Manganese(III) Porphyrin-based Potentiometric Sensors for Diclofenac Assay in Pharmaceutical Preparations
Abstract
:1. Introduction
2. Experimental Section
Reagents
Membrane preparation and electrode construction
Apparatus and electrodes
Sample preparation
3. Results and Discussion
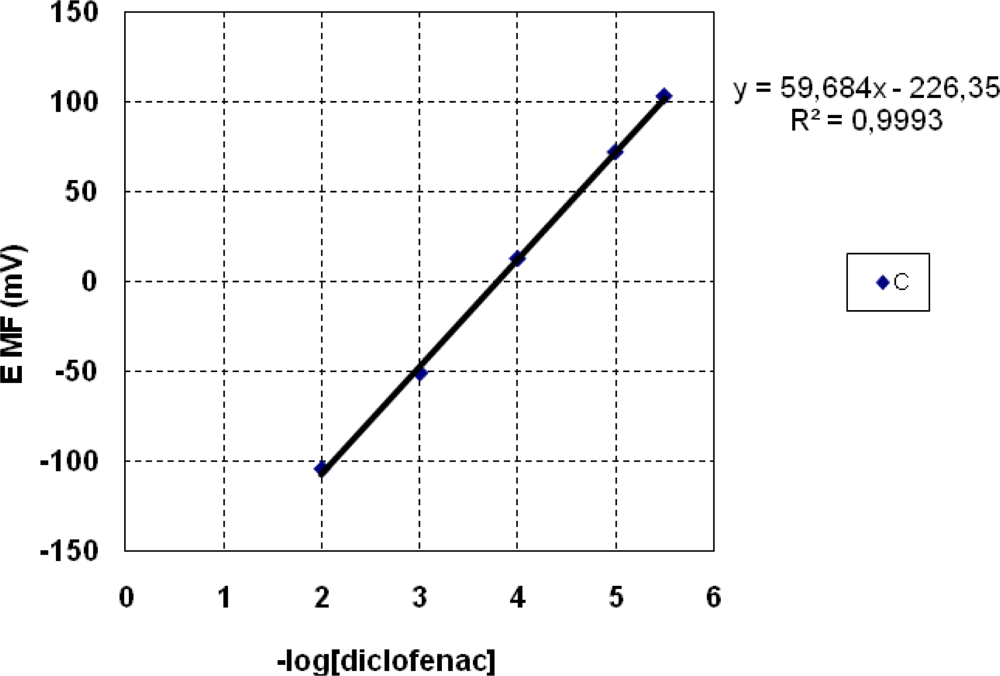
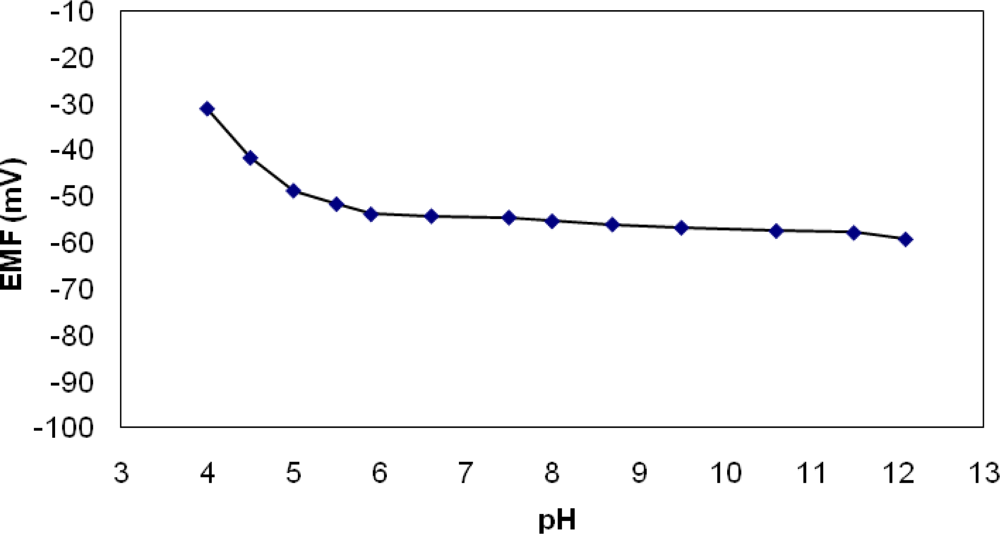
3.1. Potentiometric Measurements
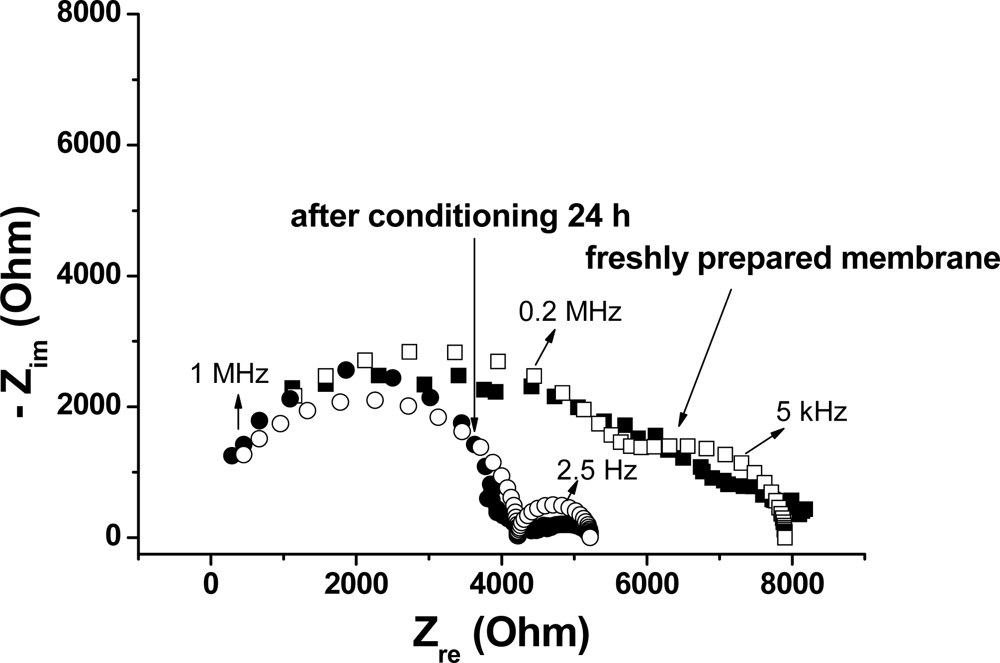
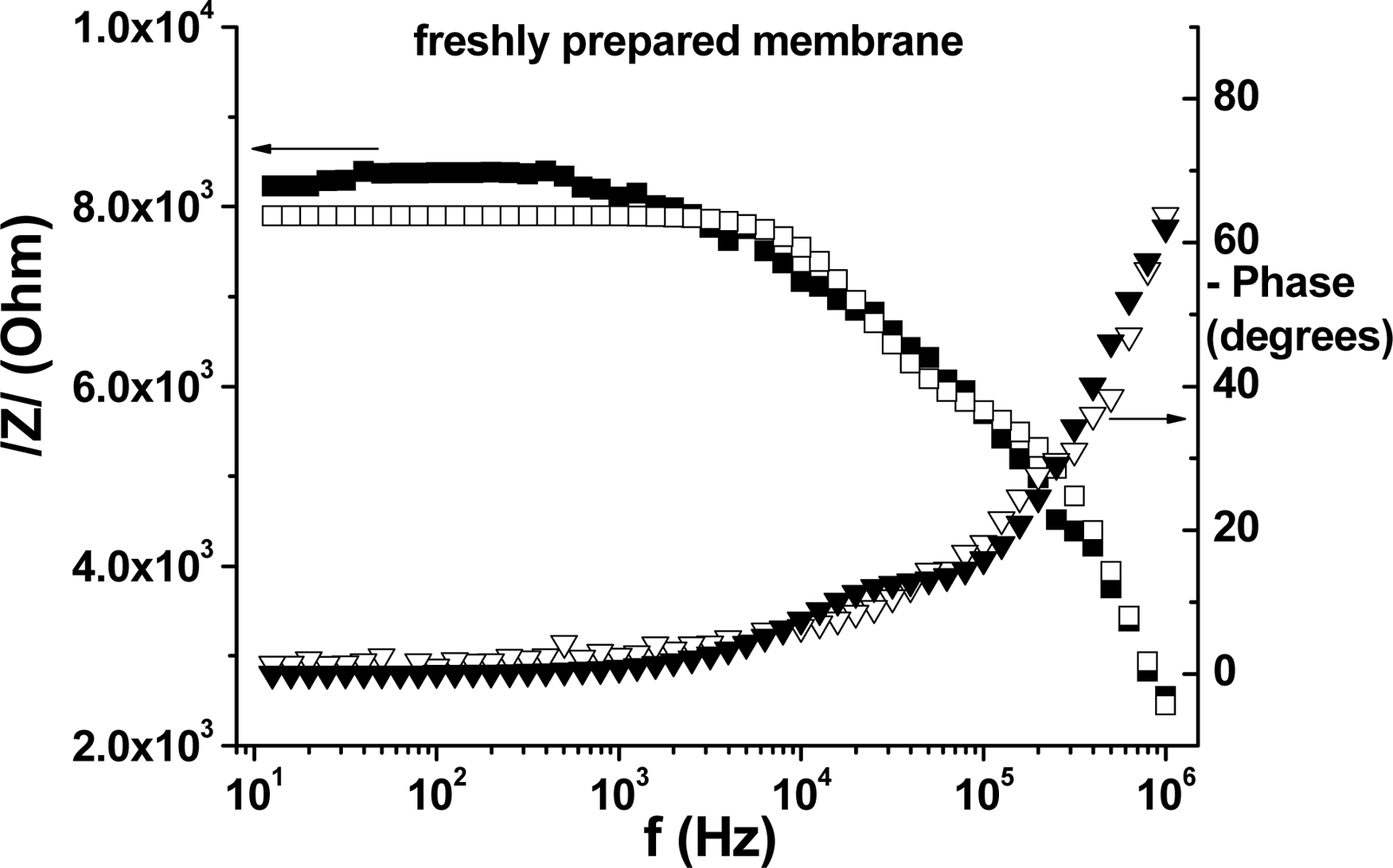
3.2. Electrochemical Impedance Spectroscopy (EIS)
3.3. Analytical Applications
4. Conclusions
Acknowledgments
References and Notes
- Gupta, VK; Jain, AK; Maheshwari, G; Lang, H; Ishtaiwi, Z. Copper(II)-selective potentiometric sensors based on porphyrins in PVC matrix. Sens. Actuat. B 2006, 117, 99–106. [Google Scholar]
- Gupta, VK; Chauhan, DK; Saini, VK; Agarwal, S; Antonijevic, MM; Lang, H. A porphyrin based potentiometric sensor for Zn2+ determination. Sensors 2003, 3, 223–235. [Google Scholar]
- Vlascici, D; Făgădar-Cosma, E; Bizerea Spiridon, O. A New Composition for Co(II)-porphyrin-based membranes used in thiocyanate-selective electrodes. Sensors 2006, 6, 892–900. [Google Scholar]
- Vlascici, D; Fagadar, CE; Pica, EM; Cosma, V; Bizerea, O; Mihailescu, G; Olenic, L. Free base porphyrins as ionophores for heavy metal sensors. Sensors 2008, 8, 4995–5004. [Google Scholar]
