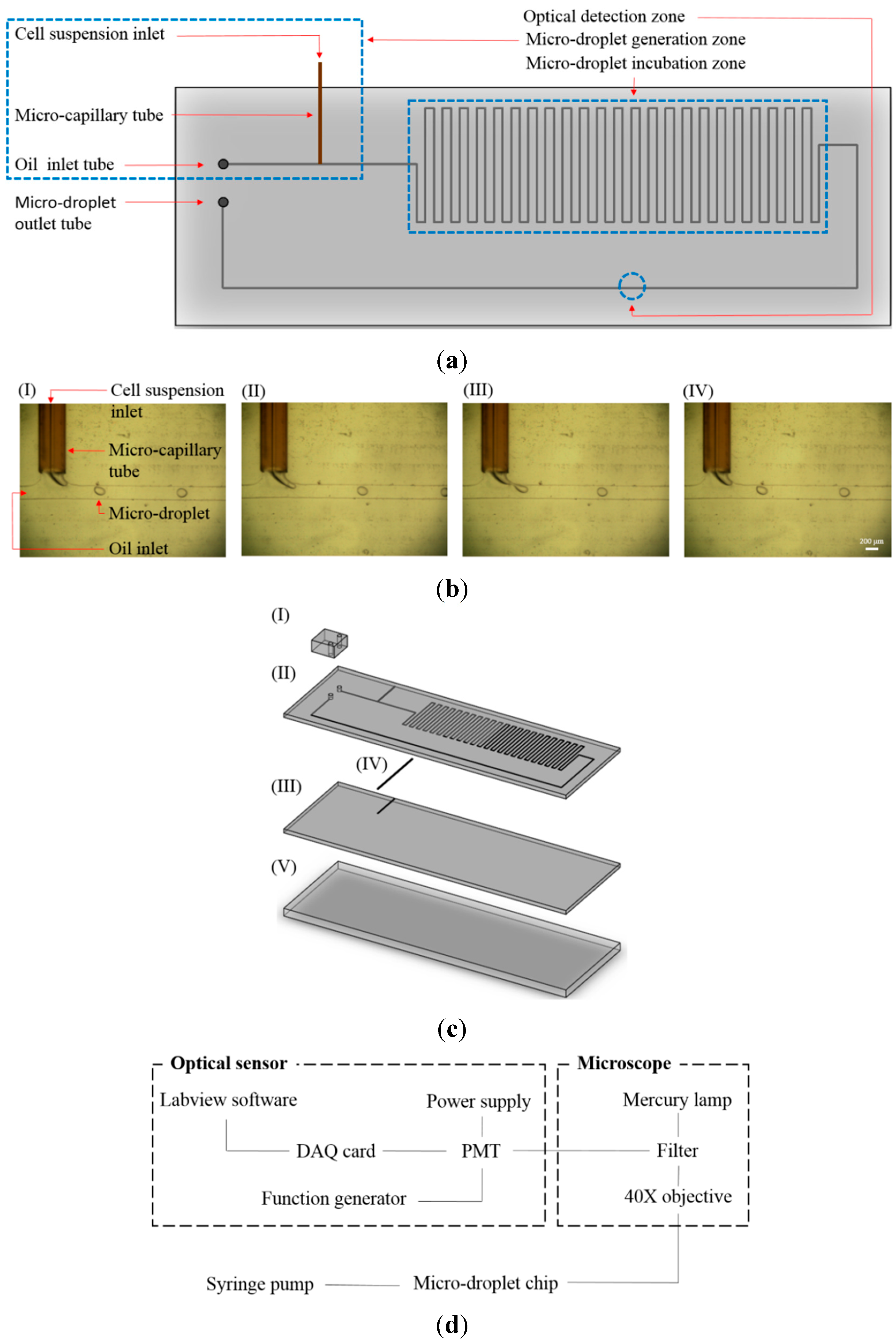

2.2. Fabrication and Experimental Setup
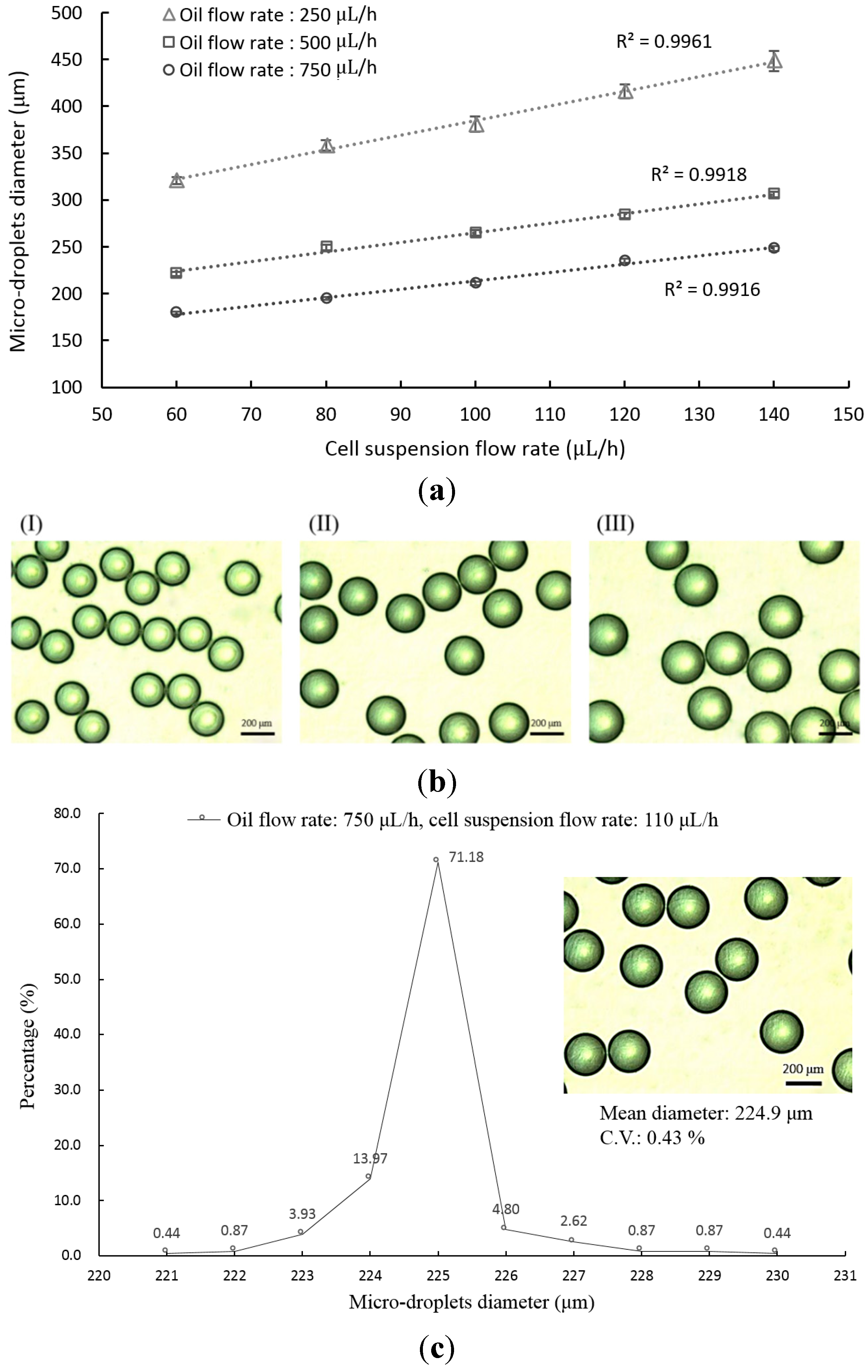
2.3. Evaluation of Size and Uniformity of Micro-Droplets
2.4. Evaluation of Cell Viability
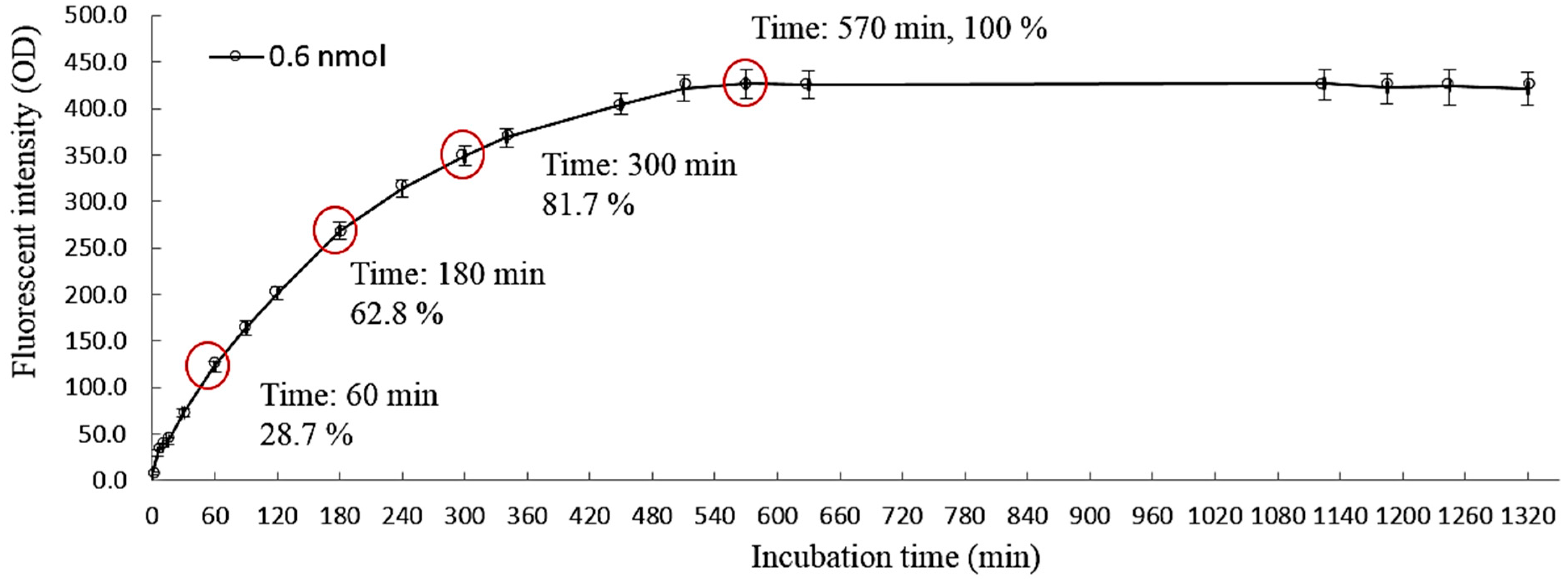
2.5. Quantification of the Live Cancer Cells through Fluorescence-Based Optical Detection of Lactic Acid
2.6. Statistical Analysis
3. Results and Discussion
3.1. The Application of the Proposed Microfluidic Device for Micro-Droplet Generation and Microencapsulation of Cells
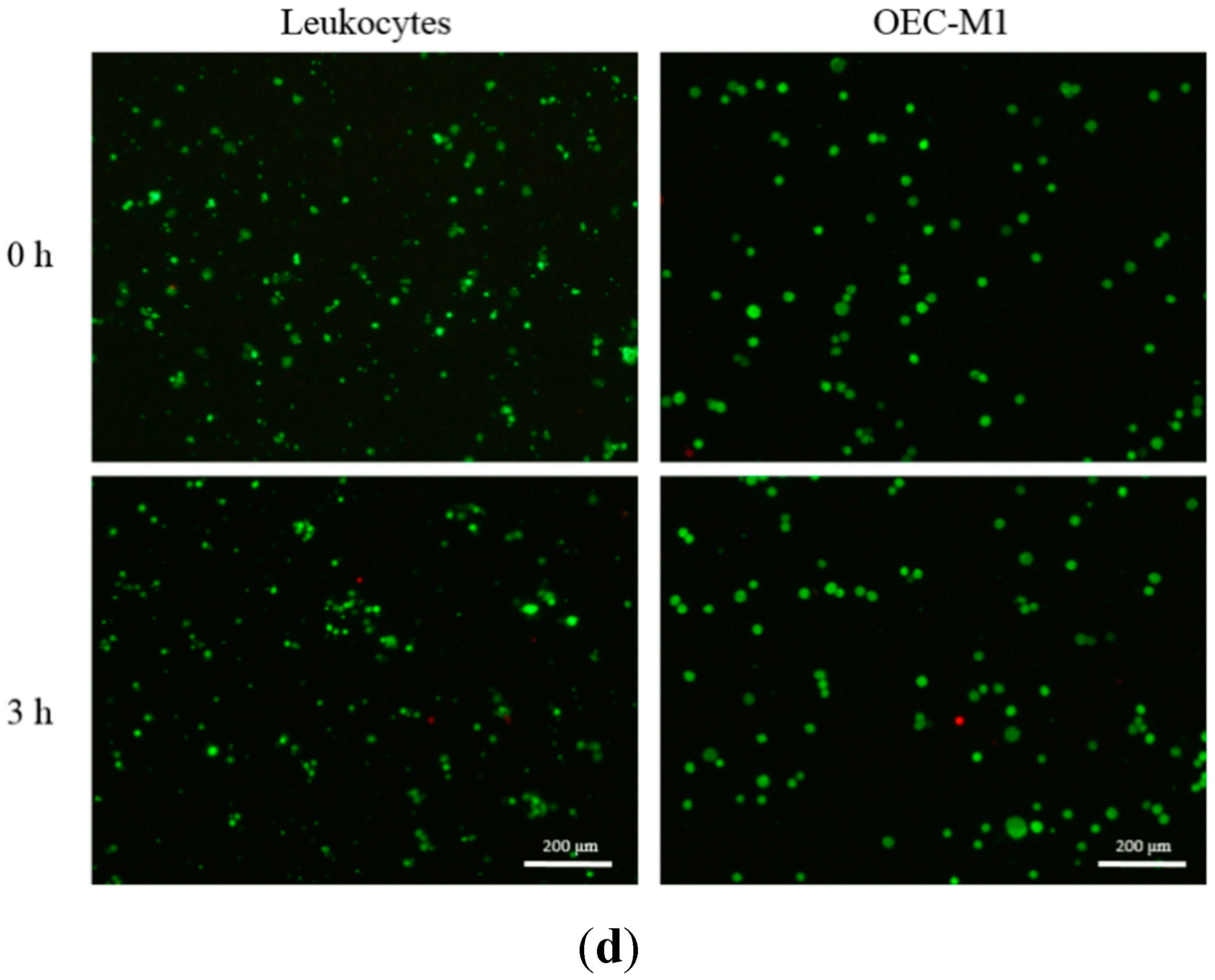
3.2. The Feasibility of Using the Proposed Microfluidic Optical Sensing Device for Live Cancer Cell Detection
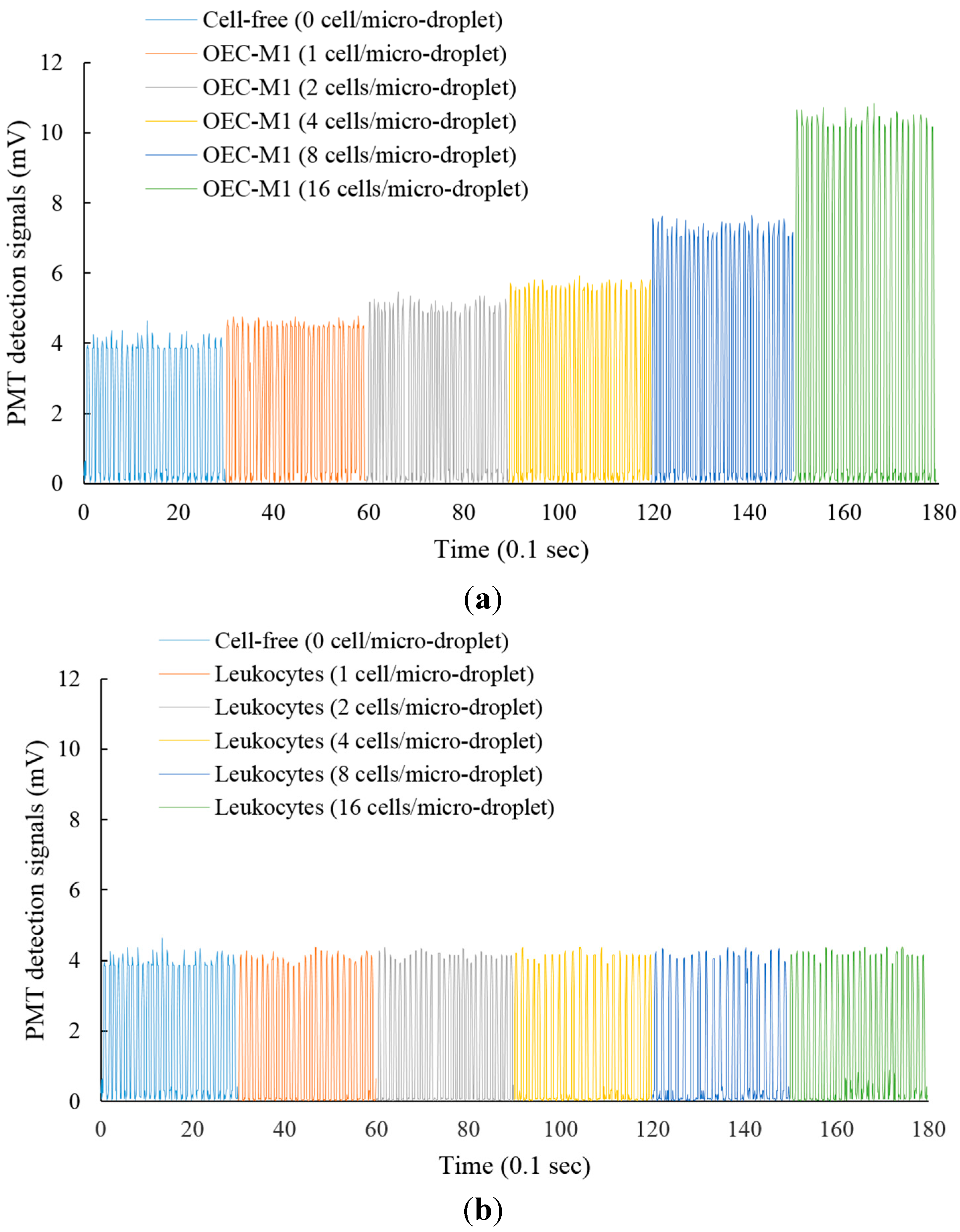
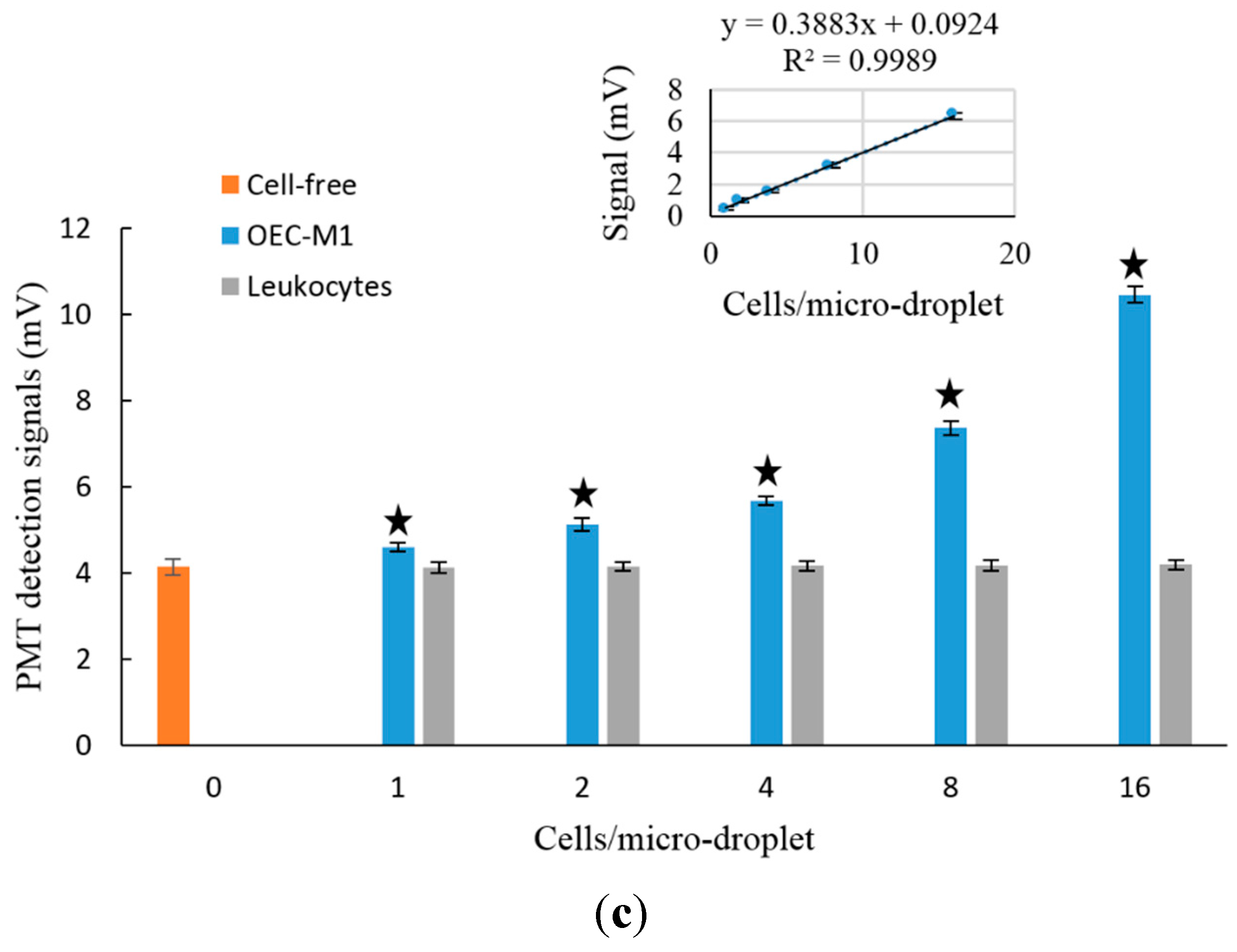
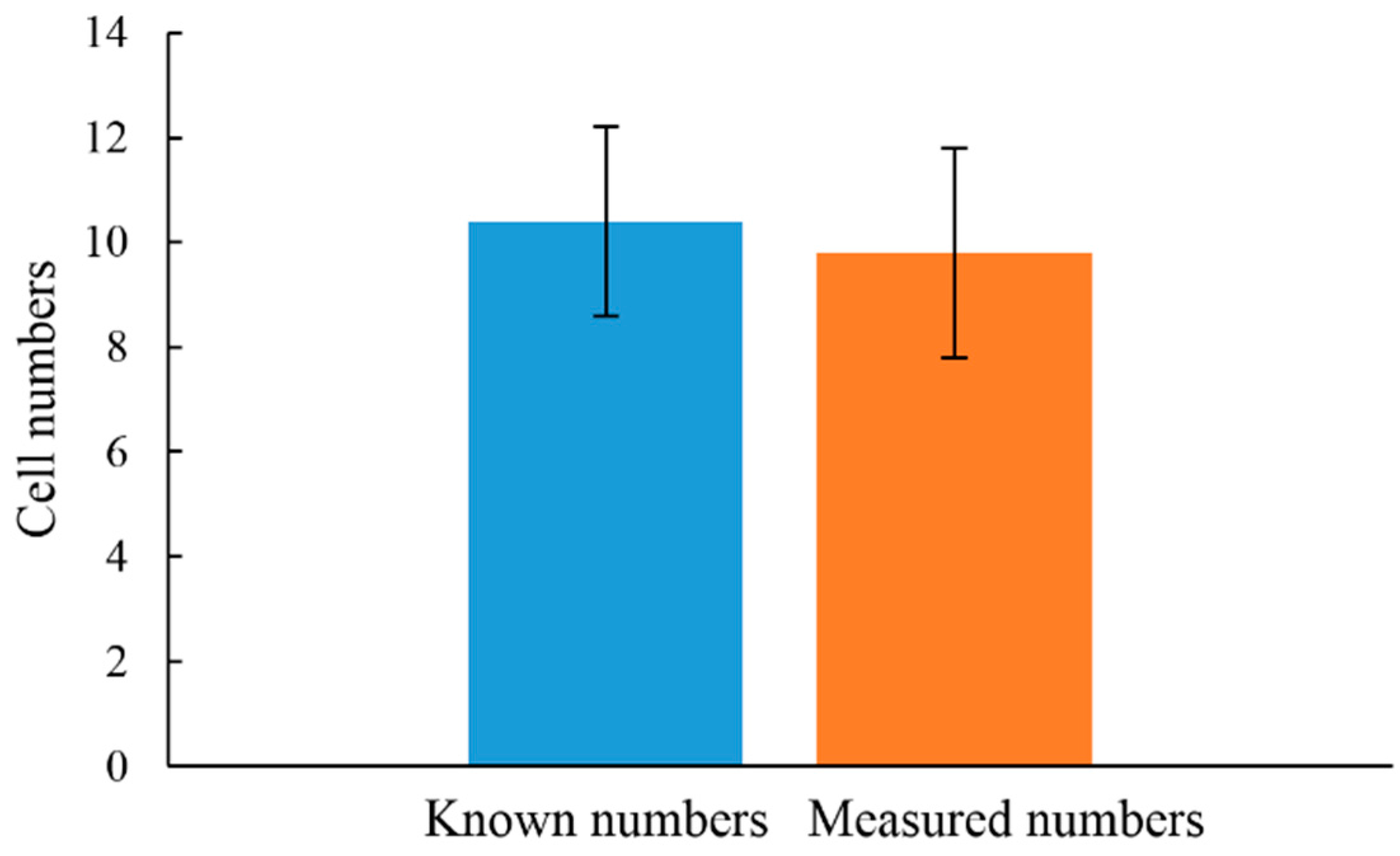
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Helling, T.S.; Martin, M. Cause of Death from Liver Metastases in Colorectal Cancer. Ann. Surg. Oncol. 2014, 21, 501–506. [Google Scholar] [CrossRef]
- Ashworth, T.R. A case of cancer in which cells similar to those in the tumours were seen in the blood after death. Aust. Med. J. 1869, 14, 146–149. [Google Scholar]
- Sinha, G. Circulating tumor cells in early-stage breast cancer. J. Natl. Cancer Inst. 2012, 104, 1693–1694. [Google Scholar] [CrossRef]
- Dawood, S.; Broglio, K.; Valero, V. Circulating tumor cells in metastatic breast cancer: From prognostic stratification to modification of the staging system? Cancer 2008, 113, 2422–2430. [Google Scholar] [CrossRef]
