Harnessing Genetic Diversity of Wild Gene Pools to Enhance Wheat Crop Production and Sustainability: Challenges and Opportunities
Abstract
1. Global Crop Demand: the Need for New Strategies
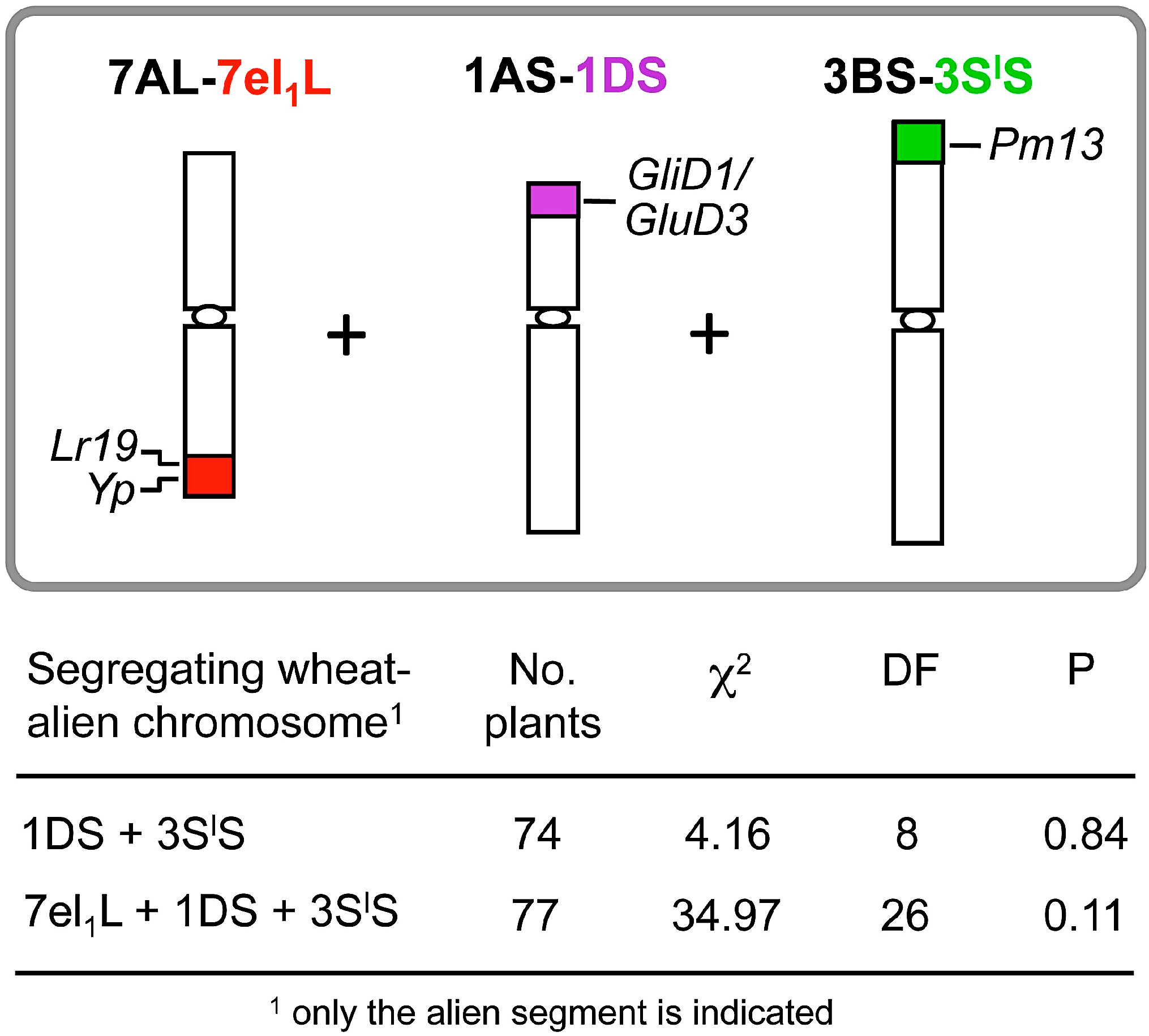
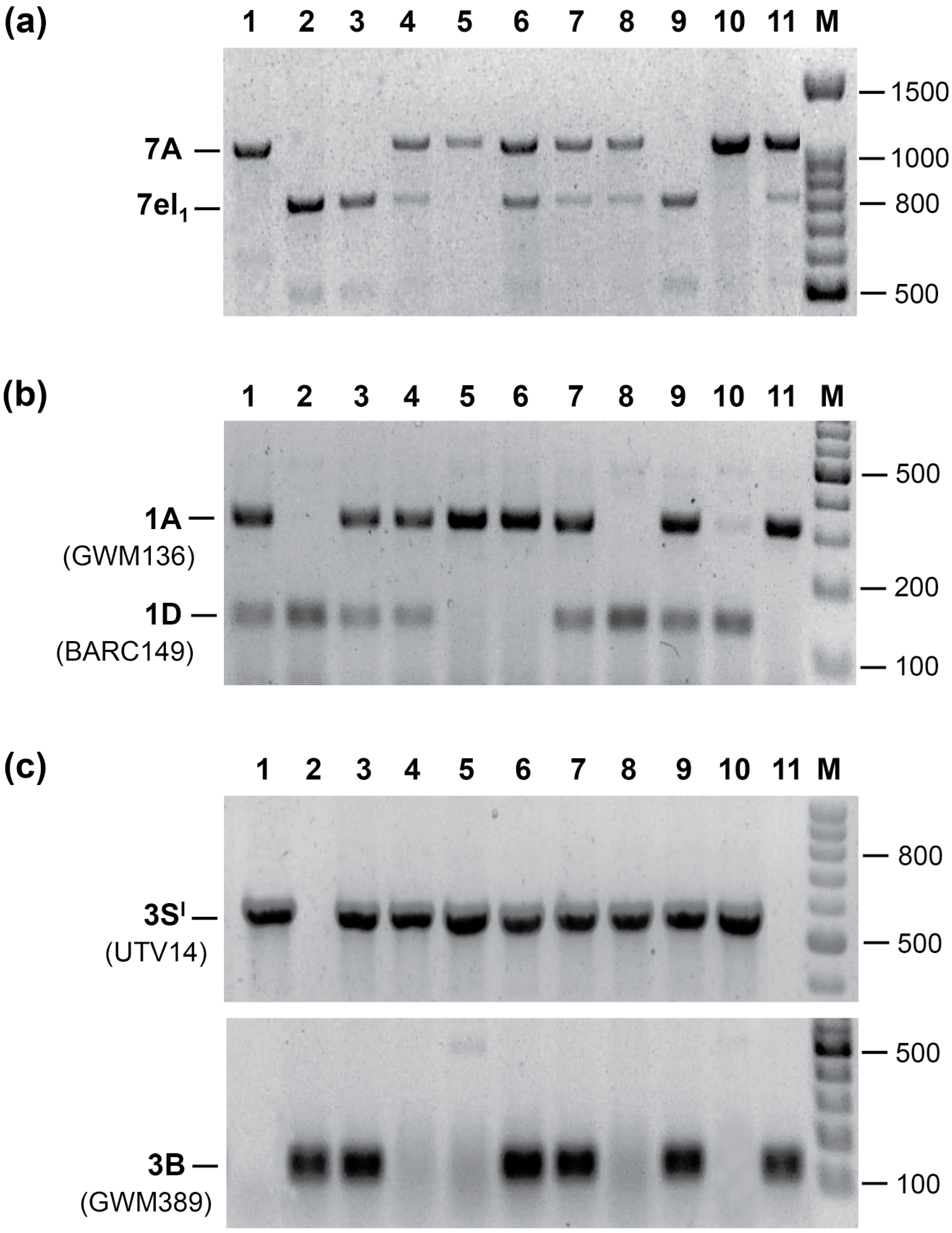
2. Stacking Different Alien Segments