- Vlascici, D; Bizerea Spiridon, O; Făgădar-Cosma, E. Thiocyanate-selective electrode based on rhodium porphyrin derivatives. J. Optoelectron. Adv. Mat 2006, 8, 883–887. [Google Scholar]
- Suzuki, H; Nakagawa, H; Mifune, M; Saita, Y. Triiodide ion-selective electrode based on manganese(III)-tetraphenylporphine. Anal. Sci 1993, 9, 351–354. [Google Scholar]
- Khorasani, JH; Amini, MK; Motaghi, H; Tangestaninejad, S; Moghadam, M. Manganese porphyrin derivatives as ionophores for thiocyanate-selective electrodes; the influence of porphyrin substituents and additives on the response properties. Sens. Act. B 2002, 87, 448–456. [Google Scholar]
- Santos, EMG; Araújo, AN; Couto, CMCM; Montenegro, MCBSM; Kejzlarova, A; Solich, P. Ion selective electrodes for penicillin-G based on Mn(III)TPP-Cl and their application in pharmaceutical formulations control by sequential injection analysis. J. Pharm. Biomed. Anal 2004, 36, 701–709. [Google Scholar]
- Santos, EMG; Araújo, AN; Couto, CMCM; Montenegro, MCBSM. Construction and evaluation of PVC and sol-gel sensor membranes based on Mn(III)TPP-Cl. Application to valproate determination in pharmaceutical preparations. Anal. Bioanal. Chem 2006, 384, 867–875. [Google Scholar]
- Saraswathyamma, B; Pajak, M; Radecki, J; Maes, W; Dehaen, W; Kumar, KG; Radecka, H. PVC supported liquid membrane and carbon paste potentiometric sensors incorporating a Mn(III)-porphyrin for the direct determination of undissociated paracetamol. Electroanalysis 2008, 20, 2009–2015. [Google Scholar]