- Nesteruk, D.; Rutkowski, A.; Fabisiewicz, S.; Pawlak, J.; Siedlecki, J.A.; Fabisiewicz, A. Evaluation of prognostic significance of circulating tumor cells detection in rectal cancer patients treated with preoperative radiotherapy: Prospectively collected material data. BioMed Res. Int. 2014. [Google Scholar] [CrossRef]
- Li, M.; Zhang, B.; Zhang, Z.; Liu, X.; Qi, X.; Zhao, J.; Jiang, Y.; Zhai, H.; Ji, Y.; Luo, D. Stem cell-like circulating tumor cells indicate poor prognosis in gastric cancer. BioMed Res. Int. 2014. [Google Scholar] [CrossRef]
- Jiang, Z.F.; Cristofanilli, M.; Shao, Z.M.; Tong, Z.S.; Song, E.W.; Wang, X.J.; Liao, N.; Hu, X.C.; Liu, Y.; Wang, Y.; et al. Circulating tumor cells predict progression-free and overall survival in Chinese patients with metastatic breast cancer, HER2-positive or triple-negative (CBCSG004): A multicenter, double-blind, prospective trial. Ann. Oncol. 2013, 24, 2766–2772. [Google Scholar] [CrossRef]
- Smerage, J.B.; Barlow, W.E.; Hortobagyi, G.N.; Winer, E.P.; Jones, B.L.; Srkalovic, G.; Tejwani, S.; Schott, A.F.; O’Rourke, M.A.; Lew, D.L.; et al. Circulating Tumor Cells and Response to Chemotherapy in Metastatic Breast Cancer: SWOG S0500. J. Clin. Oncol. 2014. [Google Scholar] [CrossRef]
- Liotta, L.A.; Kleinerman, J.; Saidel, G.M. The significance of hematogenous tumor cell clumps in the metastatic process. Cancer Res. 1976, 36, 889–894. [Google Scholar]
- Yu, M.; Stott, S.; Toner, M.; Maheswaran, S.; Haber, D.A. Circulating tumor cells: Approaches to isolation and characterization. J. Cell Biol. 2011, 192, 373–382. [Google Scholar] [CrossRef]