2.1. Agronomic Evaluation of Multiple Recombinant Lines
2.2. Breeding Potential of the Triple Recombinant
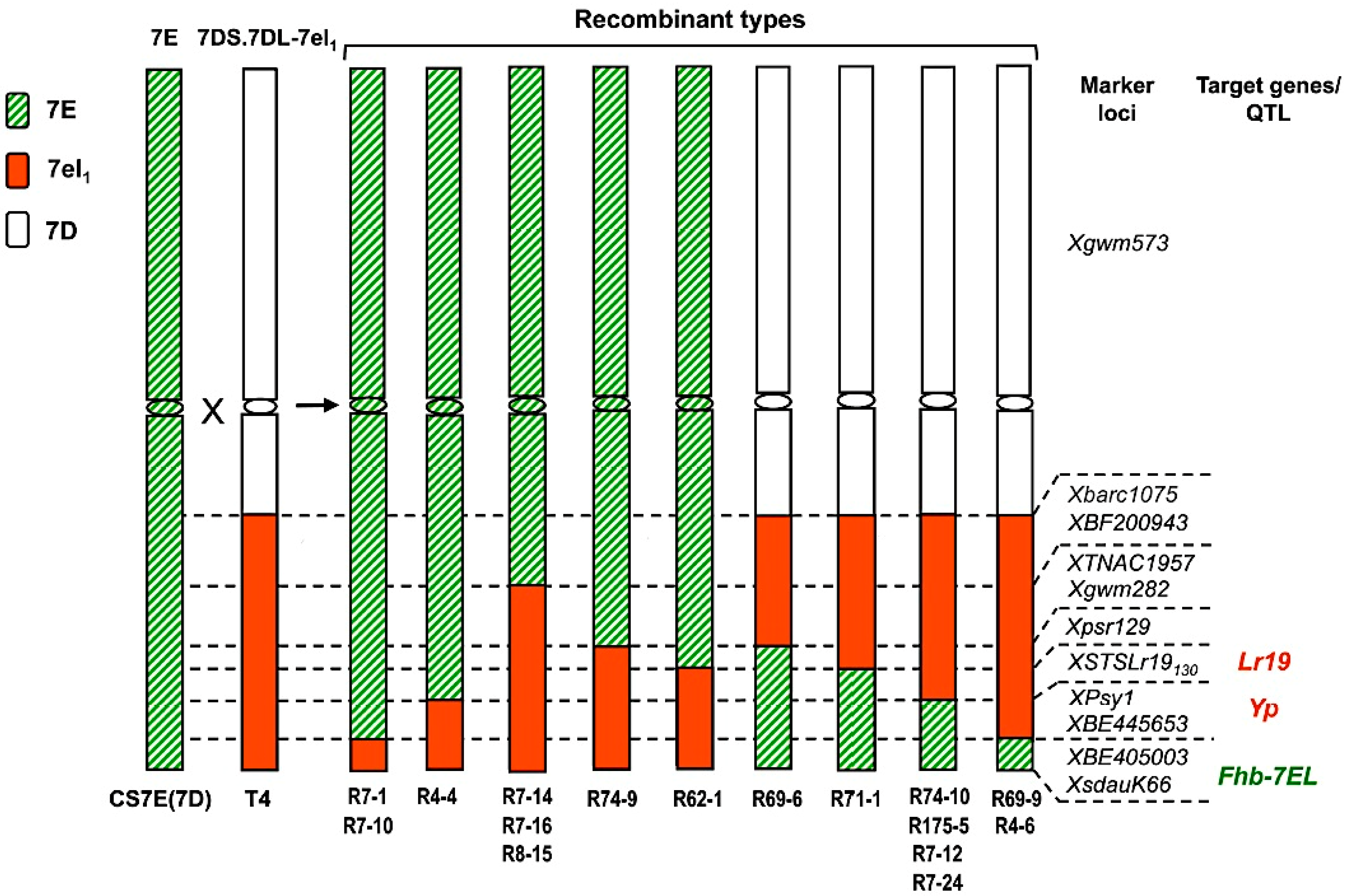
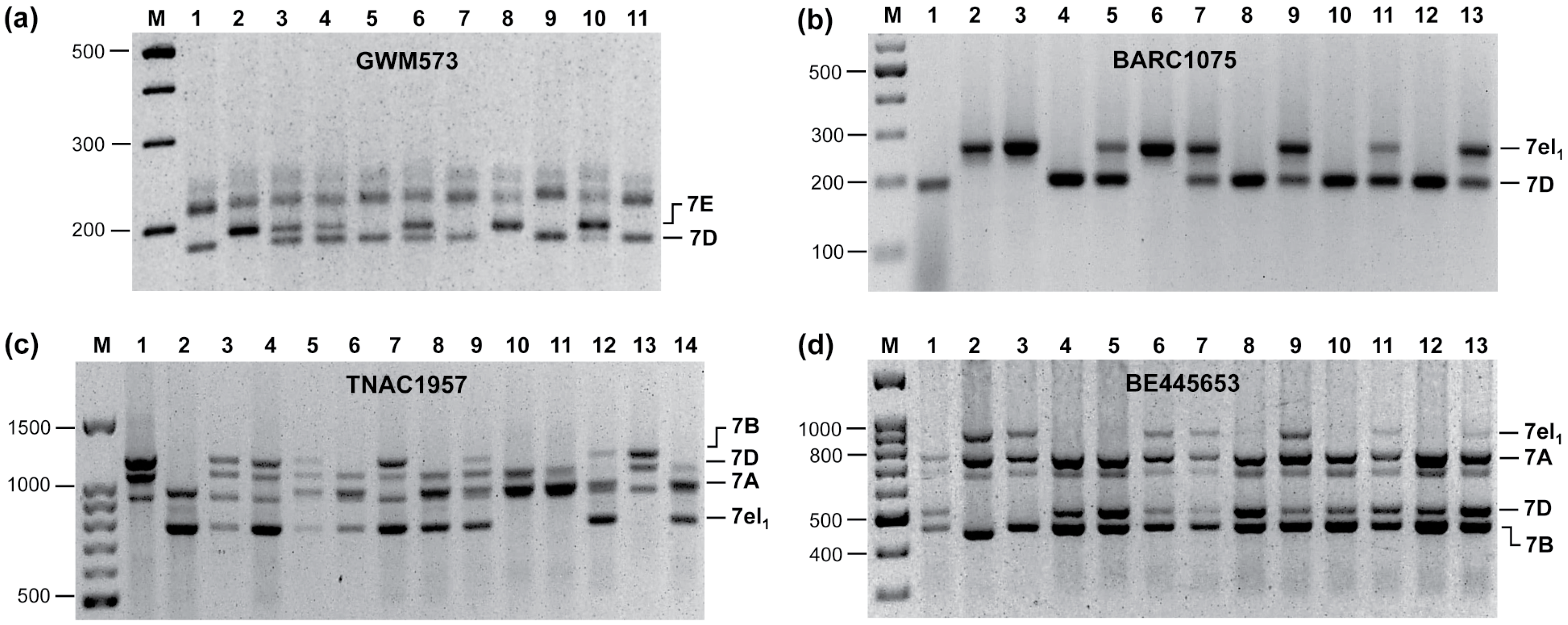
3. Assembling Genes from Different Alien Sources into the Same Segment
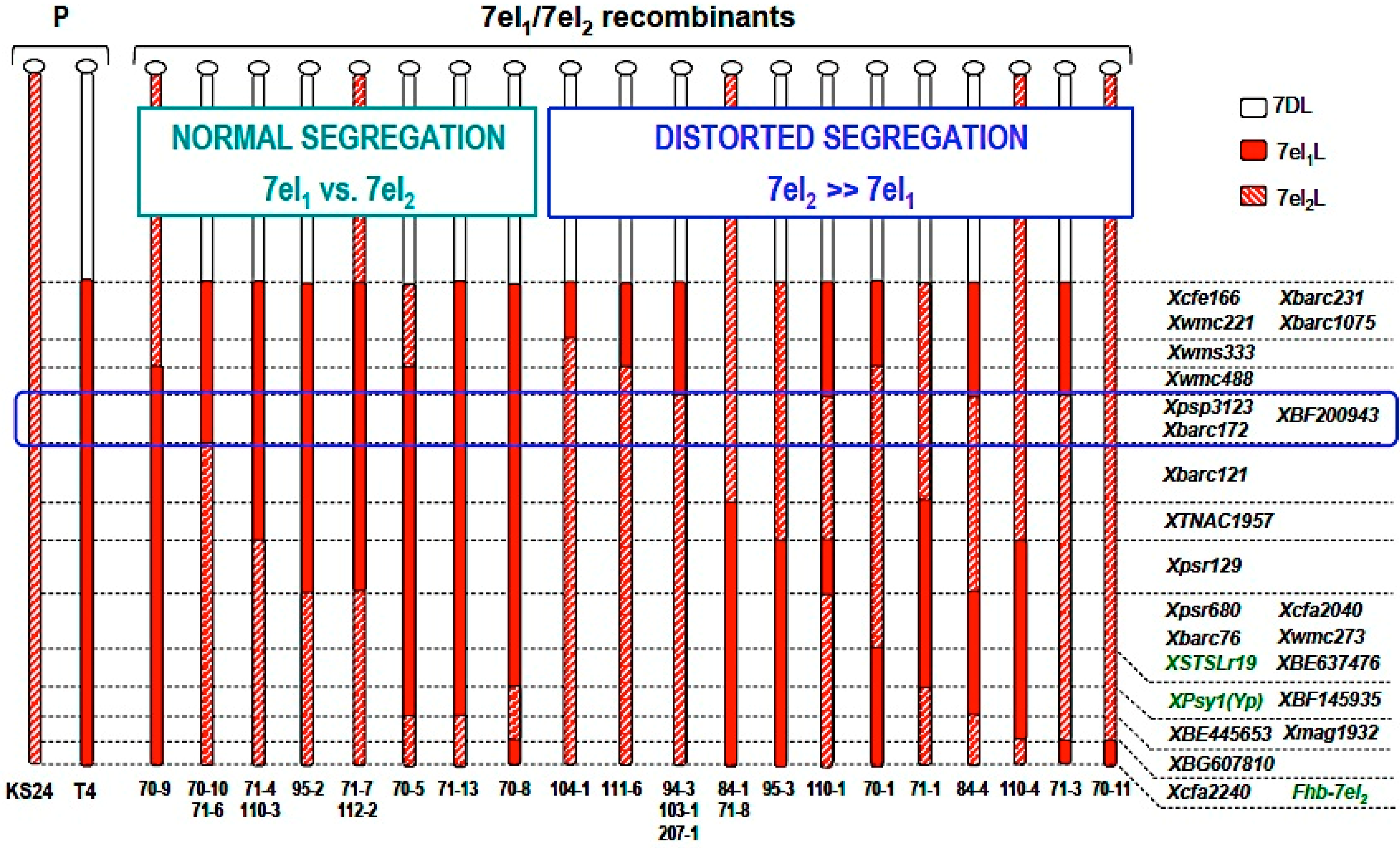
4. An Intriguing Issue in Wheat-Alien Gene Transfer: Segregation Distortion
5. The Way Forward
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Statista—The Statistical Portal: Global Grain Acreage by Type—2011/12 to 2016/17. Available online: https://www.statista.com/statistics/272536/acreage-of-grain-worldwide-by-type/ (accessed on 30 July 2017).