- Agrawal, YK; Shivramchandra, K. Spectrophotometric determination of diclofenac sodium in tablets. J. Pharm. Biomed. Anal 1991, 9, 97–100. [Google Scholar]
- Arancibia, JA; Boldrini, MA; Escandar, GM. Spectrofluorimetric determination of diclofenac in the presence of α-cyclodextrin. Talanta 2000, 52, 261–268. [Google Scholar]
- Kasperek, R. Determination of diclofenac sodium and papaverine hydrochloride in tablets by HPLC method. Acta Pol. Pharm 2008, 65, 403–408. [Google Scholar]
- Kormosh, Z; Hunka, I; Bazel, Y; Kormosh, N; Laganovski, A; Mazurenko, I. A new diclofenac membrane sensor based on its ion associate with crystal violet. Application to diclofenac determination in urine and pharmaceuticals. J. Iran. Chem. Soc 2007, 4, 408–413. [Google Scholar]
- Hassan, SSM; Mahmoud, WH; Elmosallamy, MAF; Almarzooqi, MH. Iron(II)-phtalocyanine as a novel recognition sensor for selective potentiometric determination of diclofenac and warfarin drugs. J. Pharm. Biomed. Anal 2005, 39, 315–321. [Google Scholar]
- Shamsipur, M; Jalali, F; Ershad, S. Preparation of a diclofenac potentiometric sensor and its application to pharmaceutical analysis and to drug recovery from biological fluids. J. Pharm. Biomed. Anal 2005, 37, 943–947. [Google Scholar]