- Yusa, A.; Toneri, M.; Masuda, T.; Ito, S.; Yamamoto, S.; Okochi, M.; Kondo, N.; Iwata, H.; Yatabe, Y.; Ichinosawa, Y.; et al. Development of a New Rapid Isolation Device for Circulating Tumor Cells (CTCs) Using 3D Palladium Filter and Its Application for Genetic Analysis. PLoS One 2014. [Google Scholar] [CrossRef]
- Huang, S.B.; Wu, M.H.; Lin, Y.H.; Hsieh, C.H.; Yang, C.L.; Lin, H.C.; Tseng, C.P.; Lee, G.B. High purity and label free isolation of circulating tumor cells (CTCs) in a microfluidic platform by using optically-induced-dielectrophoretic (ODEP) force. Lab Chip 2013, 13, 1371–1383. [Google Scholar] [CrossRef]
- Mohamed, H.; Murray, M.; Turner, J.N.; Caggana, M. Isolation of tumor cells using size and deformation. J. Chromatogr. A 2009, 1216, 8289–8295. [Google Scholar] [CrossRef]
- Li, J.; Chen, L.; Zhang, X.; Zhang, Y.; Liu, H.; Sun, B.; Zhao, L.; Ge, N.; Qian, H.; Yang, Y.; et al. Detection of Circulating Tumor Cells in Hepatocellular Carcinoma Using Antibodies against Asialoglycoprotein Receptor, Carbamoyl Phosphate Synthetase 1 and Pan-Cytokeratin. PLoS One 2014. [Google Scholar] [CrossRef]
- Coumans, F.A.W.; Dalum, G.V.; Beck, M.; Terstappen, L.W.M.M. Filter Characteristics Influencing Circulating Tumor Cell Enrichment from Whole Blood. PLoS One 2013. [Google Scholar] [CrossRef]
- Mikolajczyk, S.D.; Millar, L.S.; Tsinberg, P.; Coutts, S.M; Zomorrodi, M.; Pham, T.; Bischoff, F.Z.; Pircher, T.J. Detection of EpCAM-negative and cytokeratin-negative circulating tumor cells in peripheral blood. J. Oncol. 2011. [Google Scholar] [CrossRef]