- Giorgi, B.; Porfiri, O. (Eds.) The Varieties of Strampelli: A Milestone in Wheat Breeding in Italy and in the World. In Proceedings of the Special Session of the 41st Annual Congress of the Italian Society of Agricultural Genetics (SIGA), Abbadia di Fiastra, Tolentino, Italy, 26 September 1997; Tipografia San Giuseppe: Pollenza, Italy, 1998; pp. 1–63, (In Italian with English Abstracts). [Google Scholar]
- Borlaug, N.E. Sixty-two years of fighting hunger: Personal recollections. Euphytica 2007, 157, 287–297. [Google Scholar] [CrossRef]
- Calderini, D.F.; Slafer, G.A. Changes in yield and yield stability in wheat during the 20th century. Field Crops Res. 1998, 57, 335–347. [Google Scholar] [CrossRef]
- De Vita, P.; Nicosia, O.L.D.; Nigro, F.; Platani, C.; Riefolo, C.; Di Fonzo, N.; Cattivelli, L. Breeding progress in morpho-physiological, agronomical and qualitative traits of durum wheat cultivars released in Italy during the 20th century. Eur. J. Agron. 2007, 26, 39–53. [Google Scholar] [CrossRef]
- Fischer, T.; Byerlee, D.; Edmeades, G. Crop Yields and Global Food Security: Will Yield Increase Continue to Feed the World? ACIAR Monograph No. 158; Australian Centre for International Agricultural Research: Canberra, Australia, 2014; p. xxii + 634. [Google Scholar]
- Lobell, D.B.; Gourdji, S.M. The influence of climate change on global crop productivity. Plant Physiol. 2012, 160, 1686–1697. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Balzer, C.; Hill, J.; Befort, B.L. Global food demand and the sustainable intensification of agriculture. Proc. Natl. Acad. Sci. USA 2011, 108, 20260–20264. [Google Scholar] [CrossRef] [PubMed]
- Hawkesford, M.; Araus, J.; Park, R.; Calderini, D.; Miralles, D.; Shen, T.; Zhang, J.; Parry, M.A.J. Prospects of doubling global wheat yields. Food Energy Secur. 2013, 2, 34–48. [Google Scholar] [CrossRef]
- Mujeeb-Kazi, A.; Kazi, A.G.; Dundas, I.; Rasheed, A.; Ogbonnaya, F.; Kishii, M.; Bonnett, D.; Wang, R.R.-C.; Xu, S.; Chen, P.; et al. Genetic diversity for wheat improvement as a conduit to food security. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Burlington, NI, USA, 2013; Volume 122, pp. 179–257. [Google Scholar]
- Molnár-Láng, M.; Ceoloni, C.; Doležel, J. (Eds.) Alien Introgression in Wheat—Cytogenetics, Molecular Biology, and Genomics; Springer: Cham, Switzerland, 2015; pp. 1–385. [Google Scholar] [CrossRef]
- Sears, E.R. Transfer of alien genetic material to wheat. In Wheat Science-Today and Tomorrow; Evans, L.T., Peacock, W.J., Eds.; Cambridge University Press: Cambridge, UK, 1981; pp. 75–89. [Google Scholar]
- Naranjo, T.; Benavente, E. The mode and regulation of chromosome pairing in wheat-alien hybrids (Ph genes, an updated view). In Alien Introgression in Wheat—Cytogenetics, Molecular Biology, and Genomics; Molnár-Láng, M., Ceoloni, C., Doležel, J., Eds.; Springer: Cham, Switzerland, 2015; pp. 133–162. [Google Scholar]
- Ceoloni, C.; Biagetti, M.; Ciaffi, M.; Forte, P.; Pasquini, M. Wheat chromosome engineering at the 4x level: The potential of different alien gene transfers into durum wheat. Euphytica 1996, 89, 87–97. [Google Scholar] [CrossRef]
- Ceoloni, C.; Jauhar, P.P. Chromosome engineering of the durum wheat genome: Strategies and applications of potential breeding value. In Genetic Resources, Chromosome Engineering, and Crop Improvement: Cereals; Singh, R.J., Jauhar, P.P., Eds.; CRC Press: Boca Raton, FL, USA, 2006; pp. 27–59. [Google Scholar]
- Qi, L.; Friebe, B.; Zhang, P.; Gill, B.S. Homoeologous recombination, chromosome engineering and crop improvement. Chromosome Res. 2007, 15, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Ayala-Navarrete, L.; Mechanicos, A.A.; Gibson, J.M.; Singh, D.; Bariana, H.S.; Fletcher, J.; Shorter, S.; Larkin, P.J. The Pontin series of recombinant alien translocations in bread wheat: Single translocations integrating combinations of Bdv2, Lr19 and Sr25 disease-resistance genes from Thinopyrum intermedium and Th. ponticum. Theor. Appl. Genet. 2013, 126, 2467–2475. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.; Klindworth, D.L.; Friesen, T.L.; Chao, S.; Ohm, J.B.; Xu, S.S. Development and characterization of wheat lines carrying stem rust resistance gene Sr43 derived from Thinopyrum ponticum. Theor. Appl. Genet. 2014, 127, 969–980. [Google Scholar] [CrossRef] [PubMed]
- Danilova, T.V.; Zhang, G.; Liu, W.; Friebe, B.; Gill, B.S. Homoeologous recombination-based transfer and molecular cytogenetic mapping of a wheat streak mosaic virus and Triticum mosaic virus resistance gene Wsm3 from Thinopyrum intermedium. Theor. Appl. Genet. 2017, 130, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Koo, D.-H.; Xia, Q.; Li, C.; Bai, F.; Song, Y.; Friebe, B.; Gill, B.S. Homoeologous recombination-based transfer and molecular cytogenetic mapping of powdery mildew-resistant gene Pm57 from Aegilops searsii into wheat. Theor. Appl. Genet. 2017, 130, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Ceoloni, C.; Pasquini, M.; Simeone, R. The cytogenetic contribution to the analysis and manipulation of the durum wheat genome. In Durum Wheat Breeding: Current Approaches and Future Strategies; Royo, C., Nachit, M.N., Di Fonzo, N., Araus, J.L., Pfeiffer, W.H., Slafer, G.A., Eds.; Haworth Press: New York, NY, USA, 2005; pp. 165–196. [Google Scholar]
- Ceoloni, C.; Forte, P.; Gennaro, A.; Micali, S.; Carozza, R.; Bitti, A. Recent developments in durum wheat chromosome engineering. Cytogenet. Genome Res. 2005, 109, 328–344. [Google Scholar] [CrossRef] [PubMed]