- Santini, AO; Pezza, HR; Pezza, L. Determination of diclofenac in pharmaceutical preparations using a potentiometric sensor immobilized in a graphite matrix. Talanta 2006, 68, 636–642. [Google Scholar]
- Kormosh, Z; Hunka, I; Bazel, Y; Laganovski, A; Mazurenko, I; Kormosh, N. Determination of diclofenac in pharmaceuticals and urine samples using a membrane sensor based on the ion associate of diclofenac with Rhodamine B. Cent. Eur. J. Chem 2007, 5, 813–823. [Google Scholar]
- Santos, EMG; Araújo, AN; Couto, CMCM; Montenegro, MCBSM. Potentiometric behaviour of ion selective electrodes based on iron porphyrins: The influence of porphyrin substituents on the response properties and analytical determination of diclofenac in pharmaceutical formulations. J. Pharm. Biomed. Anal 2006, 42, 535–542. [Google Scholar]
- Fagadar-Cosma, E; Mirica, M; Balcu, I; Bucovicean, C; Cretu, C; Armeanu, I; Fagadar-Cosma, G. Syntheses, Spectroscopic and AFM characterization of some manganese porphyrins and their Hybrid Silica Nanomaterials. Molecules 2009, 14, 1370–1388. [Google Scholar]
- Umezawa, Y; Buhlmann, P; Umezawa, K; Tohda, K. Potentiometric selectivity coefficients of ion-selective electrodes. Part I Inorganic cations. Pure Appl. Chem 2000, 72, 1851–2082. [Google Scholar]
- Egorov, VV; Rakhman’ko, EM; Rat’ko, AA. Metalloporphyrin-based anion-selective electrodes with unusual selectivity. J. Anal. Chem 2002, 57, 46–53. [Google Scholar]