- Bauer, S.K.; Hoffmann, O.; Wallwiener, D.; Kimmig, R.; Fehm, T. Expression of stem cell and epithelial-mesenchymal transition markers in primary breast cancer patients with circulating tumor cells. Breast Cancer Res. 2012, 14. [Google Scholar] [CrossRef]
- Danila, D.C.; Heller, G.; Gignac, G.A.; Gonzalez-Espinoza, R.; Anand, A.; Tanaka, E.; Lilja, H.; Schwartz, L.; Larson, S.; Fleisher, M.; et al. Circulating tumor cell number and prognosis in progressive castration-resistant prostate cancer. Clin. Cancer Res. 2007, 13, 7053–7058. [Google Scholar] [CrossRef]
- Lambrechts, A.C.; Bosma, A.J.; Klaver, S.G.; Top, B.; Perebolte, L.; van’t Veer, L.J.; Rodenhuis, S. Comparison of immunocytochemistry, reverse transcriptase polymerase chain reaction, and nucleic acid sequence-based amplification for the detection of circulating breast cancer cells. Breast Cancer Res. Treat 1999, 46, 219–231. [Google Scholar]
- Lin, H.C.; Hsieh, C.H.; Hsu, H.C.; Wu, M.H.; Tseng, C.P. A negative selection system PowerMag for effective leukocyte depletion and enhanced detection of EpCAM positive and negative circulating tumor cells. Clin. Chim. Acta 2013, 419, 77–84. [Google Scholar] [CrossRef]
- Nagrath, S.; Sequist, L.V.; Maheswaran, S.; Bell, D.W.; Irimia, D.; Ulkus, L.; Smith, M.R.; Kwak, E.L.; Digumarthy, S.; Muzikansky, A.; et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 2007, 450, 1235–1239. [Google Scholar] [CrossRef]
- Gatenby, R.A.; Gillies, R.J. Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 2004, 4, 891–899. [Google Scholar] [CrossRef]