- Klindworth, D.L.; Niu, Z.; Chao, S.; Friesen, T.L.; Jin, Y.; Faris, J.D.; Cai, X.; Xu, S.S. Introgression and characterization of a goatgrass gene for a high level of resistance to Ug99 stem rust in tetraploid wheat. G3 Genes Genomes Genet. 2012, 2, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Zhang, P.; Ryan, P.R.; Rathjen, T.M.; Yan; Z.H.; Delhaize, E. Introgression of genes from bread wheat enhances the aluminium tolerance of durum wheat. Theor. Appl. Genet. 2016, 129, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Gennaro, A.; Forte, P.; Carozza, R.; Savo Sardaro, M.L.; Ferri, D.; Bitti, A.; Borrelli, G.M.; D’Egidio, M.G.; Ceoloni, C. Pyramiding different alien chromosome segments in durum wheat: Feasibility and breeding potential. Isr. J. Plant Sci. 2007, 55, 267–276. [Google Scholar] [CrossRef]
- Gennaro, A.; Forte, P.; Panichi, D.; Lafiandra, D.; Pagnotta, M.A.; D’Egidio, M.G.; Ceoloni, C. Stacking small segments of the 1D chromosome of bread wheat containing major gluten quality genes into durum wheat: Transfer strategy and breeding prospects. Mol. Breed. 2012, 30, 149–167. [Google Scholar] [CrossRef]
- Morris, C.F.; Simeone, M.N.; King, G.E.; Lafiandra, D. Transfer of soft kernel texture from Triticum aestivum to durum wheat, Triticum turgidum ssp. durum. Crop Sci. 2011, 51, 114–122. [Google Scholar] [CrossRef]
- Gazza, L.; Sgrulletta, D.; Cammerata, A.; Gazzelloni, G.; Galassi, E.; Pogna, N. Breeding and quality of soft-textured durum wheat. In Proceedings of the International Symposium on Genetics and Breeding of Durum Wheat, Rome, Italy, 27–30 May 2013; Options Méditerranéennes, Séries A; No. 110. Porceddu, E., Damania, A.B., Qualset, C.O., Eds.; 2014; pp. 511–516. [Google Scholar]
- Kuzmanović, L.; Gennaro, A.; Benedettelli, S.; Dodd, I.C.; Quarrie, S.A.; Ceoloni, C. Structural-functional dissection and characterization of yield-contributing traits originating from a group 7 chromosome of the wheatgrass species Thinopyrum ponticum after transfer into durum wheat. J. Exp. Bot. 2014, 65, 509–525. [Google Scholar] [CrossRef] [PubMed]
- Kuzmanović, L.; Ruggeri, R.; Virili, M.E.; Rossini, F.; Ceoloni, C. Effects of Thinopyrum ponticum chromosome segments transferred into durum wheat on yield components and related morpho-physiological traits in Mediterranean rain-fed conditions. Field Crops Res. 2016, 186, 86–98. [Google Scholar] [CrossRef]
- Ceoloni, C.; Margiotta, B.; Colaprico, G.; D’Egidio, M.G.; Carozza, R.; Lafiandra, D. Introgression of D-genome associated gluten protein genes into durum wheat. In Proceedings of the 10th International Wheat Genetics Symposium, Paestum, Italy, 1–6 September 2003; S.I.M.I: Rome, Italy, 2003; pp. 1320–1322. [Google Scholar]
- Micali, S.; Forte, P.; Bitti, A.; D’Ovidio, R.; Ceoloni, C. Chromosome engineering as a tool for effectively introgressing multiple useful genes from alien Triticeae into durum wheat. In Proceedings of the 10th International Wheat Genetics Symposium, Paestum, Italy, 1–6 September 2003; pp. 896–898. [Google Scholar]
- Gennaro, A.; Koebner, R.M.D.; Ceoloni, C. A candidate for Lr19, an exotic gene conditioning leaf rust resistance in wheat. Funct. Integr. Genom. 2009, 9, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Cenci, A.; D’Ovidio, R.; Tanzarella, O.A.; Ceoloni, C.; Porceddu, E. Identification of molecular markers linked to Pm13, an Aegilops longissima gene conferring resistance to powdery mildew of wheat. Theor. Appl. Genet. 1999, 98, 448–454. [Google Scholar] [CrossRef]
- Klindworth, D.L.; Hareland, G.A.; Elias, E.M.; Xu, S.S. Attempted compensation for linkage drag affecting agronomic characteristics of durum wheat 1AS/1DL translocation lines. Crop Sci. 2013, 53, 422–429. [Google Scholar] [CrossRef]
- Ceoloni, C.; Kuzmanović, L.; Gennaro, A.; Forte, P.; Giorgi, D.; Grossi, M.R.; Bitti, A. Genomes, chromosomes and genes of perennial Triticeae of the genus Thinopyrum: The value of their transfer into wheat for gains in cytogenomic knowledge and ‘precision’ breeding. In Advances in Genomics of Plant Genetic Resources; Tuberosa, R., Graner, A., Frison, E., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 333–358. [Google Scholar]
- Ceoloni, C.; Kuzmanović, L.; Forte, P.; Virili, M.E.; Bitti, A. Wheat-perennial Triticeae introgressions: Major achievements and prospects. In Alien Introgression in Wheat—Cytogenetics, Molecular Biology, and Genomics; Molnár-Láng, M., Ceoloni, C., Doležel, J., Eds.; Springer: Cham, Switzerland, 2015; pp. 273–313. [Google Scholar]
- Ayala-Navarrete, L.; Bariana, H.S.; Singh, R.P.; Gibson, J.M.; Mechanicos, A.A.; Larkin, P.J. Trigenomic chromosomes by recombination of Thinopyrum intermedium and Th. ponticum translocations in wheat. Theor. Appl. Genet. 2007, 116, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Ohm, H. Molecular mapping of Thinopyrum-derived Fusarium head blight resistance in common wheat. Mol. Breed. 2007, 20, 131–140. [Google Scholar] [CrossRef]
- Zhang, X.L.; Shen, X.R.; Hao, Y.F.; Cai, J.J.; Ohm, H.W.; Kong, L. A genetic map of Lophopyrum ponticum chromosome 7E, harboring resistance genes to Fusarium head blight and leaf rust. Theor. Appl. Genet. 2011, 122, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Forte, P.; Virili, M.E.; Kuzmanović, L.; Moscetti, I.; Gennaro, A.; D’Ovidio, R.; Ceoloni, C. A novel assembly of Thinopyrum ponticum genes into the durum wheat genome: Pyramiding Fusarium head blight resistance onto recombinant lines previously engineered for other beneficial traits from the same alien species. Mol. Breed. 2014, 34, 1701–1716. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, X.; Hou, Y.; Cai, J.; Shen, X.; Zhou, T.; Xu, H.; Ohm, H.W.; Wang, H.; Li, A.; et al. High-density mapping of the major FHB resistance gene Fhb7 derived from Thinopyrum ponticum and its pyramiding with Fhb1 by marker-assisted selection. Theor. Appl. Genet. 2015, 128, 2301–2316. [Google Scholar] [CrossRef] [PubMed]