- Horvai, G. Relationship between charge transfer resistance and exchange current density of ion transfer at the interface of two immiscible electrolyte solutions. Electroanalysis 1991, 3, 673–675. [Google Scholar]
- Armstrong, RD; Lockhart, JC; Todd, M. The mechanism of transfer of K+ between aqueous solutions and PVC membranes containing valinomycin. Electrochim. Acta 1986, 31, 591–594. [Google Scholar]
- Armstrong, RD; Todd, M. Exchange of K+ and Na+ between water and PVC in the presence of different ionophores. J. Electroanal. Chem 1988, 248, 131–138. [Google Scholar]
- Calvo, D; Bartroli, J; del Valle, M. EIS study of potentiometric membranes selective to Ca2+ employing the new ionophoric antibiotic tetronasin. Electrochim. Acta 2006, 51, 1569–1575. [Google Scholar]
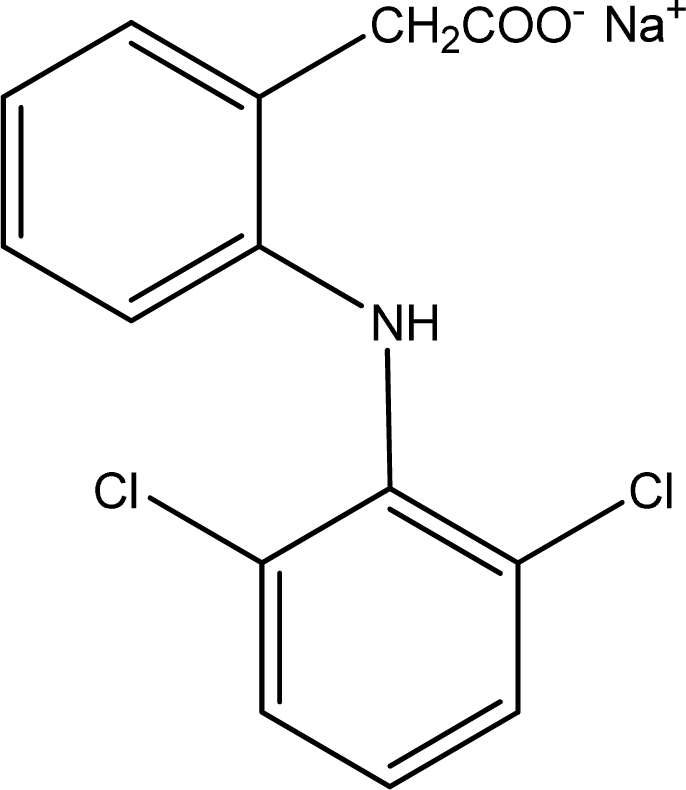
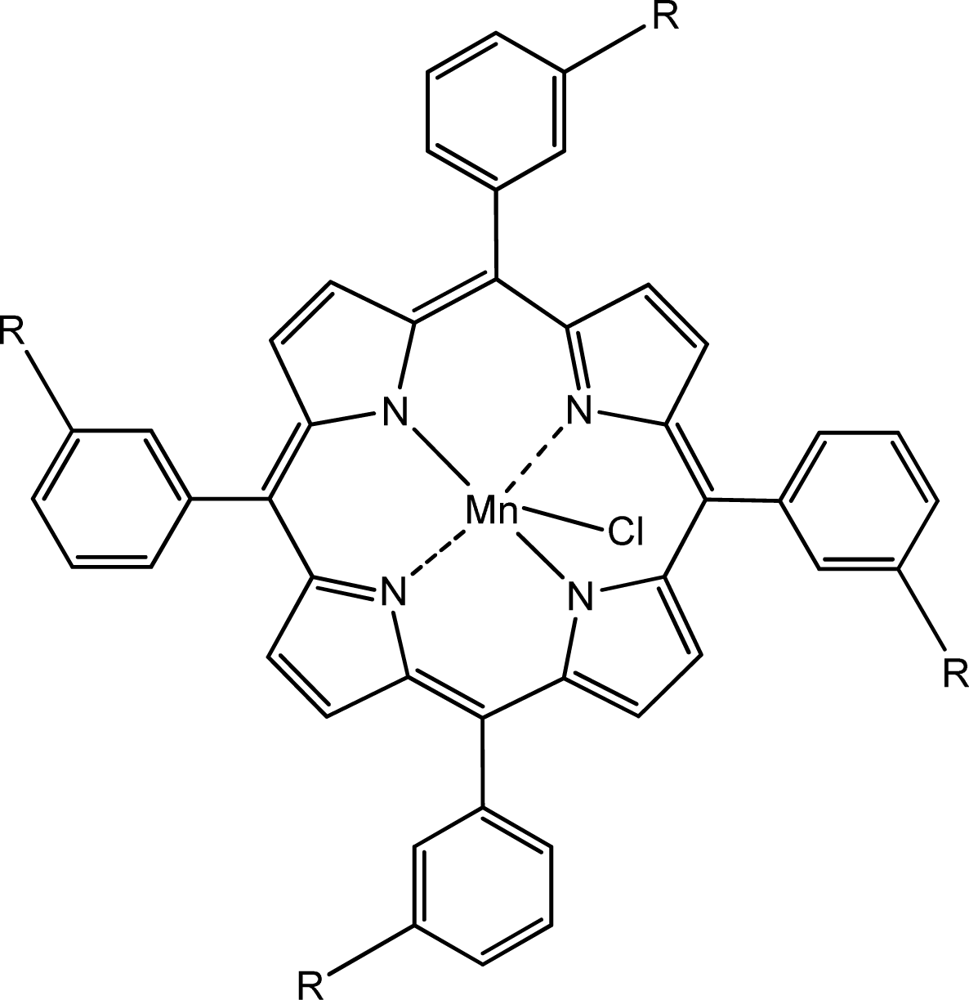
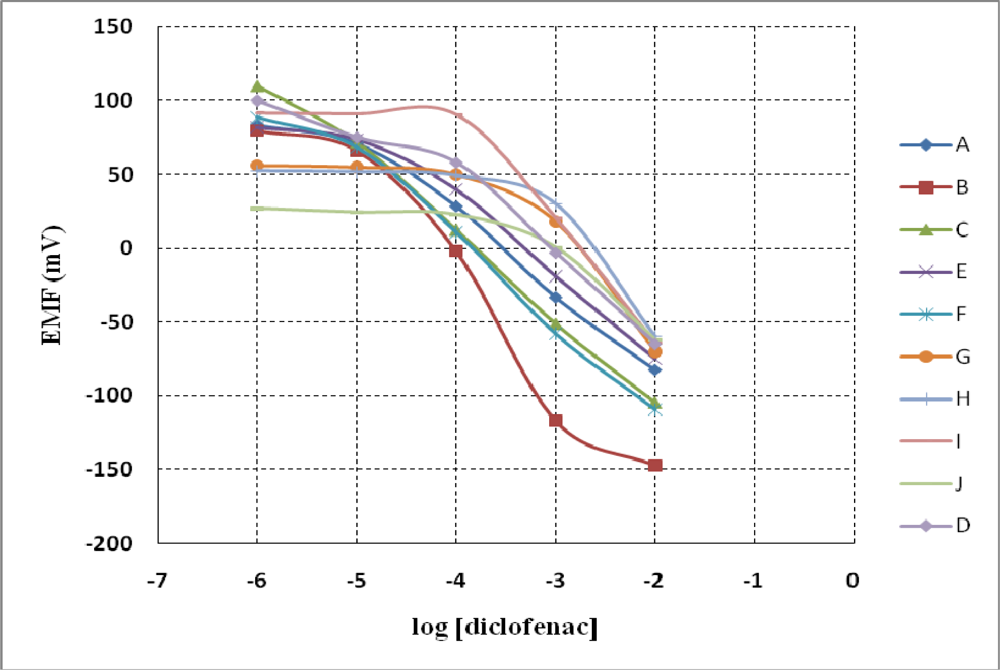


| Sensor | Ionophore (1) | Ionophore (2) | Plasticizer | PVC | NaTPB* | TDMACl* | ||
|---|---|---|---|---|---|---|---|---|
| DOP | DOS | o-NPOE | ||||||
| A | 1 | - | 66 | - | - | 33 | - | - |
| B | 1 | - | 66 | - | - | 33 | - | 20 |
| C | 1 | - | 66 | - | - | 33 | 20 | - |
| D | 1 | - | 66 | - | - | 33 | 40 | - |
| E | 1 | - | - | 66 | - | 33 | 20 | - |
| F | 1 | - | - | - | 66 | 33 | 20 | - |
| G | - | 1 | 66 | - | - | 33 | - | - |
| H | - | 1 | 66 | - | - | 33 | 20 | - |