- Kim, J.W.; Dang, C.V. Cancer’s molecular sweet tooth and the Warburg effect. Cancer Res. 2006, 66, 8927–8930. [Google Scholar] [CrossRef] [PubMed]
- Vladisavljevic, G.T.; Kobayashi, I.; Nakajima, M. Production of uniform droplets using membrane, microchannel and microfluidic emulsification devices. Microfluid. Nanofluid. 2012, 13, 151–178. [Google Scholar] [CrossRef]
- Edd, J.F.; Carlo, D.D.; Humphry, K.J.; Koster, S.; Irimia, D.; Weitz, D.A.; Toner, M. Controlled encapsulation of single-cells into monodisperse picolitre drops. Lab Chip 2008, 8, 1262–1264. [Google Scholar] [CrossRef]
- Huebner, A.; Srisa-Art, M.; Holt, D.; Abell, C.; Hollfelder, F.; deMello, A.J.; Edel, J.B. Quantitative detection of protein expression in single cells using droplet microfluidics. Chem. Commun. 2007. [Google Scholar] [CrossRef]
- Zhu, Y.; Fang, Q. Analytical detection techniques for droplet microfluidics—A review. Anal. Chim. Acta 2013, 787, 24–35. [Google Scholar] [CrossRef]
- Joensson, H.N.; Svahn, H.A. Droplet Microfluidics—A Tool for Single-Cell Analysis. Angew. Chem. Int. Ed. 2012, 51, 12176–12192. [Google Scholar] [CrossRef]
- Wu, M.H.; Wang, H.Y.; Tai, C.L.; Chang, Y.H.; Chen, Y.M.; Huang, S.B.; Chiu, T.K.; Yang, T.C.; Wang, S.S. Development of perfusion-based microbioreactor platform capable of providing tunable dynamic compressive loading to 3-D cell culture construct: Demonstration study of the effect of compressive stimulations on articular chondrocyte functions. Sens. Actuators B Chem. 2013, 176, 86–96. [Google Scholar] [CrossRef]