- Ceoloni, C.; Forte, P.; Kuzmanović, L.; Tundo, S.; Moscetti, I.; De Vita, P.; Virili, M.E.; D’Ovidio, R. Cytogenetic mapping of a major locus for resistance to Fusarium head blight and crown rot of wheat on Thinopyrum elongatum 7EL and its pyramiding with valuable genes from a Th. ponticum homoeologous arm onto bread wheat 7DL. Theor. Appl. Genet. 2017, 130, 2005–2024. [Google Scholar] [CrossRef] [PubMed]
- Lyttle, T.W. Segregation distorters. Ann. Rev. Genet. 1991, 25, 511–557. [Google Scholar] [CrossRef] [PubMed]
- Hurst, G.D.D.; Werren, J.H. The role of selfish genetic elements in eukaryotic evolution. Nat. Rev. Genet. 2001, 2, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.R.; Ingvarsson, P.K. Common features of segregation distortion in plants and animals. Genetica 2003, 117, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Guo, L.; You, J.; Liu, X.; He, Y.; Yuan, J.; Liu, G.; Feng, Z. Progress of segregation distortion in genetic mapping of plants. Res. J. Agron. 2010, 4, 78–83. [Google Scholar] [CrossRef]
- Burt, A.; Trivers, R. Selfish Genetic Elements. In Genes in Conflict—The Biology of Selfish Genetic Elements; Belknap Press: Cambridge, UK, 2006; pp. 1–18. [Google Scholar]
- Li, C.; Bai, G.; Chao, S.; Wang, Z. A High-density SNP and SSR consensus map reveals segregation distortion regions in wheat. BioMed Res. Int. 2015, 2015, 830618. [Google Scholar] [CrossRef] [PubMed]
- Seymour, D.K.; Chae, E.; Ariöz, B.I.; Koenig, D.; Weigel, D. The genetic architecture of recurrent segregation distortion in Arabidopsis thaliana. bioRxiv 2017. [Google Scholar] [CrossRef]
- Endo, T.R. The gametocidal chromosome as a tool for chromosome manipulation in wheat. Chromosome Res. 2007, 15, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Habu, Y.; Ando, T.; Ito, S.; Nagaki, K.; Kishimoto, N.; Taguchi-Shiobara, F.; Numa, H.; Yamaguchi, K.; Shigenobu, S.; Murata, M.; et al. Epigenomic modification in rice controls meiotic recombination and segregation distortion. Mol. Breed. 2015, 35, 103. [Google Scholar] [CrossRef]
- Kwiatek, M.T.; Wisniewska, H.; Slusarkiewicz-Jarzina, A.; Majka, J.; Majka, M.; Belter, J.; Pudelska, H. Gametocidal factor transferred from Aegilops geniculata Roth can be adapted for large-scale chromosome manipulations in cereals. Front. Plant Sci. 2017, 8, 409. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, H. Gametocidal genes in wheat as the inducer of chromosome breakage. In Frontiers of Wheat Bioscience; Memorial Issue, Wheat Information Service No. 100; Tsunewaki, K., Ed.; Kihara Memorial Yokohama Foundation: Yokohama, Japan, 2005; pp. 33–48. [Google Scholar]
- Endo, T.R. Gametocidal genes. In Alien Introgression in Wheat—Cytogenetics, Molecular Biology, and Genomics; Molnár-Láng, M., Ceoloni, C., Doležel, J., Eds.; Springer: Cham, Switzerland, 2015; pp. 121–131. [Google Scholar]
- Knott, D.R. The transfer of genes for disease resistance from alien species to wheat by induced translocations. In Mutation Breeding for Disease Resistance; IAEA: Vienna, Austria, 1971; pp. 67–77. [Google Scholar]
- McIntosh, R.A.; Dyck, P.L.; Green, G.J. Inheritance of leaf rust and stem rust resistance in wheat cultivars Agent and Agatha. Aust. J. Agric. Res. 1976, 28, 37–45. [Google Scholar] [CrossRef]
- Kibirige-Sebunya, I.; Knott, D.R. Transfer of stem rust resistance to wheat from an Agropyron chromosome having a gametocidal effect. Can. J. Genet. Cytol. 1983, 25, 215–221. [Google Scholar] [CrossRef]
- Scoles, G.J.; Kibirige-Sebunya, I.N. Preferential abortion of gametes in wheat induced by an Agropyron chromosome. Can. J. Genet. Cytol. 1983, 25, 1–6. [Google Scholar] [CrossRef]
- Marais, G.F. Genetic control of a response to the segregation distortion allele, Sd-1d, in the common wheat line ‘Indis’. Euphytica 1992, 60, 89–95. [Google Scholar]
- Prins, R.; Marais, G.F. A genetic study of the gametocidal effect of the Lr19 translocation of common wheat. S. Afr. J. Plant Soil 1999, 16, 10–14. [Google Scholar] [CrossRef]
- Marais, G.F.; Marais, A.S.; Groenwald, J.Z. Evaluation and reduction of Lr19-149, a recombined form of the Lr19 translocation of wheat. Euphytica 2001, 121, 289–295. [Google Scholar] [CrossRef]
- Groenwald, J.Z.; Fourie, M.; Marais, A.S.; Marais, G.F. Extention and use of a physical map of the Thinopyrum-derived Lr19 translocation. Theor. Appl. Genet. 2005, 112, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Zhang, X.; Wang, B.; Yan, M.; Qi, Y.; Kong, L. A genetic analysis of segregation distortion revealed by molecular markers in Lophopyrum ponticum chromosome 7E. J. Genet. 2011, 90, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.R. Gametocidal chromosomes and their induction of chromosome mutations in wheat. Jpn. J. Genet. 1990, 65, 135–152. [Google Scholar] [CrossRef]
- Kong, L.; Anderson, J.M.; Ohm, H.W. Segregation distortion in common wheat of a segment of Thinopyrum intermedium chromosome 7E carrying Bdv3 and development of a Bdv3 marker. Plant Breed. 2008, 128, 591–597. [Google Scholar] [CrossRef]
- Sun, Q.Y.; Breitbart, H.; Schatten, H. Role of the MAPK cascade in mammalian germ cells. Reprod. Fertil. Dev. 2000, 11, 443–450. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, S. Mitogen-activated protein kinase cascades in signaling plant growth and development. Trends Plant Sci. 2015, 20, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Knight, E.; Binnie, A.; Draeger, T.; Moscou, M.; Rey, M.-D.; Sucher, J.; Mehra, S.; King, I.; Moore, G. Mapping the ‘breaker’ element of the gametocidal locus proximal to a block of sub-telomeric heterochromatin on the long arm of chromosome 4Ssh of Aegilops sharonensis. Theor. Appl. Genet. 2015, 128, 1049–1059. [Google Scholar] [CrossRef] [PubMed]