| I | - | 1 | 66 | - | - | 33 | - | 20 |
| J | Sol–gel | - | ||||||
| Electrode | Linear concentration range (M) | Detection limit (μM) | Slope (mV/dec) |
|---|---|---|---|
| A | 1 × 10−5–1 × 10−2 | 5 | −52.3 ± 0.8 |
| B | 1 × 10−5–5 × 10−2 | 5 | −80.1 ± 1.2 |
| C | 3 × 10−6–1 × 10−2 | 1.5 | −59.7 ± 0.6 |
| D | 5 × 10−5–1 × 10−2 | 30 | −65.5 ± 0.8 |
| E | 1 × 10−5–1 × 10−2 | 8 | −50.4 ± 0.5 |
| F | 5 × 10−6–1 × 10−2 | 3 | −60.2 ± 0.9 |
| G | 5 × 10−4–1 × 10−2 | 400 | −81.1 ± 1.1 |
| H | 3 × 10−4–1 × 10−2 | 200 | −85.3 ± 1.2 |
| I | 1 × 10−4–1 × 10−2 | 150 | −77.4 ± 0.9 |
| J | 5 × 104–1 × 10−2 | 350 | −62.3 ± 0.7 |
| Interfering ion (X−) | Present research | Ref.[14] | Ref.[15] | Ref.[16] | Ref. [17] | Ref.[18] | Ref [19] |
|---|---|---|---|---|---|---|---|
| Acetate | −2.00 ± 0.02 | - | - | - | −2.9 | - | −2.45 |
| Phosphate | −4.15 ± 0.05 | - | −3.8 | - | - | - | −3.50 |
| Nitrite | −2.31 ± 0.03 | - | −3.2 | - | - | - | −2.05 |
| Benzoate | −2.38 ± 0.02 | −2.40 | −3.3 | - | −5.0 | −2.75 | |
| Tartrate | −2.43 ± 0.03 | - | −3.6 | - | −2.1 | - | −4.40 |
| Borate | −3.64 ± 0.04 | - | - | - | - | - | −2.40 |
| Thiocyanate | −1.36 ± 0.01 | - | −3.5 | - | −1.42 | - | −0.90 |
| Glycine | −2.43 ± 0.02 | - | - | −2.6 | - | −5.1 | - |
| Iodide | −2.29 ± 0.03 | - | −2.9 | - | - | −4.3 | - |
| Metabisulphite | −3.90 ± 0.06 | - | - | - | - | - | −2.50 |