- Huang, S.B.; Chang, Y.H.; Lee, H.C.; Tsai, S.W.; Wu, M.H. A pneumatically-driven microfluidic system for size-tunable generation of uniform cell-encapsulating collagen microbeads with the ultrastructure similar to native collagen. Biomed. Microdevices 2014, 16, 345–354. [Google Scholar] [CrossRef]
- Kobayashi, I.; Vladisavljevic, G.T.; Uemura, K.; Nakajima, M. CFD analysis of microchannel emulsification: Droplet generation process and size effect of asymmetric straight flow-through microchannels. Chem. Eng. Sci. 2011, 66, 5556–5565. [Google Scholar]
- Lin, Y.S.; Yang, C.H.; Wang, C.Y.; Chang, F.R.; Huang, K.S.; Hsieh, W.C. An Aluminum Microfluidic Chip Fabrication Using a Convenient Micromilling Process for Fluorescent Poly(DL-lactide-co-glycolide) Microparticle Generation. Sensors 2012, 12, 1455–1467. [Google Scholar] [CrossRef]
- Dendukuri, D.; Tsoi, K.; Hatton, T.A.; Doyle, P.S. Controlled synthesis of nonspherical microparticles using microfluidics. Langmuir 2005, 21, 2113–2116. [Google Scholar] [CrossRef] [PubMed]
- Steegmans, M.L.J.; Schroën, K.G.P.H.; Boom, R.M. Characterization of emulsification at flat microchannel Y junctions. Langmuir 2009, 25, 3396–3401. [Google Scholar] [CrossRef]
- Kim, B.I.; Jeong, S.W.; Lee, K.G.; Park, T.J.; Park, J.Y.; Song, J.J.; Lee, S.J.; Lee, C.S. Synthesis of bioactive microcapsules using a microfluidic Device. Sensors 2012, 12, 10136–10147. [Google Scholar] [CrossRef] [PubMed]
- Anna, S.L.; Bontoux, N.; Stone, H.A. Formation of dispersions using ‘‘flow focusing’’ in microchannels. Appl. Phys. Lett. 2003, 82, 364–366. [Google Scholar] [CrossRef]