- Grewal, S.; Gardiner, L-J.; Ndreca, B.; Knight, E.; Moore, G.; King, I.P.; King, J. Comparative mapping and targeted-capture sequencing of the gametocidal loci in Aegilops sharonensis. Plant Genome 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, H.; Tsunewaki, K. Hybrid dysgenesis in common wheat caused by gametocidal genes. Jpn. J. Genet. 1985, 60, 565–578. [Google Scholar] [CrossRef]
- McClintock, B. The significance of responses of the genome to challenge. Science 1984, 226, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Shaked, H.; Kashkush, K.; Ozkan, H.; Feldman, M.; Levy, A.A. Sequence elimination and cytosine methylation are rapid and reproducible responses of the genome to wide hybridization and allopolyploidy in wheat. Plant Cell 2001, 13, 1749–1759. [Google Scholar] [CrossRef] [PubMed]
- Feldman, M.; Levy, A.A. Genome evolution in allopolyploid wheat—A revolutionary reprogramming followed by gradual changes. J. Genet. Genom. 2009, 36, 511–518. [Google Scholar] [CrossRef]
- Levy, A.A. Transposons in plant speciation. In Plant Transposons and Genome Dynamics in Evolution, 1st ed.; Fedoroff, N.V., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 165–179. [Google Scholar]
- Liu, Z.; Wang, Y.; Shen, Y.; Guo, W.; Hao, S.; Liu, B. Extensive alterations in DNA methylation and transcription in rice caused by introgression from Zizania latifolia. Plant Mol. Biol. 2004, 54, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.Y.; Wang, Y.M.; Zhang, Z.J.; Shen, Y.; Lin, X.Y.; Ou, X.F.; Han, F.P.; Liu, B. Extent and pattern of DNA methylation alteration in rice lines derived from introgressive hybridization of rice and Zizania latifolia Griseb. Theor. Appl. Genet. 2006, 113, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, Z.H.; Liu, C.; Yang, Z.J.; Deng, K.J.; Peng, J.H.; Zhou, J.P.; Li, G.R.; Tang, Z.X.; Ren, Z.L. Analysis of DNA methylation variation in wheat genetic background after alien chromatin introduction based on methylation-sensitive amplification polymorphism. Sci. Bull. 2008, 53, 58–69. [Google Scholar] [CrossRef]
- Fu, S.; Sun, C.; Yang, M.; Fei, Y.; Tan, F.; Yan, B.; Ren, Z.; Tang, Z. Genetic and epigenetic variation induced by wheat-rye 2R and 5R monosomic addition lines. PLoS ONE 2013, 8, e54057. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, J. Homoeology between Agropyron elongatum chromosomes and Triticum aestivum chromosomes. Can. J. Genet. Cytol. 1980, 22, 237–259. [Google Scholar] [CrossRef]
- Marais, G.F.; Bekker, T.A.; Eksteen, A.; McCallum, B.; Fetch, T.; Marais, A.S. Attempts to remove gametocidal genes co-transferred to common wheat with rust resistance from Aegilops speltoides. Euphytica 2010, 171, 71–85. [Google Scholar] [CrossRef]
- Millet, E.; Manisterski, J.; Ben-Yehuda, P.; Distelfeld, A.; Deek, J.; Wan, A.; Chen, X.; Steffenson, B.J. Introgression of leaf rust and stripe rust resistance from Sharon goatgrass (Aegilops sharonensis Eig) into bread wheat (Triticum aestivum L.). Genome 2014, 57, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Friebe, B.; Zhang, P.; Nasuda, S.; Gill, B.S. Characterization of a knockout mutation at the Gc2 locus in wheat. Chromosoma 2003, 111, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Dempewolf, H.; Baute, G.; Anderson, J.; Kilian, B.; Smith, C.; Guarino, L. Past and future use of wild relatives in crop breeding. Crop Sci. 2017, 57, 1070–1082. [Google Scholar] [CrossRef]
- Prohens, J.; Gramazio, P.; Plazas, M.; Dempewolf, H.; Kilian, B.; Díez, M.J.; Fita, A.; Herraiz, F.J.; Rodríguez-Burruezo, A.; Soler, S.; et al. Introgressiomics: A new approach for using crop wild relatives in breeding for adaptation to climate change. Euphytica 2017, 213, 158. [Google Scholar] [CrossRef]
- King, J.; Grewal, S.; Yang, C.; Hubbart, S.; Scholefield, D.; Ashling, S.; Edwards, K.J.; Allen, A.M.; Burridge, A.; Bloor, C.; et al. A step change in the transfer of interspecific variation into wheat from Amblyopyrum muticum. Plant Biotechnol. J. 2017, 15, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Winfield, M.O.; Allen, A.M.; Burridge, A.J.; Barker, G.L.A.; Benbow, H.R.; Wilkinson, P.A.; Coghill, J.; Waterfall, C.; Davassi, A.; Scopes, G.; et al. High-density SNP genotyping array for hexaploid wheat and its secondary and tertiary gene pool. Plant Biotechnol. J. 2015, 13, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Lusser, M.; Parisi, C.; Plan, D.; Rodríguez-Cerezo, E. New Plant Breeding Techniques: State-of-the-Art and Prospects for Commercial Development; Reference Report by the Joint Research Centre of the European Commission; Publications Office of the European Union: Luxembourg, 2011. [Google Scholar] [CrossRef]
- Cardi, T. Cisgenesis and genome editing: Combining concepts and efforts for a smarter use of genetic resources in crop breeding. Plant Breed. 2016, 135, 139–147. [Google Scholar] [CrossRef]
- Lowder, L.G.; Zhang, D.; Baltes, N.J.; Paul, J.W.; Tang, X.; Zheng, X.; Voytas, D.F.; Hsieh, T.F.; Zhang, Y.; Qi, Y. A CRISPR/Cas9 toolbox for multiplexed plant genome editing and transcriptional regulation. Plant Physiol. 2015, 169, 971–985. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhang, Q.; Zhu, Q.; Liu, W.; Chen, Y.; Qiu, R.; Wang, B.; Yang, Z.; Li, H.; Lin, Y.; et al. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol. Plant 2015, 8, 1274–1284. [Google Scholar] [CrossRef] [PubMed]