| Chloride | −2.54 ± 0.01 | - | −2.3 | −2.6 | −0.36 | −4.7 | −2.60 |
| Lactate | −2.48 ± 0.02 | - | - | - | - | - | - |
| Citrate | −4.14 ± 0.07 | - | −3.8 | - | - | - | −3.45 |
| Nitrate | −2.58 ± 0.04 | −2.50 | −2.0 | −2.3 | - | −4.5 | −2.30 |
| Glucose | −2.52 ± 0.01 | - | −3.2 | −2.8 | - | −4.9 | - |
| Salicylate | −1.90 ± 0.01 | −1.00 | −2.7 | - | −2.0 | −4.7 | −0.95 |
| Sulphate | −4.18 ± 0.05 | - | −3.0 | - | −3.9 | - | −3.70 |
| Reference no. | Linear concentration range (M) | Detection limit (M) | pH range | Slope (mV/dec) |
|---|---|---|---|---|
| [14] | 5 × 10−5–5×10−2 | 2.5 × 10−5 | 6−11 | −59 ± 1 |
| [15] | 9 × 10−6–1×10−2 | 5.4 × 10−6 | 5.5−9 | −61 ± 1 |
| [16] | 1 × 10−5–1×10−2 | 4.0 × 10−6 | 6–9 | −59 ± 1 |
| [17] | 5 × 10−5–1×10−2 | 3.2 × 10−5 | 6.5–9 | −58.1 ± 0.8 |
| [18] | 1 × 10−5–5×10−2 | 5.0 × 10−6 | 2.4–7.5; 8.5−11.6 | −60 ± 1 |
| [19] | 5 × 10−5–1×10−2 | 2.0 × 10−5 | 8.8−12.8 | −59.8 ± 0.3 |
| Present research | 3 × 10−6–1×10−2 | 1.5 × 10−6 | 5.5−11.5 | −59.7 ± 0.6 |
| Sample | Label amount (mg) | Found by potentiometry (mg) | Found by HPLC (mg) | Relative error (%) |
|---|---|---|---|---|
| Diclofenac | 50.0/tablet | 49.6 ± 0.7 | 49.2 ± 0.4 | +0.8 |
| Diclotard | 100.0/tablet | 109.6 ± 1.3 | 108.4 ± 1.2 | +1.1 |
| Refen retard | 100.0/tablet | 105.9 ± 1.1 | 107.2 ± 0.9 | −1.2 |
| Voltaren | 75.0/ampoule of 3 mL | 79.2 ± 0.9 | 78.3 ± 0.8 | +1.1 |