- Brouzes, E.; Medkova, M.; Savenelli, N.; Marran, D.; Twardowski, M.; Hutchison, J.B.; Rothberg, J.M.; Link, D.R.; Perrimon, N.; Samuels, M.L. Droplet microfluidic technology for single-cell high-throughput screening. Proc. Natl. Acad. Sci. 2009, 106, 14195–14200. [Google Scholar] [CrossRef]
- Griffiths, A.D.; Tawfik, D.S. Miniaturising the laboratory in emulsion droplets. Trends Biotechnol. 2006, 24, 395–405. [Google Scholar]
- Novak, R.; Zeng, Y.; Shuga, J.; Venugopalan, G.; Fletcher, D.A.; Smith, M.T.; Mathies, R.A. Single cell multiplex gene detection and sequencing using microfluidically-generated agarose emulsions. Angew. Chem. 2011, 50, 390–395. [Google Scholar] [CrossRef]
- Thorsen, T.; Roberts, R.W.; Arnold, F.H.; Quake, S.R. Dynamic pattern formation in a vesicle-generating microfluidic device. Phys. Rev. Lett. 2001, 86, 4163–4166. [Google Scholar] [CrossRef] [PubMed]
- Link, D.R.; Anna, S.L.; Weitz, D.A.; Stone, H.A. Geometrically mediated breakup of drops in microfluidic devices. Phys. Rev. Lett. 2004, 92. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Edgar, J.S.; Jeffries, G.D.M.; Lorenz, R.M.; Shelby, J.P.; Chiu, D.T. Selective encapsulation of single cells and subcellular organelles into picoliter- and femtoliter-volume droplets. Anal. Chem. 2005, 77, 1539–1544. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Huang, Y.; Grujicic, M.; Chrisey, D.B. Study of impact-induced mechanical effects in cell direct writing using smooth particle hydrodynamic method. J. Manuf. Sci. Eng. 2008, 130. [Google Scholar] [CrossRef]