- Palmgren, M.G.; Edenbrandt, A.K.; Vedel, S.E.; Andersen, M.M.; Landes, X.; Osterberg, J.T.; Falhof, J.; Olsen, L.I.; Christensen, S.B.; Sandoe, P.; et al. Are we ready for back-to-nature crop breeding? Trends Plant Sci. 2015, 20, 155–164. [Google Scholar] [CrossRef] [PubMed]
| Genotype/Trait 1 | R117-11-20 | R117-9-11 | R117-9-71 | R117-2-21 | R117-9-59 | R117-11-8 | cv. Karur | cv. Simeto | ANCOVA p-Value 2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lr19 + Yp | + | + | + | + | + | + | − | − | |||||||||
| Glu-D3 | + | + | + | + | + | − | − | − | |||||||||
| Pm13 | + | + | + | − | − | − | − | − | |||||||||
| HD | 115.0 | b | 118.3 | a | 117.7 | ab | 116.3 | ab | 117.3 | ab | 116.3 | ab | 115.7 | ab | 106.7 | c | 0.000 *** |
| PH | 73.3 | ab | 66.7 | c | 66.7 | c | 72.3 | abc | 67.0 | c | 70.0 | bc | 71.3 | bc | 78.3 | a | 0.000 *** |
| SNM2 | 195.4 | 182.4 | 158.8 | 170.6 | 147.1 | 195.4 | 200.7 | 181.7 | 0.332 | ||||||||
| BM2 | 759.0 | 710.8 | 628.0 | 628.8 | 559.6 | 763.9 | 741.4 | 659.5 | 0.458 | ||||||||
| GYM2 | 319.8 | 281.1 | 249.6 | 266.4 | 214.2 | 323.8 | 291.1 | 248.9 | 0.254 | ||||||||
| GNM2 | 6328.5 | 6031.5 | 5192.9 | 5432.1 | 4334.1 | 6293.8 | 6133.0 | 4070.0 | 0.096 | ||||||||
| HI | 0.42 | 0.40 | 0.40 | 0.42 | 0.38 | 0.42 | 0.39 | 0.38 | 0.164 | ||||||||
| GNS | 58.6 | a | 52.0 | ab | 54.6 | a | 55.9 | a | 52.4 | ab | 52.9 | ab | 53.4 | ab | 45.2 | b | 0.001 ** |
| GYS | 3.0 | a | 2.4 | b | 2.6 | ab | 2.7 | ab | 2.6 | ab | 2.7 | ab | 2.5 | ab | 2.8 | ab | 0.035 * |
| GNSP | 3.0 | a | 2.6 | b | 2.8 | ab | 2.8 | ab | 2.6 | b | 2.9 | a | 2.5 | b | 2.8 | ab | 0.000 *** |
| TGW | 50.8 | bc | 46.6 | c | 47.3 | bc | 49.0 | bc | 49.5 | bc | 51.4 | b | 47.1 | c | 61.1 | a | 0.000 *** |
| Trait | Difference vs. Karur | Difference vs. Simeto |
|---|---|---|
| SNM2 | −3% | 8% |
| BM2 | 2% | 15% |
| GYM2 | 10% | 28% |
| GNM2 | 3% | 55% |
| HI | 8% | 11% |
| TGW | 8% | −17% |
| GNS | 10% | 30% |
| GYS | 20% | 7% |
| Rec. No. | Chrom. Type | Marker Alleles | F2 Segregation | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| BF200943 PSP3123 BARC172 | BARC 121 | TNAC 1957 | PSR 129 | HOM+ | HET | HOM− | Χ2 1:2:1 | p Value 1 | ||
| 70-5 | T4 | 7el1 | 7el1 | 7el1 | 7el1 | 9 | 19 | 7 | 0.49 | 0.78 |
| 70-8 | T4 | ” | ” | ” | ” | 6 | 24 | 13 | 2.86 | 0.24 |
| 70-9 | KS24 | ” | ” | ” | ” | 8 | 10 | 5 | 1.18 | 0.55 |
| 71-7 | KS24 | ” | ” | ” | ” | 5 | 11 | 10 | 2.54 | 0.28 |
| 71-13 | T4 | ” | ” | ” | ” | 4 | 7 | 5 | 0.38 | 0.83 |
| 95-2 | T4 | ” | ” | ” | ” | 4 | 7 | 1 | 1.83 | 0.40 |
| 110-3 | T4 | ” | ” | ” | 7el2 | 5 | 11 | 5 | 0.04 | 0.98 |
| 70-10 | T4 | ” | 7el2 | 7el2 | ” | 2 | 8 | 2 | 1.33 | 0.51 |
| 71-6 | T4 | ” | ” | ” | ” | 3 | 8 | 1 | 2.00 | 0.37 |
| 70-1 | T4 | 7el2 | ” | ” | ” | 5 | 14 | 0 | 6.89 | 0.03 * |
| 70-11 | KS24 | ” | ” | ” | ” | 9 | 13 | 0 | 8.09 | 0.02 * |
| 71-3 | T4 | ” | ” | ” | ” | 8 | 16 | 0 | 8.00 | 0.02 * |
| 84-4 | T4 | ” | ” | ” | ” | 21 | 13 | 0 | 27.82 | 0.000 *** |
| 94-3 | T4 | ” | ” | ” | ” | 9 | 18 | 0 | 9.02 | 0.01 * |
| 111-6 | T4 | ” | ” | ” | ” | 16 | 10 | 3 | 14.45 | 0.001 ** |
| 110-1 | T4 | ” | ” | ” | 7el1 | 10 | 8 | 0 | 7.33 | 0.03 * |
| 110-4 | KS24 | ” | ” | ” | ” | 10 | 13 | 1 | 6.92 | 0.03 * |
| 71-1 | T4 | ” | ” | 7el1 | ” | 8 | 11 | 0 | 7.21 | 0.03 * |
| 71-8 | KS24 | ” | ” | ” | ” | 8 | 8 | 0 | 8.00 | 0.02 * |
| 84-1 | KS24 | ” | ” | ” | ” | 9 | 9 | 0 | 6.79 | 0.03 * |
| Recomb. Genotype | Chromosome Type | F2 Segregation | ||||
|---|---|---|---|---|---|---|
| HOM+ | HET | HOM− | Χ2 1:2:1 | p Value 1 | ||
| R69-9/CS | T4/distal 7EL | 3 | 4 | 3 | 0.36 | 0.83 |
| R69-9/2*Blasco | 8 | 8 | 8 | 2.67 | 0.26 | |
| R74-10/2*Blasco | ” | 21 | 32 | 15 | 1.29 | 0.52 |
| R74-10/4*Blasco | 10 | 21 | 5 | 3.18 | 0.20 | |
| R175-5/CS | ” | 14 | 16 | 6 | 4.00 | 0.14 |
| R175-5/2*Blasco | 6 | 8 | 10 | 4.00 | 0.14 | |
| R71-1/CS | ” | 8 | 20 | 8 | 0.44 | 0.80 |
| R71-1/2*Blasco | 4 | 4 | 5 | 2.07 | 0.36 | |
| R69-6/Blasco | ” | 9 | 12 | 3 | 3.0 | 0.22 |
| R7-1/Blasco | 7E/distal 7el1L | 7 | 27 | 26 | 12.63 | 0.002 ** |
| R4-4/Blasco | ” | 8 | 21 | 26 | 14.85 | 0.000 *** |
| R62-1/CS | ” | 8 | 15 | 17 | 6.55 | 0.04 * |
| R74-9/Blasco | ” | 4 | 14 | 46 | 75.38 | 0.000 *** |
| R7-14/Blasco | ” | 6 | 20 | 21 | 10.61 | 0.005 ** |
| Parental lines | ||||||
| T4 (Thatcher)/CS | 7 | 17 | 5 | 1.14 | 0.57 | |
| T4 (Thatcher)/CS//Blasco | 6 | 10 | 8 | 1.00 | 0.61 | |
| 7E(7D) substit. (CS)/CS | 9 | 14 | 10 | 0.82 | 0.66 | |
| 7E(7D) substit. (CS)/Blasco | 11 | 14 | 10 | 1.33 | 0.51 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ceoloni, C.; Kuzmanović, L.; Ruggeri, R.; Rossini, F.; Forte, P.; Cuccurullo, A.; Bitti, A. Harnessing Genetic Diversity of Wild Gene Pools to Enhance Wheat Crop Production and Sustainability: Challenges and Opportunities. Diversity 2017, 9, 55. https://doi.org/10.3390/d9040055
Ceoloni C, Kuzmanović L, Ruggeri R, Rossini F, Forte P, Cuccurullo A, Bitti A. Harnessing Genetic Diversity of Wild Gene Pools to Enhance Wheat Crop Production and Sustainability: Challenges and Opportunities. Diversity. 2017; 9(4):55. https://doi.org/10.3390/d9040055
Chicago/Turabian StyleCeoloni, Carla, Ljiljana Kuzmanović, Roberto Ruggeri, Francesco Rossini, Paola Forte, Alessia Cuccurullo, and Alessandra Bitti. 2017. "Harnessing Genetic Diversity of Wild Gene Pools to Enhance Wheat Crop Production and Sustainability: Challenges and Opportunities" Diversity 9, no. 4: 55. https://doi.org/10.3390/d9040055
APA StyleCeoloni, C., Kuzmanović, L., Ruggeri, R., Rossini, F., Forte, P., Cuccurullo, A., & Bitti, A. (2017). Harnessing Genetic Diversity of Wild Gene Pools to Enhance Wheat Crop Production and Sustainability: Challenges and Opportunities. Diversity, 9(4), 55. https://doi.org/10.3390/d9040055