| Refen | 75.0/ampoule of 3 mL | 74.0 ± 0.8 | 73.3 ± 0.6 | +0.9 |
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vlascici, D.; Pruneanu, S.; Olenic, L.; Pogacean, F.; Ostafe, V.; Chiriac, V.; Pica, E.M.; Bolundut, L.C.; Nica, L.; Fagadar-Cosma, E. Manganese(III) Porphyrin-based Potentiometric Sensors for Diclofenac Assay in Pharmaceutical Preparations. Sensors 2010, 10, 8850-8864. https://doi.org/10.3390/s101008850
Vlascici D, Pruneanu S, Olenic L, Pogacean F, Ostafe V, Chiriac V, Pica EM, Bolundut LC, Nica L, Fagadar-Cosma E. Manganese(III) Porphyrin-based Potentiometric Sensors for Diclofenac Assay in Pharmaceutical Preparations. Sensors. 2010; 10(10):8850-8864. https://doi.org/10.3390/s101008850
Chicago/Turabian StyleVlascici, Dana, Stela Pruneanu, Liliana Olenic, Florina Pogacean, Vasile Ostafe, Vlad Chiriac, Elena Maria Pica, Liviu Calin Bolundut, Luminita Nica, and Eugenia Fagadar-Cosma. 2010. "Manganese(III) Porphyrin-based Potentiometric Sensors for Diclofenac Assay in Pharmaceutical Preparations" Sensors 10, no. 10: 8850-8864. https://doi.org/10.3390/s101008850
APA StyleVlascici, D., Pruneanu, S., Olenic, L., Pogacean, F., Ostafe, V., Chiriac, V., Pica, E. M., Bolundut, L. C., Nica, L., & Fagadar-Cosma, E. (2010). Manganese(III) Porphyrin-based Potentiometric Sensors for Diclofenac Assay in Pharmaceutical Preparations. Sensors, 10(10), 8850-8864. https://doi.org/10.3390/s101008850