- Tirella, A.; Vozzi, F.; Maria, C.D.; Vozzi, G.; Sandri, T.; Sassano, D.; Cognolato, L.; Ahluwalia, A. Substrate stiffness influences high resolution printing of living cells with an ink-jet system. J. Biosci. Bioeng. 2011, 112, 79–85. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, T.-K.; Lei, K.-F.; Hsieh, C.-H.; Hsiao, H.-B.; Wang, H.-M.; Wu, M.-H. Development of a Microfluidic-Based Optical Sensing Device for Label-Free Detection of Circulating Tumor Cells (CTCs) Through Their Lactic Acid Metabolism. Sensors 2015, 15, 6789-6806. https://doi.org/10.3390/s150306789
Chiu T-K, Lei K-F, Hsieh C-H, Hsiao H-B, Wang H-M, Wu M-H. Development of a Microfluidic-Based Optical Sensing Device for Label-Free Detection of Circulating Tumor Cells (CTCs) Through Their Lactic Acid Metabolism. Sensors. 2015; 15(3):6789-6806. https://doi.org/10.3390/s150306789
Chicago/Turabian StyleChiu, Tzu-Keng, Kin-Fong Lei, Chia-Hsun Hsieh, Hung-Bo Hsiao, Hung-Ming Wang, and Min-Hsien Wu. 2015. "Development of a Microfluidic-Based Optical Sensing Device for Label-Free Detection of Circulating Tumor Cells (CTCs) Through Their Lactic Acid Metabolism" Sensors 15, no. 3: 6789-6806. https://doi.org/10.3390/s150306789
APA StyleChiu, T.-K., Lei, K.-F., Hsieh, C.-H., Hsiao, H.-B., Wang, H.-M., & Wu, M.-H. (2015). Development of a Microfluidic-Based Optical Sensing Device for Label-Free Detection of Circulating Tumor Cells (CTCs) Through Their Lactic Acid Metabolism. Sensors, 15(3), 6789-6806. https://doi.org/10.3390/s150306789






